Effect of Genotype and Environment on Salvia miltiorrhiza Roots Using LC/MS-Based Metabolomics
Abstract
:1. Introduction
2. Results and Discussion
2.1. Identification of Metabolites
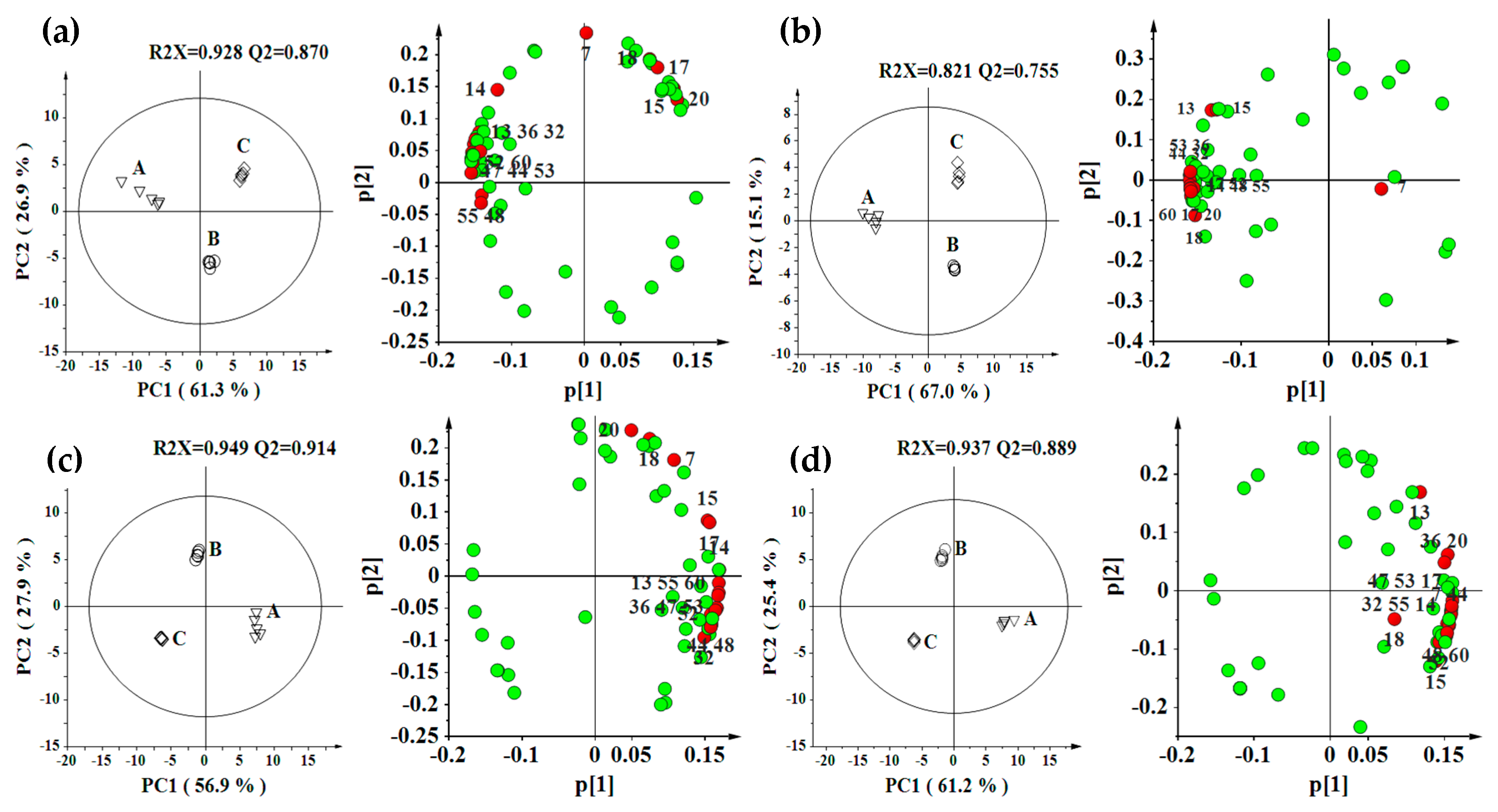
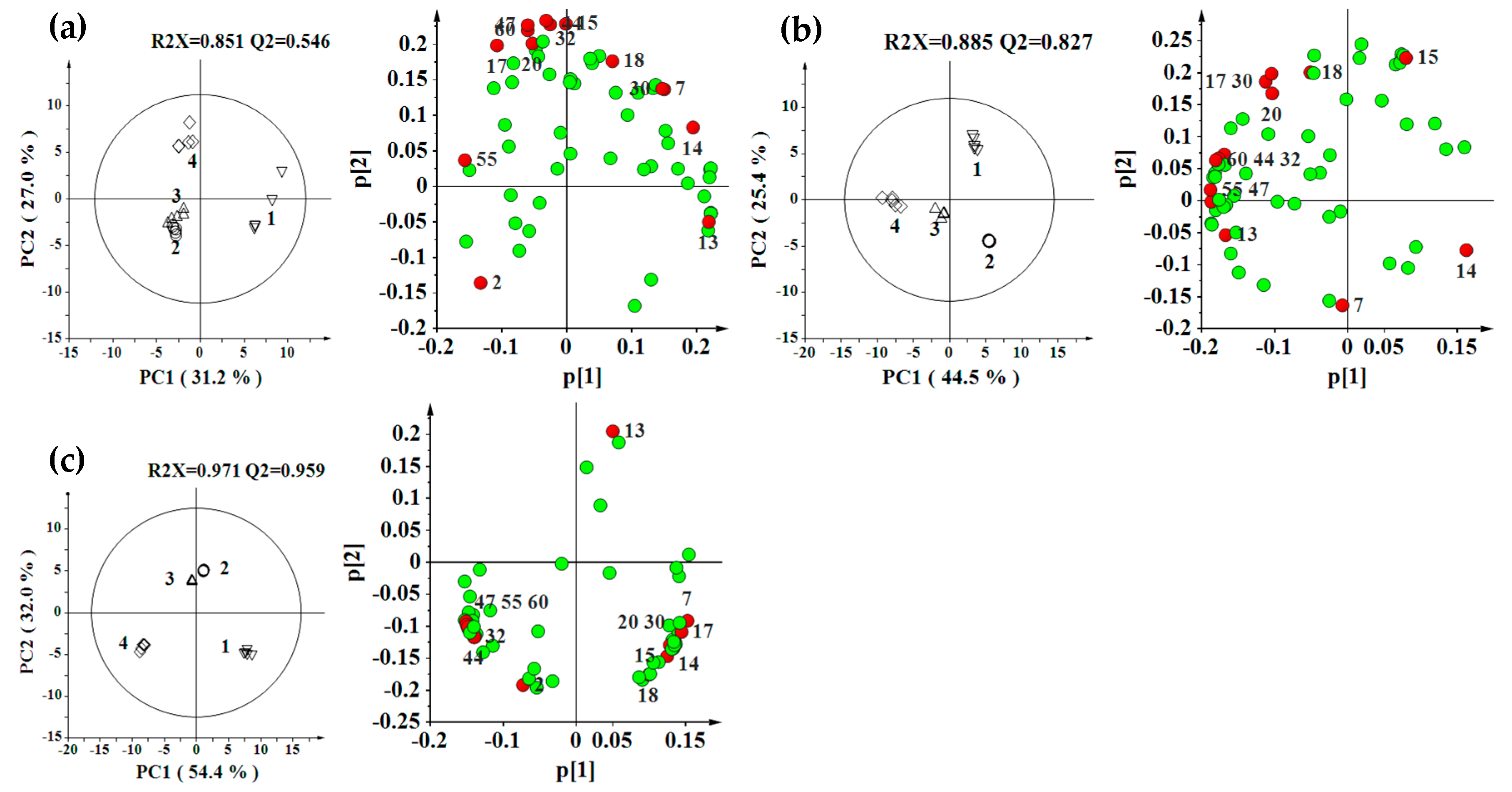
2.2. Effect of Environment, Multivariate Analysis
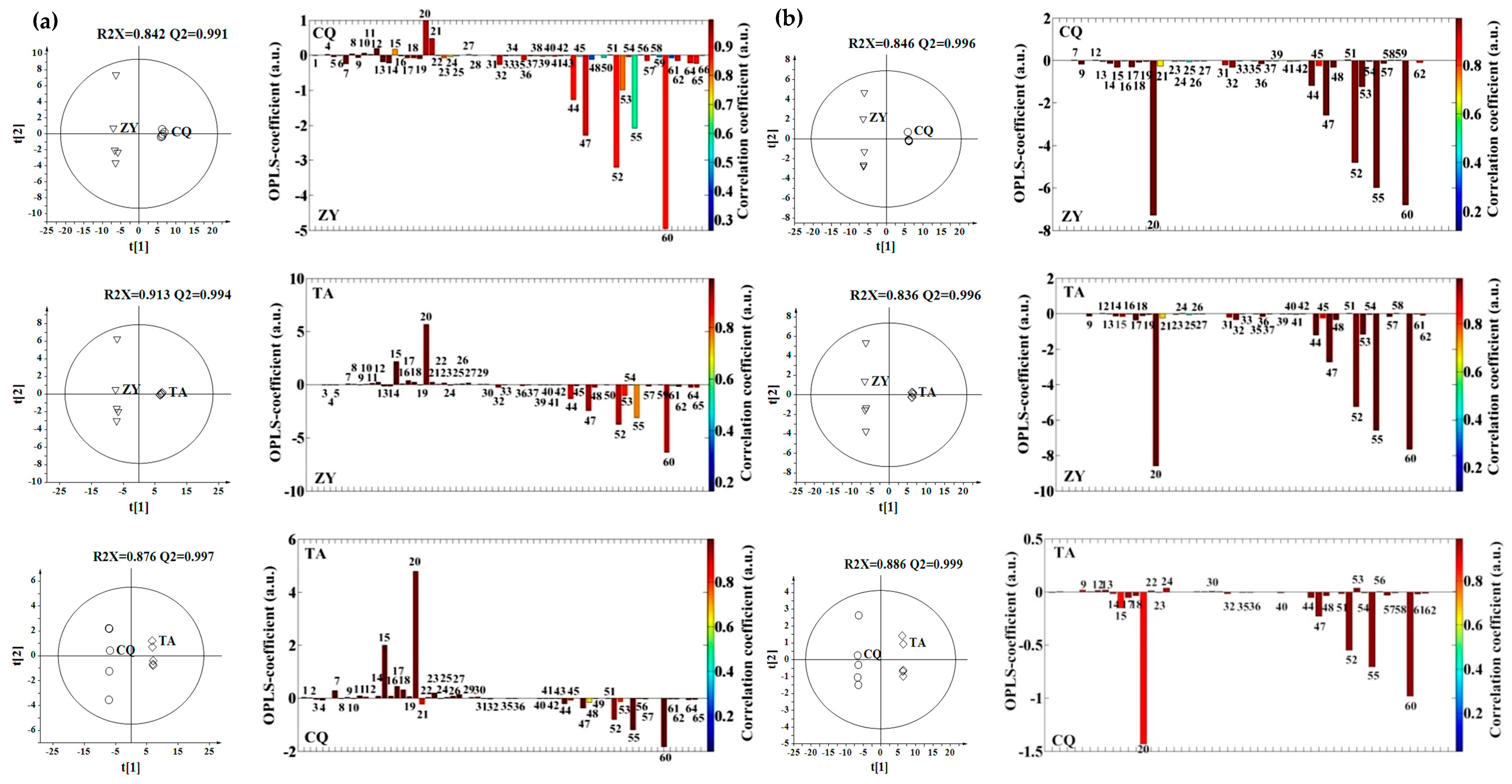
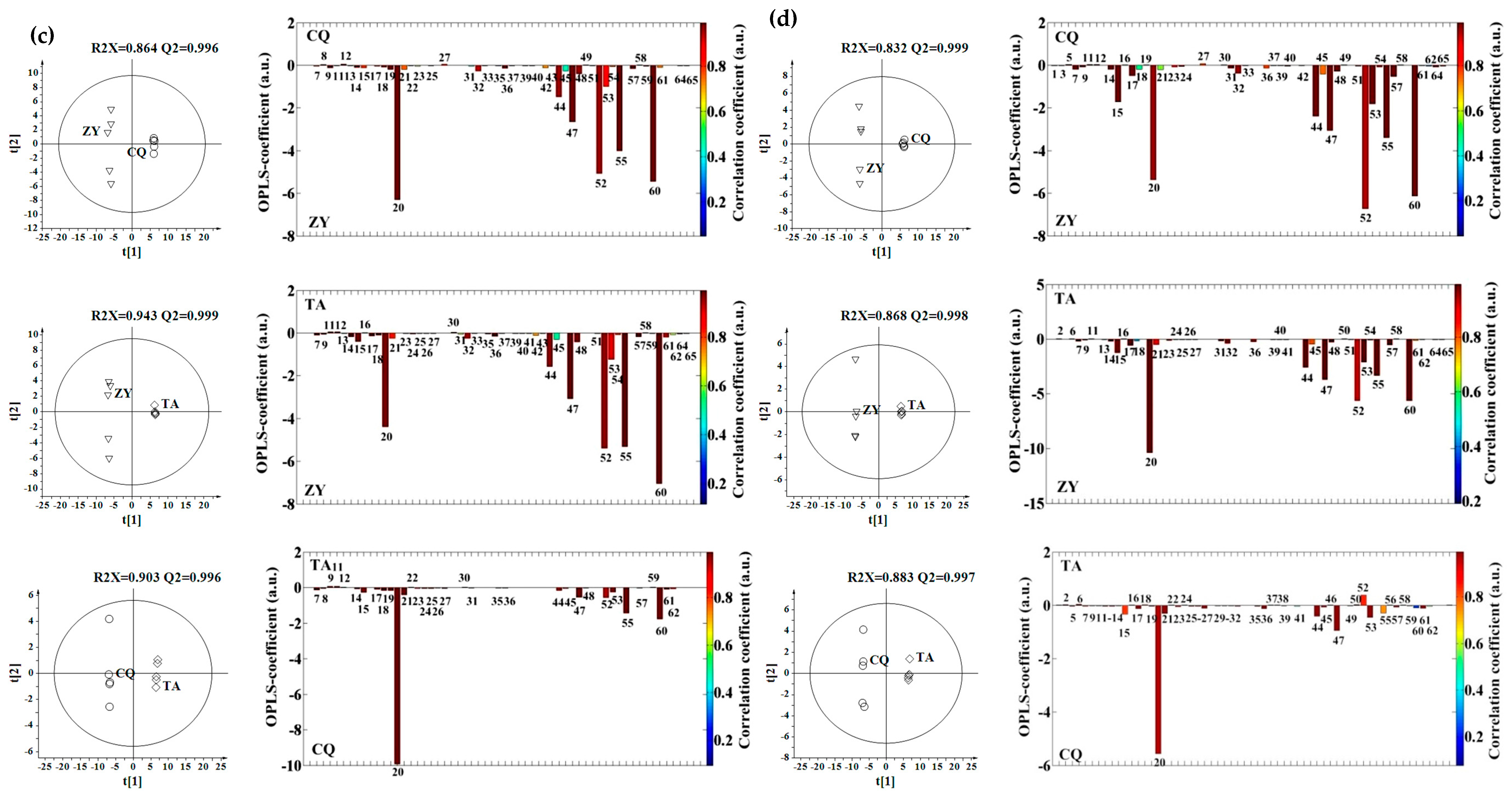
2.3. Effect of Genotype, Multivariate Analysis
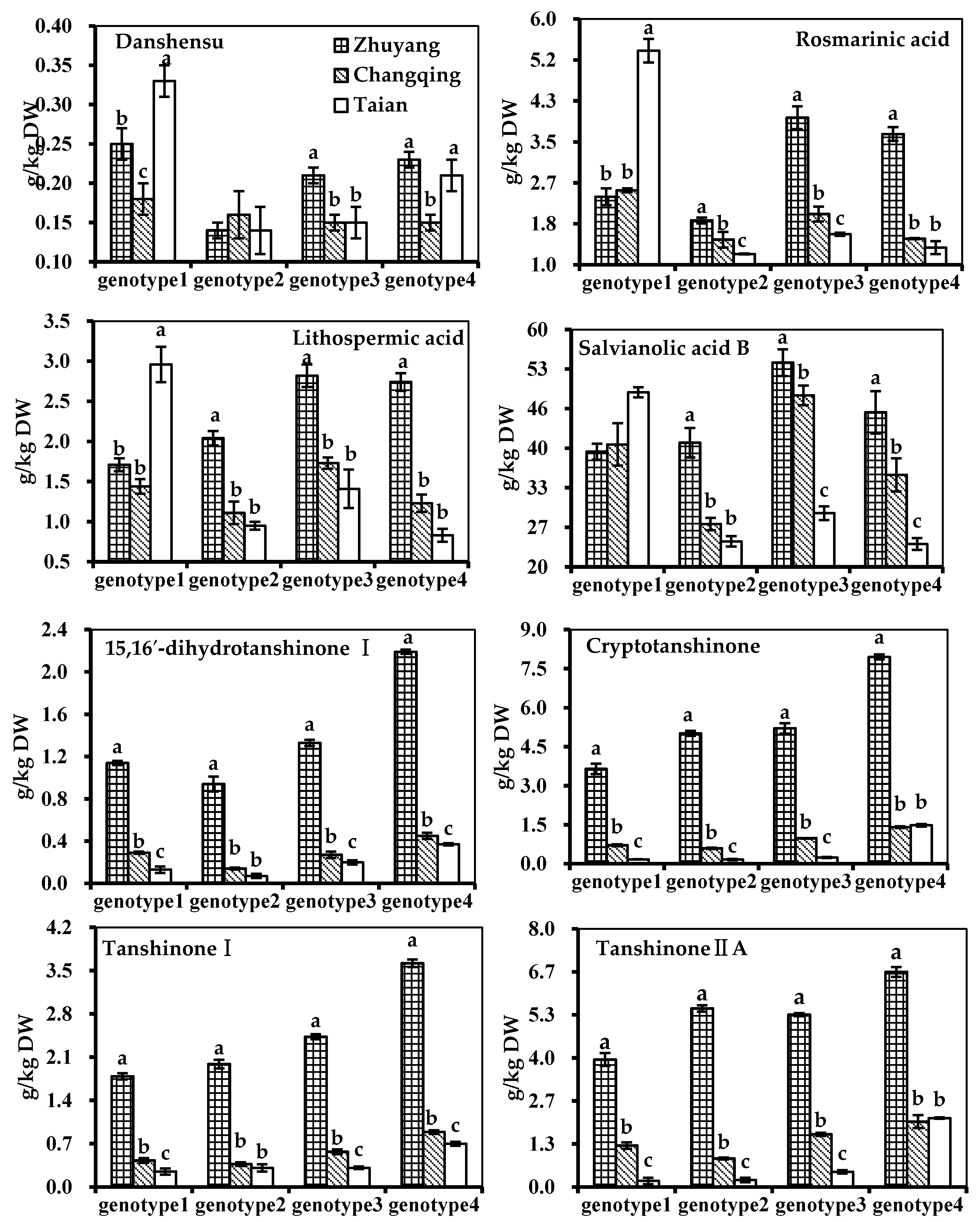
2.4. Effect of Genotype-by-Environment Interaction
3. Experimental Section
3.1. Field Experiment Design
3.2. Extraction and Analysis of Secondary Metabolites
3.3. Multivariate Data Analysis
3.4. Analysis of Enzyme Activities
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Shu, T.; Pang, M.; Rong, L.; Zhou, W.; Wang, J.; Liu, C.; Wang, X. Effects of Salvia miltiorrhiza on neural differentiation of induced pluripotent stem cells. J. Ethnopharmacol. 2014, 153, 233–241. [Google Scholar] [CrossRef] [PubMed]
- He, C.E.; Lu, L.L.; Jin, Y.; Wei, J.H.; Christie, P. Effects of nitrogen on root development and contents of bioactive compounds in Salvia miltiorrhiza Bunge. Crop Sci. 2013, 53, 2028–2039. [Google Scholar] [CrossRef]
- Zhang, H.; Yu, C.; Jia, J.Y.; Leung, S.W.; Siow, Y.L.; Man, R.Y.; Zhu, D.Y. Contents of four active components in different commercial crude drugs and preparations of danshen (Salvia miltiorrhiza). Acta Pharmacol. Sin. 2002, 23, 1163–1168. [Google Scholar] [PubMed]
- Dong, Y.; Mirris-Natschke, S.L.; Lee, K.H. Biosynthesis, total syntheses, and antitumor activity of tanshinones and their analogs as potential therapeutic agents. Nat. Prod. Rep. 2011, 28, 529–542. [Google Scholar] [CrossRef] [PubMed]
- Dat, N.T.; Jin, X.; Lee, J.H.; Lee, D.; Hong, Y.S.; Lee, K.; Kim, Y.H.; Lee, J.J. Abietane diterpenes from Salvia miltiorrhiza inhibit the activation of hypoxia-inducible factor-1. J. Nat. Prod. 2007, 70, 1093–1097. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Li, Y.; Abdolmaleky, H.M.; Li, L.; Zhou, J.R. Tanshinones inhibit the growth of breast cancer cells through epigenetic modification of Aurora A expression and function. PLoS ONE 2012, 7, e33656. [Google Scholar]
