Characterization and Analysis of 2-(2-Phenylethyl)-chromone Derivatives from Agarwood (Aquilaria crassna) by Artificial Holing for Different Times
Abstract
:1. Introduction
2. Results and Discussion
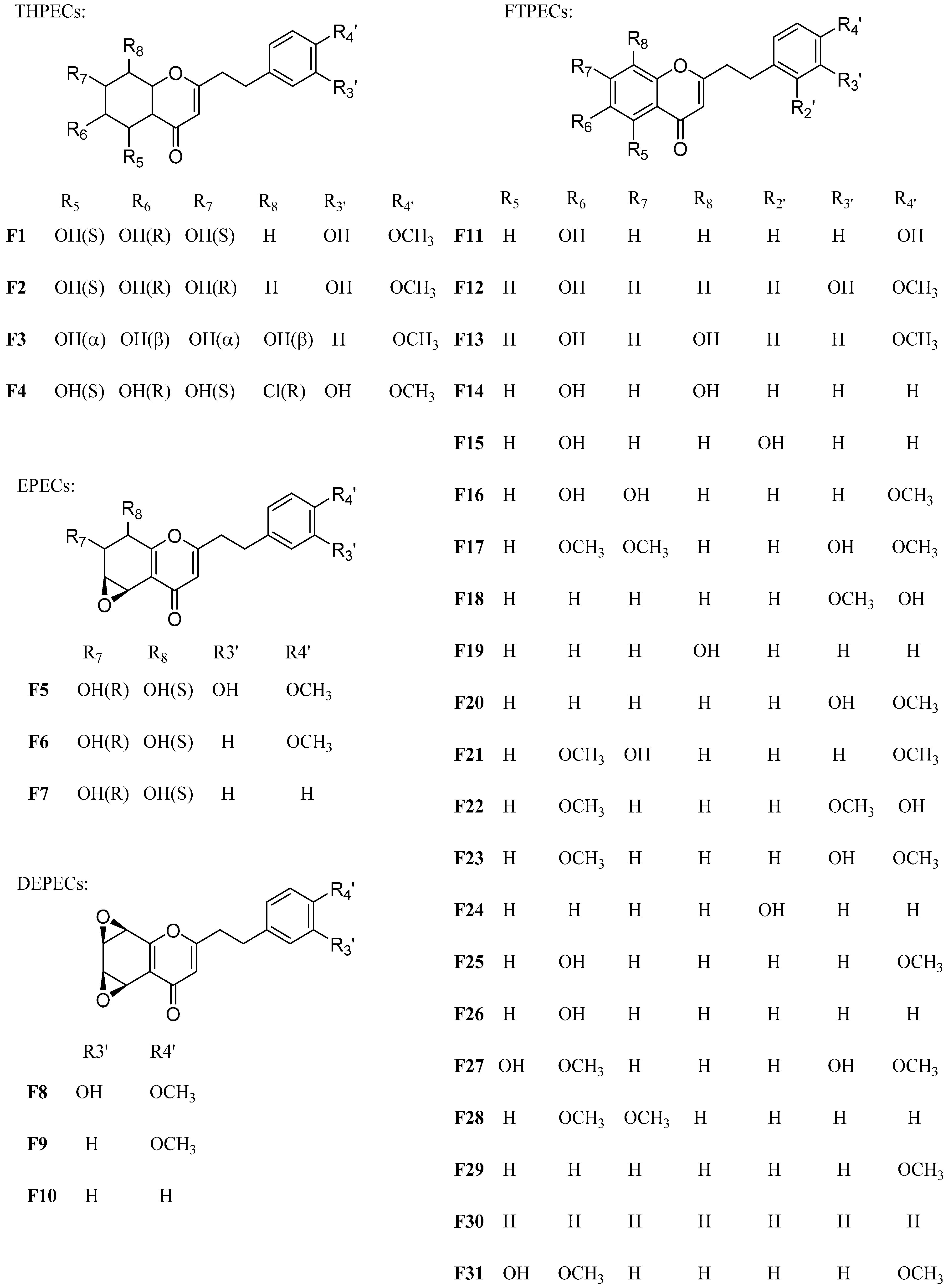
2.1. Characteristic Fragmentation Behavior of Reference Compounds F1–F31
2.2. The Method Deduced for Identification of Four Types of 2-(2-Phenylethyl)chromone Derivatives
- (1)
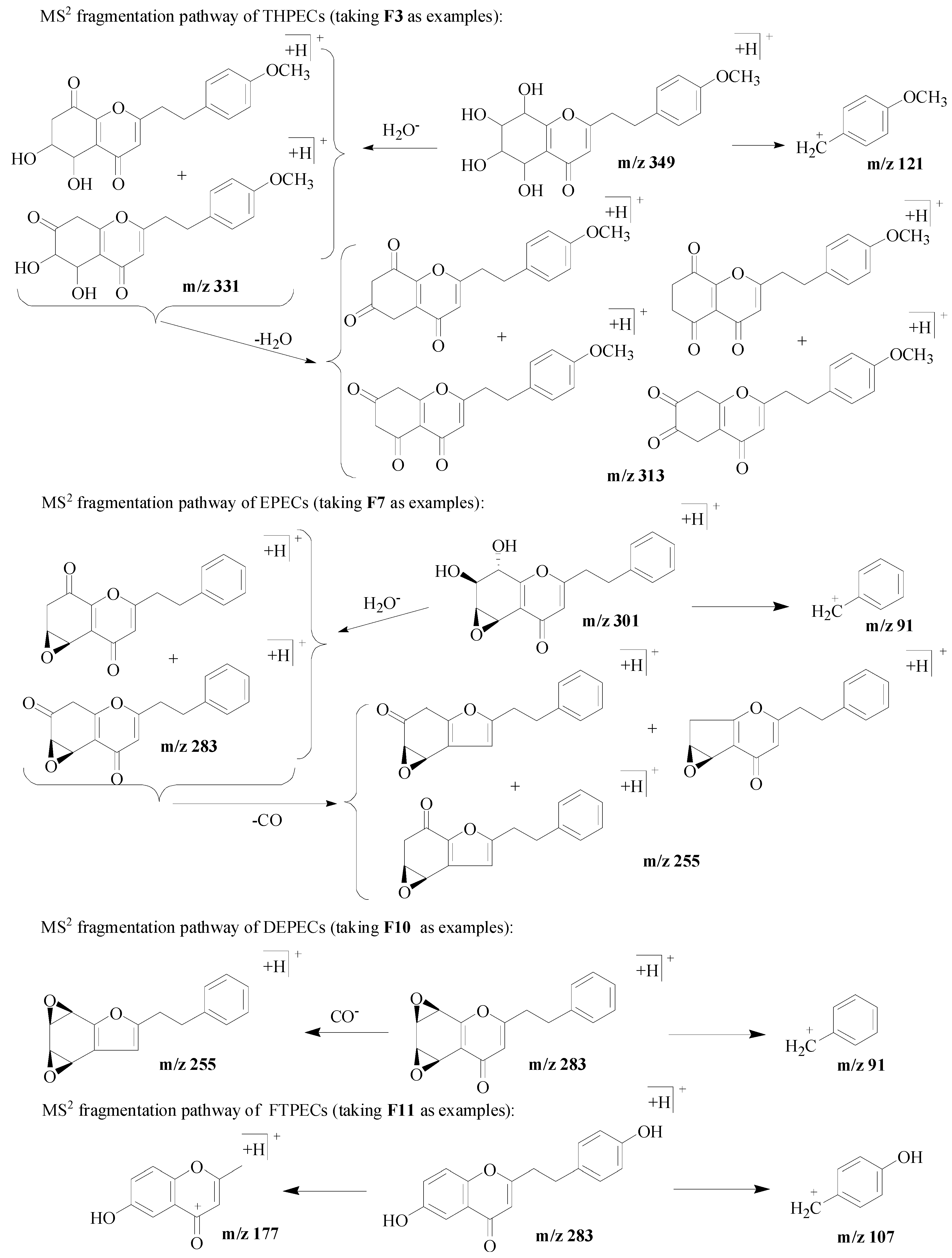
- The type of 2-(2-phenylethyl)chromone could be determined by the characteristic fragment ions as discussed above and listed in Figure 3. The present of the ions [M + H–18]+ and [M + H–18–18]+ was proposed to be THPECs. When both ions [M + H–18]+ and[M + H–28]+ were observed in the spectrum, EPECs was deduced. If only [M + H–28]+ was detected, it would be DEPECs. FTPECs only gave the peaks of benzyl ions and/or chromone moiety ions in the MS spectrum. It should be noticed that the [M + H–18]+ ion may appear as a pseudo-characteristic ion during identification, when the CH2-CH2 group between chromone moiety and phenyl moiety was substituted by a hydroxyl group (-OH) [1].
- (2)
- The four types of 2-(2-phenylethyl)chromone derivatives in agarwood have different basic skeletons (the epoxy group was assigned as a part of the basic skeletons), but substituted with similar substituent groups (mainly hydroxyl, methoxyl group and/ or chlorine atom). The molecular weights of the basic skeletons of THPECs, EPECs, DEPECs and FTPECs were 254, 268, 282 and 250, respectively. Thus, the number of different substituents of the structure could be deduced by Formula (1), where “MW” meant the molecular weight, “a” meant the number of methoxy groups, “b” represented the number of hydroxyl groups, and “c” meant the number of chlorine atom.
- (3)
