Abstract
Some aporphine alkaloids, such as crebanine, were found to present arrhythmic activity and also higher toxicity. A series of derivatives were synthesized by using three kinds of aporphine alkaloids (crebanine, isocorydine, and stephanine) as lead compounds. Chemical methods, including ring-opening reaction, bromination, methylation, acetylation, quaternization, and dehydrogenation, were adopted. Nineteen target derivatives were evaluated for their antiarrhythmic potential in the mouse model of ventricular fibrillation (VF), induced by CHCl3, and five of the derivatives were investigated further in the rat model of arrhythmia, induced by BaCl2. Meanwhile, preliminary structure-activity/toxicity relationship analyses were carried out. Significantly, N-acetamidesecocrebanine (1d), three bromo-substituted products of crebanine (2a, 2b, 2c), N-methylcrebanine (2d), and dehydrostephanine (4a) displayed antiarrhythmic effects in the CHCl3-induced model. Among them, 7.5 mg/kg of 2b was able to significantly reduce the incidence of VF induced by CHCl3 (p < 0.05), increase the number of rats that resumed sinus rhythm from arrhythmia, induced by BaCl2 (p < 0.01), and the number of rats that maintained sinus rhythm for more than 20 min (p < 0.01). Therefore, 2b showed remarkably higher antiarrhythmic activity and a lower toxicity (LD50 = 59.62 mg/kg, mice), simultaneously, indicating that 2b could be considered as a promising candidate in the treatment of arrhythmia. Structural-activity analysis suggested that variationsin antiarrhythmic efficacy and toxicity of aporphines were related to the C-1,C-2-methylenedioxy group on ring A, restricted ring B structural conformation, N-quaternization of ring B, levoduction of 6a in ring C, and the 8-, 9-, 10-methoxy groups on ring D on the skeleton.
1. Introduction
Although radio frequency ablation and other new techniques have been applied in clinical therapy for the treatment of cardiac arrhythmia, drug therapy is still an important and essential method of treatment. According to current methodology of the Vaughan Williams’ electrophysiological classification, available anti-arrhythmic drugs are divided into four categories: Na+ channel blockers (category I), β-receptor blockers (category II), K+ channel blockers (category III), and Ca+ channel blockers (category IV) [1]. Unfortunately, many of the drugs that belong to these four categories have also shown unavoidable side-effects, such as cardiac arrhythmia [2]. However, evidence shows that the new category III drugs, working through multiple ion (Ikr, Iks, Ito, INa) channels, are likely to become the most prospective candidates for the treatment of arrhythmia, due to their higher efficiency, as well as their lower risk of side-effects [3]. Therefore, searching for new components from natural products and their derivatives that act on multiple ion channels seems to be one of the more important approaches in the development of new antiarrhythmic drugs [4].
In the last 30 years, several aprophine-type compounds, such as isocorydine, l-dicentrine, and crebanine, extracted from plants in the subgenus Tuberiphania Lo et M. Yang and the genus Stephania, Menispermaceae, have been found possessing remarkable antiarrhythmic activities [5,6,7]. Based on traditional medical records, some species from this subgenus have been used as medicinal herbs for a long time in Southwest China, with medical functions such as pain-relief, heat-clearing, detoxing, and activating blood circulation [8]. Studies have shown that almost all species of this subgenus contain various types of isoquinoline alkaloids, including aprophines, morphinans, protoberberines, etc., which display extensive physiological activities. They show activity on the K+, Ca2+, and Na+ channel, and some of them have been reported to be blockers of 5-hydroxytryptamine and dopamine receptors [9]. Meanwhile, isoquinoline alkaloids, for example, berberine, l-tetrahydropalmatine, and isocorydine, have been used in clinical treatment of cardiovascular and cranial disease in China. The functions on multiple K+, Ca2+, and Na+ ion channels that these alkaloids exhibit coincides with the current leading trend in the screening of new category III anti-arrhythmic drugs [10,11,12]. In our previous observations, crebanine, an aprophine type alkaloid, showed a higher anti-arrhythmic effect on experimental arrhythmia models compared with isocroydine and l-dicentrine [13]; however, it was also found that the acute toxicity of crebanine was higher than that of isocroydine and l-dicentrine. The effective arrhythmia dosage (2.5~5.0 mg/kg) of crebanine was close to its LD50 (9.38 mg/kg) in mice [14], which implies that crebanine has a relatively narrow therapeutic window.
The present study is a further investigation regarding the structural modification and evaluation of the arrhythmic activities of aprophine alkaloids. The authors have attempted to find new aprophine derivatives with higher antiarrhythmia efficiencies and lower toxicities. A series of derivatives from three kinds of aporphine (crebanine, isocroydine, and stephanine) were designed and synthesized by connecting specific pharmacophores to enlarge the target distribution of antiarrhythmia. At the same time, hydrophobic groups or lipophilic groups were introduced in order to improve the lipid-water partition coefficient and membrane permeability. The antiarrhythmic effects of the derivatives were preliminarily evaluated in a mouse model of ventricular fibrillation (VF), induced by CHCl3, and further confirmed in a rat model of arrhythmia, induced by BaCl2. Six derivatives were obtained, and are reported here for the first time, with antiarrhythmic effects and the structure-activity relationships are discussed.
2. Results and Discussion
Nineteen products, including 13 derivatives of crebanine, four derivatives of isocorydine, and two derivatives of stephanine, were synthesized, isolated, purified, and identified (Scheme 1, Scheme 2, Scheme 3, Scheme 4 and Scheme 5), of which 11 are reported for the first time: Secocrebanine (1a), N-cyclopropylmethylsecocrebanine (1b), N-trifluoroacetamidesecocrebanine (1c), N-acetamidesecocrebanine (1d), N-methylsulfonamidesecocrebanine (1e), 3,10,11-Tribromocrebanine (2a), 10,11-Dibromocrebanine (2b), 3-Bromocrebanine (2c), 11-Methoxy-N-methylisocorydine (5a), 11-Methoxyisocorydine (5b), and 11-Ethoxyisocorydine (5c).

Scheme 1.
Synthesis of crebanine derivatives 1a, 1b, 1c, 1d, and 1e. Reagents and conditions: (i) 1-chloroethyl chloroformate, K2CO3, 1,2-CH2ClCH2Cl, 85 °C, 6 h, MeOH, 37% HCl, refluxed for 2 h; (ii) cyclopropylmethyl bromide, K2CO3, MeCN, 80 °C, 4 h; (iii) trifluoroacetic anhydride, Et3N, anhydrous CH2Cl2, 2 h, r.t.; (iv) acetic anhydride, 4-dimethylaminopyridine, Et3N, anhydous CH2Cl2, r.t., 4 h; and (v) methanesulfonyl chloride, Et3N, anhydrous CH2Cl2, r.t., 2 h.

Scheme 2.
Synthesis of crebanine analogues 2a, 2b, 2c, 2d, and 2e. Reagents and conditions: (i) N-Bromosuccinimide, trifluoroacetic acid, r.t., 18 h; (ii) ether, CH3I, MeOH, r.t., 16 h; and (iii) AlBr3, anhydrous CH2Cl2, nitrobenzene, 5 °C, argon, 18 h.

Scheme 3.
Synthesis of crebanine and stephanine derivatives 3a, 3b, 4a and 4b. Reagents and conditions: (i) 10% Pd/C, MeCN, N2, refluxed for 6 h; and (ii) NaCNBH3, EtOH, 2NHCl, r.t., 18 h.

Scheme 4.
Synthesis of isocorydine derivatives 5a, 5b, 5c, and 5d. Reagents and conditions: (i) Tetrabutylammonium bromide, K2CO3, anhydrous THF, CH3I, r.t., 8 h; (ii) N, N-diisopropylethylamine, trimethylsilyldiazomethane, MeCN, MeOH, 15 h, r.t.; and (iii) Tetrabutylammonium bromide, K2CO3, anhydrous THF, CH3CH2I, r.t., 8 h; and (iv) ether, CH3I, MeOH, r.t., 16 h.
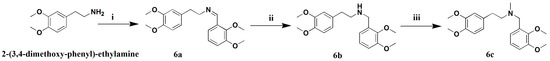
Scheme 5.
Synthesis of crebanine analogues 6c. Reagents and conditions: (i) 2,3-dimethoxy-benzoic acid , methylbenzene, 120 °C, 8 h; (ii) 5%Pd/C, MeOH, H2, 1 bar, 8 h; and (iii) K2CO3, anhydrous THF, CHI, r.t., 4 h.
The antiarrhythmic activities of the 19 products, respectively, were tested in the mouse model of ventricular fibrillation (VF), induced by CHCl3. Six compounds, 1d, 2a, 2b, 2c, N-methylcrebanine (2d), and dehydrostephanine (4a), were found to possess significant antiarrhythmic effects (p < 0.05) (Figure 1A–D). Furthermore, in the rat model of arrhythmia, induced by BaCl2, 2a, 2b, 2c, 2d, and 4a demonstrated significant antiarrhythmia activity (Table 1). Lastly, in order to evaluate toxicity, the LD50 of three active compounds, two bromo-substituted products of crebanine (2a, 2b) and N-methylcrebanine (2d), were calculated using the sequential method in mice (Table 2) as 59.62, 65.15, and 6.432 mg/kg, respectively.

