Nanoparticle-Enabled Transdermal Drug Delivery Systems for Enhanced Dose Control and Tissue Targeting
Abstract
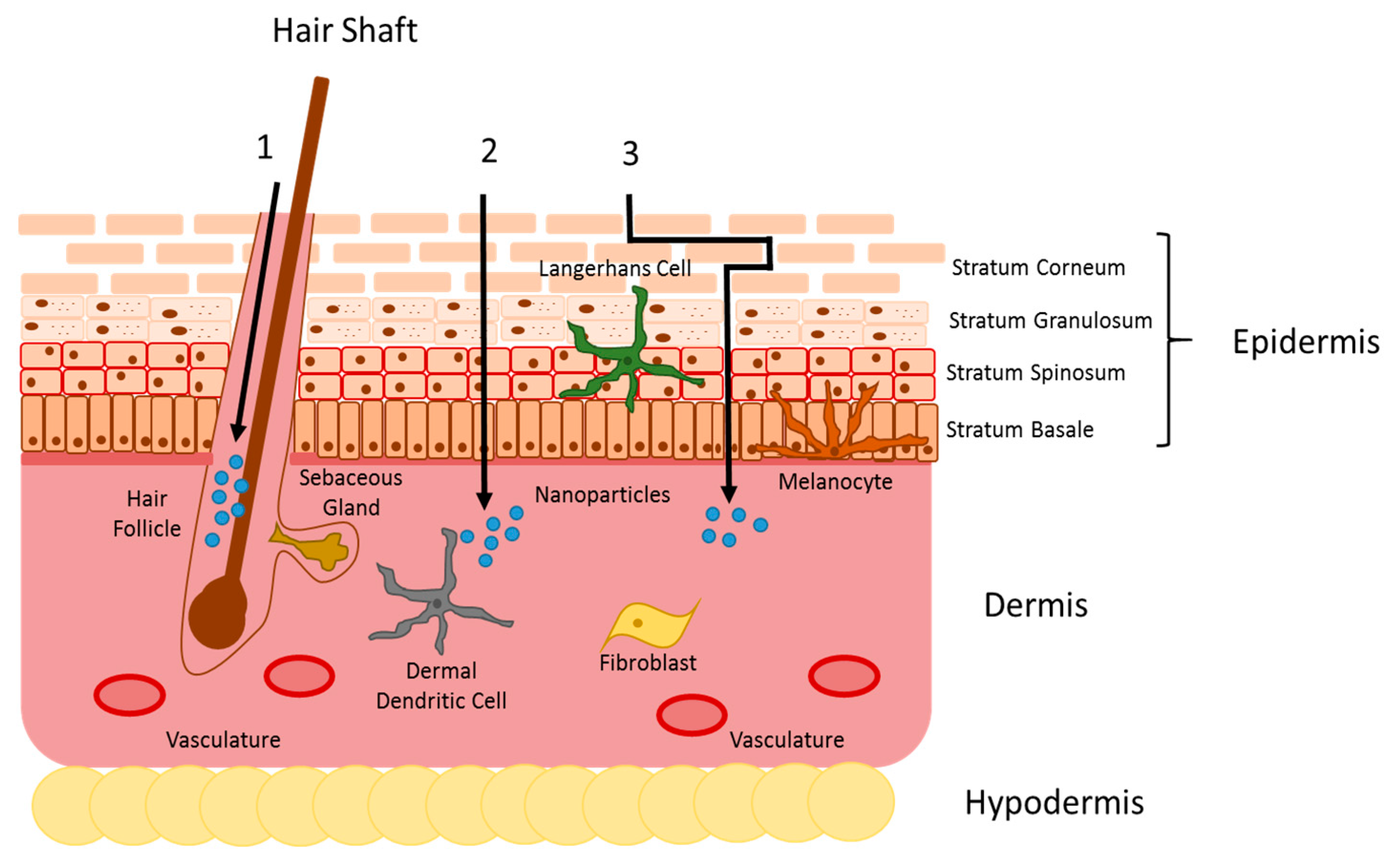
:1. Introduction
2. Skin Barrier
3. Nanocarrier Skin Penetration
4. Psoriasis and Atopic Dermatitis
5. Transdermal Nanocarriers for the Treatment of Skin Diseases
6. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Prausnitz, M.R.; Langer, R. Transdermal drug delivery. Nat. Biotechnol. 2008, 26, 1261–1268. [Google Scholar] [CrossRef] [PubMed]
- Tanner, T.; Marks, R. Delivering drugs by the transdermal route: Review and comment. Skin Res. Technol. 2008, 14, 249–260. [Google Scholar] [CrossRef] [PubMed]
- Durand, C.; Alhammad, A.; Willett, K.C. Practical considerations for optimal transdermal drug delivery. Am. J. Health. Syst. Pharm. 2012, 69, 116–124. [Google Scholar] [CrossRef] [PubMed]
- Samad, A.; Ullah, Z.; Alam, M.I.; Wais, M.; Shams, M.S. Transdermal drug delivery system: Patent reviews. Recent Pat. Drug Deliv. Formul. 2009, 3, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Gupta, H.; Babu, R.J. Transdermal delivery: Product and patent update. Recent Pat. Drug Deliv Formul 2013, 7, 184–205. [Google Scholar] [CrossRef] [PubMed]
- Graybiel, A.; Knepton, J.; Shaw, J. Prevention of experimental motion sickness by scopolamine absorbed through the skin. Aviat. Space Environ. Med. 1976, 47, 1096–1100. [Google Scholar] [PubMed]
- Larraneta, E.; McCrudden, M.T.; Courtenay, A.J.; Donnelly, R.F. Microneedles: A new frontier in nanomedicine delivery. Pharm. Res. 2016, 33, 1055–1073. [Google Scholar] [CrossRef] [PubMed]
- Oberli, M.A.; Schoellhammer, C.M.; Langer, R.; Blankschtein, D. Ultrasound-enhanced transdermal delivery: Recent advances and future challenges. Ther. Deliv. 2014, 5, 843–857. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.B.; Martin, G.P.; Jones, S.A.; Akomeah, F.K. Dermal and transdermal drug delivery systems: Current and future prospects. Drug Deliv. 2006, 13, 175–187. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.O.; Rijo, P.; Molpeceres, J.; Figueiredo, I.V.; Ascensao, L.; Fernandes, A.S.; Roberto, A.; Reis, C.P. Polymeric nanoparticles modified with fatty acids encapsulating betamethasone for anti-inflammatory treatment. Int. J. Pharm. 2015, 493, 271–284. [Google Scholar] [CrossRef] [PubMed]
- Wysocki, A.B. Skin anatomy, physiology, and pathophysiology. Nurs. Clin. N. Am. 1999, 34, 777–797. [Google Scholar]
- Matsui, T.; Amagai, M. Dissecting the formation, structure and barrier function of the stratum corneum. Int. Immunol. 2015, 27, 269–280. [Google Scholar] [CrossRef] [PubMed]
- Brandner, J.M. Tight junctions and tight junction proteins in mammalian epidermis. Eur. J. Pharm. Biopharm. 2009, 72, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Eckhart, L.; Lippens, S.; Tschachler, E.; Declercq, W. Cell death by cornification. Biochim. Biophys. Acta 2013, 1833, 3471–3480. [Google Scholar] [CrossRef] [PubMed]
- van Smeden, J.; Janssens, M.; Gooris, G.S.; Bouwstra, J.A. The important role of stratum corneum lipids for the cutaneous barrier function. Biochim. Biophys. Acta 2014, 1841, 295–313. [Google Scholar] [CrossRef] [PubMed]
- Bos, J.D.; Meinardi, M.M. The 500 dalton rule for the skin penetration of chemical compounds and drugs. Exp. Dermatol. 2000, 9, 165–169. [Google Scholar] [CrossRef] [PubMed]
- Tolleson, W.H. Human melanocyte biology, toxicology, and pathology. J. Environ. Sci Health C Environ. Carcinog. Ecotoxicol. Rev. 2005, 23, 105–161. [Google Scholar] [CrossRef] [PubMed]
- Salmon, J.K.; Armstrong, C.A.; Ansel, J.C. The skin as an immune organ. West. J. Med. 1994, 160, 146–152. [Google Scholar] [PubMed]
- Clausen, B.E.; Stoitzner, P. Functional specialization of skin dendritic cell subsets in regulating T cell responses. Front. Immunol. 2015, 6, 534. [Google Scholar] [CrossRef] [PubMed]
- Haniffa, M.; Gunawan, M.; Jardine, L. Human skin dendritic cells in health and disease. J. Dermatol. Sci. 2015, 77, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Stücker, M.; Struk, A.; Altmeyer, P.; Herde, M.; Baumgärtl, H.; Lübbers, D.W. The cutaneous uptake of atmospheric oxygen contributes significantly to the oxygen supply of human dermis and epidermis. J. Physiol. 2002, 538, 985–994. [Google Scholar] [CrossRef] [PubMed]
- Lawlor, K.T.; Kaur, P. Dermal contributions to human interfollicular epidermal architecture and self-renewal. Int. J. Mol. Sci. 2015, 16, 28098–28107. [Google Scholar] [CrossRef] [PubMed]
