Relationship between the Ingestion of a Polyphenol-Rich Drink, Hepcidin Hormone, and Long-Term Training
Abstract
:1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Chemicals
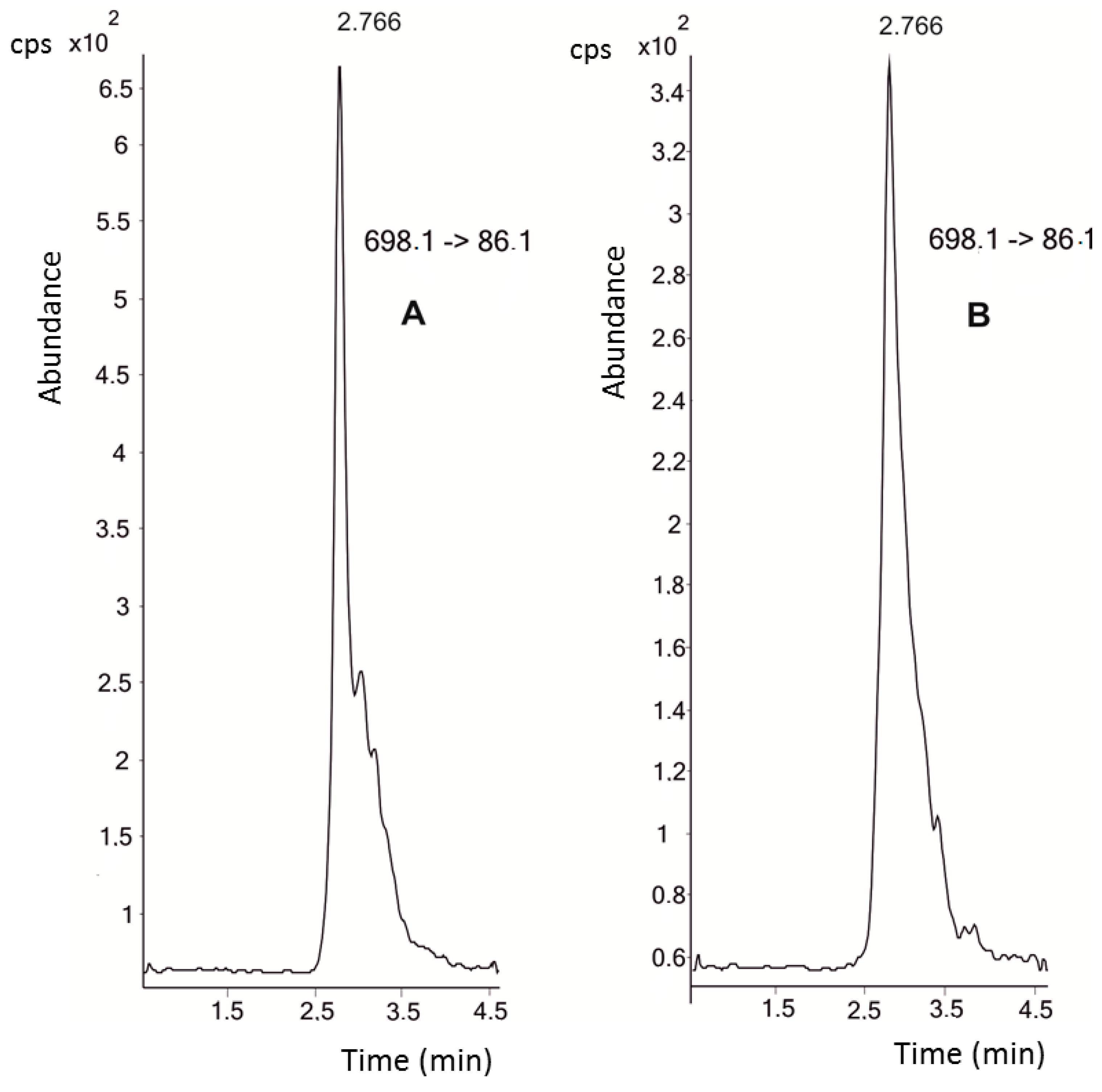
4.2. Instrumental—UHPLC-MS/MS Analysis
4.3. Extraction of Hepcidin from Human Plasma
4.4. Dietary Intervention
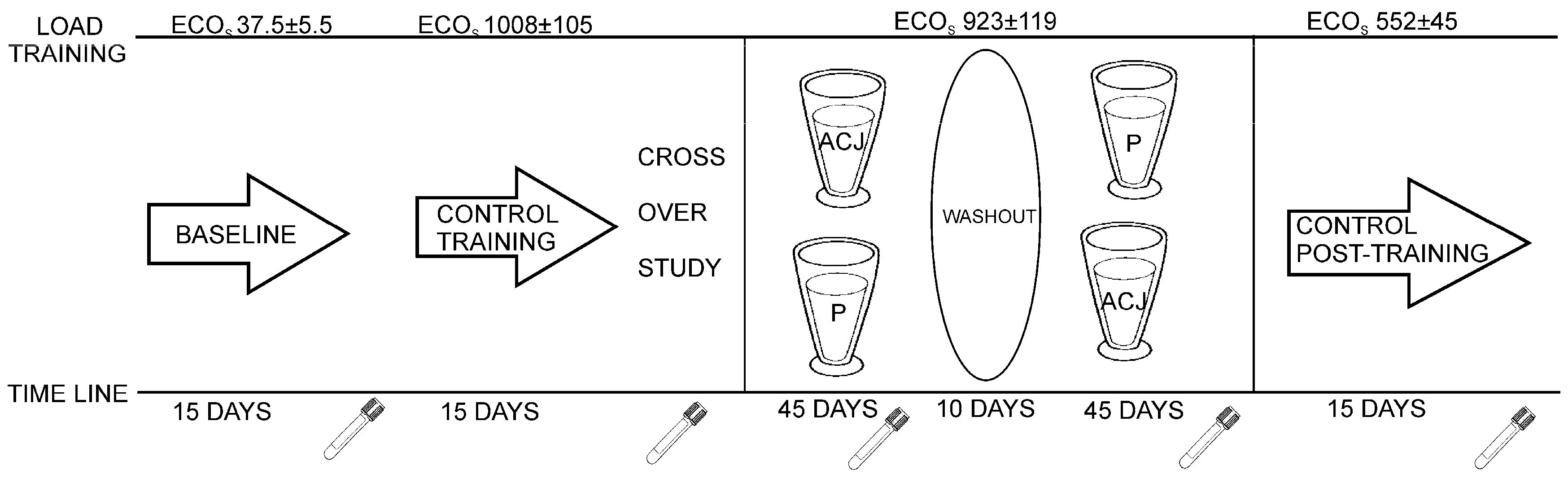
4.5. Sample Collection and Design of the Study
4.6. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Nicolas, G.; Bennoun, M.; Devaux, I.; Beaumont, C.; Grandchamp, B.; Kahn, A.; Vaulont, S. Lack of hepcidin gene expression and severe tissue iron overload in upstream stimulatory factor 2 (USF2) knockout mice. Proc. Natl. Acad. Sci. USA 2001, 98, 8780–8785. [Google Scholar] [CrossRef] [PubMed]
- Babitt, J.L.; Huang, F.W.; Wrighting, D.M.; Xia, Y.; Sidis, Y.; Samad, T.A.; Campagna, J.A.; Chung, R.T.; Schneyer, A.L.; Woolf, C.J. Bone morphogenetic protein signaling by hemojuvelin regulates hepcidin expression. Nat. Genet. 2006, 38, 531–539. [Google Scholar] [CrossRef] [PubMed]
- Álvarez, F.; Fernández-Ruiz, M.; Aguado, J.M. Hierro e infección fúngica invasiva. Rev. Iberoam. Micol. 2013, 30, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Rosolen, G.; Eberle, E.; Torres, F. Conceptos actuales sobre fisiología y patología del hierro. Hematología 2010, 14, 48–57. [Google Scholar]
- Nicolas, G.; Viatte, L.; Bennoun, M.; Beaumont, C.; Kahn, A.; Vaulont, S. Hepcidin, a new iron regulatory peptide. Blood Cell. Mol. Dis. 2002, 29, 327–335. [Google Scholar] [CrossRef]
- Ruckman, K.S.; Sherman, A.R. Effects of exercise on iron and copper metabolism in rats. J. Nutr. 1981, 111, 1593–1601. [Google Scholar] [PubMed]
- Di Santolo, M.; Stel, G.; Banfi, G.; Gonano, F.; Cauci, S. Anemia and iron status in young fertile non-professional female athletes. Eur. J. Appl. Physiol. 2008, 102, 703–709. [Google Scholar] [CrossRef] [PubMed]
- Zoller, H.; Vogel, W. Iron supplementation in athletes—First do no harm. Nutrition 2004, 20, 615–619. [Google Scholar] [CrossRef] [PubMed]
- Newlin, M.K. The Effects of Acute Exercise on Hepcidin in Women; The Florida State University: Tallahassee, FL, USA, 2009. [Google Scholar]
