Quantitative Determination of Alkaloids in Lotus Flower (Flower Buds of Nelumbo nucifera) and Their Melanogenesis Inhibitory Activity
Abstract
:1. Introduction
2. Results and Discussion
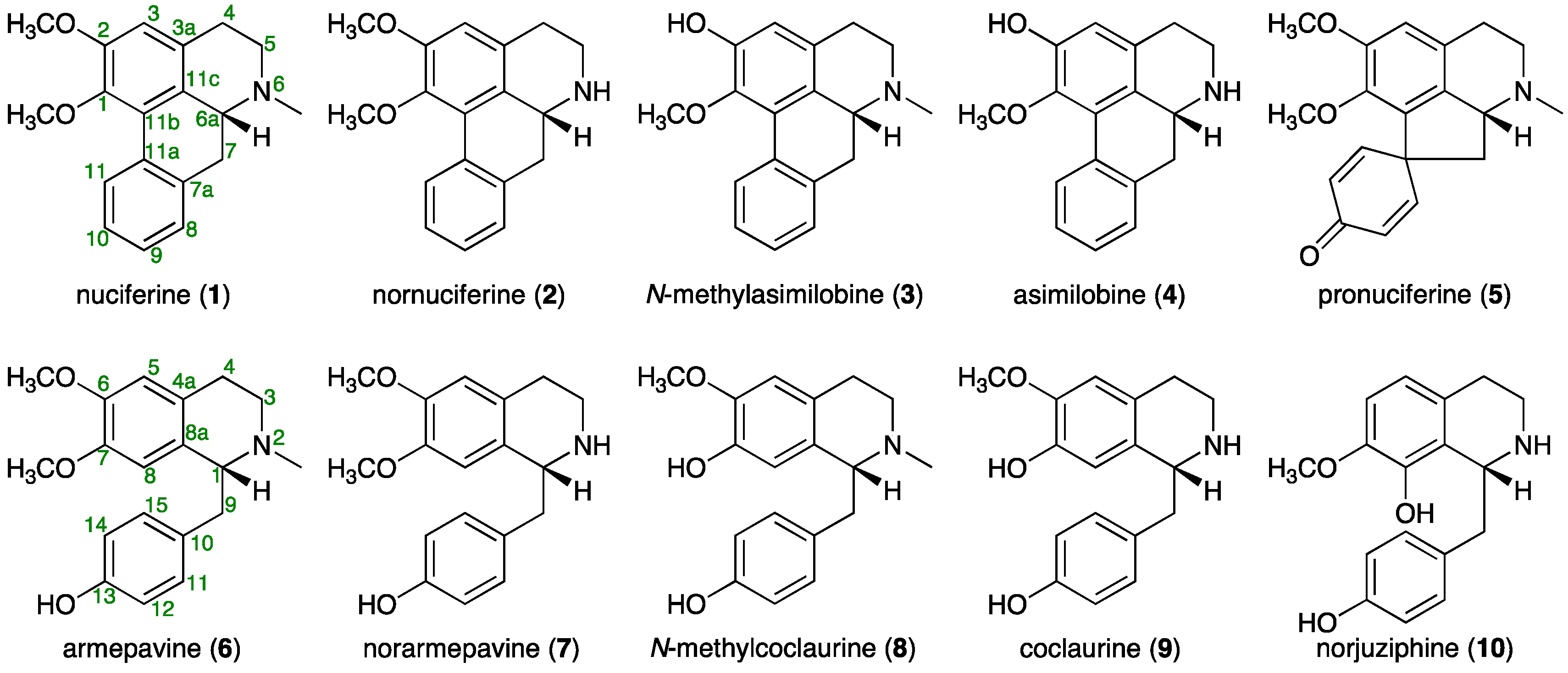
2.1. Isolation of Principal Alkaloids (1–10) from Lotus Flower
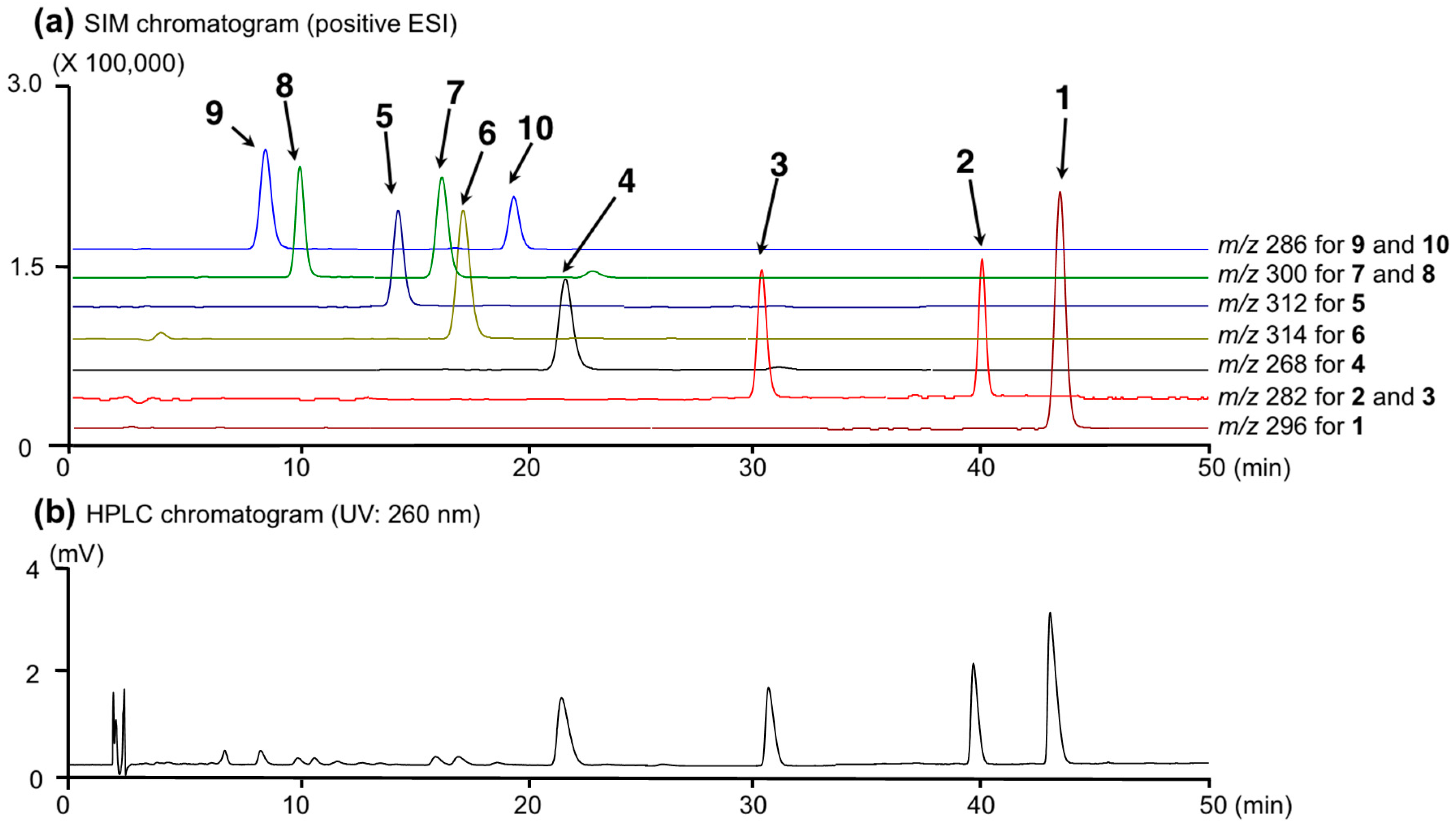
2.2. Simultaneous Quantitative Analysis of 10 Alkaloids (1–10) in Lotus Flowers
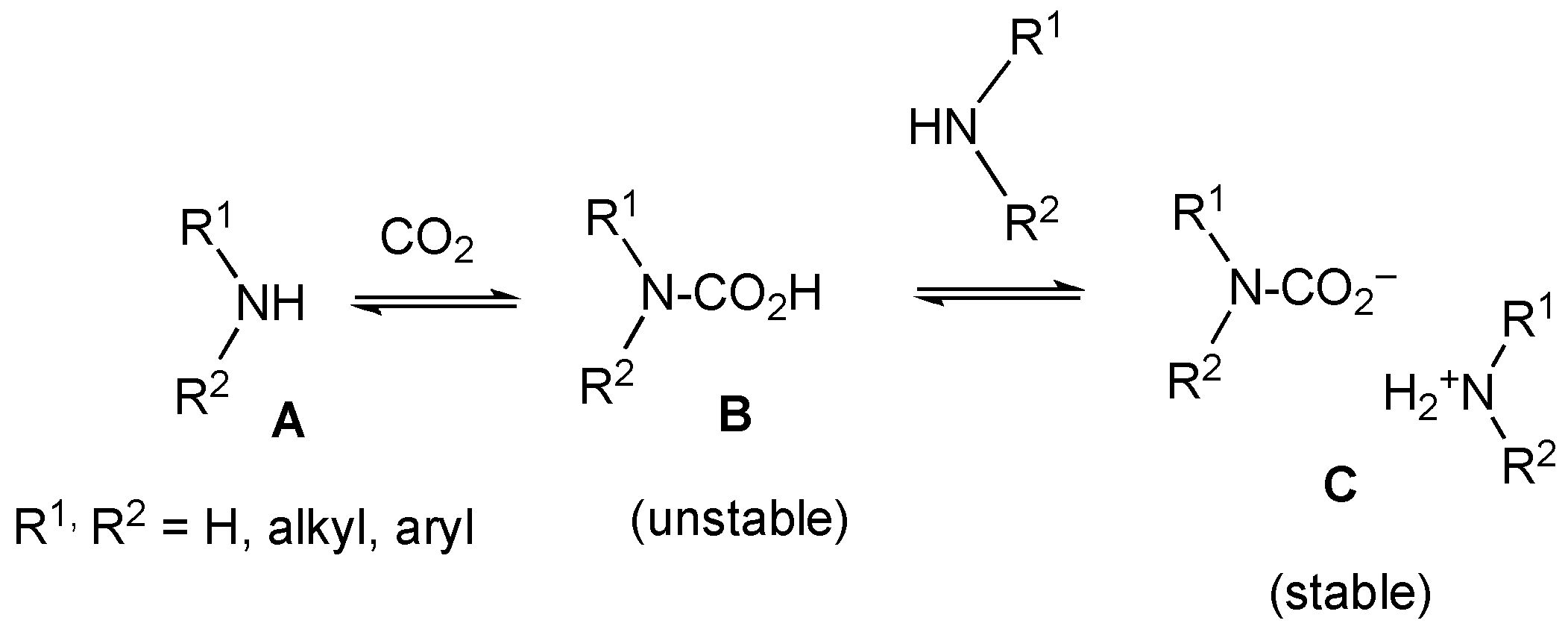
2.3. Ammonium Carbamate Salt (2′′) Formation from the Free Alkaloid (2)
2.4. Effects of the Hydrochlorides of These Alkaloids (1–10) and 2a on Theophylline-Stimulated Melanogenesis Inhibitory Activity
2.5. Effects on Mushroom Tyrosinase
2.6. Effects on Expression of Tyrosinase, TRP-1, and TRP-2
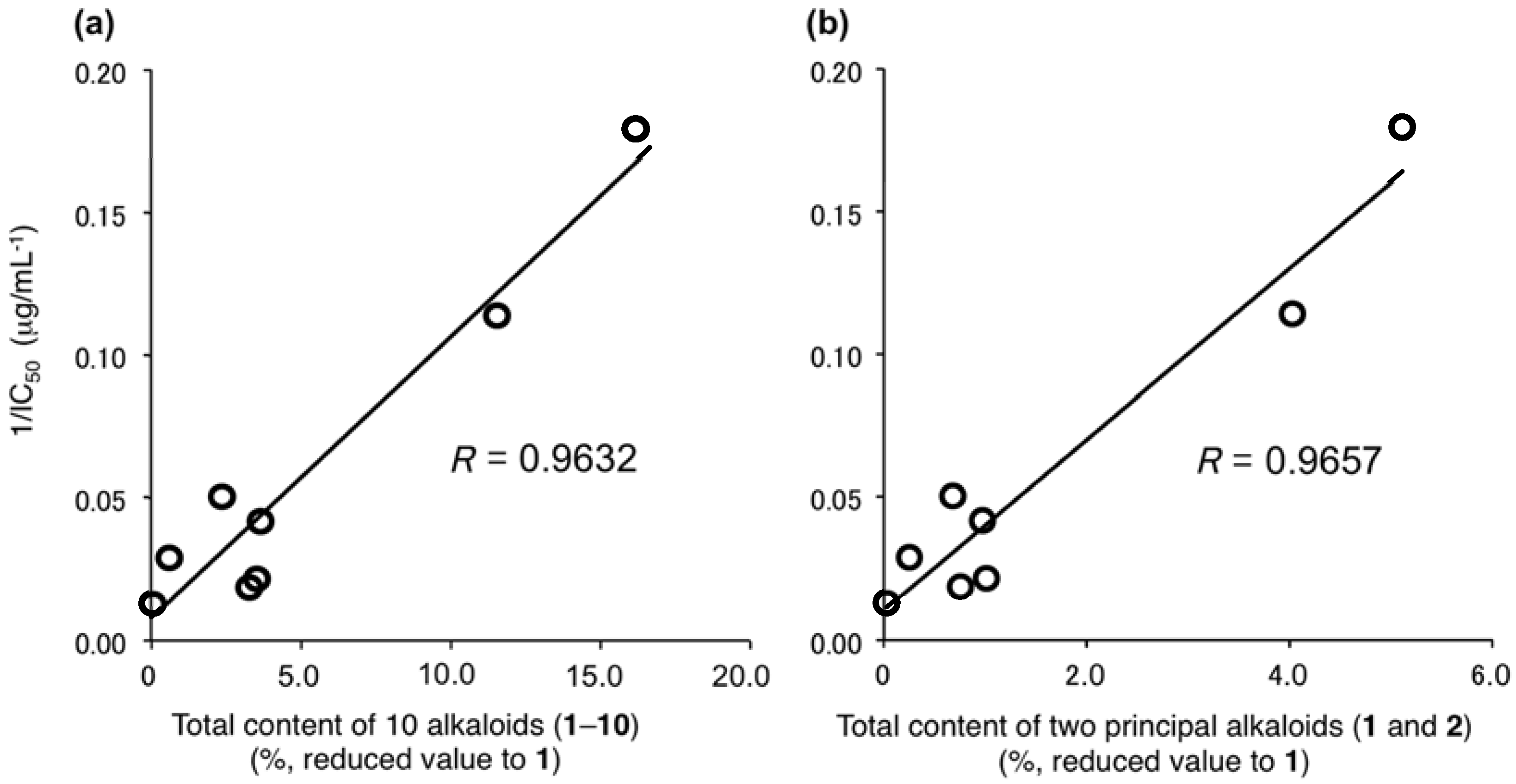
2.7. Correlation between the Melanogenesis Inhibitory Activity and Total Contents of Alkaloids (1–10) in Lotus Flower Extracts
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Plant Materials
3.3. Extraction and Isolation
3.4. Preparation of Hydrochlorides of Alkaloids (1–10)
3.5. Standard Solution Preparation
3.6. Sample Preparation
3.7. LC-MS Instruments and Conditions
3.8. Calibration and Validation
3.9. Reaction of Free Alkaloid (2) with CO2 in an Air Atmosphere
3.10. Reagents for Bioassays
3.11. Cell Culture
3.12. Melanogenesis and Cell Viability
3.13. Total Contents (%) of the 10 Alkaloids (1–10) Calculated Based on the Ratio of IC50 Values (µg/mL) Against Melanogenesis Inhibitory Activities
3.14. Mushroom Tyrosinase
3.15. Expressions of Tyrosinase, TRP-1, and TRP-2 mRNA
3.16. Statistical Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mukherjee, P.K.; Mukherjee, D.; Maji, A.K.; Rai, S.; Heinrich, M. The sacred lotus (Nelumbo nucifera)–Phytochemical and therapeutic profile. J. Pharm. Pharmacol. 2009, 61, 407–412. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, S.; Nakashima, S.; Tanabe, G.; Oda, Y.; Yokota, N.; Fujimoto, K.; Matsumoto, T.; Sakuma, R.; Ohta, T.; Ogawa, K.; et al. Alkaloid constituents from flower buds and leaves of sacred lotus (Nelumbo nucifera) with melanogenesis inhibitory activity in B16 melanoma cells. Bioorg. Med. Chem. 2013, 21, 779–787. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.-Z.; Wu, W.; Jiao, L.-L.; Yang, P.-F.; Guo, M.-Q. Analysis of flavonoids in lotus (Nelumbo nucifera) leaves and their antioxidant activity using macroprous resin chromatography coupled with LC-MS/MS and antioxidant biochemical assays. Molecules 2015, 20, 10553–10565. [Google Scholar] [CrossRef] [PubMed]
- Kredy, H.M.; Huang, D.H.; Xie, B.J.; He, H.; Yang, E.N.; Tian, B.Q.; Xiao, D. Flavonoids of lotus (Nelumbo nucifera, gaertn.) seed epicarp and their antioxidant potential. Eur. Food Res. Technol. 2010, 231, 387–394. [Google Scholar] [CrossRef]