- Amano, S. Characterization and mechanisms of photoageing-related changes in skin. Damages of basement membrane and dermal structures. Exp. Dermatol. 2016, 25, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Braverman, I.M. The role of blood vessels and lymphatics in cutaneous inflammatory processes: An overview. Br. J. Dermatol. 1983, 109, 89–98. [Google Scholar] [PubMed]
- Tansey, E.A.; Johnson, C.D. Recent advances in thermoregulation. Adv. Physiol. Educ. 2015, 39, 139–148. [Google Scholar] [CrossRef] [PubMed]
- German, G.K.; Engl, W.C.; Pashkovski, E.; Banerjee, S.; Xu, Y.; Mertz, A.F.; Hyland, C.; Dufresne, E.R. Heterogeneous drying stresses in stratum corneum. Biophys. J. 2012, 102, 2424–2432. [Google Scholar] [CrossRef] [PubMed]
- Gupta, M.; Agrawal, U.; Vyas, S.P. Nanocarrier-based topical drug delivery for the treatment of skin diseases. Expert Opin. Drug Deliv. 2012, 9, 783–804. [Google Scholar] [CrossRef] [PubMed]
- Patzelt, A.; Lademann, J. Drug delivery to hair follicles. Expert Opin. Drug Deliv. 2013, 10, 787–797. [Google Scholar] [CrossRef] [PubMed]
- Pariser, D. Topical corticosteroids and topical calcineurin inhibitors in the treatment of atopic dermatitis: Focus on percutaneous absorption. Am. J. Ther. 2009, 16, 264–273. [Google Scholar] [CrossRef] [PubMed]
- Schneider, M.; Stracke, F.; Hansen, S.; Schaefer, U.F. Nanoparticles and their interactions with the dermal barrier. Dermatoendocrinology 2009, 1, 197–206. [Google Scholar] [CrossRef]
- Jensen, L.B.; Petersson, K.; Nielsen, H.M. In vitro penetration properties of solid lipid nanoparticles in intact and barrier-impaired skin. Eur. J. Pharm. Biopharm. 2011, 79, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Mottaleb, M.M.; Moulari, B.; Beduneau, A.; Pellequer, Y.; Lamprecht, A. Surface-charge-dependent nanoparticles accumulation in inflamed skin. J. Pharm. Sci. 2012, 101, 4231–4239. [Google Scholar] [CrossRef] [PubMed]
- Lee, O.; Jeong, S.H.; Shin, W.U.; Lee, G.; Oh, C.; Son, S.W. Influence of surface charge of gold nanorods on skin penetration. Skin Res. Technol. 2013, 19, e390–e396. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M. Protective effect of fine-particle titanium dioxide on uvb-induced DNA damage in hairless mouse skin. Photodermatology 1987, 4, 209–211. [Google Scholar] [PubMed]
- Contado, C. Nanomaterials in consumer products: A challenging analytical problem. Front. Chem. 2015, 3, 48. [Google Scholar] [CrossRef] [PubMed]
- Xiao, L.; Takada, H.; Gan, X.; Miwa, N. The water-soluble fullerene derivative “radical sponge” exerts cytoprotective action against uva irradiation but not visible-light-catalyzed cytotoxicity in human skin keratinocytes. Bioorg. Med. Chem. Lett. 2006, 16, 1590–1595. [Google Scholar] [CrossRef] [PubMed]
- Watkinson, A.C.; Bunge, A.L.; Hadgraft, J.; Lane, M.E. Nanoparticles do not penetrate human skin—A theoretical perspective. Pharm. Res. 2013, 30, 1943–1946. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, R.; Smyth, N.R.; Muskens, O.L.; Nitti, S.; Heuer-Jungemann, A.; Ardern-Jones, M.R.; Kanaras, A.G. Interactions of skin with gold nanoparticles of different surface charge, shape, and functionality. Small 2015, 11, 713–721. [Google Scholar] [CrossRef] [PubMed]
- Plascencia-Villa, G.; Bahena, D.; Rodríguez, A.R.; Ponce, A.; José-Yacamán, M. Advanced microscopy of star-shaped gold nanoparticles and their adsorption-uptake by macrophages. Metall. Integr. Biometal. Sci. 2013, 5, 242–250. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Ediriwickrema, A.; Yang, F.; Lewis, J.; Girardi, M.; Saltzman, W.M. A sunblock based on bioadhesive nanoparticles. Nat. Mater. 2015, 14, 1278–1285. [Google Scholar] [CrossRef] [PubMed]
- Larese Filon, F.; Mauro, M.; Adami, G.; Bovenzi, M.; Crosera, M. Nanoparticles skin absorption: New aspects for a safety profile evaluation. Regulat. Toxicol. Pharm. 2015, 72, 310–322. [Google Scholar] [CrossRef] [PubMed]
- Baroli, B.; Ennas, M.G.; Loffredo, F.; Isola, M.; Pinna, R.; Lopez-Quintela, M.A. Penetration of metallic nanoparticles in human full-thickness skin. J. Investig. Dermatol. 2007, 127, 1701–1712. [Google Scholar] [CrossRef] [PubMed]
- Mortensen, L.J.; Oberdorster, G.; Pentland, A.P.; Delouise, L.A. In vivo skin penetration of quantum dot nanoparticles in the murine model: The effect of uvr. Nano Lett. 2008, 8, 2779–2787. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Choe, C.S.; Ahlberg, S.; Meinke, M.C.; Alexiev, U.; Lademann, J.; Darvin, M.E. Penetration of silver nanoparticles into porcine skin ex vivo using fluorescence lifetime imaging microscopy, raman microscopy, and surface-enhanced raman scattering microscopy. J. Biomed. Opt. 2015, 20, 051006. [Google Scholar] [CrossRef] [PubMed]