- The four types of chromones presented the same characteristic benzyl ion, while it could not be observed in some compounds. The benzyl moiety without substituent group was 91 [C7H7]+, therefore, the number of hydroxyl and/or methoxyl groups substituted on the benzyl moiety could be deduced according to Formula (2), where “MWbm” means the molecular weight of benzyl ion, “abm” means the number of methoxy groups on the benzyl moiety, “bbm” represents the number of hydroxyl groups on the benzyl moiety. For example, according to Formula (2), the hydroxyl- or methoxyl-substituted benzyl moiety provided characteristic ions at m/z 107 [C7H6 + OH]+, 121 [C7H6 + OCH3]+, 137 [C7H5 + OH + OCH3]+, 151 [C7H5 + 2OCH3]+:MWbm − 91 = 30abm + 16bbm
- (4)
- Except for FTPECs, the fragment ion of the chromone moiety was rarely observed in the MS2 spectra of THPECs, EPECs and DEPECs, nevertheless the number of substituent groups of the chromone moiety could still be calculated by subtracting the number of substituents on the benzyl moiety deduced by Formula (2) from the number of substituents on the whole structure deduced by Formula (1).For FTPECs with hydroxyl-substituted benzyl moieties, according to [3,6], if one of characteristic fragment ions at m/z 161 [C10H8O2 + H], 177 [C10H7O2 + OH + H], 191 [C10H7O2 + OCH3 + H], 193 [C10H6O2 + 2OH + H], 207 [C10H6O2 + OCH3 + OH + H], 221 [C10H6O2 + 2OCH3 + H] from different substituted chromone moieties was observed, a 4′/2′-OH substituent on the benzyl moiety was deduced. Otherwise, if none of them was observed, a hydroxyl group substituted at the 3′ position of the benzyl moiety was deduced, whether an ion of chromone moiety at m/z 160, 176, 190, 192, 206, 220 appeared or not. The above mentioned MS characterization has been confirmed by the FTPEC reference compounds in Table 1. The observed fragment ions of the chromone moieties of F11, F15, F18, F22 and F24 with 4′/2′-OH on the benzyl moiety were m/z 177, 177, 161, 191 and 161, respectively, which were not observed for the F12, F17, F20, F23 and F27 with 3′-OH on the benzyl moiety, while only a fragment ion at m/z 220 of F17 was observed. These chromone moiety ions could be used to suggest the position of hydroxyl groups on the benzyl moieties.
