The Powdering Process with a Set of Ceramic Mills for Green Tea Promoted Catechin Extraction and the ROS Inhibition Effect
Abstract
:1. Introduction
2. Results
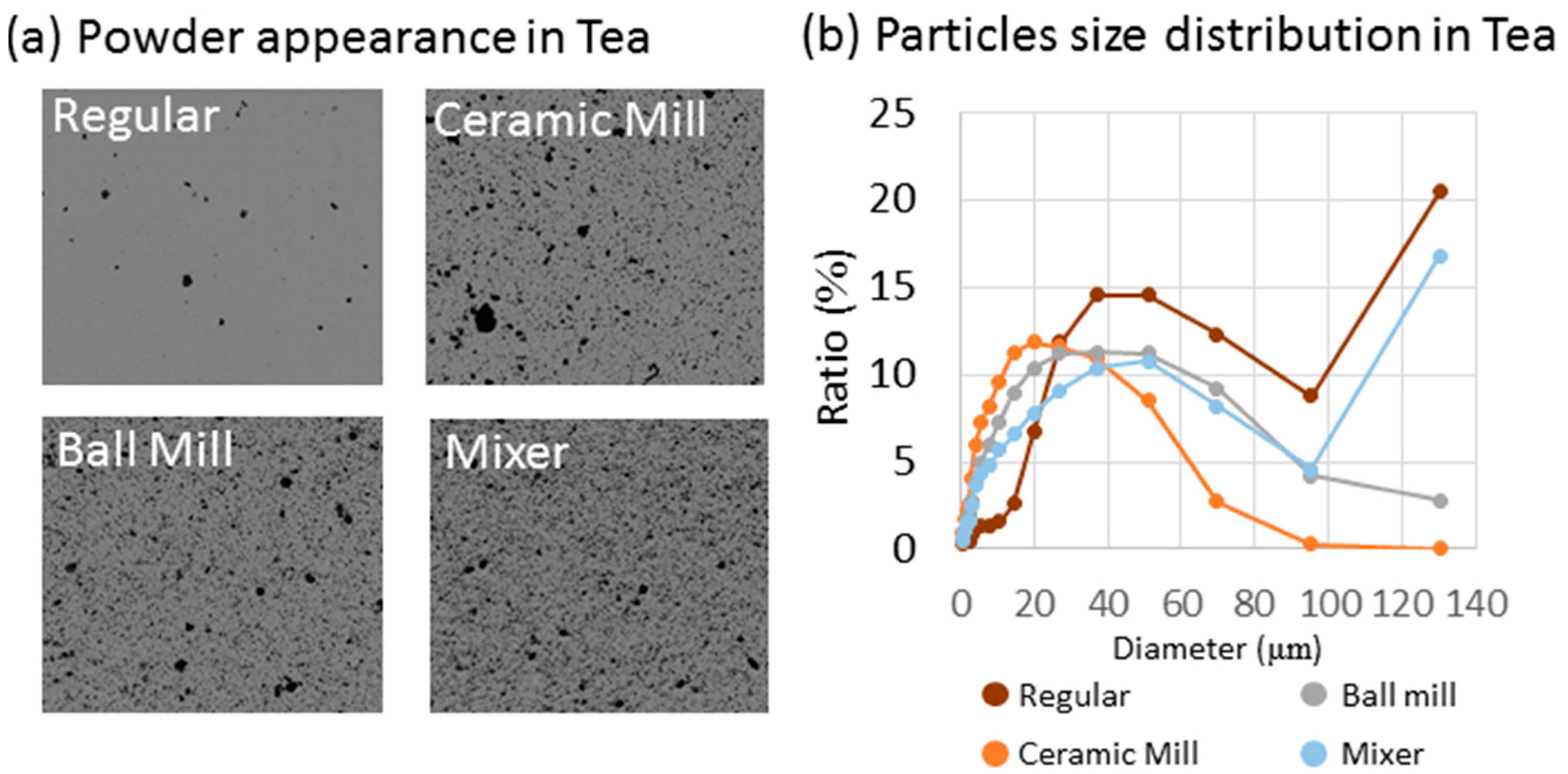
2.1. Particle Appearance in Different Powdering Methods
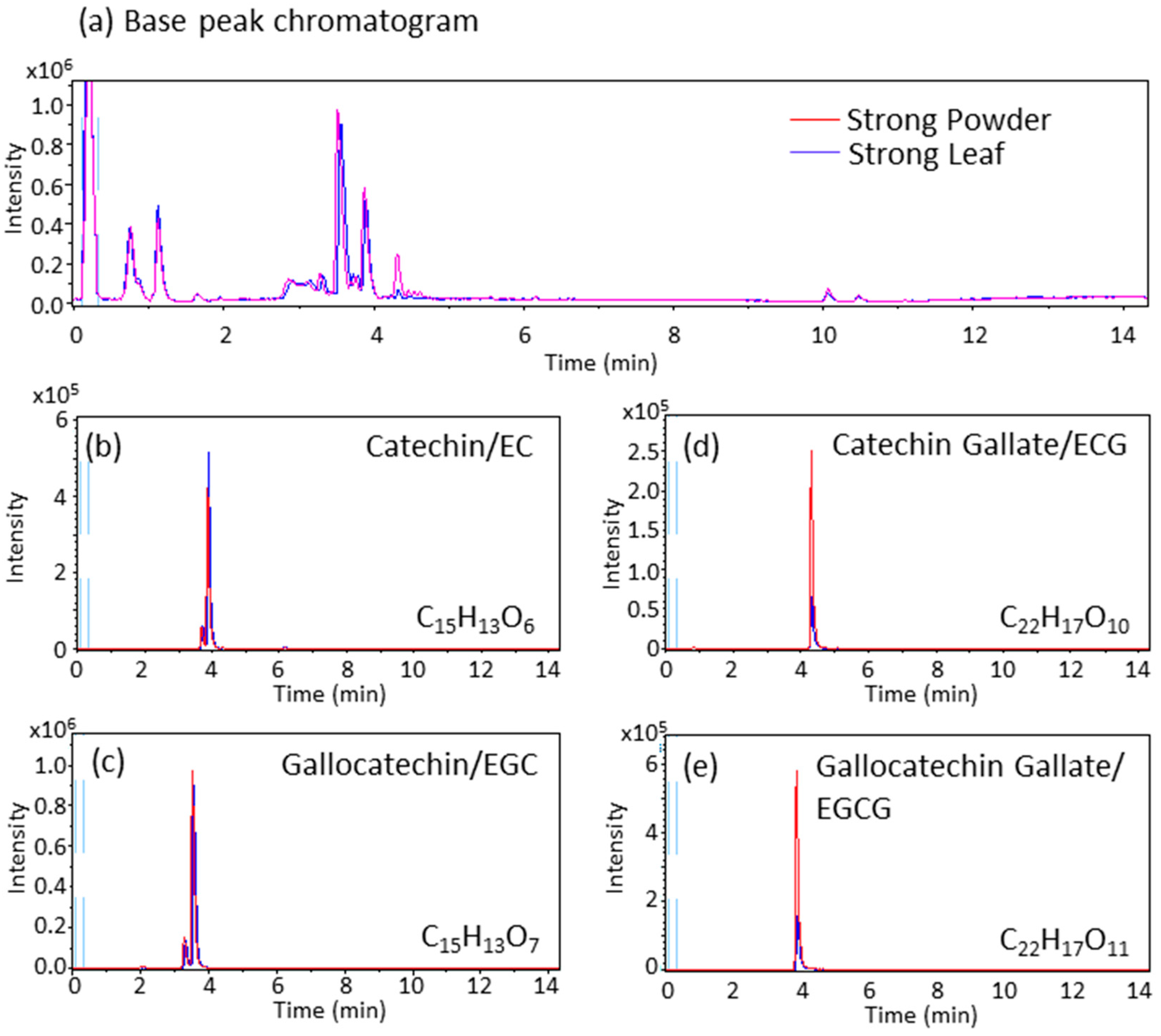
2.2. Catechin Contents of Powdered Tea and Leaf Tea
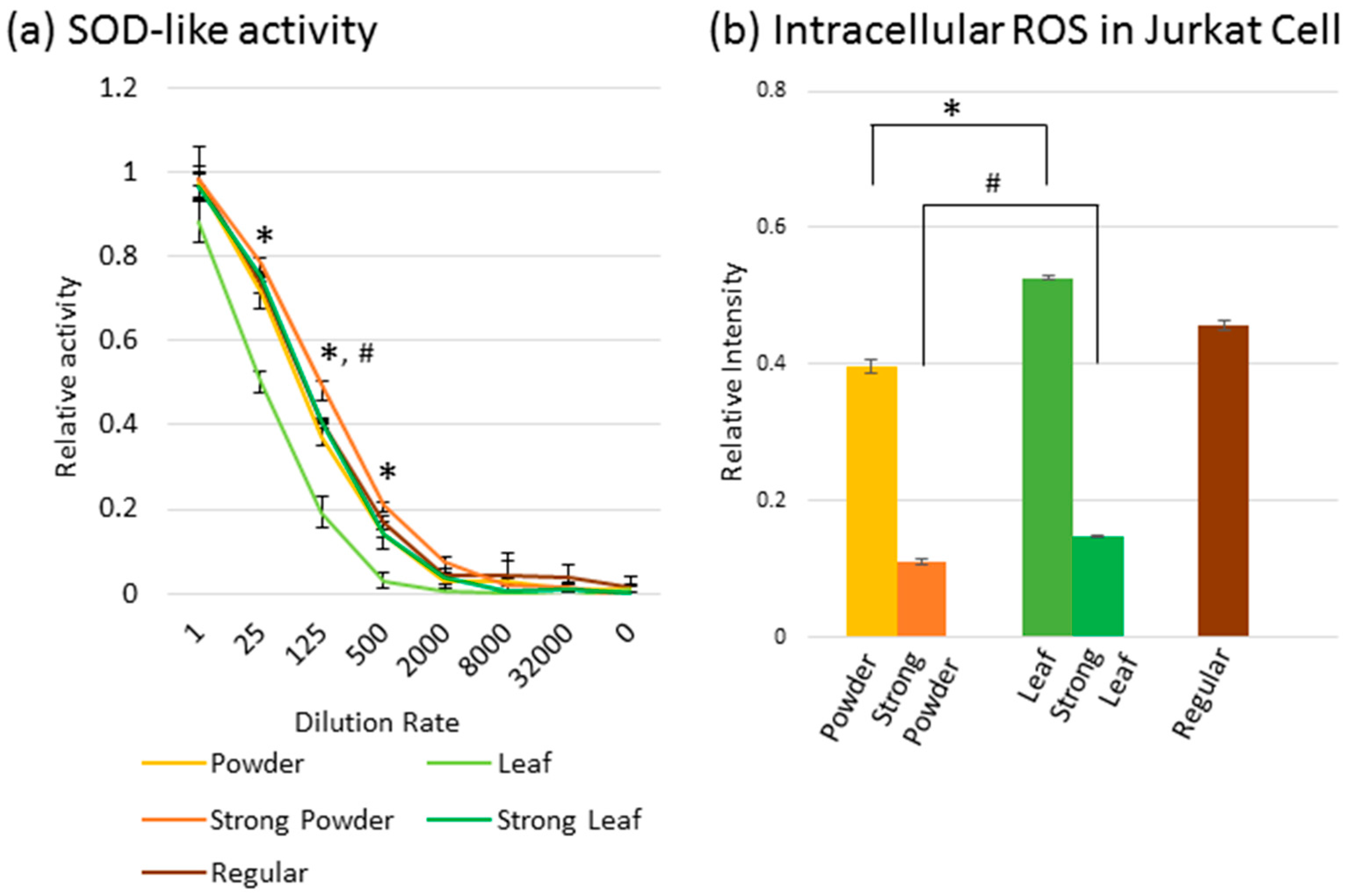
2.3. Effect of Powdered Tea and Leaf Tea on ROS
3. Discussion
3.1. Physical Properties of the Powdered Green Tea
3.2. Catechin and Caffeine Contents of the Powdered Green Tea
3.3. Effect on ROS Activity
4. Materials and Methods
4.1. Tea Preparation and Powdering Process
4.2. Chemicals
4.3. Observation of Tea Powders and Leaves with a SEM and a Multifocal Optical Microscope
4.4. Size Distribution in Tea Liquid
4.5. Catechin and Caffeine Analysis with HPLC System
4.6. Total Polyphenol Concentration
4.7. Chemical Components Analysis with LC-MS/MS System
4.8. Measurement of SOD-Like Activity
4.9. Inhibition of Intracellular ROS in a T Cell Line
4.10. Data Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AUC | Area under the concentration-time curve |
| BMI | Body Mass Index |
| Cmax | Maximum concentration |
| EC | Epicatechin |
| ECG | Epicatechin Gallate |
| EGC | Epigallocatechin |
| EGCG | Epigallocatechin Gallate |
| HPLC | High-performance Liquid Chromatography |
| LC-MS | Liquid Chromatography–Mass Spectrometry |
| LC-MS/MS | Liquid Chromatography–tandem Mass Spectrometry |
| ROS | Reactive Oxygen Species |
| SEM | Scanning Electron Microscope |
| SD | Standard Deviation |
| SOD | Superoxide Dismutase |
References
