Abstract
Zanthoxylum rhetsa is an aromatic tree, known vernacularly as “Indian Prickly Ash”. It has been predominantly used by Indian tribes for the treatment of many infirmities like diabetes, inflammation, rheumatism, toothache and diarrhea. In this study, we identified major volatile constituents present in different solvent fractions of Z. rhetsa bark using GC-MS analysis and isolated two tetrahydrofuran lignans (yangambin and kobusin), a berberine alkaloid (columbamine) and a triterpenoid (lupeol) from the bioactive chloroform fraction. The solvent fractions and purified compounds were tested for their cytotoxic potential against human dermal fibroblasts (HDF) and mouse melanoma (B16-F10) cells, using the MTT assay. All the solvent fractions and purified compounds were found to be non-cytotoxic to HDF cells. However, the chloroform fraction and kobusin exhibited cytotoxic effect against B16-F10 melanoma cells. The presence of bioactive lignans and alkaloids were suggested to be responsible for the cytotoxic property of Z. rhetsa bark against B16-F10 cells.
Keywords:
Zanthoxylum rhetsa; lignans; triterpenes; alkaloids; cytotoxicity; NMR spectroscopy; GC-MS 1. Introduction
Skin is the largest organ of the body, protecting it from many external stresses such as radiation, temperature, chemicals and microbes. It comprises the epidermis (outer layer), dermis (inner layer) and subcutaneous tissues. The dermis includes sweat glands, hair follicle, blood vessels and nerves, while the epidermis is made up of three cells namely squamous, basal and melanocytes [1]. Under normal conditions, these cells undergo systematic cell division and produce daughter cells. However, in case of abnormal cell division, the epidermal cells grow aberrantly and can cause three types of skin carcinomas based on their originating cells i.e., squamous, basal and melanoma skin cancers. The squamous and basal skin cancers, which are non-melanoma cancers, are common and not life threatening. These types of non-melanoma skin cancers can be cured if treated at an early stage. Clinical data show that skin cancer occurs primarily based on geographical areas. Basal cell carcinoma (BCC) is usually reported among Hispanics, Caucasians, Japanese and Chinese Asian, whereas squamous cell carcinoma (SCC) is more common among African, Americans and Asian Indians [2]. In comparison, melanoma cancer is less common but life threatening. Malignant melanoma is the nineteenth most commonly reported cancer in the world, and the seventh most common in the United States (US) [3]. The rate of incidence of melanoma cancer has risen exponentially as shown by the 2015 statistics that reported 73,870 cases (42,670 men and 31,200 women) as being diagnosed with the disease in the US alone [4]. Meanwhile, a total of 12,960 (7640 men and 5320 women) new cases were reported in Australia, for the year 2015. The number is expected to increase to 17,570 by 2020 [5]. The factors influencing this type of carcinoma are fair skin, a history of sunburn, continuous exposure to UV or other radiation, moles, heredity or weak immune system, etc. Current methods of treatment for melanoma cancers include cryotherapy, external surgery, radiation therapy, chemotherapy, photodynamic therapy, biological therapy and targeted drug therapy such as with vemurafenib, dabrafenib, and trametinib [2]. All of these methods of treatment have certain limitations such as high costs, side effects and reoccurrence. Thus, safer alternative remedies for skin cancer need to be found. In this context, traditional medicine and medicinal plants, in general, offer an excellent resource for the identification of new therapeutic agents for use against diseases, including skin cancer.
Zanthoxylum rhetsa (Roxb.) DC (syn. Zanthoxylum budrunga, Fam. Rutaceae) is a medium-sized aromatic tree with conical prickles on the bark of the trunk and branches. It is widely distributed in the tropical and sub-tropical regions, including India, Bangladesh, Indonesia, China and Malaysia [6]. The plant has long been valued for its medicinal uses. The Kannikar tribes from Tamil Nadu utilized a paste made from the prickly thorns of Z. rhetsa to treat breast pain and to increase lactation in breastfeeding mothers. The plant shoots are consumed as a vegetable by the Adi tribes of Arunachal Pradesh, India [7]. Meanwhile, various parts of Z. rhetsa are traditionally used as an aromatic, astringent, antimicrobial, antiseptic and antidiabetic agent, as well as used to treat snake bites, inflammatory dermatosis, cholera, rheumatism, and toothache [8,9]. Characteristic secondary metabolites of Zanthoxylum species include lignoids, alkaloids, amides, flavonoids, terpenes, sterols and coumarins [10]. Alkaloids are abundant in the trunk and root bark, and are typically of the isoquinoline and quinolone types. Lignoids are also abundant in the genus, typically of the diarylbutirolactones and furofuranic types. Previous phytochemical investigations on Z. rhetsa have shown the presence of a variety of compounds including monolignols, coumarins, alkaloids and lignans namely 3,5-dimethoxy-4-geranyloxycinnamyl alcohol, xanthyletin, 8-methoxy-N-methylflindersine and sesamin [11] as well as zanthorhetsamide [12]. Ahsan et al. [13] further reported quinolone terpene alkaloids namely, chelerybulgarine, 2′-episimulanoquinoline, 2,11-didemethoxyvepridimerine B, and rhetsidimerine, from the root bark of the plant. Moreover, GC-MS analysis of the ethanol extract of Z. rhetsa spines revealed fifteen compounds, of which 1,2-benzenedicarboxylic acid and diisooctyl ester were the major components, followed by oleic acid and n-hexadecanoic acid [14]. Likewise, the volatile constituents of the fruits, seed coat and leaf were also identified through GC-MS, wherein, sabinene, carophyllene oxide, spathulenol, α-pinene, 4-terpineol, 3-elemene, β-phellandrene, 3-pinene, γ-terpinene and myrcene were the predominant compounds [15,16,17]. The stem bark of Z. rhetsa has been shown to possess anti-inflammatory activity, which was mediated by down-regulation of TNF-α, mRNA expression of pro-inflammatory cytokines and also by inhibition of iNOS and COX-2 production [18]. The seeds of the plant have been reported to have sunscreening properties [19]. Previously, we disclosed the Ultraviolet A/Ultraviolet B (UVA/UVB) protecting properties of the bark extracts of Z. rhetsa [20]. Although previous studies have revealed the anti-inflammatory and other therapeutic activities of the plant extract, to the best of our knowledge, the potential of Z. rhetsa bark extract to protect against skin cancer has not been investigated. In this study we publicize the results of our investigation on the cytotoxic properties of Z. rhetsa bark extract, against Human Dermal Fibroblasts (HDF) and B16-F10 melanoma cells. We also report the isolation and identification of four bioactive constituents from the chloroform fraction of the Z. rhetsa bark extract.
2. Results and Discussion
2.1. Identification of Bioactive Compounds Through GC-MS Analysis
The GC-MS spectra for the crude methanol extract and the solvent fractions of Z. rhetsa bark are shown in Figure S1. The compounds present in the active fractions were identified by matching their recorded mass spectra with those retrieved from the NIST11 and WILEY229 mass spectral libraries, and by comparison with literature values [21,22,23,24,25]. Overall, a total of thirty-nine compounds were identified from the bark of Z. rhetsa. The identified compounds and their mass data are tabulated in Table 1.

Table 1.
Compounds identified in various solvent fractions of Z. rhetsa using GC-MS analysis.