- (5)
- Following the above procedure, a compound could be characterized as one of the four types of 2-(2-phenylethyl)chromones, as well as calculated the numbers of substituent groups of the chromone moiety and benzyl moiety. Furthermore, the position of substituent groups could be deduced sometimes. Besides, if more than one structure were suggested for a single peak, the comparison of retention time and MS spectra with reference compounds was performed. The compound was identified when the data were the same to the reference compound, otherwise, it might be a new compound.
2.3. Identification of 2-(2-Phenylethyl)chromones According to MS Characterization
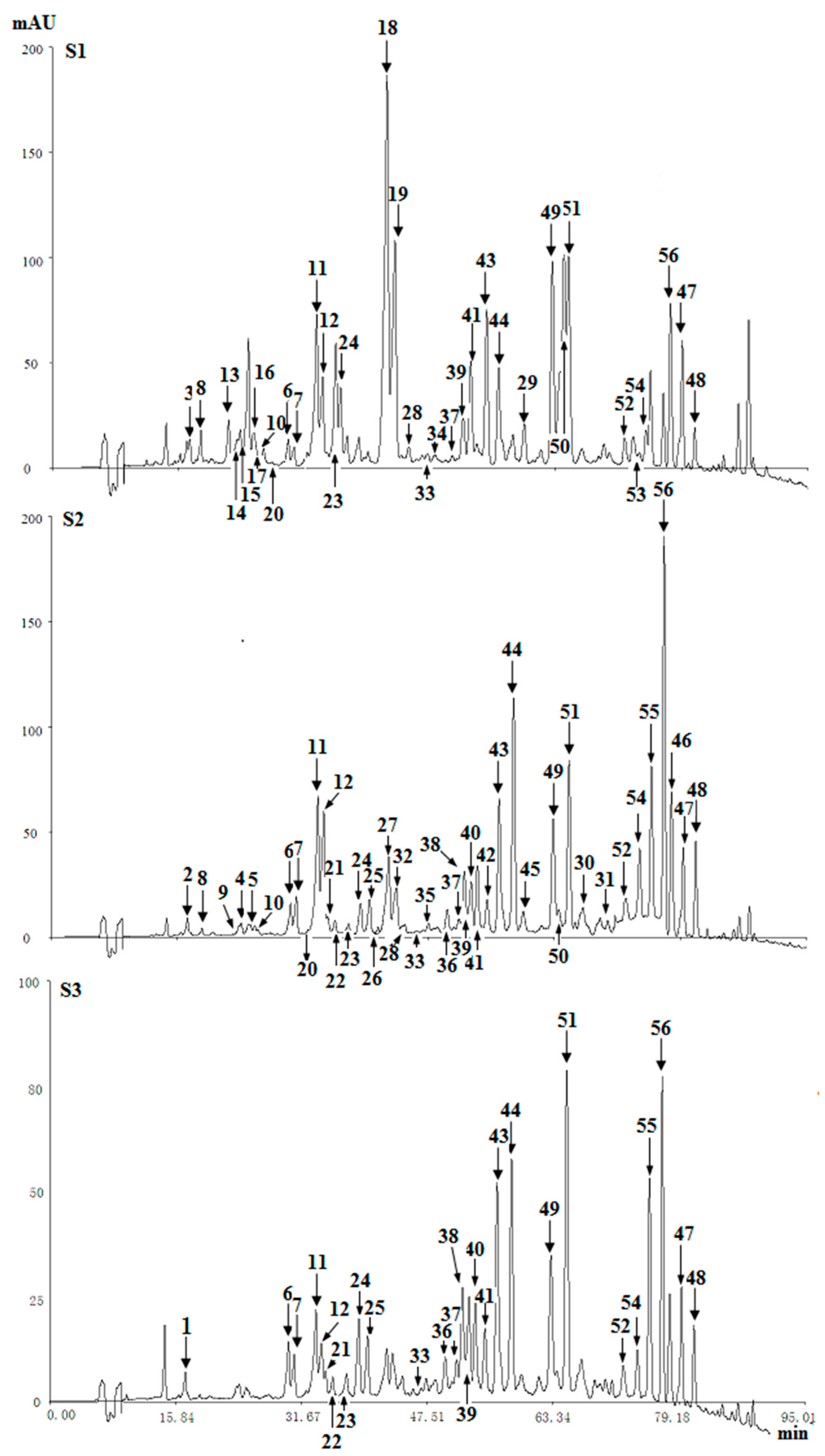
2.3.1. Structural Analysis of THPECs
2.3.2. Structural Analysis of EPECs
2.3.3. Structural Analysis of DEPECs
2.3.4. Structural Analysis of FTPECs
3. Experimental Section
3.1. Chemicals and Materials
3.2. Preparation of Sample Solutions and Reference Compound Solutions
3.3. HPLC Chromatographic Condition
3.4. MS Spectrometry
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Naef, R. The volatile and semi-volatile constituents of agarwood, the infected heartwood of Aquilaria species: A review. Flavour Fragr. J. 2011, 26, 73–87. [Google Scholar] [CrossRef]
- Espinoza, E.O.; Lancaster, C.A.; Kreitals, N.M.; Hata, M.; Cody, R.B.; Blanchette, R.A. Distinguishing wild from cultivated agarwood (Aquilaria spp.) using direct analysis in real time and time of-flight mass spectrometry. Rapid Commun. Mass Spectrom. 2014, 28, 281–289. [Google Scholar] [CrossRef] [PubMed]
- Mei, W.L.; Yang, D.L.; Wang, H.; Yang, J.L.; Zeng, Y.B.; Guo, Z.K.; Dong, W.H.; Li, W.; Dai, H.F. Characterization and Determination of 2-(2-Phenylethyl)chromones in Agarwood by GC-MS. Molecules. 2013, 18, 12324–12345. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.Q.; Wei, J.H.J.S.; Zheng, Z.; Yang, Y.; Gao, Z.H.; Sui, C.; Gong, B. Chemical Constituents of Agarwood Originating from the Endemic Genus Aquilaria Plants. Chem. Biodivers. 2012, 9, 236–250. [Google Scholar] [CrossRef] [PubMed]
- Lancaster, C.; Espinoza, E. Evaluating agarwood products for 2-(2-phenylethyl)chromones using direct analysis in real time time-of-flight mass spectrometry. Rapid Commun. Mass Spectrom. 2012, 26, 2649–2656. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.L.; Xia, B.; Jiang, Y.; Mei, W.L.; Kuckc, D. Fragmentation of protonated 2-(2-phenylethyl)chromones from agarwood: The diagnostic role of ion/neutral complexes as reactive intermediates. Eur. J. Mass Spectrom. 2015, 21, 609–621. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; Bo, W.; Sung, W.K.; Gwi, S.H.; Jeong, H.P. Eight New 2-(2-Phenylethyl)chromone (¼2-(2-Phenylethyl)-4H-1-benzopyran-4-one) Derivatives from Aquilaria malaccensis Agarwood. Helv. Chim. Acta 2012, 95, 1657–1665. [Google Scholar] [CrossRef]
