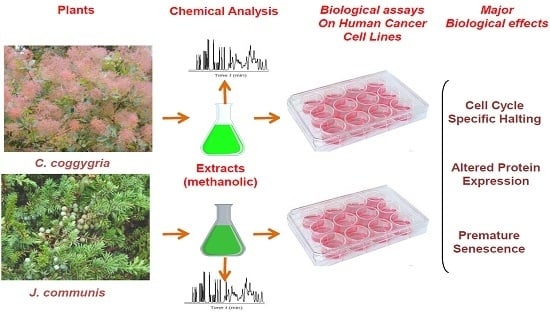
Polyphenolic Profile and Targeted Bioactivity of Methanolic Extracts from Mediterranean Ethnomedicinal Plants on Human Cancer Cell Lines
Abstract
:1. Introduction
2. Results and Discussion
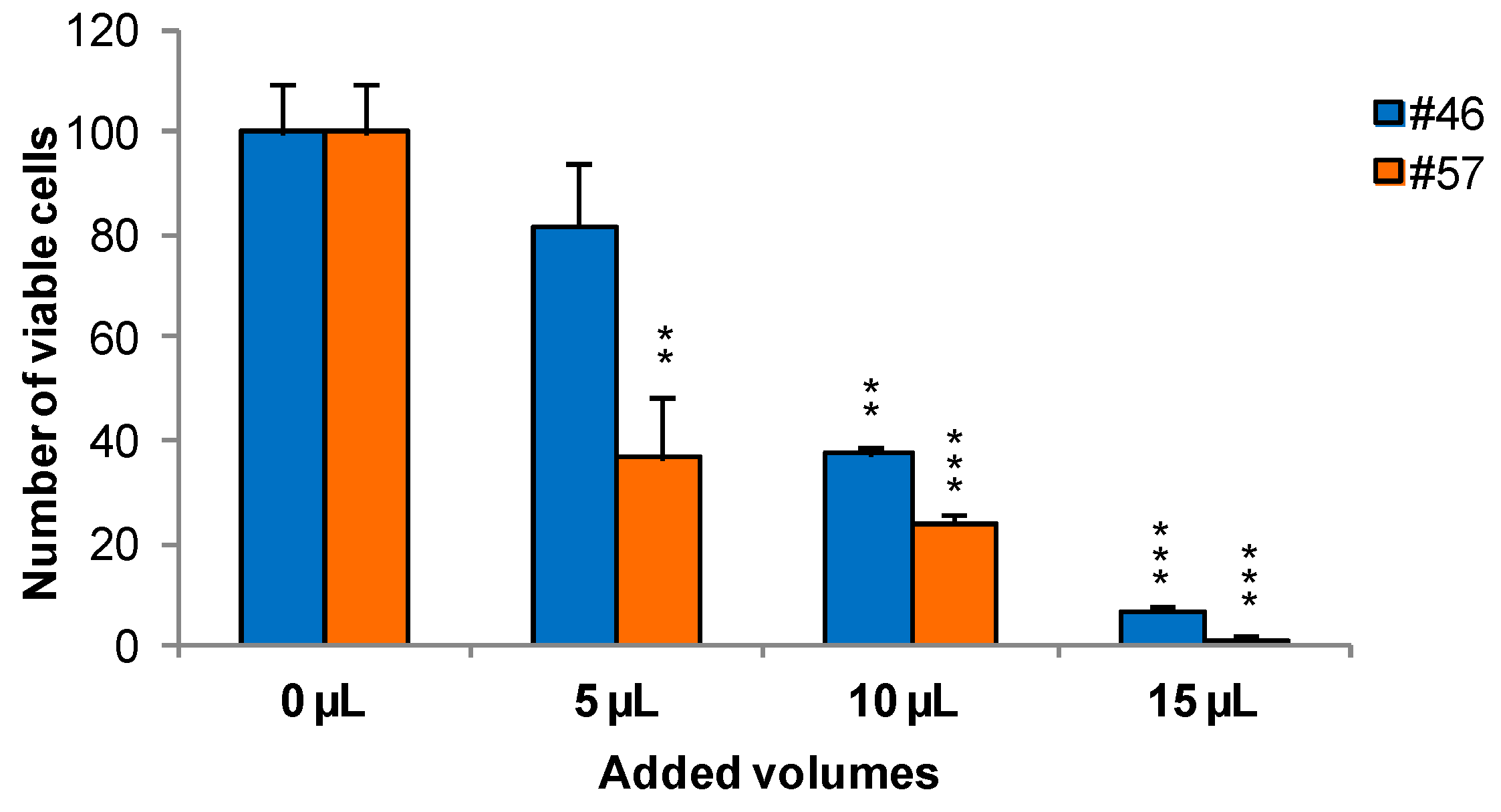
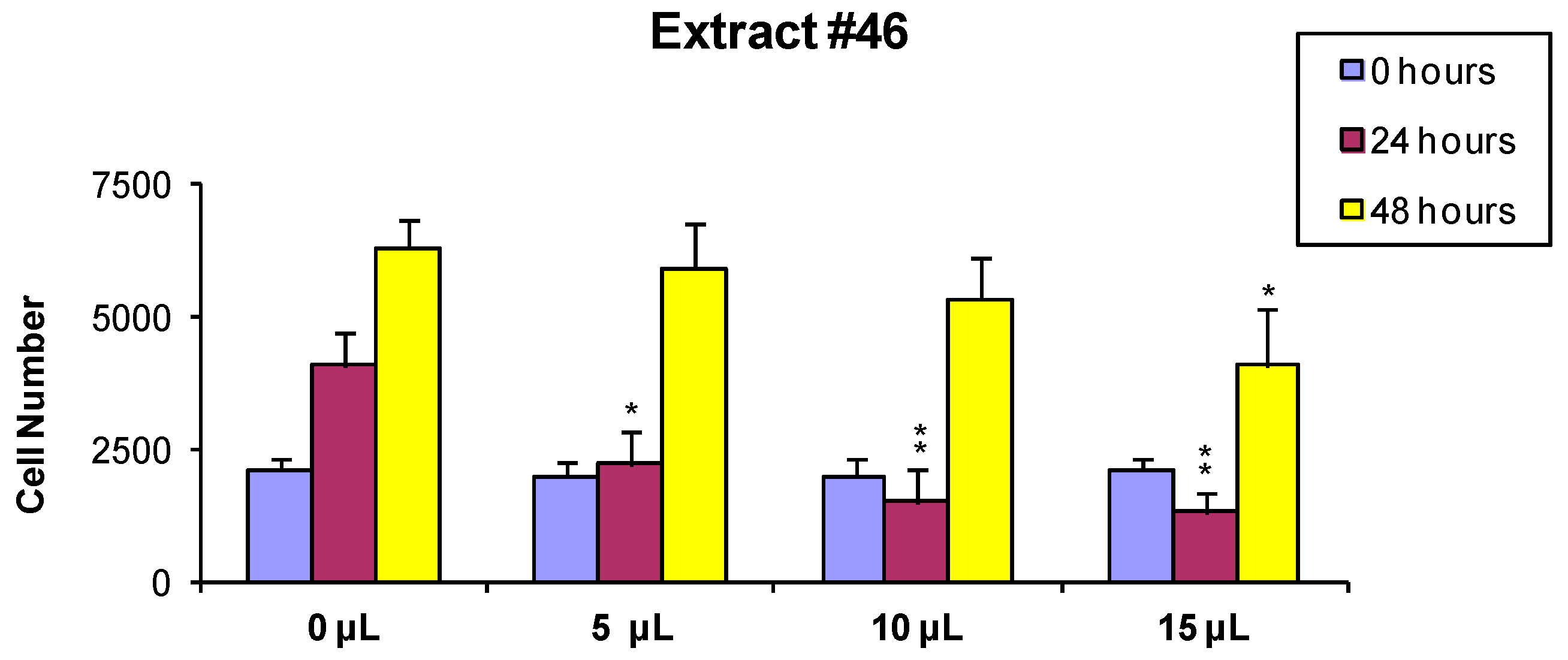
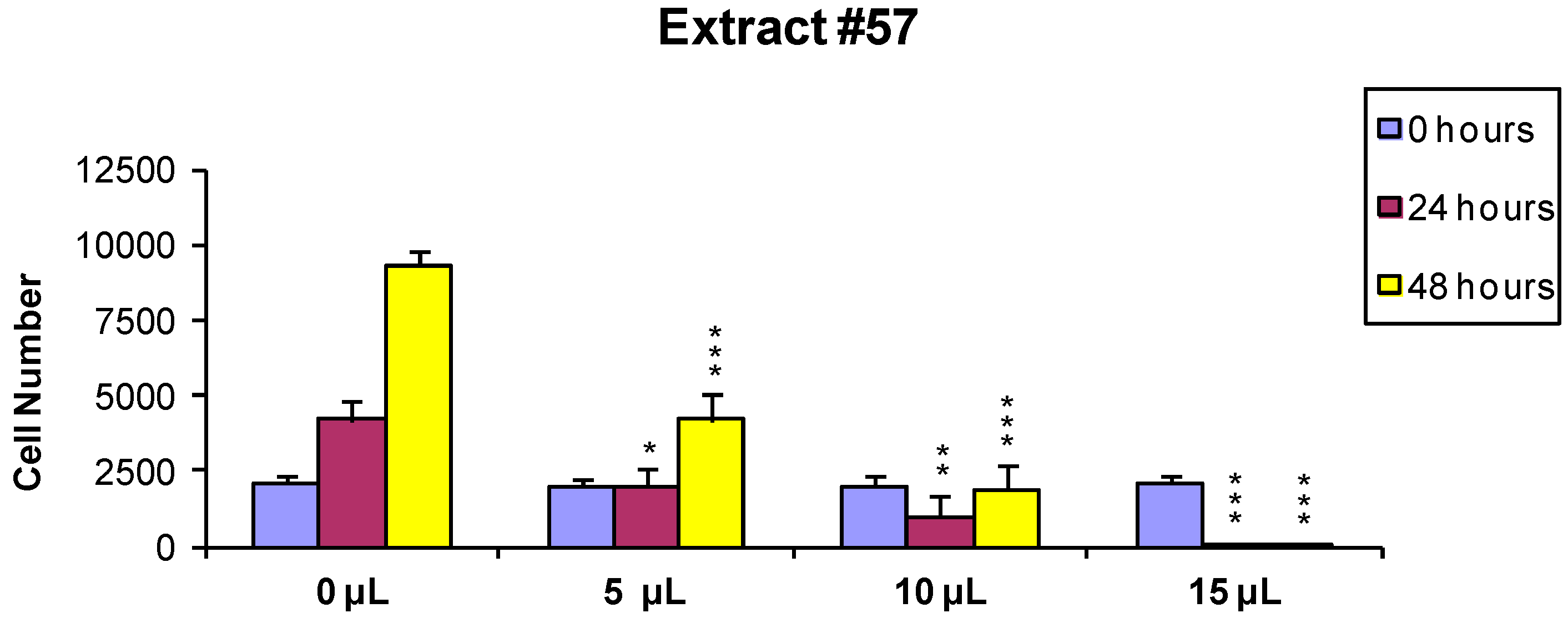
2.1. Cell Viability and Growth
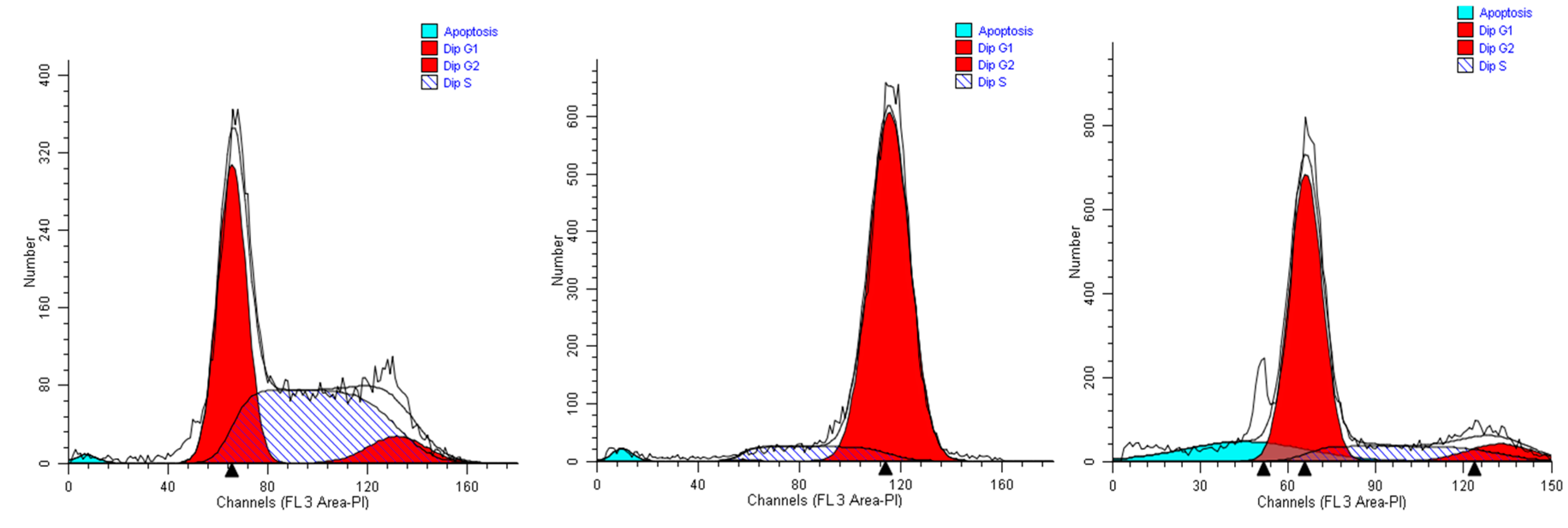
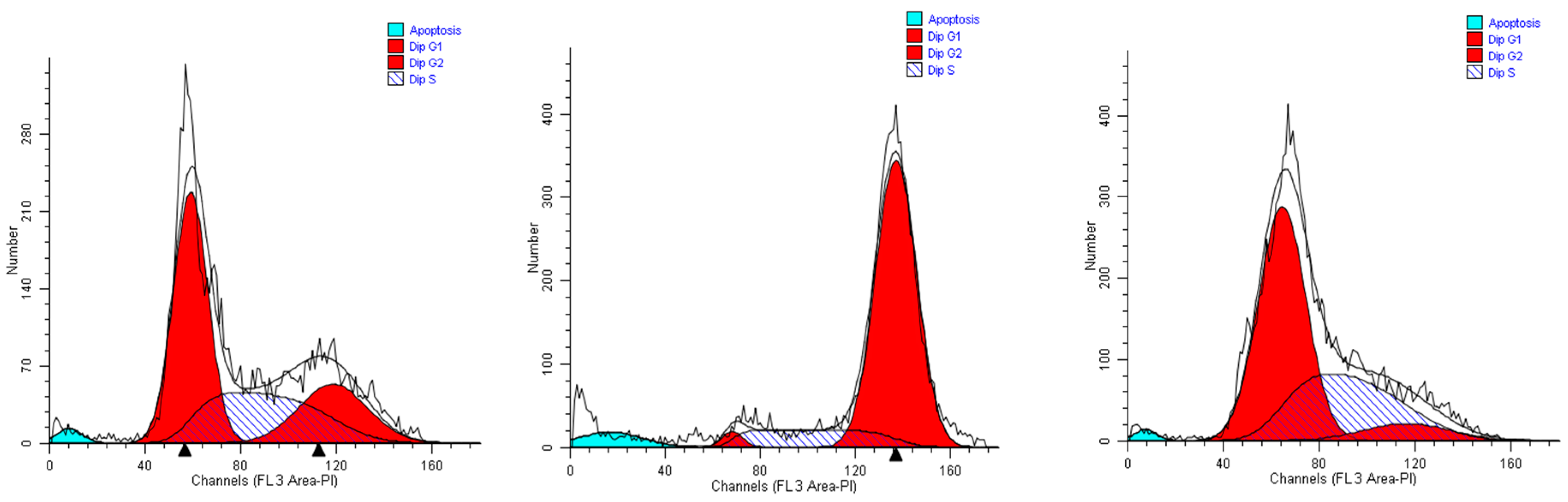
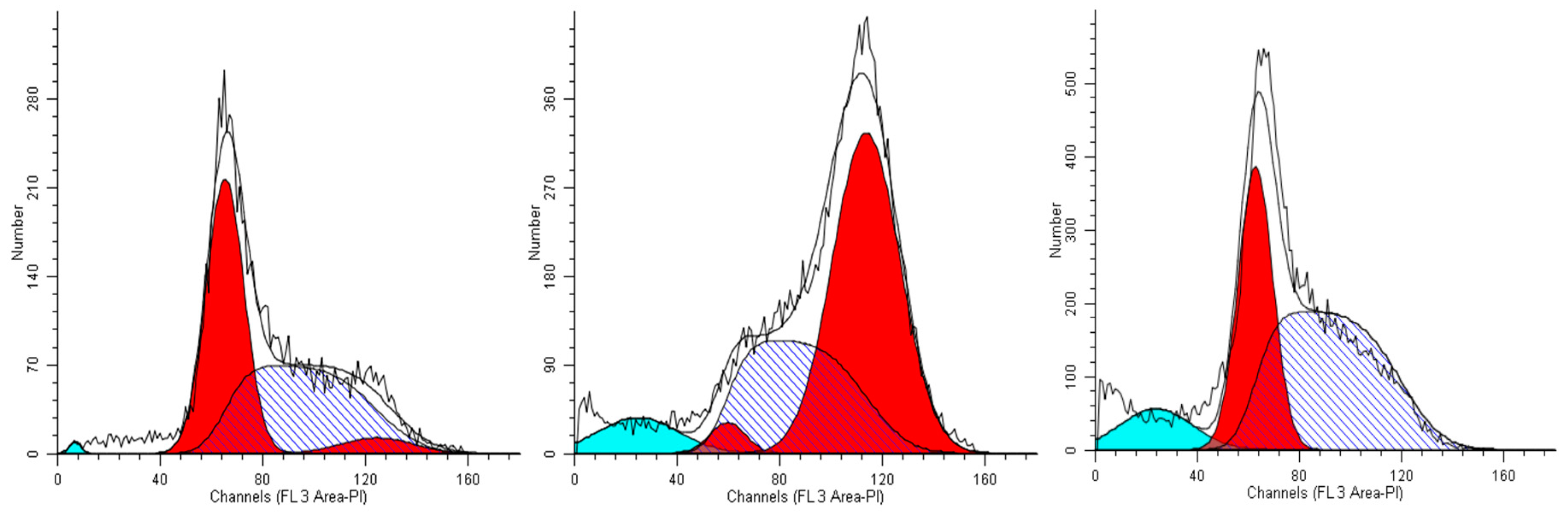
2.2. Effects of the Extracts on Cell Cycle
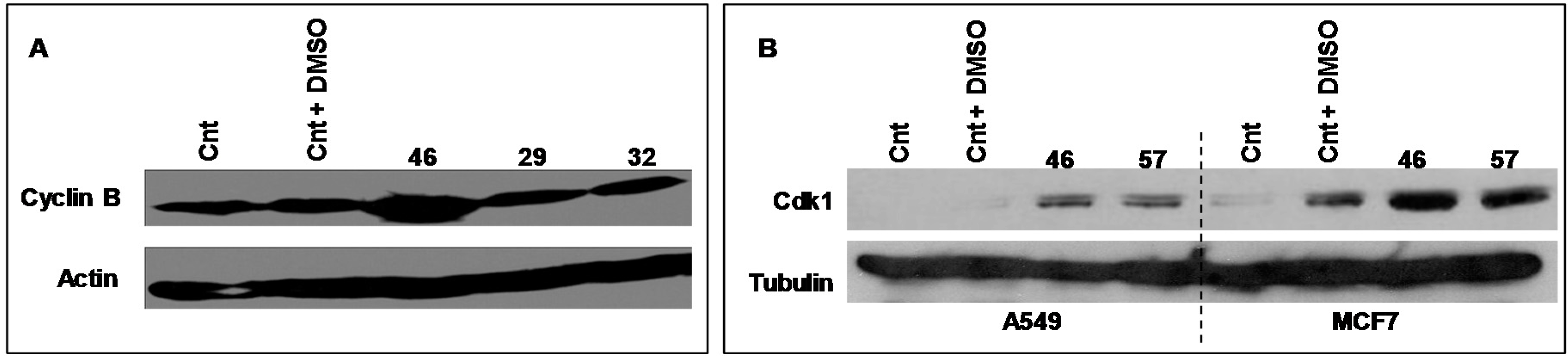
2.3. Protein Expression
2.3.1. Cyclin B, Cdk 1
2.3.2. p53
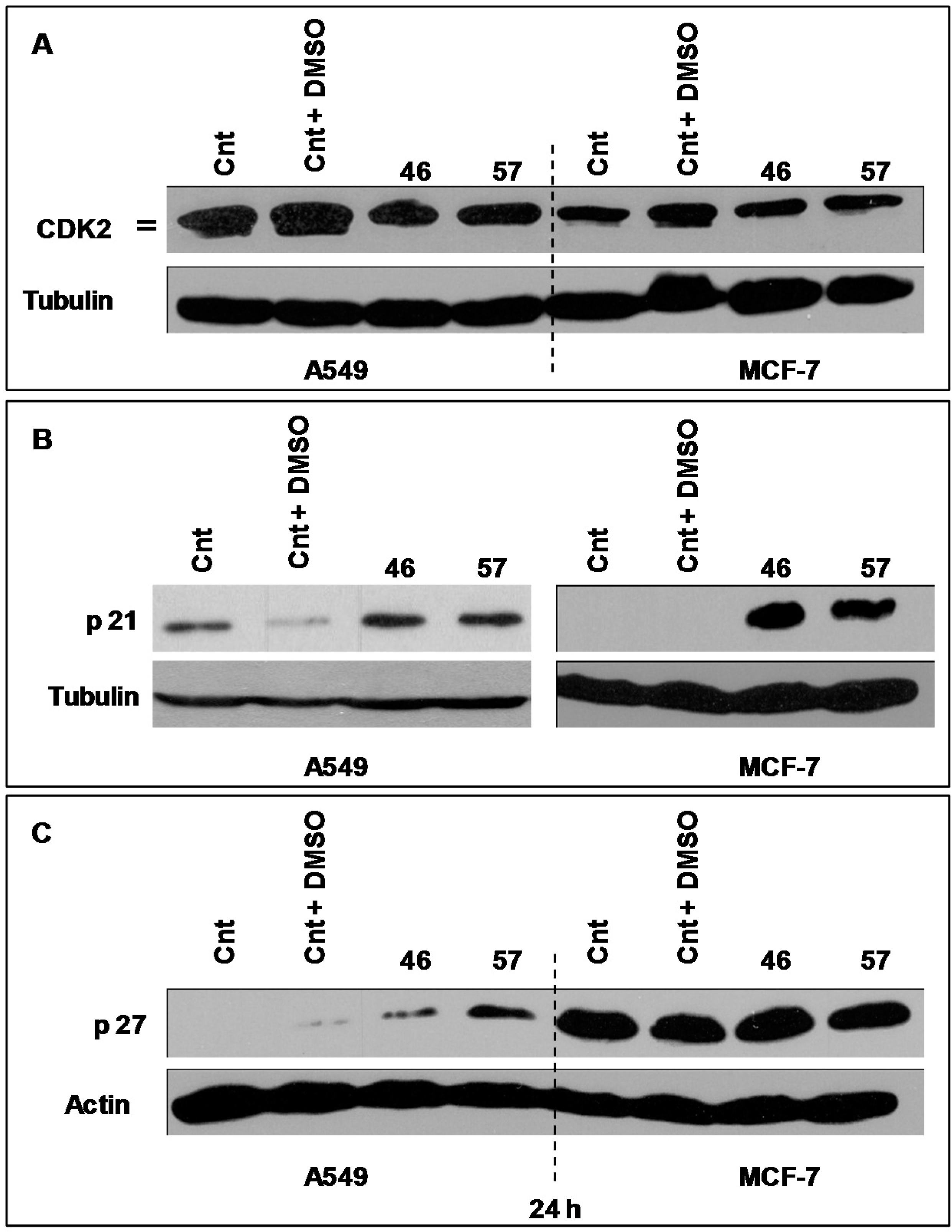
2.3.3. Cdk2, p21 and p27
2.3.4. Bcl-XL
2.4. Evaluation of Senescence in Cells
2.5. DPPH-Based Assay to Estimate the Antioxidant Capacity of J. commnunis and C. coggygria Extracts
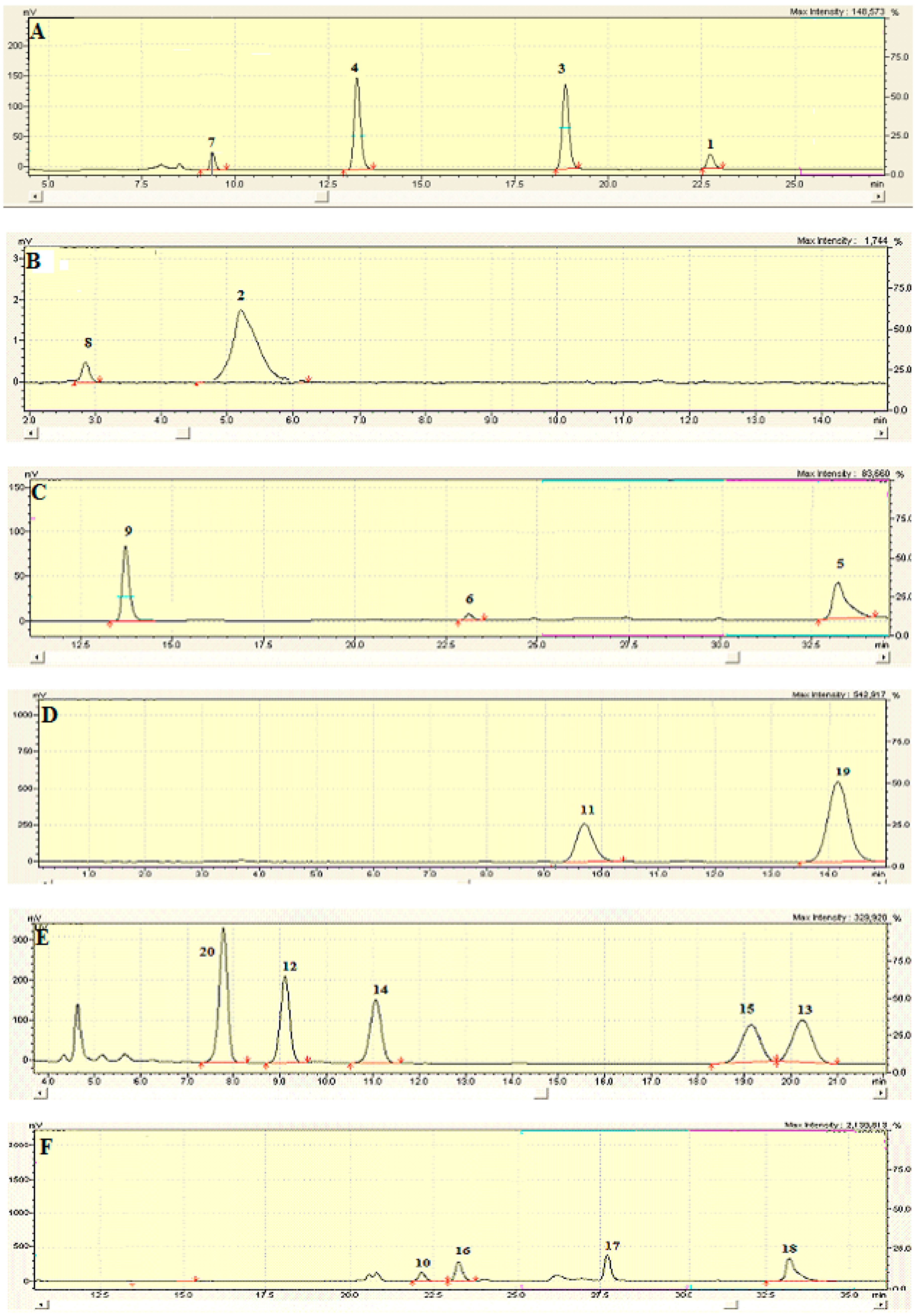
2.6. Identification and Characterization of Chemical Components of the Plant Extracts