- Nicolas, G.; Chauvet, C.; Viatte, L.; Danan, J.L.; Bigard, X.; Devaux, I.; Beaumont, C.; Kahn, A.; Vaulont, S. The gene encoding the iron regulatory peptide hepcidin is regulated by anemia, hypoxia, and inflammation. J. Clin. Investig. 2002, 110, 1037–1044. [Google Scholar] [CrossRef] [PubMed]
- Khalil, A.; Goodhand, J.R.; Wahed, M.; Yeung, J.; Ali, F.R.; Rampton, D.S. Efficacy and tolerability of intravenous iron dextran and oral iron in inflammatory bowel disease: A case-matched study in clinical practice. Eur. J. Gastroenterol. Hepatol. 2011, 23, 1029–1035. [Google Scholar] [CrossRef] [PubMed]
- Wrighting, D.M.; Andrews, N.C. Interleukin-6 induces hepcidin expression through STAT3. Blood 2006, 108, 3204–3209. [Google Scholar] [CrossRef] [PubMed]
- Song, S.-N.J.; Tomosugi, N.; Kawabata, H.; Ishikawa, T.; Nishikawa, T.; Yoshizaki, K. Down-regulation of hepcidin resulting from long-term treatment with an anti–IL-6 receptor antibody (tocilizumab) improves anemia of inflammation in multicentric Castleman disease. Blood 2010, 116, 3627–3634. [Google Scholar] [CrossRef] [PubMed]
- Peternelj, T.-T.; Coombes, J.S. Antioxidant supplementation during exercise training. Sports Med. 2011, 41, 1043–1069. [Google Scholar] [CrossRef] [PubMed]
- Medina, S.; Domínguez-Perles, R.; García-Viguera, C.; Cejuela-Anta, R.; Martínez-Sanz, J.M.; Ferreres, F.; Gil-Izquierdo, A. Physical activity increases the bioavailability of flavanones after dietary aronia-citrus juice intake in triathletes. Food Chem. 2012, 135, 2133–2137. [Google Scholar] [CrossRef] [PubMed]
- Bansal, S.S.; Abbate, V.; Bomford, A.; Halket, J.M.; Macdougall, I.C.; Thein, S.L.; Hider, R.C. Quantitation of hepcidin in serum using ultra-high-pressure liquid chromatography and a linear ion trap mass spectrometer. Rapid Commun. Mass Spectrom. 2010, 24, 1251–1259. [Google Scholar] [CrossRef] [PubMed]
- Fallon, K.E. The acute phase response and exercise: The ultramarathon as prototype exercise. Clin. J. Sport Med. 2001, 11, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Roecker, L.; Meier-Buttermilch, R.; Brechtel, L.; Nemeth, E.; Ganz, T. Iron-regulatory protein hepcidin is increased in female athletes after a marathon. Eur. J. Appl. Physiol. 2005, 95, 569–571. [Google Scholar] [CrossRef] [PubMed]
