Quantitative Variation of Flavonoids and Diterpenes in Leaves and Stems of Cistus ladanifer L. at Different Ages
Abstract
:1. Introduction
2. Results
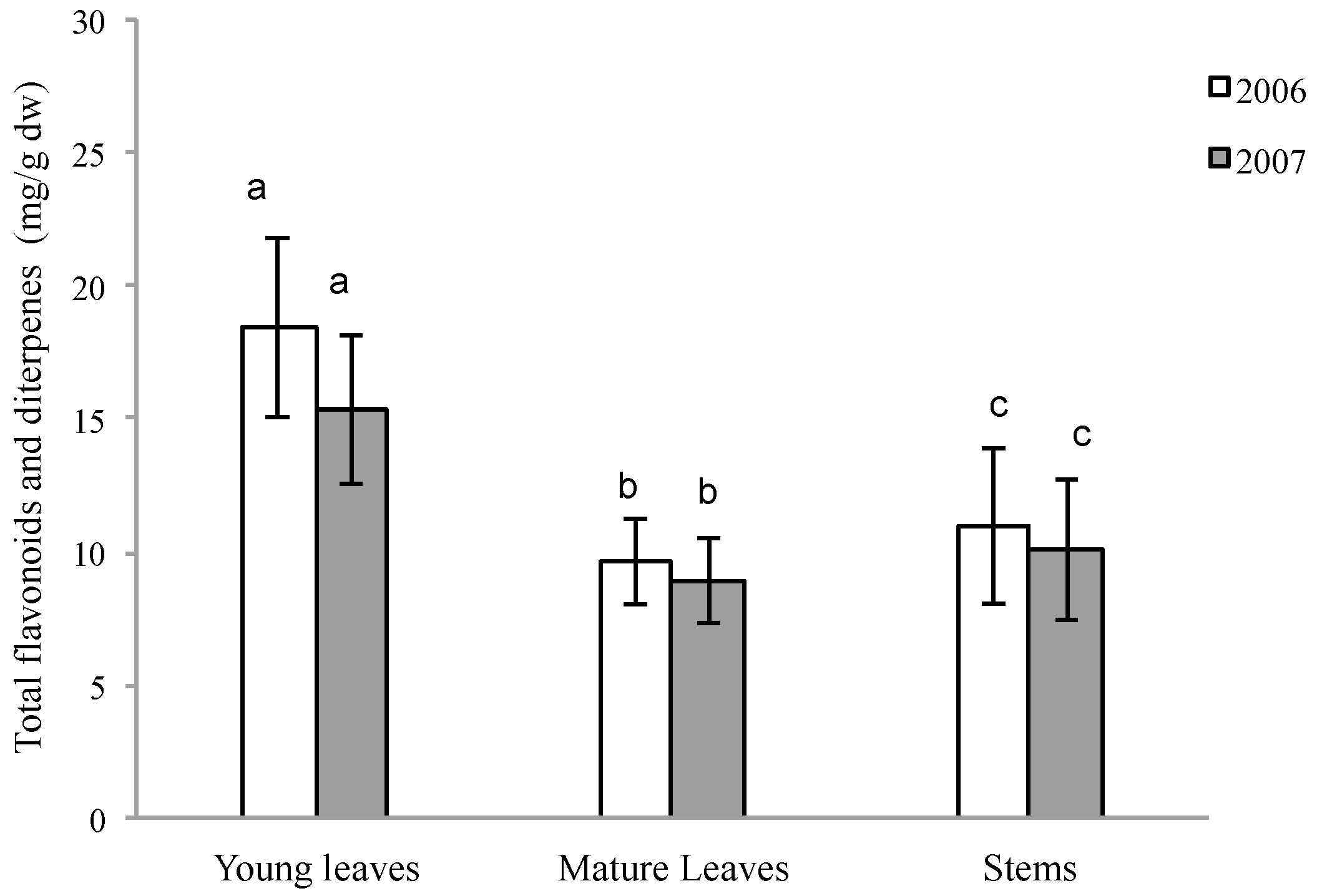
2.1. Quantitative Variation of Flavonoids and Diterpenes between Young Leaves, Mature Leaves and Stems
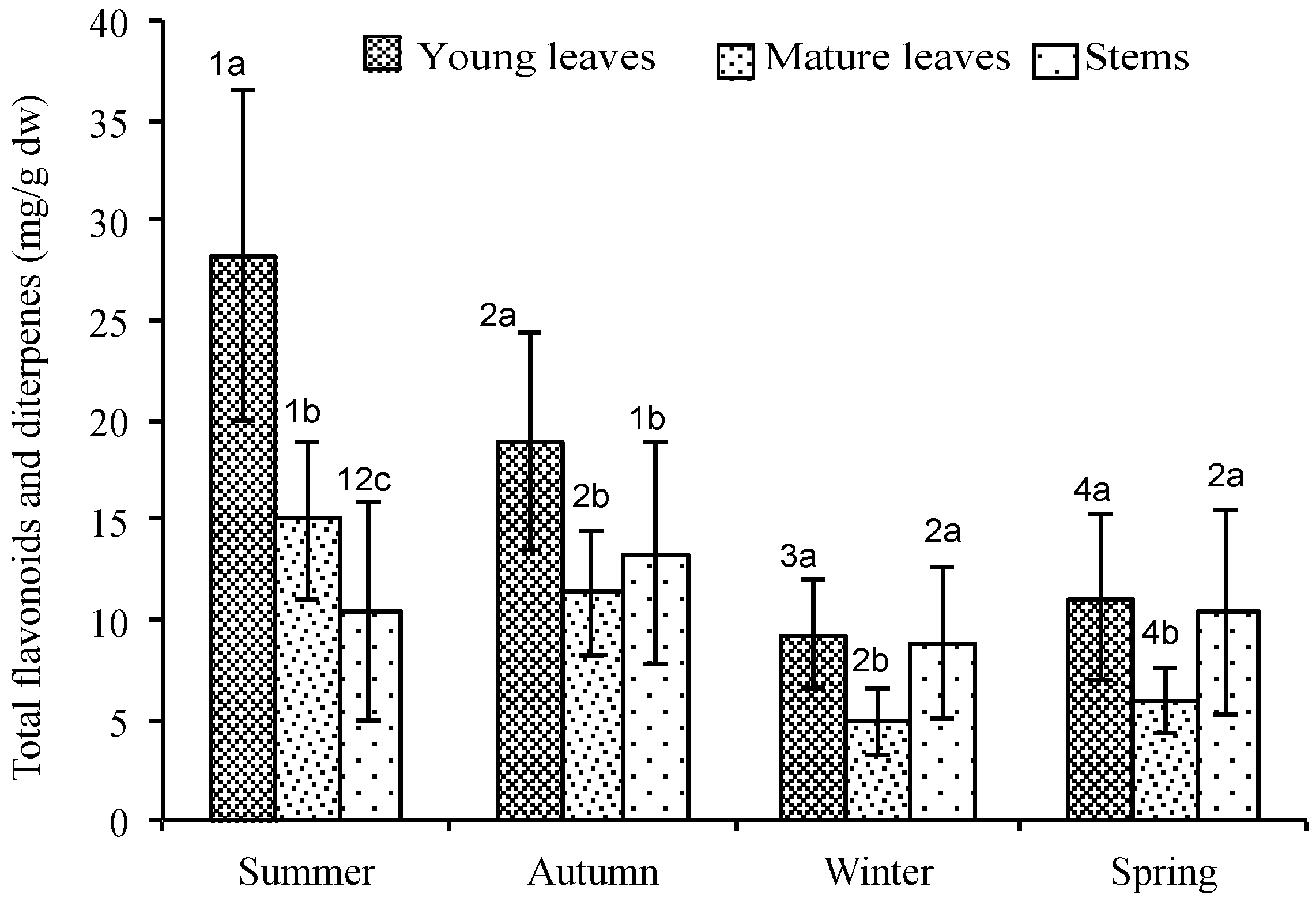
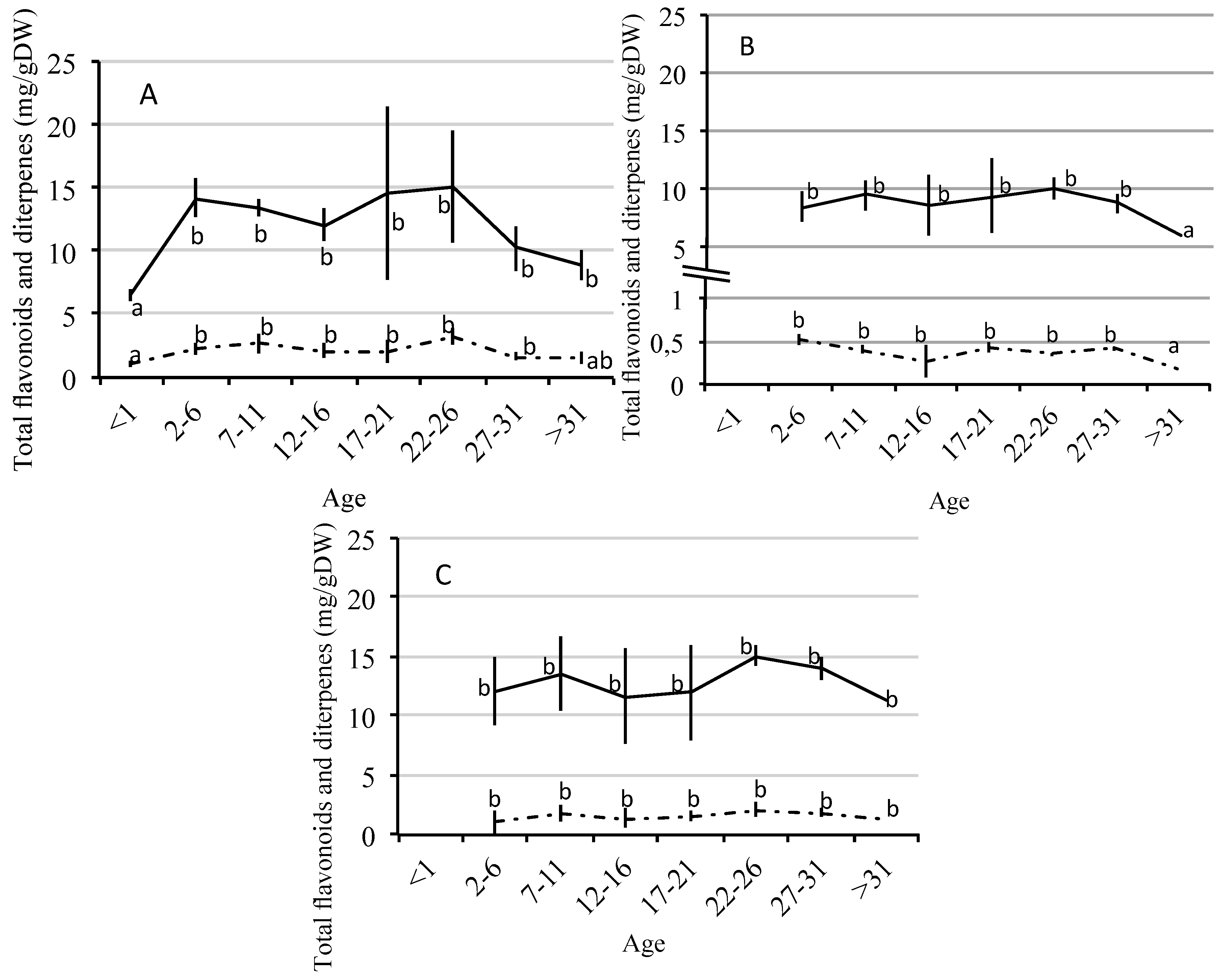
2.2. Quantitative Variation of Flavonoids and Diterpenes between Ages
3. Discussion
4. Experimental Section
4.1. Sample Collection
4.2. Age Determination
4.3. Extraction and Assay of Secondary Metabolites
4.4. Statistical Analyses
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Anaya, A.L. Ecología Química; Plaza y Valdés, S.A.: Ciudad de México, Mexico, 2003. [Google Scholar]
- Granados-Sanchez, D.; Ruiz-Puga, P.; Barrera-Escorcia, H. Ecología de la herbivoría. Rev. Chapingo 2008, 14, 51–63. [Google Scholar]
- Gross, J.; Hilker, M. Chemoecological studies of the exocrine glandular larval secretions of two chrysomelid species: Phaedon cochleariae and Chrysomela lapponica. Chemoecology 1994/1995, 5/6, 185–189. [Google Scholar] [CrossRef]
- Ikonen, A.; Tahvanainen, J.; Roininen, H. Chlorogenic acid as an antiherbivore defence of willows against leaf beetles. Entomol. Exp. Appl. 2001, 99, 47–54. [Google Scholar] [CrossRef]
- Lattanzio, V.; Lattanzio, V.M.T.; Cardinali, A. Role of phenolics in the resistance mechanisms of plants against fungal pathogens and insects. In Phytochemistry: Advances in Research. Research Signpost37/661 (2); Imperato, F., Ed.; Fort PO, Trivandrum-695 023: Kerala, India, 2006; pp. 23–67. [Google Scholar]
- Lattanzio, V. Bioactive polyphenols: Their role in quality and storability of fruit and vegetables. J. Appl. Bot. 2003, 77, 128–146. [Google Scholar]
- Zagrobelny, M.; Bak, S.; Rassmusen, A.V.; Jørgensen, B.; Lindberg Møller, B. Cyanogenic glucosides and plant–insect interactions. Phytochemistry 2004, 65, 293–306. [Google Scholar] [CrossRef] [PubMed]
- Chaves, N.; Escudero, J.C. Allelopathic effect of Cistus ladanifer on seed germination. Funct. Ecol. 1997, 11, 432–440. [Google Scholar] [CrossRef]
- Chaves, N.; Sosa, T.; Escudero, J.C. Plant growth inhibiting flavonoids in exudate of Cistus ladanifer and in associated soils. J. Chem. Ecol. 2001, 27, 623–631. [Google Scholar] [CrossRef] [PubMed]
- Chaves, N.; Sosa, T.; Alías, J.C.; Escudero, J.C. Identification and effects of the interaction of phytotoxic compounds from exudate of Cistus ladanifer leaves. J. Chem. Ecol. 2001, 27, 611–621. [Google Scholar] [CrossRef] [PubMed]
- Baas, W.J. Secondary plant compounds, their ecological significance and consequences for the carbon budget. Introduction of the carbon/nutrient cycle theory. In Causes and Consequences of Variation in Growth Rate and Productivity of Higher Plants; Lambers, H., Cambridge, M.L., Konings, H., Pons, T.L., Eds.; SPB Academic Publishing bv: The Hague, The Netherlands, 1989; pp. 313–340. [Google Scholar]
- Harborne, J.B. Biochemical plant ecology. In Plant Biochemistry; Dey, P.M., Harborne, J.B., Eds.; Academic Press: London, UK, 1997; pp. 503–516. [Google Scholar]
- Onyilagha, J.C.; Malhotra, B.; Elder, M.; French, C.J.; Towers, G.H.N. Comparative studies of inhibitory activities of chalcones on tomato ringspot virus (ToRSV). Can. J. Plant Pathol. 1997, 19, 133–137. [Google Scholar] [CrossRef]
- Zobel, A.M.; Clarke, P.A.; Lynch, J.M. Production of phenolics in response to UV irradiation and heavy metals in seedlings of Acer species. In Recent Advances in Allelopathy; Narwal, S.S., Ed.; CRC Press: Boca Raton, FL, USA, 1999; pp. 1–12. [Google Scholar]