3. Materials and Methods
3.1. Collection of Plant Samples
3.2. Preparation of Plant Extract
3.3. General Composition of Plant Extracts
3.4. Microbiological Assay
3.5. Cell Lines
3.6. Cells Growth and Viability Assay
3.7. Flow Cytometry
3.8. Western Blot Analysis
3.9. Antibodies
3.10. Senescence-Associated β-galactosidase Activity
3.11. Statistical Analysis
3.12. Chemicals
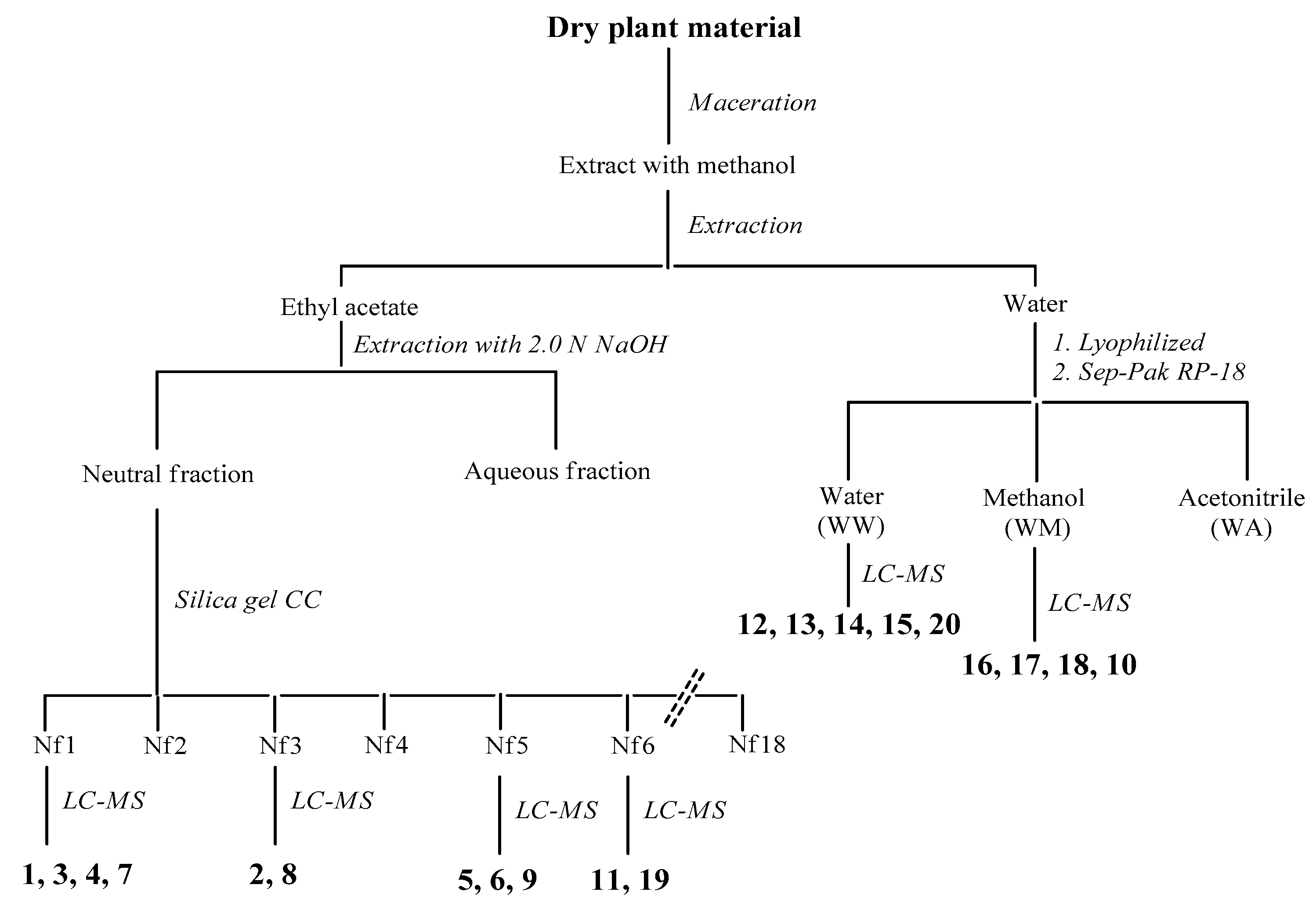
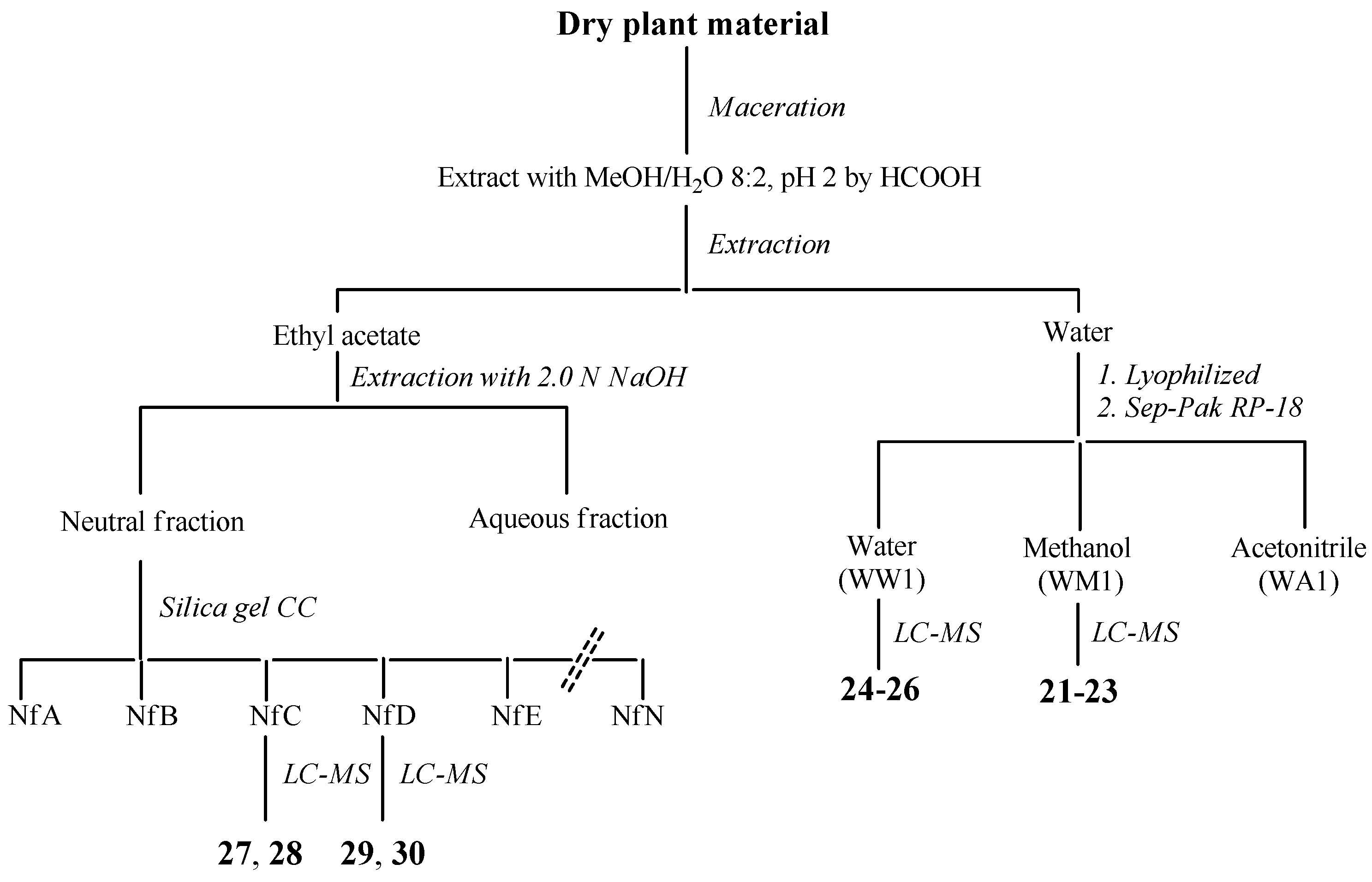
3.13. Extraction and Isolation Procedures
3.14. Determination of 2,2-Diphenyl-1-picrylhydrazyl (DPPH) Radical Scavenging Capacity
3.15. LC-MS/MS Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A
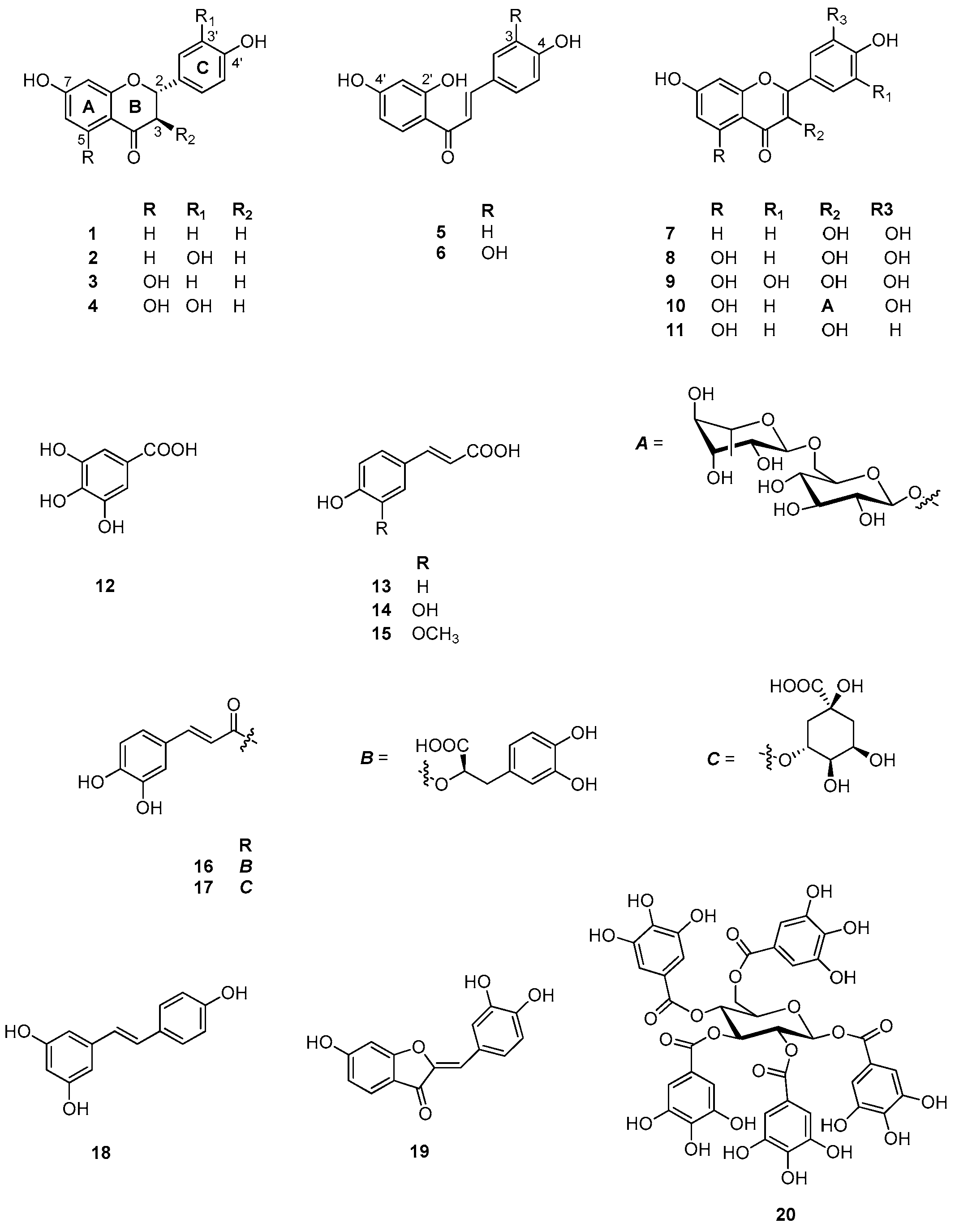
| No. | Common Name | IUPAC Name | CAS Number | Ref. |
|---|---|---|---|---|
| 1 | Liquiritigenin | 4’,7-Dihydroxyflavanone | 578-86-9 | [37] |
| 2 | Fustin | 3’,4’,7-Trihydroxyflavanol | 20725-03-5 | [38] |