Identification of the tetrahydrofuran lignans was based on their characteristic MS fragmentations via m/z 219 [(CH3O)2-C6H3-(CH2)2-(CH)2-O-CH2]+, 177 [(CH3O)2-C6H3-CH-CH=CH2]+, 165 [(CH3O)2-C6H3-CO]+ [26,27]. Previous studies also revealed the presence of alkaloids and lignans in this species [11,13]. To the best of our knowledge, this is the first report on the presence of compounds such as kobusin, yangambin, epi-eudesmin, eudesmin, 8-hydroxy-4′-methoxypinoresinol, hesperetin, magnolin reticuline, allocryptopine, usambanoline, dihydronitidine, N-methyllaurotetanine and chelerythrine in the bark of Z. rhetsa.
2.2. Evaluation of Cytotoxic Activity
MTT assay was performed to evaluate the toxic effects of the crude methanolic extract, solvent fractions and the isolated compounds, against HDF and B16-F10 melanoma cells. The extract and solvent fractions showed minimal effect on HDF cell and exhibited no toxicity against normal skin cells (Figure S2).
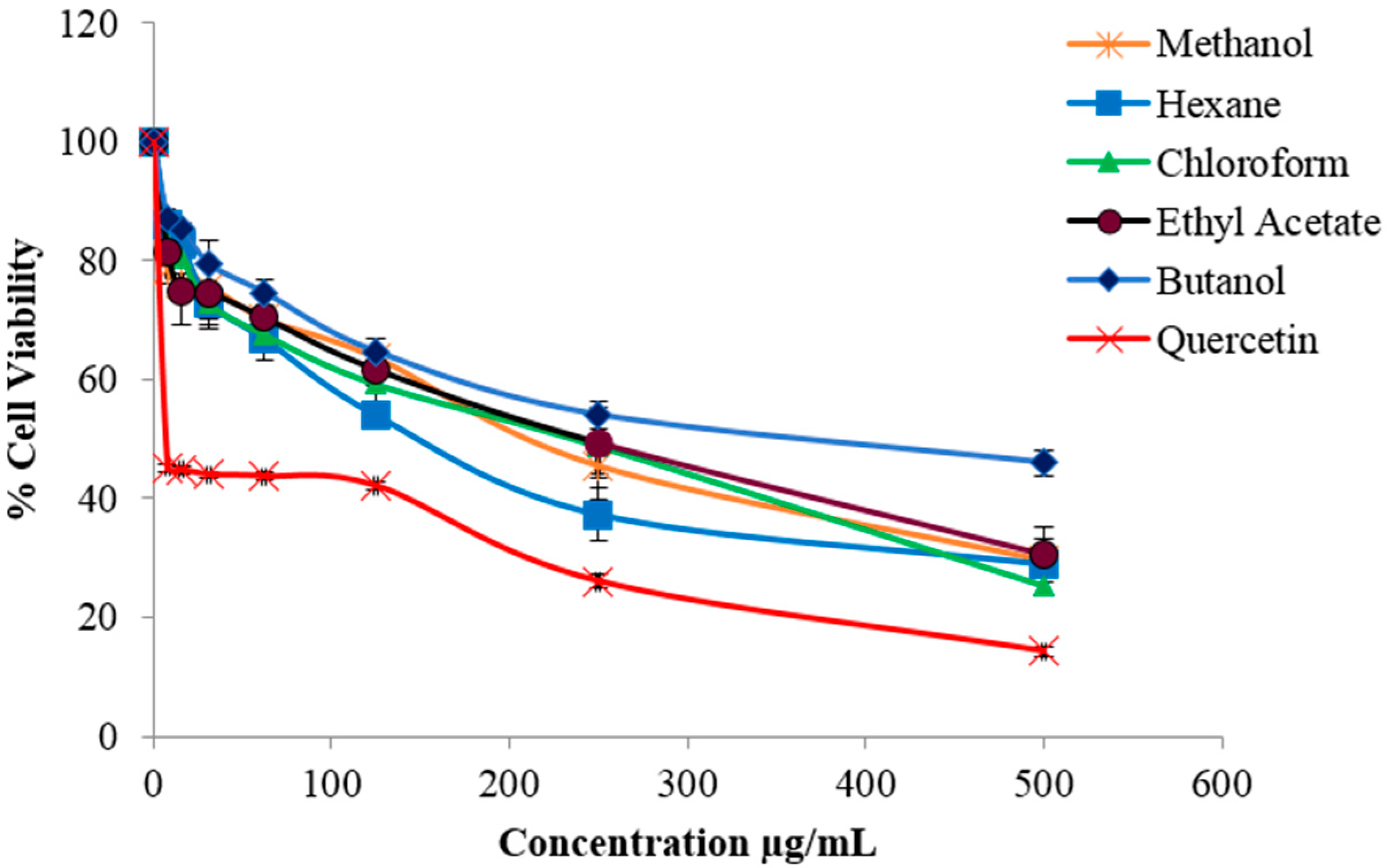
Other Zanthoxylum species have also been reported to be non-toxic to normal cell lines [28,29]. Meanwhile, the crude methanolic extract and solvent fractions were found to be toxic towards B16-F10 cells, causing significant cell death (Figure 1).The chloroform fraction exhibited the strongest cytotoxic effect followed by hexane, ethyl acetate, methanol and butanol fractions with IC50 values 156, 132.7, 174.3, 168 and 263.1 µg/mL, respectively.
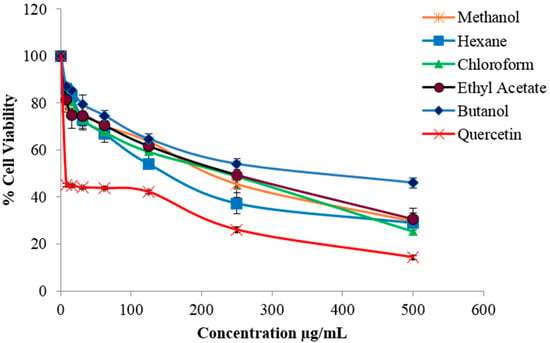
Figure 1.
Cytotoxic effect of various fractions of Z. rhetsa, at different concentrations (0–500 µg/mL), against B16-F10 melanoma cells. Data are expressed as mean ± SD of three independent experiments. Quercetin was used as the positive control.
GC-MS analysis showed that the crude methanolic extract and all the solvent fractions of Z. rhetsa bark contained a diverse array of constituents, consisting of monolignols, coumarins, alkaloids and tetrahydrofuran lignans. The GC-MS result for the hexane fraction (Table 1) showed lupeol (36.9%) to be the major constituent along with lower amounts of other constituents comprising simple ketones, sesquiterpenes, fatty acids, sterols, triterpenes and tetrahydrofuran lignans. The GC-MS result for the chloroform fraction (Table 1) showed the tetrahydrofuran lignans, yangambin (30.4%) and eudesmin (26.7%), as major constituents. Other tetrahydrofuran lignans were also observed as minor constituents together with simple ketones, triterpenes and isoquinoline alkaloids. The ethyl acetate fraction contained coniferyl alcohol (21.5%) as the major constituent along with other monolignols, coumarins, isoquinoline alkaloids and lignans. In the butanol fraction, quinolone alkaloids were predominant along with ketones, monolignols and several unknown compounds. The difference in the chemical constituents between the solvent fractions could be responsible for the difference in their cytotoxic properties as revealed by the MTT assay. Ahsan and co-workers reported that dimeric quinolone-terpene alkaloids isolated from Z. rhetsa root bark showed weak cytotoxic effect against six stomach cancer cell lines [13]. Meanwhile, according to Mukhija and his co-workers , tetrahydrofuran lignans isolated from the petroleum ether extract of Z. alatum bark were cytotoxic against lung and pancreatic carcinoma cell lines [24]. These studies suggested that lignan-rich fractions exert more cytotoxic effect in comparison to fractions rich in quinolone-terpene alkaloids. In another study, a structure-activity relationship analysis on the bioactive tetrahydrofuran lignans of Z. planispinum root, revealed that the phenolic groups in the lignans were responsible for the increase in cytotoxicity against human tumour cell lines [30]. Thus, based on these previous findings, it is highly probable that the cytotoxic property of the chloroform and hexane fractions of Z. rhetsa were due to the presence of the tetrahydrofuran lignans.