- Jiang, M.; Wang, C.; Zhang, Y.; Feng, Y.; Wang, Y.; Zhu, Y. Sparse partial-least-squares discriminant analysis for different geographical origins of Salvia miltiorrhiza by 1H-NMR-based metabolomics. Phytochem. Anal. 2014, 25, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Zhong, G.; Li, P.; Zeng, L.; Guan, J.; Li, D.; Li, S. Chemical characteristics of Salvia miltiorrhiza (Danshen) collected from different locations in China. J. Agric. Food Chem. 2009, 57, 6879–6887. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yan, Y.; Wu, Y.; Hua, W.; Chen, C.; Ge, Q.; Wang, Z. Pathway engineering for phenolic acid accumulations in Salvia miltiorrhiza by combinational genetic manipulation. Metab. Eng. 2014, 21, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Peng, H.; Shen, Y.; Zhao, R.; Huang, L. The profiling of bioactive ingredients of differently aged Salvia miltiorrhiza roots. Microsc. Res. Tech. 2013, 76, 947–954. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Wang, X.; Wang, D.; Zou, Z.; Liang, Z. Effect of drought stress on growth and accumulation of active constituents in Salvia miltiorrhiza Bunge. Ind. Crops Prod. 2011, 33, 84–88. [Google Scholar] [CrossRef]
- Xing, B.; Yang, D.; Guo, W.; Liang, Z.; Yan, X.; Zhu, Y.; Lu, Y. Ag+ as a more effective elicitor for production of tanshinones than phenolic acids in Salvia miltiorrhiza hairy roots. Molecules 2015, 20, 309–324. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, S.; Guo, L.; Huang, L. Effect of cadmium in the soil on growth, secondary metabolites and metal uptake in Salvia miltiorrhiza. Toxicol. Environ. Chem. 2013, 95, 1525–1538. [Google Scholar] [CrossRef]
- Duan, X.; Zhang, D.; Nie, L.; Zang, H. Rapid discrimination of geographical origin and evaluation of antioxidant activity of Salvia miltiorrhiza var. alba by Fourier transform near infrared spectroscopy. Spectrochim. Acta A 2014, 122, 751–757. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Wang, H.; Lu, X.; Xu, G.; Liu, B. Metabolic fingerprinting investigation of Artemisia annua L. in different stages of development by gas chromatography and gas chromatography-mass spectrometry. J. Chromatogr. A 2008, 1186, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Zhang, C.; Peng, L.; Liang, Z.; Yan, X.; Zhu, Y.; Liu, Y. Comparison of essential oil composition and phenolic acid content of selected Salvia species measured by GC-MS and HPLC methods. Ind. Crops Prod. 2015, 69, 329–334. [Google Scholar] [CrossRef]
