Development of an in Vitro System to Simulate the Adsorption of Self-Emulsifying Tea (Camellia oleifera) Seed Oil
Abstract
:1. Introduction
2. Results
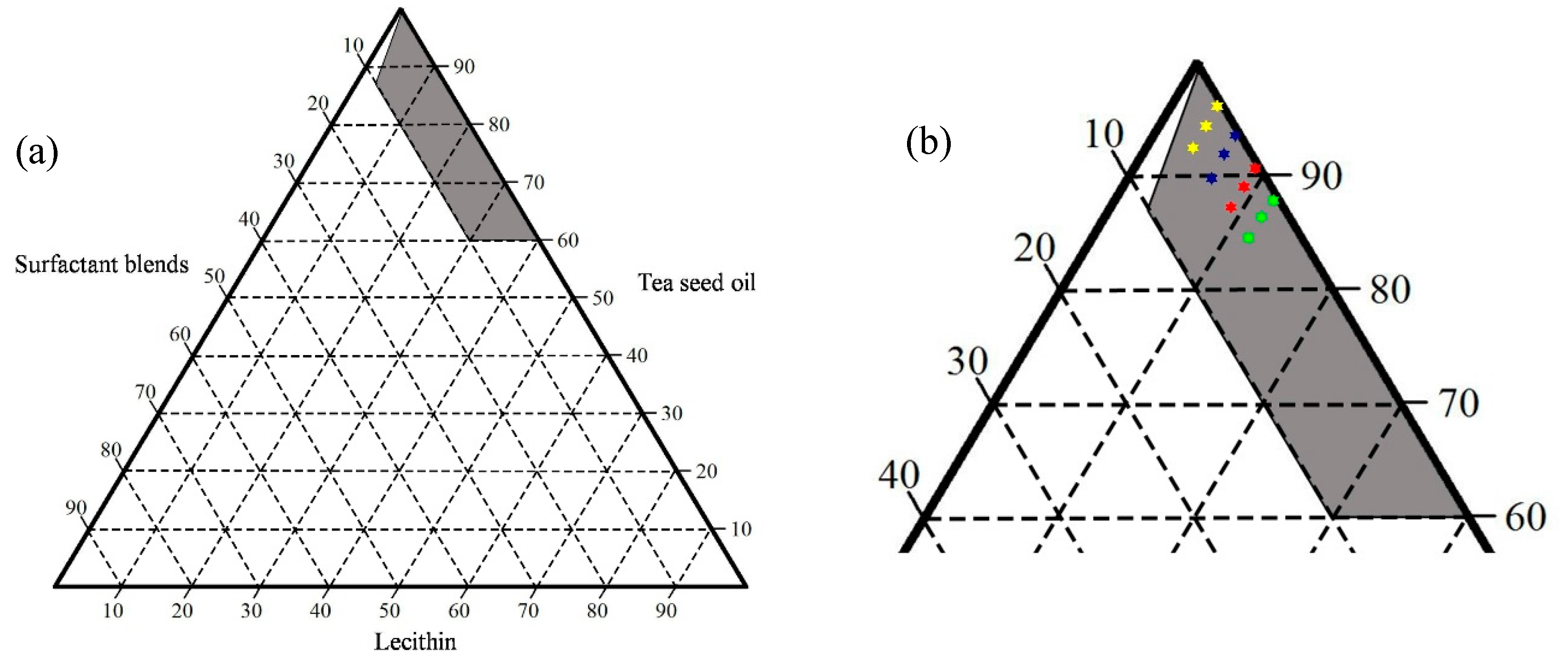
2.1. Ternary Phase Diagram
2.2. Droplet Size and Zeta Potential Determination
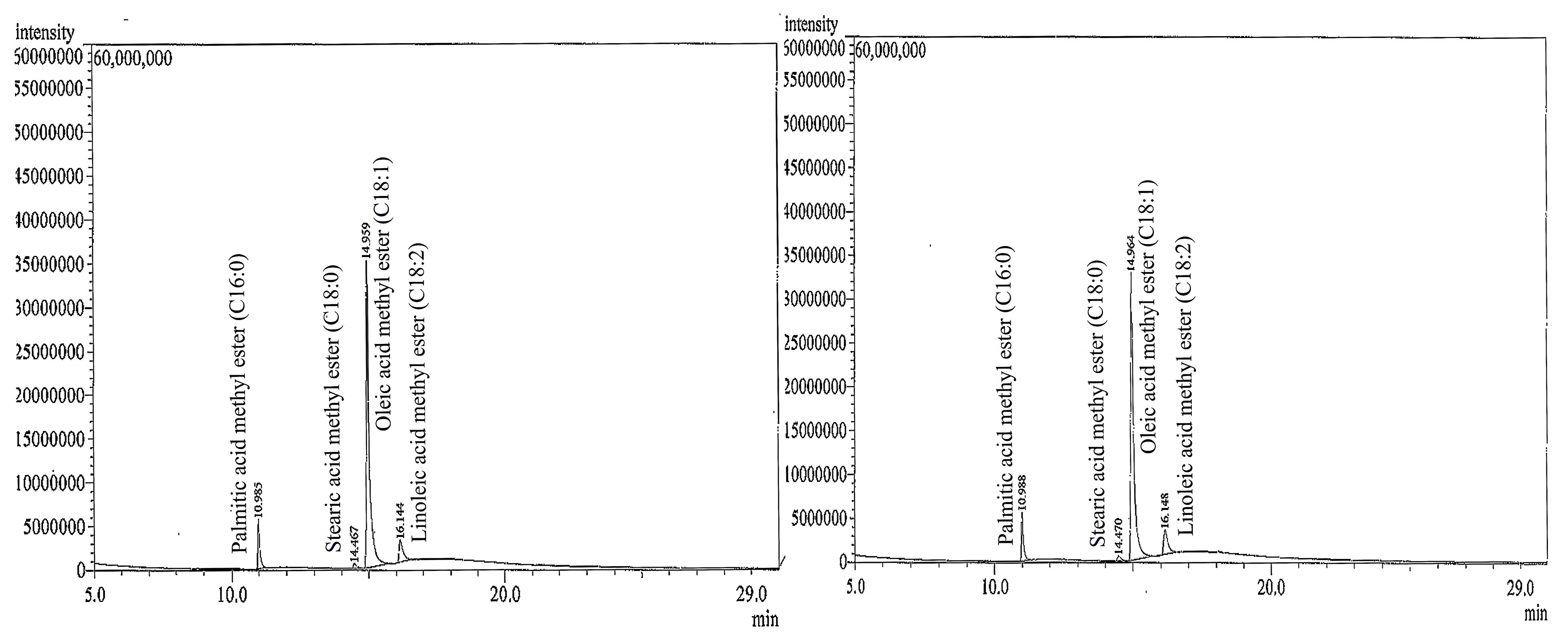
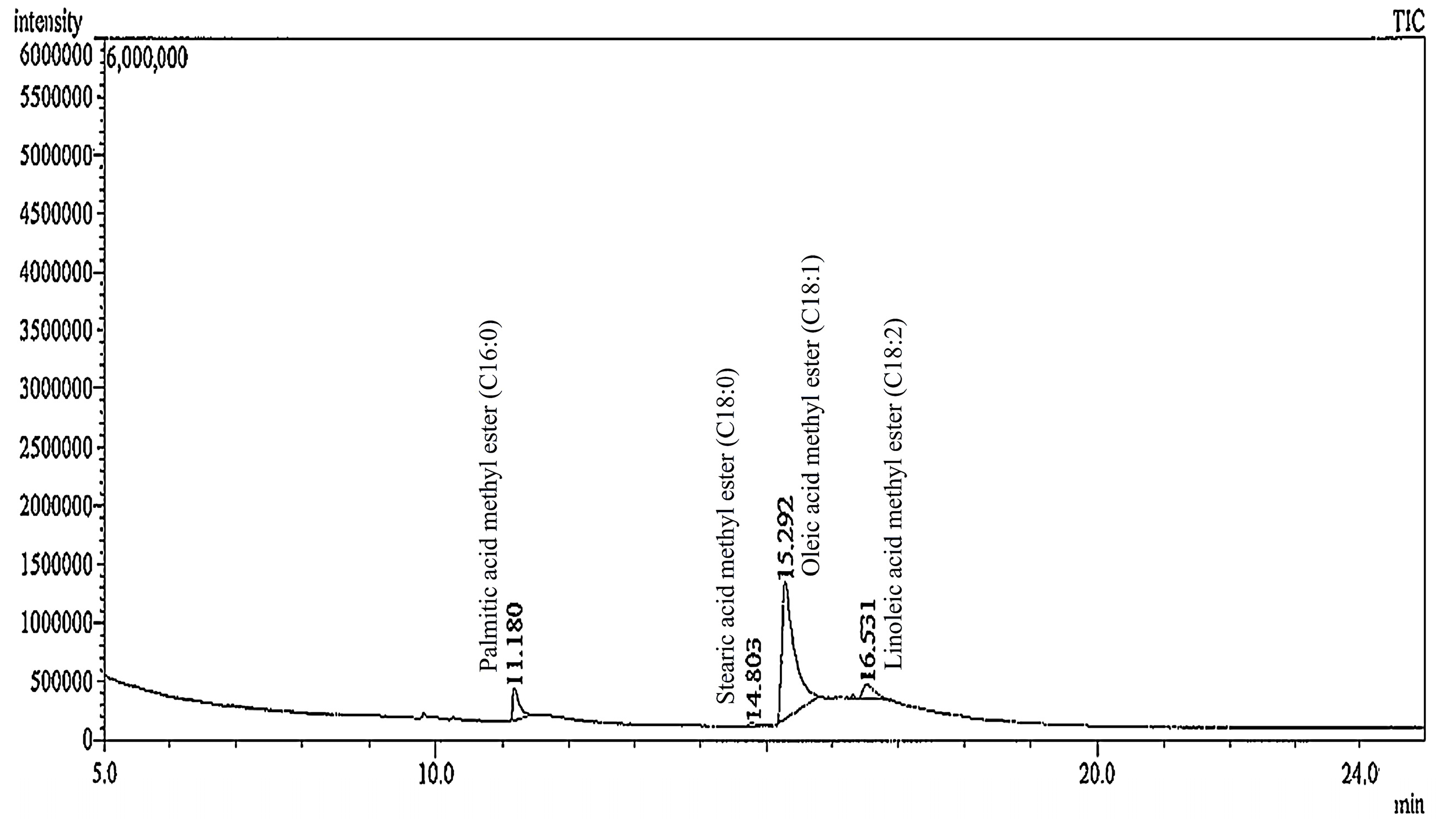
2.3. Fatty Acid Composition
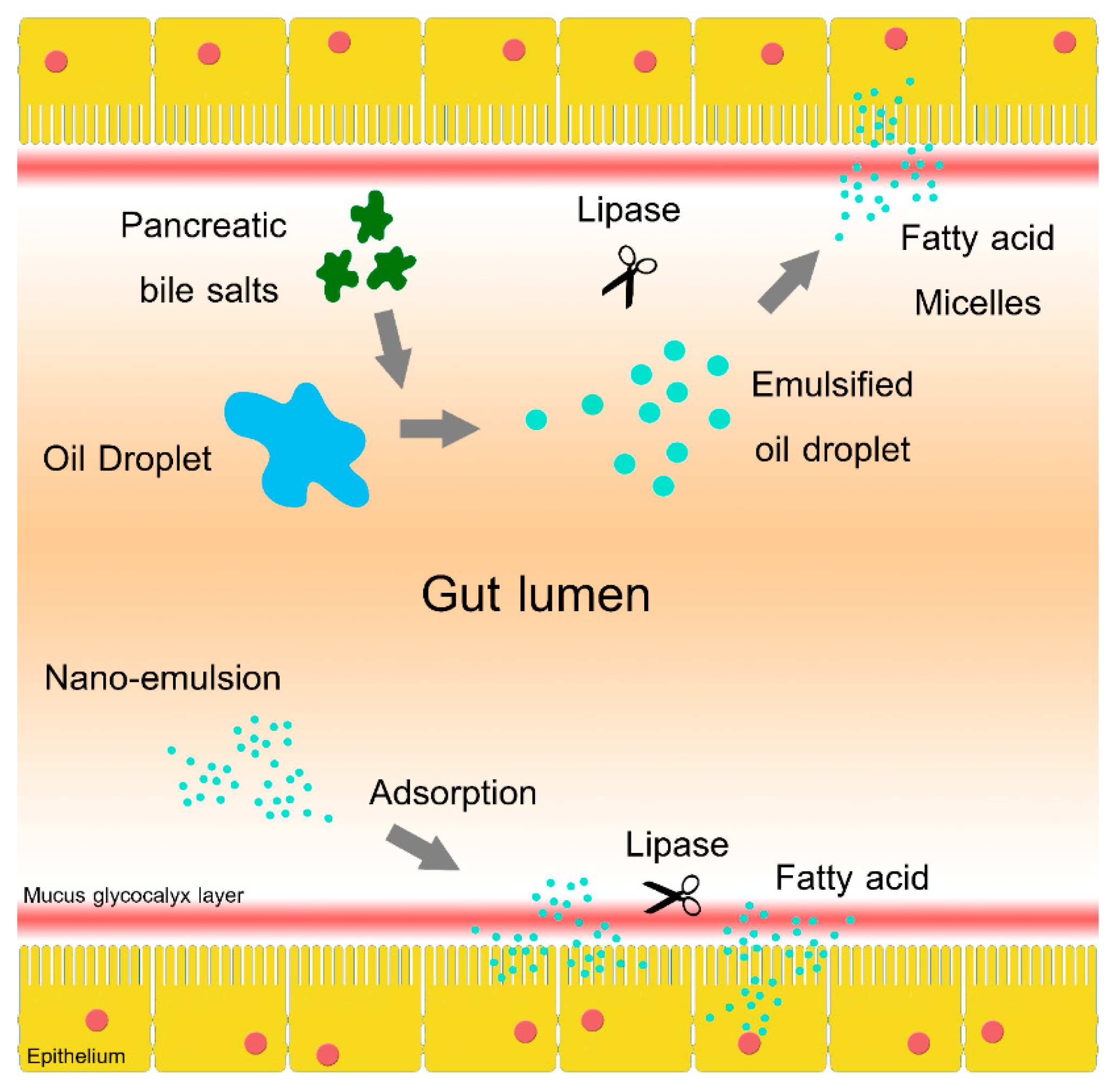
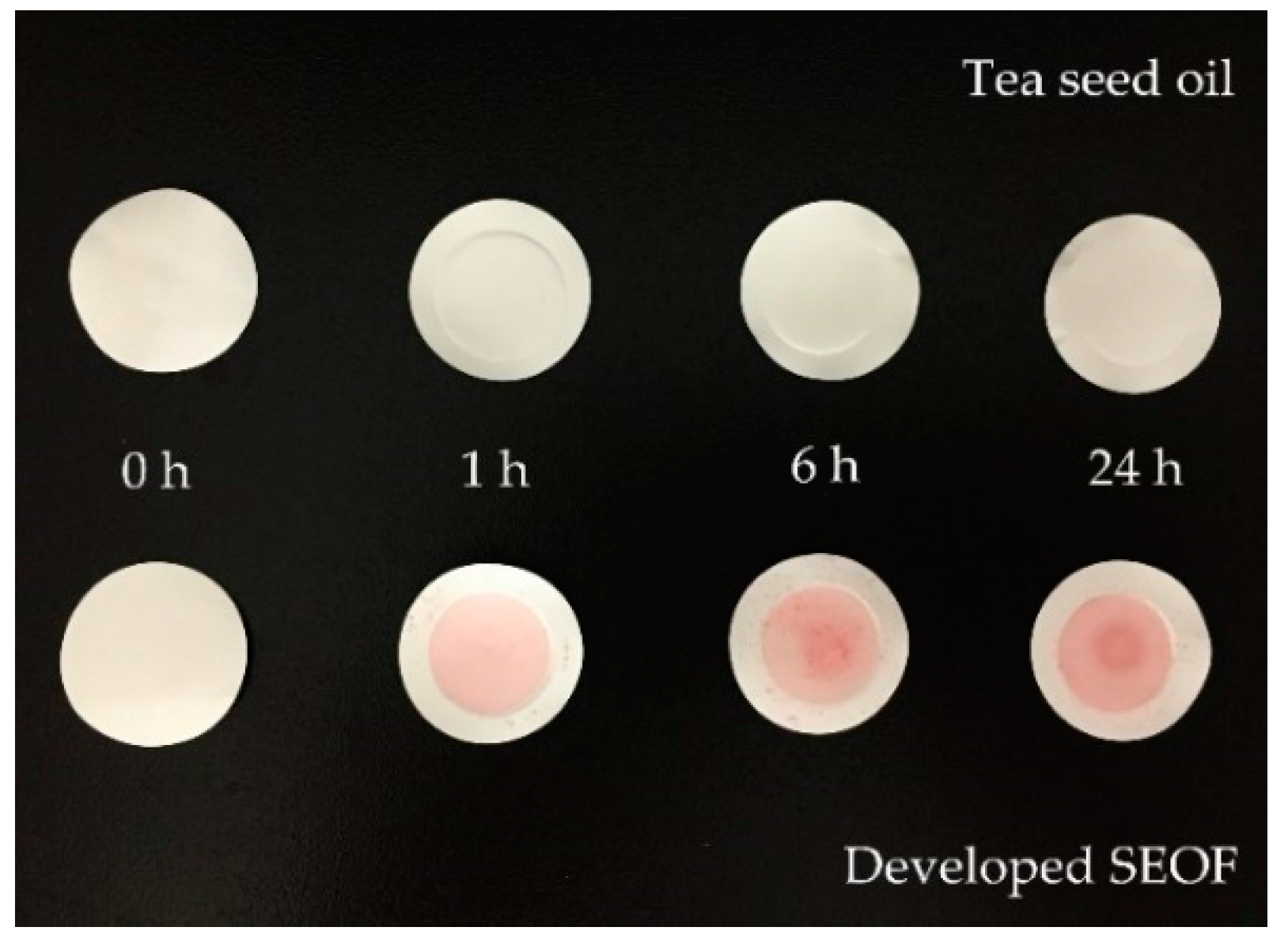
2.4. In Vitro Study of Oil Adsorption
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Construction of Ternary Phase Diagram
4.3. Preparation of Self-Emulsifying Oil Formulations (SEOF)
4.4. Emulsion Droplet Size and Zeta Potential Measurement
4.5. Chemical Composition Analysis
4.6. InVitro Study of Oil Adsorption