2.3. Structural Identification of the Isolated Compounds
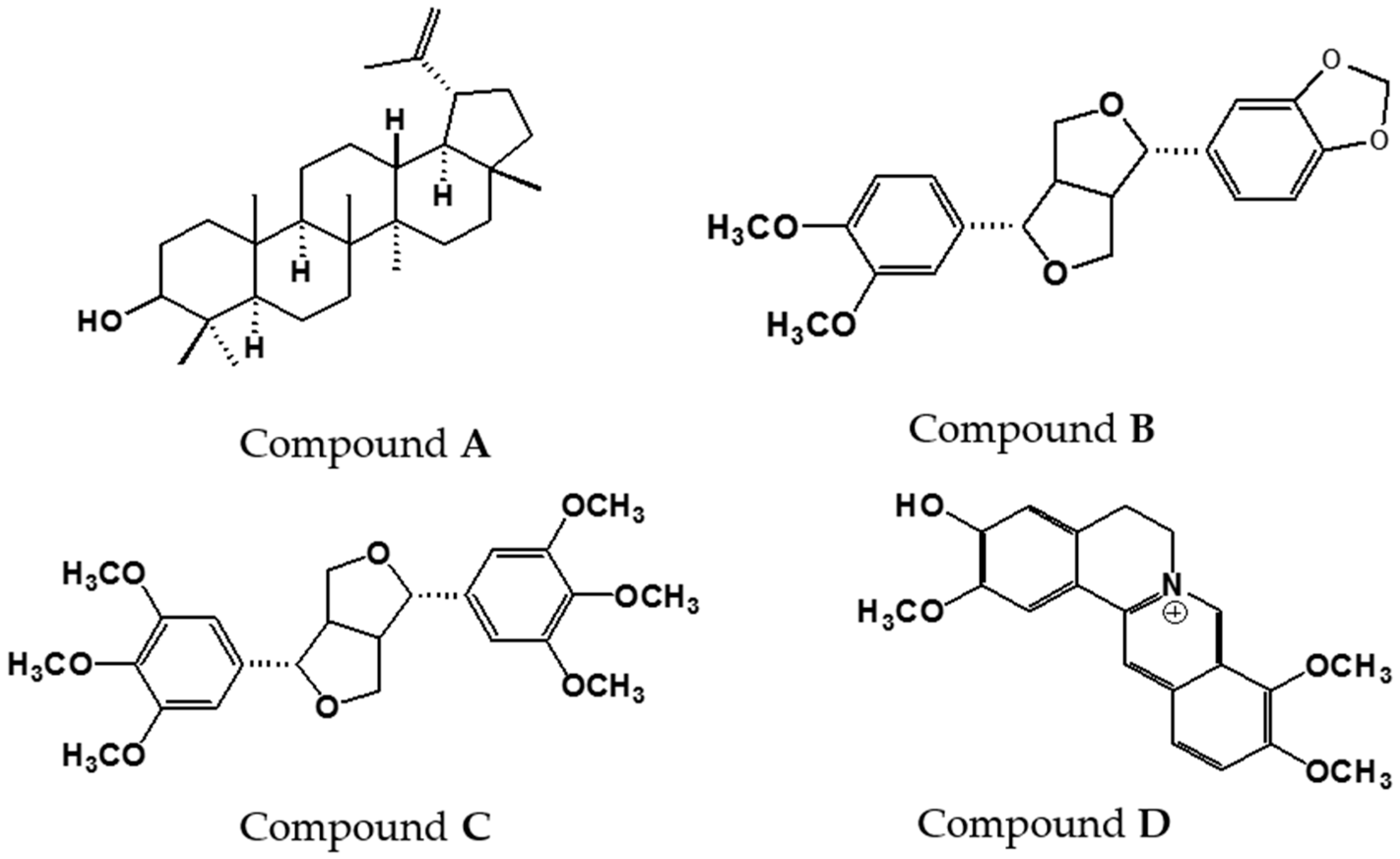
Compounds A–D, isolated from the bioactive chloroform fraction are shown in Figure 2. The structures of the compounds were elucidated using 1D and 2D NMR and mass spectral data as well as comparison with literature values. The physical and spectroscopic data of the compounds are listed as follows:
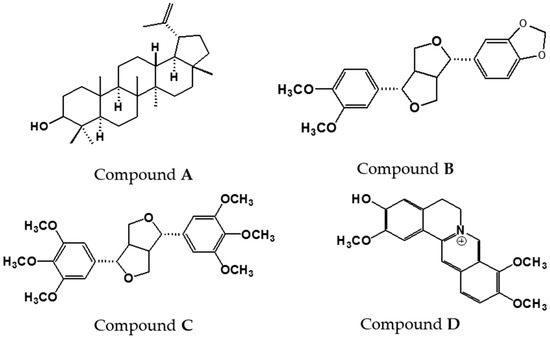
Figure 2.
Structures of compounds isolated from the chloroform fraction of Z. rhetsa bark.
Compound A (White powder): Melting point: 215–216 °C, EI-MS: m/z 426 [M]+ (calc. for C30H50O, 426.71). 1H-NMR (500 MHz, CDCl3) δ: 0.76, 0.79, 0.83, 0.95, 0.97, 1.03, 1.68 (3H, s, 7 × CH3), 3.18 (1H, dd, J = 5.5, 10.7 Hz, H-3), 4.57 (1H, s, H-29a), 4.69 (1H, s, H-29b). 13C-NMR (125 MHz, CDCl3) δ: 151.00 (C-20), 109.3 (C-29), 79.0 (C-3), 55.3 (C-5), 50.4 (C-9), 48.3 (C-18), 48.0 (C-19), 43.0 (C-17), 42.8 (C-14), 40.8 (C-8), 40.0 (C-22), 38.9 (C-13), 38.7 (C-4), 38.0 (C-1), 37.2 (C-10), 35.6 (C-16), 34.3 (C-7), 29.8 (C-21), 28.0 (C-23), 27.4 (C-12), 25.1 (C-2), 20.9 (C-11), 19.3 (C-30), 18.3 (C-6), 18.0 (C-28), 16.1 (C-25), 16.0 (C-24), 15.4 (C-27), 14.5 (C-26). Compound A was identified as lupeol [31].