- Peeling, P.; Dawson, B.; Goodman, C.; Landers, G.; Wiegerinck, E.T.; Swinkels, D.W.; Trinder, D. Effects of exercise on hepcidin response and iron metabolism during recovery. Int. J. Sport Nutr. 2009, 19, 583–597. [Google Scholar] [CrossRef]
- McClung, J.P.; Martini, S.; Murphy, N.E.; Montain, S.J.; Margolis, L.M.; Thrane, I.; Spitz, M.G.; Blatny, J.-M.; Young, A.J.; Gundersen, Y.; et al. Effects of a 7-day military training exercise on inflammatory biomarkers, serum hepcidin, and iron status. Nutr. J. 2013, 12, 141. [Google Scholar] [CrossRef] [PubMed]
- Skarpańska-Stejnborn, A.; Basta, P.; Sadowska, J.; Pilaczyńska-Szczeńniak, Ł. Effect of supplementation with chokeberry juice on the inflammatory status and markers of iron metabolism in rowers. J. Int. Soc. Sports Nutr. 2014, 11, 48. [Google Scholar] [CrossRef] [PubMed]
- Peeling, P.; Dawson, B.; Goodman, C.; Landers, G.; Wiegerinck, E.T.; Swinkels, D.W.; Trinder, D. Cumulative effects of consecutive running sessions on hemolysis, inflammation and hepcidin activity. Eur. J. Appl. Physiol. 2009, 106, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Dominguez, R.; Garnacho-Castaño, M.V.; Maté-Muñoz, J.L. Effect of hepcidin on iron metabolism in athletes. Nutr. Hosp. 2014, 30, 1218–1231. [Google Scholar] [PubMed]
- Auersperger, I.; Knap, B.; Jerin, A.; Blagus, R.; Lainscak, M.; Skitek, M.; Skof, B. The effects of 8 weeks of endurance running on hepcidin concentrations, inflammatory parameters, and iron status in female runners. Int. J. Sport Nutr. Exerc. Metab. 2012, 22, 55. [Google Scholar] [CrossRef] [PubMed]
- Medina, S.; Domínguez-Perles, R.; Cejuela-Anta, R.; Villaño, D.; Martínez-Sanz, J.M.; Gil, P.; García-Viguera, C.; Ferreres, F.; Gil, J.I.; Gil-Izquierdo, A. Assessment of oxidative stress markers and prostaglandins after chronic training of triathletes. Prostaglandins Lipid Mediat. 2012, 99, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Kasapis, C.; Thompson, P.D. The effects of physical activity on serum C-reactive protein and inflammatory markers: A systematic review. J. Am. Coll. Cardiol. 2005, 45, 1563–1569. [Google Scholar] [CrossRef] [PubMed]
- García-Flores, L.A.; Medina, S.; Cejuela-Anta, R.; Martínez-Sanz, J.M.; Abellán, Á.; Genieser, H.-G.; Ferreres, F.; Gil-Izquierdo, Á. DNA catabolites in triathletes: Effects of supplementation with an aronia–Citrus juice (polyphenols-rich juice). Food Funct. 2016, 7, 2084–2093. [Google Scholar] [CrossRef] [PubMed]