| 3 | (R,S)-Naringenin | 4’,5,7-Trihydroxyflavanone | 67604-48-2 | [39,40] |
| 4 | Taxifolin | 3’,4’,5,7-Tetrahydroxyflavanol | 480-18-2 | [40] |
| 5 | Isoliquiritigenin | trans-2’,4,4’-Trihydroxychalcone | 961-29-5 | [41] |
| 6 | Butein | trans-2’,3,4,4’-Tetrahydroxychalcone | 487-52-5 | [38] |
| 7 | Fisetin | 3’,4’,7-Trihydroxyflavonol | 528-48-3 | [39] |
| 8 | Eriodictyol | 3’,4’,5,7-Tetrahydroxyflavonol | 4049-38-1 | [39] |
| 9 | Myricetin | 3,5,7,3’,4',5’-Hexahydroxyflavone | 529-44-2 | [37] |
| 10 | Rutin | Quercetin 3-O-α-l-rhamnopyranosyl-(1→6)-β-d-glucopyranoside | 153-18-4 | [37] |
| 11 | Kaempferol | 3,5,7,4’-Tetrahydroxyflavone | 520-18-3 | [42] |
| 12 | Gallic acid | 3,4,5-Trihydroxybenzoic acid | 149-91-7 | [40] |
| 13 | Coumaric acid | 4-Hydroxycinnamic acid | 25429-38-3 | [37] |
| 14 | Caffeic acid | 3,4-Dihydroxycinnamic acid | 331-39-5 | [37] |
| 15 | Ferulic acid | 3-Methoxy-4-Hydroxycinnamic acid | 1135-24-6 | [37] |