Compound B (Colourless gum): EI-MS: m/z 370 [M]+ (calc. for C21H22O6, 370.39). 1H-NMR (500 MHz, CDCl3) δ: 6.91–6.77 (6H, m, aromatic rings), 5.95 (2H, s, OCH2O-benzo-1,3-dioxole moieties), 4.73 (2H, t, J = 10.5 Hz, H-2/6, tetrahydrofuran moieties), 4.26 (2H, d, J = 9.0 Hz, H-4b/8b, equatorial protons-tetrahydrofuran moieties, 3.89 (m, 2H, H-4a/8a, axial protons-tetrahydrofuran moieties), 3.80 (s, 3H, OCH3), 3.87 (s, 3H, OCH3), 3.08 (m, 2H, H-1/5 tetrahydrofuran moieties) 13C-NMR (125 MHz, CDCl3) δ: 149.2 (C-3′), 148.6 (C-4′), 147.8 (C-3′′), 147.1 (C-4′′), 135 (C-1′′), 133.2 (C-1′), 119.3 (C-6′′), 118.2 (C-6′), 111.1 (C-5′), 109.2 (C-2′), 108 (C-5′′), 106.4 (C-2′′), 100.9 (-OCH2O-), 85.8 (C-2), 85.7 (C-6), 71.7 (C-4), 71.6 (C-8), 56 (-OCH3), 55.9 (-OCH3), 54.3 (C-5), 54.2 (C-1). Compound B was identified as kobusin [32].
Compound C (White needles): Melting point: 119–121 °C, EI-MS: m/z 446 [M]+ (calc. for C24H30O8, 446.49). 1H-NMR (500 MHz, CDCl3) δ: 6.57 (4H, s, H-2′/6′, 2′′/6′′, aromatic rings), 4.75 (2H, d, J = 3.7 Hz, H -2/6, tetrahydrofuran moieties), 4.31 (2H, dd, J1 = 6.4 Hz, J2 = 8.6 Hz, H-4b/8b, equatorial protons-tetrahydrofuran moieties), 3.93 (2H, dd, J1 = 2.7 Hz, J2 = 9.3 Hz, H-4a/8a, axial protons-tetrahydrofuran moieties), 3.88 (12H, s, OCH3), 3.84 (6H, s, OCH3), 3.11 (2H, m, H-1/5) 13C-NMR (125 MHz, CDCl3) δ: 153.4 (C-3/3′, 5/5′), 137.5 (C-4/4′), 136.7 (C-1/1′), 102.9 (C-2/2′, 6/6′), 85.9 (C-7/7′), 72.1 (C-9/9′), 60.8 (C-4/4′, OCH3), 56.2 (C-3/3′, 5/5′, OCH3), 54.4 (C-8/8′). Compound C was identified as yangambin [33].
Compound D (Yellow powder): Melting point: 280–282 °C, EI-MS: m/z 338.4 [M]+ (calc. for C20H20NO4+, 338.37). 1H-NMR (500 MHz, CDCl3) δ: 9.72 (1H, s, H-8), 8.75 (1H, s, H-13), 8.09 (1H, d, J = 8.8 Hz, H-12), 7.98 (1H, d, J = 8.8 Hz, H-11), 7.65 (1H, s, H-1), 6.86 (1H, s, H-4), 4.91(2H, t, J = 6.3 Hz, H-6), 4.20 (3H, s, C-10, OCH3), 4.10 (3H, s, C-9, OCH3), 4.02 (3H, s, C-3, OCH3), 3.20 (2H, t, J = 6.4 Hz, H-5) 13C-NMR (125 MHz, CDCl3) δ: 150.2 (C-3), 148.3 (C-2), 144.6 (C-8), 144.3 (C-10), 138.9 (C-14), 134 (C-12a), 128.9 (C-4a), 126.7 (C-11), 122.9 (C-12), 121.7 (C-8a), 119.4 (C-13), 117.8 (C-14a), 114.5 (C-1), 108.6 (C-4), 61.1 (9-OCH3), 56.3 (10-CH3), 56.0 (C-6), 55.5 (3-OCH3), 26.3 (C-5). Compound D was identified as columbamine [34].
Zanthoxylum species, especially the bark material, have been previously reported to be rich in tetrahydrofuran lignans. These included six lignans from Z. nitidium bark [21], twelve lignans from Z. schinifolium stem [22], seven lignans from Z. armatum bark [25], three lignans from Z. budrunga bark [35], and eight lignans from the roots of Z. planispinum [30]. Apart from lignans, various alkaloids have also been reported to be abundant in the bark of several Zanthoxylum species. For example, the alkaloids N-methyl corydine, magnoflorine and berberine have been reported to be major constituents in the stem and branches of Z. punctatum and Z. monophyllum [36]. Previous studies on Z. rhetsa have also reported the presence of different types of alkaloids. This included 6-acetonyldihydrochelerythrin and arnottianamide in the conical prickles, and the quinolone-terpene alkaloids chelerybulgarine, 2,11-didemethoxyvepridimerine B, rhetsidimerine, simulanoquinoline and 2′-episimulanoquinoline in the root bark [13,37]. In addition to these compounds, the current study further reports the occurrence of kobusin, yangambin and columbamine in the bark of Z. rhetsa.
2.4. Cytotoxicity Evaluation of Isolated Compounds
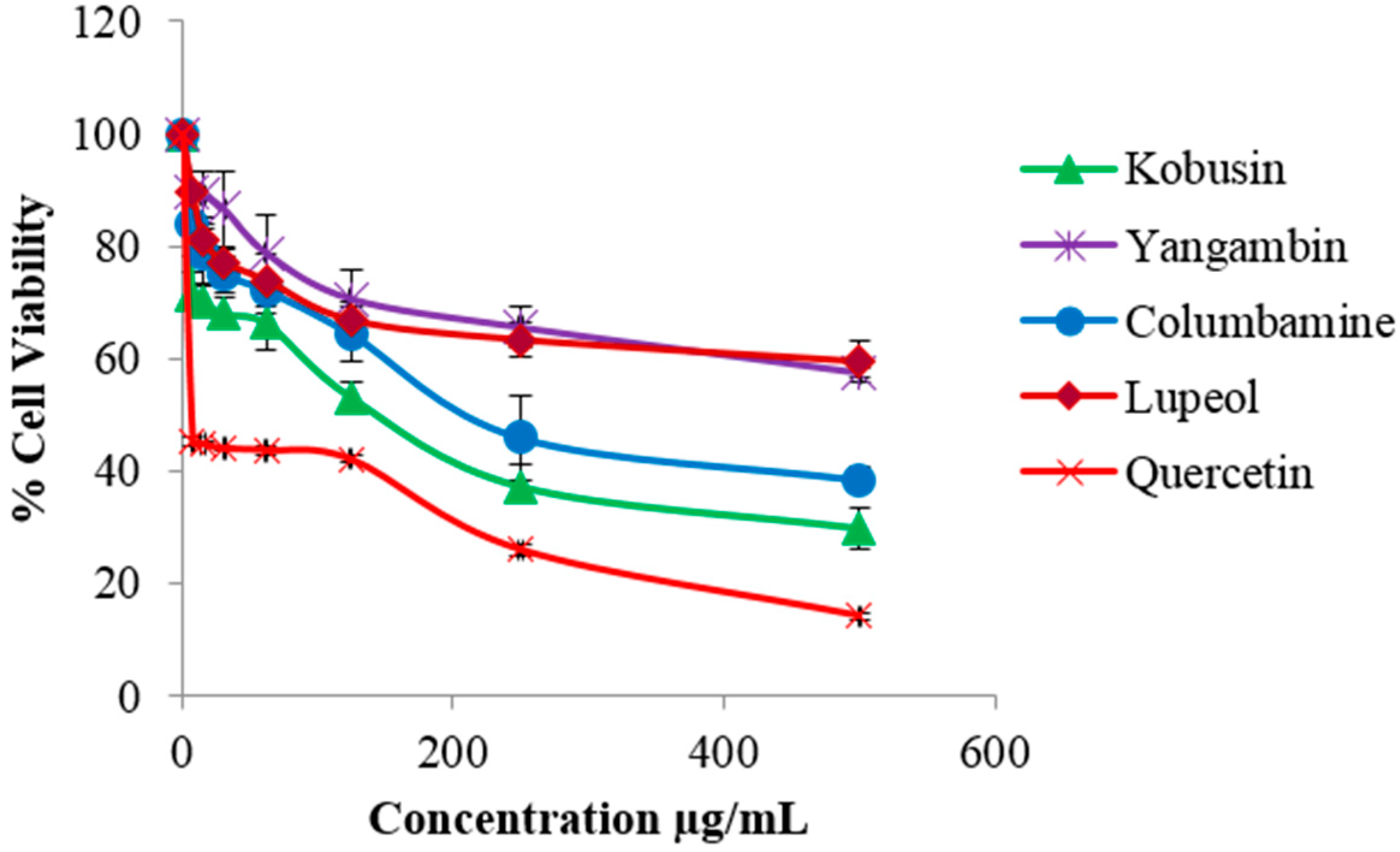