- Hurrell, R.F.; Reddy, M.; Cook, J.D. Inhibition of non-haem iron absorption in man by polyphenolic-containing beverages. Br. J. Nutr. 1999, 81, 289–295. [Google Scholar] [PubMed]
- Hurrell, R.; Egli, I. Iron bioavailability and dietary reference values. Am. J. Clin.Nutr. 2010, 91, 1461S–1467S. [Google Scholar] [CrossRef] [PubMed]
- Hider, R.C.; Liu, Z.D.; Khodr, H.H. Metal chelation of polyphenols. Methods Enzymol. 2001, 335, 190–203. [Google Scholar] [PubMed]
- Perron, N.R.; Brumaghim, J.L. A review of the antioxidant mechanisms of polyphenol compounds related to iron binding. Cell Biochem. Biophys. 2009, 53, 75–100. [Google Scholar] [CrossRef] [PubMed]
- Siegenberg, D.; Baynes, R.D.; Bothwell, T.H.; Macfarlane, B.J.; Lamparelli, R.D.; Car, N.; MacPhail, P.; Schmidt, U.; Tal, A.; Mayet, F. Ascorbic acid prevents the dose-dependent inhibitory effects of polyphenols and phytates on nonheme-iron absorption. Am. J. Clin. Nutr. 1991, 53, 537–541. [Google Scholar] [PubMed]
- Lesjak, M.; Hoque, R.; Balesaria, S.; Skinner, V.; Debnam, E.S.; Srai, S.K.; Sharp, P.A. Quercetin inhibits intestinal iron absorption and ferroportin transporter expression in vivo and in vitro. PLoS ONE 2014, 9, e102900. [Google Scholar] [CrossRef] [PubMed]
- Boato, F.; Wortley, G.M.; Liu, R.H.; Glahn, R.P. Red grape juice inhibits iron availability: Application of an in vitro digestion/Caco-2 cell model. J. Agric. Food Chem. 2002, 50, 6935–6938. [Google Scholar] [CrossRef] [PubMed]
- Martí, N.; Mena, P.; Cánovas, J.; Micol, V.; Saura, D. Vitamin C and the role of citrus juices as functional food. Natl. Prod. Commun. 2009, 4, 677–700. [Google Scholar]
- González-Molina, E.; Moreno, D.A.; García-Viguera, C. Aronia-enriched lemon juice: A new highly antioxidant beverage. J. Agric. Food Chem. 2008, 56, 11327–11333. [Google Scholar] [CrossRef] [PubMed]
- Galesloot, T.E.; Vermeulen, S.H.; Geurts-Moespot, A.J.; Klaver, S.M.; Kroot, J.J.; van Tienoven, D.; Wetzels, J.F.; Kiemeney, L.A.; Sweep, F.C.; den Heijer, M. Serum hepcidin: Reference ranges and biochemical correlates in the general population. Blood 2011, 117, e218–e225. [Google Scholar] [CrossRef] [PubMed]
