Tomato Aqueous Extract Modulates the Inflammatory Profile of Immune Cells and Endothelial Cells
Abstract
:1. Introduction
2. Results
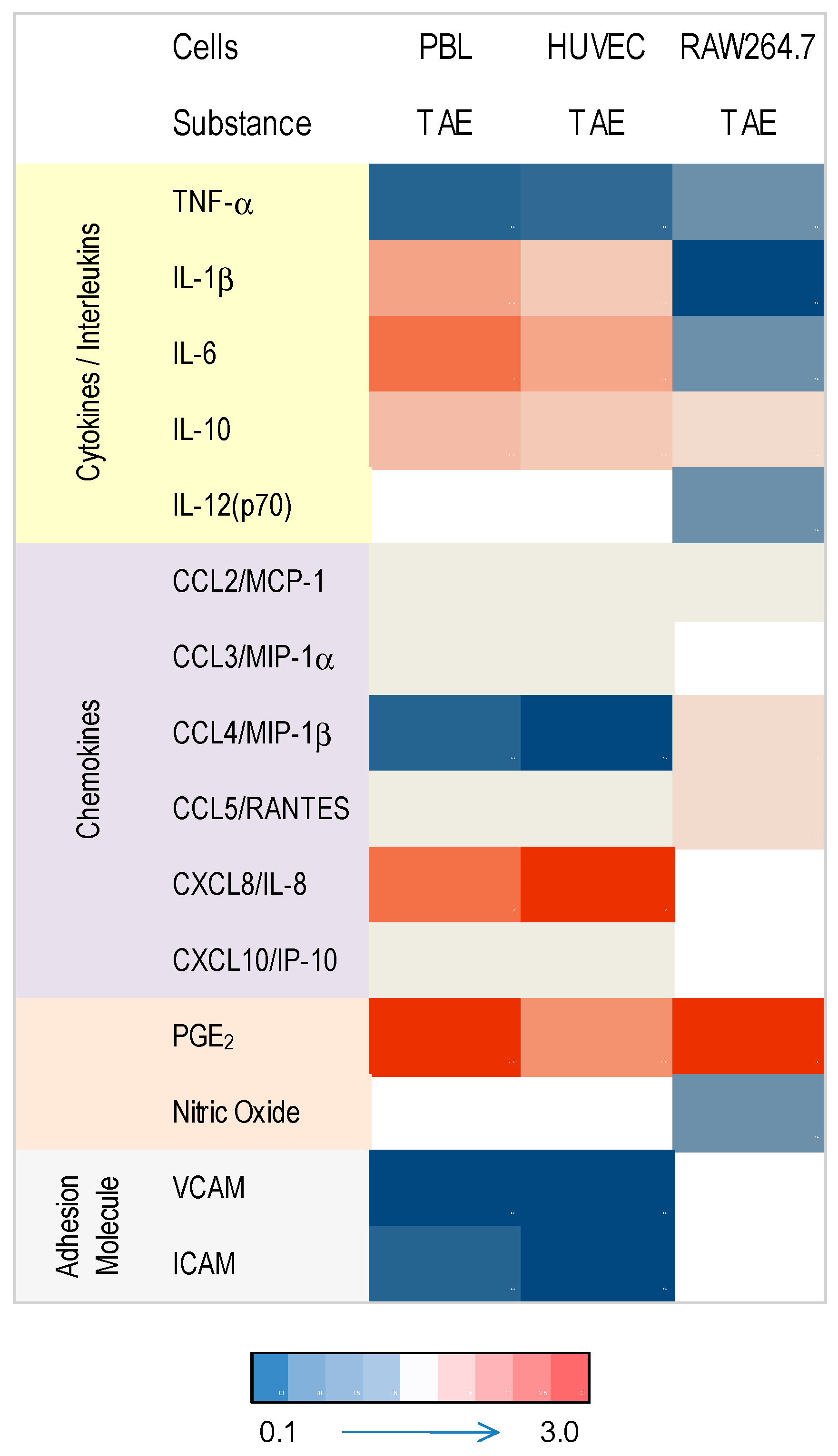
2.1. Tomato Aqueous Extract Changed the Inflammatory Profile of Murine RAW264.7 Cells
| Metabolite | LPS Stimulated | TAE (500 μg/mL) +LPS | p | Adenosine (25 μM) +LPS | p | Chlorogenic Acid (25 μM) +LPS | p | Rutin (25 μM) +LPS | p |
|---|---|---|---|---|---|---|---|---|---|
| PGE2 [pg/mL] | 7034 ± 186 | 5234 ± 260 | 0.09 | 6373 ± 44 | 0.23 | 12,016 ± 574 | 0.05 | 12,940 ± 1882 | 0.07 |
| Nitric oxide (μM) | 18.1 ± 0.2 | 13.1 ± 0.8 | 0.05 | 19.6 ± 0.9 | 0.94 | 12.2 ± 0.1 | 0.02 | 14.4 ± 0.2 | 0.04 |
| IL-6 [ng/mL] | 33.6 ± 2.4 | 36.9 ± 1.8 | 0.26 | 24.1 ± 2.1 | 0.05 | 27.0 ± 1.7 | 0.08 | 25.7 ± 2.0 | 0.07 |
| IL-1β [pg/mL] | 702 ± 120 | 579 ± 105 | 0.39 | 625 ± 78 | 0.52 | 567 ± 32 | 0.26 | 498 ± 22 | 0.14 |
| IL-12 [pg/mL] | 1083 ± 165 | 1045 ± 49 | 0.79 | 1150 ± 127 | 0.69 | 1058 ± 127 | 0.69 | 921 ± 35 | 0.31 |
| TNF-α [ng/mL] | 293 ± 33 | 290 ± 12 | 0.93 | 202 ± 5 | 0.07 | 241 ± 21 | 0.23 | 292 ± 10 | 0.98 |
| CCL2/MCP-1 [ng/mL] | 9.8 ± 1.9 | 14.9 ± 3.7 | 0.22 | 6.4 ± 2.9 | 0.30 | 8.8 ± 0.4 | 0.53 | 6.7 ± 1.7 | 0.22 |
| CCL4/MIP-1β [pg/mL] | 792 ± 50 | 1385 ± 63 | 0.01 | 753 ± 31 | 0.45 | 698 ± 36 | 0.16 | 793 ± 33 | 0.99 |
| CCL5/RANTES [ng/mL] | 34.1 ± 0.4 | 58.7 ± 1.5 | 0.002 | 28.6 ± 3.1 | 0.13 | 27.7 ± 0.8 | 0.01 | 28.4 ± 2.1 | 0.06 |
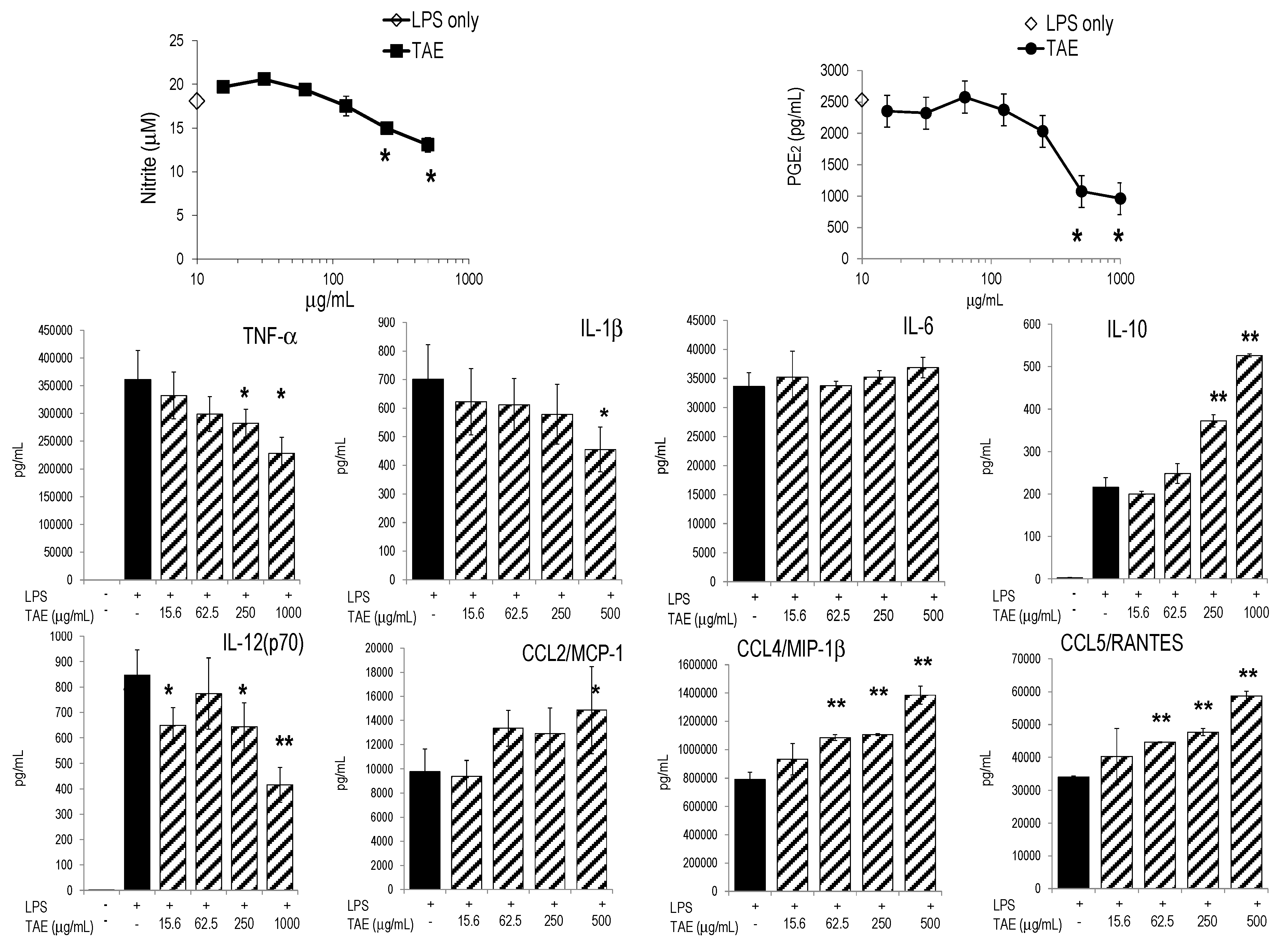
| Constituent | (In %) 1 | (μM) |
|---|---|---|
| Adenosine | 1.73 | 64.8 |
| Rutin | 0.592 | 0.96 |
| Chlorogenic Acid | 0.182 | 0.52 |
| Vitamin C | <LOD 2 | |
| Vitamin E | <LOD | |
| Lycopene | <LOD |
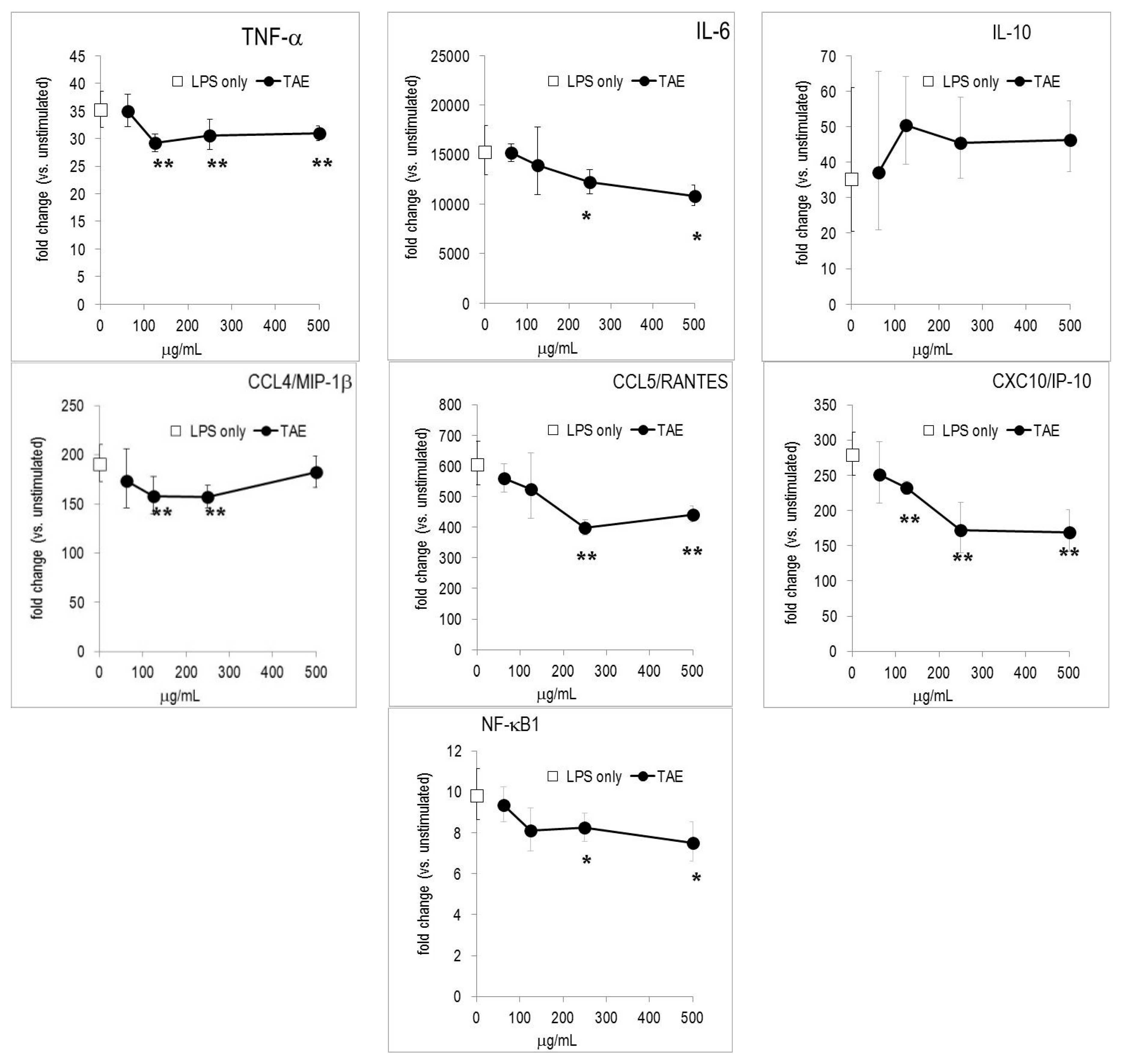
2.2. TAE Altered Inflammatory Response of Peripheral Blood Leukocytes Activated by LPS
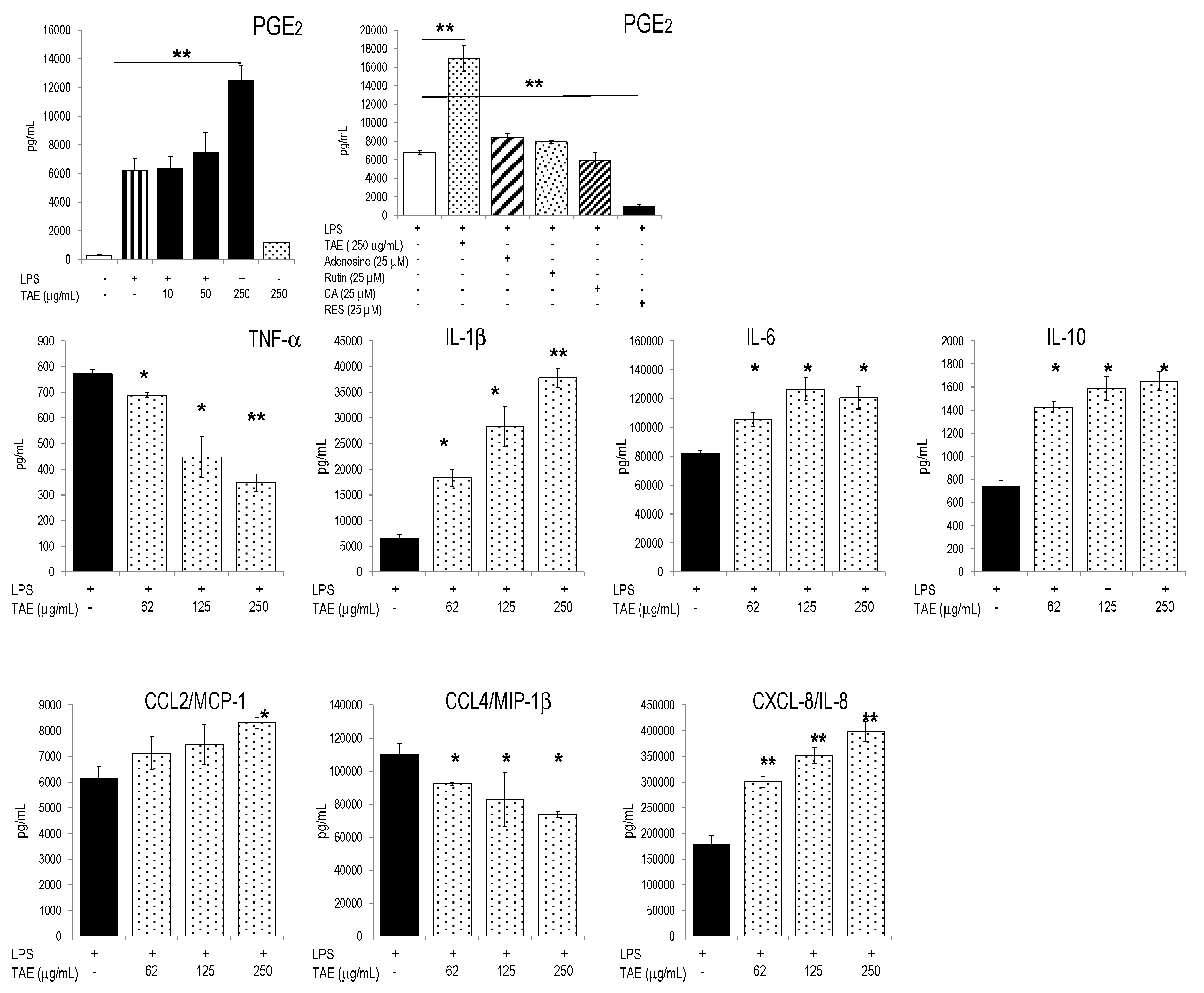
2.3. TAE Modifies Chemokine and Cytokine Gene Transcription in PBLs
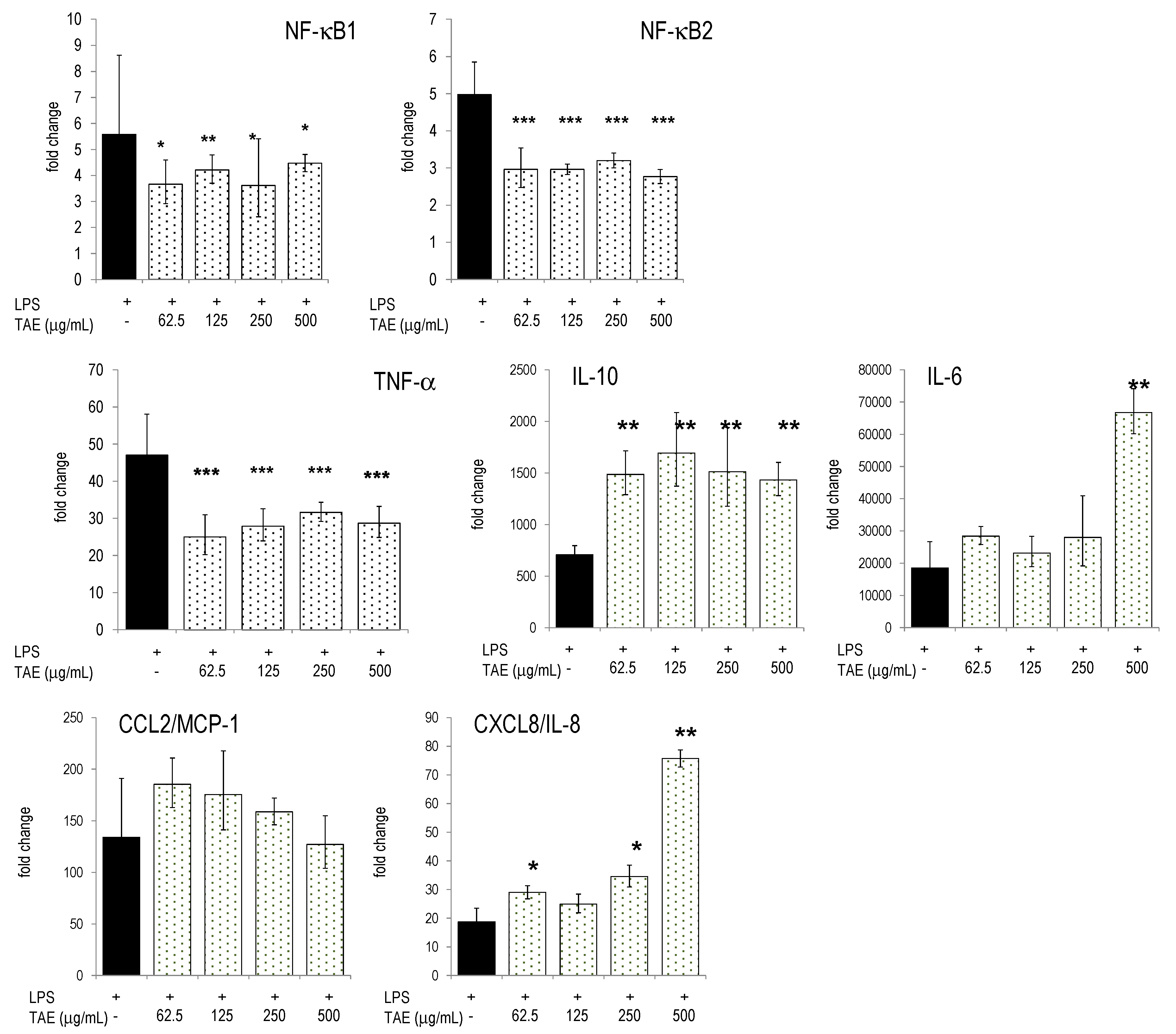