4.7. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kumar, A.; Sharma, S.; Kamble, R. Self emulsifying drug delivery system (SEDDS): Future aspects. Int. J. Pharm. Pharm. Sci. 2010, 2, 7–13. [Google Scholar]
- Shrivastava, S.; Yadav, S.K.; Verma, S. Applications of self emulsifying drug delivery systems in novel drug delivery- a review. Afr. J. Basic Appl. Sci. 2014, 6, 6–14. [Google Scholar]
- Chu, J.; Cheng, Y.L.; Rao, A.V.; Nouraei, M.; Zarate-Muñoz, S.; Acosta, E.J. Lecithin-linker formulations for self-emulsifying delivery of nutraceuticals. Int. J. Pharm. 2014, 471, 92–102. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Guo-ying, Z.; Huai-yun, Z.; Jun-ang, L. Research progress on the health function of tea oil. J. Med. Plants Res. 2011, 5, 485–489. [Google Scholar]
- Su, M.H.; Shih, M.C.; Lin, K.H. Chemical composition of seed oils in native Taiwanese Camellia species. Food Chem. 2014, 156, 369–373. [Google Scholar] [CrossRef]
- Lv, G.P.; Aoli, M.; Zhou, B.; Zhao, J. Development of a rapid and simple non-derivatization method to determine constituents and antioxidative capacity of camellia oils by HPTLC. Food Nutr. Sci. 2013, 4, 204–210. [Google Scholar] [CrossRef]
- Kalepu, S.; Manthina, M.; Padavala, V. Oral lipid-based drug delivery systems—An overview. Acta Pharm. Sin. B 2013, 3, 361–372. [Google Scholar] [CrossRef]
- O’Driscoll, C.M. Lipid-based formulations for intestinal lymphatic delivery. Eur. J. Pharm. Sci. 2002, 15, 405–415. [Google Scholar] [CrossRef]
- Mahapatra, A.K.; Murthy, P.N.; Swadeep, B.; Swain, R.P. Self-emulsifying drug delivery systems (SEDDS): An update from formulation development to therapeutic strategies. Int. J. PharmTech Res. 2014, 6, 545–568. [Google Scholar]