| 16 | Rosmarinic acid | (2R)-3-(3,4-dihydroxyphenyl)-2-[(E)-3-(3,4-dihydroxyphenyl)prop-2-enoyl]oxypropanoic acid | 20283-92-5 | [43] |
| 17 | Chlorogenic acid | 3-O-Caffeoylquinic acid | 327-97-9 | [37] |
| 18 | Resveratrol | 3,4’,5-Trihydroxystilbene | 501-36-0 | [44] |
| 19 | Sulfuretin | 3’,4’,6-Trihydroxyaurone | 50-99-7 | [38] |
| 20 | Pentagalloyl glucose | 1,2,3,4,6-Pentagalloyl-d-glucose | 50678-27-8 | [37] |
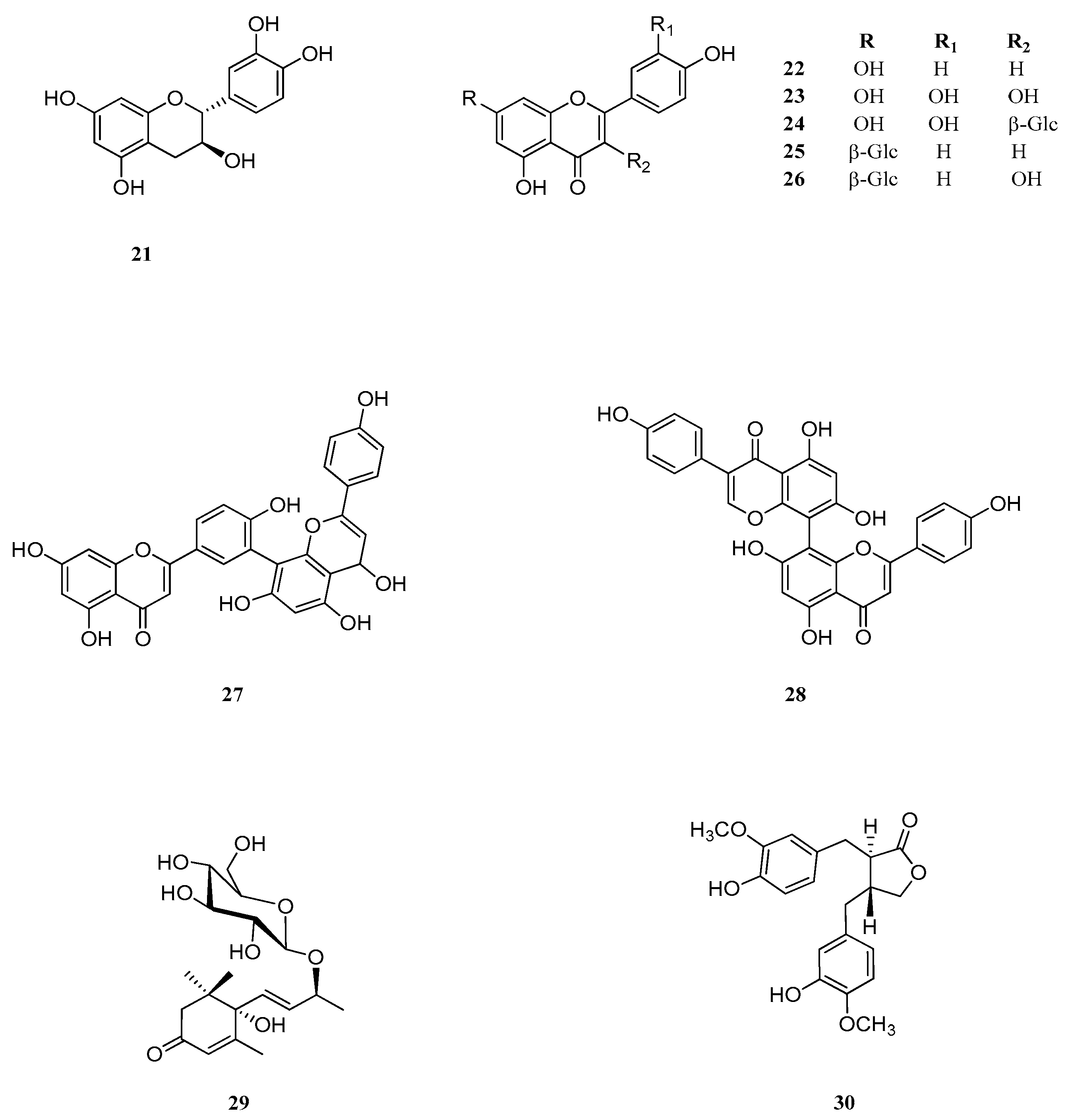
| No. | Common Name | IUPAC Name | CAS Number | Ref. |
|---|---|---|---|---|
| 21 | Catechin | 3’,4’,5,7-Tetrahydroxy-2,3-trans-flavan-3-ol | 154-23-4 | [45] |
| 22 | Apigenin | 5,7,4’-Trihydroxyflavone | 520-36-5 | [46] |
| 23 | Quercetin | 3,5,7,3’,4’-Pentahydroxyflavone | 117-39-5 | [37] |
| 24 | Isoquercetin | 3-O-β-d-Glucopyranosylquercetin | 482-35-9 | [37] |
| 25 | Apigetrin | Apigenin 7-O-β-d-Glucopiranoside | 578-74-5 | [46] |
| 26 | - | Kaempferol-7-O-β-d-Glucopiranoside | 16290-07-6 | [47] |