2.4. Modulation of Inflammatory Parameters Associated with Endothelial Dysfunction
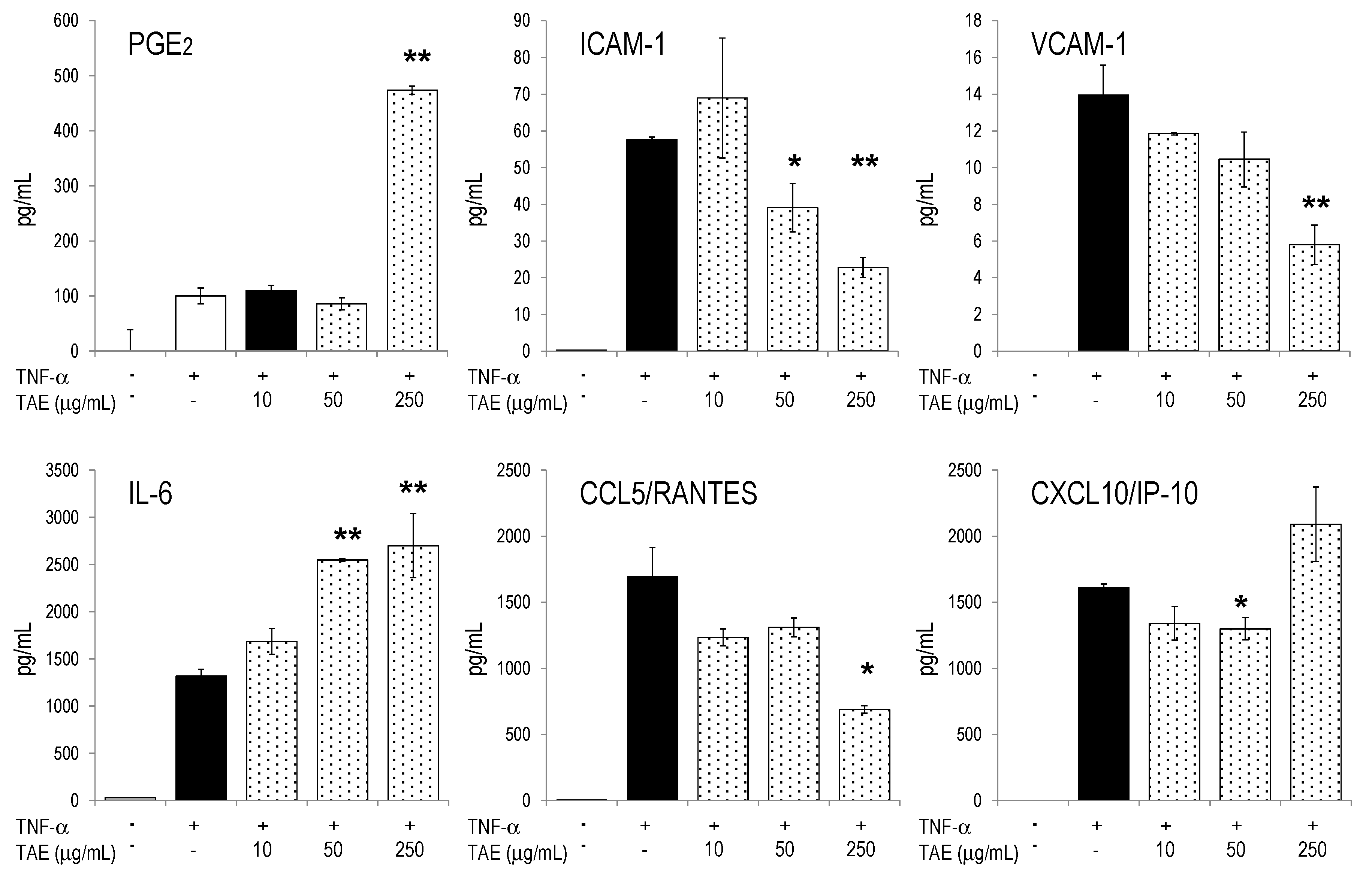
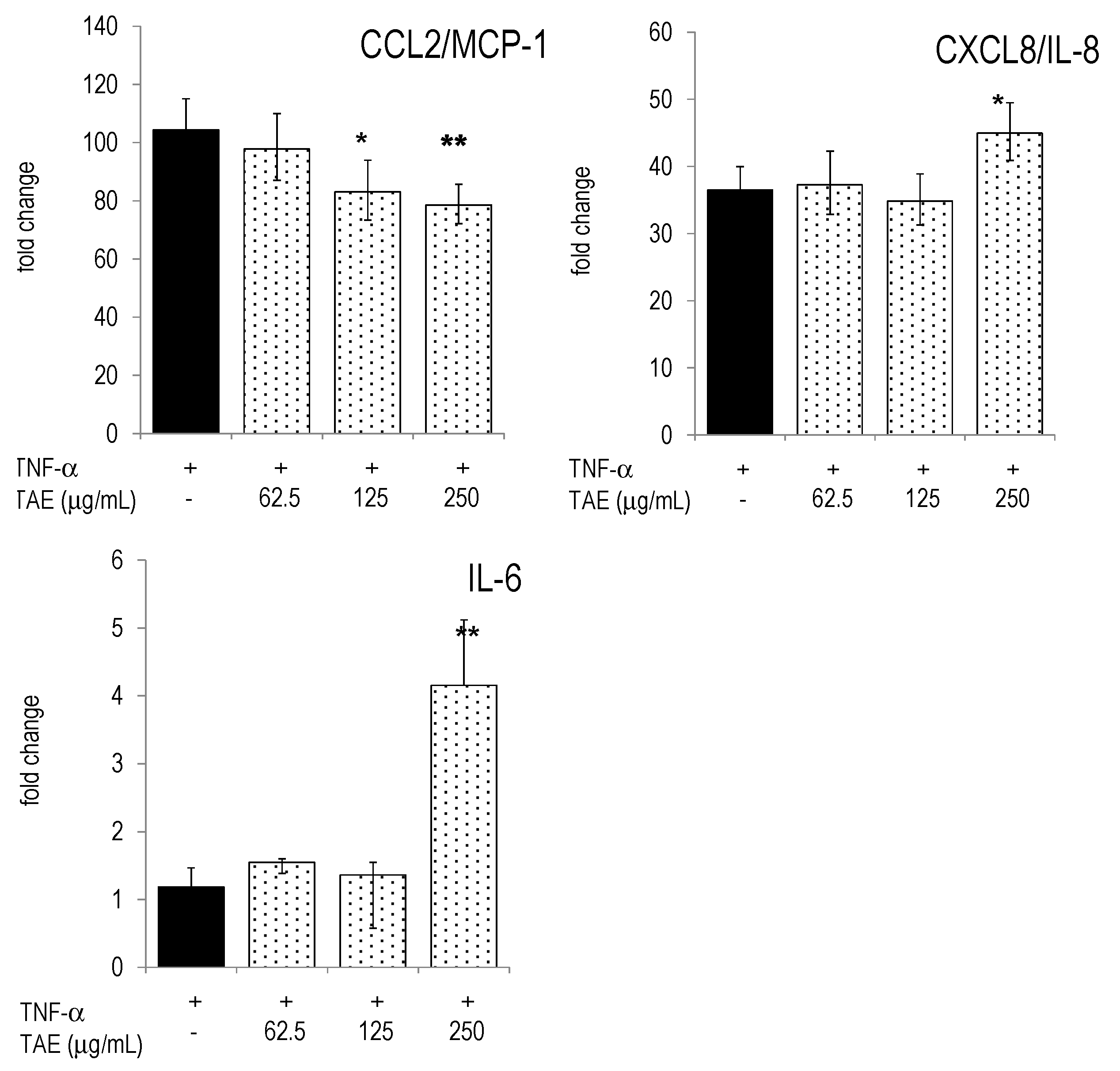
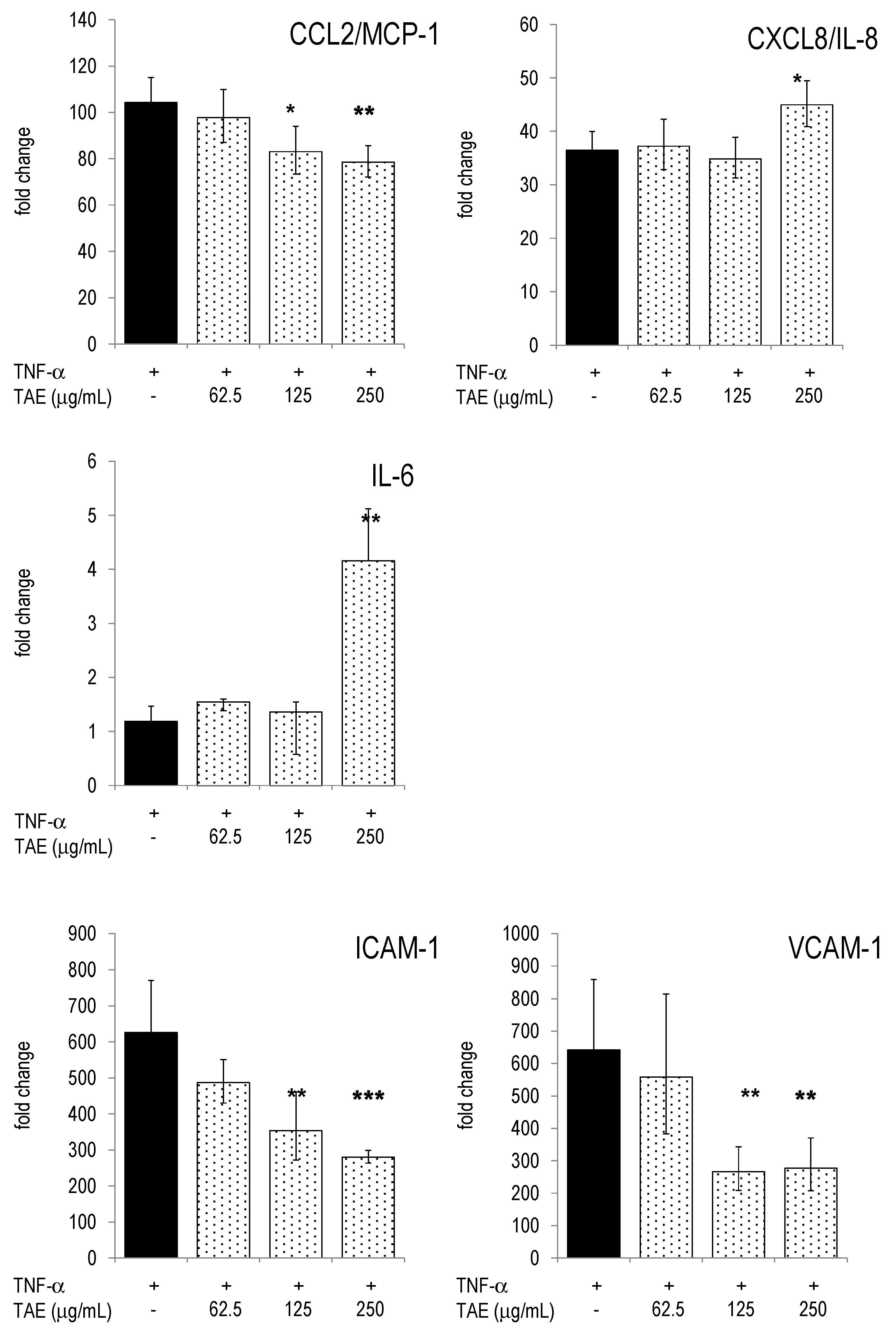
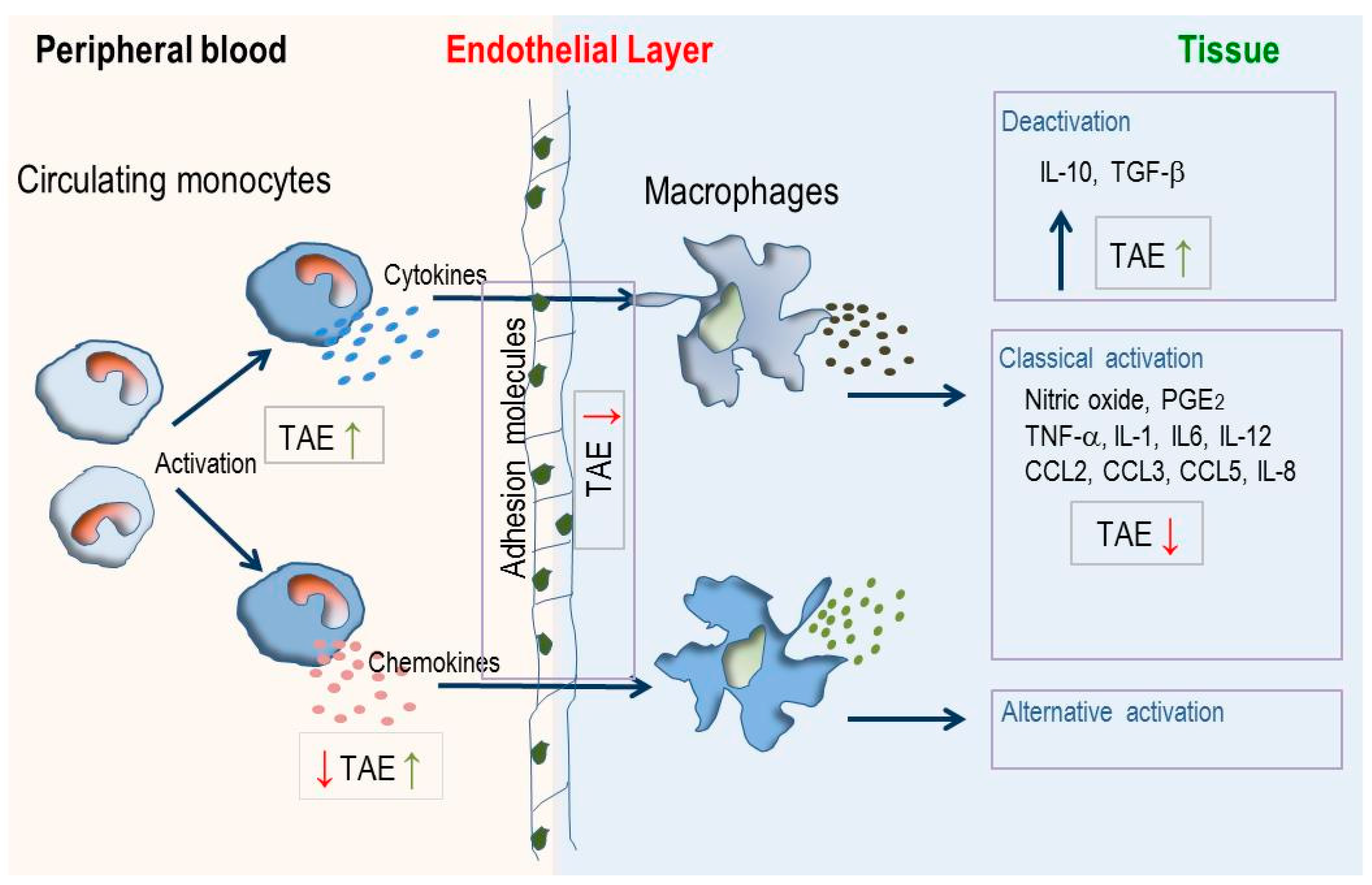
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Cell Culture
4.3. RNA Isolation, cDNA Synthesis and RT-PCR
4.4. Multiparametricanalysis of Cytokines, Chemokines and Interleukins
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| CA | chlorogenic acid |
| CK | chemokine |
| ED | endothelial dysfunction |
| HUVECs | human umbilical vein endothelial cells |
| IL | interleukin |
| NO | nitric oxide |
| PGE2 | prostaglandin E2 |
| PBLs | peripheral blood leukocytes |
| TAE | tomato aqueous extract |
| TF | transcription factor |
References
- Hotamisligil, G.S.; Erbay, E. Nutrient sensing and inflammation in metabolic diseases. Nat. Rev. Immunol. 2008, 8, 923–934. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N.; Savill, J. Resolution of inflammation: The beginning programs the end. Nat. Immunol. 2005, 6, 1191–1197. [Google Scholar] [CrossRef] [PubMed]
- Chawla, A.; Nguyen, K.D.; Goh, Y.P. Macrophage-mediated inflammation in metabolic disease. Nat. Rev. Immunol. 2011, 11, 738–749. [Google Scholar] [CrossRef] [PubMed]
- Bakker, G.C.; van Erk, M.J.; Pellis, L.; Wopereis, S.; Rubingh, C.M.; Cnubben, N.H.; Kooistra, T.; van Ommen, B.; Hendriks, H.F. An antiinflammatory dietary mix modulates inflammation and oxidative and metabolic stress in overweight men: A nutrigenomics approach. Am. J. Clin. Nutr. 2010, 91, 1044–1059. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, E.J.; Astudillo, L.A.; Gutierrez, M.I.; Contreras, S.O.; Bustamante, L.O.; Rubio, P.I.; Moore-Carrasco, R.; Alarcon, M.A.; Fuentes, J.A.; Gonzalez, D.E.; et al. Fractions of aqueous and methanolic extracts from tomato (Solanum lycopersicum L.) present platelet antiaggregant activity. Blood Coagul. Fibrinolysis 2012, 23, 109–117. [Google Scholar] [CrossRef] [PubMed]