Figure 1.
(A–D) Antiarrthymic activity of 19 aporphine derivatives in a mouse VF model, induced by CHCl3 (n = 10). Data represent the incidence percentage of VF in 10 experiments. Significantly different from the control group, * p < 0.05, ** p < 0.01. 1d, 2a, 2b, 2c, 2d, and 4a possess significant antiarrhythmic effects.

Table 1.
Antiarrhythmic activity of fiveaporphine derivatives (2a–d and 4a) in the model of arrhythmia, induced by BaCl2 in anesthetized SD rats (iv) (n = 10).

Table 2.
Acute toxicity tests of three crebanine derivatives (2b, 2c, and 2d) in mice (n = 10).
2.1. Opening up of the N-C Bondon Ring B and N-Substitution and Antiarrhythmic Activity/Toxicity
Previously, we characterized some new type III anti-arrhythmic drugs, such as dronedarone [15], most of which contained the N-substitution structure [3]. Therefore, we presumed that the N-substitution in ring B of aporphine might be one of the key reasons for their anti-arrhythmic activities. Five phenanthrene compounds were synthesized (Scheme 1) by opening up the N-C bond (1a) on ring B of crebanine, further connecting the N position with four kinds of lipophilic groups (1b–1e). The five obtained analogues were tested in the mouse model of VF, induced by CHCl3 (Figure 1A). The result showed that, except for compound 1d (10 mg/kg), which was able to return the occurrence of VF to a normal sinus rhythm in 60% of tested mice (p < 0.01) (Figure 1A), the remaining four ring B-opening derivatives (1a, 1b, 1c, and 1e) showed no significant effects on inhibiting rapid VF in mice, even though the test dosages (10–40 mg/kg) were up to eight times higher than the dosage of crebanine (5 mg/kg). Simultaneously, the tested mice also showed no obvious toxic symptoms. These results indicate that antiarrhythmic efficiency and toxicity were both decreased when ring B of crebanine was opened up. Therefore, it is speculated that a proper, closed ring B structure of aporphine was necessary, as the flat structure of the restricted ring B structural conformation could embed into myocardial cells more easily than the ring B-opening structure of phenanthrenes.
2.2. The Lipophilicity/Hydrophilicity of Crebanine Derivatives and Antiarrhythmic Activity/Toxicity
Bromo-substituted derivatives often increase the lipophilicity of compounds. Bromine atoms were first introduced into the structure of crebanine (Scheme 2) to explore the relationship between lipophilicity and the antiarrhythmic activity/toxicity of aporphines. Compared with normal saline, compounds 2a, 2b, and 2c, three kinds of crebanine bromoderivatives, exhibited a remarkable recovery in mouse VF (p < 0.05) (Figure 1B). Notably, dibromide 2b of crebanine displayed better antiarrhythmic activity than tribromide of crebanine 2a and monobromide of crebanine 2c (Table 1). After a 7.5 mg/kg dosage of 2b was given, the number of animals that recovered, and the number of animals maintaining sinus rhythm ≥20 min was obviously higher than those of 2a (15 mg/kg) and 2c (7.5 mg/kg) in the rat model of arrhythmia, induced by CHCl3 (Figure 1B). The results indicate that the different numbers of Br replacements varied the lipid solubility of crebanine and changed the level of antiarrhythmic efficiency. Furthermore, the LD50 of 2c (65.15 mg/kg) and 2b (59.62 mg/kg) in mice (Table 2) were much higher than that of crebanine (LD50 9.38 mg/kg) [14], which showed that toxicity of brominated crebanine was much lower than that of crebanine. This predicts that brominated crebanine may be a prospective modified structure and would serve as a lead compound for antiarrhythmia.
On the other hand, N-methylation (quaternization) was introduced into the structure of crebanine in order to determine how the improvement of hydrophilicity in aporphines impacted the antiarrhythmic activity and toxicity. The quaternary ammonium of crebanine (2d, Scheme 2) displayed a notable antiarrhythmic effect, even at a lower dosage of 3 mg/kg (Figure 1B), but the tested mice showed symptoms such as polypnea, mydriasis, leaping, and tremors until death, which indicate toxicity of the central nervous system (CNS) and the motor nervous system [16]. The LD50 of 2d was calculated to be 6.432 mg/kg in mice (Table 2), much lower than that of crebanine (9.382 mg/kg), which indicated that N-quaternizated aporphines could maintain anti-arrhythmic activity, but that the toxicity was increased at the same time.
2.3. Ring D O-Demethylation and Antiarrhythmic Activity
The experiments were designed to remove the O-methyl groups in ring D of crebanine in order to acquire more aporphine derivatives (Scheme 2). Table 3 shows all of reactions and their derivatives [17,18,19,20,21,22]. Since ring B was unstable and easy to open using strong acids, it was difficult to cleave the O-methyl groups in ring D when only strong acids (48%HBr/CH3COOH, 48%HBr, and IH) were used [17,18,19]. Otherwise, when crebanine was treated with BBr3 in dichloromethane, a molecular ion peak of the products was observed at m/z 300 [M + H]+, which implies that both the O-methyl groups and methylenedioxy of crebanine had been removed simultaneously. Meanwhile, the target product of the four phenolic hydroxyl groups was not acquired due to its high hydrophilicity and instability. A 9-demethyl group derivative, namely stesakine (2e, Scheme 2) [20] in Table 3, was obtained by utilizing the Lewis acids, BCl3, and AlBr3 [21,22], which indicated that the 9-methoxy group in ring D of crebanine was less stable than the 8-methoxy group.

Table 3.
O-Demethylation reaction of crebanine.
Compound 2e showed weak activity at a maximum dissolvable dosage (2.5 mg/kg) in the mouse VF model (Figure 1B), thus, the 9-methoxy group in ring D might be one of the functional groups in aporphine alkaloids. Similarly, the 8- and/or 9- or 10-methoxylation in ring D of the aporphine skeleton, such as crebanine, stephanine, and l-isocrydine, showed notable antiarrhythmic activity. On the contrary, the removal of the 8- or 9- or 10-methyl groups of aporphine alkaloids in ring D may reduce antiarrhythmic activity.
2.4. Racemization of Aporphines and Antiarrhythmic Activity
Two racemic compounds of crebanine and stephanne, namely (±) crebanine (3b) and (±) stephanine (4b), were synthesized by reduction after dehydrogenation of 6a-H (Scheme 3) [23]. Both compounds did not significantly manifest antiarrhythmic activity in the mouse model of VF induced by CHCl3 (Figure 1C). The results indicated that the levoduction of the 6a configuration was a functional group for maintaining antiarrhythmic activity.
2.5. 6a-H Dehydrogenated Derivatives of Aporphines and Antiarrhythmic Activity/Toxicity
Dehydrocrebanine (3a) was derived from dehydrogenating in 6a-H of crebanine and the formation of a double bond in ring C of crebanine (Scheme 3). 3a showed a very high toxicity in animal tests. In order to avoid animal poisoning and death, the test dosage had to be decreased to 1 mg/kg (Figure 1C); however, antiarrhythmic activity was not obviously observed in the CHCl3-induced VF model either. On the other hand, stephanine and its derivative of 6a-H dehydrogenation (dehydrostephanine, 4a) (5 mg/kg), was able to notably confront VF, caused by CHCl3, in mice (Figure 1C). In the rat model of arrhythmia induced by BaCl2, both stephanine and 4a increased the number of animals that resumed normal sinus rhythm (p < 0.01), and significantly shortened the time for recovery. However, 4a was unable to maintain a sinus rhythm, even after more than 3 min post recovery (Table 1), and most of the tested animals died during the observation period (20 min), which implies that the effective duration of 4a was much shorter and the toxicity was high. These findings may be explained by the fact that dehydrogenation compounds increase the rigidity and conjugacy of the skeleton due to ring C forming a double bond (3a, 4a), which facilitated the infusion of aporphine molecules infusing myocardial cells, and consequently producing much faster and stronger toxic effects relative to the prototype compounds.
2.6. 11-Alkylation and Quaternization of Isocorydine
Figure 1D and Table 1 show that isocorydine (10 mg/kg) displayed lower antiarrhythmic activity tendencies compared to crebanine (5 mg/kg) and stephanine (5 mg/kg). The main difference in these three compounds lies on the 1,2-dimethoxy group of ring A, which exists in crebanine and stephanine, but not in isocorydine. This increase in antiarrhythmic activity may be related to the 1,2-dimethoxy group structure, which widens the planar configuration of molecules.
All of the modified products of isocorydine, 5a, 5b, 5c, and 5d (Scheme 4) [24,25] were inactive in the model of VF induced by CHCl3 (Figure 1D) at appropriate test dosages. Meanwhile, 5a and 5d were both N-methylated as quaternary ammonium compounds, and presented such a high toxicity that the test dosages had to be reduced to 1 mg/kg (5a) and 0.8 mg/kg (5d) (Table 1) to avoid animal death. The reactions of toxicity appeared, much like those of N-methylcrebanine (2d), in the central nervous system and the motor nervous system. The experiments also evidenced that the substitution of the 11-methoxy group (5b) or 11-ethoxy group (5c) of isocorydine did not have any benefits for antiarrhythmic activity.