- Ryan, K.G.; Swinny, E.E.; Winefield, C.; Markham, K.R. Flavonoids and UV photoprotection in Arabidopsis mutants. Zeitschrift fur Naturforschung—Section C. J. Biosci. 2001, 56, 745–754. [Google Scholar]
- Subramanian, S.; Stacey, G.; Yu, O. Distinct, crucial roles of flavonoids during legume nodulation. Trends Plant Sci. 2007, 12, 282–285. [Google Scholar] [CrossRef] [PubMed]
- Croteau, R.; Ketchum, R.E.B.; Long, R.M.; Kaspera, R.; Wildung, M.R. Taxol biosynthesis and molecular genetics. Phytochem. Rev. 2006, 5, 75–97. [Google Scholar] [CrossRef] [PubMed]
- Küpeli, E.; Deliorman Orhan, D.; Yesilada, E. Effect of Cistus laurifolius L. leaf extracts and flavonoids on acetaminophen-induced hepatotoxicity in mice. J. Ethnopharmacol. 2006, 103, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Coley, P.D.; Kursar, T.A. Anti-herbivore defenses of young tropical leaves: Physiological constraints and ecological tradeoffs. In Tropical Forest Plant Ecophysiology; Mulkey, S.S., Chazdon, R., Smith, A.P., Eds.; Chapman and Hall: New York, NY, USA, 1996; pp. 305–336. [Google Scholar]
- Coley, P.D.; Barone, J.A. Ecology of Defenses. In Encyclopedia of Biodiversity; Levin, S., Ed.; Academic Press: San Diego, CA, USA, 2001; Volume 2, pp. 11–21. [Google Scholar]
- Brenes-Arguedas, T.; Coley, P.D. Phenotypic variation and spatial structure of secondary chemistry in a natural population of a tropical tree species. Oikos 2005, 108, 410–420. [Google Scholar] [CrossRef]
- Waterman, P.G.; Mole, S. Analysis of Phenolic Plant Metabolites; Blackwell Scientific Publications: Oxford, UK, 1994. [Google Scholar]
- Del Valle, J.C.; Buide, M.L.; Casimiro-Soriguer, I.; Whittall, J.B.; Narbona, E. On flavonoid accumulation in different plant parts: Variation patterns hmong individuals and populations in the shore campion (Silene littorea). Front. Plant Sci. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Herrera, C.M. Tipos morfológicos y funcionales en plantas del matorral mediterráneo del sur de España. Stvdia Oecol. 1984, 5, 7–34. [Google Scholar]
- Nuñez, E. Ecología del jaral de Cistus ladanifer L. Ph.D. Thesis, Facultad de Ciencias, Universidad de Extremadura, Badajoz, Spain, 1989. [Google Scholar]
- Braun-Blanquet, J.; Molinier, R.; Warner, H. Prodome des Groupements Vegetaux; Imprimerie de la Charite: Montpellier, Francia, 1941. [Google Scholar]
- Devesa, J.A. Vegetación y Flora de Extremadura; Universitas Editorial: Badajoz, España, 2008. [Google Scholar]
- Garcia-Martin, D.; Garcia-Vallejo, C. Contribuition a la connaissance de l’huile essentielle de Cistus ladanifer var. maculatus Dun (Ciste commun-jara d´Espagne). Parf. Cosm. Savon 1969, 12, 283–290. [Google Scholar]
- Crespo, J.M.; Cardenal, J.A.; Peral, D.; Vallejo, J.R. Jara pringosa (Cistus ladanifer), usos, utilidades y curiosidades en Extremadura. Rev. Estud. Extremeños 2009, 65, 1637–1650. [Google Scholar]
- Becerro de Bengoa, G. Aprovechamiento de Cistus ladanifer L. Congreso Nacional de Medio Ambiente; Universidad Católica de Ávila: Ávila, España, 2014. [Google Scholar]
- Pascual, T.; Urones, J.G.; Gonzalez, M. Terpenoides monohidroxilados de la gomorresina de Cistus ladaniferus L. An. Quim. 1977, 73, 1024–1028. [Google Scholar]
- Proksch, P.; Gülz, P.G. Methylated flavonoids from Cistus ladanifer and Cistus palhinhae and their taxonomic implications. Phytochemistry 1984, 23, 470–471. [Google Scholar] [CrossRef]