- O’Kennedy, N.; Crosbie, L.; van Lieshout, M.; Broom, J.I.; Broom, J.I.; Webb, D.J.; Duttaroy, A.K. Effects of antiplatelet components of tomato extract on platelet function in vitro and ex vivo: A time-course cannulation study in healthy humans. Am. J. Clin. Nutr. 2006, 84, 570–579. [Google Scholar] [PubMed]
- Rodriguez-Azua, R.; Treuer, A.; Moore-Carrasco, R.; Cortacans, D.; Gutierrez, M.; Astudillo, L.; Fuentes, E.; Palomo, I. Effect of tomato industrial processing (different hybrids, paste, and pomace) on inhibition of platelet function in vitro, ex vivo, and in vivo. J. Med. Food 2014, 17, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Bessler, H.; Salman, H.; Bergman, M.; Alcalay, Y.; Djaldetti, M. In vitro effect of lycopene on cytokine production by human peripheral blood mononuclear cells. Immunol. Investig. 2008, 37, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Raiola, A.; Rigano, M.M.; Calafiore, R.; Frusciante, L.; Barone, A. Enhancing the health-promoting effects of tomato fruit for biofortified food. Mediat. Inflamm. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Seok, J.; Warren, H.S.; Cuenca, A.G.; Mindrinos, M.N.; Baker, H.V.; Xu, W.; Richards, D.R.; McDonald-Smith, G.P.; Gao, H.; Hennessy, L.; et al. Genomic responses in mouse models poorly mimic human inflammatory diseases. Proc. Natl. Acad. Sci. USA 2013, 110, 3507–3512. [Google Scholar] [CrossRef] [PubMed]
- Takao, K.; Miyakawa, T. Genomic responses in mouse models greatly mimic human inflammatory diseases. Proc. Natl. Acad. Sci. USA 2015, 112, 1167–1172. [Google Scholar] [CrossRef] [PubMed]
- Armoza, A.; Haim, Y.; Bashiri, A.; Wolak, T.; Paran, E. Tomato extract and the carotenoids lycopene and lutein improve endothelial function and attenuate inflammatory NF-kappaB signaling in endothelial cells. J. Hypertens. 2013, 31, 521–529. [Google Scholar] [CrossRef] [PubMed]
