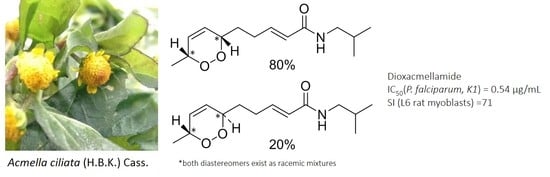
A New Alkamide with an Endoperoxide Structure from Acmella ciliata (Asteraceae) and Its in Vitro Antiplasmodial Activity
Abstract
:1. Introduction
2. Results and Discussion
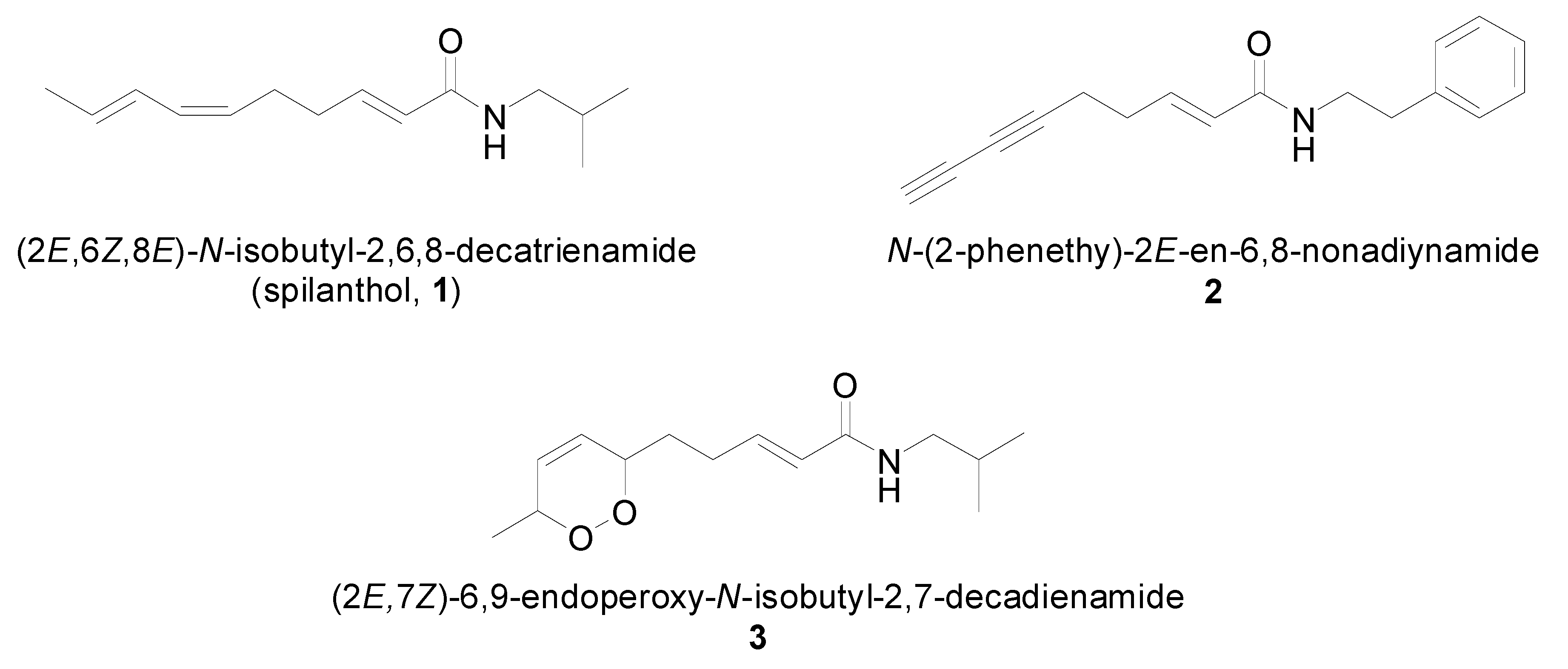
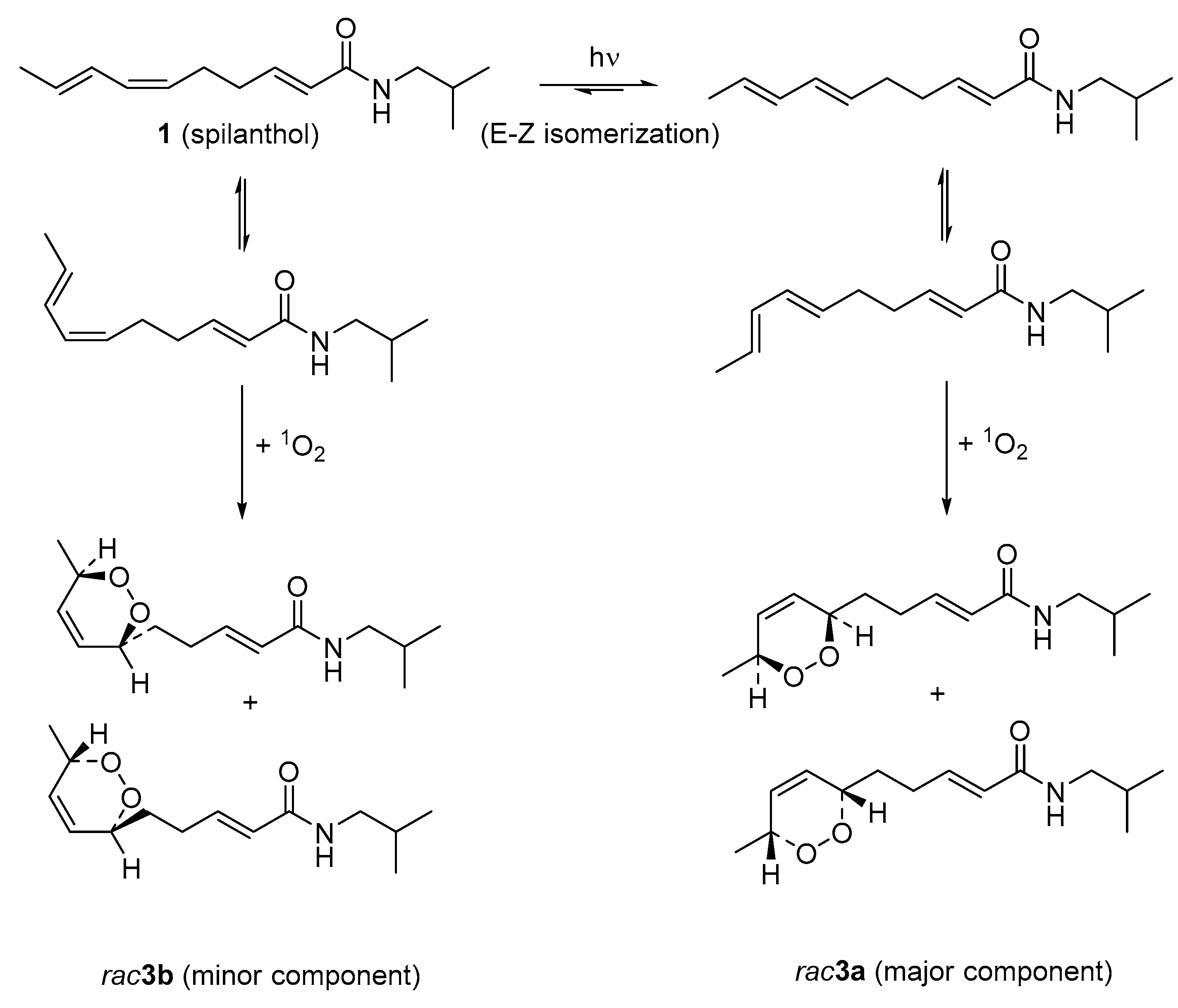
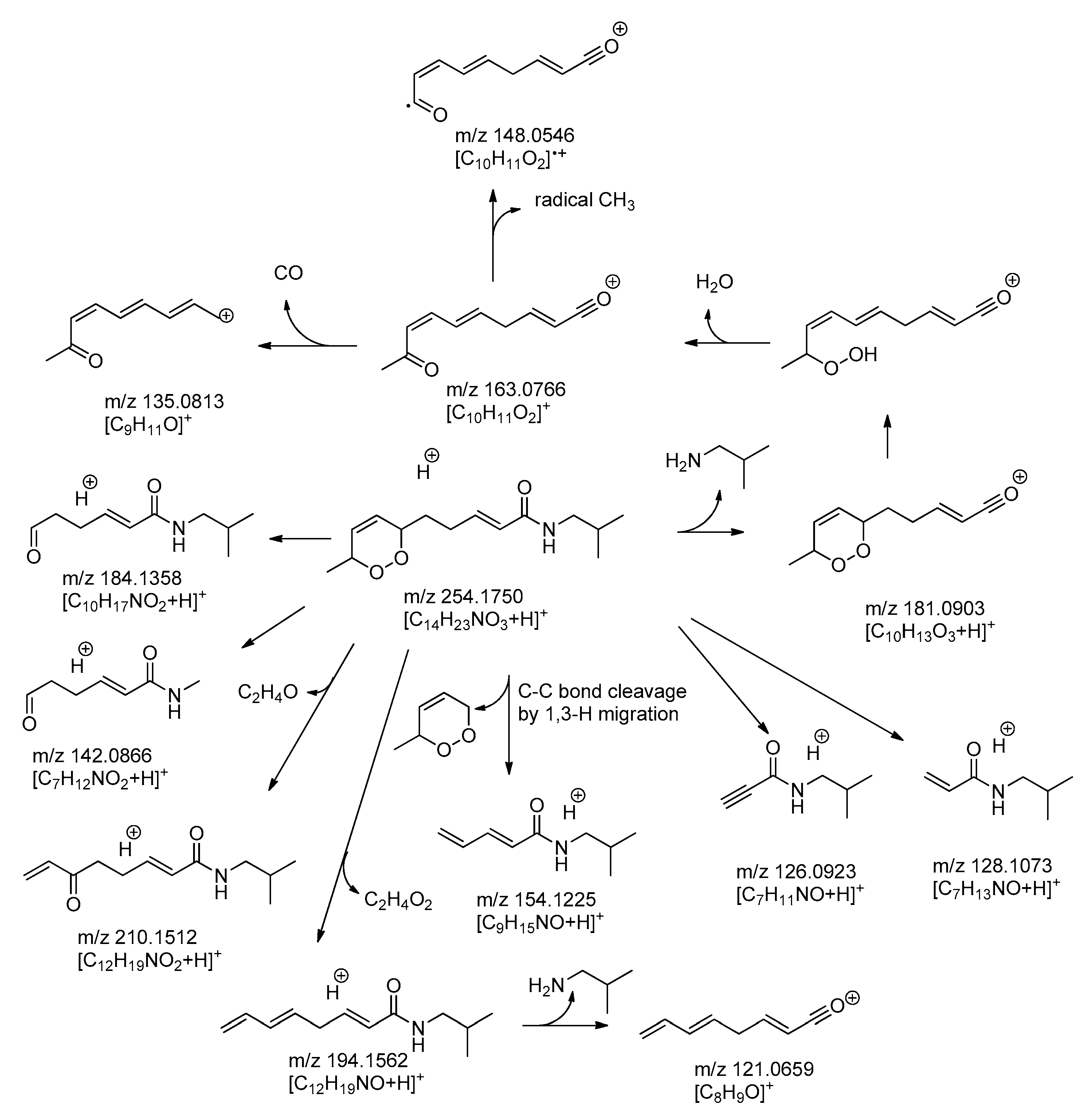
2.1. Isolation and Characterization of Alkamides
2.2. In Vitro Antiplasmodial Activity of Isolated Alkamides
3. Materials and Methods
3.1. General Procedures
3.2. Botanical Material
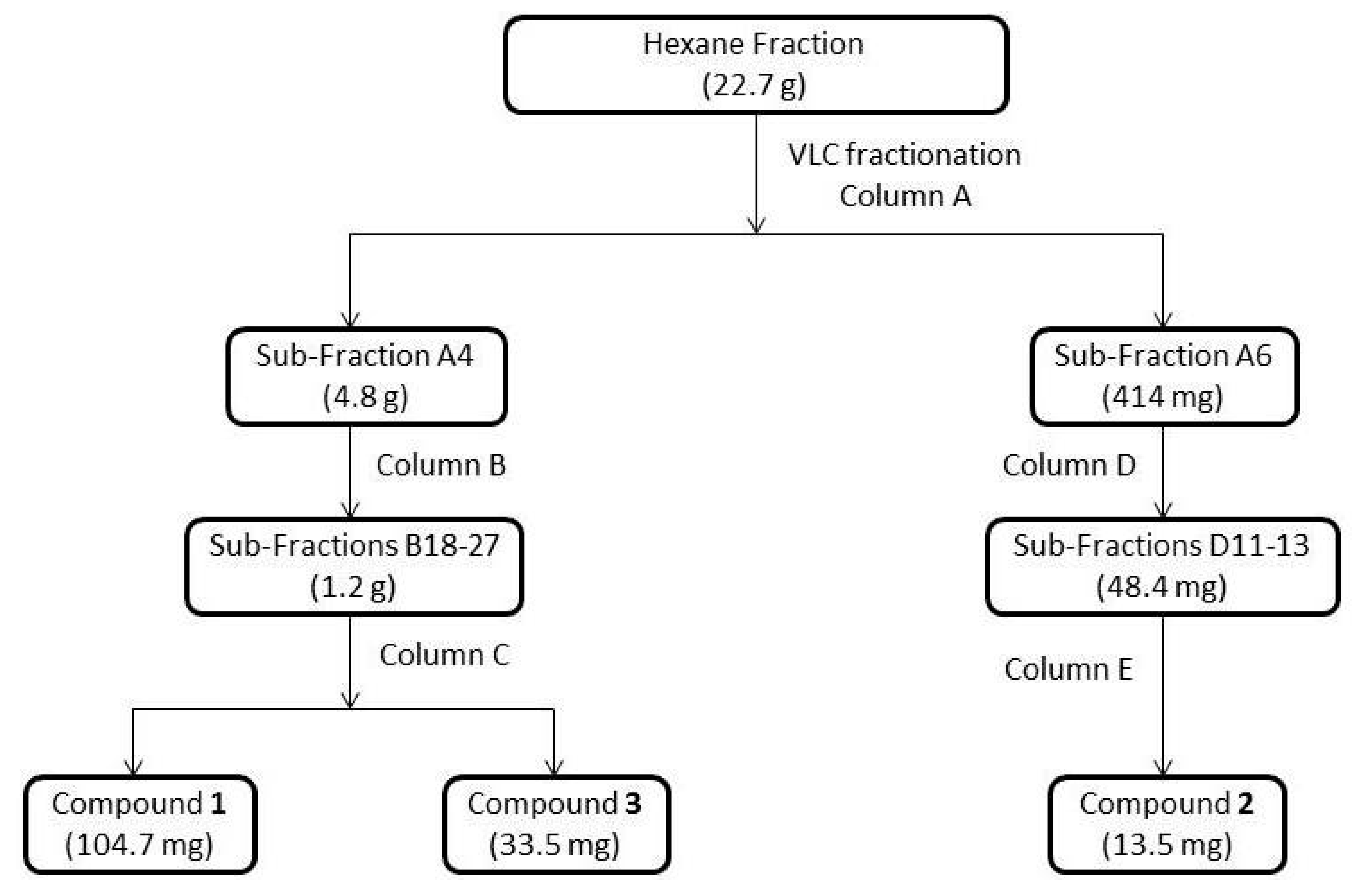
3.3. Extraction and Isolation
3.4. Biological Activity Tests
3.5. Molecular Modeling and NMR Shift Calculations for Compound 3
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jansen, R.K. The systematics of Acmella (Asteraceae-Heliantheae). Syst. Bot. Monogr. 1985, 8, 115. [Google Scholar] [CrossRef]