The isolated compounds were evaluated for cytotoxicity against HDF and B16-F10 melanoma cells. The isolated compounds were found to be non-toxic to HDF cells (Figure S3). However kobusin caused a high percentage of cell death against B16-F10 cells followed by columbamine, lupeol and yangambin, giving IC50 values of 112.2, 195.6, 377.8 and 442.4 µg/mL, respectively. Quercetin, the positive control, gave an IC50 value of 22.72 µg/mL. The results of the cytotoxicity test on the isolated compounds are shown in Figure 3. These results were in good agreement with previous reports where kobusin, isolated from Z. alatum bark, showed strong cytotoxic effect, with IC50 values of 34.71 µg/mL and 32.86 µg/mL, against A549 (lung) and MIA-PaCa (pancreatic) cell lines, respectively [24]. On the other hand, columbamine, previously isolated from Rhizoma coptidis, has been found to be moderately cytotoxic with an IC50 value of 226.54 µg/mL against HepG2 cells [38]. Meanwhile, lupeol, isolated from Grewia tiliaefolia, was reported to be weakly cytotoxic with an IC50 value of 330 µg/mL against B16-F10 cells [39]. It was also found to mediate anticancer activity against malignant melanoma cells by altering the level of Bcl-2, Bax protein and Wnt/β-catenin signaling [40]. In the case of yangambin, previous report of its cytotoxic effect had only been reported on murine macrophages. The toxic effect was found to be low. Structure-activity relationship studies have associated the higher cytotoxic effect of kobusin and columbamine with the presence of phenylmethylenedioxy and phenolic groups, respectively [30,38].
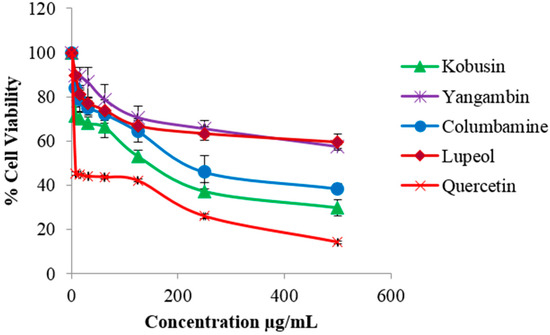
Figure 3.
Cytotoxic effect of isolated compounds from Z. rhetsa at various concentrations (0–500 µg/mL) against B16-F10 melanoma cells. Data are expressed as mean ± SD of three independent experiments. Quercetin was used as the positive control.
3. Materials and Methods
3.1. General Experimental Procedures
Melting points were recorded on a Koffler hot-stage apparatus (Electrothermal 9100, Dubuque, IA, USA) and were uncorrected. GC-MS analysis were carried out on a QP-2010 Ultra GCMS spectrometer (Shimadzu, Kyoto, Japan) equipped with a flame ionization detector (FID). 1D and 2D NMR spectra were recorded on a Unity INOVA 500 MHz spectrometer (Varian, Palo Alto, CA, USA) using standard pulse programs. Chloroform-d (CDCl3) and methanol-d4 (CD3OD) were used as NMR solvents, and TMS was utilized for internal referencing. Solvents used for extraction and isolation were of analytical grade and obtained from R & M chemicals (Edmonton, AB, Canada). Kieselgel 60 (0.040–0.063 mm) and Lichroprep RP-18 (40–63 µm) were purchased from Merck (Darmstadt, Germany) while Sephadex LH-20 was purchased from Sigma (St. Louis, MO, USA). Dulbecco’s Modified Eagle Medium (DMEM), fetal bovine serum (FBS), trypsin–EDTA, penicillin/streptomycin were purchased from GIBCO (Lifetechnologies, Grand Island, NY, USA). Trypan blue and MTT (3-(4,5-dimethylthiazole-2-yl)-2,5-biphenyl tetrazolium bromide) were purchased from Sigma. A SpectraMax Plus (Molecular Devices, Sunnyvale, CA, USA) microplate reader was used in the bioassays.
3.2. Sample Collection
The trunk barks of Z. rhetsa were collected from Pangkor Island, Malaysia. A voucher specimen (SK2226/13) was deposited at the Herbarium of the Institute of Bioscience, Universiti Putra Malaysia.
3.3. Extraction
The air dried, powdered bark material (910 g) of Z. rhetsa was subjected to ultrasound-assisted extraction with 100% methanol and dried under vacuum at 40 °C to yield the crude methanolic extract (65 g). The extract was then subjected to solvent-solvent extraction using organic solvents of increasing polarities. The resulting solvent fractions were dried under vacuum, lyophilized to yield 14 g hexane, 17.4 g chloroform, 2 g ethyl acetate and 5.3 g butanol fractions. All the samples were stored at −20 °C prior to further analysis [20].
3.4. Cell Culture
Both HDF and B16-F10 cells were provided by the Laboratory of Vaccines and Immunotherapeutics (LIVES), Institute of Bioscience, UPM. Human dermal fibroblasts (HDF) cells (passage 11–15) were maintained in DMEM medium containing 5% fetal bovine serum, 1% penicillin (100 U/mL) and streptomycin (100 μg/mL). Mouse melanoma (B16-F10B) cells (passage 11–15) were also maintained in DMEM medium with 10% fetal bovine serum, 1% penicillin (100 U/mL) and streptomycin (100 μg/mL). Cells were sub-cultured, incubated and maintained in a humidified 5% CO2 incubator at 37 °C. Only the cells that attained more than 80% confluence (growth phase) were used for cell seeding [41].
