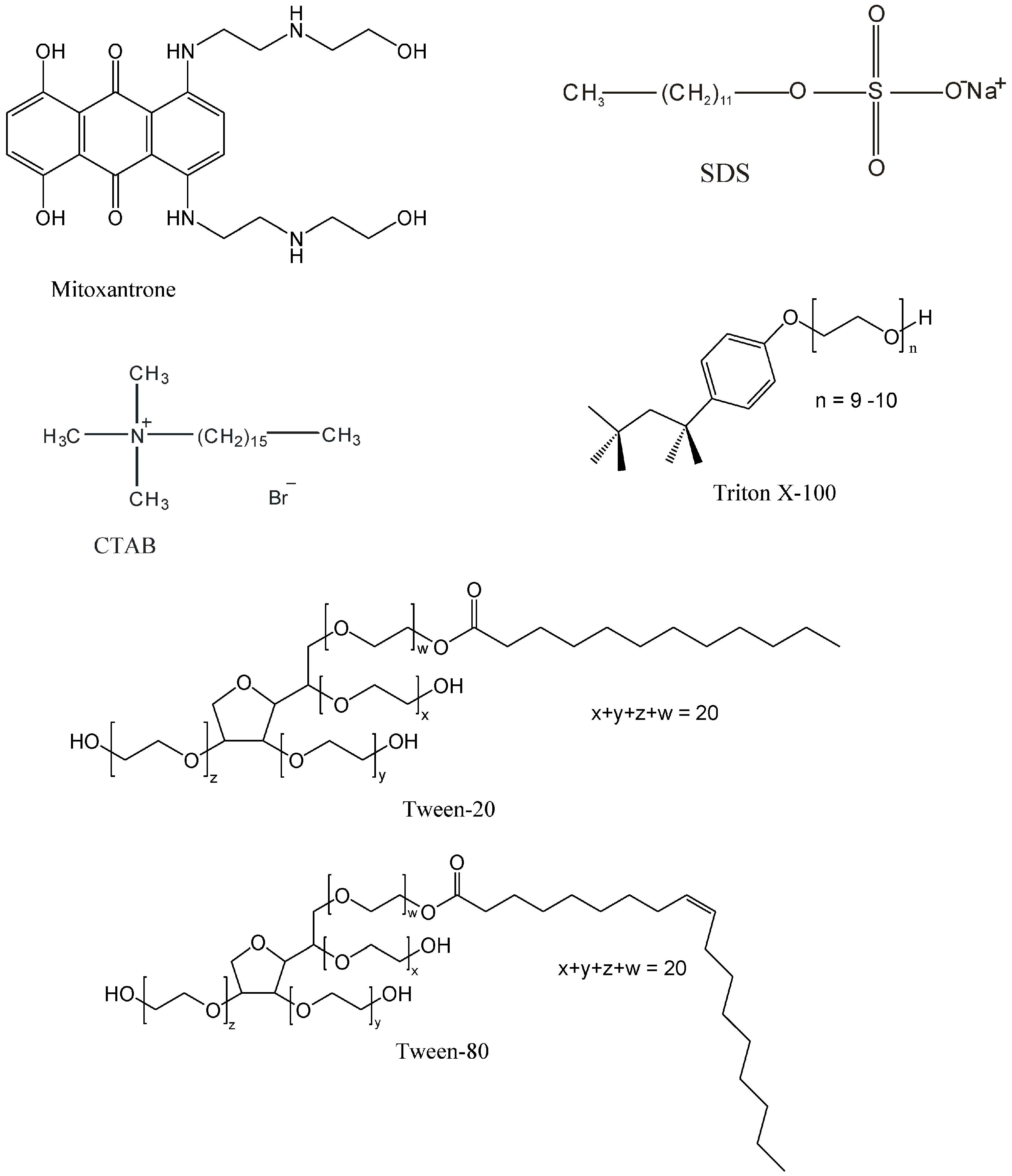
Mitoxantrone-Surfactant Interactions: A Physicochemical Overview
Abstract
:1. Introduction
2. UV-Vis Absorption Studies
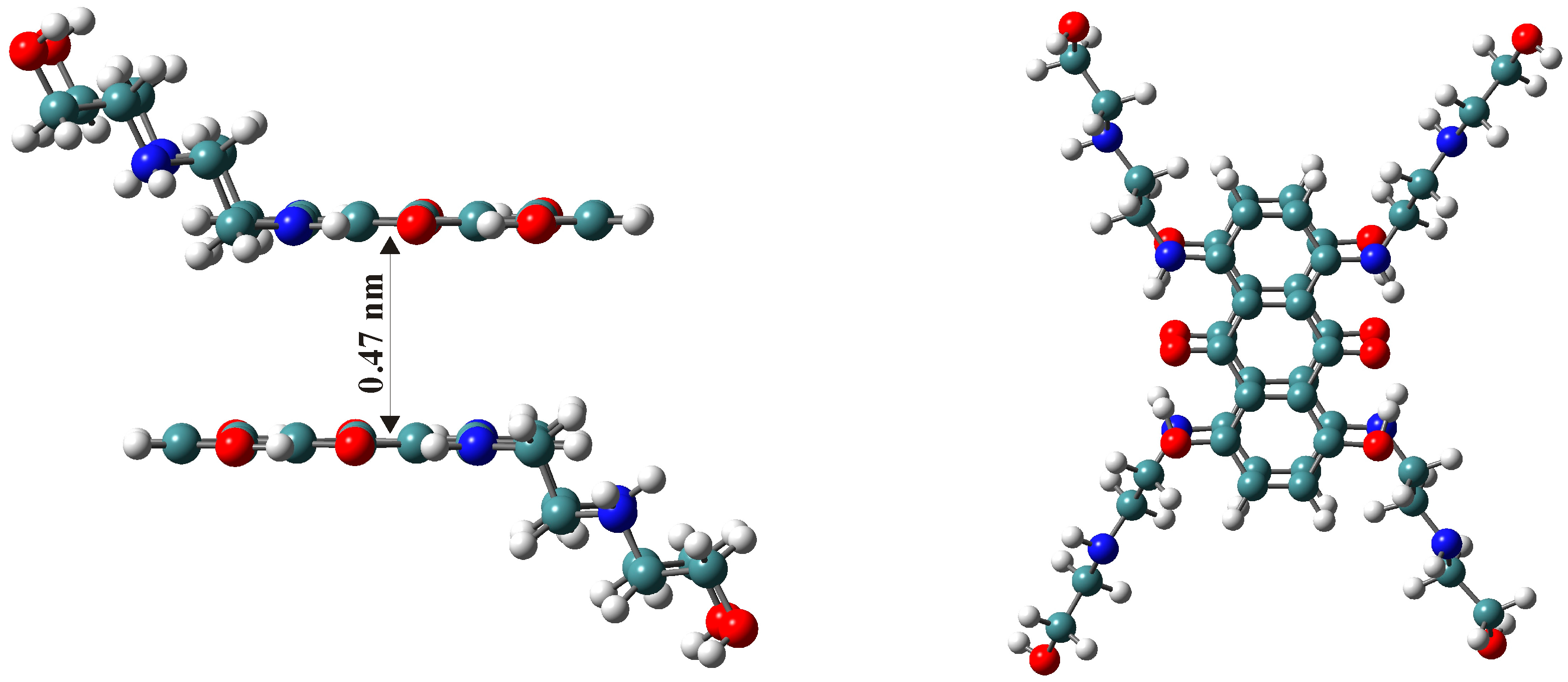
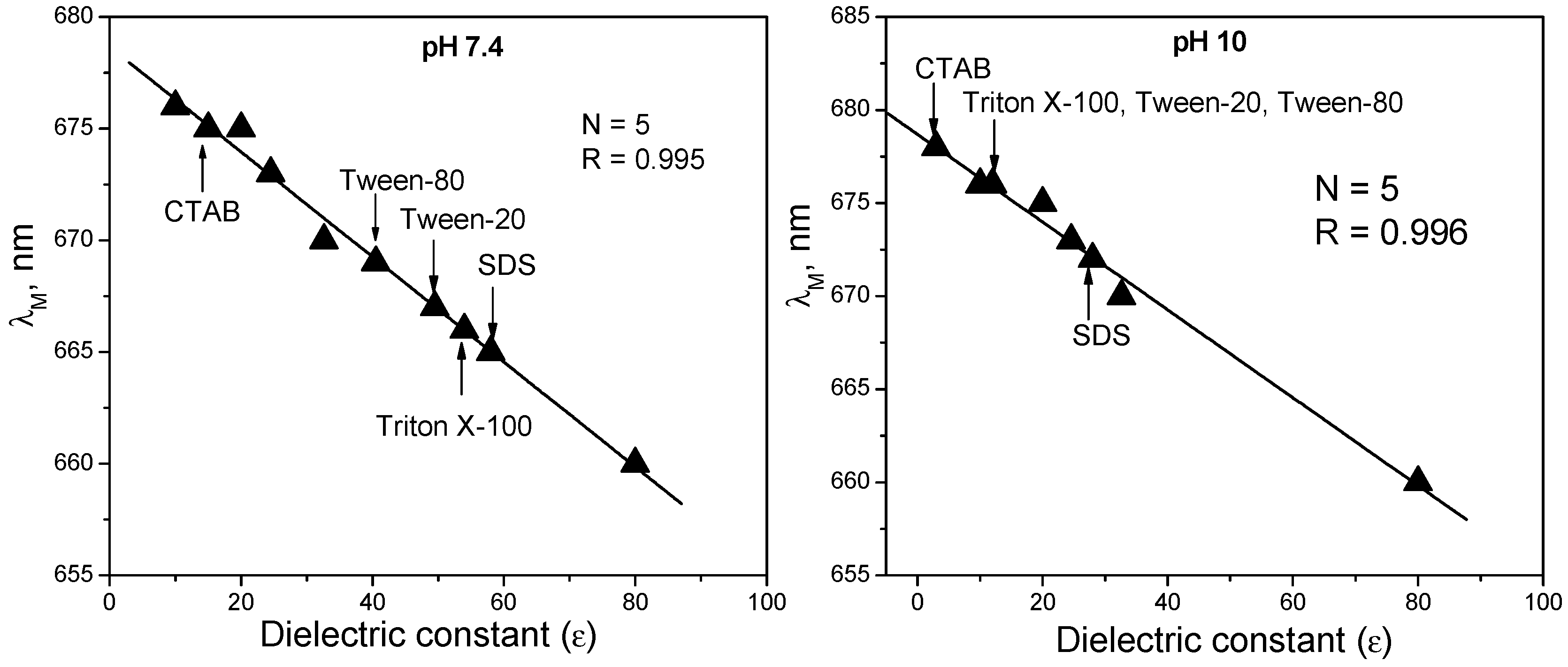
3. Polarity of the Micellar Environment and Probable Location of Mitoxantrone
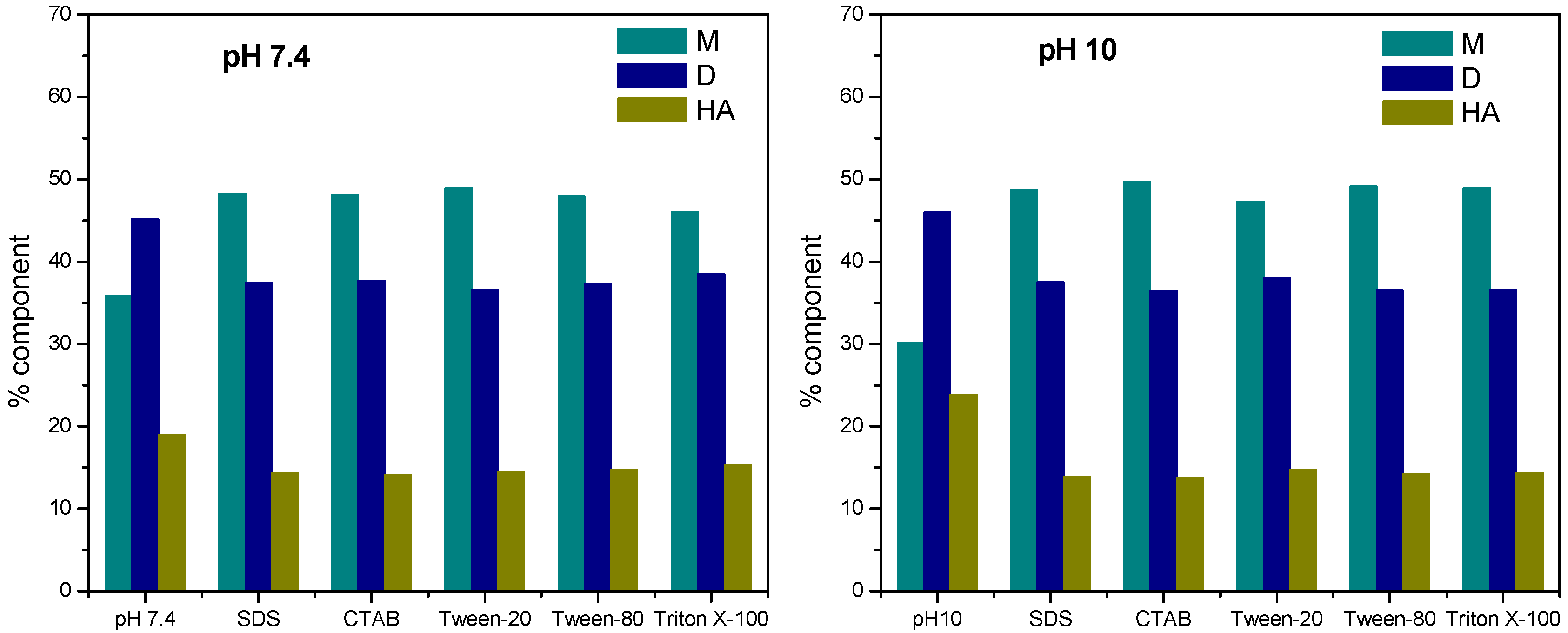
4. Binding Parameters
4.1. Binding Constant
4.2. Partition Coefficient
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fox, E.J. Mechanism of action of mitoxantrone. Neurology 2004, 63, S15–S18. [Google Scholar] [CrossRef] [PubMed]
- Rossato, L.G.; Costa, V.M.; de Pinho, P.G.; Arbo, M.D.; de Freitas, V.; Vilain, L.; de Lourdes Bastos, M.; Palmeira, C.; Remiao, F. The metabolic profile of mitoxantrone and its relation with mitoxantrone-induced cardiotoxicity. Arch. Toxicol. 2013, 87, 1809–1820. [Google Scholar] [CrossRef] [PubMed]
- Hande, K.R. Topoisomerase II inhibitors. Update Cancer Ther. 2008, 3, 13–26. [Google Scholar] [CrossRef]
- Neuhaus, O.; Kieseier, B.C.; Hartung, H.P. Therapeutic role of mitoxantrone in multiple sclerosis. Pharmacol. Ther. 2006, 109, 198–209. [Google Scholar] [CrossRef] [PubMed]
- Van Dalen, E.C.; van der Pal, H.J.H.; Bakker, P.J.M.; Caron, H.N.; Kremer, L.C.M. Cumulative incidence and risk factors of mitoxantrone-induced cardiotoxicity in children: A systematic review. Eur. J. Cancer 2004, 40, 643–652. [Google Scholar] [CrossRef] [PubMed]
- Avasarala, J.R.; Cross, A.H.; Clifford, D.B.; Singer, B.A.; Siegel, B.A.; Abbey, E.E. Rapid onset mitoxantrone-induced cardiotoxicity in secondary progressive multiple sclerosis. Mult. Scler. 2003, 9, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Dores-Sousa, J.L.; Duarte, J.A.; Seabra, V.; de Lourdes Bastos, M.; Carvalho, F.; Costa, V.M. The age factor for mitoxantrone’s cardiotoxicity: Multiple doses render the adult mouse heart more susceptible to injury. Toxicology 2015, 329, 106–119. [Google Scholar] [CrossRef] [PubMed]
- Minotti, G.; Menna, P.; Salvatorelli, E.; Cairo, G.; Gianni, L. Anthracyclines: Molecular advances and pharmacologic developments in antitumor activity and cardiotoxicity. Pharmacol. Rev. 2004, 56, 185–229. [Google Scholar] [CrossRef] [PubMed]
- Menna, P.; Salvatorelli, E.; Minotti, G. Cardiotoxicity of antitumor drugs. Chem. Res. Toxicol. 2008, 21, 978–989. [Google Scholar] [CrossRef] [PubMed]
- Šimùnek, T.; Štěrba, M.; Popelová, O.; Adamcová, M.; Hrdina, R.; Geršl, V. Anthracycline-induced cardiotoxicity: Overview of studies examining the roles of oxidative stress and free cellular iron. Pharmacol. Rep. 2009, 61, 154–171. [Google Scholar] [CrossRef]
- Robison, T.W.; Giri, S.N. Effects of chronic administration of doxorubicin on myocardial beta-adrenergic receptors. Life Sci. 1986, 39, 731–736. [Google Scholar] [CrossRef]