- Zhenjia, Z.; Minglin, W.; Daijie, W.; Wenjuan, D.; Xiao, W.; Chengchao, Z. Preparative separation of alkaloids from Nelumbo nucifera leaves by conventional and pH-zone-refining counter-current chromatography. J. Chromatogr. B 2010, 878, 1647–1651. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Song, Z.; Xue, W.; Sheng, J.; Shu, Z.; Shi, Y.; Liang, J.; Yao, X. Synthesis and structure-activity relationship of nuciferine derivatives as potential acetylcholinesterase inhibitors. Med. Chem. Res. 2014, 23, 3178–3186. [Google Scholar] [CrossRef]
- Hu, R.; Dai, X.; Lu, Y.; Pan, Y. Preparative separation of isoquinoline alkaloids from Stephania yunnanensis by pH-zone-refining counter-current chromatography. J. Chromatogr. B 2010, 878, 1881–1884. [Google Scholar] [CrossRef] [PubMed]
- Fischer, D.C.H.; Gonçalves, M.I.; Oliveira, F.; Alvarenga, M.A. Constituents from Siparuna apiosyce. Fitoterapia 1999, 70, 322–323. [Google Scholar] [CrossRef]
- De Wet, H.; van Heerden, F.R.; van Wyk, B.-E. Alkaloidal variation in Cissampelos capensis (Menispermaceae). Molecules 2011, 16, 3001–3009. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Olalla, A.; Würdemann, M.A.; Wanner, M.J.; Ingemann, S.; van Maarseveen, J.H.; Hiemstra, H. Organocatalytic enantioselective pictet–spengler approach to biologically relevant 1-benzyl-1,2,3,4-tetrahydroisoquinoline alkaloids. J. Org. Chem. 2015, 80, 5125–5132. [Google Scholar] [CrossRef] [PubMed]
- Baxendale, I.R.; Davidson, T.D.; Ley, S.V.; Perni, R.H. Enantioselective synthesis of the tetrahydrobenzylisoquinoline alkaloid (−)-norarmepavine using polymer supported reagents. Heterocycles 2003, 60, 2707–2715. [Google Scholar] [CrossRef]
- Tomita, M.; Shingu, T.; Fujitani, K.; Furukawa, H. Studies on the alkaloids of menispermaceous plants. CCXVI. Nuclear magnetic resonance spectra of benzylisoquinoline derivatives. (1). n-methylcoclaurine type bases. Chem. Pharm. Bull. 1965, 13, 921–926. [Google Scholar] [CrossRef] [PubMed]
- Faust, J.; Preiss, A.; Ripperger, H.; Sandoval, D.; Schreiber, K. Alkaloids aus Annona cristalensis. Pharmazie 1981, 36, 713. [Google Scholar]
- Chen, J.-J.; Chang, Y.-L.; Teng, C.-M.; Lin, W.-Y.; Chen, Y.-C.; Chen, I.-S. A new tetrahydroprotoberberine N-oxide alkaloids and anti-pletelet aggregation constituents of Corydalis tashiroi. Planta Med. 2001, 67, 423–427. [Google Scholar] [CrossRef] [PubMed]
- Fichter, F.; Becker, B. Über die bildung ysmmetrisch dialkylierter harnstoffe durch erthitzen der entsprechenden carbaminate. Ber. Dtsch. Chem. Ges. 1911, 44, 3481–3485. [Google Scholar] [CrossRef]
- Werner, E.A. CXVII.-the constitution of carbamides. Part XI. The methanism of the synthesis of urea from ammonium carbamate. The preparation of certain mixed tri-substituted carbamates and dithiocarbamates. J. Chem. Soc. Trans. 1920, 117, 1046–1053. [Google Scholar] [CrossRef]
- Jensen, A.; Faurholt, C. Studies on carbamates. V. The carbamates of alpha-alanine and beta-alanine. Acta Chem. Scand. 1952, 6, 385–394. [Google Scholar] [CrossRef]
- Olsen, J.; Vejlby, K.; Faurholt, C. Studies on carbamates. VI. The carbamate of glycine. Acta Chem. Scand. 1952, 6, 395–397. [Google Scholar]