- Lademann, J.; Knorr, F.; Richter, H.; Blume-Peytavi, U.; Vogt, A.; Antoniou, C.; Sterry, W.; Patzelt, A. Hair follicles—An efficient storage and penetration pathway for topically applied substances. Summary of recent results obtained at the center of experimental and applied cutaneous physiology, charite-universitatsmedizin berlin, germany. Skin Pharmacol. Physiol. 2008, 21, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Lademann, J.; Richter, H.; Teichmann, A.; Otberg, N.; Blume-Peytavi, U.; Luengo, J.; Weiss, B.; Schaefer, U.F.; Lehr, C.M.; Wepf, R.; et al. Nanoparticles—An efficient carrier for drug delivery into the hair follicles. Eur. J. Pharm. Biopharm. 2007, 66, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Filon, F.L.; Crosera, M.; Adami, G.; Bovenzi, M.; Rossi, F.; Maina, G. Human skin penetration of gold nanoparticles through intact and damaged skin. Nanotoxicology 2011, 5, 493–501. [Google Scholar] [CrossRef] [PubMed]
- Campbell, C.S.; Contreras-Rojas, L.R.; Delgado-Charro, M.B.; Guy, R.H. Objective assessment of nanoparticle disposition in mammalian skin after topical exposure. J. Control. Release 2012, 162, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Miquel-Jeanjean, C.; Crepel, F.; Raufast, V.; Payre, B.; Datas, L.; Bessou-Touya, S.; Duplan, H. Penetration study of formulated nanosized titanium dioxide in models of damaged and sun-irradiated skins. Photochem. Photobiol. 2012, 88, 1513–1521. [Google Scholar] [CrossRef] [PubMed]
- Alnasif, N.; Zoschke, C.; Fleige, E.; Brodwolf, R.; Boreham, A.; Ruhl, E.; Eckl, K.M.; Merk, H.F.; Hennies, H.C.; Alexiev, U.; et al. Penetration of normal, damaged and diseased skin—an in vitro study on dendritic core-multishell nanotransporters. J. Control. Release 2014, 185, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Pasparakis, M.; Haase, I.; Nestle, F.O. Mechanisms regulating skin immunity and inflammation. Nat. Rev. Immunol. 2014, 14, 289–301. [Google Scholar] [CrossRef] [PubMed]
- Monteiro-Riviere, N.A.; Bristol, D.G.; Manning, T.O.; Rogers, R.A.; Riviere, J.E. Interspecies and interregional analysis of the comparative histologic thickness and laser doppler blood flow measurements at five cutaneous sites in nine species. J. Investig. Dermatol. 1990, 95, 582–586. [Google Scholar] [CrossRef] [PubMed]
- Mortensen, L.J.; Jatana, S.; Gelein, R.; De Benedetto, A.; De Mesy Bentley, K.L.; Beck, L.A.; Elder, A.; Delouise, L.A. Quantification of quantum dot murine skin penetration with uvr barrier impairment. Nanotoxicology 2013, 7, 1386–1398. [Google Scholar] [CrossRef] [PubMed]
- Tang, L.; Zhang, C.; Song, G.; Jin, X.; Xu, Z. In vivo skin penetration and metabolic path of quantum dots. Sci. China Life Sci. 2013, 56, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Hirai, T.; Yoshikawa, T.; Nabeshi, H.; Yoshida, T.; Akase, T.; Yoshioka, Y.; Itoh, N.; Tsutsumi, Y. Dermal absorption of amorphous nanosilica particles after topical exposure for three days. Pharmazie 2012, 67, 742–743. [Google Scholar] [PubMed]
- Mardhiah Adib, Z.; Ghanbarzadeh, S.; Kouhsoltani, M.; Yari Khosroshahi, A.; Hamishehkar, H. The effect of particle size on the deposition of solid lipid nanoparticles in different skin layers: A histological study. Adv. Pharm. Bull. 2016, 6, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Xie, G.; Lu, W.; Lu, D. Penetration of titanium dioxide nanoparticles through slightly damaged skin in vitro and in vivo. J. Appl. Biomater. Funct Mater. 2015, 13, e356–e361. [Google Scholar] [CrossRef] [PubMed]
- Goon, A.T.; Yosipovitch, G.; Chan, Y.H.; Goh, C.L. Barrier repair in chronic plaque-type psoriasis. Skin Res. Technol. 2004, 10, 10–13. [Google Scholar] [CrossRef] [PubMed]
- Czarnowicki, T.; Krueger, J.G.; Guttman-Yassky, E. Skin barrier and immune dysregulation in atopic dermatitis: An evolving story with important clinical implications. J. Allergy Clin. Immunol. Pract. 2014, 2, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Dommasch, E.D.; Li, T.; Okereke, O.I.; Li, Y.; Qureshi, A.A.; Cho, E. Risk of depression in women with psoriasis: A cohort study. Br. J. Dermatol. 2015, 173, 975–980. [Google Scholar] [CrossRef] [PubMed]
- Baviera, G.; Leoni, M.C.; Capra, L.; Cipriani, F.; Longo, G.; Maiello, N.; Ricci, G.; Galli, E. Microbiota in healthy skin and in atopic eczema. BioMed Res. Int. 2014, 2014, 436921. [Google Scholar] [CrossRef] [PubMed]