- Den Elzen, W.P.; de Craen, A.J.; Wiegerinck, E.T.; Westendorp, R.G.; Swinkels, D.W.; Gussekloo, J. Plasma hepcidin levels and anemia in old age. The Leiden 85-Plus Study. Haematologica 2013, 98, 448–454. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uijterschout, L.; Swinkels, D.W.; Domellöf, M.; Lagerqvist, C.; Hudig, C.; Tjalsma, H.; Vos, R.; van Goudoever, J.B.; Brus, F. Serum hepcidin measured by immunochemical and mass-spectrometric methods and their correlation with iron status indicators in healthy children aged 0.5–3 y. Pediatr. Res. 2014, 76, 409–414. [Google Scholar] [CrossRef] [PubMed]
- Auersperger, I.; Škof, B.; Leskošek, B.; Knap, B.; Jerin, A.; Lainscak, M. Exercise-induced changes in iron status and hepcidin response in female runners. PLoS ONE 2013, 8, e58090. [Google Scholar] [CrossRef] [PubMed]
- Fernández, C.; Santos, H.M.; Ruíz-Romero, C.; Blanco, F.J.; Capelo-Martínez, J.L. A comparison of depletion versus equalization for reducing high-abundance proteins in human serum. Electrophoresis 2011, 32, 2966–2974. [Google Scholar] [CrossRef] [PubMed]
- Itkonen, O.; Stenman, U.-H.; Parkkinen, J.; Soliymani, R.; Baumann, M.; Hämäläinen, E. Binding of hepcidin to plasma proteins. Clin. Chem. 2012, 58, 1158–1160. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Murphy, A.T.; Witcher, D.R.; Luan, P.; Wroblewski, V.J. Quantitation of hepcidin from human and mouse serum using liquid chromatography tandem mass spectrometry. Blood 2007, 110, 1048–1054. [Google Scholar] [CrossRef] [PubMed]
- Kobold, U.; Dülffer, T.; Dangl, M.; Escherich, A.; Kubbies, M.; Röddiger, R.; Wright, J.A. Quantification of hepcidin-25 in human serum by isotope dilution micro-HPLC–tandem mass spectrometry. Clin. Chem. 2008, 54, 1584–1586. [Google Scholar] [CrossRef] [PubMed]
- Lainé, F.; Laviolle, B.; Ropert, M.; Bouguen, G.; Morcet, J.; Hamon, C.; Massart, C.; Westermann, M.; Deugnier, Y.; Loréal, O. Early effects of erythropoietin on serum hepcidin and serum iron bioavailability in healthy volunteers. Eur. J. Appl. Physiol. 2012, 112, 1391–1397. [Google Scholar] [CrossRef] [PubMed]
- Cejuela Anta, R.; Esteve-Lanao, J. Training load quantification in triathlon. J. Hum. Sport Exerc. 2011, 6. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the aronia-citrus juice and placebo juice are available from the authors.