- Vogt, T.; Proksch, P.; Gültz, P.G. Epicuticular flavonoids in the genus Cistus, Cistaceae. J. Plant Physiol. 1987, 131, 25–36. [Google Scholar] [CrossRef]
- Chaves, N.; Escudero, J.C.; Gutierrez-Merino, C. Seasonal variation of exudate of Cistus ladanifer. J. Chem. Ecol. 1993, 19, 2577–2591. [Google Scholar] [CrossRef] [PubMed]
- Alías, J.C. Influencia de los Factores Climáticos en la Síntesis y Actividad de Compuestos Fitotóxicos Secretados por Cistus ladanifer L. Dissertation, Universidad de Extremadura, Cáceres, España, 2006. [Google Scholar]
- Pascual, T.; Urones, J.G.; Basage, P.; Marcos, I.S.; Montaña, A. Nuevo estudio sobre componentes de Cistus ladanifer L. Stud. Chem. 1984, 31, 31–47. [Google Scholar]
- Chaves, N.; Ríos, J.L.; Gutiérrez, C.; Escudero, J.C.; Alías, J.M. Analysis of secreted flavonoids of Cistus ladanifer L. by high-performance liquid chromatographyparticle beam mass spectrometry. J. Chromatogr. A 1998, 799, 111–115. [Google Scholar] [CrossRef]
- Sosa, T.; Chaves, N.; Alías, J.C.; Escudero, J.C.; Henao, F.; Gutiérrez-Merino, C. Inhibition of mouth skeletal muscle relaxation by flavonoids of Cistus ladanifer L.: A plant defense mechanism against herbivores. J. Chem. Ecol. 2004, 30, 1087–1101. [Google Scholar] [CrossRef] [PubMed]
- Chaves, N.; Escudero, J.C. Variation of flavonoid synthesis induced by ecological factors. In Principles and Practices in Plant Ecology; Inderjit, Dakshini, K.M.M., Foy, C.L., Eds.; Allelochemicals Interactions, CRC Press: Boca Raton, FL, USA, 1999; pp. 267–285. [Google Scholar]
- Chaves, N.; Alías, J.C.; Sosa, T.; Escudero, J.C. Alelopathic potential of Cistus ladanifer chemicals in response to variations of light and temperature. Chemoecology 2002, 12, 139–145. [Google Scholar] [CrossRef]
- Chaves, N.; Escudero, J.C.; Gutiérrez-Merino, C. Role of ecological variables in the seasonal variation of flavonoid content of Cistus ladanifer exudate. J. Chem. Ecol. 1997, 23, 579–603. [Google Scholar] [CrossRef]
- Chaves, N.; Sosa, T.; Alías, J.C.; Escudero, J.C. Germination inhibition of herbs in Cistus ladanifer L. soil: Possible involvement of allelochemicals. Allelopath. J. 2003, 11, 31–42. [Google Scholar]
- Barrajón-Catalán, E.; Fernández-Arroyo, S.; Saura, D.; Guillén, E.; Fernández-Gutiérrez, A.; Segura-Carretero, A. Cistaceae aqueous extracts containing ellagitannins show antioxidant and antimicrobial capacity, and cytotoxic activity against human cancer cells. Food Chem. Toxicol. 2010, 48, 2273–2282. [Google Scholar] [CrossRef] [PubMed]
- Barros, L.; Dueñas, M.; Alves, C.T.; Silva, S.; Henriques, M.; Santos-Buelga, C. Antifungal activity and detailed chemical characterization of Cistus ladanifer phenolic extracts. Ind. Crops Prod. 2013, 41, 41–45. [Google Scholar] [CrossRef]
- Davies, K.M.; Albert, N.W.; Schwinn, K.E. From land in glights tomimicry: The molecular regulation off lower colouration and mechanisms for pigmentation patterning. Funct. Plant Biol. 2012, 39, 619–638. [Google Scholar] [CrossRef]
- Munne’-Bosch, S.; Alegre, L. Subcellular compartmentation of the diterpene carnosic acid and its derivatives in the leaves of rosemary. Plant Physiol. 2001, 125, 1094–1102. [Google Scholar] [CrossRef]
- Krischik, V.A.; Denno, R.F. Individual population and geographic patterns in plant defense. In Variable Plants and Herbivores in Natural and Managed Systems; Denno, R.F., Mcclure, M., Eds.; Academic Press: New York, NY, USA, 1983; pp. 463–512. [Google Scholar]
- Gamble, L.R.; Bergin, T.M. Western kingbird (Tyrannus verticalis). In The Birds of North America; Poole, A., Grill, F., Eds.; The Academy of Natural Sciences: Philadelphia, PA, USA; The American Ornithologists’ Union: Washington, DC, USA, 1996. [Google Scholar]