| 27 | Amentoflavone | Didemethyl ginkgetin | 1617-53-4 | [48] |
| 28 | Cupressoflavon | - | - | [48] |
| 29 | Corchoionoside C | (6S,9S)-Roseoside A | 185414-25-9 | [49] |
| 30 | Matairesinol | Dibenzylbutyrolactone lignanolide | 580-72-3 | [50] |
| Compound | Precursor Ion (m/z) | Daughter Ion (m/z) | Ref. |
|---|---|---|---|
| 1 | 255 [M − H]− | 135 | [37] |
| 2 | 271 [M − H]− | - | [38], standard |
| 3 | 271 [M − H]− | 177, 151, 119, 107 | [39,40], standard |
| 4 | 303 [M − H]− | 285, 251, 235, 217, 179, 177, 125, 113, 101 | [40] |
| 5 | 279 [M + Na]+ | - | [41] |
| 6 | 271 [M − H]− | - | [38] |
| 7 | 285 [M − H]− | 257, 241, 229, 163, 135, | [39] |
| 8 | 287 [M − H]− | 151, 135, 125, 107 | [39] |
| 9 | 317 [M − H]− | 271, 245, 179, 152, 151, 137, 124 | [40] |
| 10 | 609 [M − H]− | 301, 300 | [37] |
| 11 | 285 [M − H]− | 257, 229, 213 | [42] |
| 12 | 169 [M − H]− | 125, 127, 107 | [40] |
| 13 | 163 [M − H]− | 119 | [37] |