- Jensen, A.; Jensen, B.J.; Faurholt, C. Studies on carbamates. VII. The carbamates of n-propylamine and isopropylamine. Acta Chem. Scand. 1952, 6, 398–403. [Google Scholar] [CrossRef]
- Combes, J.R.; White, L.D.; Tripp, C.P. Chemical modification of metal oxide surfaces in supercritical CO2: In situ infrared studies of the adsorption and reaction of organosilanes on silica. Langmuir 1999, 15, 7870–7875. [Google Scholar] [CrossRef]
- Kreher, U.P.; Rosamilia, A.E.; Raston, C.L.; Scott, J.L.; Strauss, C.R. Self-associated, “distillable” ionic media. Molecules 2004, 9, 387–393. [Google Scholar] [CrossRef] [PubMed]
- Mase, N.; Horibe, T. Organocatalytic Knoevenagel condensations by means of carbamic acid ammonium salts. Org. Lett. 2013, 15, 1854–1857. [Google Scholar] [CrossRef] [PubMed]
- Murohy, L.J.; Robertson, K.N.; Kemp, R.A.; Tuononen, H.M.; Clyburne, J.A.C. Structurally simple complexes of CO2. Chem. Commun. 2015, 51, 3942–3956. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, F.S.; Kitchens, C.L. Reduced reactivity of amines against nucleophilic substitution via reversible reaction with carbon dioxide. Molecules 2016, 21, 24. [Google Scholar] [CrossRef] [PubMed]
- Morikawa, T.; Nakanishi, Y.; Ninomiya, K.; Matsuda, H.; Nakashima, S.; Miki, H.; Miyashita, Y.; Yoshikawa, M.; Hayakawa, T.; Muraoka, O. Dimeric pyrrolidinoindoline-type alkaloids with melanogenesis inhibitory activity in flower buds of Chimonanthus praecox. J. Nat. Med. 2014, 68, 539–549. [Google Scholar] [CrossRef] [PubMed]
- Morikawa, T.; Ninomiya, K.; Kuramoto, H.; Kamei, I.; Yoshikawa, M.; Muraoka, O. Phenylethanoid andphenylpropanoid glycosides with melanogenesis inhibitory activity from the flowers of Narcissus tazetta var. chinensis. J. Nat. Med. 2016, 70, 89–101. [Google Scholar] [CrossRef] [PubMed]
- Ninomiya, K.; Matsumoto, T.; Chaipech, S.; Miyake, S.; Katsuyama, Y.; Tsuboyama, A.; Pongpiriyadacha, Y.; Hayakawa, T.; Muraoka, O.; Morikawa, T. Simultaneous quantitative analysis of 12 methoxyflavones with melanogenesis inhibitory activity from the rhizomes of Kaempferia parviflora. J. Nat. Med. 2016, 70, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Tanabe, G.; Sugano, Y.; Sonoda, N.; Tsutsui, N.; Morikawa, T.; Ninomiya, K.; Yoshikawa, M.; Muraoka, O. Total synthesis of 4,5-didehydroguadiscine: A potent melanogeneisi inhibitor from the Brazilian medicinal herb, Hornschuchia oblique. J. Nat. Prod. 2015, 78, 1536–1542. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds 1–10 are available from the authors.

| Treatment a | Inhibition (%) | IC50 (µg/mL) | ||||
|---|---|---|---|---|---|---|
| 0 µg/mL | 3 µg/mL | 10 µg/mL | 30 µg/mL | 100 µg/mL | ||
| MeOH ext. | 0.0 ± 1.3 (100.0 ± 9.3) | 28.3 ± 3.7 (110.9 ± 2.7) | 68.9 ± 3.1 ** (124.2 ± 5.1) | 96.4 ± 3.1 ** (126.5 ± 5.3) | 97.0 ± 3.3 ** (103.3 ± 7.1) | 5.6 |
| EtOAc-soluble fraction | 0.0 ± 6.1 (100.0 ± 7.2) | 11.0 ± 1.7 (99.3 ± 9.8) | 51.5 ± 5.3 ** (106.8 ± 5.1) | 83.0 ± 2.9 ** (110.9 ± 4.2) | 100.9 ± 3.4 ** (118.3 ± 10.4) | 11.1 |