- Hopper, W.; Mahadevan, A. Utilization of catechin and its metabolites by Bradyrhizobium japonicum. Appl. Microbiol. Biotechnol. 1991, 35, 411–415. [Google Scholar] [CrossRef]
- Nilsson, M.C.; Gallet, C.; Wallstedt, A. Temporal variability of phenolics and batatasin-III in Empetrum hermaphroditum leaves over an eight-year period: Interpretations of ecological function. Oikos 1998, 81, 6–16. [Google Scholar] [CrossRef]
- Gallet, C.; Nilsson, M.C.; Zackrisson, O. Phenolic metabolites of ecological significance in Empetrum hermaphroditum leaves and associated humus. Plant Soil 1999, 210. [Google Scholar] [CrossRef]
- Laitinen, M.L.; Julkunen-Tiitto, R.; Tahvanainen, J.; Heinonen, J.; Rousi, M. Variation in birch (Betula pendula) shoot secondary chemistry due to genotype, environment and ontogeny. J. Chem. Ecol. 2005, 31, 697–717. [Google Scholar] [CrossRef] [PubMed]
- Çirak, C.; Radusiene, J.; Ivanauskas, L.; Janulis, V. Variation of bioactive secondary metabolites in Hypericum origanifolium during its phenological cycle. Acta Physiol. Plant 2007, 29, 197–203. [Google Scholar] [CrossRef]
- Covelo, F.; Gallardo, A. Green and senescent leaf phenolics showed spatial autocorrelation in a Quercus robur population in northwestern Spain. Plant Soil 2004, 259, 267–276. [Google Scholar] [CrossRef]
- Karabourniotis, G.; Fasseas, C. The dense indumentum with its polyphenol content may replace the protective role of the epidermis in some young xeromorphic leaves. Can. J. Bot. 1996, 74, 347–351. [Google Scholar] [CrossRef]
- Ehleringer, J. The influence of water stress and temperature on leaf pubescence development in Encelia farinoza. Am. J. Bot. 1982, 69, 670–675. [Google Scholar] [CrossRef]
- Mauseth, J.D. Plant Anatomy; Benjamin/Cummings Publishing Company: Menlo Park, CA, USA, 1988. [Google Scholar]
- Levin, D.A. The role of trichomes in plant defence. Q. Rev. Biol. 1973, 48, 3–15. [Google Scholar] [CrossRef]
- Juniper, B.E.; Jefree, C.E. Plant Surfaces; Edward Arnold: London, UK, 1983. [Google Scholar]
- Wagner, G.J. Secreting glandular trichomes: More than just hairs. Plant Physiol. 1991, 96, 675–679. [Google Scholar] [CrossRef] [PubMed]
- Allen, E.A.; Hoch, H.C.; Steadman, J.R.; Stavely, R.J. Influence of leaf surface features on spore deposition and the epiphytic growth of phytopathogenic fungi. In Microbial Ecology of Leaves; Andrews, J.H., Hirano, S.S., Eds.; Springer-Verlag: New York, NY, USA, 1991; pp. 87–110. [Google Scholar]
- Day, T.A.; Martin, G.; Vogenmann, T.C. Penetration of UV-B radiation in foliage: Evidence that the epidermis behaves as a non-uniform filter. Plant Cell Environ. 1993, 16, 735–741. [Google Scholar] [CrossRef]
- Karabourniotis, G.; Kyparissis, A.; Manetas, Y. Leaf hairs of Olea europaea L protect underlying tissues against ultraviolet-B radiation damage. Environ. Exp. Bot. 1993, 33, 341–345. [Google Scholar] [CrossRef]
- Tattini, M.; Gravano, E.; Pinelli, P.; Mulinacci, N.; Romani, A. Flavonoids accumulate in leaves and glandular trichomes of Phillyrea latifolia exposed to excess solar radiation. New Phytol. 2000, 148, 69–77. [Google Scholar] [CrossRef]
- Werker, E. Trichome diversity and development. Adv. Bot. Res. 2000, 31. [Google Scholar]
- Maffei, M.; Chialva, F.; Sacco, T. Glandular trichomes and essential oils in developing peppermint leaves. New Phytol. 1989, 111, 707–716. [Google Scholar] [CrossRef]