- Dai, H.F.; Liu, J.; Zeng, Y.B.; Han, Z.; Wang, H.; Mei, W.L. A New 2-(2-Phenylethyl)Chromone from Chinese Eaglewood. Molecules 2009, 14, 5165–5168. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Xu, Z.R.; Chai, X.Y.; Zeng, K.W.; Jia, Y.X.; Bi, D.; Ma, Z.Z.; Tu, P.F. Nine 2-(2-Phenylethyl)chromone Derivatives from the Resinous Wood of Aquilaria sinensis and Their Inhibition of LPS-Induced NO Production in RAW 264.7 Cells. Eur. J. Org. Chem. 2012, 2012, 5389–5397. [Google Scholar] [CrossRef]
- Yagura, T.; Shibayama, N.; Ito, M.; Kiuchib, F.; Honda, G. Three novel diepoxy tetrahydrochromones from agarwood artificially produced by intentional wounding. Tetrahedron Lett. 2005, 46, 4395–4398. [Google Scholar] [CrossRef]
- Wei, L.; Cai, H.C.; Wen, H.D.; Zhi, K.G.; Hao, W.; Wen, L.M.; Hao, F.D. 2-(2-Phenylethyl)chromone derivatives from Chinese agarwood induced by artificial holing. Fitoterapia 2014, 98, 117–123. [Google Scholar]
- Xia, B.; Li, J.R.; Yang, D.L.; Mei, W.L.; Ding, L.S.; Zhou, Y. A rapid and highly specific method to evaluate the presence of 2-(2-phenylethyl)chromones in agarwood by supercritical fluid chromatography-mass spectrometry. Eur. J. Mass Spectrom. 2014, 20, 395–402. [Google Scholar] [CrossRef]
- Li, J.; Chen, D.; Jiang, Y.; Zhang, Q.; Zhang, L.; Tu, P.F. Identification and quantification of 5,6,7,8-tetrahydro-2-(2-phenylethyl)chromones in Chinese eaglewood by HPLC with diode array detection and MS. J. Sep. Sci. 2013, 36, 3733–3740. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wu, J.; Zhao, Y.X.; De, Y.Y.; Mei, W.L.; Dai, H.F. A new cytotoxic 2-(2-phenylethyl)chromone from Chinese eaglewood. Chin. Chem. Lett. 2008, 9, 934–936. [Google Scholar] [CrossRef]
- Yang, D.L.; Mei, W.L.; Zeng, Y.B.; Guo, Z.K.; Zhao, Y.X.; Wang, H.; Zuo, W.J.; Dong, W.H.; Wang, Q.H.; Dai, H.F. 2-(2-phenylethyl)chromone derivatives in Chinese Agarwood “Qi-Nan” from Aquilaria sinensis. Planta Med. 2013, 79, 1329–1334. [Google Scholar] [CrossRef] [PubMed]
- Dai, H.F.; Liu, J.; Han, Z.; Zeng, Y.B.; Wang, H. Two new 2-(2-phenylethyl)chromones from Chinese eaglewood. J. Asian Nat. Prod. Res. 2010, 12, 134–137. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.L.; Wang, H.; Guo, Z.K.; Dong, W.H.; Mei, W.L.; Dai, H.F. A new 2-(2-phenylethyl)chromone derivative in Chinese agarwood ‘Qi-Nan’ from Aquilaria sinensis. J. Asian Nat. Prod. Res. 2014, 16, 770–776. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Not available.