- Lee, J.; Jung, Y.; Shin, J.; Kim, H.K.; Moon, B.C.; Ryu, D.H.; Hwang, G. Secondary metabolite profiling of Curcuma species grown at different locations using GC/TOF and UPLC/Q-TOF MS. Molecules 2014, 19, 9535–9551. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.; Kim, K.; Choi, B.Y.; Lee, D.; Shin, Y.; Bang, K.; Cha, S.; Lee, J.W.; Choi, H.; Jang, D.S.; et al. Metabolomic approach for age discrimination of Panax ginseng using UPLC-Q-Tof MS. J. Agric. Food Chem. 2011, 59, 10435–10441. [Google Scholar] [CrossRef] [PubMed]
- Boulila, A.; Sanaa, A.; Salem, I.B.; Rokbeni, N.; M’rabet, Y.; Hosni, K.; Fernandez, X. Antioxidant properties and phenolic variation in wide populations of Marrubium vulgare L. (Lamiaceae). Ind. Crops Prod. 2015, 76, 616–622. [Google Scholar] [CrossRef]
- Grayer, R.J.; Vieira, R.F.; Price, A.M.; Kite, G.C.; Simon, J.E.; Paton, A.J. Characterization of cultivars within species of Ocimum by exudate flavonoid profiles. Biochem. Syst. Ecol. 2004, 32, 901–913. [Google Scholar] [CrossRef]
- Song, Z.; Li, X. Expression profiles of rosmarinic acid biosynthesis genes in two Salvia miltiorrhiza lines with differing water-soluble phenolic contents. Ind. Crops Prod. 2015, 71, 24–30. [Google Scholar] [CrossRef]
- Baiano, A.; Terracone, C.; Viggiani, I.; Nobile, M.A.D. Effects of cultivars and location on quality, phenolic content and antioxidant activity of extra-virgin olive oils. J. Am. Oil Chem. Soc. 2013, 90, 103–111. [Google Scholar] [CrossRef]
- Fang, X.; Wang, J.; Zhang, S.; Zhao, Q.; Zheng, Z.; Song, Z. Simultaneous extraction of hydrosoluble phenolic acids and liposoluble tanshinones from Salviae miltiorrhizae radix by an optimized microwave-assisted extraction method. Sep. Purif. Technol. 2012, 86, 149–156. [Google Scholar] [CrossRef]