- O’Kennedy, N.; Crosbie, L.; Whelan, S.; Luther, V.; Horgan, G.; Broom, J.I.; Webb, D.J.; Duttaroy, A.K. Effects of tomato extract on platelet function: A double-blinded crossover study in healthy humans. Am. J. Clin. Nutr. 2006, 84, 561–569. [Google Scholar] [PubMed]
- Richard, N.; Porath, D.; Radspieler, A.; Schwager, J. Effects of resveratrol, piceatannol, tri-acetoxystilbene, and genistein on the inflammatory response of human peripheral blood leukocytes. Mol. Nutr. Food Res. 2005, 9, 431–442. [Google Scholar] [CrossRef] [PubMed]
- Gasparini, C.; Feldmann, M. NF-kappaB as a target for modulating inflammatory responses. Curr. Pharm. Des. 2012, 18, 5735–5745. [Google Scholar] [CrossRef] [PubMed]
- Biswas, S.K.; Mantovani, A. Macrophage plasticity and interaction with lymphocyte subsets: Cancer as a paradigm. Nat. Immunol. 2010, 11, 889–896. [Google Scholar] [CrossRef] [PubMed]
- Ostertag, L.M.; O’Kennedy, N.; Kroon, P.A.; Duthie, G.G.; de Roos, B. Impact of dietary polyphenols on human platelet function—A critical review of controlled dietary intervention studies. Mol. Nutr. Food Res. 2010, 54, 60–81. [Google Scholar] [CrossRef] [PubMed]
- Furie, M.B.; McHugh, D.D. Migration of neutrophils across endothelial monolayers is stimulated by treatment of the monolayers with interleukin-1 or tumor necrosis factor-alpha. J. Immunol. 1989, 143, 3309–3317. [Google Scholar] [PubMed]
- May, L.T.; Torcia, G.; Cozzolino, F.; Ray, A.; Tatter, S.B.; Santhanam, U.; Sehgal, P.B.; Stern, D. Interleukin-6 gene expression in human endothelial cells: RNA start sites, multiple IL-6 proteins and inhibition of proliferation. Biochem. Biophys. Res. Commun. 1989, 159, 991–998. [Google Scholar] [CrossRef]