- Ui, A.; Kuriyama, S.; Kakizaki, M.; Sone, T.; Nakaya, N.; Ohmori-Matsuda, K.; Hozawa, A.; Nishino, Y.; Tsuji, I. Green tea consumption and the risk of liver cancer in Japan: The Ohsaki Cohort study. Cancer Causes Control 2009, 20, 1939–1945. [Google Scholar] [CrossRef] [PubMed]
- Inoue, M.; Sasazuki, S.; Wakai, K.; Suzuki, T.; Matsuo, K.; Shimazu, T.; Tsuji, I.; Tanaka, K.; Mizoue, T.; Nagata, C.; et al. Green tea consumption and gastric cancer in Japanese: A pooled analysis of six cohort studies. Gut 2009, 58, 1323–1332. [Google Scholar] [CrossRef] [PubMed]
- Maeda-Yamamoto, M.; Ema, K.; Shibuichi, I. In vitro and in vivo anti-allergic effects of ‘benifuuki’ green tea containing O-methylated catechin and ginger extract enhancement. Cytotechnology 2007, 55, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Ide, K.; Yamada, H.; Takuma, N.; Park, M.; Wakamiya, N.; Nakase, J.; Ukawa, Y.; Sagesaka, Y.M. Green tea consumption affects cognitive dysfunction in the elderly: A pilot study. Nutrients 2014, 6, 4032–4042. [Google Scholar] [CrossRef] [PubMed]
- Fukino, Y.; Ikeda, A.; Maruyama, K.; Aoki, N.; Okubo, T.; Iso, H. Randomized controlled trial for an effect of green tea-extract powder supplementation on glucose abnormalities. Eur J. Clin Nutr. 2008, 62, 953–960. [Google Scholar] [CrossRef] [PubMed]
- Thielecke, F.; Boschmann, M. The potential role of green tea catechins in the prevention of the metabolic syndrome—A review. Phytochemistry 2009, 70, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Kokubo, Y.; Iso, H.; Saito, I.; Yamagishi, K.; Yatsuya, H.; Ishihara, J.; Inoue, M.; Tsugane, S. The impact of green tea and coffee consumption on the reduced risk of stroke incidence in Japanese population: The Japan public health center-based study cohort. Stroke 2013, 44, 1369–1374. [Google Scholar] [CrossRef] [PubMed]