| n-BuOH-soluble fraction | 0.0 ± 13.3 (100.0 ± 11.2) | 17.6 ± 7.0 (104.6 ± 11.6) | 35.9 ± 7.1 (104.9 ± 2.6) | 72.3 ± 4.7 ** (114.8 ± 13.9) | 94.7 ± 3.5 ** (104.3 ± 3.8) | 13.7 |
| Treatment | Inhibition (%) | IC50 (µg/mL) | ||||
| 0 µg/mL | 0.1 µg/mL | 0.3 µg/mL | 1 µg/mL | 3 µg/mL | ||
| CHCl3-soluble fraction | 0.0 ± 1.7 (100.0 ± 3.5) | 19.8 ± 6.8 (101.5 ± 6.1) | 35.6 ± 6.6 ** (103.1 ± 6.1) | 79.2 ± 2.3 ** (114.7 ± 4.6) | 107.2 ± 3.0 ** (107.4 ± 9.5) | 0.37 |
| Extraction Method | Extraction Yield (%) | Contents (mg/g in Dry Material) a | Total | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | |||
| Methanol, reflux | 15.0 | 1.76 (100) | 1.75 (100) | 0.07 (100) | 0.63 (100) | 0.69 (100) | 0.83 (100) | 1.45 (100) | 5.73 (100) | 1.30 (100) | 0.75 (100) | 14.96 (100) |
| 50% Methanol, reflux | 25.3 | 1.09 (62) | 1.35 (77) | 0.05 (71) | 0.50 (79) | 0.61 (88) | 0.78 (94) | 1.35 (93) | 3.79 (66) | 0.94 (73) | 0.56 (75) | 11.02 (74) |
| H2O, reflux | 23.1 | 0.24 (14) | 0.35 (20) | n.d. b | 0.21 (33) | 0.18 (26) | 0.38 (45) | 0.78 (54) | 2.57 (45) | 0.66 (51) | 0.29 (38) | 5.66 (38) |
| Methanol, sonication | 9.6 | 0.88 (50) | 1.11 (64) | 0.03 (44) | 0.39 (62) | 0.33 (48) | 0.47 (56) | 0.97 (67) | 2.77 (48) | 0.70 (54) | 0.42 (56) | 8.07 (54) |
| 50% Methanol, sonication | 22.0 | 0.98 (56) | 1.27 (73) | 0.04 (58) | 0.49 (78) | 0.47 (69) | 0.80 (96) | 1.38 (95) | 3.93 (69) | 0.97 (75) | 0.59 (79) | 10.92 (73) |
| H2O, sonication | 19.3 | 0.14 (8) | 0.21 (12) | n.d. b | 0.12 (20) | 0.08 (11) | 0.25 (30) | 0.53 (37) | 1.91 (33) | 0.48 (37) | 0.19 (26) | 3.91 (26) |
| Analyte | Regression Equation a | Correlation Coefficient | Detection Limit b (ng) | Quantitation Limit b (ng) | Precision c (RSD, %) | |
|---|---|---|---|---|---|---|
| Intra-Day | Inter-Day | |||||
| Nuciferine (1) | y = 7477635x − 1302 | 0.9998 | 0.17 | 0.51 | 0.25 | 0.59 |
| Nornuciferine (2) | y = 2698708x − 10941 | 1.0000 | 0.71 | 2.16 | 0.79 | 0.43 |
| N-Methylasimilobine (3) | y = 7054297x + 243961 | 0.9996 | 0.32 | 0.99 | 1.36 | 1.40 |
| Asimilobine (4) | y = 2076494x − 36021 | 0.9999 | 0.70 | 2.13 | 0.63 | 0.57 |
| Pronuciferine (5) | y = 3522995x + 101328 | 0.9998 | 0.73 | 2.18 | 0.95 | 1.08 |
| Armepavine (6) | y = 2076494x − 36021 | 0.9999 | 0.32 | 0.97 | 0.68 | 1.10 |
| Norarmepavine (7) | y = 1998354x − 15296 | 0.9999 | 0.81 | 2.47 | 0.54 | 0.73 |
| N-Methylcoclaurine (8) | y = 1595194x + 53314 | 0.9999 | 0.90 | 2.71 | 0.59 | 0.86 |
| Coclaurine (9) | y = 1878370x + 16838 | 0.9999 | 0.44 | 1.33 | 0.98 | 0.39 |
| Norjuziphine (10) | y = 1745634x + 15240 | 1.0000 | 0.88 | 2.65 | 0.64 | 0.66 |
| Add (µg/mL) | Recovery a (%) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | |
| 10 | 98.7 ± 0.6 | 101.4 ± 0.7 | 95.3 ± 1.0 | 99.0 ± 0.9 | 93.2 ± 0.7 | 98.5 ± 0.3 | 104.0 ± 0.7 | 94.7 ± 1.4 | 97.4 ± 0.2 | 97.4 ± 1.0 |
| 15 | 92.3 ± 0.4 | 101.7 ± 0.2 | 97.5 ± 0.7 | 101.8 ± 0.1 | 98.1 ± 0.5 | 102.2 ± 0.6 | 99.4 ± 1.1 | 99.8 ± 1.6 | 104.0 ± 0.2 | 100.2 ± 0.8 |