- Kim, Y.I.; Mohri, S.; Hirai, S.; Lin, S.; Goto, T.; Ohyane, C.; Sakamoto, T.; Takahashi, H.; Shibata, D.; Takahashi, N.; et al. Tomato extract suppresses the production of proinflammatory mediators induced by interaction between adipocytes and macrophages. Biosci. Biotechnol. Biochem. 2015, 79, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Simone, R.E.; Russo, M.; Catalano, A.; Monego, G.; Froehlich, K.; Boehm, V.; Palozza, P. Lycopene inhibits NF-κB-mediated IL-8 expression and changes redox and PPARgamma signalling in cigarette smoke-stimulated macrophages. PLoS ONE 2011, 6, e19652. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.; Ma, Y.; Liu, D. Rutin suppresses palmitic acids-triggered inflammation in macrophages and blocks high fat diet-induced obesity and fatty liver in mice. Pharm. Res. 2013, 30, 2940–2950. [Google Scholar] [CrossRef] [PubMed]
- Shan, J.; Fu, J.; Zhao, Z.; Kong, X.; Huang, H.; Luo, L.; Yin, Z. Chlorogenic acid inhibits lipopolysaccharide-induced cyclooxygenase-2 expression in RAW264.7 cells through suppressing NF-kappaB and JNK/AP-1 activation. Int. Immunopharmacol. 2009, 9, 1042–1048. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Dong, L.; Jiang, J.; Zhao, J.; Jiang, J.; Wang, Y.; Lu, X.; Guo, X. Chlorogenic acid reduces liver inflammation and fibrosis through inhibition of toll-like receptor 4 signaling pathway. Toxicology 2013, 303, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.H.; Wu, C.F.; Huang, H.W.; Jao, Y.C.; Yen, G.C. Naturally occurring flavonoids attenuate high glucose-induced expression of proinflammatory cytokines in human monocytic THP-1 cells. Mol. Nutr. Food Res. 2009, 53, 984–995. [Google Scholar] [CrossRef] [PubMed]
- Hasko, G.; Cronstein, B. Regulation of inflammation by adenosine. Front. Immunol. 2013, 4, 85. [Google Scholar] [CrossRef] [PubMed]
- Hasko, G.; Kuhel, D.G.; Chen, J.F.; Schwarzschild, M.A.; Deitch, E.A.; Mabley, J.G.; Marton, A.; Szabo, C. Adenosine inhibits IL-12 and TNF-α production via adenosine A2a receptor-dependent and independent mechanisms. FASEB J. 2000, 14, 2065–2074. [Google Scholar] [CrossRef] [PubMed]