- Saito, E.; Inoue, M.; Sawada, N.; Shimazu, T.; Yamaji, T.; Iwasaki, M.; Sasazuki, S.; Noda, M.; Iso, H.; Tsugane, S.; et al. Association of green tea consumption with mortality due to all causes and major causes of death in a Japanese population: The Japan Public Health Center-based Prospective Study (JPHC Study). Ann. Epidemiol. 2015, 25, 512–518. [Google Scholar] [CrossRef] [PubMed]
- Sawamura, S. Particle Size and Taste of Matcha Produced before the Middle Ages. J. Cook. Sci. Japan 2011, 44, 231–237. [Google Scholar]
- Yamada, T. Kissa no Rekishi. In Cha Daihyakka; Kyokai, N.-G.B., Ed.; Nousan Gyoson Bunka Kyokai: Tokyo, Japan, 2008; Volume 1, pp. 67–81. [Google Scholar]
- Zhang, J. Seicha no Shurui, Gijutsu, Seichasha. In Sado to chanoyu: Nihoncha bunka shiron; Tankosha: Kyoto, Japan, 2004; pp. 153–200. [Google Scholar]
- Ikegaya, K.; Takayanagi, H.; Anan, T. Chemical Composition of Mat-cha. Tea Res. J. 1984, 60, 79–81. [Google Scholar] [CrossRef]
- Matsuzaki, T.; Hara, Y. Antioxidative activity of tea leaf catechins. Nippon Nogeikagaku Kaishi 1985, 59, 129–134. [Google Scholar] [CrossRef]
- Shimada, K. Determination of Tea-Leaf Saponins and Water-Soluble Pectin in a Green Tea Infusion. J. Home Econ. Japan 2003, 54, 957–962. [Google Scholar]
- Saijo, R.; Takeda, Y. HPLC Analysis of Catechins in Various Kinds of Green Teas Produced in Japan and Abroad. Nippon Shokuhin Kagaku Kogaku Kaishi 1999, 46, 138–147. [Google Scholar] [CrossRef]