- Solem, L.E.; Henry, T.R.; Wallace, K.B. Disruption of mitochondrial calcium homeostasis following chronic doxorubicin administration. Toxicol. Appl. Pharmacol. 1994, 129, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Boucek, R.J., Jr.; Miracle, A.; Anderson, M.; Engelman, R.; Atkinson, J.; Dodd, D.A. Persistent effects of doxorubicin on cardiac gene expression. J. Mol. Cell. Cardiol. 1999, 31, 1435–1446. [Google Scholar] [CrossRef] [PubMed]
- Štěrba, M.; Popelová, O.; Len, J.; Fučíková, A.; Brčáková, E.; Mazurová, Y.; Jirkovský, E.; Šimůnek, T.; Adamcová, M.; Mičuda, S.; et al. Proteomic insights into chronic anthracycline cardiotoxicity. J. Mol. Cell. Cardiol. 2011, 50, 849–862. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Ma, Y.; Yang, C.; Guo, L.; Yang, X. Interaction of anticancer drug mitoxantrone with DNA analyzed by electrochemical and spectroscopic methods. Biophys. Chem. 2005, 116, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Lown, J.W.; Morgan, A.R.; Yen, S.F.; Wang, Y.H.; Wilson, W.D. Characteristics of the binding of the anticancer agents mitoxantrone and ametantrone and related structures to deoxyribonucleic acids. Biochemistry 1985, 24, 4028–4035. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, S.; Jangir, D.K.; Mehrotra, R. Spectroscopic studies of the effects of anticancer drug mitoxantrone interaction with calf-thymus DNA. J. Photochem. Photobiol. B 2013, 120, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Enache, M.; Volanschi, E. Electrochemical and spectral study of the interaction of antitumoral drug mitoxantrone with DNA. Rev. Roum. Chim. 2005, 50, 131–140. [Google Scholar]
- Rosenberg, L.S.; Carvlin, M.J.; Krugh, T.R. The antitumor agent mitoxantrone binds cooperatively to DNA: Evidence for heterogeneity in DNA conformation. Biochemistry 1986, 25, 1002–1008. [Google Scholar] [CrossRef] [PubMed]
- Kapuscinski, J.; Darzynkiewicz, Z. Relationship between the pharmacological activity of antitumor drugs Ametantrone and Mitoxantrone (Novatrone) and their ability to condense nucleic acids. Proc. Nat. Acad. Sci. USA 1986, 83, 6302–6306. [Google Scholar] [CrossRef] [PubMed]
- Hajihassan, Z.; Rabbani-Chadegani, A. Interaction of mitoxantrone, as an anticancer drug, with chromatin proteins, core histones and H1, in solution. Int. J. Biol. Macromol. 2011, 48, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Pommier, Y.; Leo, E.; Zhang, H.L.; Marchand, C. DNA topoisomerases and their poisoning by anticancer and antibacterial drugs. Chem. Biol. 2010, 17, 421–433. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, J.; Basu, A.; Kumar, G.S. Intercalative interaction of the anticancer drug mitoxantrone with double stranded DNA: A calorimetric characterization of the energetics. J. Chem. Thermodyn. 2014, 75, 45–51. [Google Scholar] [CrossRef]