- Kasper, J.; Melzig, M.F.; Jenett-Siems, K. New phenolic compounds of Acmella ciliata. Planta Med. 2010, 76, 633–635. [Google Scholar] [CrossRef] [PubMed]
- Martin, R.; Becker, H. Spilanthol-related amides from Acmella ciliata. Phytochem. 1984, 23, 1781–1783. [Google Scholar] [CrossRef]
- Martin, R.; Becker, H. Amides and other constituents from Acmella ciliata. Phytochem. 1985, 24, 2295–2300. [Google Scholar] [CrossRef]
- Rios, M.Y.; Guadarrama, A.B.A.; Gutiérrez, M.C. Analgesic activity of affinin, an alkamide from Heliopsis longipes (Compositae). J. Ethnopharmacol. 2007, 110, 364–367. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.C.; Fan, N.C.; Lin, M.H.; Chu, I.R.; Huang, H.J.; Hu, C.Y.; Han, S.Y. Anti-inflammatory effect of spilanthol from Spilanthes acmella on murine macrophage by down-regulating LPS-induced inflammatory mediators. J. Agric. Food Chem. 2008, 56, 2341–2349. [Google Scholar] [CrossRef] [PubMed]
- Spelman, K.; Depoix, D.; McCray, M.; Mouray, E.; Grellier, P. The traditional medicine Spilanthes acmella, and the alkylamides spilanthol and undeca-2E-ene-8,10-diynoic acid isobutylamide, demonstrate in vitro and in vivo antimalarial activity. Phytother. Res. 2011, 25, 1098–1101. [Google Scholar] [CrossRef] [PubMed]
- Gasquet, M.; Delmas, F.; Timon-David, P.; Keita, A.; Guindo, M.; Koita, N.; Diallo, D.; Doumbo, O. Evaluation in vitro and in vivo of a traditional antimalarial, “Malarial 5”. Fitoterapia 1993, 64, 423–426. [Google Scholar]
- World Health Organization. World Malaria Report 2015. Available online: http://www.who.int/malaria/publications/world-malaria-report-2015/report/en (accessed on 26 May 2016).
- Kong, L.Y.; Tan, R.X. Artemisinin, a miracle of Traditional Chinese Medicine. Nat. Prod. Rep. 2015, 32, 1617–1621. [Google Scholar] [CrossRef] [PubMed]