| 20 | 98.4 ± 0.1 | 105.8 ± 0.7 | 101.2 ± 0.8 | 102.0 ± 0.7 | 95.9 ± 0.8 | 105.1 ± 0.6 | 99.9 ± 0.9 | 96.3 ± 1.1 | 105.8 ± 0.3 | 95.3 ± 0.8 |
| Sample No. | Part | Loss of Drying a (%) | Extraction Yield b (%) | Contents (mg/g in Dry Material) a | Total | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | |||||
| NN-1 | whole flowers | 10.3 | 15.0 | 1.76 | 1.75 | 0.07 | 0.63 | 0.69 | 0.83 | 1.45 | 5.73 | 1.30 | 0.75 | 14.96 |
| NN-2 | petals | 8.6 | 16.6 | 1.99 | 2.41 | 0.07 | 1.22 | 0.62 | 1.02 | 1.74 | 5.85 | 1.60 | 0.52 | 17.04 |
| NN-3 | receptacles | 10.0 | 8.6 | 0.06 | 0.07 | n.d. c | 0.01 | 0.01 | 0.01 | 0.01 | 0.20 | 0.04 | n.d. c | 0.41 |
| NN-4 | stamens | 8.6 | 20.0 | 0.58 | 0.68 | n.d. c | 0.25 | 0.23 | 0.47 | 0.64 | 2.88 | 0.60 | 0.32 | 6.65 |
| NN-5 | whole flowers | 8.1 | 16.3 | 0.56 | 0.27 | n.d. c | 0.04 | n.d. c | 0.23 | 0.34 | 1.74 | 0.19 | 0.16 | 3.53 |
| NN-6 | petals | 7.3 | 19.0 | 0.80 | 0.34 | n.d. c | n.d. c | 0.01 | 0.36 | 0.52 | 3.14 | 0.32 | 0.27 | 5.76 |
| NN-7 | receptacles | 9.2 | 7.4 | 0.27 | 0.53 | n.d. c | n.d. c | 0.05 | 0.03 | 0.03 | 0.74 | 0.27 | n.d.c | 1.92 |
| NN-8 | stamens | 7.9 | 15.5 | 0.01 | n.d. c | n.d. c | n.d. c | n.d. c | n.d. c | n.d. c | 0.03 | 0.01 | 0.03 | 0.08 |
| Position | 2′′ (anion part) | Position | 2′′ (cation part) | ||
|---|---|---|---|---|---|
| δH (J in Hz) | δC | δH (J in Hz) | δC | ||
| 1 | 146.0 a | 1′ | 146.2 a | ||
| 2 | 152.5 b | 2′ | 153.7 b | ||
| 3 | 6.69 (s) | 111.4 | 3′ | 6.67 (s) | 111.4 |
| 3a | 128.7 | 3a′ | 125.7 | ||
| 4 | 2.74 (br d, ca. 15)
2.98 (br d, ca. 15) | 29.9, 30.2 | 4′ | 2.95 (dd, 3.8, 16.8)
3.62 (ddd, 5.7, 13.5, 16.8) | 25.5 |
| 5 | 3.20–3.35 (m)
4.62 (br d, ca. 12.5) | 41.8, 44.4 | 5′ | 3.24 (br ddd-like, ca. 13.5, 13.5, 13.5)
3.87 (br dd-like, ca. 5.7, 13.5) | 41.4 |
| 6a | 4.92 (br d, ca. 13) | 54.9, 55.8 | 6a′ | 4.29 (br dd-like, ca. 13.5, 13.5) | 53.0 |
| 7 | 2.86–2.90 (m)
3.02–3.14 (m) | 33.9, 35.9 | 7′ | 3.38 (dd, 13.5, 13.5)
3.45 (dd, 4.5, 13.5) | 34.0 |
| 7a | 135.7 | 7a′ | 133.1 | ||
| 8 | 7.24–7.30 (m) | 128.0 c | 8′ | 7.24–7.30 (m) | 128.3 c |
| 9 | 7.24–7.30 (m) | 127.4 c | 9′ | 7.24–7.30 (m) | 128.2 c |
| 10 | 7.35 (m) | 127.3 c | 10′ | 7.35 (m) | 127.8 c |
| 11 | 8.44 (br s-like) | 128.5 c | 11′ | 8.41 (d, 7.9) | 128.6 c |
| 11a | 131.3 | 11a′ | 131.3 | ||
| 11b | 127.5 d | 11b′ | 127.0 d | ||
| 11c | 124.8, 124.9 | 11c′ | 121.4 | ||
| 1-OCH3 | 3.667 e (s) | 60.3 f | 1′-OCH3 | 3.673 e (s) | 60.0 f |
| 2-OCH3 | 3.90 g (s) | 56.0 h | 2′-OCH3 | 3.91 g (s) | 55.9 h |
| N-COO | 157.5, 160.1 | NH2 | 9.96 (br ddd-like, ca. 13.5, 13.5, 13.5 )
10.84 (br d-like, ca. 13.5) | ||
| Position | 2a | Position | 2 | ||
| δH (J in Hz) | δC | δH (J in Hz) | δC | ||
| 1 | 145.7 | 1 | 145.3 | ||
| 2 | 152.1 | 2 | 152.2 | ||
| 3 | 6.67 (s) | 111.5 | 3 | 6.65 (s) | 111.8 |
| 3a | 126.1 | 3a | 128.5 | ||
| 4 | 2.65 (br d, ca. 15)
2.88 (m) | 30.3 | 4 | 2.71 (d, 13.1)
3.05 (m) | 28.9 |
| 5 | 3.00 (br dd, ca. 11, 13)