- Nieciecka, D.; Krolikowska, A.; Krysinski, P. Probing the interactions of mitoxantrone with biomimetic membranes with electrochemical and spectroscopic techniques. Electrochim. Acta 2015, 165, 430–442. [Google Scholar] [CrossRef]
- Deleu, M.; Crowet, J.M.; Nasir, M.N.; Lins, L. Complementary biophysical tools to investigate lipid specificity in the interaction between bioactive molecules and the plasma membrane: A review. Biochim. Biophys. Acta 2014, 1838, 3171–3190. [Google Scholar] [CrossRef] [PubMed]
- Eeman, M.; Deleu, M. From biological membranes to biomimetic model membranes. Biotechnol. Agron. Soc. Environ. 2010, 14, 719–736. [Google Scholar]
- Peetla, C.; Stine, A.; Labhasetwar, V. Biophysical interactions with model lipid membranes: Applications in drug discovery and drug delivery. Mol. Pharm. 2009, 6, 1264–1276. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, H.; Sarkar, M. Interaction of piroxicam with micelles: Effect of hydrophobic chain length on structural switchover. Biophys. Chem. 2005, 117, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Torchilin, V.P. Structure and design of polymeric surfactant-based drug delivery systems. J. Control. Release 2001, 73, 137–172. [Google Scholar] [CrossRef]
- Florence, A.T.; Hussain, N. Transcytosis of nanoparticle and dendrimer delivery systems: Evolving vistas. Adv. Drug Deliv. Rev. 2001, 50, S69–S89. [Google Scholar] [CrossRef]
- Rangel-Yagui, C.O.; Pessoa, A., Jr.; Tavares, L.C. Micellar solubilization of drugs. J. Pharm. Pharm. Sci. 2005, 8, 147–165. [Google Scholar] [PubMed]
- Caetano, W.; Tabak, M. Interaction of chlorpromazine and trifluoroperazine with ionic micelles: Electronic absorption spectroscopy studies. Spectrochim. Acta A 1999, 55, 2513–2528. [Google Scholar] [CrossRef]
- Cudina, O.; Brboric, J.; Jankovic, I.; Karljikovic-Raji, K.; Vladimirov, S. Study of valsartan interaction with micelles as a model system for biomembranes. Colloids Surf. B 2008, 65, 80–84. [Google Scholar] [CrossRef] [PubMed]
- Borissevitch, I.E.; Borges, C.P.F.; Yushmanov, V.E.; Tabak, M. Localization of dipyridamole molecules in ionic micelles: Effect of micelle and drug charges. Biochim. Biophys. Acta 1995, 1238, 57–62. [Google Scholar] [CrossRef]
- Cudina, O.; Karljikovic-Rajic, K.; Ruvarac-Bugarcic, I.; Jankovic, I. Interaction of hydrochlorothiazide with cationic surfactant micelles of cetyltrimethylammonium bromide. Colloids Surf. A 2005, 256, 225–232. [Google Scholar] [CrossRef]
- Lazaro, G.S.; Meneses, A.L., Jr.; de Macedo, O.F.L.; de Fatima Gimenez, I.; da Costa, N.B., Jr.; Barreto, L.S.; Almeida, L.E. Interaction of pyrimethamine and sulfadiazine with ionic and neutral micelles: Electronic absorption and fluorescence studies. Colloids Surf. A 2008, 324, 98–104. [Google Scholar] [CrossRef]
- Adlakha-Hutcheon, G.; Bally, M.B.; Shew, C.R.; Madden, T.D. Controlled destabilization of a liposomal drug delivery system enhances mitoxantrone antitumor activity. Nat. Biotechnol. 1999, 17, 775–779. [Google Scholar] [PubMed]