| 14 | 179 [M − H]− | 135, 117 | [37] |
| 15 | 193 [M − H]− | 178, 149, 134 | [37] |
| 16 | 359 [M − H]− | 197, 160 | [43] |
| 17 | 353 [M − H]− | 191, 179, 173, 161, 135 | [37] |
| 18 | 227 [M − H]− | 185, 159, 158, 143, 119, | [44] |
| 19 | 269 [M − H]− | - | [38] |
| 20 | 939 [M − H]− | 787, 769, 617 | [37] |
| Compound | Precursor Ion (m/z) | Daughter Ion (m/z) | Ref. |
|---|---|---|---|
| 21 | 289 | 248, 227, 217, 203, 188, 164, 151, 125, 123 | [45] |
| 22 | 269 | 227, 195, 183, 151, 121, 117, 107 | [46] |
| 23 | 301 | 273, 229, 178, 151 | [37] |
| 24 | 463 | 300, 301, 303 | [37] |
| 25 | 431 | 269, 225, 197, 183, 169, 151, 121, 117 | [46] |
| 26 | 447 | 285, 254 | [47] |
| 27 | 537 | [48], standard | |
| 28 | 537 | - | [48], standard |
| 29 | 385 | [49] | |
| 30 | 357 | 342, 313, 298, 209 | [50] |
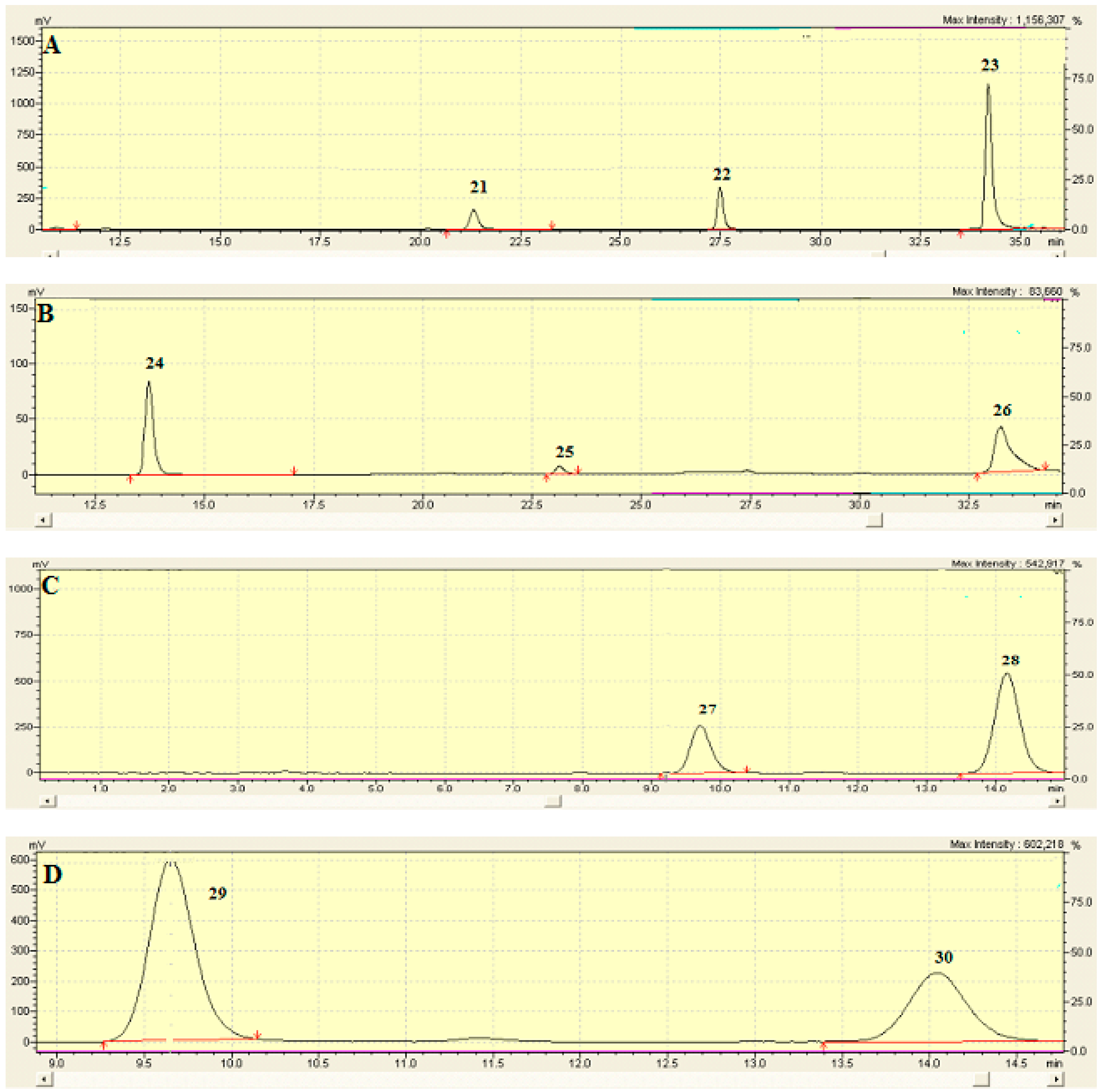
References
- DNP—Dictionary of Natural Products. Avaible online: http://dnp.chemnetbase.com (accessed on 15 June 2015).