- Nemeth, Z.H.; Lutz, C.S.; Csoka, B.; Deitch, E.A.; Leibovich, S.J.; Gause, W.C.; Tone, M.; Pacher, P.; Vizi, E.S.; Hasko, G. Adenosine augments IL-10 production by macrophages through an A2B receptor-mediated posttranscriptional mechanism. J. Immunol. 2005, 175, 8260–8270. [Google Scholar] [CrossRef] [PubMed]
- Barczyk, K.; Ehrchen, J.; Tenbrock, K.; Ahlmann, M.; Kneidl, J.; Viemann, D.; Roth, J. Glucocorticoids promote survival of anti-inflammatory macrophages via stimulation of adenosine receptor A3. Blood 2010, 116, 446–455. [Google Scholar] [CrossRef] [PubMed]
- Hasko, G.; Szabo, C.; Nemeth, Z.H.; Kvetan, V.; Pastores, S.M.; Vizi, E.S. Adenosine receptor agonists differentially regulate IL-10, TNF-alpha, and nitric oxide production in RAW264.7 macrophages and in endotoxemic mice. J. Immunol. 1996, 157, 4634–4640. [Google Scholar] [PubMed]
- Baggiolini, M. Chemokines and leukocyte traffic. Nature 1998, 392, 565–568. [Google Scholar] [CrossRef] [PubMed]
- Phillipson, M.; Kubes, P. The neutrophil in vascular inflammation. Nat. Med. 2011, 17, 1381–1390. [Google Scholar] [CrossRef] [PubMed]
- Hunter, C.A.; Jones, S.A. IL-6 as a keystone cytokine in health and disease. Nat. Immunol. 2015, 16, 448–457. [Google Scholar] [CrossRef] [PubMed]
- Hassanian, S.M.; Dinarvand, P.; Rezaie, A.R. Adenosine regulates the proinflammatory signaling function of thrombin in endothelial cells. J. Cell. Physiol. 2014, 229, 1292–1300. [Google Scholar] [CrossRef] [PubMed]
- Kaneider, N.; Mosheimer, B.; Reinisch, N.; Patsch, J.R.; Wiedermann, C.J. Inhibition of thrombin-induced signaling by resveratrol and quercetin: Effects on adenosine nucleotide metabolism in endothelial cells and platelet-neutrophil interactions. Thromb. Res. 2004, 114, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Gordon, S.; Taylor, P.R. Monocyte and macrophage heterogeneity. Nat. Rev. Immunol. 2005, 5, 953–964. [Google Scholar] [CrossRef] [PubMed]
- Richmond, A. Nf-kappa B, chemokine gene transcription and tumour growth. Nat. Rev. Immunol. 2002, 2, 664–674. [Google Scholar] [CrossRef] [PubMed]