- Dai, H.; Xiao, C.; Liu, H.; Hao, F.; Tang, H. Combined NMR and LC-DAD-MS analysis reveals comprehensive metabonomic variations for three phenotypic cultivars of Salvia miltiorrhiza Bunge. J. Proteome Res. 2010, 9, 1565–1578. [Google Scholar] [CrossRef] [PubMed]
- Dai, H.; Xiao, C.; Liu, H.; Tang, H. Combined NMR and LC-MS analysis reveals the metabonomic changes in Salvia miltiorrhiza Bunge induced by water depletion. J. Proteome Res. 2010, 9, 1460–1475. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.; Liu, Y.; Yang, M.; Guo, H.; Guan, S.; Sun, J.; Guo, D. Development of the fingerprints for the quality of the roots of Salvia miltiorrhiza and its related preparations by HPLC-DAD and LC-MSn. J. Chromatogr. B 2007, 846, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, P.M.; Niemeyer, E.D. Effects of nitrogen fertilization on the phenolic composition and antioxidant properties of basil (Ocimum basilicum L.). J. Agric. Food Chem. 2008, 56, 8685–8691. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.D.; Yang, M.S.; Supanjani; Smith, D.L. Fertilizer effect on the yield and terpene components from the flowerheads of Chrysanthemum boreale M. (Compositae). Agron. Sustain. Dev. 2005, 25, 205–211. [Google Scholar] [CrossRef]
- Gao, W.; Sun, H.; Xiao, H.; Cui, G.; Hillwig, M.L.; Jackson, A.; Wang, X.; Shen, Y.; Zhao, N.; Zhang, L.; et al. Combining metabolomics and transcriptomics to characterize tanshinone biosynthesis in Salvia miltiorrhiza. BMC Genomics 2014, 15. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.Y.; Cui, G.H.; Huang, L.Q.; Qiu, D.Y. Effects of methyl jasmonat on accumulation and release of tanshinones in suspension cultures of Salvia miltiorrhiza hairy root. Zhongguo Zhong Yao Za Zhi 2007, 32, 300–302. [Google Scholar] [PubMed]
- Wu, Y.; Liu, C.; Kuang, J.; Ge, Q.; Zhang, Y.; Wang, Z. Overexpression of SmLEA enhance salt and drought tolerance in Escherichia coli and Salvia miltiorrhiza. Protoplasma 2014, 251, 1191–1199. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Zhao, J.; Pan, F.; Li, D.; Chen, H.; Wang, K. Changes in nitrogen and phosphorus limitation during succession in a karst region in southwest China. Plant Soil 2015, 391, 77–91. [Google Scholar] [CrossRef]