- Werker, E.; Putievsky, E.; Ravid, U.; Dudai, N.; Katzir, I. Glandular hairs, secretory cavities, and the essential oil in developing leaves of Ocimum basilicum L. (Lamiaceae). Ann. Bot. Lond. 1993, 71, 43–50. [Google Scholar] [CrossRef]
- Alías, J.C.; Sosa, T.; Valares, C.; Escudero, J.C.; Chaves, N. Seasonal variation of Cistus ladanifer L. diterpenes. Plants 2012, 1, 6–15. [Google Scholar] [CrossRef]
- Riipi, M.; Haukioja, E.; Lempa, K.; Ossipov, V.; Ossipova, S.; Pihlaja, K. Ranking of individual mountain birch trees in terms of leaf chemistry: Seasonal and annual variation. Chemoecology 2004, 14, 31–43. [Google Scholar] [CrossRef]
- Brennan, R.M.; Robertson, G.W.; McNicol, J.W.; Fyffe, L.; Hall, J.E. The use of metabolic profiling in the identification of gall mite (Cecidophyopsis ribis Westw.)—Resistant blackcurrant (Ribes nigrum L.) genotypes. Ann. Appl. Biol. 1992, 121, 503–509. [Google Scholar] [CrossRef]
- Brignolas, F.; Lieutier, F.; Sauvard, D.; Christiansen, E.; Berryman, A.A. Phenolic predictors for Norway spruce resistance to the bark beetle ipsipographus (Coleoptera: Scolytidae) and an associated fungus, Ceratocystis polonica. Can. J. For. Res. 1998, 28, 720–728. [Google Scholar] [CrossRef]
- Smith, M. Plant Resistance to Arthropods: Molecular and Conventional Approaches; Springer: Berlin, Germany, 2005. [Google Scholar]
- Ranger, C.M.; Singh, A.P.; Johnson-Cicalese, J.; Polavarapu, S.; Vorsa, N. Intraspecific variation in aphid resistance and constitutive phenolics exhibited by the wild blueberry Vaccinium darrowi. J. Chem. Ecol. 2007, 33, 711–729. [Google Scholar] [CrossRef] [PubMed]
- Lattanzio, V.; Arpaia, S.; Cardinali, A.; Di Venere, D.; Linsalata, V. Role of endogenous flavonoids in resistance mechanism of Vigna to aphids. J. Agric. Food Chem. 2000, 48, 5316–5320. [Google Scholar] [CrossRef] [PubMed]
- Gould, K.S.; Markham, K.R.; Smith, R.H.; Goris, J.J. Functional role of anthocyanins in the leaves of Quintinia serrata A. Cunn. J. Exp. Bot. 2000, 51, 1107–1115. [Google Scholar] [CrossRef] [PubMed]
- Tattini, M.; Landi, M.; Brunetti, C.; Giordano, C.; Remorini, D.; Gould, K.S. Epidermal coumaroyl anthocyanins protects weet basil against excess Light stress: Multiple consequence so flight attenuation. Physiol. Plant 2014, 152, 585–598. [Google Scholar] [CrossRef] [PubMed]
- Jansen, M.A.; Gaba, V.; Greenberg, B.M. Higher plants and UV- B radiation: Balancing damage, repair and acclimation. Trends Plant Sci. 1998, 3, 131–135. [Google Scholar] [CrossRef]
- Halliwell, B. Oxidative dange, lipid peroxidation and antioxidant protection in chloroplasts. Chem. Phys. Lipids 1987, 44, 327–340. [Google Scholar] [CrossRef]
- Iturbe-Ormaetxe, I.; Escuredo, P.R.; Arrese-Igor, C.; Becana, M. Oxidative dange in pea plants exposed to wáter déficit or paraquat. J. Plant Physiol. 1998, 116, 173–181. [Google Scholar] [CrossRef]
- Perl-Treves, R.; Perl, A. Oxidative stress: An introduction. In Oxidative Stress in Plants; Inzé, D., Van Montagu, M., Eds.; Taylor and Francis: London, UK, 2002; pp. 1–32. [Google Scholar]
- Arbona, V.; Flors, V.; Jacas, J.; García-Agustín, P.; Gomez-Cadenas, A. Enzymatic and no enzymatic antioxidant responses of Carrizo citrange, a salt-sensitive citrus rootstock, to different levels of salinity. Plant Cell Physiol. 2003, 44, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi-Shinozahi, K.; Shinozaki, K. Transcripcional regulatory networks in celular responses and tolerance to dehydration and cold stresses. Annu. Rev. Plant Biol. 2006, 57, 781–803. [Google Scholar] [CrossRef] [PubMed]