- Althaus, J.B.; Kaiser, M.; Brun, R.; Schmidt, T.J. Antiprotozoal activity of Achillea ptarmica (Asteraceae) and its main alkamide constituents. Molecules 2014, 19, 6428–6438. [Google Scholar] [CrossRef] [PubMed]
- Nakatani, N.; Nagashima, M. Pungent alkamides from Spilanthes acmella L. var oleracea Clarke. Biosci. Biotechnol. Biochem. 1992, 56, 759–762. [Google Scholar] [CrossRef]
- Constantino, M.G.; Silva, G.V.J.; Heleno, V.C.G.; Borin, I.A. Simulação de desdobramentos complexos de espectros de RMN de 1H. Quim. Nova 2006, 29, 160–167. [Google Scholar] [CrossRef]
- Martínez, S.L.; Guadarrama, A.B.A.; Rios, M.Y. Minor alkamides from Heliopsis longipes S.F. Blake (Asteraceae) fresh roots. Phytochem. Lett. 2011, 4, 275–279. [Google Scholar] [CrossRef]
- Devkota, K.P.; Wilson, J.; Henrich, C.J.; McMahon, J.B.; Reilly, K.M.; Beutler, J.A. Isobutylhydroxyamides from the pericarp of nepalese Zanthoxylum armatum inhibit NF1-defective tumor cell line growth. J. Nat. Prod. 2013, 76, 59–63. [Google Scholar] [CrossRef] [PubMed]
- McEwen, C.N.; McKay, R.G.; Larsen, B.S. Analysis of solids, liquids, and biological tissues using solids probe introduction at atmospheric pressure on commercial LC/MS instruments. Anal. Chem. 2005, 77, 7826–7831. [Google Scholar] [CrossRef] [PubMed]
- Neff, W.E.; Frankel, E.N. Photosensitized oxidation of methyl linolenate monohydroperoxides: Hydroperoxy cyclic peroxides, dihydroperoxides and hydroperoxy bis-cyclic peroxides. Lipids 1984, 19, 952–957. [Google Scholar] [CrossRef]
- Schmidt, T.J.; Nour, A.M.M.; Khalid, S.A.; Kaiser, M.; Brun, R. Quantitative structure-antiprotozoal activity relationships of sesquiterpene lactones. Molecules 2009, 14, 2062–2076. [Google Scholar] [CrossRef] [PubMed]
- Molecular Operating Environment (MOE); 2013.08; Chemical Computing Group Inc.: Montreal, QC, Canada, 2016.