- Kadunce, D.P.; Krueger, G.G. Pathogenesis of psoriasis. Dermatol. Clin. 1995, 13, 723–737. [Google Scholar] [PubMed]
- Worldwide variation in prevalence of symptoms of asthma, allergic rhinoconjunctivitis, and atopic eczema: Isaac. The international study of asthma and allergies in childhood (isaac) steering committee. Lancet 1998, 351, 1225–1232.
- Harrop, J.; Chinn, S.; Verlato, G.; Olivieri, M.; Norback, D.; Wjst, M.; Janson, C.; Zock, J.P.; Leynaert, B.; Gislason, D.; et al. Eczema, atopy and allergen exposure in adults: A population-based study. Clin. Exp. Allergy 2007, 37, 526–535. [Google Scholar] [CrossRef] [PubMed]
- Helmick, C.G.; Lee-Han, H.; Hirsch, S.C.; Baird, T.L.; Bartlett, C.L. Prevalence of psoriasis among adults in the US: 2003–2006 and 2009–2010 national health and nutrition examination surveys. Am. J. Prev. Med. 2014, 47, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Merola, J.F.; Li, T.; Li, W.Q.; Cho, E.; Qureshi, A.A. Prevalence of psoriasis phenotypes among men and women in the USA. Clin. Exp. Dermatol. 2016, 41, 486–489. [Google Scholar] [CrossRef] [PubMed]
- Harris, J.M.; Williams, H.C.; White, C.; Moffat, S.; Mills, P.; Newman Taylor, A.J.; Cullinan, P. Early allergen exposure and atopic eczema. Br. J. Dermatol. 2007, 156, 698–704. [Google Scholar] [CrossRef] [PubMed]
- Martin, L. [what’s new in dermatological treatments?]. Ann. Dermatol. Venereol. 2010, 137, S165–S176. [Google Scholar] [CrossRef]
- Menter, A.; Korman, N.J.; Elmets, C.A.; Feldman, S.R.; Gelfand, J.M.; Gordon, K.B.; Gottlieb, A.; Koo, J.Y.; Lebwohl, M.; Leonardi, C.L.; et al. Guidelines of care for the management of psoriasis and psoriatic arthritis: Section 6. Guidelines of care for the treatment of psoriasis and psoriatic arthritis: Case-based presentations and evidence-based conclusions. J. Am. Acad. Dermatol. 2011, 65, 137–174. [Google Scholar] [CrossRef] [PubMed]
- Aslam, I.; Sandoval, L.F.; Feldman, S.R. What’s new in the topical treatment of allergic skin diseases. Curr. Opin. Allergy Clin. Immunol. 2014, 14, 436–450. [Google Scholar] [CrossRef] [PubMed]
- Saeki, H.; Nakahara, T.; Tanaka, A.; Kabashima, K.; Sugaya, M.; Murota, H.; Ebihara, T.; Kataoka, Y.; Aihara, M.; Etoh, T.; et al. Clinical practice guidelines for the management of atopic dermatitis 2016. J. Dermatol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Ellis, C.N.; Drake, L.A.; Prendergast, M.M.; Abramovits, W.; Boguniewicz, M.; Daniel, C.R.; Lebwohl, M.; Stevens, S.R.; Whitaker-Worth, D.L.; Cheng, J.W.; et al. Cost of atopic dermatitis and eczema in the united states. J. Am. Acad. Dermatol. 2002, 46, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Brezinski, E.A.; Dhillon, J.S.; Armstrong, A.W. Economic burden of psoriasis in the united states: A systematic review. JAMA Dermatol. 2015, 151, 651–658. [Google Scholar] [CrossRef] [PubMed]
- D’Souza, L.S.; Payette, M.J. Estimated cost efficacy of systemic treatments that are approved by the us food and drug administration for the treatment of moderate to severe psoriasis. J. Am. Acad. Dermatol. 2015, 72, 589–598. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, H.; Kubo, A.; Sasaki, T.; Amagai, M. Loss-of-function mutations within the filaggrin gene and atopic dermatitis. Curr. Probl. Dermatol. 2011, 41, 35–46. [Google Scholar] [PubMed]
- Sandilands, A.; Terron-Kwiatkowski, A.; Hull, P.R.; O’Regan, G.M.; Clayton, T.H.; Watson, R.M.; Carrick, T.; Evans, A.T.; Liao, H.; Zhao, Y.; et al. Comprehensive analysis of the gene encoding filaggrin uncovers prevalent and rare mutations in ichthyosis vulgaris and atopic eczema. Nat. Genet. 2007, 39, 650–654. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, H.; Nagao, K.; Kubo, A.; Hata, T.; Shimizu, A.; Mizuno, H.; Yamada, T.; Amagai, M. Altered stratum corneum barrier and enhanced percutaneous immune responses in filaggrin-null mice. J. Allergy Clin. Immunol. 2012, 129, 1538–1546. [Google Scholar] [CrossRef] [PubMed]
- Seykora, J.; Dentchev, T.; Margolis, D.J. Filaggrin-2 barrier protein inversely varies with skin inflammation. Exp. Dermatol. 2015, 24, 720–722. [Google Scholar] [CrossRef] [PubMed]
- De Benedetto, A.; Rafaels, N.M.; McGirt, L.Y.; Ivanov, A.I.; Georas, S.N.; Cheadle, C.; Berger, A.E.; Zhang, K.; Vidyasagar, S.; Yoshida, T.; et al. Tight junction defects in patients with atopic dermatitis. J. Allergy Clin. Immunol. 2011, 127, 773–786. [Google Scholar] [CrossRef] [PubMed]