2.7. Design of An Analogue of Crebanine
An analogue of crebanine, compound 6c, was designed and synthesized according to the key group of crebanine (Scheme 5), which contained two basic sites, a tertiary amine group and a dimethoxy-benzene group. However, animal tests showed that 6c could not significantly inhibit the occurrence of VF induced in mice. Therefore, it is necessary to design a series of dimethoxy-phenylamine derivatives in order to seek out the potential active compound.
3. Experimental Section
3.1. Synthesis
3.1.1. General Information
Isocorydine, crebanine, and stephanine were extracted from the plant, identified as Stephania yunnanensis Lo by Professor Yunshu Ma, Yunnan University of Traditional Chinese Medicine, and as reported previously [26,27]. Solvents and reagents used in syntheses were of analytical grade, were purchased from commercial sources, and used without further purification.
All reactions were carried out under an atmosphere of argon, with magnetic stirring, in flame dried or oven-dried glassware. Analytical thin layer chromatography was performed on 0.25-mm silica gel GF 254 plates (Qingdao Haiyang Chemical Co., Ltd., Qingdao, China). Visualization was accomplished with UV light and bismuth potassium iodide solution staining followed by heating. 1H-NMR spectra were recorded on a 400 MHz spectrometer (Amersham Pharmacia Biotech AB Inc., Tokyo, Japan) in CDCl3 or CD3OD at ambient temperature, using the solvent signal as an internal standard. Data are reported as: (br/broad, s/singlet, d/doublet, t/triplet, q/quartet, m/multiplet; integration; coupling constant(s) in hertz). 13C-NMR spectra were recorded on a 400 MHz spectrometer (Amersham Pharmacia Biotech AB Inc., Tokyo, Japan) in CDCl3 at ambient temperature, using the solvent signal as an internal standard. Electrospray ionization mass spectrometry was detected with a Brukerama Zon SL (Bruker Daltonics Inc., Leipzig, Bremen, Germany); HR-MS was detected with 6200 series TOF/6500 series (Agilent Technologies Inc., Santa Clara, UT, USA); rotation was detected with a WZZ-three digital automatic polarimeter (Shanghai Shengguang Technology Co., Ltd., Shanghai, China).
3.1.2. Preparation of Secocrebanine (1a)
A mixture of crebanine (2 g, 5.9 mmol) and K2CO3 (1.7 g, 1.2 mmol) in 1,2-dichloroethane (20 mL) was stirred and 1-chloroethyl chloroformate (1.0 g, 7.1 mmol) was added slowly. The mixture was heated to 85 °C for 6 h, the 1,2-dichloroethane-insoluble material was filtered off, and the filtrate was concentrated to give the residue, which was dissolved in MeOH (20 mL) and five drops of 37% HCl, and refluxed for 2 h. The solution was concentrated and neutralized with saturated Na2CO3, and the residue was extracted with CH2Cl2. The combined extracts were dried over anhydrous Na2SO4, filtered, and concentrated to get a crude product, which was crystallized from EtOAC at 4 °C, yielding compound 1a (1.2 g, 60%) as a pink-white solid. 1H-NMR (400 MHZ, CDCl3, δ; ppm) 8.83 (d, J = 9.2 HZ, 1H, 5-H), 7.93 (d, J = 9.6 HZ, 1H, 10-H), 7.87 (d, J = 9.6 HZ, 1H, 9-H), 7.29 (d, J = 8.8 HZ, 1H, 6-H), 7.10 (s, 1H, 2-H), 6.22 (s, 2H, -OCH2O-), 4.02 (s, 3H, 8-OCH3), 4.00 (s, 3H, 9-OCH3), 3.28–3.24 (t, J = 7.2 HZ, 2H, -CH2α), 2.97–2.93 (t, J = 7.2 HZ, 2H, -CH2β), 2.47 (s, 3H, -NCH3); 13C-NMR (100 MHZ, CDCl3, δ; ppm) 33.87 (t, -CH2β), 36.43 (q, N-CH3), 53.12 (t, -CH2α), 56.30 (q, 7-OCH3), 61.32 (q, 8-OCH3), 100.95 (t, -OCH2O-), 110.05 (d, C-2), 112.60 (d, C-6), 117.11 (s, C-8a), 118.38 (d, C-5), 123.26 (s, C-4a), 123.64 (d, C-9), 123.79 (d, C-10), 125.19 (s, C-1a), 127.41 (s, C-5a), 130.88 (s, C-1), 141.76 (s, C-4), 143.23 (s, C-7), 144,98 (s, C-3), 149.96 (s, C-8), Positive ESI-MS m/z: 340.2 [M + H]+. HR-MS for C20H21NO4 [M + H]+; calcd. 340.1543, found: 340.1542.
3.1.3. Preparation of N-Cyclopropylmethylsecocrebanine (1b)
Product 1a (200 mg, 0.59 mmol), K2CO3 (163 mg, 1.18 mmol), and cyclopropylmethyl bromide (96 mg, 0.71 mmol) were added to MeCN (20 mL) and heated at 80 °C for 4 h. The reaction mixture was filtered and concentrated to give a residue, which was purified by silica column chromatography (CHCl3/MeOH 80:1), yielding compound 1b (180 mg, 78%) as a white solid. 1H-NMR (400 MHZ, CDCl3, δ; ppm) 8.81(d, J = 9.2 HZ, 1H, 5-H), 8.04 (d, J = 9.6 HZ, 1H, 10-H), 7.93 (d, J = 9.2 HZ, 1H, 9-H), 7.31 (d, J = 8.4 HZ, 1H, 6-H), 7.16 (s, 1H, 2-H), 6.23 (s, 2H, -OCH2O-), 4.02 (s, 3H, 8-OCH3), 3.99 (s, 3H, 9-OCH3), 3.84–3.78 (m, 1H, CH2α), 3.66–3.60 (m, 1H, -CH2β), 3.47–3.42 (m, 1H, -CH2β), 3.18–3.15 (m, 2H, N-CH2), 2.97 (s, 3H, -NCH3), 2.93–2.89 (m, 1H, CH2α), 1.30–1.25 (m, 1H, -CH), 0.85–0.82 (m, 2H, -CH2), 0.50–0.47 (m, 1H, -CH2). .13C-NMR (100 MHZ, CDCl3, δ; ppm) 100.62 (t, -OCH2O-), 52.82 (t, -CH2β), 29.59 (t, CH2α), 29.26 (t, -CH2), 29.05 (t, -CH2), 61.00 (q, 8-OCH3), 56.02 (q, 8-OCH3), 43.97 (q, N-CH3), 115.77 (d, C-5), 114.88 (d, C-6),62.40(d, -CH), 152.55 (s, C-8), 145.17 (s, C-3), 145.02 (s, C-7), 141.99 (s, C-4), 133.19 (s, C-1),130.69 (s, C-5a), 125.73(s, C-1a), 124.66 (s, C-4a), 114.88(s, C-8a). Positive ESI-MS m/z: 394.2 [M + H]+. HR-MS for C24H27NO4 [M + H]+; calcd. 394.2013, found: 394.2026.
3.1.4. Preparation of N-Trifluoroacetamidesecocrebanine (1c)
A solution of 1a (200 mg, 0.59 mmol) and Et3N (72 mg, 0.71 mmol) in anhydrous CH2Cl2 (30 mL), trifluoroacetic anhydride (149 mg, 0.71 mmol) was slowly added under an ice-bath, then, the solution was stirred for 2 h at room temperature. The mixture was diluted with water (20 mL) and extracted with CH2Cl2 (3 × 10 mL). The combined extracts were dried over anhydrous Na2SO4, filtered and concentrated to give a crude product, which was purified by silica gel column chromatography (CH2Cl2/MeOH 100:1), and yielding compound 1c (210 mg, 82%) as a white solid. 1H-NMR (400 MHZ, CDCl3, δ; ppm) 8.83 (d, J = 9.2 HZ, 1H, 5-H), 7.99 (d, J = 9.2 HZ, 1H, 10-H), 7.94 (d, J = 9.6 HZ, 1H, 9-H), 7.31 (d, J = 9.2 HZ, 1H, 6-H), 7.07 (s, 1H, 2-H), 6.24 (s, 1H, -OCH2O-), 6.23 (s, 1H, -OCH2O-), 4.03 (s, 3H, 8-OCH3), 4.00 (s, 3H, 9-OCH3), 3.73–3.69 (t, J = 7.4 HZ, 2H, -CH2α), 3.36–3.32 (t, J = 7.2 HZ, 2H, -CH2β), 3.11 (s, 1H, -NCH3), 2.99 (s, 2H, -NCH3). 13C-NMR (100 MHZ, CDCl3, δ; ppm) 30.67 (t, CH2α), 36.04 (q, N-CH3), 51.79 (t, -CH2β ), 56.32 (q, 7-OCH3), 61.33(q, 8-OCH3), 101.21 (t, -OCH2O-), 110.13 (d, C-2), 112.91 (d, C-6), 117.15 (s, C-8a), 119.12 (s, C-4a), 119.28 (d, C-5), 122.08 (s, CF3), 123.77 (d, C-9), 123.81 (d, C-10), 125.39 (C-1a), 127.47 (C-5a), 128.83 (s, C-1), 142.27 (s, C-4), 143.32 (s, C-7), 145.08 (s, C-3), 150.15 (s, C-8). Positive ESI-MS m/z: 458.2 [M + Na]+. HR-MS for C22H20F3NO5 [M + Na]+; calcd. 458.1186, found: 458.1188.