| Type | No. | tR/min | Fragment Ions (m/z) | Name | |||||
|---|---|---|---|---|---|---|---|---|---|
| [M + H]+ | [M + H–18]+ | [M + H–18–18]+ | [M + H–18–28]+ | CM | BM | ||||
| THPECs | F1 | 12.9 | 349 | 331 | 313 | 137 | (5S*,6R*,7S*)-5,6,7-Trihydroxy-2-[2-(3-hydroxy-4-methoxyphenyl)ethyl]-5,6,7,8-tetrahydrochromone | ||
| F2 | 13.2 | 349 | 331 | 313 | 137 | (5S*,6R*,7R*)-5,6,7-Trihydroxy-2-[2-(3-hydroxy-4-methoxyphenyl)ethyl]-5,6,7,8-tetrahydrochromone | |||
| F3 | 15.2 | 349 | 331 | 313 | 121 | 5α,6β,7α,8β-Tetrahydroxy-2-[2-(4-methoxyphenyl)-ethyl]-5,6,7,8-tetrahydrochromone | |||
| F4 | 17.3 | 383 | 365 | 347 | 137 | (5S*,6R*,7S*,8R*)-8-Chloro-5,6,7-trihydroxy-2-[2-(3-hydroxy-4-methoxyphenyl)ethyl]-5,6,7,8-tetrahydro-chromone | |||
| EPECs | F5 | 18.3 | 347 | 329 | 301 | 137 | 5,6-Edroxy-7,8-dihydroxy-2-[2-(3-hydroxy-4-methoxy)phenylethyl]-5,6,7,8-tetrahydrochromone | ||
| F6 | 33.1 | 331 | 313 | 285 | 121 | 5,6-Edroxy-7,8-dihydroxy-2-[2-(4-methoxy)phenyl-ethyl]-5,6,7,8-tetrahydrochromone | |||
| F7 | 33.8 | 301 | 283 | 255 | 91 | 5,6-Edroxy-7,8-dihydroxy-2-(2-phenylethyl)-5,6,7,8-tetrahydrochromone | |||
| DEPECs | F8 | 24.4 | 329 | 301 | 137 | 5,6:7,8-Diedroxy-2-[2-(3-hydroxy-4-methoxy)phenyl-ethyl]-5,6,7,8-tetrahydrochromone | |||
| F9 | 42.6 | 313 | 285 | 121 | Oxidoagarochromones B | ||||
| F10 | 43.5 | 283 | 255 | 91 | Oxidoagarochromones A | ||||
| FTPECs | F11 | 34.7 | 283 | 177 | 107 | 6-Hydroxy-2-[2-(4-hydroxy)phenylethyl]chromone | |||
| F12 | 36.6 | 313 | 137 | 6-Hydroxy-2-[2-(3-hydroxy-4-methoxy)phenylethyl]-chromone | |||||
| F13 | 38.3 | 313 | 192 | 121 | 6,8-Dihydroxy-2-[2-(4-methoxy)phenylethyl]chromone | ||||
| F14 | 39.5 | 283 | 192 | 91 | 6,8-Dihydroxy-2-(2-phenylethyl)chromone | ||||
| F15 | 41.5 | 283 | 177 | 107 | 6-Hydroxy-2-[2-(2-hydroxy)phenylethyl]chromone | ||||
| F16 | 42.4 | 313 | 121 | 6,7-Dihydroxy-2-[2-(4-methoxy)phenylethyl]chromone | |||||
| F17 | 43.2 | 357 | 220 | 137 | 6,7-Dimethoxy-2-[2-(3-hydroxy-4-methoxy)phenyl-ethyl]chromone | ||||
| F18 | 47.5 | 297 | 161 | 137 | 2-[2-(3-Methoxy-4-hydroxy)phenylethyl]chromone | ||||
| F19 | 49.8 | 267 | 176 | 91 | 8-Hydroxy-2-(2-phenylethyl)chromone | ||||
| F20 | 50.4 | 296 | 137 | 2-[2-(3-Hydroxy-4-methoxy)phenylethyl]chromone | |||||
| F21 | 51.2 | 327 | 121 | 6-Methoxy-7-hydroxy-2-[2-(4-methoxy)phenylethyl]-chromone | |||||
| F22 | 52.3 | 327 | 191 | 137 | 6-Methoxy-2-[2-(3-methoxy-4-hydroxy)phenylethyl]-chromone | ||||
| F23 | 54.3 | 327 | 137 | 6-Methoxy-2-[2-(3-hydroxy-4-methoxy)phenylethyl]-chromone | |||||
| F24 | 55.2 | 267 | 161 | 107 | 2-[2-(2-Hydroxy)phenylethyl]chromone | ||||
| F25 | 56.5 | 297 | 121 | 6-Hydroxy-2-[2-(4-methoxy)phenylethyl]chromone | |||||
| F26 | 58.3 | 267 | 176 | 91 | 6-Hydroxy-2-(2-phenylethyl)chromone | ||||
| F27 | 59.1 | 343 | 137 | 5-Hydroxy-6-methoxy-2-[2-(3-hydroxy-4-methoxy)-phenylethyl]chromone | |||||
| F28 | 64.9 | 311 | 220 | 91 | 6,7-Dimethoxy-2-(2-phenylethyl)chromone | ||||