- van Drongelen, V.; Haisma, E.M.; Out-Luiting, J.J.; Nibbering, P.H.; El Ghalbzouri, A. Reduced filaggrin expression is accompanied by increased staphylococcus aureus colonization of epidermal skin models. Clin. Exp. Allergy 2014, 44, 1515–1524. [Google Scholar] [CrossRef] [PubMed]
- Jinnestal, C.L.; Belfrage, E.; Back, O.; Schmidtchen, A.; Sonesson, A. Skin barrier impairment correlates with cutaneous staphylococcus aureus colonization and sensitization to skin-associated microbial antigens in adult patients with atopic dermatitis. Int. J. Dermatol. 2014, 53, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Nomura, I.; Goleva, E.; Howell, M.D.; Hamid, Q.A.; Ong, P.Y.; Hall, C.F.; Darst, M.A.; Gao, B.; Boguniewicz, M.; Travers, J.B.; et al. Cytokine milieu of atopic dermatitis, as compared to psoriasis, skin prevents induction of innate immune response genes. J. Immunol. 2003, 171, 3262–3269. [Google Scholar] [CrossRef] [PubMed]
- Brandt, E.B.; Sivaprasad, U. Th2 cytokines and atopic dermatitis. J. Clin. Cell. Immunol. 2011, 2. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, K.; Mizutani, H. The role of cytokines/chemokines in the pathogenesis of atopic dermatitis. Curr. Probl. Dermatol. 2011, 41, 80–92. [Google Scholar] [PubMed]
- Chandran, V. The genetics of psoriasis and psoriatic arthritis. Clin. Rev. Allergy Immunol. 2013, 44, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Guttman-Yassky, E.; Nograles, K.E.; Krueger, J.G. Contrasting pathogenesis of atopic dermatitis and psoriasis—Part I: Clinical and pathologic concepts. J. Allergy Clin. Immunol. 2011, 127, 1110–1118. [Google Scholar] [CrossRef] [PubMed]
- Hanel, K.H.; Cornelissen, C.; Luscher, B.; Baron, J.M. Cytokines and the skin barrier. Int. J. Mol. Sci. 2013, 14, 6720–6745. [Google Scholar] [CrossRef] [PubMed]
- Wolf, R.; Orion, E.; Ruocco, E.; Ruocco, V. Abnormal epidermal barrier in the pathogenesis of psoriasis. Clin. Dermatol. 2012, 30, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Sticherling, M. Psoriasis and autoimmunity. Autoimmun. Rev. 2016, 15, 1167–1170. [Google Scholar] [CrossRef] [PubMed]
- Fry, L.; Baker, B.S.; Powles, A.V.; Engstrand, L. Psoriasis is not an autoimmune disease? Exp. Dermatol. 2015, 24, 241–244. [Google Scholar] [CrossRef] [PubMed]
- Hanifin, J.M.; Cooper, K.D.; Ho, V.C.; Kang, S.; Krafchik, B.R.; Margolis, D.J.; Schachner, L.A.; Sidbury, R.; Whitmore, S.E.; Sieck, C.K.; et al. Guidelines of care for atopic dermatitis, developed in accordance with the american academy of dermatology (aad)/american academy of dermatology association “administrative regulations for evidence-based clinical practice guidelines”. J. Am. Acad. Dermatol. 2004, 50, 391–404. [Google Scholar] [CrossRef] [PubMed]
- Korting, H.C.; Schafer-Korting, M. Carriers in the topical treatment of skin disease. Handb. Exp. Pharmacol. 2010, 197, 435–468. [Google Scholar]
- Dreier, J.; Sorensen, J.A.; Brewer, J.R. Superresolution and fluorescence dynamics evidence reveal that intact liposomes do not cross the human skin barrier. PLoS ONE 2016, 11, e0146514. [Google Scholar] [CrossRef] [PubMed]
- Madheswaran, T.; Baskaran, R.; Thapa, R.K.; Rhyu, J.Y.; Choi, H.Y.; Kim, J.O.; Yong, C.S.; Yoo, B.K. Design and in vitro evaluation of finasteride-loaded liquid crystalline nanoparticles for topical delivery. AAPS PharmSciTech 2013, 14, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Lapteva, M.; Mondon, K.; Moller, M.; Gurny, R.; Kalia, Y.N. Polymeric micelle nanocarriers for the cutaneous delivery of tacrolimus: A targeted approach for the treatment of psoriasis. Mol. Pharm. 2014, 11, 2989–3001. [Google Scholar] [CrossRef] [PubMed]
- Goebel, A.S.; Neubert, R.H.; Wohlrab, J. Dermal targeting of tacrolimus using colloidal carrier systems. Int. J. Pharm. 2011, 404, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Pople, P.V.; Singh, K.K. Targeting tacrolimus to deeper layers of skin with improved safety for treatment of atopic dermatitis-part ii: In vivo assessment of dermatopharmacokinetics, biodistribution and efficacy. Int. J. Pharm. 2012, 434, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Pople, P.V.; Singh, K.K. Development and evaluation of colloidal modified nanolipid carrier: Application to topical delivery of tacrolimus, part II—In vivo assessment, drug targeting, efficacy, and safety in treatment for atopic dermatitis. Eur. J. Pharm. Biopharm. 2013, 84, 72–83. [Google Scholar] [CrossRef] [PubMed]
- Thapa, R.K.; Yoo, B.K. Evaluation of the effect of tacrolimus-loaded liquid crystalline nanoparticles on psoriasis-like skin inflammation. J. Dermatol. Treat. 2014, 25, 22–25. [Google Scholar] [CrossRef] [PubMed]