- Zong, C.; Song, Z.; Chen, H.; Liu, C.; Wang, J.; Guo, L.; Liu, T.; Pan, Y. Construction of the first genetic linkage map of Salvia miltiorrhiza Bge. using SSR, SRAP and ISSR markers. Acta Pharm. Sin. 2015, 50, 360–366. [Google Scholar]
- Morrison, T.A.; Kessler, J.R.; Hatfield, R.D.; Buxton, D.R. Activity of two lignin biosynthesis enzymes during development of a maize internode. J. Sci. Food Agric. 1994, 65, 133–139. [Google Scholar] [CrossRef]
- Kofalvi, S.A.; Nassuth, A. Influence of wheat streak mosaic virus infection on phenylpropanoid metabolism and the accumulation of phenolics and lignin in wheat. Physiol. Mol. Plant Pathol. 1995, 47, 365–377. [Google Scholar] [CrossRef]
- Knobloch, K.H.; Hahlbrock, K. Isoenzymes of p-coumarate: CoA ligase from cell suspension cultures of Glycine max. Eur. J. Biochem. 1975, 52, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
| Peak No. | Molecular Weight | λmax (nm) | RT (min) | Putative Annotation | Negative Ion Mode | Positive Ion Mode | Ref. | Annot. Level § | |||
|---|---|---|---|---|---|---|---|---|---|---|---|
| [M − H]− | Major Fragments | [M + Na]+ | [M + H]+ | Major Fragments | |||||||
| 1 | 198 | 282(sh)–326 | 3.9 | Danshensu | 197 | 395, 257, 179, 135 | 221 | 199 | 181, 163, 153 | abcd | 1 |
| 2 | 154 | 228–261–280 | 4.7 | Procatechuic acid | 153 | 213, 307,109 | - † | - | - | be | 2 |
| 4 | 138 | 232–281–313 | 6.6 | Procatechuic aldehyde | 137 | 275 | 161 | - | - | be | 2 |
| 5 | 168 | 219–294–330 | 7.6 | Vanillic acid | 167 | - | - | - | - | b | 2 |
| 6 | 180 | 240–281–326 | 8.2 | Caffeic acid | 179 | 359, 239, 161, 151, 135 | - | 181 | 163 | bce | 2 |
| 7 | 314 | 275–318 | 12.4 | Salvianolic acid F | 313 | 269, 159, 109 | - | - | - | bd | 2 |
| 10 | 194 | 290(sh)–323 | 18.5 | Ferulic acid | 193 | 175, 147 | - | 195 | 177, 163 | bce | 2 |
| 13 | 538 | 288 | 20.0 | Salvianolic acid I | 537 | 559, 493, 339, 295 | 561 | 539 | 521, 323 | bcd | 2 |
| 14 | 718 | 214–299(sh)–329 | 22.1 | Salvianolic acid E | 717 | 519 | 741 | - | - | b | 2 |
| 15 | 360 | 331(sh) | 23.1 | Rosmarinic acid | 359 | 719, 419, 341, 315, 197, 179, 161 | 383 | 361 | 181, 163 | abcde | 1 |
| 16 | 286 | 259–299–320–373 | 23.6 | Luteolin | 285 | 133 | - | - | - | ND | 2 |