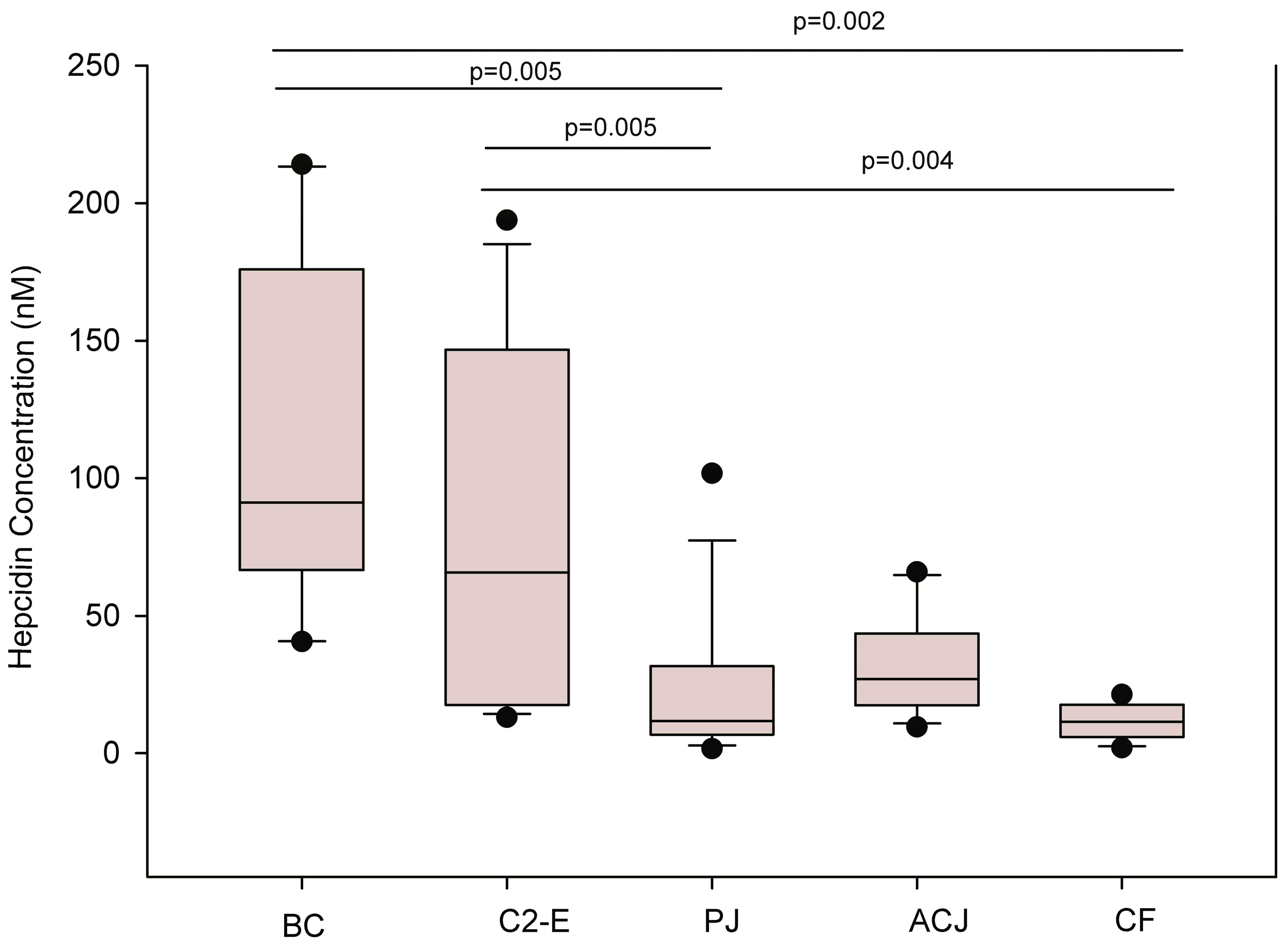
| Groups | Mean | STD | Confidence Intervals (CI) |
|---|---|---|---|
| Baseline Control | 116.6 | 63.3 | 78.29–154.83 |
| Control 2-Exercise | 78.7 | 71.1 | 44.23–124.91 |
| Placebo Juice | 15.2 | 12.6 | 5.60–38.04 |
| Aronia-Citrus juice | 29.4 | 19.6 | 18.61–40.27 |
| Final Control | 10.0 | 7.5 | 5.78–14.12 |
| Mean | SD | |
|---|---|---|
| Flavanones (mg) | ||
| Eriocitrin | 11.45 | 0.08 |
| Hesperidin | 13.54 | 0.14 |
| Flavones (mg) | ||
| Diosmetin-6,8-di-O-glucoside | 7.75 | 0.19 |
| Diosmin | <0.25 | |
| Vicenin-2 | 0.59 | 0.02 |
| Anthocyanins (mg) | ||
| Cyanidin 3-O-glucoside | 1.31 | 0.02 |
| Cyanidin 3-O-arabinoside | 9.19 | 0.20 |
| Cyanidin 3-O-galactoside | 15.08 | 0.10 |
| Cyanidin 3-O-xyloside | 1.11 | 0.03 |
| Total Anthocyanins | 26.70 | 0.35 |
| Hydroxycinnamic acids (mg) | ||
| Neochlorogenic acid | 19.72 | 0.17 |
| Chlorogenic acid | 14.69 | 0.13 |
| Σ Quercetin derivatives * (mg) | 4.31 | 0.13 |
| Mean | SD | |
|---|---|---|
| Energy intake (kcal·d−1) | 2446.3 | 528.5 |
| Carbohydrate (g·d−1) | 268.7 | 81.2 |
| Dietary fibre (g·d−1) | 21.4 | 8.3 |
| Sugar (g·d−1) | 100.9 | 28.8 |
| Proteins (g·d−1) | 108.5 | 35.6 |
| Total lipids (g·d−1) | 110.4 | 4.7 |
| SFA a (g·d−1) | 31.6 | 2.8 |
| MUFA b (g·d−1) | 56.7 | 0.1 |
| PUFA c (g·d−1) | 16.4 | 0.7 |
| Vitamin C (mg·d−1) | 156.9 | 30.9 |
| Vitamin A (µg·d−1) | 2198.7 | 1090.8 |
| Vitamin E (mg·d−1) | 17.5 | 5.0 |
| Vitamin D (mg·d−1) | 869.9 | 167.2 |
| Iron (mg·d−1) | 17.9 | 4.2 |
| Selenium (mg·d−1) | 126.4 | 33.1 |
| Mean | SD | |
|---|---|---|
| Weight (kg) | 62.3 | 10.5 |
| Height (m) | 1.7 | 0.1 |
| BMI a (kg·m−2) | 21.7 | 0.7 |
| Total fat (kg) | 9.0 | 0.4 |
