Terpenoids from the Marine-Derived Fungus Aspergillus fumigatus YK-7
Abstract
:1. Introduction
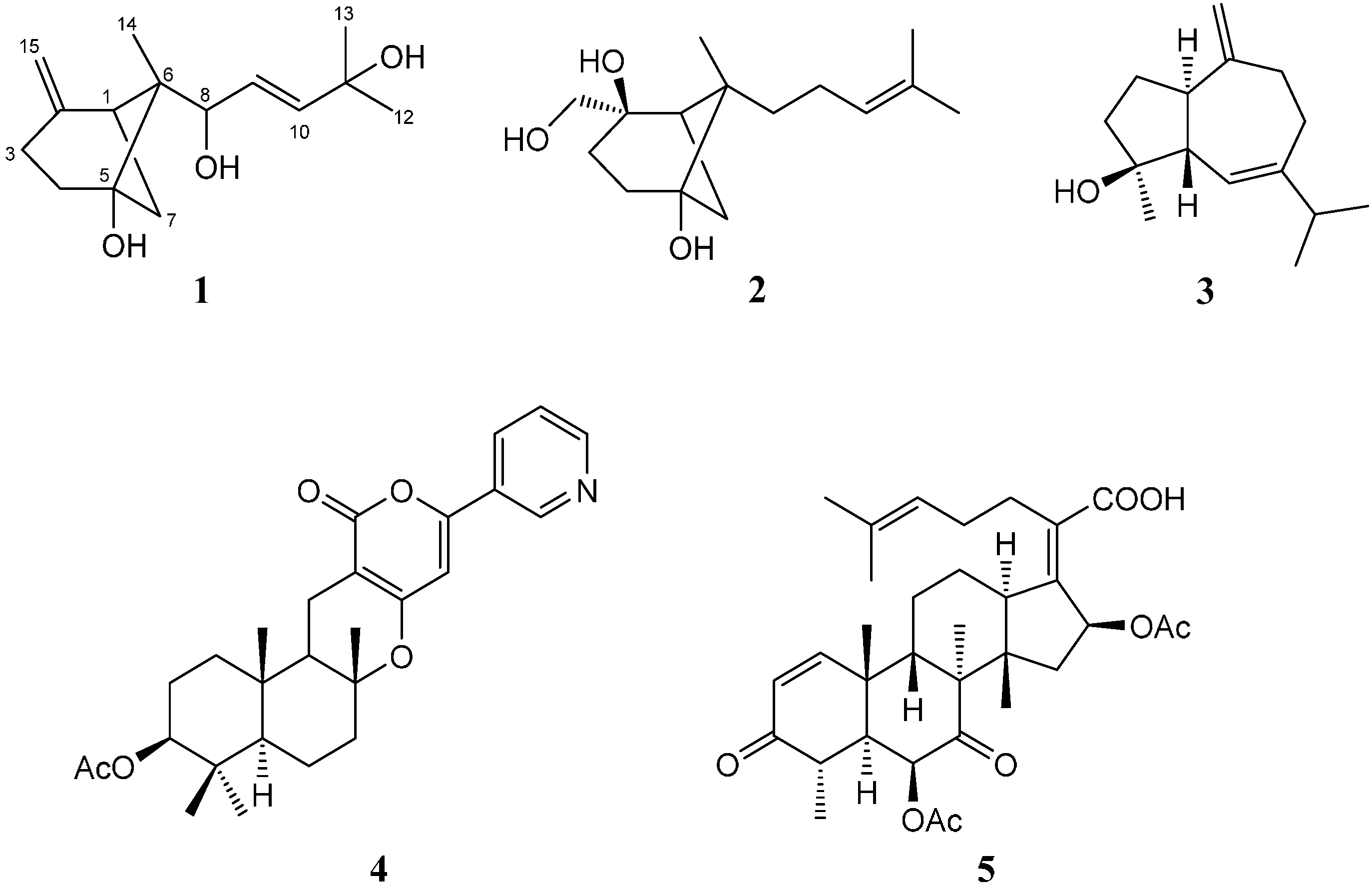
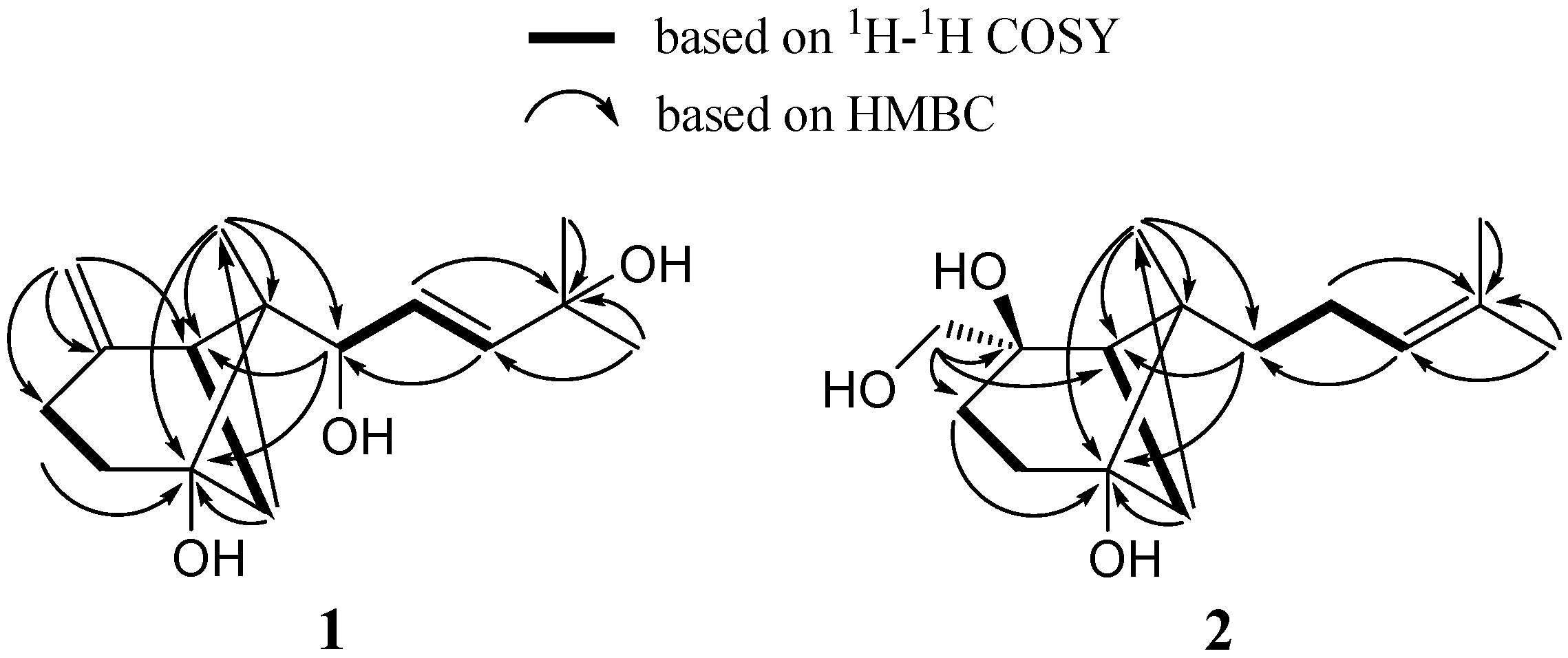
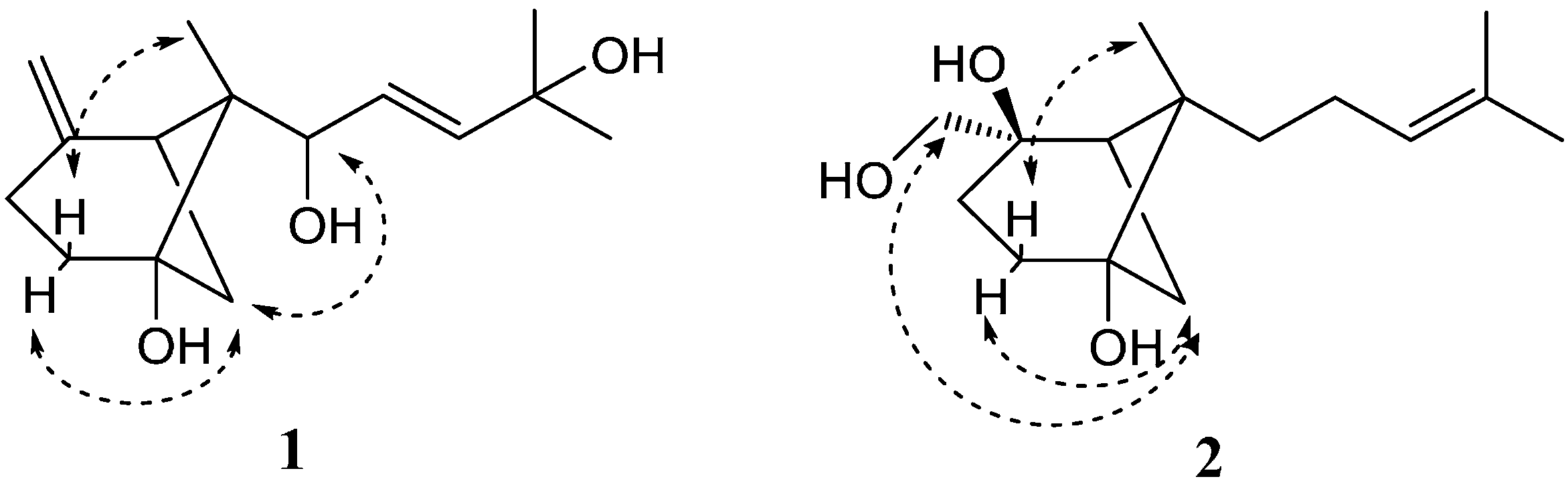
2. Results and Discussion
| 1 | 2 | |||
|---|---|---|---|---|
| Position | δC a | δH b (J in Hz) | δC a | δH b (J in Hz) |
| 1 | 42.2 | 2.33, d (7.5) | 39.7 | 2.15, m |
| 2 | 147.9 | 76.5 | ||
| 3 | 25.3 | 2.32, 2.61, m | 29.4 | 1.82, 1.86, m |
| 4 | 31.5 | 1.79 (α), 1.98 (β), m | 30.8 | 1.75 (α), 2.05 (β), m |
| 5 | 76.7 | 76.2 | ||
| 6 | 52.5 | 46.9 | ||
| 7 | 36.1 | 1.91, d (10.0) | 36.1 | 1.47, 2.17, m |
| 2.47, dd (10.0, 7.5) | ||||
| 8 | 74.5 | 4.85, d (6.1) | 34.5 | 1.45, 1.70, m |
| 9 | 125.4 | 5.66, dd (15.7, 6.1) | 23.2 | 2.03, 2.10, m |
| 10 | 140.5 | 5.88, d (15.7) | 124.9 | 5.15, t (7.1) |