| F29 | 72.0 | 281 | 121 | 2-[2-(4-Methoxy)phenylethyl]chromone | |||||
| F30 | 73.8 | 251 | 160 | 91 | 2-(2-Phenylethyl)chromone | ||||
| F31 | 78.7 | 326 | 121 | 5-Hydroxy-6-methoxy-2-[2-(4-methoxy)phenylethyl]-chromone | |||||
| No. | tR (min) | [M + H]+ (m/z) | (+)Fragment Ions (m/z) | OCH3 | OH | Identification | RFC | RC (%) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| [M + H–18]+ | [M + H–18–18]+ | [M + H–18–28]+ | CM | BM | S1 | S2 | S3 | |||||||
| THPECs | ||||||||||||||
| 1 | 15.9 | 349 | 331 | 313 | 121 | 1 | 4 | 5α,6β,7α,8β-Tetrahydroxy-2-[2-(4-methoxyphenyl)ethyl]-5,6,7,8-tetrahydrochromone | F3 | 0.51 | ||||
| 2 | 16.0 | 319 | 301 | 283 | 4 | (5S,6S,7R)-5,6,7-Trihydroxy-2-[2-(2-hydroxyphenyl)-ethyl]-5,6,7,8-tetrahydrochromone or isomers | 0.15 | |||||||
| 3 | 16.7 | 349 | 331 | 313 | 1 | 4 | Isomer of F3 | 0.50 | ||||||
| 4 | 23.7 | 303 | 285 | 267 | 3 | rel-(5R,6S,7R)-5,6,7-Trihydroxy-2-(2-phenylethyl)-5,6,7,8-tetrahydrochromone | 0.09 | |||||||
| 5 | 24.2 | 317 | 299 | 281 | 121 | 1 | 2 | New compound (two hydroxy on CM, one methoxy on BM) | 0.08 | |||||
| 6 | 29.4 | 367 | 349 | 331 | 121 | 1 | 3 | rel-(5R,6S,7S,8R)-8-Chloro-5,6,7-trihydroxy-2-[2-(4-methoxyphenyl)ethyl]-5,6,7,8-tetrahydrochromone | 0.65 | 0.65 | 1.16 | |||
| 7 | 30.1 | 337 | 319 | 301 | 3 | (5S,6S,7S,8R)-8-Chloro-2-(2-phenylethyl)-5,6,7-trihydroxy-5,6,7,8-tetrahydrochromone or new compound | 0.43 | 0.76 | 0.80 | |||||
| EPECs | ||||||||||||||
| 8 | 18.5 | 347 | 329 | 301 | 137 | 1 | 3 | 5,6-Edroxy-7,8-dihydroxy-2-[2-(3-hydroxy-4-methoxy)-phenylethyl]-5,6,7,8-tetrahydrochromone | F5 | 1.13 | 0.12 | |||
| 9 | 23.3 | 285 | 267 | 239 | 1 | New compound (one hydroxy on CM) | 0.09 | |||||||
| 10 | 25.0 | 331 | 313 | 285 | 121 | 1 | 2 | New compound (isomer of F6) | 0.94 | 0.15 | ||||
| 11 | 33.0 | 331 | 313 | 285 | 121 | 1 | 2 | 5,6-Edroxy-7,8-dihydroxy-2-[2-(4-methoxy)phenylethyl]-5,6,7,8-tetrahydrochromone | F6 | 7.25 | 4.95 | 2.11 | ||
| 12 | 33.7 | 301 | 283 | 255 | 2 | 5,6-Edroxy-7,8-dihydroxy-2-(2-phenylethyl)-5,6,7,8-tetrahydrochromone | F7 | 3.26 | 2.76 | 0.85 | ||||
| DEPECs | ||||||||||||||
| 13 | 21.8 | 329 | 301 | 137 | 1 | 1 | New compound (isomer of F8) | 1.00 | ||||||
| 14 | 23.0 | 329 | 301 | 193 | 137 | 1 | 1 | New compound (isomer of F8) | 0.50 | |||||
| 15 | 23.3 | 329 | 301 | 137 | 1 | 1 | New compound (isomer of F8) | 0.28 | ||||||
| 16 | 24.2 | 329 | 301 | 137 | 1 | 1 | 5,6:7,8-Diedroxy-2-[2-(3-hydroxy-4-methoxy)phenylethyl]-5,6,7,8-tetrahydrochromone | F8 | 0.47 | |||||
| 17 | 24.6 | 329 | 301 | 137 | 1 | 1 | New compound (isomer of F8) | 0.02 | ||||||
| 18 | 42.0 | 313 | 285 | 121 | 1 | Oxidoagarochromones B | F9 | 4.85 | ||||||
| 19 | 42.9 | 283 | 255 | Oxidoagarochromones A | F10 | 0.89 | 0.21 | |||||||
| No. | tR (min) | [M + H]+ (m/z) | Fragment Ions (m/z) | CM | BM | Identification | RFC | RC (%) | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CM | BM | OCH3 | OH | OCH3 | OH | S1 | S2 | S3 | |||||