- Gershanik, T.; Benita, S. Self-dispersing lipid formulations for improving oral absorption of lipophilic drugs. Eur. J. Pharm. Biopharm. 2000, 50, 179–188. [Google Scholar] [CrossRef]
- Eid, A.M.M.; Baie, S.H.; Arafat, O.M. The effect of surfactant blends on the production of a novel Swietenia macrophylla oil self-nanoemulsifying system. Int. J. Pharm. Pharm. Sci. 2012, 4, 481–486. [Google Scholar]
- Athas, J.C.; Jun, K.; McCafferty, C.; Owoseni, O.; John, V.T.; Raghavan, S.R. An effective dispersant for oil spills based on food-grade amphiphiles. Langmuir 2014, 30, 9285–9294. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.S.; Ansari, M.; Samaan, M.; Acosta, E.J. Linker-based lecithin microemulsions for transdermal delivery of lidocaine. Int. J. Pharm. 2008, 349, 130–143. [Google Scholar] [CrossRef] [PubMed]
- Acosta, E.J.; Nguyen, T.; Witthayapanyanon, A.; Harwell, J.H.; Sabatini, D.A. Linker-based bio- compatible microemulsions. Environ. Sci. Technol. 2005, 39, 1275–1282. [Google Scholar] [CrossRef] [PubMed]
- Al-Sabagh, A.M.; Emara, M.M.; El-Din, M.R.N.; Aly, W.R. Formation of water-in-diesel oil nano-emulsions using high energy method and studying some of their surface active properties. Egypt. J. Petrol. 2011, 20, 17–23. [Google Scholar] [CrossRef]
- Patel, R.B.; Patel, M.R.; Bhatt, K.K.; Patel, B.G. Formulation consideration and characterization of microemulsion drug delivery system for transnasal administration of carbamazepine. Bull. Fac. Pharm. Cairo Univ. 2013, 51, 243–253. [Google Scholar] [CrossRef]
- Avachat, A.M.; Patel, V.G. Self nanoemulsifying drug delivery system of stabilized ellagic acid–phospholipid complex with improved dissolution and permeability. Saudi Pharm. J. 2015, 23, 276–289. [Google Scholar] [CrossRef] [PubMed]
- Kohli, K.; Chopra, S.; Dhar, D.; Arora, S.; Khar, R.K. Self-emulsifying drug delivery systems: An approach to enhance oral bioavailability. Drug Discov. Today 2010, 15, 958–965. [Google Scholar] [CrossRef] [PubMed]
- Zanchetta, B.; Chaud, M.V.; Santana, M.H.A. Self-emulsifying drug delivery systems (SEDDS) in pharmaceutical development. J. Adv. Chem. Eng. 2015, 5, 1–7. [Google Scholar]
- Jantratid, E.; Janssen, N.; Reppas, C.; Dressman, J.B. Dissolution media simulating conditions in the proximal human gastrointestinal tract: An update. Pharm. Res. 2008, 25, 1663–1676. [Google Scholar] [CrossRef] [PubMed]
- Cascone, S.; Lamberti, G.; Titomanlio, G. Designing in vitro systems to simulate the in vivo permeability of drugs. Transl. Med. UniSa 2014, 10, 18–21. [Google Scholar] [PubMed]
- Sample Availability: Not Available.