- Ma, Y.; Zhou, L.; Zheng, H.; Xing, L.; Li, C.; Cui, J.; Che, S. pH-responsive mitoxantrone (MX) delivery using mesoporous silica nanoparticels (MSN). J. Mater. Chem. 2011, 21, 9483–9486. [Google Scholar] [CrossRef]
- Ugwu, S.; Zhang, A.; Parmar, M.; Miller, B.; Sardone, T.; Peikov, V.; Ahmad, I. Preparation, characterization, and stability of liposome-based formulations of mitoxantrone. Drug Dev. Ind. Pharm. 2005, 31, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Cui, J.; Wang, C.; Wang, J.; Li, Y.; Zhang, L.; Guo, W.; Wang, Y. Lipid composition and grafted PEG affect in vivo activity of liposomal mitoxantrone. Int. J. Pharm. 2008, 362, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Enache, M.; Anghelache, I.; Volanschi, E. Coupled spectral and electrochemical evaluation of the anticancer drug mitoxantrone-sodium dodecyl sulfate interaction. Int. J. Pharm. 2010, 390, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Enache, M.; Volanschi, E. Spectral studies on the molecular interaction of anticancer drug mitoxantrone with CTAB micelles. J. Pharm. Sci. 2011, 100, 558–565. [Google Scholar] [CrossRef] [PubMed]
- Enache, M.; Volanschi, E. Spectroscopic investigations of the molecular interaction of anticancer drug mitoxantrone with non-ionic surfactant micelles. J. Pharm. Pharmacol. 2012, 64, 688–696. [Google Scholar] [CrossRef] [PubMed]
- Borges, C.P.F.; Borissevitch, I.E.; Tabak, M. Charge- and pH-dependent binding sites of dipyridamole in ionic micelles: A fluorescence study. J. Lumin. 1995, 65, 105–112. [Google Scholar] [CrossRef]
- Raghunand, N.; Mahoney, B.P.; Gillies, R.J. Tumor acidity, ion trapping and chemotherapeutics II. pH-dependent partition coefficients predict importance of ion trapping on pharmacokinetics of weakly basic chemotherapeutic agents. Biochem. Pharmacol. 2003, 66, 1219–1229. [Google Scholar] [CrossRef]
- Ariga, K. Interfaces working for biology: Solving biological mysteries and opening up future nanoarchitectonics. ChemNanoMat 2016, 2, 333–343. [Google Scholar] [CrossRef]
- Schreier, S.; Malheiros, S.V.P.; de Paula, E. Surface active drugs: Self-association and interaction with membranes and surfactants. Physicochemical and biological aspects. Biochim. Biophys. Acta 2000, 1508, 210–234. [Google Scholar] [CrossRef]
- Feofanov, A.; Sharonov, S.; Kudelina, I.; Fleury, F.; Nabiev, I. Localization and molecular interactions of mitoxantrone within living K562 cells as probed by confocal spectral imaging analysis. Biophys. J. 1997, 73, 3317–3327. [Google Scholar] [CrossRef]
- Enache, M.; Volanschi, E. Spectral characterization of self-association of antitumor drug mitoxantrone. Rev. Roum. Chim. 2010, 55, 255–262. [Google Scholar]
