Antimicrobial Activity and Chromatographic Analysis of Extracts from Tropaeolum pentaphyllum Lam. Tubers
Abstract
:1. Introduction
2. Results
2.1. Extraction Methods
2.2. Antimicrobial Activity
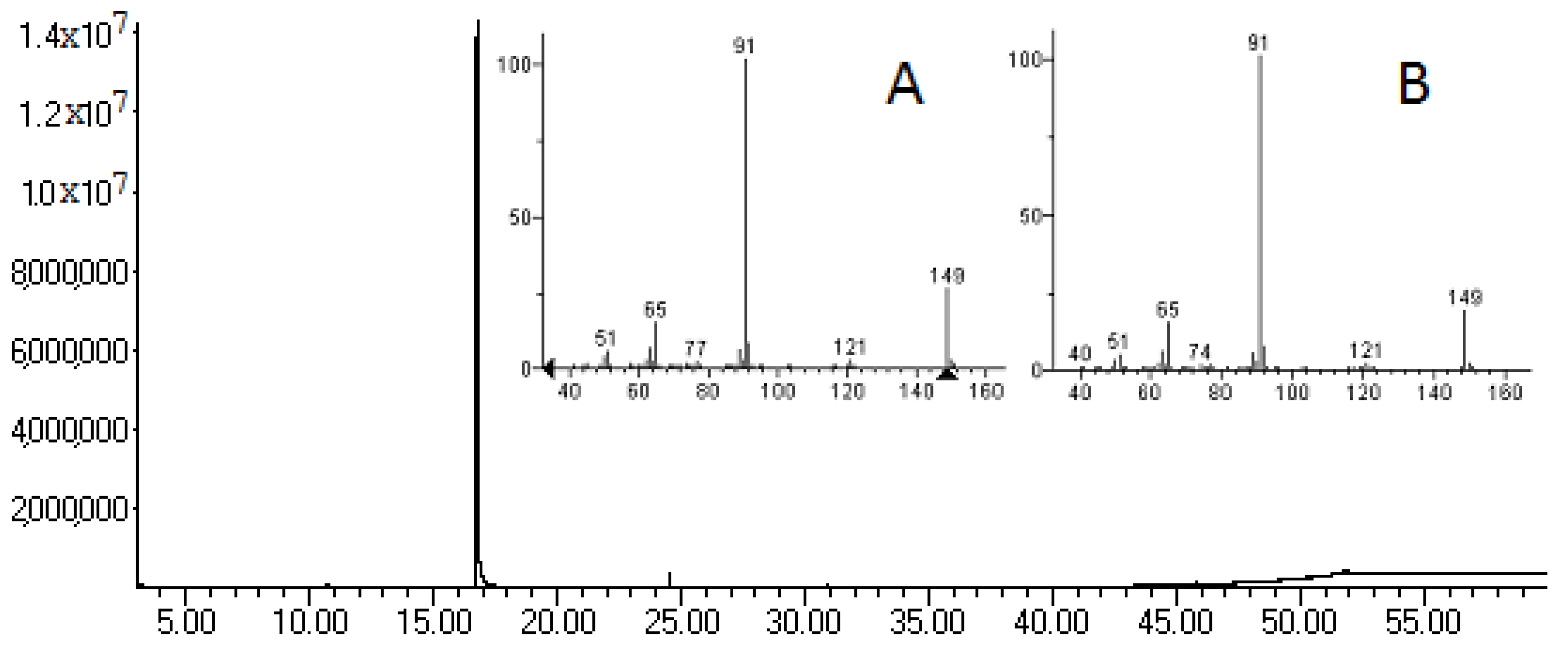
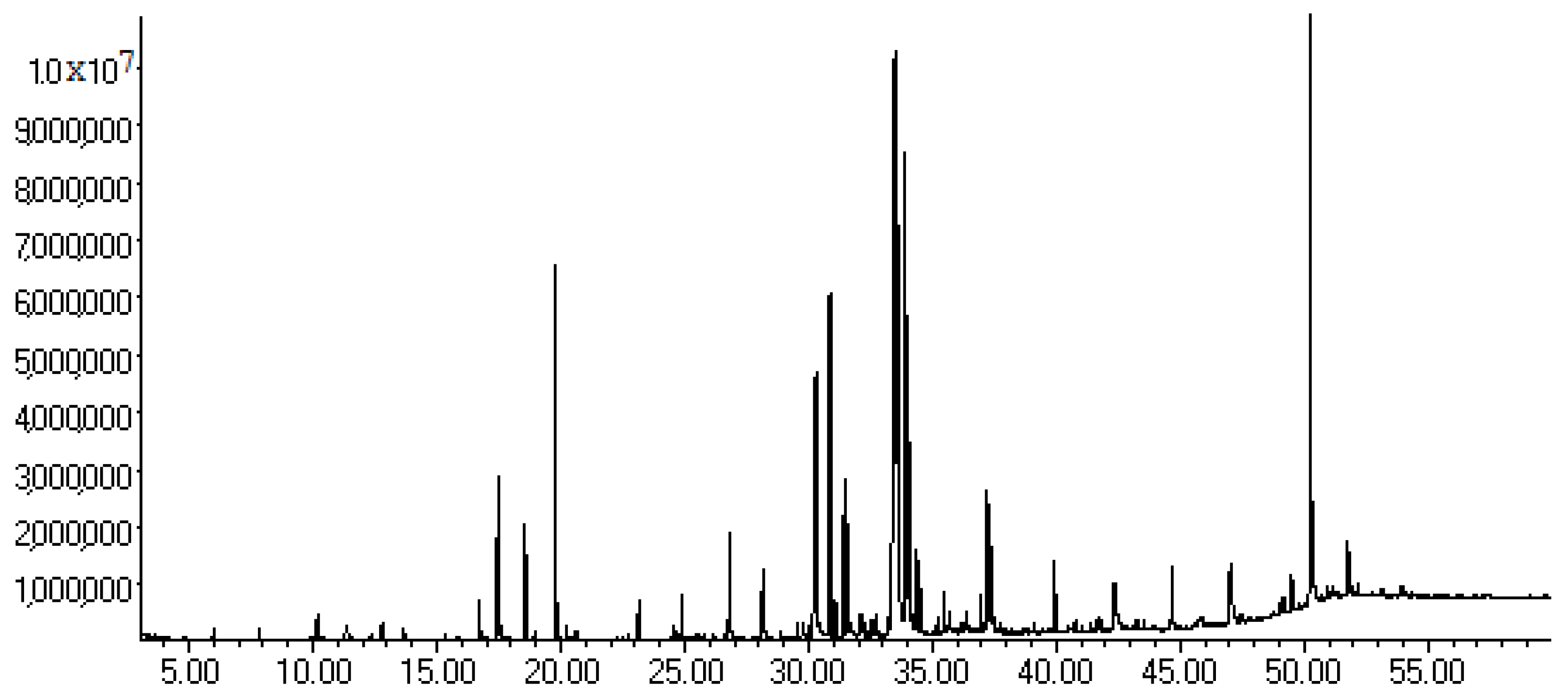
2.3. Chromatographic Analysis
3. Discussion
4. Materials and Methods
4.1. Plant Material and Extraction Methods
4.2. Antimicrobial Assays
4.2.1. Antibacterial Activity
4.2.2. Antifungal Activity
4.3. Chromatographical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rix, M. Tropaeolaceae: 687. Tropaeolum pentaphyllum. Curtis’s Bot. Mag. 2010, 27, 296–300. [Google Scholar] [CrossRef]
- Trojan-Rodrigues, M.; Alves, T.L.S.; Soares, G.L.G.; Ritter, M.R. Plants used as antidiabetics in popular medicine in Rio Grande do Sul, southern Brazil. J. Ethnopharmacol. 2012, 139, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Zuchiwschi, E.; Fantini, A.C.; Alves, A.C.; Peroni, N. Limitações ao uso de espécies florestais nativas pode contribuir com a erosão do conhecimento ecológico tradicional e local de agricultores familiares. Acta Bot. Bras. 2010, 24, 270–282. [Google Scholar] [CrossRef]
- Agneta, R.; Möllers, C.; Rivelli, A.R. Horseradish (Armoracia rusticana), a neglected medical and condiment species with a relevant glucosinolate profile: A review. Genet. Resour. Crop. Evol. 2013, 60, 1923–1943. [Google Scholar] [CrossRef]
- Fenner, R.; Betti, A.H.; Mentz, L.A.; Rates, S.M.K. Plantas utilizadas na medicina popular brasileira com potencial atividade antifúngica. Braz. J. Pharm. Sci. 2006, 42, 369–394. [Google Scholar] [CrossRef]
- Aly, R. Microbial infections of skin and nail. In Medical Microbiology, 4th ed.; Baron, S., Ed.; University of Texas Medical Branch: Galveston, TX, USA, 1996. [Google Scholar]
- Cos, P.; Vlietinck, A.J.; Berghe, V.D.; Maes, L. Anti-infective potential of natural products: How to develop a stronger in vitro ‘proof-of-concept’. J. Ethnopharmacol. 2006, 106, 290–302. [Google Scholar] [CrossRef] [PubMed]
- Pubchem Azithramycine. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/55185?from=summary#section=Top (accessed on 20 February 2016).