- Ballaré, C.L.; Caldwell, M.M.; Flint, S.D.; Robinson, S.A.; Bornman, J.F. Effects of solar ultravioleta radiation on terrestrial ecosystems. Patterns, mechanisms, and interactions with climate change. Photochem. Photobiol. Sci. 2011, 10, 226–241. [Google Scholar] [CrossRef] [PubMed]
- Lichtenthaler, H.K.; Schwender, J.; Disch, A.; Rohmer, M. Biosynthesis of isoprenoids in higher plant chloroplasts proceeds via a mevalonate-independent pathway. FEBS Lett. 1997, 400, 271–274. [Google Scholar] [CrossRef]
- Haraguchi, H.; Saito, T.; Okamura, N.; Yagi, A. Inhibition of lipid peroxidation and superoxide generation by diterpenoids from Rosmarinus officinalis. Planta Med. 1995, 61, 333–336. [Google Scholar] [CrossRef] [PubMed]
- Munne’-Bosch, S.; Schwarz, K.; Alegre, L. Enhanced formation of a-tocopherol and highly oxidized abietane diterpenes in water-stressed rosemary plants. Plant Physiol. 1999, 121, 1047–1052. [Google Scholar] [CrossRef]
- Munne’-Bosch, S.; Alegre, L. Changes in carotenoids, tocopherols and diterpenes during drought and recovery, and the biological significance of chlorophyll loss in Rosmarinus officinalis plants. Planta 2000, 210, 925–931. [Google Scholar] [CrossRef]
- Bryant, J.; Julkunen-Tiitto, R. Ontogenetic development of chemical defense by seedling resin birch: Energy cost of defense production. J. Chem. Ecol. 1995, 21, 883–896. [Google Scholar] [CrossRef] [PubMed]
- Fico, G.; Bilia, A.R.; Morelli, I.; Tome, F. Flavonoid distribution in Pyracantha coccinea plants at different growth phases. Biochem. Syst. Ecol. 2000, 28, 673–678. [Google Scholar] [CrossRef]
- Valares, C. Variación Del Metabolismo Secundario En Plantas Debida Al Genotipo Y Al Ambiente. Ph.D. Thesis, Facultad de Ciencias, Universidad de Extremadura, Badajoz, España, 2011. [Google Scholar]
- Cabezas, J.; Escudero, J.C. Estudio Termométrico de la Provincia de Badajoz; Dirección General de Investigación, Extensión y Capacitación Agraria: Badajoz, España, 1989. [Google Scholar]
- Cabezas, J.; Nuñez, E.; Escudero, J.C.; Marroquin, A. Estudio Pluviométrico de la Provincia de Badajoz; Consejeria de Agricultura y Comercio: Badajoz, Spain, 1983. [Google Scholar]
- Patón, D.; Azocar, P.; Tovar, J. Growth and productivity in forage biomass in relation to the age assessed by dendrochronology in the evergreen shrub Cistus ladanifer L. using different regression models. J. Arid. Environ. 1998, 38, 221–235. [Google Scholar] [CrossRef]
- Hostettmamm, K.; Domon, B.; Schaufelberger, D.; Hostettmann, M. On Line High-Performance liquid chromatography: Ultraviolet visible spectroscopy of phenolic compounds in plant extrats using post column derivatization. J. Chromatogr. 1984, 283, 137–147. [Google Scholar]
- Markham, K.R. Techniques of Flavonoids Identification; Academic Press: London, UK, 1982. [Google Scholar]
- Chaves, N. Variación Cualitativa y Cuantitativa de los Flavonoides del Exudado de Cistus ladanifer L. como Respuesta a Diferentes Factores Ecológicos. Dissertation, Universidad de Extremadura, Badajoz, España, 1994. [Google Scholar]
- Vogth, T.; Gülz, P.G. Isocratic column liquid chromatographic separation of a complex mixture of epicuticular flavonoid aglycones and intracellelar flavonol glycosides from Cistus laurifolius L. J. Chromatogr. 1991, 537, 453–459. [Google Scholar] [CrossRef]
- Sosa, T.; Valares, C.; Alías, J.; Chaves, N. Persistence of flavonoids in Cistus ladanifer soils. Plant Soil 2010, 337, 51–63. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are available from the authors.
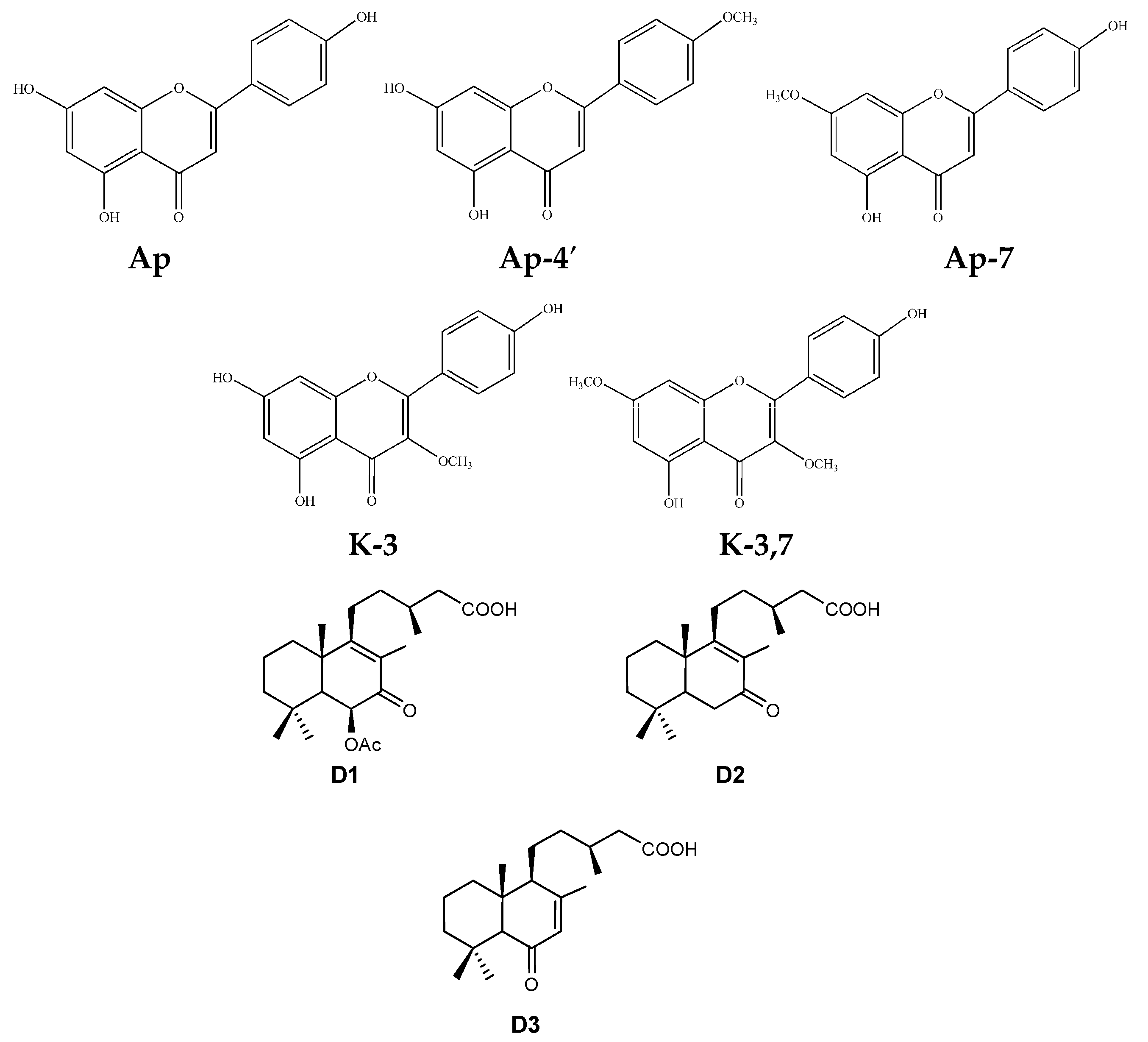