- Goto, T.; Nagashima, H.; Yoshida, Y.; Kiso, M. Contents of Individual Tea Catechins and Caffeine in Japanese Green Tea. Tea Res. J. 1996, 83, 21–28. [Google Scholar] [CrossRef]
- Nanjo, F.; Mori, M.; Goto, K.; Hara, Y. Radical scavenging activity of tea catechins and their related compounds. Biosci. Biotechnol. Biochem. 1999, 63, 1621–1623. [Google Scholar] [CrossRef] [PubMed]
- Legeay, S.; Rodier, M.; Fillon, L.; Faure, S.; Clere, N. Epigallocatechin Gallate: A Review of Its Beneficial Properties to Prevent Metabolic Syndrome. Nutrients 2015, 7, 5443–5468. [Google Scholar] [CrossRef] [PubMed]
- Onishi, I.; Morimoto, T.; Togashi, Y. Grinding Method of Tencha and Physical Properties of Powdered Green Tea (Ceremony Tea). Tea Res. J. 1973, 39, 23–28. [Google Scholar] [CrossRef] [Green Version]
- Sawamura, S.; Haraguchi, Y.; Ikeda, H.; Sonoda, J. Properties and Shapes of Matcha with Various Milling Method. Nippon Shokuhin Kagaku Kogaku Kaishi. 2010, 57, 304–309. [Google Scholar] [CrossRef]
- Haraguchi, Y.; Imada, Y.; Sawamura, S. Production and Characterization of Fine Matcha for Processed Food. Nippon Shokuhin Kagaku Kogaku Kaishi. 2003, 50, 468–473. [Google Scholar] [CrossRef]
- Maeda-Yamamoto, M.; Ema, K.; Tokuda, Y.; Monobe, M.; Tachibana, H.; Sameshima, Y.; Kuriyama, S. Effect of green tea powder (Camellia sinensis L. cv. Benifuuki) particle size on O-methylated EGCG absorption in rats; The Kakegawa Study. Cytotechnology 2011, 63, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Shishikura, Y.; Khokhar, S. Factors affecting the levels of catechins and caffeine in tea beverage: Estimated daily intakes and antioxidant activity. J. Sci. Food Agric. 2005, 85, 2125–2133. [Google Scholar] [CrossRef]