3.5. MTT Proliferation Assay
Cells were seeded in 96 well plates (1 × 104 cells/well) and incubated in 5% CO2 incubator at 37 °C for 24 h. The cells were treated with serial dilutions of Z. rhetsa extract and positive control ranging from 500, 250, 125, 62.5, 31.25, 15.625, 7.81 µg/mL. The cells treated with medium without the sample served as negative control. Quercetin which is known to inhibit cell growth, induce apoptosis and is cytotoxic against B-16 melanoma cells, served as positive control [42,43]. All the samples and positive control were dissolved in DMEM medium with a small amount of DMSO (the final concentration of DMSO was lower than 0.1%). Negative control was also treated with the same solvent vehicle without the sample. After 24 h, cell viability was analyzed using MTT assay wherein 20 µL of MTT (5 mg/mL PBS) was added to each well and incubated at 37 °C for 3–4 h. MTT enters the mitochondria of the viable cells and gets reduced to insoluble formazan (dark purple) product. The formazan product was pelleted by centrifuging the plates at 1200 rpm for 5 min. Afterwards, the cells were treated with DMSO to dissolve the formazan product and subsequently its absorbance was measured spectrophotometrically after 30 min at 570 nm, using microplate reader (SpectraMax Plus). All experiments were performed in triplicates with three different passage cells:
Cell Viability (%) = (Treated cells/Untreated cells) × 100
3.6. GC-MS Analysis of Solvent Fractions
The crude methanolic extract and the solvent fractions were subjected to GC-MS analysis using a BPx5 column (30 m × 0.25 mm × 0.25 µm). The oven temperature program was set to 50 °C for 0 min, heated to 330 °C at the rate of 3 °C/min and held constant at 330 °C for a further 5 min. Helium was used as carrier gas with the following conditions: total flow—11.8 mL/min, column flow—0.8 mL/min, linear velocity—32.4 cm/s, purge flow—3.0 mL/min, split ratio—10. Mass spectra were recorded with ion source temperature of 200 °C and interface temperature of 250 °C. The mass scan parameters included a start time of 2.5 min and end time of 95.0 min. The acquisition (ACQ) parameters were as follows: Scan event time—0.10 s, scan speed—10000, mass range—40 m/z to 700 m/z.
3.7. Isolation of Chemical Constituents from the Bioactive Chloroform Fraction
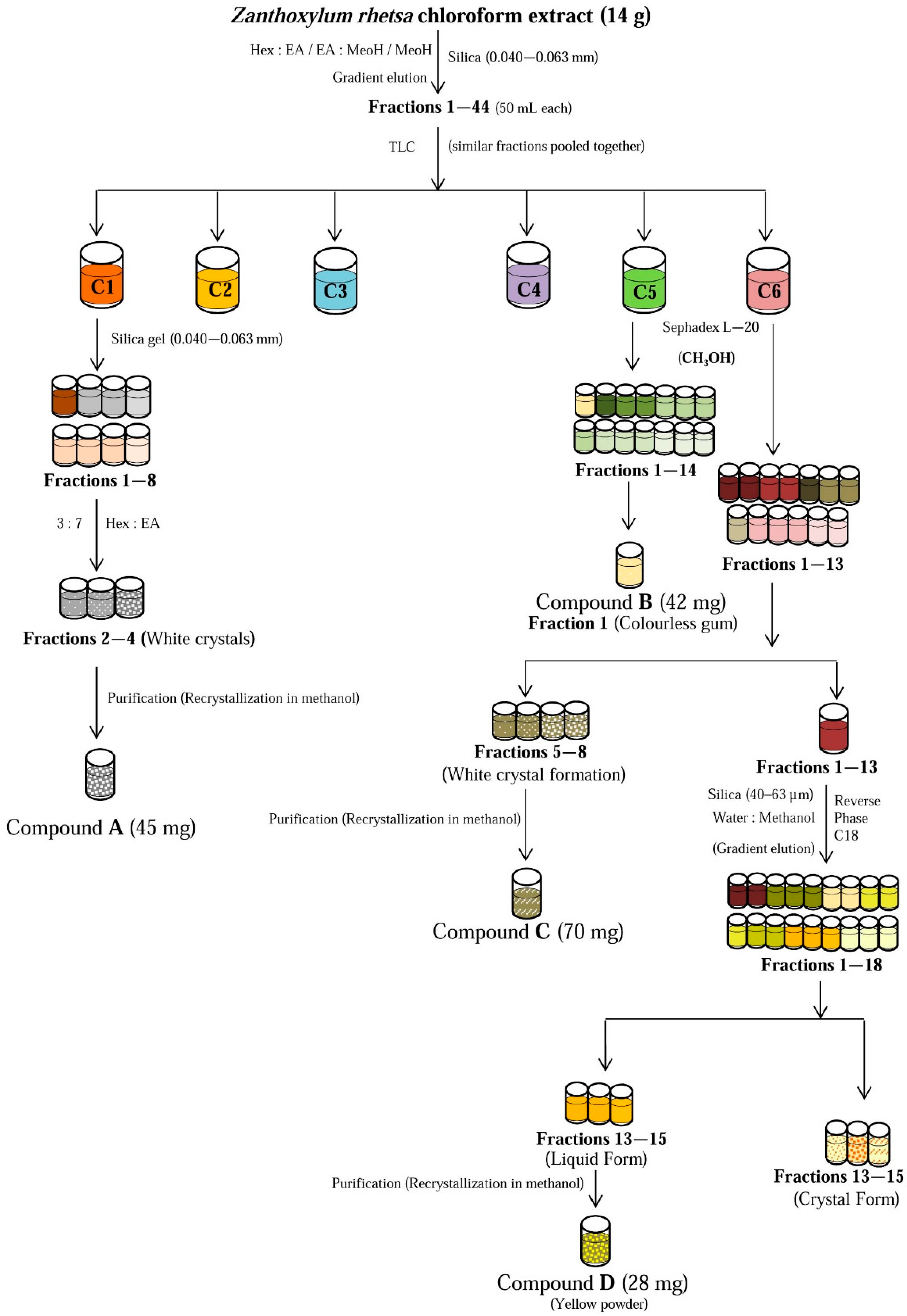
The scheme for the isolation of chemical constituents from the chloroform fraction is illustrated in Figure 4.
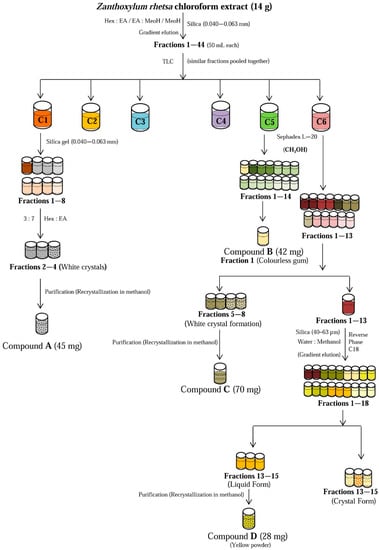
Figure 4.
Isolation of compounds from the chloroform fraction of Z. rhetsa extract using column chromatography.
The dried chloroform extract (14 g) was subjected to column chromatography (CC) over silica gel (0.040–0.063 mm) with solvent systems hexane:ethyl acetate (9:1, 8:2 7:3, 6:4, 5:5, 0:10) followed by ethyl acetate:methanol (9:1) and 100% methanol, to give 44 sub-fractions (~50 mL each). Sub-fractions with similar TLC profiles were pooled together and relabeled as C1, C2, C3, C4, C5, C6. Compound A (lupeol) was isolated as white powder (45 mg) from sub-fraction C1 using normal phase CC, where hexane and ethyl acetate were used as eluent at the ratio of 3:7. Compound B (42 mg, kobusin) was isolated from sub-fraction C5 by first passing it through a Sephadex LH-20 column, eluted with methanol. Sub-faction C6 was similarly pretreated with Sephadex LH-20 to give thirteen sub-fractions. White needle like crystals were filtered from sub-fractions 5–8, which yielded 70 mg of Compound C (yangambin) after further purification by recrystallization in methanol. The remaining sub-fraction C6 was subjected to CC over reverse phase silica (40–63 µm), eluted with solvent system water:methanol at varying ratios (9:1, 7:3, 5:5, 4:6, 3:7, 0:10), to give 18 sub-fractions. Sub-fractions 13–15 were pooled and further purified via repeated CC and recrystallization steps to yield 28 mg of Compound D (columbamine).