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Montgomery, J.A., Jr.; Vreven, T.; Kudin, K.N.; Burant, J.C.; et al. Gaussian 03, Revision B.05; Gaussian, Inc.: Wallingford, CT, USA, 2004. [Google Scholar]
- Sample Availability: Samples of the compounds 1–3 are available from the authors.
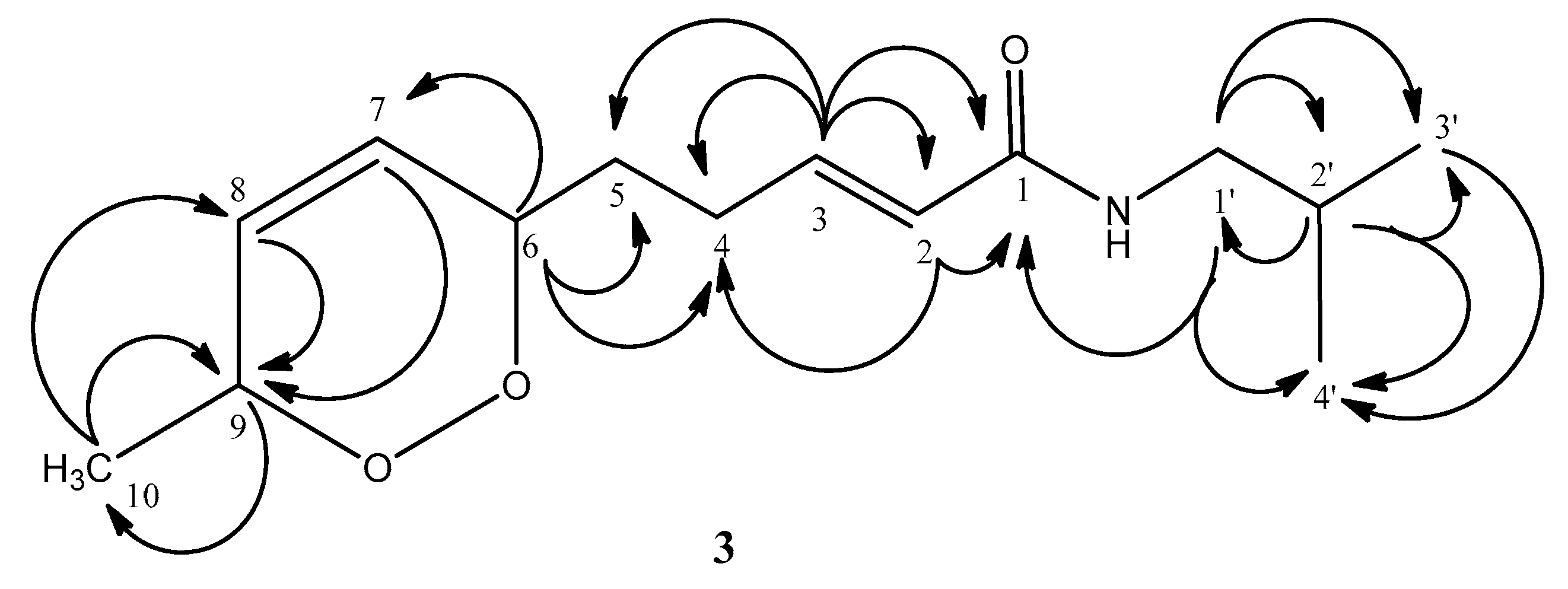
| Position | 3a | 3b |
|---|---|---|
| δH, Mult. (J in Hz) | δH, Mult. (J in Hz) | |
| 1 | ||
| 2 | 5.82 ddd (15.2, 1.4 and 1.4) | 5.82 ddd (15.2, 1.4 and 1.4) |
| 3 | 6.81 ddd (15.2, 7.4 and 6.6) | 6.81 ddd (15.2, 7.4 and 6.6) |
| 4 | 2.32 ddddd (14.9, 8.8, 7.4, 7.2 and 1.4) | 2.32 ddddd (14.9, 8.8, 7.4, 7.2 and 1.4) |
| 2.36 ddddd (14.9, 9.1, 6.6, 5.3 and 1.4) | 2.36 ddddd (14.9, 9.1, 6.6, 5.3 and 1.4) | |
| 5 | 1.69 dddd (14.2, 9.1, 7.2 and 4.0) | 1.67 dddd (14.2, 9.1, 7.2 and 4.0) |
| 1.87 dddd (14.2, 8.9, 8.8 and 5.3) | 1.87 dddd (14.2, 8.9, 8.8 and 5.3) | |
| 6 | 4.38 ddddd (8.9, 4.0, 2.4, 1.3 and 1.2) | 4.59 ddddd (8.9, 4.0, 2.4, 1.3 and 1.2) |
| 7 | 5.83 ddd (10.3, 2.4 and 1.5) | 5.78 ddd (10.3, 2.4 and 1.5) |
| 8 | 5.86 ddd (10.3, 1.7 and 1.2) | 5.83 ddd (10.3, 1.7 and 1.2) |
| 9 | 4.66 qddd (6.7, 1.7, 1.5 and 1.3) | 4.70 qddd (6.7, 1.7, 1.5 and 1.3) |
| 10 | 1.23 d (6.7) | 1.20 d (6.7) |
| 1’ | 3.14 dd (6.8 and 6.2) | 3.14 dd (6.8 and 6.2) |
| 2’ | 1.80 th (6.8 and 6.7) | 1.80 th (6.8 and 6.7) |
| 3’ | 0.92 d (6.7) | 0.92 d (6.7) |
| 4’ | 0.92 d (6.7) | 0.92 d (6.7) |
| Position | 3a | 3b | ||
|---|---|---|---|---|
| δC | HMBC | δC | HMBC | |
| 1 | 166.0 | 165.9 | ||