| Compounds | Summer | Autumn | Winter | Spring | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Young Leaves | Mature Leaves | Stems | Young Leaves | Mature Leaves | Stems | Young Leaves | Mature Leaves | Stems | Young Leaves | Mature Leaves | Stems | |
| Flavonoids | ||||||||||||
| Ap | 0.12(0.04) a | 0.08(0.03) b | 0.11(0.06) a,b | 0.15(0.11) a | 0.07(0.02) b | 0.16(0.07) a | 0.10(0.05) a | 0.03(0.01) b | 0.11(0.06) a | 0.08(0.04) a | 0.05(0.02) b | 0.15(0.15) a |
| K-3 | 1.95(0.95) a | 1.09(0.51) b | 1.25(0.86) c | 1.66(1.08) a | 0.91(0.46) b | 2.02(0.96) c | 0.75(0.53) a | 0.27(0.20) b | 0.80(0.52) a | 0.83(0.45) a | 0.32(0.16) b | 1.13(0.90) c |
| Ap-4′ | 1.37(0.28) a | 0.78(0.19) b | 1.06(0.43) c | 2.06(1.03) a | 0.69(0.16) b | 1.30(0.66) c | 1.28(0.25) a | 0.56(0.11) b | 1.48(0.36) c | 1.01(0.42) a | 0.75(0.25) b | 1.79(0.81) c |
| Ap-7 | 2.49(0.58) a | 1.41(0.24) b | 1.42(0.54) b | 2.56(1.12) a | 1.21(0.51) b | 1.68(0.69) c | 1.46(0.33) a | 0.67(0.13) b | 1.40(0.64) a | 1.43(0.49) a | 1.07(0.38) b | 1.64(0.75) c |
| K-3.7 | 20.72(6.62) a | 10.82(3.30) b | 6.68(3.50) c | 9.25(4.01) a | 7.85(2.42) a | 7.46(3.40) a | 3.22(1.64) a | 2.91(1.29) b | 3.64(2.29) c | 5.95(2.66) a | 3.27(1.09) b | 4.48(3.59) c |
| Total | 26.65(7.90) a | 13.99(3.86) b | 9.90(5.25) c | 15.59(5.09) a | 10,71(2.99) b | 12.58(5.29) a,b | 6.78(2.10) a | 4.45(1.59) b | 7.51(3.05) a | 9.29(3.67) a | 5.45(1.41) b | 9.18(4.85) a |
| Diterpenes | ||||||||||||
| D1 | 1.44(0.43) a | 0.75(0.24) b | 0.53(0.26) c | 3.03(1.61) a | 0.48(0.19) b | 0.66(0.40) c | 2.14(1.03) a | 0.42(0.33) b | 1.21(1.45) c | 1.56(0.62) a | 0.57(0.32) b | 1.15(0.60) c |
| D2 | 0.08(0.03) a | 0.04(0.03) b | 0.02(0.01) c | 0.17(0.16) a | 0.02(0.01) b | 0.03(0.01) b | 0.12(0.07) a | 0.02(0.01) b | 0.06(0.05) c | 0.09(0.03) a | 0.03(0.02) b | 0.05(0.03) b |
| D3 | 0.04(0.02) a | 0.02(0.02) b | 0.02(0.02) b | 0.13(0.06) a | 0.02(0.01) b | 0.04(0.02) c | 0.35(0.28) a | 0.05(0.04) b | 0.13(0.10) c | 0.09(0.04) a | 0.03(0.02) b | 0.07(0.05) c |
| Total | 1.56(0.46) a | 0.81(0.26) b | 0.56(0.30) c | 3.32(1.75) a | 0.52(0.20) b | 0.72(0.43) c | 2.60(1.11) a | 0.48(0.36) b | 1.46(1.55) c | 1.73(0.70) a | 0.62(0.41) b | 1.26(0.66) c |
| Compounds | Age | <1 | 2–6 | 7–11 | 12–16 | 17–21 | 22–26 | 27–31 | >31 |
|---|---|---|---|---|---|---|---|---|---|
| Flavonoids (mg/g dw) | |||||||||
| Ap | Young leaves | 0.05 (0.01) a | 0.10 (0.07) b | 0.15 (0.09) c | 0.09 (0.02) b,c | 0.23 (0.07) c,d | 0.30 (0.02) d | 0.12 (0.04) b,c | 0.09 (0.02) b,c |
| Madures leaves | 0.04 (0.02) a | 0.06 (0.02) b | 0.04 (0.01) a,b | 0.06 (0.04) a,b | 0.06 (0.01) a,b | 0.06 (0.01) a,b | 0.03 (0.00) a | ||
| Stems | 0.17 (0.07) a,b | 0.17 (0.05) a | 0.12 (0.06) b | 0.15 (0.06) a,b | 0.17 (0.01) a | 0.19 (0.04) a | 0.21 (0.01) a | ||
| K-3 | Young leaves | 1.64 (0.69) a | 3.32 (1.50) b | 3.66 (1.07) b | 2.84 (0.90) b | 4.78 (1.99) b,c | 5.07(0.36) c | 2.72 (0.78) b | 2.23 (0.44) b,c |
| Madures leaves | 1.69 (0.68) a | 2.02 (1.08) a | 1.43 (0.31) a | 2.13 (1.50) a | 2.24 (0.22) a | 1.85 (0.10) a | 1.18 (0.32) a | ||
| Stems | 4.43 (1.41) a,b | 5.25 (1.66) a,b | 3.92 (2.07) a | 4.68 (2.22) a,b | 6.05 (0.42) b | 4.91 (0.90) a,b | 4.77 (0.76) a,b | ||
| Ap-4′ | Young leaves | 0.87 (0.39) a | 1.24 (0.46) b | 1.20 (0.30) b | 1.29 (0.04) b | 1.39 (0.77) a | 1.99 (0.52) b | 1.07(0.29) a,b | 0.98 (0.09) a,b |
| Madures leaves | 0.75 (0.25) a | 0.61 (0.07) a,b | 0.64 (0.11) a,b | 0.77 (0.32) a | 0.77 (0.22) a | 0.65 (0.08) a,b | 0.54 (0.06) b | ||