3.1.5. Preparation of N-Acetamidesecocrebanine (1d)
To a stirred solution of 1a (200 mg, 0.59 mmol), 4-dimethylaminopyridine (144 mg, 1.18 mmol), and Et3N (72 mg, 0.71 mmol) in anhydrous CH2Cl2 (20 mL), acetic anhydride (72 mg, 0.71mmol) was slowly added and stirred for 4 h at room temperature. The mixture was diluted with water (10 mL) and extracted with CH2Cl2 (3 × 10mL). The combined extracts were dried over anhydrous Na2SO4, filtered, and concentrated to get a crude product, which was purified by silica gel column chromatography (CH2Cl2/MeOH 100:1), yielding compound 1d (180 mg, 80%) as a white solid. 1H-NMR (400 MHZ, CDCl3, δ; ppm) 8.83 (d, J = 8.4 HZ, 1H, 5-H), 8.00 (d, J = 10.0 HZ, 1H, 10-H) 7.96 (d, J = 9.6 HZ, 1H, 9-H), 7.32 (d, J = 9.2 HZ, 1H, 6-H), 7.09 (s, 1H, 2-H), 6.23 (s, 1H, -OCH2O-), 6.22 (s, 1H, -OCH2O-), 4.03 (s, 6H, 8-OCH3), 4.00 (s, 6H, 9-OCH3), 3.67–3.61 (dd, J = 15.6 HZ, J = 8.0 HZ, 2H, -CH2α), 3.31–3.27 (t, J = 7.4 HZ, 2H, -CH2β), 3.00 (s, 1H, -NCH3), 2.81 (s, 2H, -NCH3), 2.09 (s, 2H, -NOCH3), 1.85 (s, 1H, -NOCH3). 13C-NMR (100 MHZ, CDCl3, δ; ppm) 22.03 (q, -NOCH3), 32.57 (t, -CH2α), 37.39 (q, N-CH3), 51.99 (t, -CH2β), 56.33 (q, 7-OCH3), 61.32 (q, 8-OCH3), 100.08 (t, -OCH2O-), 110.28 (d, C-2), 112.88 (d, C-6), 117.17 (s, C-8a), 119.10 (d, C-5),123.76 (d, C-9), 123.81 (d, C-10), 125.55 (s, C-4a), 127.50 (C-1a), 128.72 (C-5a), 130.31 (s, C-1), 142.30 (s, C-4), 143.28 (s, C-7), 145.14 (s, C-3), 150.11 (s, C-8), 170.69 (s, -C=O).Positive ESI-MS m/z: 404.2 [M + Na]+.HR-MS for C22H23NO5 [M + Na]+; calcd. 404.1468, found: 404.1474.
3.1.6. Preparation of N-Methylsulfonamidesecocrebanine (1e)
To a mixture of 1a (200 mg, 0.59 mmol) and Et3N (72 mg, 0.71 mmol) in anhydrous CH2Cl2 (30 mL) under an ice-bath, methanesulfonyl chloride (81 mg, 0.71 mmol) was slowly added and stirred for 2 h at room temperature. The mixture was diluted with water (20 mL) and extracted with CH2Cl2 (3 × 10 mL). The combined extracts were dried over anhydrous Na2SO4, filtered, and concentrated to give a crude product, which was purified by silica gel column chromatography (CH2Cl2/MeOH 100:1), yielding 1e (210 mg, 82%) as a white solid. 1H-NMR (400 MHZ, CDCl3, δ; ppm) 8.83 (d, J = 9.2 HZ, 1H, 5-H), 7.98 (d, J = 9.6 HZ, 1H, 10-H), 7.85 (d, J = 9.6 HZ, 1H, 9-H), 7.31(d, J = 9.2 HZ, 1H, 6-H), 7.11 (s, 1H, 2-H), 6.23 (s, 2H, -OCH2O-), 4.03 (s, 3H, 8-OCH3), 4.00 (s, 3H, 9-OCH3), 3.46–3.42 (m, 2H, -CH2α), 3.37–3.34 (m, 2H, -CH2β), 2.92 (s, 3H, -NCH3), 2.74 (s, 3H, -NSO2CH3). 13C-NMR (100 MHZ, CDCl3, δ; ppm) 33.26 (t, -CH2α), 35.38 (t, -NSO2CH3), 51.56 (t, -CH2β), 56.31 (q, 7-OCH3), 61.34 (q, 8-OCH3), 101.10 (t, -OCH2O-), 110.30 (d, C-2), 112.76 (d, C-6),117.13 (s, C-8a), 119.06 (d, C-5),122.72 (d, C-9),123.77 (d, C-10),125.21 (s, C-4a),127.40 (C-1a), 128.88 (s, C-1), 142.20 (s, C-4),143.29 (s, C-7), 145.07 (s, C-3),150.07 (s, C-8). Positive ESI-MS m/z: 440.2 [M + Na]+. HR-MS for C21H23NO6S [M + Na]+; calcd. 440.1138, found: 440.1140.
3.1.7. Preparation of 3,10,11-Tribromocrebanine (2a), 10,11-Dibromocrebanine (2b) and 3-Bromocrebanine (2c)
Crebanine (200 mg, 0.59 mmol) and N-Bromosuccinimide (213.6 mg, 1.2 mmol) were added to trifluoroacetic acid (5 mL), the mixture was stirred for 18hat room temperature, diluted with water (10 mL), and neutralized with saturated NaHCO3 and extracted with CH2Cl2 (3 × 10 mL). The combined CH2Cl2 solution was brined with water, dried over anhydrous Na2SO4, filtered, and concentrated to give a crude product, which was purified by silica gel column chromatography (CH2Cl2/MeOH 200:1→100:1), yielding compound 2a (52 mg, 27%), compound 2b (50 mg, 17%), and compound 2c (80 mg, 33%).
Compound 2a, pale yellow solid. 1H-NMR (400 MHZ, CDCl3, δ; ppm) 6.16 (1H, s, 9, -OCH2O-), 6.06 (1H, s, -OCH2O-), 3.93 (3H, s, 8-OCH3), 3.87 (3H, s, 9-OCH3), 3.54 (1H, d, J = 12.8 HZ, 6a-H), 3.16–3.09 (m, 1H, 7-Ha), 2.99–2.91 (m, 2H, 5-Ha, 7-Hb), 2.77–2.72 (m, 1H, 5-Ha), 2.60 (s, 3H, -NCH3), 2.53–2.50 (m, 1H, 4-Ha), 2.15–2.02 (m, 1H, 4-Hb). 13C-NMR (100 MHZ, CDCl3, δ; ppm), 29.29 (t, C-7), 29.33 (t, C-4), 52.78 (t, C-5), 44.02 (t, -NCH3), 60.82 (q, 9-OCH3), 61.17 (q, 8-OCH3), 62.18 (d, C-6a), 100.77 (t, -OCH2O-),102.96 (s, C-3), 115.29 (s, C-10), 118.63 (s, C-11), 125.78 (s, C-7a), 129.83 (s, C-1b), 130.73 (s, C-3a), 132.67 (s, C-11a), 142.26 (s, C-1), 145.28 (s, C-8), 149.72 (s, C-2), 150.88 (s, C-9a). Positive ESI-MS m/z: 579.2 [M + 3]+. HR-MS for C20H18Br3NO4 [M + H]+; calcd.573.8859, found:573.8853.
Compound 2b, white solid. 1H-NMR (400 MHZ, CDCl3, δ; ppm) 7.13 (s, 1H, 3-H), 6.14 (s, 1H, -OCH2O-), 6.05 (s, 1H, -OCH2O-), 3.89 (s, 3H, 8-OCH3), 3.79 (s, 3H, 9-OCH3), 3.61–3.57 (m, 1H, 6a-H), 3.11–3.07 (m, 1H, 7-Ha), 2.97–2.87 (m, 2H, 5-Ha, 7-Hb), 2.80–2.66 (m, 1H, 5-Hb), 2.56 (s, 3H, -NCH3), 2.52–2.47 (m, 1H, 4-Ha), 2.11–2.04 (t, J = 13.6 HZ, 1H, 4-Hb). 13C-NMR (100 MHZ, CDCl3, δ; ppm) 100.62 (t, -OCH2O-), 52.82 (t, C-5), 29.26 (t, C-4), 29.05 (t, C-7), 62.40 (d, C-6a), 115.95 (d, C-3), 152,55 (s, C-9), 145.17 (s, C-2), 145.09 (s, C-8), 141.99 (s, C-1), 133.19 (s, C-11a), 130.69 (s, C-3a), 125.73 (s, C-7a), 124.66 (s, C-1a). Positive ESI-MS m/z: 498.0 [M + H]+. HR-MS for C20H19Br2NO4 [M + H]+; calcd. 495.9754, found: 495.9759.