| 17 | 538 | 255–308 | 24.3 | Lithospermic acid | 537 | 493, 449, 339, 313, 295 | 561 | 539 | 521, 493, 341 | abcd | 1 |
| 18 | 670 | 260–280 | 26.0 | Prolithospermic acid derivative | 669 | 551, 519 | 693 | 671 | 521, 433, 373 | cd | 2 |
| 20 | 718 | 289–310 | 27.2 | Salvianolic acid B | 717 | 339, 321, 295, 279, 277 | 741 | 719 | 521 | abce | 1 |
| 21 | 718 | 225(sh)–285–305 | 27.6 | Isosalvianolic acid B | 717 | 519 | 741 | - | - | bc | 2 |
| 23 | 718 | 225(sh)–285–305 | 30.0 | Salvianolic acid L | 717 | 519, 501, 339, 295, 197 | 741 | 719 | 521, 323, 295 | bcd | 2 |
| 24 | 494 | 225–285–310 | 31.1 | Salvianolic acid A | 493 | 295 | - | - | - | abe | 2 |
| 26 | 312 | 226–265–310 | 34.2 | Tanshindiol C | - | - | - | 313 | 373, 295, 267 | b | 2 |
| 29 | 344 | 260–340(sh) | 38.7 | Royleanone-4 | - | - | 367 | 345 | 327, 309 | b | 2 |
| 30 | 312 | 310–340(sh) | 39.6 | 7α-hydroxyallyl-royleanone | - | - | 353 | 331 | 313 | b | 2 |
| 32 | 310 | 275(sh)–254 | 43.5 | Tanshinone II B | - | - | - | 311 | 293, 283, 265, 251 | bcd | 2 |
| 33 | 310 | 225–265 | 44.0 | 1-ketoisocryptotanshinone | - | - | 333 | 311 | 293 | cd | 2 |
| 34 ‡ | 488 | 225–275–325 | 45.3 | Tormentic acid | 487 | 470, 469, 467, 423 | - | - | - | cd | 2 |
| 36 | 340 | 275(sh)–380 | 45.4 | Trijuganone C | - | - | 363 | 341 | 309, 281, 273 | bcd | 2 |
| 37 | 296 | 230–260–330 | 45.9 | Danshenxinkun A | 295 | 265 | - | - | - | c | 2 |
| 39 | 414 | 230(sh)–255–330 | 46.1 | β-sitosterol | - | - | 437 | 415 | - | cd | 2 |
| 40 | 308 | 235(sh)–380–395 | 46.4 | Tanshinaldehyde | - | - | 331 | 309 | - | cd | 2 |
| 42 | 310 | 240(sh)–270 | 47.1 | Przewa tanshinone A | - | - | - | 311 | 293, 275, 247 | b | 2 |
| 44 | 278 | 245(sh)–285–330 | 47.8 | 15,16′-dihydrotanshinone I | - | - | 301 | 279 | 261, 237, 233, 209 | abce | 1 |
| 46 | 314 | 250(sh)–380 | 48.7 | Neocryptotanshinone | 313 | - | - | 315 | 295, 279 | bc | 2 |
| 47 | 338 | 255(sh)–275 | 49.0 | Methyltanshinonate | - | - | - | 339 | 307, 297, 279, 278, 261 | b | 2 |
| 48 | 280 | 274(sh)–320 | 49.3 | Trijuganone B | - | - | 303 | 281 | 263, 235 | cd | 2 |
| 49 | 294 | 250(sh) | 49.7 | Trijuganone A | - | - | 317 | 295 | 280 | b | 2 |