- Doktorovova, S.; Araujo, J.; Garcia, M.L.; Rakovsky, E.; Souto, E.B. Formulating fluticasone propionate in novel peg-containing nanostructured lipid carriers (peg-nlc). Colloids Surf. B Biointerfaces 2010, 75, 538–542. [Google Scholar] [CrossRef] [PubMed]
- Marchiori, M.L.; Lubini, G.; Dalla Nora, G.; Friedrich, R.B.; Fontana, M.C.; Ourique, A.F.; Bastos, M.O.; Rigo, L.A.; Silva, C.B.; Tedesco, S.B.; et al. Hydrogel containing dexamethasone-loaded nanocapsules for cutaneous administration: Preparation, characterization, and in vitro drug release study. Drug Dev. Ind. Pharm. 2010, 36, 962–971. [Google Scholar] [CrossRef] [PubMed]
- Fontana, M.C.; Rezer, J.F.; Coradini, K.; Leal, D.B.; Beck, R.C. Improved efficacy in the treatment of contact dermatitis in rats by a dermatological nanomedicine containing clobetasol propionate. Eur. J. Pharm. Biopharm. 2011, 79, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Eroglu, I.; Azizoglu, E.; Ozyazici, M.; Nenni, M.; Gurer Orhan, H.; Ozbal, S.; Tekmen, I.; Ertam, I.; Unal, I.; Ozer, O. Effective topical delivery systems for corticosteroids: Dermatological and histological evaluations. Drug Deliv. 2016, 23, 1502–1513. [Google Scholar] [PubMed]
- Siddique, M.I.; Katas, H.; Amin, M.C.; Ng, S.F.; Zulfakar, M.H.; Jamil, A. In Vivo dermal pharmacokinetics, efficacy, and safety of skin targeting nanoparticles for corticosteroid treatment of atopic dermatitis. Int. J. Pharm. 2016, 507, 72–82. [Google Scholar] [CrossRef] [PubMed]
- Ourique, A.F.; Pohlmann, A.R.; Guterres, S.S.; Beck, R.C. Tretinoin-loaded nanocapsules: Preparation, physicochemical characterization, and photostability study. Int. J. Pharm. 2008, 352, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Ourique, A.F.; Melero, A.; Silva, C.d.B.d.; Schaefer, U.F.; Pohlmann, A.R.; Guterres, S.S.; Lehr, C.-M.; Kostka, K.-H.; Beck, R.C.R. Improved photostability and reduced skin permeation of tretinoin: Development of a semisolid nanomedicine. Eur. J. Pharm. Biopharm. 2011, 79, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Ridolfi, D.M.; Marcato, P.D.; Justo, G.Z.; Cordi, L.; Machado, D.; Duran, N. Chitosan-solid lipid nanoparticles as carriers for topical delivery of tretinoin. Colloids Surf. B Biointerfaces 2012, 93, 36–40. [Google Scholar] [CrossRef] [PubMed]
- Manconi, M.; Sinico, C.; Caddeo, C.; Vila, A.O.; Valenti, D.; Fadda, A.M. Penetration enhancer containing vesicles as carriers for dermal delivery of tretinoin. Int. J. Pharm. 2011, 412, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Raza, K.; Singh, B.; Lohan, S.; Sharma, G.; Negi, P.; Yachha, Y.; Katare, O.P. Nano-lipoidal carriers of tretinoin with enhanced percutaneous absorption, photostability, biocompatibility and anti-psoriatic activity. Int. J. Pharm. 2013, 456, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Bessar, H.; Venditti, I.; Benassi, L.; Vaschieri, C.; Azzoni, P.; Pellacani, G.; Magnoni, C.; Botti, E.; Casagrande, V.; Federici, M.; et al. Functionalized gold nanoparticles for topical delivery of methotrexate for the possible treatment of psoriasis. Colloids Surf. B Biointerfaces 2016, 141, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Srisuk, P.; Thongnopnua, P.; Raktanonchai, U.; Kanokpanont, S. Physico-chemical characteristics of methotrexate-entrapped oleic acid-containing deformable liposomes for in vitro transepidermal delivery targeting psoriasis treatment. Int. J. Pharm. 2012, 427, 426–434. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.K.; Huang, Z.R.; Zhuo, R.Z.; Fang, J.Y. Combination of calcipotriol and methotrexate in nanostructured lipid carriers for topical delivery. Int J. Nanomed. 2010, 5, 117–128. [Google Scholar]
- Singka, G.S.; Samah, N.A.; Zulfakar, M.H.; Yurdasiper, A.; Heard, C.M. Enhanced topical delivery and anti-inflammatory activity of methotrexate from an activated nanogel. Eur. J. Pharm. Biopharm. 2010, 76, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Avasatthi, V.; Pawar, H.; Dora, C.P.; Bansod, P.; Gill, M.S.; Suresh, S. A novel nanogel formulation of methotrexate for topical treatment of psoriasis: Optimization, in vitro and in vivo evaluation. Pharm. Dev. Technol. 2016, 21, 554–562. [Google Scholar] [PubMed]
- Romero, G.B.; Arntjen, A.; Keck, C.M.; Muller, R.H. Amorphous cyclosporin a nanoparticles for enhanced dermal bioavailability. Int. J. Pharm. 2016, 498, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.T.; Jang, D.J.; Kim, J.H.; Park, J.Y.; Lim, J.S.; Lee, S.Y.; Lee, K.M.; Lim, S.J.; Kim, C.K. Topical administration of cyclosporin a in a solid lipid nanoparticle formulation. Pharmazie 2009, 64, 510–514. [Google Scholar] [PubMed]