3.8. Statistical Analysis
Data were expressed as mean ± SD of three independent experiments. Statistical analysis were performed through GraphPad Prism software Version 5 using one way ANOVA followed by Turkey’s t- test. Differences were considered to be significant when p values < 0.05.
4. Conclusions
Our findings suggest that the presence of compounds such as lignans and alkaloids plays an important role in the overall cytotoxic effects of Z. rhetsa bark extract towards the melanoma B16-F10 cell line. In contrast, all the solvent fractions of the bark extract and the isolated compounds were found to be relatively non-toxic to HDF cells. The selective cytotoxicity of the extract and isolated compounds may be due to the metabolic defects in the cancerous cells or the peculiar reaction of the compounds against the melanoma cells [44,45,46,47]. Nevertheless, the mechanism of action underlying the selective cytotoxicity is worthy of further study to fully understand the exact mechanism. Overall, the results of this study indicated that Z. rhetsa bark extract may have potential use as a dermo protective ingredient in skincare or cosmeceutical products.
Supplementary Materials
Supplementary materials can be accessed at: http://www.mdpi.com/1420-3049/21/6/652/s1.
Acknowledgments
The authors thank Dr. Shamsul Khamis for plant collection and Universiti Putra Malaysia (UPM) for grant provided under the Research University Grant Scheme (RUGS 05-02-12-1863RU).
Author Contributions
R.K.S. and K.S. designed, conducted the experiment and purified the compounds. R.K.S., K.S., and S.A. analyzed the data. R.K.S., M.T.A. and K.S wrote the article. S.A., Y.R., F.A., I.S.I., K.S. supervised and provided intellectual input throughout the research work.
Conflicts of Interest
The authors have no competing interests to declare.
References
- Kolarsick, P.A.J.; Kolarsick, M.A.; Goodwin, C. Anatomy and physiology of the skin. J. Dermatol. Nurses Assoc. 2011, 3, 203–213. [Google Scholar] [CrossRef]
- The Skin Cancer Foundation. Available online: http://www.skincancer.org/skin-cancer-information/skin-cancer-facts#melanoma (accessed on 29 March 2016).
- Melanoma Skin Cancer. Available online: http://www.cancer.org/acs/groups/cid/documents/webcontent/003120-pdf.pdf/ (accessed on 1 November 2015).
- Skin Cancer Facts. Available online: http://www.cancer.org/cancer/cancercauses/sunanduvexposure/skin-cancer-facts/ (accessed on 18 May 2015).
- Melanoma of the skin statistics. Available online: http://canceraustralia.gov.au/affected-cancer/cancer-types/melanoma-skin/melanoma-skin-statistics/ (accessed on 10 October 2015).
- Hartley, T.G. A revision of the Malaysian species of Zanthoxylum (Rutaceae). J. Arnold Arboretum. 1970, 51, 423–426. [Google Scholar]
- Payum, T.; Das, A.K.; Shankar, R.; Tamuly, C.; Hazarika, M. Folk use and antioxidant potential determination of Zanthoxylum rhetsa DC. shoot-a highly utilized hot spice folk vegetable of Arunachal Pradesh, India. Int. J. Pharm. Sci. Res. 2013, 4, 1000–1005. [Google Scholar]
- Singh, A.; Singh, R.K.; Bharadwaj, R.; Singh, A.K. Adaptations of culturally and nutritionally important foods in Eastern Himalaya: A case study with Adi women of Arunachal Pradesh. Indian J. Tradit. Knowl. 2014, 11, 623–633. [Google Scholar]
- Nimachow, G.; Rawar, J.S.; Arunachalam, A.; Oyi, D. Ethno-medicines of Aka tribe, West Kameng District, Arunachal Pradesh (India). Sci. Cult. 2011, 77, 149–155. [Google Scholar]
- Patiño, L.O.J.; Prieto, R.J.A.; Cuca, S.L.E. Zanthoxylum Genus as Potential Source of Bioactive Compounds. In Bioactive Compounds in Phytomedicine; Rasooli, I., Ed.; InTech: Rijeka, Croatia, 2008; pp. 185–218. [Google Scholar]
- Ahsan, M.; Zaman, T.A.; Hasan, C.M.; Ito, C.; Islam, S.K.N. Constituents and cytotoxicity of Zanthoxylum rhesta stem bark. Fitoterapia 2000, 71, 697–700. [Google Scholar] [CrossRef]
- Tantapakul, C.; Phakhodee, W.; Ritthiwigrom, T.; Yossathera, K.; Deachathai, S.; Laphookhieo, S. Antibacterial compounds from Zanthoxylum rhetsa. Arch. Pharm. Res. 2012, 35, 1139–1142. [Google Scholar] [CrossRef] [PubMed]
- Ahsan, M.; Haque, M.R.; Hossain, M.B.; Islam, S.N.; Gray, A.I.; Hasan, C.M. Cytotoxic dimeric quinolone-terpene alkaloids from the root bark of Zanthoxylum rhetsa. Phytochemistry 2014, 103, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Lalitharani, S.; Kalpanadevi, V.; Mohan, V.R. Pharmacognostic studies on the spine of Zanthoxylum rhetsa (Roxb.) DC. Biosci. Discov. 2013, 4, 5–11. [Google Scholar]
- Shankaracharya, N.B.; Naik, J.P.; Nagalakshmi, S.; Rao, L.J.M. Chemical composition and flavour quality of tirphal Zanthoxylum rhetsa. PAFAI J. 1994, 16, 15–21. [Google Scholar]
- Shafi, P.M.; Saidutty, A.; Clery, R.A. Volatile Constituents of Zanthoxylum rhetsa Leaves and Seeds. J. Essent. Oil Res. 2000, 12, 179–182. [Google Scholar]
- Rana, V.S.; Blazquez, M.A. Volatile Constituents of the Seed Coat of Zanthoxylum rhetsa (Roxb.) DC. J. Essent. Oil Res. 2010, 22, 430–432. [Google Scholar] [CrossRef]
- Thu, N.B.; Trung, T.N.; Ha, D.T. Zanthoxylum rhetsa Stem bark extract inhibits LPS-induced COX-2 and iNOS expression in RAW 264.7 cells via the NF-κB inactivation. Nat. Prod. Sci. 2010, 16, 265–270. [Google Scholar]
- Kale, S.S.; Rajmane, A.H.; Urunkar, V.C.; Gaikwad, M.K.; Bhandare, S.B. Formulation and In-vitro Evaluation of Sun Protection Factor of Methanolic Extract of Zanthoxylum rhetsa DC Sunscreen lotion. Res. J. Pharmacogn. Phytochem. 2011, 3, 206–210. [Google Scholar]
- Santhanam, R.; Ahmad, S.; Abas, F.; Ismail, I.S.; Rukayadi, Y.; Shaari, K. Photoprotective properties of Zanthoxylum rhetsa: An in vitro analysis. J. Chem. Pharm. Res. 2013, 5, 1512–1520. [Google Scholar]