- Dai, J.; Mumper, R.J. Plant phenolics: Extraction, analysis and their antioxidant and anticancer properties. Molecules 2010, 15, 7313–7352. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.H. Potential synergy of phytochemicals in cancer prevention: Mechanism of action. J. Nutr. 2004, 134, 3479–3485. [Google Scholar]
- Ferrazzano, G.F.; Roberto, L.; Catania, M.R.; Chiaviello, A.; de Natale, A.; Roscetto, E.; Pinto, G.; Pollio, A.; Ingenito, A.; Palumbo, G. Screening and scoring of antimicrobial and biological activities of Italian vulnerary plants against major oral pathogenic bacteria. Evid. Based Complement. Altern. Med. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Van Slambrouck, S.; Daniels, A.L.; Hooten, C.J.; Brock, S.L.; Jenkins, A.R.; Ogasawara, M.A.; Baker, J.M.; Adkins, G.; Elias, E.M.; Agustin, V.J.; et al. Effects of crude aqueous medicinal plant extracts on growth and invasion of breast cancer cells. Oncol. Rep. 2007, 17, 1487–1492. [Google Scholar] [CrossRef] [PubMed]
- Savikin, K.; Zdunic, G.; Jankovic, T.; Stanojkovic, T.; Juranic, Z.; Menkovic, N. In vitro cytotoxic and antioxidative activity of Cornus mas and Cotinus coggygria. Nat. Prod. Res. 2009, 23, 1731–1739. [Google Scholar] [CrossRef] [PubMed]
- Cmielová, J.; Rezáčová, M. p21Cip1/Waf1 protein and its function based on a subcellular localization. J. Cell Biochem. 2011, 112, 3502–3506. [Google Scholar] [CrossRef] [PubMed]
- Levkau, B.; Koyama, H.; Raines, E.W.; Clurman, B.E.; Herren, B.; Orth, K.; Roberts, J.M.; Ross, R. Cleavage of p21Cip1/Waf1 and p27Kip1 mediates apoptosis in endothelial cells through activation of Cdk2: Role of a caspase cascade. Mol. Cell 1998, 1, 553–563. [Google Scholar] [CrossRef]
- Tillhon, M.; Cazzalini, O.; Dutto, I.; Stivala, L.A.; Prosperi, A. P21CDKN1A and DNA Repair Systems: Recent Findings and Future Perspectives, New Research Directions in DNA Repair; Chen, C., Ed.; InTech: Rijeka, Croatia, 2013. [Google Scholar]
- Besson, A.; Dowdy, S.F.; Roberts, J.M. CDK inhibitors: Cell cycle regulators and beyond. Dev. Cell 2008, 14, 159–169. [Google Scholar] [CrossRef] [PubMed]
- Johnson, B.W.; Cepero, E.; Boise, L.H. Bcl-xL inhibits cytochrome c release but not mitochondrial depolarization during the activation of multiple death pathways by tumor necrosis factor-alpha. J. Biol. Chem. 2000, 275, 31546–31553. [Google Scholar] [CrossRef] [PubMed]
- Crescenzi, E.; Palumbo, G.; Brady, H.J.M. BCl-2 activates a programme of premature senescence in human carcinoma cells. Biochem. J. 2003, 375, 263–274. [Google Scholar] [CrossRef] [PubMed]
- Kuntz, S.; Wenzel, U.; Daniel, H. Comparative analysis of the effects of flavonoids on proliferation, cytotoxicity, and apoptosis in human colon cancer cell lines. Eur. J. Nutr. 1999, 38, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Noh, E.M.; Park, Y.J.; Kim, J.M.; Kim, M.S.; Kim, H.R.; Song, H.K.; Hong, O.Y.; So, H.S.; Yang, S.H.; Kim, J.S.; et al. Fisetin regulates TPA-induced breast cell invasion by suppressing matrix metalloproteinase-9 activation via the PKC/ROS/MAPK pathways. Eur. J. Pharmacol. 2015, 764, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.M.; Noh, E.M.; Kwon, K.B.; Kim, J.S.; You, Y.O.; Hwang, J.K.; Hwang, B.M.; Kim, M.S.; Lee, S.J.; Jung, S.H.; et al. Suppression of TPA-induced tumor cell invasion by sulfuretin via inhibition of NF-κB-dependent MMP-9 expression. Oncol. Rep. 2013, 29, 1231–1237. [Google Scholar] [PubMed]
- Maggiolini, M.; Statti, G.; Vivacqua, A.; Gabriele, S.; Rago, V.; Loizzo, M.; Menichini, F.; Amdò, S. Estrogenic and antiproliferative activities of isoliquiritigenin in MCF7 breast cancer cells. J. Steroid Biochem. Mol. Biol. 2002, 82, 315–322. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, M.; Yu, L.; Zhao, Y.; He, N.; Yang, X. Antitumor activities of quercetin and quercetin-5′,8-disulfonate in human colon and breast cancer cell lines. Food Chem. Toxicol. 2012, 50, 1589–1599. [Google Scholar] [CrossRef] [PubMed]
- Mamadalieva, N.Z.; Herrmann, F.; El-Readi, M.Z.; Tahrani, A.; Hamoud, R.; Egamberdieva, D.R.; Azimova, S.S.; Wink, M. Flavonoids in Scutellaria immaculata and S. ramosissima (Lamiaceae) and their biological activity. J. Pharm. Pharmacol. 2011, 63, 1346–1357. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Lee, M.S.; Oh, W.K.; Sul, J.Y. Fatty acid synthase inhibition by amentoflavone induces apoptosis and antiproliferation in human breast cancer cells. Biol. Pharm. Bull. 2009, 32, 1427–1432. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Sul, J.Y.; Park, J.B.; Lee, M.S.; Cha, E.Y.; Song, I.S.; Kim, J.R.; Chang, E.S. Fatty acid synthase inhibition by amentoflavone suppresses HER2/neu (erbB2) oncogene in SKBR3 human breast cancer cells. Phytother. Res. 2013, 27, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Bouzaienea, N.N.; Jaziria, S.K.; Kovacic, H.; Chekir-Ghedira, L.; Ghedira, K.; Luis, J. The effects of caffeic, coumaric and ferulic acids on proliferation, superoxide production, adhesion and migration of human tumor cells in vitro. Eur. J. Pharmacol. 2015, 766, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Deiab, S.; Mazzio, E.; Eyunni, S.; McTier, O.; Mateeva, N.; Elshami, F.; Soliman, K.F. 1,2,3,4,6-Penta-O-galloylglucose within Galla chinensis inhibits human LDH-A and attenuates cell proliferation in MDA-MB-231 breast cancer cells. Evid. Based Complement. Altern. Med. 2015, 2015. [Google Scholar] [CrossRef]
- Thompson, L.U.; Boucher, B.A.; Liu, Z.; Cotterchio, M.; Kreiger, N. Phytoestrogen content of foods consumed in Canada, including isoflavones, lignans, and coumestan. Nutr. Cancer 2006, 54, 184–201. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, T.C.; Juan, G.; Darzynkiewicz, Z.; Wu, J.M. Resveratrol increases nitric oxide synthase, induces accumulation of p53 and p21(WAF1/ CIP1), and suppresses cultured bovine pulmonary artery endothelial cell proliferation by perturbing progression through S and G2. Cancer Res. 1999, 59, 2596–2601. [Google Scholar] [PubMed]
- She, Q.B.; Bode, A.M.; Ma, W.Y.; Chen, N.Y.; Dong, Z. Resveratrol-induced activation of p53 and apoptosis is mediated by extracellular-signal regulated protein kinases and p38 kinase. Cancer Res. 2001, 61, 1604–1610. [Google Scholar] [PubMed]
- Ji, B.C.; Hsu, W.H.; Yang, J.S.; Hsia, T.C.; Lu, C.C.; Chiang, J.H.; Yang, J.L.; Lin, C.H.; Lin, J.J.; Suen, L.J.W.; et al. Gallic Acid Induces Apoptosis via Caspase-3 and Mitochondrion-Dependent Pathways in Vitro and Suppresses Lung Xenograft Tumor Growth in Vivo. J. Agric. Food Chem. 2009, 57, 7596–7604. [Google Scholar] [CrossRef] [PubMed]
- Casagrande, F.; Darbon, J.M. Effects of structurally related flavonoids on cell cycle progression of human melanoma cells: Regulation of cyclin-dependent kinases CDK2 and CDK1. Biochem. Pharmacol. 2001, 61, 1205–1215. [Google Scholar] [CrossRef]
- Chien, S.Y.; Wu, Y.C.; Chung, J.G.; Yang, J.S.; Lu, H.F.; Tsou, M.F.; Wood, W.G.; Kuo, S.J.; Che, D.R. Quercetin-induced apoptosis acts through mitochondrial- and caspase-3-dependent pathways in human breast cancer MDA-MB-231 cells. Hum. Exp. Toxicol. 2009, 28, 493–503. [Google Scholar] [CrossRef] [PubMed]
- Aalinkeel, R.; Bindukumar, B.; Reynolds, J.L.; Sykes, D. E.; Mahajan, S.D.; Chadha, K.C.; Schwartz, S.A. The dietary bioflavonoid, quercetin, selectively induces apoptosis of prostate cancer cells by down-regulating the expression of heat shock protein 90. Prostate 2008, 68, 1773–1789. [Google Scholar] [CrossRef] [PubMed]
- Koehn, F.E.; Carter, G.T. The evolving role of natural products in drug discovery. Nature 2005, 4, 206–220. [Google Scholar] [CrossRef] [PubMed]
- Solowey, E.; Lichtenstein, M.; Sallon, S.; Paavilainen, H.; Solowey, E.; Lorberboum-Galski, H. Evaluating medicinal plants for anticancer activity. Sci. World J. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Postiglione, I.; Chiaviello, A.; Aloj, S.M.; Palumbo, G. 5-Aminolaevulinic acid/photo-dynamic therapy and gefitinib in non-small cell lung cancer cell lines: A potential strategy to improve gefitinib therapeutic efficacy. Cell Prolif. 2013, 46, 382–395. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1971, 227, 680–685. [Google Scholar] [CrossRef]
- Dimri, G.P.; Lee, X.; Basile, G.; Acosta, M.; Scott, G.; Roskelley, C.; Medrano, E.E.; Linskens, M.; Rubelj, I.; Pereira-Smith, O.; et al. A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc. Natl. Acad. Sci. USA. 1995, 92, 9363–9367. [Google Scholar] [CrossRef] [PubMed]
- GraphPad QuickCalcs. Available online: http://www.graphpad.com/quickcalcs/ (accessed on 1 November 2010).