- Ohmori, Y.; Hamilton, T.A. Requirement for STAT1 in LPS-induced gene expression in macrophages. J. Leukoc. Biol. 2001, 69, 598–604. [Google Scholar] [PubMed]
- Ito, S.; Ansari, P.; Sakatsume, M.; Dickensheets, H.; Vazquez, N.; Donnelly, R.P.; Larner, A.C.; Finbloom, D.S. Interleukin-10 inhibits expression of both interferon alpha- and interferon gamma- induced genes by suppressing tyrosine phosphorylation of STAT1. Blood 1999, 93, 1456–1463. [Google Scholar] [PubMed]
- Baty, F.; Facompre, M.; Wiegand, J.; Schwager, J.; Brutsche, M.H. Analysis with respect to instrumental variables for the exploration of microarray data structures. BMC Bioinform. 2006, 7, 422. [Google Scholar] [CrossRef] [PubMed]
- Diehl, S.; Rincon, M. The two faces of IL-6 on Th1/Th2 differentiation. Mol. Immunol. 2002, 39, 531–536. [Google Scholar] [CrossRef]
- Heusinkveld, M.; de Vos van Steenwijk, P.J.; Goedemans, R.; Ramwadhdoebe, T.H.; Gorter, A.; Welters, M.J.; van Hall, T.; van der Burg, S.H. M2 macrophages induced by prostaglandin E2 and IL-6 from cervical carcinoma are switched to activated M1 macrophages by CD4+ Th1 cells. J. Immunol. 2011, 187, 1157–1165. [Google Scholar] [CrossRef] [PubMed]
- Mayer, A.; Debuisson, D.; Denanglaire, S.; Eddahri, F.; Fievez, L.; Hercor, M.; Triffaux, E.; Moser, M.; Bureau, F.; Leo, O.; et al. Antigen presenting cell-derived IL-6 restricts Th2-cell differentiation. Eur. J. Immunol. 2014, 44, 3252–3262. [Google Scholar] [PubMed]
- Mantovani, A.; Sozzani, S.; Locati, M.; Allavena, P.; Sica, A. Macrophage polarization: Tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol. 2002, 23, 549–555. [Google Scholar] [CrossRef]
- Mauer, J.; Chaurasia, B.; Goldau, J.; Vogt, M.C.; Ruud, J.; Nguyen, K.D.; Theurich, S.; Hausen, A.C.; Schmitz, J.; Bronneke, H.S.; et al. Signaling by IL-6 promotes alternative activation of macrophages to limit endotoxemia and obesity-associated resistance to insulin. Nat. Immunol. 2014, 15, 423–430. [Google Scholar] [CrossRef] [PubMed]