- Kjaer, A.; Madsen, Ø.; Maeda, Y. Seed volatiles within the family Tropaeolaceae. Phytochemistry 1978, 17, 1285–1287. [Google Scholar] [CrossRef]
- Sofrata, A.; Santagelo, E.M.; Azeem, M.; Borg-Karlson, A.-K.; Gustafsson, A.; Pütsep, K. Benzyl isothiocyanate, a major component from the roots of Salvadora persica is highly active against Gram-negative bacteria. PLoS ONE 2011, 6, e23045. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.G.; Lee, H.S. Growth-inhibiting activities of phenethyl isothiocyanate and its derivatives and against intestinal bacteria. J. Food. Sci. 2009, 74, 467–471. [Google Scholar] [CrossRef] [PubMed]
- Dias, C.; Aires, A.; Saavedra, M.J. Antimicrobial activity of isothiocyanates from cruciferous plants against Methicillin-Resistant Staphylococcus aureus (MRSA). Int. J. Mol. Sci. 2014, 15, 19552–19561. [Google Scholar] [CrossRef] [PubMed]
- Dufour, V.; Stahl, M.; Baysse, C. The antibacterial properties of isothiocyanates. Microbiology 2015, 161, 229–243. [Google Scholar] [CrossRef] [PubMed]
- Borges, A.; Serra, S.; Abreu, A.C.; Saavedra, M.J.; Salgado, A.; Simões, M. Evaluation of the effects of selected phytochemicals on quorum sensing inhibition and in vitro cytotoxicity. Biofouling 2014, 30, 183–195. [Google Scholar] [CrossRef] [PubMed]
- Dufour, V.; Alazzam, B.; Ermel, G.; Thepaut, M.; Rossero, A.; Tresse, O.; Baysse, C. Antimicrobial activities of isothiocyanates against Campylobacter jejuni isolates. Front. Cell Infect. Microbiol. 2012, 2, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Dufour, V.; Stahl, M.; Rosenfeld, E.; Stintzi, A.; Baysse, C. Insights into the mode of action of benzyl isothiocyanate on Campylobacter jejuni. Appl. Environ. Microbiol. 2013, 79, 6958–6968. [Google Scholar] [CrossRef] [PubMed]
- Khalil, A.T. Benzylamides from Salvadora persica. Arch. Pharm. Res. 2006, 29, 952–956. [Google Scholar] [CrossRef] [PubMed]
- Kawakishi, S.; Namiki, M. Decomposition of allyl isothiocyanate in aqueous solution. Agric. Biol. Chem. 1968, 33, 452–459. [Google Scholar] [CrossRef]
- Pecháček, R.; Velišek, J.; Hrabcová, H. Decomposition products of allyl isothiocyanate in aqueous solution. J. Agric. Food Chem. 1997, 45, 4584–4588. [Google Scholar] [CrossRef]
- Libenson, L.; Hadley, F.P.; McIlroy, A.P.; Wetzel, V.M.; Mellon, RR. Antibacterial effect of elemental sulfur. J. Infect. Dis. 1953, 93, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Kabara, J.J.; Swieczkowski, D.M.; Conley, A.J.; Truant, J.P. Fatty acids and derivatives as antimicrobial agents. Antimicrob. Agents Chemother. 1972, 2, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Tamokou, J.D.; Mpetga, D.J.S.; Lunga, P.K.; Tene, M.; Tane, P.; Kuiate, J.R. Antioxidant and antimicrobial of ethyl acetate extract, fractions and compounds from stem bark of Albizia adianthifolia (Mimosoideae). BMC Complement. Altern. Med. 2012, 12, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Drobnica, L.; Zemanová, M.; Nemec, P.; Antos, K.; Kristián, P.; Stullerová, A.; Knoppová, V.; Nemec, P., Jr. Antifungal activity of isothiocyanates and related compounds I. Naturally ocurring isothiocyanates and their analogues. Appl. Environ. Microb. 1967, 15, 701–709. [Google Scholar]