- Enache, M.; Toader, A.M. Insights into molecular interaction of anticancer drug mitoxantrone with anionic surfactant sodium dodecyl sulfate at different temperatures. J. Surf. Deterg. 2016. in preparation. [Google Scholar]
- Liu, S.; Wang, F.; Liu, Z.; Hu, X.; Yi, A.; Duan, H. Resonance Rayleigh scattering spectra for studying the interaction of anthracycline antineoplastic antibiotics with some anionic surfactants and their analytical applications. Anal. Chim. Acta 2007, 601, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Manna, K.; Panda, A.K. Spectral studies on the interaction of pinacyanol chloride with binary surfactants in aqueous medium. Spectrochim. Acta A 2009, 74, 1268–1274. [Google Scholar] [CrossRef] [PubMed]
- Sabate, R.; Gallardo, M.; de la Maza, A.; Estelrich, J. A spectroscopy study of the interaction of pinacyanol with n-dodecyltrimethylammonium bromide micelles. Langmuir 2001, 17, 6433–6437. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Montgomery, J.A., Jr.; Vreven, T.; Kudin, K.N.; Burant, J.C.; et al. Gaussian 03; Gaussian, Inc.: Pittsburgh, PA, USA, 2003. [Google Scholar]
- Barone, V.; Cossi, M. Quantum Calculation of Molecular Energies and Energy Gradients in Solution by a Conductor Solvent Model. J. Phys. Chem. A 1998, 102, 1995–2001. [Google Scholar] [CrossRef]
- Riahi, S.; Ganjali, M.R.; Dinarvand, R.; Karamdoust, S.; Bagherzadeh, K.; Norouzi, P. A theoretical study on interactions between mitoxantrone as an anticancer drug and DNA: Application in drug design. Chem. Biol. Drug. Des. 2008, 71, 474–482. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Cui, P.; Liu, C.-B.; Yuan, S.-L. Molecular dynamics simulation of pyrene solubilized in a sodium dodecyl sulfate micelle. Langmuir 2012, 28, 4931–4938. [Google Scholar] [CrossRef] [PubMed]
- Davies, D.B.; Veselkov, D.A.; Evstigneev, M.P.; Veselkov, A.N. Self-association of the antitumour agent novatrone (mitoxantrone) and its hetero-association with caffeine. J. Chem. Soc. Perkin Trans. 2001, 2, 61–67. [Google Scholar] [CrossRef]
- Goyal, P.S.; Aswal, V.K. Micellar structure and inter-micelle interactions in micellar solutions: Results of small angle neutron scattering studies. Curr. Sci. 2001, 80, 972–979. [Google Scholar]
- Sarkar, M.; Poddar, S. Studies of the interaction of surfactants with cationic dye by absorption spectroscopy. J. Colloid Interface Sci. 2000, 221, 181–185. [Google Scholar] [CrossRef] [PubMed]
- Erdinc, N.; Gokturk, S.; Tuncay, M. Interaction of epirubicin HCl with surfactants: Effect of NaCl and glucose. J. Pharm. Sci. 2004, 93, 1566–1576. [Google Scholar] [CrossRef] [PubMed]
- Zafar, F.; Shah, A.; Khan, A.M.; Rana, U.A.; Ahmad, Z.; Siddiq, M.; Ali, S. A spectroscopic and electrochemical investigation of interactions of anticancer uracil derivatives with cationic and anionic surfactants. J. Chem. Soc. Pak. 2015, 37, 290–299. [Google Scholar]