4.73 (br d, ca. 13) | 38.8 | 5 | 3.01 (m)
3.40 (br q, ca. 6) | 43.0 |
| 6a | 4.46 (br s) | 51.4 | 6a | 3.85 (br dd, ca. 5, 14) | 53.5 |
| 7 | 2.86 (m)
2.98 (dd, 12.8, 15.8) | 35.2 | 7 | 2.77 (t, 13.8)
2.87 (dd, 4.6, 13.8) | 37.3 |
| 7a | 136.8 | 7a | 135.9 | ||
| 8 | 7.25 (dd, 1.6, 7.8) | 128.2 | 8 | 7.24 (m) | 127.8 |
| 9 | 7.27 (br dd, ca. 8, 8) | 127.5 | 9 | 7.21 (ddd, 1.1, 7.1, 7.1) | 127.4 |
| 10 | 7.32 (ddd, 1.6, 7.8, 8.0) | 126.9 | 10 | 7.30 (m) | 127.0 |
| 11 | 8.44 (br d, ca. 8) | 128.3 | 11 | 8.39 (br d, ca. 8) | 128.4 |
| 11a | 131.7 | 11a | 132.1 | ||
| 11b | 127.6 | 11b | 126.5 | ||
| 11c | 129.7 | 11c | 128.7 | ||
| 1-OCH3 | 3.66 (s) | 60.2 | 1-OCH3 | 3.67 (s) | 60.2 |
| 2-OCH3 | 3.90 (s) | 56.0 | 2-OCH3 | 3.88 (s) | 55.9 |
| N-COO | 156.0 | ||||
| N-CO2CH3 | 3.76 (s) | 52.6 | |||
| Treatment | Inhibition (%) | IC50 | |||||
|---|---|---|---|---|---|---|---|
| 0 µM | 3 µM | 10 µM | 30 µM | 100 µM | (µM) | (µg/mL) b | |
| Nuciferine·HCl (1) a | 0.0 ± 4.0
(100.0 ± 2.6) | 28.3 ± 4.2 **
(112.1 ± 2.6) | 57.8 ± 2.3 **
(112.1 ± 2.5) | 89.7 ± 1.7 **
(100.9 ± 5.8) | 91.8 ± 2.6 **
(45.9 ± 3.3 #) | 7.1 | 2.4 |
| Nornuciferine·HCl (2) a | 0.0 ± 3.4
(100.0 ± 4.9) | 44.6 ± 3.4 **
(99.5 ± 8.2) | 70.6 ± 3.6 **
(93.9 ± 4.1) | 94.3 ± 3.2 **
(70.6 ± 4.8 #) | — (1.9 ± 0.1 #) | 3.9 | 1.2 |
| N-Methylasimilobine·HCl (3) a | 0.0 ± 4.7
(100.0 ± 3.9) | 3.3 ± 2.9
(96.6 ± 2.6) | 19.5 ± 2.4 **
(100.4 ± 3.0) | 63.4 ± 5.2 **
(119.3 ± 9.4) | 89.8 ± 2.1 **
(57.7 ± 1.3 #) | 43.1 | 13.7 |
| Asimilobine·HCl (4) a | 0.0 ± 8.1
(100.0 ± 5.7) | 16.2 ± 12.8
(96.5 ± 7.5) | 31.9 ± 2.2 **
(98.9 ± 10.0) | 87.2 ± 3.3 **
(76.6 ± 6.3) | — (13.7 ± 2.2 #) | 11.3 | 3.4 |
| Pronuciferine·HCl (5) a | 0.0 ± 10.7
(100.0 ± 4.5) | 23.1 ± 3.4
(104.2 ± 3.4) | 18.8 ± 1.2
(97.4 ± 0.7) | 37.2 ± 2.6 **
(98.4 ± 1.8) | 88.4 ± 3.7 **
(89.1 ± 9.4) | 47.1 | 16.3 |
| Armepavine·HCl (6) a | 0.0 ± 6.0
(100.0 ± 2.5) | 33.9 ± 0.8 **
(104.0 ± 3.3) | 58.5 ± 7.1 **
(104.0 ± 7.7) | 81.8 ± 3.0 **
(104.0 ± 7.2) | 97.4 ± 0.4 **
(78.1 ± 2.0) | 6.5 | 3.4 |
| Norarmepavine·HCl (7) a | 0.0 ± 3.1
(100.0 ± 2.6) | 32.6 ± 4.4 **
(86.4 ± 4.0) | 53.5 ± 8.5 **
(81.2 ± 4.0) | 81.6 ± 1.4 **
(83.3 ± 2.2) | 90.5 ± 1.2 **
(68.8 ± 1.4 #) | 7.5 | 2.5 |
| N-Methylcoclaurine·HCl (8) a | 0.0 ± 5.4
(100.0 ± 3.0) | 38.6 ± 2.4 **
(97.1 ± 1.5) | 55.7 ± 3.4 **
(92.8 ± 4.1) | 74.7 ± 2.0 **
(96.4 ± 4.2) | —
— | 6.5 | 2.2 |
| Coclaurine·HCl (9) a | 0.0 ± 2.9
(100.0 ± 2.9) | 45.6 ± 7.7 **
(97.0 ± 7.7) | 65.4 ± 2.5 **
(96.2 ± 5.0) | 82.4 ± 3.5 **
(86.5 ± 7.7) | 68.0 ± 6.4 **
(53.1 ± 8.3 #) | 3.9 | 1.3 |
| Norjuziphine·HCl (10) a | 0.0 ± 5.4
(100.0 ± 5.3) | 18.5 ± 4.0 *
(98.9 ± 6.0) | 36.8 ± 4.5 **
(91.3 ± 4.9) | 94.4 ± 2.0 **
(83.9 ± 8.3) | 106.0 ± 2.0 **
(57.0 ± 2.4 #) | 14.4 | 4.6 |
| 2a | 0.0 ± 7.3
(100.0 ± 4.7) | 13.1 ± 8.9
(109.8 ± 4.3) | 43.1 ± 7.8 **
(127.5 ± 4.8) | 54.5 ± 4.3 **
(129.1 ± 2.8) | — — | 19.9 | 6.7 |
| Treatment | Inhibition (%) | IC50 | |||||
| 0 µM | 30 µM | 100 µM | 300 µM | 1000 µM | (µM) | (µg/mL) | |
| Arbutin [25,26,27] | 0.0 ± 1.4