- Manici, L.M.; Lazzeri, L.; Palmieri, S. In vitro Fungitoxic activity of some glucosinolates and their enzyme-derived products toward plant pathogenic fungi. J. Agric. Food Chem. 1997, 45, 2768–2773. [Google Scholar] [CrossRef]
- Tweedy, B.G. Inorganic sulfur as a fungicide. In Residue Reviews; Gunther, F.A., Gunther, J.D., Eds.; Springer: New York, NY, USA, 1981; pp. 43–68. [Google Scholar]
- Thillairajasekar, K.; Duraipandiyan, V.; Perumal, P.; Ignacimuthu, S. Antimicrobial activity of Trichodesmium erythraeum (Ehr) (microalga) from South East coast of Tamil Nadu, India. Int. J. Integr. Biol. 2009, 5, 167–170. [Google Scholar]
- Pohl, C.H.; Kock, J.L.F.; Thibane, V.S. Antifungal free fatty acids: A review. In Science against Microbial Pathogens: Communicating Current Research and Technological Advances; Méndez-Vilaz, A., Ed.; Formatex: Badajoz, Spain, 2011; pp. 61–71. [Google Scholar]
- Avis, T.J.; Bélanger, R.B. Specificity and mode of action of the antifungal fatty acid cis-9-Heptadecenoic acid produced by Pseudozyma flocculosa. Appl. Environ. Microbiol. 2001, 67, 956–960. [Google Scholar] [CrossRef] [PubMed]
- Moser, K.; Kriwet, K.; Naik, A.; Kalia, Y.N.; Guy, R.H. Passive skin penetration enhancement and its quantification in vitro. Eur. J. Pharm. Biopharm. 2001, 52, 103–112. [Google Scholar] [CrossRef]
- Fox, L.T.; Gerber, M.; Du Pleiss, J.; Hamman, J.H. Transdermal drug delivery enhancement by compounds of natural origin. Molecules 2011, 16, 10507–10540. [Google Scholar] [CrossRef]
- Fang, J.-Y.; Chiu, H.-C.; Wu, J.-T.; Chiang, Y.-R.; Hsu, S.-H. Fatty acids in Botryococcus braunii accelerate topical delivery of flurbiprofen into and across skin. Int. J. Pharm. 2004, 276, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Kezutyte, T.; Desbenoit, N.; Brunelle, A.; Briedis, V. Studying the penetration of fatty acids into human skin by ex vivo TOF-SIMS imaging. Biointerphases 2013, 8, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Clinical and Laboratory Standards Institute. Methods for Dilution Antimicrobial Susceptibility Test for Bacteria that Grow Aerobically. Approved Standard, M07-A9, 9th ed.; CLSI: Wayne, MI, USA, 2012. [Google Scholar]
- Clinical and Laboratory Standards Institute. Methods for Dilution Antimicrobial Susceptibility Test for Bacteria that Grow Aerobically. Approved Standard, M27-A3, 3th ed.; CLSI: Wayne, MI, USA, 2008. [Google Scholar]
- Clinical and Laboratory Standards Institute. Reference Method for Broth Dilution Antifungals Susceptibility Testing of Conidium-Forming Filamentous Fungi: Approved Standard, M38-A2, 2nd ed.; CLSI: Wayne, MI, USA, 2008. [Google Scholar]
- Sample Availability: Samples of all the extracts and the essential oil are available from the authors.