- Xu, W.; Huang, M.; Li, H.; Chen, X.; Zhang, Y.; Lia, J.; Xu, W.; Chu, K.; Chen, L. Chemical profiling and quantification of Gua-Lou-Gui-Zhi decoction by high performance liquid chromatography/quadrupole-time-of-flight mass spectrometry and ultra-performance liquid chromatography/triple quadrupole mass spectrometry. J. Chromatogr. B Analyt. Technol. Biomed. Life. Sci. 2015, 986, 69–84. [Google Scholar] [CrossRef] [PubMed]
- Junior, G.M.V.; de Sousa, C.M.; Cavalheiro, A.J.; Lago, J.H.G.; Chaves, M.H. Phenolic derivatives from fruits of Dipteryx lacunifera Ducke and evaluation of their antiradical activities. Helv. Chim. Acta 2008, 11, 2159–2167. [Google Scholar] [CrossRef]
- Nicolas, N.; Rustan, I.; de Hoffman, E.; Quetin-Leclercq, J. Determination of flavone, flavonol, and flavanone aglycones by negative ion liquid chromatography electrospray ion trap mass spectrometry. J. Am. Soc. Mass Spectrom. 2001, 12, 707–715. [Google Scholar]
- Sun, Z.; Zhao, L.; Zuo, L.; Qi, C.; Zhao, P.; Hou, X. A UHPLC–MS/MS method for simultaneous determination of six flavonoids, gallic acid and 5,8-dihydroxy-1,4-naphthoquinone in rat plasma and its application to a pharmacokinetic study of cortex Juglandis mandshuricae extract. J. Chromatogr. B 2014, 958, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Salem, M.M.; Werbovetz, K.A. Isoflavonoids and other compounds from Psorothamnus arborescens with antiprotozoal activities. J. Nat. Prod. 2006, 69, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Matic, S.; Stanic, S.; Bogojevic, D.; Vidakovic, M.; Grdovic, N.; Dinic, S.; Solujic, S.; Mladenovic, M.; Stankovic, N.; Mihailovi, M. Methanol extract from the stem of Cotinus coggygria Scop., and its major bioactive phytochemical constituent myricetin modulate pyrogallol-induced DNA damage and liver injury. Mutat. Res. 2013, 755, 81–89. [Google Scholar] [PubMed]
- Xiaochuan, L.; Chen, Y.; Weikang, S.; Gangyi, L.; Jingying, J.; Yiping, W. Simultaneous determination of magnesium lithospermate B, rosmarinic acid, and lithospermic acid in beagle dog serum by liquid chromatography/tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2004, 18, 2878–2882. [Google Scholar]
- Careri, M.; Corradini, C.; Elviri, L.; Nicoletti, I.; Zagnoni, I. Liquid chromatography-electrospray tandem mass spectrometry of cis-resveratrol and trans-resveratrol: Development, validation, and application of the method to red wine, grape, and winemaking byproducts. J. Agric. Food Chem. 2004, 52, 6868–6874. [Google Scholar] [CrossRef] [PubMed]
- Guaratini, T.; Armelini, A.I.P.V.; Ferrari, C.R.; Schefer, R.R.; Neto, A.P.; Navas, R.; Reigada, J.B.; Silva, D.B. Application of matrix-assisted laser-desorption/ionization time-of-flight LIFT for identification of cocoa condensed tannins. J. Mass Spectrom. 2014, 49, 251–255. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; He, L.; Lu, L.; Liu, Y.; Dong, G.; Miao, J.; Luo, P. Characterization and quantification of the chemical compositions of Scutellariae Barbatae herba and differentiation from its substitute by combining UHPLC–PDA–QTOF–MS/MS with UHPLC–MS/MS. J. Pharm. Biomed. Anal. 2015, 109, 62–66. [Google Scholar] [CrossRef] [PubMed]
- Das Neves Costa, F.; Jerz, G.; de Souza Figueiredo, F.; Winterhalter, P.; Leitão, G.G. Solvent system selectivities in countercurrent chromatography using Salicornia gaudichaudiana metabolites as practical example with off-line electrospray mass-spectrometry injection profiling. J. Chromatogr. A. 2015, 1385, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Innocenti, M.; Michelozzi, M.; Giaccherini, C.; Ieri, F.; Vincieri, F.F.; Mulinacci, N. Flavonoids and biflavonoids in Tuscan berries of Juniperus communis L.: detection and quantitation by HPLC/DAD/ESI/MS. J. Agric. Food Chem. 2007, 55, 6596–6602. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, T.; Iida, N.; Inatomi, Y.; Murata, H.; Inada, A.; Murata, J.; Lang, F.A.; Iinuma, M.; Tanaka, T.; Sakagami, Y. A monoterpene glucoside and three megastigmane glycosides from Juniperus communis var. depressa. Chem. Farm. Bull. 2005, 53, 783–787. [Google Scholar] [CrossRef]
- Eklund, P.C.; Backman, M.J.; Kronberg, L.A.; Annika, I.; Smeds, A.I.; Sjoholm, R.E. Identification of lignans by liquid chromatography-electrospray ionization ion-trap mass spectrometry. J. Mass Spectrom. 2008, 43, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Extracts from J. communis and C. Coggygria. can be obtained from the authors on request.




| Ethyl Acetate Extracts | Water Extracts | ||
|---|---|---|---|
| C. coggygria | J. communis | C. coggygria | J. communis |
| Liquiritigenin (1) | Amentoflavone (27) | Rutin (10) | Catechin (21) |
| Fustin (2) | Cupressoflavon (28) | Gallic acid (12) | Apigenin (22) |
| Naringenin (3) | Corchoionoside C (29) | Coumaric acid (13) | Quercetin (23) |
| Taxifolin (4) | Matairesinol (30) | Caffeic acid (14) | Isoquercetin (24) |
| Isoliquiritigenin (5) | Ferulic acid (15) | Apigetrin (23) | |
| Butein (6) | Rosmarinic acid (16) | Kaempferol-7-O-α-d-Glucopiranoside (26) | |
| Fisetin (7) | Chlorogenic acid (17) | ||
| Eriodictyol (8) | Resveratrol (18) | ||
| Myricetin (9) | Pentagalloyl glucose (20) | ||
| Kaempferol (11) | |||
| Sulfuretin (19) | |||
| Extracts (Undiluted Stocks) 15 µL | Incubation Time (h) (in DMEM) | Bacterial Strain |
|---|---|---|
| J. communis | 72 | Sterile |
| C. coggygria | 24 | B. licheniformis B. subtilis B. sonorensis |
| P. scolopendrium | 72 | B. licheniformis B. subtilis |
| E. hyemale | 72 | B. licheniformis B. subtilis |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pollio, A.; Zarrelli, A.; Romanucci, V.; Di Mauro, A.; Barra, F.; Pinto, G.; Crescenzi, E.; Roscetto, E.; Palumbo, G. Polyphenolic Profile and Targeted Bioactivity of Methanolic Extracts from Mediterranean Ethnomedicinal Plants on Human Cancer Cell Lines. Molecules 2016, 21, 395. https://doi.org/10.3390/molecules21040395
Pollio A, Zarrelli A, Romanucci V, Di Mauro A, Barra F, Pinto G, Crescenzi E, Roscetto E, Palumbo G. Polyphenolic Profile and Targeted Bioactivity of Methanolic Extracts from Mediterranean Ethnomedicinal Plants on Human Cancer Cell Lines. Molecules. 2016; 21(4):395. https://doi.org/10.3390/molecules21040395
Chicago/Turabian StylePollio, Antonino, Armando Zarrelli, Valeria Romanucci, Alfredo Di Mauro, Federica Barra, Gabriele Pinto, Elvira Crescenzi, Emanuela Roscetto, and Giuseppe Palumbo. 2016. "Polyphenolic Profile and Targeted Bioactivity of Methanolic Extracts from Mediterranean Ethnomedicinal Plants on Human Cancer Cell Lines" Molecules 21, no. 4: 395. https://doi.org/10.3390/molecules21040395
APA StylePollio, A., Zarrelli, A., Romanucci, V., Di Mauro, A., Barra, F., Pinto, G., Crescenzi, E., Roscetto, E., & Palumbo, G. (2016). Polyphenolic Profile and Targeted Bioactivity of Methanolic Extracts from Mediterranean Ethnomedicinal Plants on Human Cancer Cell Lines. Molecules, 21(4), 395. https://doi.org/10.3390/molecules21040395