- Yang, C.H.; Cheng, M.J.; Lee, S.J.; Yang, C.W.; Chang, H.S.; Chen, I.S. Secondary metabolites and cytotoxic activities from the stem bark of Zanthoxylum nitidum. Chem. Biodivers. 2009, 6, 846–857. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Sun, Y.N.; Yan, X.T.; Yang, S.Y.; Kim, E.J.; Kang, H.K.; Kim, Y.H. Coumarins and lignans from Zanthoxylum schinifolium and their anticancer activities. J. Agric. Food Chem. 2013, 61, 10730–10740. [Google Scholar] [CrossRef] [PubMed]
- Gaya, C.H.; Kawaka, J.F.; Muchugi, A.; Ngeranwa, J.J. Variation of alkaloids in the Kenyan Zanthoxylum gilletii (De Wild Waterman). Afr. J. Plant Sci. 2013, 7, 438–444. [Google Scholar] [CrossRef]
- Mukhija, M.; Dhar, K.L.; Kalia, A.N. Bioactive Lignans from Zanthoxylum alatum Roxb. stem bark with cytotoxic potential. J. Ethnopharmacol. 2014, 152, 106–112. [Google Scholar] [PubMed]
- Kumar, V.; Kumar, S.; Singh, B.; Kumar, N. Quantitative and structural analysis of amides and lignans in Zanthoxylum armatum by UPLC-DAD-ESI-QTOF-MS/MS. J. Pharm. Biomed. Anal. 2014, 94, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Pelter, A. The mass spectra of oxygen heterocycles. Part IV. The mass spectra of some complex lignans. J. Chem. Soc. C Org. 1967. [Google Scholar] [CrossRef]
- Tonelli, F.M.P.; de Siqueira, J.M.; Mala, G.A.S.; Soares, L.F.; da Silva, D.B.; Carolla, C.A.; Sartori, A.L.B. Bioautography as a search tool to identify the allelopathic compounds in Virola sebifera. Allelopath J. 2014, 33, 277–288. [Google Scholar]
- Paik, S.Y.; Koh, K.H.; Beak, S.M.; Paek, S.H.; Kim, J.A. The essential oils from Zanthoxylum schinifolium pericarp induce apoptosis of HepG2 human hepatoma cells through increased production of reactive oxygen species. Biol. Pharm. Bull. 2005, 28, 802–807. [Google Scholar] [CrossRef] [PubMed]
- Lan, Y.; Li, H.; Chen, Y.Y.; Zhang, Y.W.; Liu, N.; Zhang, Q.; Wu, Q. Essential oil from Zanthoxylum bungeanum Maxim. and its main components used as transdermal penetration enhancers: A comparative study. J. Zhejiang Univ. Sci B 2014, 15, 940–952. [Google Scholar] [PubMed]
- Su, G.Y.; Wang, K.W.; Wang, X.Y.; Wu, B. Bioactive lignans from Zanthoxylum planispinum with cytotoxic potential. Phytochem. Lett. 2015, 11, 120–126. [Google Scholar] [CrossRef]
- Jain, P.S.; Bari, S.B. Isolation of lupeol, stigmasterol and campesterol from petroleum ether extract of woody stem of Wrightia tinctoria. Asian J. Plant Sci. 2010, 9, 163–167. [Google Scholar] [CrossRef]
- Chang, S.W.; Kim, K.H.; Lee, I.K.; Choi, S.U.; Lee, K.R. Phytochemical Constituents of Geranium eriostemon. Nat. Prod. Sci. 2009, 15, 151–155. [Google Scholar]
- Kim, J.Y.; Lim, H.J.; Lee, D.Y.; Kim, J.S.; Kim, D.H.; Lee, H.J.; Kim, H.D.; Jeon, R.; Ryu, J.H. In vitro anti-inflammatory activity of lignans isolated from Magnolia fargesii. Bioorg. Med. Chem. Lett. 2009, 19, 937–940. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, T.J.; Chia, Y.C.; Wu, Y.C.; Chen, C.Y. Chemical Constituents from the Stems of Mahonia Japonica. J. Chin. Chem. Soc. 2004, 51, 443–446. [Google Scholar] [CrossRef]
- Rahman, M.M.; Islam, M.A.; Khondkar, P.; Gray, A.I. Alkaloids and lignans from Zanthoxylum budrunga (Rutaceae). Biochem. Syst. Ecol. 2005, 33, 91–96. [Google Scholar] [CrossRef]
- Stermitz, F.R.; Sharifi, I.A. Alkaloids of Zanthoxylum monophyllum and Zanthoxylum punctatum. Phytochemistry 1977, 16, 2003–2006. [Google Scholar] [CrossRef]
- Sreelekha, M.; Anto, N.P.; Anto, R.J.; Shafi, R.J. Cytotoxicity of 6-acetonyldihydro-chelerythrin, arnottianamide and 6-(2-hydroxypropyl)-dihydrochelerythrine towards human cancer cell lines. Indian J. Chem. Sect. B 2014, 53, 647–651. [Google Scholar]
- Zhang, L.L.; Ma, L.N.; Yan, D.; Zhang, C.E.; Gao, D.; Xiong, Y.; Sheng, F.Y.; Dong, X.P.; Xiao, X.H. Dynamic monitoring of the cytotoxic effects of protoberberine alkaloids from Rhizoma coptidis on HepG2 cells using the xCELLigence system. Chin. J. Nat. Med. 2014, 12, 428–435. [Google Scholar] [CrossRef]
- Badami, S.; Vijayan, P.; Mathew, N.; Chandrasekhar, R.; Godavarthi, A.; Dhanaraj, S.A.; Suresh, B. In vitro cytotoxic properties of Grewia tiliaefolia bark and lupeol. Indian J. Pharmacol. 2003, 35, 250–251. [Google Scholar]
- Saleem, M. Lupeol, a novel anti-inflammatory and anti-cancer dietary triterpene. Cancer Lett. 2009, 285, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Sung, J.H.; Park, S.H.; Seo, D.H.; Lee, J.H.; Hong, S.W.; Hong, S.S. Antioxidative and skin-whitening effect of an aqueous extract of Salicornia herbacea. Biosci. Biotechnol. Biochem. 2009, 73, 552–556. [Google Scholar] [CrossRef] [PubMed]
- Iwashita, K.; Kobori, M.; Yamaki, K.; Tsushida, T. Flavonoids inhibit cell growth and induce apoptosis in B16 melanoma 4A5 cells. Biosci. Biotechnol. Biochem. 2000, 64, 1813–1820. [Google Scholar] [CrossRef] [PubMed]
- Chinembirim, T.N.; du Plessis, L.H.; Gerber, M.; Hamman, J.H.; du Plessis, J. Review of natural compounds for potential skin cancer treatment. Molecules 2014, 19, 11679–11721. [Google Scholar] [CrossRef] [PubMed]
- Albert, A. Fundamental aspects of selective toxicity. Ann. N. Y. Acad. Sci. 1965, 123, 5–18. [Google Scholar] [CrossRef]
- Milajerdi, A.; Djafarian, K.; Hosseini, B. The toxicity of saffron (Crocus sativus L.) and its constituents against normal and cancer cells. J. Nutr. Intermed. Metab. 2016, 3, 23–32. [Google Scholar] [CrossRef]
- Sandjo, L.P.; Kuete, V.; Tchangna, R.S.; Efferth, T.; Ngadjui, B.T. Cytotoxic Benzophenanthridine and Furoquinoline Alkaloids from Zanthoxylum buesgenii (Rutaceae). Chem. Cent. J. 2014, 8. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-H.; Jang, Y.-O.; Kim, B.-T.; Hwang, K.-J.; Lee, J.-C. Induction of caspase-dependent apoptosis in melanoma cells by the synthetic compound (E)-1-(3,4-dihydroxyphenethyl)-3-styrylurea. BMB Rep. 2009, 42, 806–811. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Pure compounds not available, solvent fractions are available from the authors.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).