| Bacteria | MIC/MBC (µg/mL) | ||||
|---|---|---|---|---|---|
| EO | CfF | EaF | BITC | Azithromycin | |
| Enterococcus faecalis ATCC 91299 | 40/640 | 80/640 | 80/1280 | 40/640 | 8/- |
| Escherichia coli ATCC 5922 | 10/320 | 10/80 | 20/640 | 10/320 | 8/- |
| Klebsiella pneumoniae ATCC 700603 | 40/1280 | 40/640 | 320/- | 40/640 | 2/- |
| Pseudomonas aeruginosa ATCC 27853 | 20/320 | 40/640 | 40/640 | 20/320 | 4/- |
| Salmonella pullorum ATCC 9140 | 40/640 | 10/- | 80/1280 | 40/640 | 8/- |
| Staphylococcus aureus ATCC 29213 | 40/1280 | 40/640 | 640/- | 40/320 | 2/- |
| Fungus | MIC/MFC (µg/mL) | ||||
|---|---|---|---|---|---|
| EO | CfF | EaF | BITC | Fluconazole | |
| Candida albicans ATCC 14053 | 40/320 | 20/20 | 40/320 | 40/160 | 4/- |
| Candida dubliniensis CBS7987 | 40/320 | 10/10 | 40/640 | 40/160 | 8/- |
| Candida dubliniensis CI FS | 40/160 | 20/20 | 40/640 | 40/160 | 2/- |
| Candida dubliniensis CI FR | 40/160 | 20/20 | 40/640 | 40/160 | 64/- |
| Candida glabrata ATCC 2001 | 20/80 | 40/40 | 40/320 | 20/80 | 32/- |
| Candida glabrata CI FS | 20/160 | 40/40 | 40/640 | 20/160 | 16/- |
| Candida glabrata CI FR | 20/160 | 40/40 | 20/640 | 20/160 | 128/- |
| Candida glabrata CI CR | NT | 160/1280 | NT | 160/640 | 4/- |
| Candida guilliermondii CI | 80/320 | 10/10 | 80/320 | 80/320 | 8/- |
| Candida parapsilosis ATCC 22018 | 80/320 | 10/20 | 80/640 | 80/320 | 16/- |
| Candida parapsilosis CI CR | NT | 320/1280 | NT | 320/1280 | 8/- |
| Candida tropicalis CI | 320/- | 10/10 | 1280/- | 320/- | 8/- |
| Cryptococcus neoformans ATCC 90012 | 10/80 | 2.5/2.5 | 10/320 | 5/80 | 16/- |
| Sacharomyces cerevisiae ATCC 2601 | 40/320 | 2.5/2.5 | 80/320 | 40/160 | 4/- |
| Fungus | MIC/MFC (µg/mL) | ||||
|---|---|---|---|---|---|
| EO | CfF | EaF | BITC | Itraconazole | |
| Aspergillus fumigatus CI | 80/1280 | 160/1280 | 320/- | 80/1280 | 8/- |
| Aspergillus fumigatus EI | 20/1280 | 80/640 | 40/- | 20/1280 | 16/- |
| Aspergillus flavus CI | NT | 1280/- | NT | 160/1280 | 1/- |
| Aspergillus niger CI | NT | 1280/- | NT | 160/1280 | 2/- |
| Trichophyton rubrum CI | NT | 2.5/320 | NT | 2.5/40 | 2/- |
| Microsporum canis CI | NT | 20/160 | NF | 20/80 | 8- |
| Fonsecaea pedrosoi CI | NT | 80/640 | NT | 40/320 | 1/- |
| Pseudallescheria boydii CI | NT | 80/1280 | NT | 40/640 | 4/- |
| Fusarium solani CI | NT | 320/- | NT | 160/1280 | >16/- |
| Sporothrix schenckii CI | NT | 40/320 | NT | 80/320 | 4/- |
| Compound | RT (min) | Relative Area (%) | (M+), m/z (%) | Major Fragment Ions, m/z (%) |
|---|---|---|---|---|
| Benzyl isothiocyanate | 16.72 | 0.65% | 149 (16) | 91 (100) 65 (14) 92 (7) 89 (5) 63 (5) 51 (4) 50 (3) 90 (3) 62 (2) |