| 50 | 300 | 255 | 50.1 | Miltipolone | 299 | - | - | - | - | cd | 2 |
| 52 | 296 | 265(sh)–355 | 51.7 | Cryptotanshinone | - | - | 319 | 297 | 615, 279, 251, 237 | abcde | 1 |
| 53 | 276 | 250(sh) | 53.6 | Tanshinone I | - | - | 299 | 277 | 575, 259, 249, 231 | abcde | 1 |
| 55 | 278 | 210–230–290(sh) | 56.0 | 1,2′-dihydrotanshinone I | - | - | 301 | 279 | 339, 261, 233 | cd | 2 |
| 59 | 280 | 245(sh)–275 | 58.1 | Dehydromiltirone | - | - | - | 281 | 461, 266 | b | 2 |
| 60 | 294 | 270(sh) | 58.5 | Tanshinone IIA | - | - | 317 | 295 | 611, 280, 262, 249 | abcde | 1 |
| 62 | 282 | 255(sh)–285 | 59.72 | Miltirone | - | - | 305 | 283 | 587, 268, 240 | bcd | 2 |
| Putative Annotation | Genotype 1 | Genotype 2 | Genotype 3 | Genotype 4 |
|---|---|---|---|---|
| (a) Zhuyang, g/kg DW | ||||
| Danshensu | 0.25 ± 0.02 a | 0.14 ± 0.01 c | 0.21 ± 0.01 b | 0.23 ± 0.01 b |
| Rosmarinic acid | 2.38 ± 0.17 c | 1.90 ± 0.06 d | 3.98 ± 0.23 a | 3.65 ± 0.14 b |
| Lithospermic acid | 1.71 ± 0.08 c | 2.04 ± 0.09 b | 2.82 ± 0.14 a | 2.74 ± 0.11 a |
| Salvianolic acid B | 39.23 ± 1.33 c | 40.73 ± 2.46 c | 54.08 ± 2.24 a | 45.78 ± 3.55 b |
| 15,16′-dihydrotanshinone I | 1.14 ± 0.02 c | 0.94 ± 0.07 d | 1.33 ± 0.03 b | 2.19 ± 0.02 a |
| Cryptotanshinone | 3.65 ± 0.20 c | 5.02 ± 0.10 b | 5.21 ± 0.20 b | 7.95 ± 0.10 a |
| Tanshinone I | 1.79 ± 0.05 d | 1.99 ± 0.07 c | 2.43 ± 0.04 b | 3.62 ± 0.06 a |
| Tanshinone IIA | 3.94 ± 0.20 c | 5.52 ± 0.10 b | 5.33 ± 0.05 b | 6.65 ± 0.15 a |
| (b) Changqing, g/kg DW | ||||
| Danshensu | 0.18 ± 0.02 | 0.16 ± 0.03 | 0.15 ± 0.01 | 0.15 ± 0.01 |
| Rosmarinic acid | 2.51 ± 0.04 a | 1.51 ± 0.16 c | 2.03 ± 0.15 b | 1.53 ± 0.01 c |
| Lithospermic acid | 1.44 ± 0.09 b | 1.11 ± 0.14 c | 1.73 ± 0.07 a | 1.23 ± 0.11 c |
| Salvianolic acid B | 40.42 ± 3.54 b | 27.16 ± 1.03 d | 48.61 ± 1.65 a | 35.36 ± 2.76 c |
| 15,16′-dihydrotanshinone I | 0.29 ± 0.01 b | 0.14 ± 0.01 c | 0.27 ± 0.03 b | 0.45 ± 0.03 a |
| Cryptotanshinone | 0.71 ± 0.04 c | 0.59 ± 0.03 d | 0.98 ± 0.01 b | 1.41 ± 0.03 a |
| Tanshinone I | 0.43 ± 0.04 c | 0.37 ± 0.03 c | 0.57 ± 0.04 b | 0.89 ± 0.03 a |
| Tanshinone IIA | 1.28 ± 0.10 c | 0.88 ± 0.03 d | 1.63 ± 0.05 b | 2.02 ± 0.20 a |