| Stems | 1.77 (o.46) a | 1.45 (0.20) b | 1.50 (0.35) b | 1.64 (0.32) a,b | 2.20 (0.58) c | 1.98 (0.39) a,c | 1.60 (0.33) a,b | ||
| Ap-7 | Young leaves | 1.13 (0.52) a | 1.83 (0.56) b | 1.81 (0.46) b | 1.39 (0.26) a | 1.94 (0.97) a,b | 2.20 (0.72) a,b | 1.47 (0.17) a | 1.50 (0.49) a,b |
| Madures leaves | 1.24 (0.44) a | 1.16 (0.37) a | 0.96(0.06) a | 1.24 (0.47) a | 1.29 (0.45) a | 1.19 (0.15) a | 0.88 (0.35) a | ||
| Stems | 1.51 (0.39 ) a | 1.40 (0.13) a | 1.27 (0.18) a | 1.45 (0.21) a | 1.67 (0.66) a | 1.69 (0.02) a | 1.50 (0.59) a | ||
| K-3.7 | Young leaves | 2.79 (1.58) a | 1.67 (3.21) b | 6.65 (1.40) b | 6.44 (1.14) b | 6.20 (1.78) b | 5.59 (1.01) b | 4.84 (0.54) b | 4.07 (0.36) a,b |
| Madures leaves | 4.77 (1.09) a | 5.64 (1.80) a | 5.53 (1.86) a | 5.17 (1.25) a | 5.75 (0.98) a | 5.09 (1.00) a | 3.31 (0.59) b | ||
| Stems | 4.12 (1.39) a | 5.26 (2.35) a | 4.78 (2.14) a | 4.00 (1.29) a | 4.94 (0.89) a | 5.22 (0.48) a | 3.24 (0.89) a | ||
| Diterpenes (mg/g dw) | |||||||||
| D1 | Young leaves | 0.47 (0.43) a | 0.80 (0.32) b | 0.71 (0.25) a,b | 0.87 (0.33) b | 0.91 (0.68) b | 1.50 (0.46) c | 0.76 (0.12) a,b | 0.41 (0.11) a |
| Madures leaves | 0.24 (0.14) a | 0.14 (0.08) b,c | 0.15 (0.05) a,b,c | 0.22 (0.10) a,b | 0.19 (0.05) a,b | 0.22 (0.09) a,b | 0.08 (0.02) c | ||
| Stems | 0.43 (0.15) a | 0.42 (0.17) a,b | 0.52 (0.31) a,b | 0.64 (0.18) b | 0.92 (0.36) c | 0.80 (0.05) c | 0.37 (0.13) a | ||
| D2 | Young leaves | 0.23 (0.19) a | 0.58 (0.25) b | 0.66 (0.18) b | 0.63 (0.28) b | 0.61 (0.62) a,b | 1.07 (0.34) c | 0.52 (0.01) b | 0.43 (0.21) b |
| Madures leaves | 0.11 (0.05) a | 0.09 (0.05) a,b | 0.06 (0.03) b | 0.09 (0.04) a,b | 0.09 (0.03) a | 0.12 (0.00) a | 0.06 (0.00) b | ||
| Stems | 0.20 (0.11) a | 0.38 (0.15) b | 0.34 (0.10) b | 0.38 (0.24) b | 0.55 (0.31) c | 0.45 (0.06) b,c | 0.42 (0.12) b,c | ||
| D3 | Young leaves | 0.35 (0.17) a | 0.88 (048) b | 1.27 (0.74) b | 0.66 (0.21) b | 0.52 (0.15) a,b | 0.74 (0.37) b | 0.37 (0.16) a | 0.70 (0.31) b |
| Madures leaves | 0.16 (0.09) a | 0.16 (0.11) a,b | 0.06 (0.02) b,c | 0.12 (0.07) a,b | 0.08 (0.03) b,c | 0.07 (0.06) b,c | 0.04 (0.00) c | ||
| Stems | 0.47 (0.23) a | 1.01 (0.71) b | 0.52 (0.28) a,b | 0.55 (0.35) a,b | 0.64 (0.25) a,b | 0.57 (0.19) a,b | 0.53 (0.24) a,b | ||
| Climatic Parameters | Sprint | Summer | Autumn | Winter |
|---|---|---|---|---|
| P (mm) | 46.7 | 11.8 | 359.4 | 155.7 |
| Tmax (°C) | 25.8 | 35.3 | 16.5 | 15.3 |
| Tmin (°C) | 11.4 | 17.4 | 8 | 5.2 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valares Masa, C.; Sosa Díaz, T.; Alías Gallego, J.C.; Chaves Lobón, N. Quantitative Variation of Flavonoids and Diterpenes in Leaves and Stems of Cistus ladanifer L. at Different Ages. Molecules 2016, 21, 275. https://doi.org/10.3390/molecules21030275
Valares Masa C, Sosa Díaz T, Alías Gallego JC, Chaves Lobón N. Quantitative Variation of Flavonoids and Diterpenes in Leaves and Stems of Cistus ladanifer L. at Different Ages. Molecules. 2016; 21(3):275. https://doi.org/10.3390/molecules21030275
Chicago/Turabian StyleValares Masa, Cristina, Teresa Sosa Díaz, Juan Carlos Alías Gallego, and Natividad Chaves Lobón. 2016. "Quantitative Variation of Flavonoids and Diterpenes in Leaves and Stems of Cistus ladanifer L. at Different Ages" Molecules 21, no. 3: 275. https://doi.org/10.3390/molecules21030275
APA StyleValares Masa, C., Sosa Díaz, T., Alías Gallego, J. C., & Chaves Lobón, N. (2016). Quantitative Variation of Flavonoids and Diterpenes in Leaves and Stems of Cistus ladanifer L. at Different Ages. Molecules, 21(3), 275. https://doi.org/10.3390/molecules21030275