| 20 | 26.1 | 329 | 137 | 2 | 1 | 3′- | 6,8-Dihydroxy-2-[2-(3-hydroxy-4-methoxyl)phenylethyl]-chromone | 0.28 | |||||
| 21 | 34.2 | 283 | 177 | 107 | 6- | 4′- | 6-Hydroxy-2-[2-(4-hydroxy)phenylethyl]chromone | F11 | 8.6 | 0.03 | |||
| 22 | 35.0 | 313 | 177 | 137 | 1 | 1 | 4′or 2′- | 6-Hydroxy-2-[2-(3-methoxyl-4-hydroxy)phenylethyl]-chromone | 0.26 | 0.26 | |||
| 23 | 36.8 | 313 | 137 | 6- | 4′- | 3′- | 6-Hydroxy-2-[2-(3-hydroxy-4-methoxyl)phenylethyl]-chromone | F12 | 0.66 | 0.23 | 0.41 | ||
| 24 | 38.4 | 313 | 121 | 6,8- | 4′- | 6,8-Dihydroxy-2-[2-(4-methoxy)phenylethyl]chromone | F13 | 0.66 | 0.87 | 2.32 | |||
| 25 | 39.5 | 283 | 192 | 91 | 6,8- | 6,8-Dihydroxy-2-(2-phenylethyl)chromone | F14 | 0.93 | 1.81 | ||||
| 26 | 41.3 | 283 | 177 | 107 | 6- | 2′- | 6-Hydroxy-2-[2-(2-hydroxy)phenylethyl]chromone | F15 | 0.05 | ||||
| 27 | 42.7 | 313 | 121 | 6,7- | 4′- | 6,7-Dihydroxy-2-[2-(4-methoxy)phenylethyl]chromone | F16 | 2.80 | 0.46 | 0.45 | |||
| 28 | 44.0 | 283 | 192 | 91 | 2 | New compound (isomer of F14) | 1.33 | 0.52 | |||||
| 29 | 59.1 | 343 | 137 | 6- | 5- | 3′- | 4′- | 5-Hydroxy-6-methoxy-2-[2-(3-methoxy-4-hydroxy)phenyl-ethyl]chromone | F27 | 1.18 | |||
| 30 | 66.3 | 313 | 121 | 2 | 1 | 5,8-Dihydroxy-2-[2-(4-methoxy)phenylethyl]chromone | 1.82 | ||||||
| 31 | 68.5 | 283 | 192 | 91 | 2 | 5,8-Dihydroxy-2-(2-phenylethyl)chromone | 0.78 | ||||||
| 32 | 43.1 | 357 | 220 | 137 | 6,7- | 4′- | 3′- | 6,7-Dimethoxy-2-[2-(3-hydroxy-4-methoxyl)phenylethyl]-chromone | F17 | 0.46 | |||
| 33 | 46.8 | 267 | 161 | 107 | 1 | 2-[2-(4-Hydroxy)phenylethyl]chromone | 0.27 | 0.21 | 0.20 | ||||
| 34 | 47.5 | 297 | 161 | 137 | 1 | 4′- | 2-[2-(3-Methoxy-4-hydroxy)phenylethyl]chromone | F18 | 0.22 | ||||
| 35 | 48.5 | 297 | 191 | 107 | 1 | 2′ or 4′- | 6-Methoxy-2-[2-(4-hydroxy)phenylethyl]chromone or new compound | 0.16 | |||||
| 36 | 49.2 | 267 | 176 | 91 | 8- | 8-Hydroxy-2-(2-phenylethyl)chromone | F19 | 0.78 | 1.16 | ||||
| 37 | 50.3 | 297 | 121 | 1 | 1 | 6-Hydroxy-2-[2-(4-methoxy)phenylethyl]chromone or new compound | 0.16 | 0.04 | 1.16 | ||||
| 38 | 51.4 | 327 | 121 | 6- | 7- | 4′- | 6-Methoxy-7-hydroxy-2-[2-(4-methoxy)phenylethyl]-chromone | F20 | 1.27 | 1.57 | 2.55 | ||
| 39 | 52.3 | 327 | 191 | 137 | 6- | 3′- | 4′- | 6-Methoxy-2-[2-(3-methoxy-4-hydroxy)phenylethyl]-chromone | F22 | 2.88 | 0.85 | 1.88 | |
| 40 | 53.0 | 297 | 206 | 1 | 1 | 6-Hydroxy-7-methoxy-2-(2-phenylethyl)chromone | 1.74 | 2.32 | |||||
| 41 | 54.3 | 327 | 137 | 6- | 4′- | 3′- | 6-Methoxy-2-[2-(3-hydroxy-4-methoxy)phenylethyl]-chromone | F23 | 5.43 | 2.06 | 1.98 | ||
| 42 | 54.9 | 267 | 161 | 107 | 2′- | 2-[2-(2-Hydroxy)phenylethyl]chromone | F24 | 1.06 | |||||
| 43 | 55.8 | 297 | 121 | 1 | 1 | 6-Hydroxy-2-[2-(4-methoxy)phenylethyl]chromone or new compound | F25 | 4.25 | 5.36 | 8.17 | |||
| 44 | 57.6 | 267 | 176 | 91 | 6- | 6-Hydroxy-2-(2-phenylethyl)chromone | F26 | 1.42 | 6.64 | 8.17 | |||
| 45 | 58.7 | 297 | 191 | 107 | 1 | 2′ or 4′- | 6-Methoxy-2-[2-(4-hydroxy)phenylethyl]chromone or new compound | 0.27 | |||||