- Westerterp-Plantenga, M.S.; Lejeune, M.P.; Kovacs, E.M. Body weight loss and weight maintenance in relation to habitual caffeine intake and green tea supplementation. Obes. Res. 2005, 13, 1195–1204. [Google Scholar] [CrossRef] [PubMed]
- Hursel, R.; Westerterp-Plantenga, M.S. Catechin- and caffeine-rich teas for control of body weight in humans. Am. J. Clin. Nutr. 2013, 98, 1682S–1693S. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, K.; Hirose, S.; Nagaoka, S.; Yanase, E. Interaction between Tea Polyphenols and Bile Acid Inhibits Micellar Cholesterol Solubility. J. Agric. Food Chem. 2016, 64, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Nishizawa, M.; Inoue, N.; Hosoya, T.; Yoshida, M.; Ukawa, Y.; Sagesaka, Y.M.; Doi, T.; Nakayama, T.; Kumazawa, S.; et al. Epigallocatechin gallate decreases the micellar solubility of cholesterol via specific interaction with phosphatidylcholine. J. Agric. Food Chem. 2014, 62, 2881–2890. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (poly)phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxid. Redox Signal. 2013, 18, 1818–1892. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Mateos, A.; Vauzour, D.; Krueger, C.G.; Shanmuganayagam, D.; Reed, J.; Calani, L.; Mena, P.; Del Rio, D.; Crozier, A. Bioavailability, bioactivity and impact on health of dietary flavonoids and related compounds: An update. Arch. Toxicol. 2014, 88, 1803–1853. [Google Scholar] [CrossRef] [PubMed]
- Stalmach, A.; Troufflard, S.; Serafini, M.; Crozier, A. Absorption, metabolism and excretion of Choladi green tea flavan-3-ols by humans. Mol. Nutr. Food Res. 2009, 53 Supplement, S44–S53. [Google Scholar] [CrossRef] [PubMed]