- Bhattacharjee, J.; Verma, G.; Aswal, V.K.; Date, A.A.; Nagarsenker, M.S.; Hassan, P.A. Tween 80–sodium deoxycholate mixed micelles: Structural characterization and application in doxorubicin delivery. J. Phys. Chem. B 2010, 114, 16414–16421. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, J.; Verma, G.; Aswal, V.K.; Hassan, P.A. Small angle neutron scattering study of doxorubicin–surfactant complexes encapsulated in block copolymer micelles. Pramana J. Phys. 2008, 71, 991–995. [Google Scholar] [CrossRef]
- Serbanescu, I.; Bulcu, D.; Volanschi, E. Electrochemical study of the interaction of doxorubicin with anionic surfactants (sodium lauryl sulfate). An. Univ. Bucuresti. Chim. 2005, I-II, 369–376. [Google Scholar]
- Das, A.; Roy, S.; Mondal, P.; Datta, A.; Mahali, K.; Loganathan, G.; Dharumadurai, D.; Sengupta, P.S.; Akbarsha, M.A.; Guin, P.S. Studies on the interaction of 2-amino-3-hydroxy-anthraquinone with surfactant micelles reveal its nucleation in human MDA-MB-231 breast adinocarcinoma cells. RSC Adv. 2016, 6, 28200–28212. [Google Scholar] [CrossRef]
- Enache, M.; Bulcu, D.; Volanschi, E. Spectral studies of anticancer drug actinomycin D in aqueous solutions of different surfactants. J. Colloid Surf. Chem. 2008, 8, 43–51. [Google Scholar]
- Enache, M.; Bulcu, D.; Serbanescu, I.; Volanschi, E. Interaction of actinomycin D with anionic surfactant, sodium dodecyl sulfate: Spectral and electrochemical investigations. Rev. Roum. Chim. 2007, 52, 725–731. [Google Scholar]
- Enache, M.; Ionescu, S.; Volanschi, E. Studies on the anticancer drug mitoxantrone–DNA–sodium dodecyl sulfate system. J. Mol. Liq. 2015, 208, 333–341. [Google Scholar] [CrossRef]
- Digman, J.D.; Qu, X.; Ren, J.; Chaires, J.B. Daunomycin binding to detergent micelles: A model system for evaluating the hydrophobic contribution to drug–DNA intercations. J. Phys. Chem. B 2007, 111, 11576–11584. [Google Scholar]
- Westerlund, F.; Wilhelmsson, L.M.; Norden, B.; Lincoln, P. Micelle-sequestered dissociation of cationic DNA-intercalated drugs: Unexpected surfactant-induced rate enhancement. J. Am. Chem. Soc. 2003, 125, 3773–3779. [Google Scholar] [CrossRef] [PubMed]
- Mitra, R.K.; Sinha, S.S.; Pal, S.K. Interactions of Nile Blue with micelles, reverse micelles and a genomic DNA. J. Fluoresc. 2008, 18, 423–432. [Google Scholar] [CrossRef] [PubMed]
- Mitra, R.K.; Sinha, S.S.; Maiti, S.; Pal, S.K. Sequence dependent ultrafast electron transfer of Nile Blue in oligonucleotides. J. Fluoresc. 2009, 19, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Izumrudov, V.A.; Zhirykova, M.V.; Goulko, A.A. Ethidium bromide as a promising probe for studying DNA interaction with cationic amphiphiles and stability of the resulting complexes. Langmuir 2002, 18, 10348–10356. [Google Scholar] [CrossRef]
- Patra, A.; Hazra, S.; Kumar, G.S.; Mitra, R.K. Entropy contribution toward micelle driven deintercalation of drug–DNA complex. J. Phys. Chem. B 2014, 118, 901–908. [Google Scholar] [CrossRef] [PubMed]