| 2 | 124.4 | C-1, C-4 | 124.4 | C-1, C-4 |
| 3 | 143.3 | C-1, C-2, C-4, C-5 | 143.2 | C-1, C-2, C-4, C-5 |
| 4 | 27.9 | C-2, C-3, C-5, C-6 | 27.5 | C-2, C-3, C-5, C-6 |
| C-2, C-3, C-5, C-6 | C-2, C-3, C-5, C-6 | |||
| 5 | 31.5 | C-3, C-4, C-6, C-7 | 31.0 | C-3, C-4, C-6, C-7 |
| C-3, C-4, C-6, C-7 | C-3, C-4, C-6, C-7 | |||
| 6 | 77.2 | C-4, C-5, C-7, C-8 | 76.8 | C-4, C-5, C-7, C-8 |
| 7 | 127.0 | C-6, C-9 | 127.2 | C-6, C-9 |
| 8 | 129.3 | C-6, C-9 | 129.5 | C-6, C-9 |
| 9 | 74.3 | C-7, C-8, C-10 | 74.2 | C-7, C-8, C-10 |
| 10 | 18.1 | C-8, C-9 | 17.7 | C-8, C-9 |
| 1’ | 46.8 | C-1, C-2’, C-3’, C-4’ | 46.7 | C-1, C-2’, C-3’, C-4’ |
| 2’ | 28.5 | C-1’, C-3’, C-4’ | 28.5 | C-1’, C-3’, C-4’ |
| 3’ | 20.1 | C-1’, C-2’, C-4’ | 20.1 | C-1’, C-2’, C-4’ |
| 4’ | 20.1 | C-1’, C-2’, C-3’ | 20.1 | C-1’, C-2’, C-3’ |
| Position H- | Calc. Δ | Δδ Calc | Exp. Δ | Δδ Exp | ||
|---|---|---|---|---|---|---|
| 6S,9R-cis | 6R,9R-trans | Major Isomer | Minor Isomer | |||
| 5a | 1.426 | 1.486 | 0.059 | 1.873 | 1.873 | 0.000 |
| 5b | 2.149 | 1.510 | −0.638 | 1.693 | 1.669 | −0.024 |
| 6 | 4.030 | 4.622 | 0.591 | 4.377 | 4.586 | 0.209 |
| 7 | 6.098 | 5.859 | −0.240 | 5.830 | 5.782 | −0.048 |
| 8 | 6.032 | 5.950 | −0.082 | 5.860 | 5.830 | −0.030 |
| 9 | 4.807 | 4.849 | 0.042 | 4.663 | 4.696 | 0.033 |
| 10 | 0.982 | 0.998 | 0.016 | 1.232 | 1.203 | −0.029 |
| Compound | Tbr | Pf (NF54) | Pf (K1) | L6 |
|---|---|---|---|---|
| 1 | 2.88 ± 0.06 (13.0) | 0.99 ± 0.12 (4.5) | n.t. | 39.9 ± 1.9 (180.3) |