- Yoshino, K.; Hara, Y.; Sano, M.; Tomita, I. Antioxidative effects of black tea theaflavins and thearubigin on lipid peroxidation of rat liver homogenates induced by tert-butyl hydroperoxide. Biol. Pharm. Bull. 1994, 17, 146–149. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, F.; Ono, M.; Masuoka, C.; Ito, Y.; Sakata, Y.; Shimizu, K.; Nonaka, G.; Nishioka, I.; Nohara, T. Evaluation of the anti-oxidative effect (in vitro) of tea polyphenols. Biosci. Biotechnol. Biochem. 2003, 67, 396–401. [Google Scholar] [CrossRef] [PubMed]
- Terada, S.; Maeda, Y.; Masui, T.; Suzuki, Y.; Ina, K. Comparison of caffeine and catechin components in infusion of various tea (green, oolong and black tea) and tea drinks. Nippon Shokuhin Kagaku Kogaku Kaishi. 1987, 34, 20–27. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of Total Phenolics with Phosphomolybdic-Phosphotungstic Acid Reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Kamitani, I.; Fujioka, K.; Kamada, M.; Ikeda, K.; Manome, Y. Functional Investigation of Sweet Red Bean Pastes as Anti-oxidant Food: Polyphenol Contents, Superoxide Dismutase Activity, and Inhibitory Effects on Reactive Oxygen Species. Nippon Shokuhin Kagaku Kogaku Kaishi. 2015, 62, 349–353. [Google Scholar] [CrossRef]
- Human Metabolome Database. Available online: http://www.hmdb.ca/ (accessed on 1 February 2016).
- Bass, D.A.; Parce, J.W.; Dechatelet, L.R.; Szejda, P.; Seeds, M.C.; Thomas, M. Flow cytometric studies of oxidative product formation by neutrophils: A graded response to membrane stimulation. J. Immunol. 1983, 130, 1910–1917. [Google Scholar] [PubMed]
- Sample Availability: Not available.