| (c) Taian, g/kg DW | ||||
| Danshensu | 0.33 ± 0.02 a | 0.14 ± 0.03 c | 0.15 ± 0.02 c | 0.21 ± 0.02 b |
| Rosmarinic acid | 5.34 ± 0.24 a | 1.22 ± 0.01 c | 1.62 ± 0.03 b | 1.35 ± 0.13 c |
| Lithospermic acid | 2.96 ± 0.22 a | 0.95 ± 0.05 a | 1.41 ± 0.24 b | 0.83 ± 0.08 c |
| Salvianolic acid B | 49.13 ± 0.85 a | 24.27 ± 0.88 a | 28.96 ± 1.16 c | 23.82 ± 0.97 c |
| 15,16′-dihydrotanshinone I | 0.13 ± 0.03 c | 0.07 ± 0.02 d | 0.20 ± 0.02 b | 0.37 ± 0.01 a |
| Cryptotanshinone | 0.17 ± 0.01 c | 0.16 ± 0.03 c | 0.24 ± 0.02 b | 1.48 ± 0.05 a |
| Tanshinone I | 0.25 ± 0.05 b | 0.31 ± 0.06 b | 0.31 ± 0.02 b | 0.70 ± 0.03 a |
| Tanshinone IIA | 0.19 ± 0.10 c | 0.22 ± 0.07 c | 0.47 ± 0.05 b | 2.13 ± 0.02 a |
| Paired Genotypes | ZY vs. CQ | ZY vs. TA | CQ vs. TA |
|---|---|---|---|
| Genotype 1 vs. Genotype 2 | location a | location | location |
| Genotype 1 vs. Genotype 3 | location | location | location |
| Genotype 1 vs. Genotype 4 | location | location | genotype b |
| Genotype 2 vs. Genotype 3 | location | location | genotype |
| Genotype 2 vs. Genotype 4 | location | location | genotype |
| Genotype 3 vs. Genotype 4 | location | location | location |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, Q.; Song, Z.; Fang, X.; Pan, Y.; Guo, L.; Liu, T.; Wang, J. Effect of Genotype and Environment on Salvia miltiorrhiza Roots Using LC/MS-Based Metabolomics. Molecules 2016, 21, 414. https://doi.org/10.3390/molecules21040414
Zhao Q, Song Z, Fang X, Pan Y, Guo L, Liu T, Wang J. Effect of Genotype and Environment on Salvia miltiorrhiza Roots Using LC/MS-Based Metabolomics. Molecules. 2016; 21(4):414. https://doi.org/10.3390/molecules21040414
Chicago/Turabian StyleZhao, Qi, Zhenqiao Song, Xinsheng Fang, Yuling Pan, Linlin Guo, Tian Liu, and Jianhua Wang. 2016. "Effect of Genotype and Environment on Salvia miltiorrhiza Roots Using LC/MS-Based Metabolomics" Molecules 21, no. 4: 414. https://doi.org/10.3390/molecules21040414
APA StyleZhao, Q., Song, Z., Fang, X., Pan, Y., Guo, L., Liu, T., & Wang, J. (2016). Effect of Genotype and Environment on Salvia miltiorrhiza Roots Using LC/MS-Based Metabolomics. Molecules, 21(4), 414. https://doi.org/10.3390/molecules21040414