Compound 2c, white solid. Positive ESI-MS m/z: 418 [M]+, 420.0 [M + 2]+. 1H-NMR (400 MHZ, CDCl3, δ; ppm) 7.76 (d, J = 8.8 HZ, 1H, 11-H), 6.90 (d, J = 8.8 HZ, 1H, 10-H), 6.22 (s, 1H, -OCH2O-), 6.07(s, 1H, -OCH2O-), 3.91 (s, 3H, 8-OCH3), 3.84 (s, 3H, 9-OCH3), 3.82–3.80 (m, 1H, 6a-H), 3.09-3.04 (m, 2H, 5-Ha, 7-Hb), 2.90–2.82 (m, 2H, 5-Ha, 7-Hb), 2.17 (s, 3H, -NCH3), 1.95–1.87 (m, 1H, 4-Ha), 1.79–1.72 (m, 1H, 4-Hb).
3.1.8. Preparation of N-Methylcrebanine (2d)
A mixture of crebanine (200 mg, 0.59 mmol), ether (109 mg, 1.48 mmol) and CH3I (184.5 mg, 1.3 mmol) in MeOH (10 mL) was stirred for 16 h at room temperature. The reaction mixture was concentrated and extracted with CHCl3 (3 × 10 mL). The combined extracts were dried over anhydrous Na2SO4, filtered, and concentrated to give a crude product, which was purified by silica gel column chromatography (CH2Cl2/MeOH 30:1), yielding compound 2d (180 mg, 86%) as a white solid. 1H-NMR (400 MHZ, CDCl3, δ; ppm) 7.70 (d, J = 8.8 Hz, 1H, 11-H), 7.05 (d, J = 8.8 Hz, 1H, 10-H), 6.78 (s, 1H, 3-H), 6.18 (s, 1H, -OCH2O-), 5.98 (s, 1H, -OCH2O-), 4.74–4.70 (m, 1H, 6a-H), 3.80 (s, 3H, 8-OCH3), 3.76–3.73 (m, 1H, 7-Ha), 3.72 (s, 3H, 9-OCH3), 3.64–3.55 (m, 2H, 7-Hb,5-Ha), 3.33 (s, 3H, -NCH3), 3.18–3.09 (m, 1H, 5-Hb), 2.97 (s, 3H, -NCH3), 2.94–2.89 (m, 1H, 4-Ha), 2.70–2.63 (t, J = 14.2 Hz, 1H, 4Hb). Positive ESI-MS m/z: 354.2 [M]+.
3.1.9. Preparation of Stesakine (2e)
Crebanine (200 mg, 0.59 mmol) in 2mL of anhydrous CH2Cl2 was slowly added into a suspension of AlBr3 (320 mg, 1.2 mmol) in nitrobenzene (5 mL) at 5 °C under argon. The solution was stirred for 18 h, diluted with water (20 mL) and neutralized with saturated NaHCO3. The mixture was extracted with CH2Cl2 (3 × 10 mL), and the combined CH2Cl2 solution was brined with water, dried with anhydrous Na2SO4, filtered, and concentrated to give a crude product, which was purified by silica gel column chromatography (CH2Cl2/MeOH 50:1), yieldingcompound 2e (56 mg, 29%) as a colorless oil. 1H-NMR (400 MHZ, CDCl3, δ; ppm) 7.63 (d, J = 8.8 HZ, 1H, 11-H), 6.83 (d, J = 8.8 HZ, 1H, 10-H), 6.53 (s, 1H, 3-H), 6.06 (s, 1H, -OCH2O-), 5.92 (s, 1H, -OCH2O-), 5.77 (s, 1H, 9-OH), 3.93 (s, 3H, 8-CH3), 3.69–3.64 (dd, J = 16.0 HZ, J = 4.0 HZ, 1H, 6a-H), 3.19–3.03 (m, 3H, 7-H, 5-Ha), 2.65–2.61 (m, 1H, 5-Hb), 2.60 (s, 3H, -NCH3), 2.56–2.50 (m, 1H, 4-Ha), 2.32–2.25 (t, J = 10.4 HZ, 1H, 4-Hb), 13C-NMR (100 MHZ, CDCl3, δ; ppm) 142.2 (s, C-1), 116.7 (s, C-1a), 126.5 (s, C-1b), 146.6 (s, C-2), 106.7 (d, C-3), 126.7 (s, C-3a), 26.3 (t, C-4), 56 (t, C-5), 61.9 (d, 6a), 29.2 (t, C-7), 124.8 (s, C-7a), 156.4 (s, C-8), 145.8 (s, C-9), 108.5 (d, t-10), 118.7 (d, C-11), 126.6 (s, C-11a), 100.1 (t, -CH2O-), 44.0 (q, -NCH3), 56.0 (q, 8-OCH3). Positive ESI-MS m/z: 326 [M + H]+.
3.1.10. Preparation of Dehydrocrebanine (3a)
Pd/C (10%, 210 mg) was added to a suspension of crebanine (200 mg, 0.59 mmol) in MeCN (20 mL), and the solution was refluxed for 6 h under N2. The mixture reaction solution was filtered and the filtrate was evaporated to give compound 3a (181 mg, 91%) as a yellowish-green solid. 1H-NMR (400 MHZ, CDCl3, δ; ppm) 8.66 (d, J = 8.8 Hz, 1H, 11-H), 7.03 (d, J = 9.2 Hz, 1H, 10-H), 6.89 (s, 2H, 3-H, 7-H), 6.19 (s, 2H, -OCH2O-), 3.99 (s, 3H, 8-OCH3), 3.97 (s, 3H, 9-OCH3), 3.34–3.37 (t, J = 5.8 Hz, 2H, 5-H), 3.24–3.31 (t, J = 5.8 Hz, 2H, 4-H), 3.13 (s, 3H, -NCH3). Positive ESI-MS m/z: 348.2 [M + H]+.
3.1.11. Preparation of (±) Crebanine (3b)
A mixture of 3a (150 mg, 0.44 mmol) and NaCNBH3 (36 mg, 0.57 mmol) in absolute EtOH was stirred, and a mixture of EtOH and 2NHCl was added until the pH approached 3.0 (within 4 h). Then, the solution was continuously stirred for 18 h at room temperature. After evaporation of the reaction mixture, the pH was adjusted to 8 with saturated Na2CO3 and the mixture solution was extracted with CH2Cl2 (3 × 10 mL). The combined extracts were dried over anhydrous Na2SO4, filtered, and concentrated to get a crude product, which was purified by silica gel column chromatography (CH2Cl2/MeOH 50:1), yielding compound 3b (120 mg, 80%) as a yellow oil. : 0° (C 0.2, MeOH). 1H-NMR (400 MHZ, CDCl3, δ; ppm) 7.81 (d, J = 8.8 Hz, 1H, 11-H), 6.88 (d, J = 8.8 Hz, 1H, 10-H), 6.53 (s, 1H, 3-H), 6.07 (s, 1H, -OCH3O-), 5.90 (s, 1H, -OCH3O-), 3.90 (s, 3H, 8-OCH3), 3.81 (s, 3H, 9-OCH3), 3.70–3.65 (dd, J = 14.8 Hz, J = 4.4 Hz, 1H, 6a-H), 3.18–3.04 (m, 3H, 7-H, 5-Ha), 2.66–2.61 (m, 1H, 5-Hb), 2.59 (s, 3H, -NCH3), 2.56–2.49 (m, 1H, 4-Ha), 2.33–2.26 (t, J = 14.2 Hz, 1H, 4-Hb). Positive ESI-MS m/z: 340.2 [M + H]+.
3.1.12. Preparation of Dehydrostephanine (4a)
In a similar manner used in the preparation of 3a, stephanine (200 mg, 0.65 mmol) was subjected to a dehydrogenation reaction to give compound 4a (162 mg, 81%) as a yellowish-green solid. 1H-NMR (400 MHZ, CDCl3, δ; ppm) 8.56 (d, J = 8.4 HZ, 1H, 11-H), 8.29 (d, J = 8.0 HZ, 10-H), 7.08 (s, 1H, 9-H), 6.96 (s, H, 3-H), 6.93 (s, H, 7-H), 6.21 (s, 2H, -OCH2O-), 4.02 (s, 3H, 8-OCH3), 3.40–3.37 (t, J = 5.6 HZ, 2H, 5-CH2), 3.27–3.24 (m, 2H, 5-CH2), 3.12 (s, 3H). Positive ESI-MS m/z: 308.2 [M + H]+.
3.1.13. Preparation of (±) Stephanine (4b)
In a similar manner used for the preparation of 3b, 4a (150 mg, 0.49 mmol) was subjected to a racemization reaction to give compound 4b (125 mg, 83%) as a yellowish-green solid. : 0° (C 0.2, MeOH). 1H-NMR (400 MHZ, CDCl3, δ; ppm) 7.70 (d, J = 8.0 Hz, 1H, 11-H), 7.31–7.26 (t, J = 8.0 Hz, 1H, 10-H), 6.85 (d, J = 8.0 Hz, 1H, 9-H), 6.58 (s, 1H, 3-H), 6.10 (s, 1H, -OCH2O-), 5.95 (s, 1H, -OCH2O-), 3.86 (s, 1H, 8-OCH3), 3.78–3.74 (dd, J = 14.8 Hz, J = 3.6 Hz, 1H, 6a-H), 3.40–3.34 (m, 3H, 7-H, 5a-H), 2.90–2.83 (m, 1H, 5a-H), 2.80 (s, 3H, -NCH3), 2.77–2.72 (m, 1H, 4a-H), 2.58–2.51 (t, J = 14.4 Hz, 1H, 4b-H). Positive ESI-MS m/z: 310.2 [M + H]+.