| Lean weight (kg) | 26.1 | 7.5 |
| Subescapular skinfold (mm) | 11.2 | 2.2 |
| Tricipital skinfold (mm) | 12.6 | 5.2 |
| Bicipital skinfold (mm) | 7.9 | 3.5 |
| Ileocrestal skinfold (mm) | 15.9 | 5.4 |
| Supraespinal skinfold (mm) | 11.7 | 3.7 |
| Abdominal skinfold (mm) | 19.8 | 4.7 |
| Thigh skinfold (mm) | 21.1 | 8.7 |
| Calf skinfold (mm) | 11.9 | 4.1 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Villaño, D.; Vilaplana, C.; Medina, S.; Algaba-Chueca, F.; Cejuela-Anta, R.; Martínez-Sanz, J.M.; Ferreres, F.; Gil-Izquierdo, A. Relationship between the Ingestion of a Polyphenol-Rich Drink, Hepcidin Hormone, and Long-Term Training. Molecules 2016, 21, 1333. https://doi.org/10.3390/molecules21101333
Villaño D, Vilaplana C, Medina S, Algaba-Chueca F, Cejuela-Anta R, Martínez-Sanz JM, Ferreres F, Gil-Izquierdo A. Relationship between the Ingestion of a Polyphenol-Rich Drink, Hepcidin Hormone, and Long-Term Training. Molecules. 2016; 21(10):1333. https://doi.org/10.3390/molecules21101333
Chicago/Turabian StyleVillaño, Débora, Cristina Vilaplana, Sonia Medina, Francisco Algaba-Chueca, Roberto Cejuela-Anta, Jose Miguel Martínez-Sanz, Federico Ferreres, and Angel Gil-Izquierdo. 2016. "Relationship between the Ingestion of a Polyphenol-Rich Drink, Hepcidin Hormone, and Long-Term Training" Molecules 21, no. 10: 1333. https://doi.org/10.3390/molecules21101333
APA StyleVillaño, D., Vilaplana, C., Medina, S., Algaba-Chueca, F., Cejuela-Anta, R., Martínez-Sanz, J. M., Ferreres, F., & Gil-Izquierdo, A. (2016). Relationship between the Ingestion of a Polyphenol-Rich Drink, Hepcidin Hormone, and Long-Term Training. Molecules, 21(10), 1333. https://doi.org/10.3390/molecules21101333