- Shah, P.P.; Desai, P.R.; Patel, A.R.; Singh, M.S. Skin permeating nanogel for the cutaneous co-delivery of two anti-inflammatory drugs. Biomaterials 2012, 33, 1607–1617. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.M.; Yoon, G.H.; Lee, H.C.; Jung, M.H.; Yu, S.I.; Yeon, S.J.; Min, S.K.; Kwon, Y.S.; Hwang, J.H.; Shin, H.S. Thermodynamic insights and conceptual design of skin-sensitive chitosan coated ceramide/plga nanodrug for regeneration of stratum corneum on atopic dermatitis. Sci. Rep. 2015, 5, 18089. [Google Scholar] [CrossRef] [PubMed]
- Keck, C.M.; Anantaworasakul, P.; Patel, M.; Okonogi, S.; Singh, K.K.; Roessner, D.; Scherrers, R.; Schwabe, K.; Rimpler, C.; Muller, R.H. A new concept for the treatment of atopic dermatitis: Silver-nanolipid complex (snlc). Int. J. Pharm. 2014, 462, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Ilinskaya, A.N.; Dobrovolskaia, M.A. Immunosuppressive and anti-inflammatory. Br. J. Pharmacol 2014, 171, 3988–4000. [Google Scholar] [CrossRef] [PubMed]
- Shershakova, N.; Baraboshkina, E.; Andreev, S.; Purgina, D.; Struchkova, I.; Kamyshnikov, O.; Nikonova, A.; Khaitov, M. Anti-inflammatory effect of fullerene c60 in a mice model of atopic dermatitis. J. Nanobiotechnol. 2016, 14, 8. [Google Scholar] [CrossRef] [PubMed]
- Ilves, M.; Palomaki, J.; Vippola, M.; Lehto, M.; Savolainen, K.; Savinko, T.; Alenius, H. Topically applied zno nanoparticles suppress allergen induced skin inflammation but induce vigorous ige production in the atopic dermatitis mouse model. Part. Fibre Toxicol. 2014, 11, 38. [Google Scholar] [CrossRef] [PubMed]
- Zheng, D.; Giljohann, D.A.; Chen, D.L.; Massich, M.D.; Wang, X.Q.; Iordanov, H.; Mirkin, C.A.; Paller, A.S. Topical delivery of sirna-based spherical nucleic acid nanoparticle conjugates for gene regulation. Proc. Natl. Acad. Sci. USA 2012, 109, 11975–11980. [Google Scholar] [CrossRef] [PubMed]
- Desmet, E.; Bracke, S.; Forier, K.; Taevernier, L.; Stuart, M.C.; De Spiegeleer, B.; Raemdonck, K.; van Gele, M.; Lambert, J. An elastic liposomal formulation for rnai-based topical treatment of skin disorders: Proof-of-concept in the treatment of psoriasis. Int. J. Pharm. 2016, 500, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Bracke, S.; Carretero, M.; Guerrero-Aspizua, S.; Desmet, E.; Illera, N.; Navarro, M.; Lambert, J.; Del Rio, M. Targeted silencing of defb4 in a bioengineered skin-humanized mouse model for psoriasis: Development of sirna secosome-based novel therapies. Exp. Dermatol. 2014, 23, 199–201. [Google Scholar] [CrossRef] [PubMed]
- Keren, A.; David, M.; Gilhar, A. Novel nanosome delivery system combined with sirna targeting the antimicrobial gene dfb4: A new approach for psoriasis management? Exp. Dermatol. 2014, 23, 464–465. [Google Scholar] [CrossRef] [PubMed]
- Depieri, L.V.; Borgheti-Cardoso, L.N.; Campos, P.M.; Otaguiri, K.K.; Vicentini, F.T.; Lopes, L.B.; Fonseca, M.J.; Bentley, M.V. Rnai mediated il-6 in vitro knockdown in psoriasis skin model with topical sirna delivery system based on liquid crystalline phase. Eur. J. Pharm. Biopharm. 2016, 105, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Kanazawa, T.; Hamasaki, T.; Endo, T.; Tamano, K.; Sogabe, K.; Seta, Y.; Ohgi, T.; Okada, H. Functional peptide nanocarriers for delivery of novel anti-rela rna interference agents as a topical treatment of atopic dermatitis. Int. J. Pharm. 2015, 489, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Hansen, S.; Lehr, C.M. Nanoparticles for transcutaneous vaccination. Microb. Biotechnol. 2012, 5, 156–167. [Google Scholar] [CrossRef] [PubMed]
- Gungor, S.; Ozsoy, Y. Systemic delivery of antihypertensive drugs via skin. Ther. Deliv. 2012, 3, 1101–1116. [Google Scholar] [CrossRef] [PubMed]
- Azeem, A.; Ahmad, F.J.; Khar, R.K.; Talegaonkar, S. Nanocarrier for the transdermal delivery of an antiparkinsonian drug. AAPS PharmSciTech 2009, 10, 1093–1103. [Google Scholar] [CrossRef] [PubMed]
- Sack, M.; Alili, L.; Karaman, E.; Das, S.; Gupta, A.; Seal, S.; Brenneisen, P. Combination of conventional chemotherapeutics with redox-active cerium oxide nanoparticles—A novel aspect in cancer therapy. Mol. Cancer Ther. 2014, 13, 1740–1749. [Google Scholar] [CrossRef] [PubMed]