3.1.14. Preparation of 11-Methoxy-N-Methylisocorydine (5a)
To a mixture of isocorydine (200 mg, 0.59 mmol), Tetrabutylammonium bromide (16 mg, 0.03 mmol), and K2CO3 (163 mg, 1.18 mmol) in anhydrous THF (10 mL) CH3I (185 mg, 1.30 mmol) was successively added over a 10-min period and stirred for 8 h at room temperature. The reaction mixture was condensed and extracted with CHCl3. The combined extracts were dried over anhydrous Na2SO4, filtered, and concentrated to give a crude product, which was purified by silica gel column chromatography (CH2Cl2/MeOH 50:1), yielding compound 5a (154 mg, 71%) as a white solid. 1H-NMR (400 MHZ, CDCl3, δ; ppm) 6.95 (d, J = 8.0 HZ, 1H, 8-H), 6.84 (d, J = 8.4 HZ, 1H, 9-H), 6.66 (s, 1H, 3-H), 3.88 (d, J = 4.0 HZ, 6H, 1-OCH3,2-OCH3), 3.87 (s, 3H,11-OCH3), 3.72 (s, 3H, 10-OCH3), 3.17–3.10 (m, 1H, 6a-H), 3.04–2.99 (m, 2H, 7-Ha, 5-Ha), 2.87 (d, J = 12.4 HZ, 1H, 7-Hb), 2.69 (d, J = 16.8 HZ, 1H, 5-Hb), 2.53–2.48 (m, 4H, -NCH3, 4-Ha), 2.40–2.33 (t, J = 13.0 HZ, 1H, 4-Hb).13C-NMR (100 MHZ, CDCl3, δ; ppm) 29.73 (t, C-4), 23.03 (t, C-7), 42.94 (q, N-CH3), 53.07 (q, N-CH3), 55.83 (q, 10-OCH3), 55.97(q, 2-OCH3), 60.00 (t, C-5), 60.21 (q, 1-OCH3), 60.40 (q, 11-OCH3), 111.46 (d, C-3), 112.72 (d, C-9), 121.63 (s, C-11a). 122.36 (d, C-8), 123.98 (s, C-1a), 123.96 (s, C-7a), 126.18 (s, C-1b), 146.35 (s, C-1), 147.04 (s, C-10), 152.20 (s, C-11), 152.54 (s, C-20). Positive ESI-MS m/z: 370.2 [M + H]+. HR-MS for C22H28NO4 [M + H]+; calcd. 370.2013, found: 370.2026.
3.1.15. Preparation of 11-Methoxyisocorydine (5b)
A solution of isocorydine (200 mg, 0.59 mmol) in MeCN (5 mL) and MeOH (5 mL) was treated with N,N-diisopropylethylamine (91 mg, 0.71 mmol) and trimethylsilyldiazomethane (0.36 mL, 2M in hexane), the mixture was stirred at room temperature for 15 h, condensed, and extracted with CHCl3. The combined extracts were dried over anhydrous Na2SO4, filtered, and concentrated to give a crude product, which was purified by silica gel column chromatography (CHCl3–MeOH = 80:1), yielding compound 5b (51 mg, 24%) as a colorless oil. 1H-NMR (400 MHZ, CDCl3, δ; ppm) 6.95 (d, J = 8.0 HZ, 1H, 8-H), 6.84 (d, J = 8.4 HZ, 1H, 9-H), 6.66 (s, 1H, 3-H), 3.88 (d, J = 4.0 HZ, 6H, 1-OCH3,2-OCH3), 3.87 (s, 3H,11-OCH3), 3.72 (s, 3H, 10-OCH3), 3.17–3.10 (m, 1H, 6a-H), 3.04–2.99 (m, 2H, 7-Ha, 5-Ha), 2.87 (d, J = 12.4 HZ, 1H, 7-Hb), 2.69 (d, J = 16.8 HZ, 1H, 5-Hb), 2.53–2.48 (m, 4H, -NCH3, 4-Ha), 2.40–2.33 (t, J = 13.0 HZ, 1H, 4-Hb). Positive ESI-MS m/z: 356.2 [M + H]+. HR-MS for C21H25NO4 [M + H]+; calcd. 356.1856, found: 356.1864.
3.1.16. Preparation of 11-Ethoxyisocorydine (5c)
A mixture of isocorydine (200 mg, 0.59 mmol), Tetrabutylammonium bromide (16 mg, 0.03 mmol), and K2CO3 (163 mg, 1.18 mmol) in anhydrous THF (10 mL) was successively added CH3CH2I (203 mg, 1.30 mmol) over a 10-min period and stirred for 8 h at room temperature. The reaction mixture was condensed and extracted with CHCl3. The combined extracts were dried over anhydrous Na2SO4, filtered, and concentrated to get a crude product, which was purified by silica gel column chromatography (CH2Cl2/MeOH 50:1), yielding compound 5c (80 mg, 23%) as a colorless oil. 1H-NMR (400 MHZ, CDCl3, δ; ppm) 6.95 (d, J = 8.0 HZ, 1H, 8-H), 6.83 (d, J = 8.0 HZ,1H, 9-H), 6.66 (s, 1H, 3-H), 3.87 (d, J = 2.8 HZ, 6H, 1-OCH3, 2-OCH3), 3.84–3.79 (m, 1H, 6a-H), 3.66 (s, 3H, 10-OCH3), 3.19–3.14 (m, 1H, 7-Ha), 3.05–2.98 (m, 2H,11-CH2), 2.87–2.83 (m, 1H, 7-Hb), 2.71–2.67 (m 1H, 5-Ha), 2.54 (s, 3H, -NCH3), 2.50–2.47 (m, 4-Ha), 2.40–2.34 (t, 1H, J = 12.8 HZ, 4-Hb), 1.14–1.10 (t, J = 7.0 HZ, 3H, 11-CH3). 13C-NMR (100 MHZ, CDCl3, δ; ppm) 15.79 (q, 11-CH2CH3), 25.87 (t, C-7), 33.34 (t, C-4). 56.21 (q, 10-OCH3), 60.96 (q, 1-OCH3), 61.14 (q, 2-OCH3), 68.89 (t, 11-CH2CH3), 111.41 (d, C-3), 112.72 (d, C-9), 122.04 (d, C-8), 124.00 (C-7a), 125.01 (s, C-1a), 127.00 (s, C-1b), 146 .89 (s, C-1), 147.04 (s, C-10), 152.82 (s, C-11), 153.42 (s, C-2). Positive ESI-MS m/z: 370.2 [M + H]+. HR-MS for C22H27NO4 [M + H]+; calcd. 370.2013, found: 370.2022.
3.1.17. Preparation of N-Methylisocorydine (5d)
In a similar manner used in the preparation of 2d, isocorydine (200 mg, 0.59 mmol) was subjected to a N-methylation reaction to give compound 5d (183 mg, 88% yield) as a white solid. 1H-NMR (400 MHZ, CDCl3, δ; ppm) 8.60 (s, 1H, 11-OH), 7.01 (d, J = 8.0 HZ, 1H, 8-H), 6.89 (d, J = 8.0 Hz, 2H, 9-H), 6.86 (s, 1H, 3-H), 4.77–4.73 (dd, J = 12.0 HZ, J = 5.2 HZ, 1H, 6a-H), 4.24 (d, J = 13.2, 1H, 5a-H), 3.96 (s, 3H, 1-OCH3), 3.89 (s, 3H, 2-OCH3), 3.76 (s, 3H, 10-OCH3), 3.74 (s, 3H, -NCH3), 3.65–3.58 (m, 2H, 7a-H), 3.54–3.41 (m, 2H, 5b-H, 7a-H), 3.33 (s, 3H, -NCH3), 3.11–3.07 (m, 1H, 4-Ha), 2.88–2.81 (t, J = 12.0 HZ, 1H, 4-Hb). Positive ESI-MS m/z: 356.2 [M]+.