(100.0 ± 2.1) | 20.4 ± 0.5
(82.4 ± 3.0) | 38.1 ± 0.9 **
(78.1 ± 1.9) | 61.5 ± 0.6 **
(79.8 ± 2.2) | 83.7 ± 0.5 **
(53.1 ± 1.8 #) | 174 | 47.4 |
| Substrate:Treatment | Inhibition (%) | |||||
|---|---|---|---|---|---|---|
| l-Tyrosine | l-DOPA | |||||
| 0 µM | 10 µM | 100 µM | 0 µM | 10 µM | 100 µM | |
| Nuciferine·HCl (1) a | 0.0 ± 3.2 | 6.5 ± 3.3 | 30.4 ± 1.9 ** | 0.0 ± 2.9 | 4.4 ± 3.8 | 11.0 ± 3.4 |
| Nornuciferine·HCl (2) a | 0.0 ± 2.6 | 1.0 ± 0.7 | 14.2 ± 1.5 ** | 0.0 ± 3.1 | 15.4 ± 4.1 | 8.7 ± 4.1 |
| N-Methylasimilobine·HCl (3) a | 0.0 ± 3.6 | 7.2 ± 7.3 | 0.8 ± 3.5 | 0.0 ± 1.5 | 2.3 ± 1.5 | 3.1 ± 3.9 |
| Asimilobine·HCl (4) a | 0.0 ± 1.8 | 10.5 ± 2.5 * | 14.0 ± 1.2 ** | 0.0 ± 3.3 | 3.2 ± 2.4 | 5.7 ± 3.0 |
| Pronuciferine·HCl (5) a | 0.0 ± 5.3 | −0.4 ± 2.4 | −4.4 ± 8.1 | 0.0 ± 2.0 | 6.5 ± 2.0 | 5.3 ± 5.2 |
| Armepavine·HCl (6) a | 0.0 ± 4.4 | −1.9 ± 0.9 | 40.2 ± 3.7 ** | 0.0 ± 0.5 | 4.5 ± 0.8 | 2.9 ± 1.2 |
| Norarmepavine·HCl (7) a | 0.0 ± 1.8 | −1.7 ± 1.5 | 23.3 ± 1.6 ** | 0.0 ± 2.7 | −1.3 ± 1.4 | 1.5 ± 1.5 |
| N-Methylcoclaurine·HCl (8) a | 0.0 ± 5.2 | −5.0 ± 5.7 | 15.3 ± 4.3 | 0.0 ± 2.3 | 6.1 ± 1.4 | 7.4 ± 0.8 |
| Coclaurine·HCl (9) a | 0.0 ± 2.3 | 9.0 ± 0.7 * | 35.1 ± 1.7 ** | 0.0 ± 2.5 | −4.9 ± 1.1 | 4.4 ± 1.1 |
| Norjuziphine·HCl (10) a | 0.0 ± 2.5 | 5.1 ± 1.1 | 27.4 ± 3.0 ** | 0.0 ± 2.0 | 1.8 ± 0.7 | 22.3 ± 3.6 ** |
| 2a | 0.0 ± 1.5 | 5.3 ± 1.7 | 2.9 ± 0.7 | 0.0 ± 2.7 | 3.2 ± 1.1 | 6.6 ± 2.3 |
| Substrate: l-Tyrosine | Inhibition (%) | |||||
| Treatment | 0 µM | 10 µM | 30 µM | 100 µM | 300 µM | IC50 (µM) |
| Kojic acid [25,26,27] | 0.0 ± 2.4 | 12.2 ± 3.3 | 46.4 ± 2.6 ** | 66.5 ± 2.1 ** | 96.8 ± 0.9 ** | 43.6 |
| Substrate: l-DOPA | Inhibition (%) | |||||
| Treatment | 0 µM | 10 µM | 30 µM | 100 µM | 300 µM | IC50 (µM) |
| Kojic acid [25,26,27] | 0.0 ± 0.9 | 22.3 ± 2.1 ** | 50.6 ± 0.6 ** | 78.2 ± 0.7 ** | 89.3 ± 0.3 ** | 29.6 |
| Treatment | Tyrosinase mRNA/-actin mRNA | ||
|---|---|---|---|
| 0 µM | 3 µM | 10 µM | |
| Nuciferine·HCl (1) a | 1.00 ± 0.15 | 0.59 ± 0.03 * | 0.45 ± 0.05 * |
| Nornuciferine·HCl (2) a | 1.00 ± 0.19 | 0.76 ± 0.05 | 0.51 ± 0.10 |
| Armepavine·HCl (6) a | 1.00 ± 0.14 | 0.86 ± 0.13 | 0.74 ± 0.02 |
| Norarmepavine·HCl (7) a | 1.00 ± 0.24 | 0.81 ± 0.08 | 1.00 ± 0.11 |
| Coclaurine·HCl (9) a | 1.00 ± 0.14 | 0.82 ± 0.21 | 0.52 ± 0.05 |
| Treatment | TRP-1 mRNA/-actin mRNA | ||
| 0 µM | 3 µM | 10 µM | |
| Nuciferine·HCl (1) a | 1.00 ± 0.12 | 1.18 ± 0.17 | 1.18 ± 0.25 |
| Nornuciferine·HCl (2) a | 1.00 ± 0.10 | 1.21 ± 0.18 | 1.15 ± 0.19 |
| Armepavine·HCl (6) a | 1.00 ± 0.22 | 0.98 ± 0.32 | 0.83 ± 0.15 |
| Norarmepavine·HCl (7) a | 1.00 ± 0.09 | 0.87 ± 0.22 | 1.03 ± 0.25 |
| Coclaurine·HCl (9) a | 1.00 ± 0.24 | 0.51 ± 0.08 | 0.66 ± 0.13 |
| Treatment | TRP-2 mRNA/-actin mRNA | ||
| 0 µM | 3 µM | 10 µM | |
| Nuciferine·HCl (1) a | 1.00 ± 0.16 | 1.33 ± 0.35 | 0.87 ± 0.16 |
| Nornuciferine·HCl (2) a | 1.00 ± 0.12 | 0.92 ± 0.20 | 1.36 ± 0.09 |
| Armepavine·HCl (6) a | 1.00 ± 0.05 | 1.07 ± 0.22 | 0.81 ± 0.22 |