| Formula | Lecithin (%) | Surfactant Blends (%) | Droplet Size (nm) NS | PDI | ZP (mV) |
|---|---|---|---|---|---|
| A1 | 3 | 0.1 | 216.34 ± 16.38 | 0.271 ± 0.058 ab | −17.98 ± 1.63 a |
| A2 | 2 | 208.20 ± 7.03 | 0.277 ± 0.048 b | −7.78 ± 0.73 cd | |
| A3 | 4 | 220.42 ± 15.77 | 0.244 ± 0.029 ab | −6.16 ± 0.63 d | |
| B1 | 6 | 0.1 | 225.29 ± 29.39 | 0.264 ± 0.022 ab | −18.86 ± 2.57 a |
| B2 | 2 | 209.30 ± 8.49 | 0.239 ± 0.022 ab | −11.03 ± 1.77 bc | |
| B3 | 4 | 221.60 ± 20.00 | 0.195 ± 0.018 ab | −7.85 ± 0.71 cd | |
| C1 | 9 | 0.1 | 222.74 ± 15.13 | 0.259 ± 0.022 ab | −18.87 ± 1.45 a |
| C2 | 2 | 212.42 ± 8.34 | 0.204 ± 0.031 ab | −11.97 ± 1.30 bc | |
| C3 | 4 | 214.84 ± 9.15 | 0.190 ± 0.020 ab | −8.99 ± 0.73 bcd | |
| D1 | 12 | 0.1 | 224.91 ± 23.79 | 0.239 ± 0.031 ab | −20.39 ± 2.57 a |
| D2 | 2 | 206.40 ± 3.00 | 0.175 ± 0.040 a | −13.08 ± 1.44 b | |
| D3 | 4 | 208.51 ± 10.27 | 0.197 ± 0.010 ab | −10.39 ± 1.35 bc |
| Sample | Palmitic Acid (C16:0) | Stearic Acid (C18:0) | Oleic Acid (C18:1) | Linoleic Acid (C18:2) |
|---|---|---|---|---|
| Tea seed oil | 8.83 ± 0.13 | 1.37 ± 0.05 | 82.17 ± 0.10 | 7.64 ± 0.13 |
| SEOF | 9.01 ± 0.04 | 1.52 ± 0.07 | 80.64 ± 0.32 | 8.83 ± 0.29 |
| Time (Month) | Fatty Acid Content (g/100 g of SEOF) | |||
|---|---|---|---|---|
| Palmitic Acid (C16:0) | Stearic Acid (C18:0) | Oleic Acid (C18:1) | Linoleic Acid (C18:2) | |
| 0 | 8.41 ± 0.03 | 1.63 ± 0.01 | 61.58 ± 0.46 | 11.34 ± 0.15 |
| 1 | 9.63 ± 0.09 | 1.80 ± 0.04 | 61.52 ± 0.35 | 11.50 ± 0.05 |
| 2 | 9.26 ± 0.07 | 1.91 ± 0.04 | 61.05 ± 0.55 | 11.66 ± 0.10 |
| 3 | 9.76 ± 0.05 | 1.81 ± 0.06 | 61.75 ± 0.54 | 11.57 ± 0.22 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sramala, I.; Pinket, W.; Pongwan, P.; Jarussophon, S.; Kasemwong, K. Development of an in Vitro System to Simulate the Adsorption of Self-Emulsifying Tea (Camellia oleifera) Seed Oil. Molecules 2016, 21, 479. https://doi.org/10.3390/molecules21050479
Sramala I, Pinket W, Pongwan P, Jarussophon S, Kasemwong K. Development of an in Vitro System to Simulate the Adsorption of Self-Emulsifying Tea (Camellia oleifera) Seed Oil. Molecules. 2016; 21(5):479. https://doi.org/10.3390/molecules21050479
Chicago/Turabian StyleSramala, Issara, Wichchunee Pinket, Pawinee Pongwan, Suwatchai Jarussophon, and Kittiwut Kasemwong. 2016. "Development of an in Vitro System to Simulate the Adsorption of Self-Emulsifying Tea (Camellia oleifera) Seed Oil" Molecules 21, no. 5: 479. https://doi.org/10.3390/molecules21050479
APA StyleSramala, I., Pinket, W., Pongwan, P., Jarussophon, S., & Kasemwong, K. (2016). Development of an in Vitro System to Simulate the Adsorption of Self-Emulsifying Tea (Camellia oleifera) Seed Oil. Molecules, 21(5), 479. https://doi.org/10.3390/molecules21050479