- Patra, A.; Hazra, S.; Samanta, N.; Kumar, G.S.; Mitra, R.K. Micelle induced dissociation of DNA-ligand complexes: The effect of ligand binding specificity. Int. J. Biol. Macromol. 2016, 82, 418–424. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.J.; Im, S.S.; Oh, S.G. Investigation on the solubilization locus of aniline-HCl salt in SDS micelles with 1H NMR spectroscopy. Langmuir 2001, 17, 565–566. [Google Scholar] [CrossRef]
- Burns, C.P.; Haugstad, B.N.; Mossman, C.J.; North, J.A.; Ingraham, L.M. Membrane lipid alteration: Effect on cellular uptake of mitoxantrone. Lipids 1988, 23, 393–397. [Google Scholar] [CrossRef] [PubMed]
- Sabate, R.; Gallardo, M.; Estelrich, J. Location of pinacyanol in micellar solutions of N-alkyl trimethylammonium bromide surfactants. J. Colloid Interface Sci. 2001, 233, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Mukerjee, P.; Ray, A. Charge-transfer interactions and the polarity at the surface of micelles of long-chain pyridinium iodides. J. Phys. Chem. 1966, 70, 2144–2149. [Google Scholar] [CrossRef] [PubMed]
- Sarpal, R.S.; Belletete, M.; Durocher, G. Fluorescence probing and proton-transfer equilibrium reactions in water, SDS, and CTAB using 3,3-dimethyl-2-phenyl-3H-indole. J. Phys. Chem. 1993, 97, 5007–5013. [Google Scholar] [CrossRef]
- Mukerjee, P.; Cardinal, J.R. Benzene derivatives and naphthalene solubilized in micelles. Polarity of microenvironments, location and distribution in micelles, and correlation with surface activity in hydrocarbon-water systems. J. Phys. Chem. 1978, 82, 1620–1627. [Google Scholar] [CrossRef]
- Singh, S.K.; Kishore, N. Volumetric properties of amino acids and hen-egg white lysozyme in aqueous Triton X-100 at 298.15 K. J. Solut. Chem. 2004, 33, 1411–1427. [Google Scholar] [CrossRef]
- Shen, X.; Belletete, M.; Durocher, G. Study of the interactions between substituted 2,2′-bithiophenes and cyclodextrins. Chem. Phys. Lett. 1998, 298, 201–210. [Google Scholar] [CrossRef]
- Usman, M.; Siddiq, M. Surface and micellar properties of chloroquine diphosphate and its interactions with surfactants and human serum albumin. J. Chem. Thermodyn. 2013, 58, 359–366. [Google Scholar] [CrossRef]
- Sepulveda, L.; Lissi, E.; Quina, F. Interactions of neutral molecules with ionic micelles. Adv. Colloid Int. Sci. 1986, 25, 1–57. [Google Scholar] [CrossRef]
- Kawamura, H.; Manabe, M.; Miyamoto, Y.; Fujita, Y.; Tokunaga, S. Partititon coefficients of homologous ω-phenylalkanols between water and sodium dodecyl sulfate micelles. J. Phys. Chem. 1989, 93, 5536–5540. [Google Scholar] [CrossRef]
- Reynolds, J.A.; Stoeckenius, W. Molecular weight of bacteriorhodopsin solubilized in Triton X-100. Proc. Natl. Acad. Sci. USA 1977, 74, 2803–2804. [Google Scholar] [CrossRef]