| Norarmepavine·HCl (7) a | 1.00 ± 0.18 | 0.80 ± 0.24 | 0.77 ± 0.14 |
| Coclaurine·HCl (9) a | 1.00 ± 0.11 | 1.02 ± 0.22 | 1.07 ± 0.07 |
| Sample No. | Inhibition (%) | IC50 | ||||
|---|---|---|---|---|---|---|
| 0 µg/mL | 3 µg/mL | 10 µg/mL | 30 µg/mL | 100 µg/mL | (µg/mL) | |
| NN-1 | 0.0 ± 1.3 (100.0 ± 9.3) | 28.3 ± 3.7 (110.9 ± 2.7) | 68.9 ± 3.1 ** (124.2 ± 5.1) | 96.4 ± 3.1 ** (126.5 ± 5.3) | 97.0 ± 3.3 ** (103.3 ± 7.1) | 5.6 |
| NN-2 | 0.0 ± 5.7 (100.0 ± 2.7) | 15.7 ± 8.2 (113.3 ± 2.2) | 49.9 ± 12.0 ** (114.6 ± 1.8) | 108.2 ± 4.1 ** (128.9 ± 2.8) | 103.8 ± 6.7 ** (116.2 ± 2.5) | 8.8 |
| NN-3 | 0.0 ± 5.9 (100.0 ± 6.8) | 18.6 ± 4.2 (104.7 ± 4.3) | 26.1 ± 5.0 ** (102.6 ± 2.9) | 72.9 ± 0.7 ** (119.3 ± 5.4) | 86.8 ± 4.1 ** (108.2 ± 3.6) | 34.7 |
| NN-4 | 0.0 ± 10.0 (100.0 ± 5.3) | 10.7 ± 8.1 (99.4 ± 7.1) | 28.3 ± 4.7 (99.1 ± 11.1) | 95.7 ± 1.6 ** (98.0 ± 5.0) | 107.0 ± 2.1 ** (92.4 ± 6.3) | 24.0 |
| NN-5 | 0.0 ± 7.3 (100.0 ± 3.7) | 23.7 ± 4.0 ** (99.6 ± 2.2) | 34.4 ± 5.1 ** (102.7 ± 4.3) | 86.6 ± 2.6 ** (99.6 ± 2.4) | 103.4 ± 1.9 ** (92.5 ± 5.0) | 19.9 |
| NN-6 | 0.0 ± 6.0 (100.0 ± 4.5) | −6.9 ± 13.7 (87.8 ± 4.7) | −6.3 ± 12.0 (88.1 ± 1.0) | 77.4 ± 5.4 ** (90.8 ± 4.0) | 104.6 ± 2.1 ** (74.1 ± 3.3 #) | 54.1 |
| NN-7 | 0.0 ± 4.4 (100.0 ± 1.6) | −9.5 ± 6.9 (94.7 ± 4.1) | 9.0 ± 7.6 (101.2 ± 2.2) | 79.4 ± 2.1 ** (102.3 ± 2.8) | 84.2 ± 4.2 ** (84.9 ± 0.9) | 46.6 |
| NN-8 | 0.0 ± 1.7 (100.0 ± 4.6) | −3.3 ± 4.8 (100.8 ± 7.4) | 1.3 ± 7.9 (93.5 ± 3.2) | 57.6 ± 3.1 ** (95.6 ± 3.2) | 64.9 ± 2.3 ** (97.8 ± 3.8) | 78.9 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morikawa, T.; Kitagawa, N.; Tanabe, G.; Ninomiya, K.; Okugawa, S.; Motai, C.; Kamei, I.; Yoshikawa, M.; Lee, I.-J.; Muraoka, O. Quantitative Determination of Alkaloids in Lotus Flower (Flower Buds of Nelumbo nucifera) and Their Melanogenesis Inhibitory Activity. Molecules 2016, 21, 930. https://doi.org/10.3390/molecules21070930
Morikawa T, Kitagawa N, Tanabe G, Ninomiya K, Okugawa S, Motai C, Kamei I, Yoshikawa M, Lee I-J, Muraoka O. Quantitative Determination of Alkaloids in Lotus Flower (Flower Buds of Nelumbo nucifera) and Their Melanogenesis Inhibitory Activity. Molecules. 2016; 21(7):930. https://doi.org/10.3390/molecules21070930
Chicago/Turabian StyleMorikawa, Toshio, Niichiro Kitagawa, Genzoh Tanabe, Kiyofumi Ninomiya, Shuhei Okugawa, Chiaki Motai, Iyori Kamei, Masayuki Yoshikawa, I-Jung Lee, and Osamu Muraoka. 2016. "Quantitative Determination of Alkaloids in Lotus Flower (Flower Buds of Nelumbo nucifera) and Their Melanogenesis Inhibitory Activity" Molecules 21, no. 7: 930. https://doi.org/10.3390/molecules21070930
APA StyleMorikawa, T., Kitagawa, N., Tanabe, G., Ninomiya, K., Okugawa, S., Motai, C., Kamei, I., Yoshikawa, M., Lee, I.-J., & Muraoka, O. (2016). Quantitative Determination of Alkaloids in Lotus Flower (Flower Buds of Nelumbo nucifera) and Their Melanogenesis Inhibitory Activity. Molecules, 21(7), 930. https://doi.org/10.3390/molecules21070930