- Weissig, V.; Pettinger, T.K.; Murdock, N. Nanopharmaceuticals (part 1): Products on the market. Int. J. Nanomed. 2014, 9, 4357–4373. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Magaye, R.; Castranova, V.; Zhao, J. Titanium dioxide nanoparticles: A review of current toxicological data. Particle Fibre Toxicol. 2013, 10. [Google Scholar] [CrossRef] [PubMed]
- Kononenko, V.; Narat, M.; Drobne, D. Nanoparticle interaction with the immune system. Arh. Hig. Rada Toksikol. 2015, 66, 97–108. [Google Scholar] [CrossRef] [PubMed]
| Reference | Model System | Nanocarrier Formulation | Findings |
|---|---|---|---|
| [97,98] | DNFB-induced AD mouse model | Tacrolimus loaded 20–100-nm lipid nanocarriers | Increased skin targeting of drug and decreased AD like symptoms in mouse model |
| [99] | Imiquimod-induced psoriasis mouse model | Tacrolimus loaded 150–200-nm liquid crystalline nanoparticles | Increased skin penetration and effectiveness treating psoriasis model compared to drug without nanocarrier |
| [102] | Nickel sulfate-induced dermatitis model in Wistar rats | Clobetasol propionate loaded 200-nm lipid core nanocapsules | Better controlled drug release and improved skin outcomes compared to drug without nanocarrier |
| [103] | DNFB-induced AD model in Wistar rats | Betamethasone valerate and diflucortolone valerate loaded 220–350-nm liposomes | Better dermal outcomes with a 10 times reduced dose compared to drug without nanocarrier |
| [109] | Mouse tail model of psoriasis | Tretinoin loaded <200-nm nanostructured lipid carriers and liposomes | Increased orthokeratosis observed in the mouse tail, indicative of better psoriasis outcomes |
| [114] | Imiquimod-induced psoriasis mouse model | Methotrexate loaded 278-nm nanostructured lipid carriers | Decreased psoriatic area and severity index |
| [116] | Ovalbumin-induced AD mouse model | Cyclosporin A loaded 73-nm solid lipid nanocarriers | Decreased local IL-4 and IL-5 cytokine levels |
| [117] | Imiquimod-induced psoriasis mouse model | Ketoprofen loaded 200-nm chitosan nanoparticles | Increased skin penetration and better dermal outcomes compared to drug without nanocarrier |
| [118] | SDS-induced AD model in SD rats | Ceramide loaded 200-nm chitosan nanoparticles | Ceramide loaded particles displayed efficacy in regenerating the stratum corneum |
| [119] | DNFB-induced AD model in mice | Silver ion loaded 200-nm nanolipid complex | Silver ions displayed anti-microbial and anti-inflammatory effects in model of AD |
| [121] | Ovalbumin-induced AD mouse model | Aqueous dispersion of nC60 | Nanoparticle treatment reduced IgE and cytokine production and led to better histological skin outcomes |
| [122] | Ovalbumin and Staphylococcal enterotoxin B-induced AD mouse model | Nanosized zinc oxide particles (<50 nm) | Decreased ear swelling responses, but heightened systemic IgE levels |
| [123] | Human keratinocytes and hairless mouse skin (SKH1-E) | EGFR siRNA surrounding a 13-nm gold nanoparticle | siRNA linked nanoparticles led to reduction of EGFR both in culture and after topical delivery in the mouse |
| [124] | Psoriatic and healthy human skin biopsies | Beta-defensin 2 siRNA loaded 100-nm liposomal nanocarrier | Proof of concept beta-defensin 2 knockdown in in vitro keratinocytes from psoriasis patients |
| [127] | TritonX-100-induced dermatitis in reconstructed human epidermis and porcine ears for penetration analysis | IL-6 siRNA loaded 200-270-nm liquid crystalline nanodispersions | Observed penetration of IL-6 siRNA nanocarriers and the subsequent decrease in IL-6 extracellular release |
| [128] | DNFB-induced AD mouse model | RelA siRNA loaded nanosized protein carrier | Decreased ear swelling responses, indicating better dermal outcomes |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palmer, B.C.; DeLouise, L.A. Nanoparticle-Enabled Transdermal Drug Delivery Systems for Enhanced Dose Control and Tissue Targeting. Molecules 2016, 21, 1719. https://doi.org/10.3390/molecules21121719
Palmer BC, DeLouise LA. Nanoparticle-Enabled Transdermal Drug Delivery Systems for Enhanced Dose Control and Tissue Targeting. Molecules. 2016; 21(12):1719. https://doi.org/10.3390/molecules21121719
Chicago/Turabian StylePalmer, Brian C., and Lisa A. DeLouise. 2016. "Nanoparticle-Enabled Transdermal Drug Delivery Systems for Enhanced Dose Control and Tissue Targeting" Molecules 21, no. 12: 1719. https://doi.org/10.3390/molecules21121719
APA StylePalmer, B. C., & DeLouise, L. A. (2016). Nanoparticle-Enabled Transdermal Drug Delivery Systems for Enhanced Dose Control and Tissue Targeting. Molecules, 21(12), 1719. https://doi.org/10.3390/molecules21121719