- Garstecki, P.; Fuerstman, M.J.; Whitesides, G.M. Nonlinear dynamics of a flow-focusing bubble generator: An inverted dripping faucet. Phys. Rev. Lett. 2005, 94, 234502–234504. [Google Scholar] [CrossRef] [PubMed]
- Hillgren, A.; Evertsson, H.; Alden, M. Interaction between lactate dehydrogenase and Tween 80 in aqueous solution. Pharm. Res. 2002, 19, 105–110. [Google Scholar] [CrossRef]
- Agrawal, P.; Barthwal, S.K.; Barthwal, R. Studies on self-aggregation of anthracycline drugs by restrained molecular dynamics approach using nuclear magnetic resonance spectroscopy supported by absorption, fluorescence, diffusion ordered spectroscopy and mass spectrometry. Eur. J. Med. Chem. 2009, 44, 1437–1451. [Google Scholar] [CrossRef] [PubMed]
- Evstigneev, M.P.; Khomich, V.V.; Davies, D.B. Self-association of daunomycin antibiotic in various buffer solutions. Russ. J. Phys. Chem. 2006, 80, 741–746. [Google Scholar] [CrossRef]
- Sample Availability: Not Available.


| Mitoxantrone | pH 7.4 | pH 10 | References | ||
|---|---|---|---|---|---|
| λD | λM | λD | λM | ||
| Buffer | 610 | 660 | 614 | 666 | [45] |
| CTAB | 623 | 675 | 624 | 678 | [42] |
| SDS | 614 | 665 | 619 | 672 | [41] |
| Triton X-100 | 615 | 666 | 623 | 676 | [43] |
| Tween-20 | 617 | 667 | 624 | 676 | [43] |
| Tween-80 | 618 | 669 | 624 | 676 | [43] |
| Surfactant | pH 7.4 | pH 10 | References | ||
|---|---|---|---|---|---|
| Kb/M−1 | ∆Gb0/kJ/mol | Kb/M−1 | ∆Gb0/kJ/mol | ||
| CTAB | 2933 | −19.78 | 4365 | −20.76 | [42] |
| SDS | 982 | −16.77 | 1193 | −17.25 | [41] |
| Triton X-100 | 30 | −8.52 | 472 | −15.25 | [43] |
| Tween-20 | 52 | −9.79 | 610 | −15.89 | [43] |
| Tween-80 | 71 | −10.56 | 798 | −16.56 | [43] |
| Surfactant | pH 7.4 | pH 10 | References | ||
|---|---|---|---|---|---|
| Kx | ∆Gx0/kJ/mol | Kx | ∆Gx0/kJ/mol | ||
| CTAB | 1.72 × 105 | −29.86 | 2.65 × 105 | −30.93 | [42] |
| SDS | 4.79 × 104 | −26.24 | 6.98 × 104 | −27.16 | [41] |
| Triton X-100 | 8.31 × 103 | −22.34 | 1.33 × 105 | −29.22 | [43] |
| Tween-20 | 1.73 × 103 | −18.40 | 3.64 × 104 | −26.01 | [43] |
| Tween-80 | 3.11 × 103 | −19.91 | 5.61 × 104 | −27.08 | [43] |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Enache, M.; Toader, A.M.; Enache, M.I. Mitoxantrone-Surfactant Interactions: A Physicochemical Overview. Molecules 2016, 21, 1356. https://doi.org/10.3390/molecules21101356
Enache M, Toader AM, Enache MI. Mitoxantrone-Surfactant Interactions: A Physicochemical Overview. Molecules. 2016; 21(10):1356. https://doi.org/10.3390/molecules21101356
Chicago/Turabian StyleEnache, Mirela, Ana Maria Toader, and Madalin Iancu Enache. 2016. "Mitoxantrone-Surfactant Interactions: A Physicochemical Overview" Molecules 21, no. 10: 1356. https://doi.org/10.3390/molecules21101356
APA StyleEnache, M., Toader, A. M., & Enache, M. I. (2016). Mitoxantrone-Surfactant Interactions: A Physicochemical Overview. Molecules, 21(10), 1356. https://doi.org/10.3390/molecules21101356