| 2 | 17.1 ± 2.3 (68.1) | 22.1 ± 4.0 (88.0) | n.t. | 60.1 ± 9.4 (239.2) |
| 3 | 5.60 ± 0.67 (13.0) | 1.29 ± 0.20 (5.1) | 0.54 ± 0.14 (2.1) | 42.6 ± 1.7 (168.2) |
| Pos. contr. | 0.004 ± 0.001 a | 0.002 ± 0.001 b | 0.09 ± 0.006 b | 0.004 ± 0.001 c |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silveira, N.; Saar, J.; Santos, A.D.C.; Barison, A.; Sandjo, L.P.; Kaiser, M.; Schmidt, T.J.; Biavatti, M.W. A New Alkamide with an Endoperoxide Structure from Acmella ciliata (Asteraceae) and Its in Vitro Antiplasmodial Activity. Molecules 2016, 21, 765. https://doi.org/10.3390/molecules21060765
Silveira N, Saar J, Santos ADC, Barison A, Sandjo LP, Kaiser M, Schmidt TJ, Biavatti MW. A New Alkamide with an Endoperoxide Structure from Acmella ciliata (Asteraceae) and Its in Vitro Antiplasmodial Activity. Molecules. 2016; 21(6):765. https://doi.org/10.3390/molecules21060765
Chicago/Turabian StyleSilveira, Narjara, Julia Saar, Alan Diego C. Santos, Andersson Barison, Louis P. Sandjo, Marcel Kaiser, Thomas J. Schmidt, and Maique W. Biavatti. 2016. "A New Alkamide with an Endoperoxide Structure from Acmella ciliata (Asteraceae) and Its in Vitro Antiplasmodial Activity" Molecules 21, no. 6: 765. https://doi.org/10.3390/molecules21060765
APA StyleSilveira, N., Saar, J., Santos, A. D. C., Barison, A., Sandjo, L. P., Kaiser, M., Schmidt, T. J., & Biavatti, M. W. (2016). A New Alkamide with an Endoperoxide Structure from Acmella ciliata (Asteraceae) and Its in Vitro Antiplasmodial Activity. Molecules, 21(6), 765. https://doi.org/10.3390/molecules21060765