3.1.18. Preparation of (Z)-N-2,3-Dimethoxybenzylidene-3,4-Dimethoxy-2-Phenyl-Ethanamine (6a)
A mixture of 2-(3,4-dimethoxy-phenyl)-ethylamine (1 g, 5.5 mmol) and 2,3-dimethoxy-benzoic acid (1.1 g, 6.1 mmol) in methylbenzene (50 mL) was heated to 120 °C for 8 h. The reaction mixture was condensed and extracted with EtOAC. The combined extracts were washed with water three times, dried over anhydrous Na2SO4, filtered, and concentrated. The crude product was drawn off by a vacuum pump over 4 h to give compound 6a (1.78 g, 92% Purity, 91% yield) as a brown-red oil. 1H-NMR (400 MHZ, CDCl3, δ; ppm) 7.54 (d, J = 7.6 HZ 1H, N-CH2CH2Ph-2H), 7.09–7.05 (t, J = 8.0 HZ, 1H, N-CH2Ph-4H), 6.96 (d, J = 8.0 HZ, 1H, N-CH2CH2Ph-5H), 6.76 (d, J = 12.0 HZ, 3H, N-CH2CH2Ph-6H, N-CH2Ph-3H, N-CH2Ph-5H), 3.90–3.88 (m, 1H, N-CH2β), 3.87–3.76 (m, 12H, 4×-OCH3), 2.98–2.94 (t, J = 7.0 HZ, 2H, N-CH2α). Positive ESI-MS m/z: 330.2 [M + H]+.
3.1.19. Preparation of N-2,3-Dimethoxybenzyl-3,4-Dimethoxy-2-Phenylethanamine (6b)
Amixture of 6a (1.5 g, 4.6 mmol) and Pd/C(5%, 150 mg) in MeOH (50 mL) was stirred under a hydrogen atmosphere (1 bar) for 8 h. The MeOH-insoluble material was filtered off and the filtrate was evaporated to give a crude product, which was purified by silica gel column chromatography (CH2Cl2/petroleum ether 1:1), yielding compound 6b (1.3 g, 87%) as a yellow oil. 1H-NMR (400 MHZ, CDCl3, δ; ppm), 7.01–6.97 (t, J = 8.0 HZ, 1H, N-CH2Ph-4H), 6.83 (d, J = 6.4 HZ, 2H, N-CH2CH2Ph-5H, N-CH2CH2Ph-6H), 6.78 (d, J = 8.0 HZ, 1H, N-CH2CH2Ph-5H), 6.73 (d, 2H, J = 11.6, N-CH2Ph-3H, N-CH2Ph-5H), 3.84–3.77 (m, 12H, 4×-OCH3), 2.87–2.83 (t, J = 7.0 HZ, 2H, N-CH2α), 2.78–2.74 (t, J = 6.8 HZ, 2H, N-CH2β), 1.89 (s, 2H, N-CH2). Positive ESI-MS m/z: 310.2 [M + H]+.
3.1.20. Preparation of N-Methyl-N-2,3-Dimethoxybenzyl-3,4-Dimethoxy-2-Phenyl-Ethanamine (6c)
To a mixture of 6b (1.2 g, 3.6 mmol) and K2CO3 (1.0 g, 7.2 mmol) in anhydrous THF (20 mL),CHI (610.6 mg, 4.3 mmol) was slowly added under an ice-bath over a 10-min period. The solution was stirred for 4 h at room temperature, condensed, and extracted with EtOAC. The combined extracts were dried over anhydrous Na2SO4 and filtered, and the filtrate was concentrated to give a crude product, which was purified by silica gel column chromatography (CH2Cl2/petroleum ether 10:1), yielding compound 6c (950 mg, 76% yield) as a colorless oil. 1H-NMR (400 MHZ, CDCl3, δ; ppm) 7.06–7.00 (dd, 2H, J = 9.6 HZ, J = 4.8 HZ, N-CH2CH2Ph-5H, N-CH2CH2Ph-6H), 6.86 (t, 1H, J = 4.2 HZ, N-CH2Ph-4H), 6.78 (d, 1H, J = 8.4, N-CH2CH2Ph-2H), 6.73 (d, 2H, J = 7.2, N-CH2Ph-5H, N-CH2Ph-3H), 3.86–3.82 (m, 12H, 4×-OCH3), 3.74 (s, 2H, N-CH2), 2.87 (t, 2H, J = 7.2, N-CH2α), 2.77–2.71 (m, 2H, N-CH2β), 2.37 (s, 3H). Positive ESI-MS m/z: 346.2 [M + H]+.
3.2. Pharmacological Evaluation
3.2.1. Materials
Verapamil hydrochloride (Shanghai Harvest Pharmaceutical Co., Ltd., Shanghai, China), lidocaine hydrochloride (Shanghai ZhaohuiPharmaceutical Co., Ltd., Shanghai, China), isocorydine, crebanine, and stephanine were used as positive control groups. Normal saline (NS) (Kunming Nanjiang Pharmaceutical Co., Ltd., Kunming, China) was used as negative control group. Other chemicals used—chloral hydrate, barium chloride, chloroform, sodium hydroxide, and hydrochloric acid were of analytical grade.
All aporphine alkaloid derivatives were used for the tested drug groups, and were synthesized, purified, and determined according to Section 3.1.2, Section 3.1.3, Section 3.1.4, Section 3.1.5, Section 3.1.6, Section 3.1.7, Section 3.1.8, Section 3.1.9, Section 3.1.10, Section 3.1.11, Section 3.1.12, Section 3.1.13, Section 3.1.14, Section 3.1.15, Section 3.1.16, Section 3.1.17, Section 3.1.18, Section 3.1.19 and 3.1.20 in this paper. The compounds were dissolved in 10% hydrochloric acid solution and were adjusted to pH5–7 with 40% sodium hydroxide.
3.2.2. Animals
The experiments were carried out on Kunming mice (28–35 g) and SD rats (180–210 g), which were provided by Hunan SJA Laboratory Animal Co., Ltd. (SYXK (Dian) K 2011-0011, Changsha, China). Half of the animals were male and half were female, and they were housed in a controlled environment (temperature 22 ± 1 °C; 60% ± 10% humidity; in a 12/12 h light-dark cycle) with free access to standard pellet artificial diet and tap water. All animal experiments performed were in compliance with the Animal Experimental Care and Ethic Committee of Yunnan University of Traditional Chinese Medicine.
Determination of the dosage for tested drugs: The animals were first given 5 mg/kg of tested drugs, based on the effective dose of crebanine in mice and rat [7]. The dosage was reduced or increased according to the exhibited symptoms of poisoning, or death, and the antiarrhythmic effect on the animals.
3.2.3. Statistical Analyses
Data are expressed as the means ± SD and incidence ratio. Statistical significance was calculated using the Chi-square test. Differences were considered significant when p < 0.05. LD50 was determined using a related formulate of up-and-down procedure (UDP) [28].
3.2.4. Antiarrhythmic Activity Test of Aporphine Derivatives in a Mouse Model of Ventricular Fibrillation Induced by CHCl3
Mice were randomly divided into a negative control group (NS), 4 positive control groups (verapamil hydrochloride, crebanine, iscorydine, and stephanine), and 19 tested groups for aporphine derivatives (Figure 1). Tested drugs were injected though the caudal vein at a chosen dose. Five minutes later, each mouse was placed in a 500-mL inverted beaker, which contained a cotton ball that had been soaked with 1mL of chloroform, and then were removed immediately after respiratory arrest. The chest of the asphyxiated mouse was opened in order to observe the condition of ventricular fibrillation (VF) [7,29]. The occurrence ratios of VF in mice are listed in Figure 1.
3.2.5. Anti-Arrhythmic Activity of Five Kinds of Aporphine Derivatives in a Rat Model of Arrhythmia Induced by BaCl2
Rats were randomly divided into 10 groups: an NS group, 4 groups of positive control (lidocaine, crebanine, iscorydine and stephanine) groups, and 5 tested groups of active derivatives (2a–d and 4a) (Table 1). First, rats were anesthetized with 10% choral hydrate (ip, 300 mg/kg), fixed on their backs and connected to a physiological signal acquisition system in order to record normal II lead electrocardiogram (ECG). Then, a barium chloride solution was injected into the caudal vein of each rat (4 mg/kg, at a volume of 1 mL/kg) to trigger arrhythmia. Three minutes later, rats were injected with different treatments though the lingual vein at a certain doses. Then, ECG was recorded. The number of rats that recovered to a normal sinus rhythm, the duration needed to recover, and number of rats that maintained normal rhythm for more than 3 min, 5 min, and 20 min, in each group, were calculated [7,29] (Table 1).
3.2.6. Acute Toxicity Test in Mice
Mice were injected with the test compounds via the caudal vein at a certain dose. The Dm (100% death) and Dn (0% death) of the compounds were found in preliminary tests; then, 5–6 dose groups (n = 10) of the compounds were set up, according to the requirements of the up-and-down procedure (UDP) method, death number was recorded and LD50 was determined [28].
4. Conclusions
In this study, derivatives of three kinds of aporphine alkaloids, crebanine, isocorydine, and stephanine, were designed and synthesized through the reactions of ring-opening, N-substitution, demethylation, methylation, acetylation, quaternization, and dehydrogenation. The use of NBS for the bromination reaction was successfully applied to the synthesis of the three kinds of bromocrebanines. Among these derivatives, compound 2b, dibromocrebanine, proved to be the most promising candidate, showing maximum potency against arrhythmia and minimum toxicity compared to the other compounds prepared in this family. Derivatives 2a, 2c, 2d, and 4a also displayed impressive potency. Finally, derivative 1d exhibited a lower, though still significant, activity.