| 2-Phenylacetamide | 17.49 | 4.37% | 135 (18) | 91 (100) 92 (91) 65 (20) 44 (12) 63 (8) 89 (7) 51 (5) 93 (5) 90 (4) |
| N-benzylacetamide | 18.56 | 2.21% | 149 (71) | 106 (100) 91 (28) 43 (20) 107 (15) 77 (13) 79 (13) 51 (8) 65 (7) 150 (7) |
| Benzylcarbamide | 19.80 | 6.23% | 150 (50) | 106 (100) 79 (41) 91 (34) 77 (34) 51 (23) 107 (20) 78 (15) 104 (15) 65 (13) |
| Palmitic acid | 30.29 | 6.75% | 256 (30) | 73 (100) 60 (84) 43 (73) 57 (63) 55 (57) 41 (56) 129 (43) 71 (37) 69 (31) 83 (24) |
| Ethyl palmitate | 30.86 | 4.66% | 284 (7) | 88 (100) 101 (58) 43 (26) 55 (22) 41 (20) 89 (17) 57 (16) 70 (16) 157 (15) 73 (15) |
| Elemental sulfur | 31.61 | 1.62% | 256 (70) | 64 (100) 160 (53) 128 (52) 192 (41) 258 (25) 32 (21) 96 (21) 224 (18) 66 (11) |
| Oleic acid | 33.60 | 10.25% | 282 (1) | 55 (100) 69 (72) 41 (67) 83 (58) 97 (49) 67 (45) 43 (45) 81 (37) 84 (34) 57 (32) |
| Ethyl-9,12-octadecadienoate | 33.90 | 6.98% | 308 (8) | 67 (100) 81 (90) 95 (65) 55 (61) 41 (46) 79 (43) 82 (42) 68 (38) 96 (35) 54 (34) |
| Ethyl oleate | 34.02 | 5.88% | 310 (6) | 55 (100) 69 (71) 41 (63) 83 (60) 88 (57) 97 (51) 84 (49) 43 (47) 96 (45) 101 (45) |
| Butyl palmitate | 34.38 | 1.03% | 312 (6) | 56 (100) 57 (47) 257 (41) 43 (35) 41 (32) 55 (28) 73 (24) 60 (24) 239 (20) 129 (20) |
| Unknown phytosterol | 50.26 | 11.35% | 414 (100) | 43(95) 107 (68) 105 (66) 145 (65) 95 (62) 55 (61) 81 (60) 329 (59) 303 (58) |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Da Cruz, R.C.; Denardi, L.B.; Mossmann, N.J.; Piana, M.; Alves, S.H.; De Campos, M.M.A. Antimicrobial Activity and Chromatographic Analysis of Extracts from Tropaeolum pentaphyllum Lam. Tubers. Molecules 2016, 21, 566. https://doi.org/10.3390/molecules21050566
Da Cruz RC, Denardi LB, Mossmann NJ, Piana M, Alves SH, De Campos MMA. Antimicrobial Activity and Chromatographic Analysis of Extracts from Tropaeolum pentaphyllum Lam. Tubers. Molecules. 2016; 21(5):566. https://doi.org/10.3390/molecules21050566
Chicago/Turabian StyleDa Cruz, Ritiel Corrêa, Laura Bedin Denardi, Natalia Jank Mossmann, Mariana Piana, Sydney Hartz Alves, and Marli Matiko Anraku De Campos. 2016. "Antimicrobial Activity and Chromatographic Analysis of Extracts from Tropaeolum pentaphyllum Lam. Tubers" Molecules 21, no. 5: 566. https://doi.org/10.3390/molecules21050566
APA StyleDa Cruz, R. C., Denardi, L. B., Mossmann, N. J., Piana, M., Alves, S. H., & De Campos, M. M. A. (2016). Antimicrobial Activity and Chromatographic Analysis of Extracts from Tropaeolum pentaphyllum Lam. Tubers. Molecules, 21(5), 566. https://doi.org/10.3390/molecules21050566