- Walle, T.; Hsieh, F.; DeLegge, M.H.; Oatis, J.E., Jr.; Walle, U.K. High absorption but very low bioavailability of oral resveratrol in humans. Drug Metab. Dispos. 2004, 32, 1377–1382. [Google Scholar] [CrossRef] [PubMed]
- Gallucci, S.; Matzinger, P. Danger signals: SOS to the immune system. Curr. Opin. Immunol. 2001, 13, 114–119. [Google Scholar] [CrossRef]
- Skoberne, M.; Beignon, A.S.; Bhardwaj, N. Danger signals: A time and space continuum. Trends Mol. Med. 2004, 10, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Schwager, J.; Hoeller, U.; Wolfram, S.; Richard, N. Rose hip and its constituent galactolipids confer cartilage protection by modulating cytokine, and chemokine expression. BMC Complement. Altern. Med. 2011, 11, 105–119. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the TAE are available from the authors.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schwager, J.; Richard, N.; Mussler, B.; Raederstorff, D. Tomato Aqueous Extract Modulates the Inflammatory Profile of Immune Cells and Endothelial Cells. Molecules 2016, 21, 168. https://doi.org/10.3390/molecules21020168
Schwager J, Richard N, Mussler B, Raederstorff D. Tomato Aqueous Extract Modulates the Inflammatory Profile of Immune Cells and Endothelial Cells. Molecules. 2016; 21(2):168. https://doi.org/10.3390/molecules21020168
Chicago/Turabian StyleSchwager, Joseph, Nathalie Richard, Bernd Mussler, and Daniel Raederstorff. 2016. "Tomato Aqueous Extract Modulates the Inflammatory Profile of Immune Cells and Endothelial Cells" Molecules 21, no. 2: 168. https://doi.org/10.3390/molecules21020168
APA StyleSchwager, J., Richard, N., Mussler, B., & Raederstorff, D. (2016). Tomato Aqueous Extract Modulates the Inflammatory Profile of Immune Cells and Endothelial Cells. Molecules, 21(2), 168. https://doi.org/10.3390/molecules21020168