| Leaf/Powder Contents (g/80 mL) | Total Polyphenol (μg CE/mL) | EC (μg/mL) | ECG (μg/mL) | EGC (μg/mL) | EGCG (μg/mL) | Caffeine (μg/mL) | |
|---|---|---|---|---|---|---|---|
| Regular | 2.00 | 1275.5 ± 13.91 a | 94.5 ± 2.45 a | 31.4 ± 3.55 a | 354.8 ± 6.9 a | 195.6 ± 3.77 a | 191.1 ± 1.85 a |
| Powder | 0.36 | 1121.1 ± 28.96 b | 56.3 ± 0.42 b | 35.7 ± 5.69 a | 191.3 ± 8.75 b | 210.6 ± 23.41 a | 121.5 ± 1.51 b |
| Leaf | 0.36 | 373.0 ± 7.20 c | 37.0 ± 0.91 c | 18.1 ± 1.40 a | 38.8 ± 3.14 c | 57.9 ± 1.96 b | 77.8 ± 0.64 c |
| Strong Powder | 0.96 | 3055.1 ± 26.35 d | 131.1 ± 1.27 d | 98.0 ± 21.22 b | 513.7 ± 1.74 d | 615.1 ± 88.36 c | 311.9 ± 4.32 d |
| Strong Leaf | 0.96 | 1890.9 ± 11.24 e | 136.0 ± 0.28 e | 33.3 ± 7.67 a | 501.2 ± 5.84 d | 200.1 ± 6.94 a | 244.0 ± 1.88 e |
| Retention Time (min) | ― | ― | 20.2 | 30.5 | 13.0 | 22.0 | 10.6 |
| Search Compound | Retention Time (min) | [M − H]− | Area | |
|---|---|---|---|---|
| Strong Powder | Strong Leaf | |||
| Catechin/EC | 3.9 | 289.07 | 2,684,378 ± 89,484.5 | 3,241,299 ± 192,287.7 * |
| Gallocatechin/EGC | 3.5 | 305.07 | 6,418,883 ± 149,286.8 * | 6,041,930 ± 16,410.1 |
| Catechin gallate/ECG | 4.3 | 441.08 | 1,364,591 ± 16,486.5 * | 358,874 ± 19,209.7 |
| Gallocatechin gallate/EGCG | 3.9 | 457.08 | 3,590,762 ± 128,970.1 * | 1,043,276 ± 67,495.9 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fujioka, K.; Iwamoto, T.; Shima, H.; Tomaru, K.; Saito, H.; Ohtsuka, M.; Yoshidome, A.; Kawamura, Y.; Manome, Y. The Powdering Process with a Set of Ceramic Mills for Green Tea Promoted Catechin Extraction and the ROS Inhibition Effect. Molecules 2016, 21, 474. https://doi.org/10.3390/molecules21040474
Fujioka K, Iwamoto T, Shima H, Tomaru K, Saito H, Ohtsuka M, Yoshidome A, Kawamura Y, Manome Y. The Powdering Process with a Set of Ceramic Mills for Green Tea Promoted Catechin Extraction and the ROS Inhibition Effect. Molecules. 2016; 21(4):474. https://doi.org/10.3390/molecules21040474
Chicago/Turabian StyleFujioka, Kouki, Takeo Iwamoto, Hidekazu Shima, Keiko Tomaru, Hideki Saito, Masaki Ohtsuka, Akihiro Yoshidome, Yuri Kawamura, and Yoshinobu Manome. 2016. "The Powdering Process with a Set of Ceramic Mills for Green Tea Promoted Catechin Extraction and the ROS Inhibition Effect" Molecules 21, no. 4: 474. https://doi.org/10.3390/molecules21040474
APA StyleFujioka, K., Iwamoto, T., Shima, H., Tomaru, K., Saito, H., Ohtsuka, M., Yoshidome, A., Kawamura, Y., & Manome, Y. (2016). The Powdering Process with a Set of Ceramic Mills for Green Tea Promoted Catechin Extraction and the ROS Inhibition Effect. Molecules, 21(4), 474. https://doi.org/10.3390/molecules21040474