| 46 | 78.2 | 357 | 121 | 2 | 1 | 1 | New compound (two methoxyls and one hydroxyl on CM, one methoxyl on BM) | 1.00 | |||||
| 47 | 78.7 | 327 | 121 | 6- | 5- | 4′- | 5-Hydroxy-6-methoxy-2-[2-(4-methoxy)phenylethyl]-chromone | F31 | 4.31 | 2.79 | 3.47 | ||
| 48 | 80.3 | 297 | 206 | 91 | 1 | 1 | 5-Hydroxy-6-methoxy-2-(2-phenylethyl)chromone | 1.36 | 2.58 | 2.23 | |||
| 49 | 62.5 | 341 | 121 | 2 | 1 | 6,7-Dimethoxy-2-[2-(4-methoxy)phenylethyl]chromone | 5.67 | 5.06 | 4.43 | ||||
| 50 | 63.3 | 341 | 151 | 1 | 2 | New compound (one methoxyl on CM, two methoxyls on BM) | 1.28 | 0.51 | |||||
| 51 | 64.8 | 311 | 220 | 91 | 6,7- | 6,7-Dimethoxy-2-(2-phenylethyl)chromone | F28 | 5.58 | 6.41 | 11.93 | |||
| 52 | 71.7 | 281 | 121 | 4′- | 2-[2-(4-Methoxy)phenylethyl]chromone | F29 | 0.62 | 0.98 | 1.18 | ||||
| 53 | 73.2 | 281 | 190 | 91 | 1 | 6-Methoxy-2-(2-phenylethyl)chromone or new compound | 0.17 | ||||||
| 54 | 73.4 | 251 | 160 | 91 | 2-(2-phenylethyl)chromone | F30 | 0.17 | 1.05 | 1.52 | ||||
| 55 | 74.7 | 311 | 121 | 1 | 1 | 6-Methoxy-2-[2-(3-methoxy)phenylethyl]chromone or 6-methoxy-2-[2-(4-methoxy)phenylethyl]chromone | 4.89 | 6.59 | |||||
| 56 | 76.3 | 281 | 190 | 91 | 1 | 6-Methoxy-2-(2-phenylethyl)chromone or new compound | 2.28 | 10.6 | 9.34 | ||||
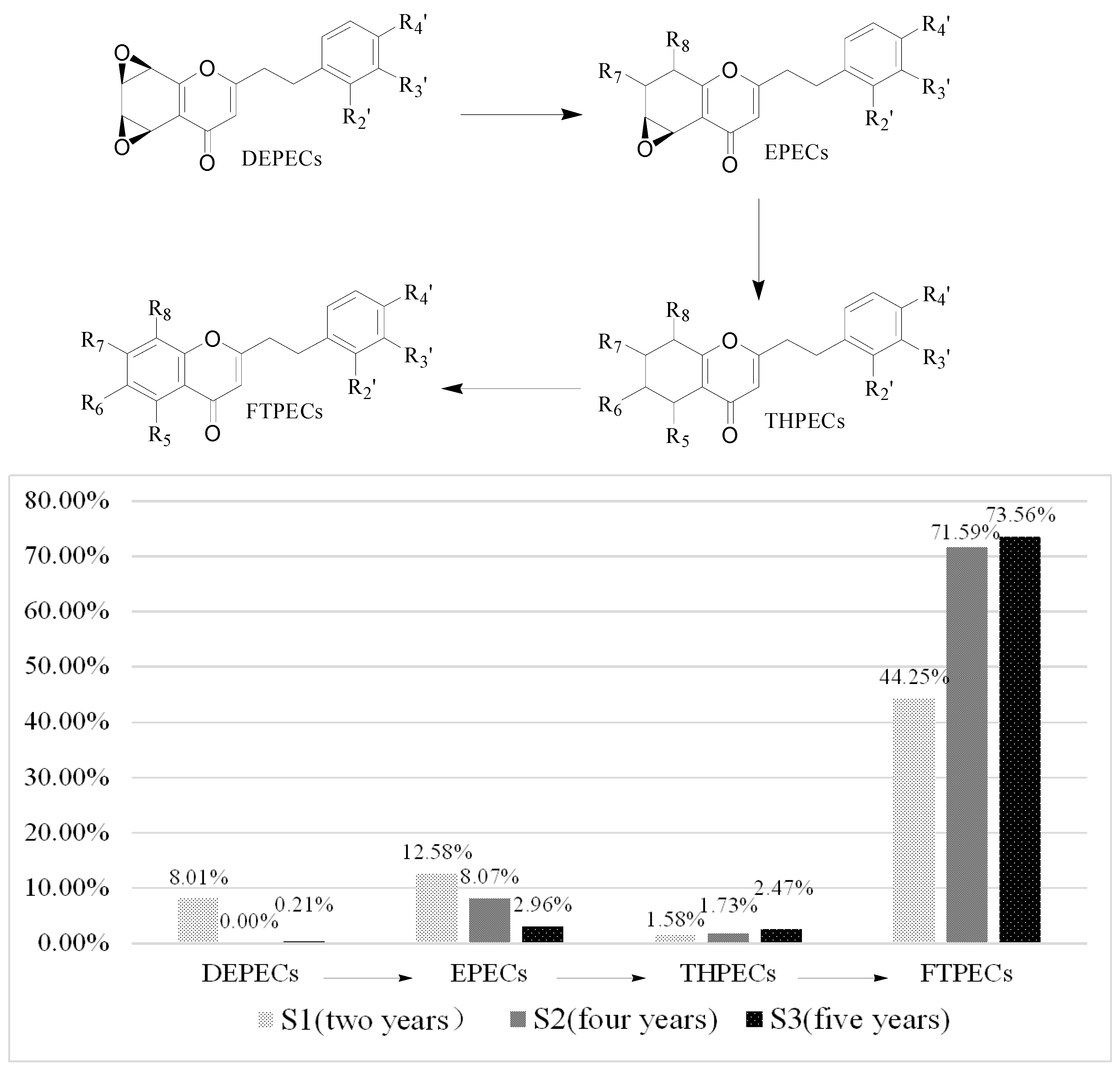
| N(RC/%) | DEPECs | EPECs | Total (E) | THPECs | FTPECs | Total (all) |
|---|---|---|---|---|---|---|
| S1 | 7 (8.01%) | 4 (12.58%) | 11 (20.59%) | 3 (1.58%) | 23 (44.25%) | 37 (66.42%) |
| S2 | 0 (0%) | 5 (8.07%) | 5 (8.07%) | 5 (1.73%) | 33 (71.59%) | 43 (81.39%) |
| S3 | 1 (0.21%) | 2 (2.96%) | 3 (3.17%) | 3 (2.47%) | 23 (73.56%) | 29 (79.20%) |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, J.L.; Dong, W.H.; Kong, F.D.; Liao, G.; Wang, J.; Li, W.; Mei, W.L.; Dai, H.F. Characterization and Analysis of 2-(2-Phenylethyl)-chromone Derivatives from Agarwood (Aquilaria crassna) by Artificial Holing for Different Times. Molecules 2016, 21, 911. https://doi.org/10.3390/molecules21070911
Yang JL, Dong WH, Kong FD, Liao G, Wang J, Li W, Mei WL, Dai HF. Characterization and Analysis of 2-(2-Phenylethyl)-chromone Derivatives from Agarwood (Aquilaria crassna) by Artificial Holing for Different Times. Molecules. 2016; 21(7):911. https://doi.org/10.3390/molecules21070911
Chicago/Turabian StyleYang, Jin Ling, Wen Hua Dong, Fan Dong Kong, Ge Liao, Jun Wang, Wei Li, Wen Li Mei, and Hao Fu Dai. 2016. "Characterization and Analysis of 2-(2-Phenylethyl)-chromone Derivatives from Agarwood (Aquilaria crassna) by Artificial Holing for Different Times" Molecules 21, no. 7: 911. https://doi.org/10.3390/molecules21070911
APA StyleYang, J. L., Dong, W. H., Kong, F. D., Liao, G., Wang, J., Li, W., Mei, W. L., & Dai, H. F. (2016). Characterization and Analysis of 2-(2-Phenylethyl)-chromone Derivatives from Agarwood (Aquilaria crassna) by Artificial Holing for Different Times. Molecules, 21(7), 911. https://doi.org/10.3390/molecules21070911