The relationship of the structure–antiarrhythmic activities of aporphines are summarized as: (1) C-1,C-2-methylenedioxy group on ring A of aporphines increases the planar configuration of molecules and plays an important role in both activity and toxicity; (2) a closed structure of the B ring is essential for both antiarrhythmic efficacy and toxicity; additionally, N-quaternization of ring B could increase toxicity; (3) levoduction of 6a in ring C is an active group; and (4) 8-, 9-, and 10-methoxy groups on ring D is an important functional groups. Further explorations are underway to evaluate the biological activities and the ion channel mechanisms of these aporphine derivatives.
Acknowledgments
The National Natural Science Foundation of China (81260651) and the Science Foundation of Education Department of Yunnan Province (2015J102) are gratefully acknowledged for their financial support. We acknowledge members of Minglong Yuan’s lab, Yunnan Minzu University, for chemical modifications. We also thank. Qing Zhao, Hongping He and Aixue Zuo for chemical structure analyses, and Jia Chen for language assistance.
Author Contributions
Yunshu Ma and Hui Wang conceived and designed the experiments; Hui Wang and Shujun Kong performed the experiments; Hui Wang, Zixian Yang and Hongmei Wang analyzed the data; Qiuyan Huang, Jingyu Li and Cheng Chen contributed reagents and materials; Yunshu Ma, Hui Wang and Xin Cheng wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Scholz, H. Classification and Mechanism of Action of Antiarrhythmic Drugs. Fundam. Clin. Pharmacol. 1994, 8, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Podrid, P.J. Aggravation of Arrhythmia by Antiarrhythmic Drugs (Proarrhythmia). Cardiac. Electrophysiol. Clin. 2010, 2, 459–470. [Google Scholar] [CrossRef]
- Yang, Q.; Wang, X.J.; Tang, Y.-Q.; You, Q.-D. The Research Practice of Anti-arrhythmic Agents Targeting on Potassium Ion Channel. Acta Pharm. Sin. 2011, 46, 12–18. [Google Scholar]
- Yao, S.B.; Hao, J.H.; Li, X.G.; Fan, L.; Liu, Y. Anti-arrhythmic Targets of Natural Products: Research Advances. Chin. J. Pharmacol. Toxicol. 2016, 2, 151–157. [Google Scholar]
- Ren, J.H.; Chen, L.H. The Pharmacological Action and Clinical Application of Isocorydine. Chin. Pharm. 1999, 7, 10–11. [Google Scholar]
- Su, Y.H.; Li, H.L. Effective Comparison of l-dicentrine on Arrhythmia Caused by Different Kinds of Cardiac Glycoside. Chin. J. Ethnomed. Ethnopharm. 2002, 58, 301–302. [Google Scholar]
- Yu, Z.F.; Ma, Y.S.; Zhao, Z.X.; Wang, F.C. Antiarrhymic Effects of Crebanine. Chin. J. Chin. Mat. Med. 1992, 11, 685–687. [Google Scholar]
- Huang, J.X.; Chen, Y. Studies on the Alkaloids of Stephania Species. I. Isolation and identification of alkaloids from Stephniaepigeae. Acta Pharm. Sin. 1979, 14, 612–616. [Google Scholar]
- Moreno, L.; Cabedo, N.; Ivorra, M.D.; Sanz, M.J.; Castel, A.L.; Álvarez, C.; Cortes, D. 3,4-Dihydroxy- and 3,4-methylenedioxy-phenanthrene-type alkaloids with high selectivity for D2 dopamine receptor. Bioorg. Med. Chem. Lett. 2013, 23, 4824–4827. [Google Scholar] [CrossRef] [PubMed]
- Kathofer, S.; Thomas, D.; Karle, C.A. The Novel Antiarrhythmic Drug Dronedarone: Comparison with Amiodarone. Cardiovasc. Drug Rev. 2005, 23, 217–230. [Google Scholar] [CrossRef] [PubMed]
- Touboul, P.; Brugada, J.; Capucci, A.; Touboul, P.; Crijns, N.; Edvardsson, H.J.G.M.; Hohnloser, S.H. Dronedarone for Prevention of Atrial Fibrillation: A Dose-ranging Study. Eur. Heart J. 2003, 24, 1481–1487. [Google Scholar] [CrossRef]
- Ehrlich, J.R.; Hohnloser, S.H. Milestones in the Management of Atrialfibrillation. Heart Rhythm. 2009, 6, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Li, W.L.; Yu, Z.F.; Lin, Q.; Ma, Y.S.; Luo, Q.Y. Effects of Crebanine on Arrhythmias Induced by Ischemia and Reperfusion in Rats. Chin. Pharmacol. Bull. 2005, 21, 1019–1020. [Google Scholar]
- Ma, Y.S.; Wu, H.P.; Zhang, Z.L. Acute Toxicity and Antiarrhythmia Effect of Crebanine. Chin. J. Mod. Appl. Pharm. 2005, 22, 368–369. [Google Scholar]
- Lei, Z.B. Adverse Reactions and Interactions of the Antiarrhythmic Drug Dronedarone. Chin. J. New. Drugs 2012, 21, 1826–1830. [Google Scholar]
- Zhao, Q.Z.; Zhao, Y.M. Progress in Biological Activities of Aporphinoid Alkaloids. Nat. Prod. Res. Dev. 2006, 18, 316–324. [Google Scholar]
- Gao, Y.; Zong, R.; Campbell, A.; Kula, N.S.; Baldessarini, R.J.; Neumeyer, J.L. Synthesis and Dopamine Agonist and Antagonist Effects of R-(−)- and S-(+)-11-hydroxy-N-n-propylnoraporphine. J. Med. Chem. 1988, 31, 1392–1396. [Google Scholar] [CrossRef] [PubMed]
- Hedberg, M.H.; Johansson, A.M.; Nordvall, G.; Yliniemelä, A.; Li, H.B.; Martin, A.R.; Hjorth, S.; Unelius, L.; Sundell, S.; Hacksell, U. (R)-11-Hydroxy- and (R)-11-Hydroxy-10-methylaporphine: Synthesis, Pharmacology, and Modeling of D2A and 5-HT1A Receptor Interactions. J. Med. Chem. 1995, 38, 647–658. [Google Scholar] [CrossRef] [PubMed]
- Neumeyer, J.L.; Granchelli, F.; Little, A.D.; Aporphines, V.I. A novel synthesis of 7-hydroxyaporphine, 7-hydroxynoraporphine and 6a,7-dehydronoraporphine Granchelli. Tetrahedron Lett. 1970, 11, 5261–5264. [Google Scholar] [CrossRef]
- Wang, X.K.; Zhao, T.F. Distribution and Biological Activities of Alkaloids in plants of Genus Stephania. Chin. Pharm. J. 1990, 25, 3–6. [Google Scholar]
- Gao, Y.G.; Baldessarini, R.J.; Kula, N.S.; Neumeyer, J.L. Synthesis and Dopamine Receptor Affinities of Enantiomers of 2-substituted Apomorphines and Their N-n-propyl Analogs. J. Med. Chem. 1990, 33, 1800–1805. [Google Scholar] [CrossRef] [PubMed]
- Manabu, N.; Kiyoharu, N.; Kaoru, F.; Eiichi, F.J. Demethylation of Methyl Ethers of Alcohol and Phenol with an Aluminum Halide-Thiol System. Org. Chem. 1980, 45, 4275–4277. [Google Scholar]
- Gao, Y.; Ram, V.J.; Campbell, A.; Kula, N.S.; Baldessarini, R.J.; Neumeyer, J.L. Synthesis and structural requirements of N-substituted norapomorphines for affinity and activity at dopamine D-1, D-2, and agonist receptor sites in rat brain. J. Med. Chem. 1990, 33, 39–44. [Google Scholar] [CrossRef]
- Shamma, M.; Slusarchyk, W.A. The Aporphine Alkaloids. Chem. Rev. 1964, 64, 59–79. [Google Scholar] [CrossRef]
- Nakamura, S.; Nakashima, S.; Tanabe, G.; Oda, Y.; Yokota, N.; Fujimoto, K.; Matsumoto, T.; Sakuma, R.; Ohta, T.; Ogawa, K. Alkaloid constituents from flower buds and leaves of sacred lotus (Nelumbonucifera, Nymphaeaceae) with melanogenesis inhibitory activity in B16 melanoma cells. Bioorg. Med. Chem. Lett. 2013, 21, 779–879. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Fang, S.D.; Liang, D.; Jiang, F.Y. Studies on the Alkaloids of Stephania Yunnanensis. Acta Bot. Sin. 1989, 31, 296–299. [Google Scholar]
- Zuo, A.X.; Ma, Y.S.; Rao, G.X.; Chin, J. Akaloids from Roots of Stephania Mashanica. Chin. Mat. Med. 2013, 4, 574–577. [Google Scholar]
- OECD. Acute oral toxicity-up-and-down procedure. In OECD Guidelines for the Testing of Chemicals; Organization for Economic Cooperation and Development: Paris, France, 2001; Volume 425, pp. 1–26. [Google Scholar]
- Li, Y.K. Experimental Methodology of TCM Pharmacology; Shanghai Scientific and Tchnical Press: Shanghai, China, 2006; pp. 1001–1007. [Google Scholar]
- Sample Availability: The compounds 1a, 2a, 2b, 1e, 2d and 6b are avilable, other compounds are not provided from us.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).