| 11 | 70.9 | 131.8 | ||
| 12 | 29.8 | 1.33, s | 17.8 | 1.62, s |
| 13 | 30.1 | 1.33, s | 25.8 | 1.68, s |
| 14 | 10.9 | 0.80, s | 17.8 | 1.18, s |
| 15 | 108.0 | 4.57, br. s | 69.4 | 3.34, d (10.8) |
| 4.67, br. s | 3.47, d (10.8) | |||
| Compound | U937 Cells | PC-3 Cells |
|---|---|---|
| 1 | 84.9 ± 2.4 | >100 |
| 2 | >100 | >100 |
| 3 | 67.1 ± 1.9 | >100 |
| 4 | 4.2 ± 0.3 | >100 |
| 5 | 57.5 ± 3.2 | >100 |
| Doxorubicin hydrochloride | 0.021 ± 0.002 | 0.73 ± 0.04 |
3. Experimental Section
3.1. General Procedures
3.2. Fungal Material
3.3. Extraction and Isolation
3.4. Cell Culture and Growth-Inhibition Assay
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Xu, L.; Meng, W.; Cao, C.; Wang, J.; Shan, W.; Wang, Q. Antibacterial and antifungal compounds from marine fungi. Mar. Drugs 2015, 13, 3479–3513. [Google Scholar] [CrossRef] [PubMed]
- Gribble, G.-W. Biological activity of recently discovered halogenated marine natural products. Mar. Drugs 2015, 13, 4044–4136. [Google Scholar] [CrossRef] [PubMed]
- Frisvad, J.-C.; Rank, C.; Nielsen, K.-F.; Larsen, T.-O. Metabolomics of Aspergillus fumigatus. Med. Mycol. 2009, 47, S53–S71. [Google Scholar] [CrossRef] [PubMed]
- Molina, J.-M.; Tourneur, M.; Sarfati, C.; Chevret, S.; de Gouvello, A.; Gobert, J.-G.; Balkan, S.; Derouin, F. Fumagillin treatment of intestinal microsporidiosis. N. Engl. J. Med. 2002, 346, 1963–1969. [Google Scholar] [CrossRef] [PubMed]
- Killough, J.-H.; Magill, G.-B.; Smith, R.-C. The treatment of amebiasis with fumagillin. Science 1952, 115, 71–72. [Google Scholar] [CrossRef] [PubMed]
- Kruger, E.-A.; Figg, W.-D. TNP-470: An angiogenesis inhibitor in clinical development for cancer. Expert Opin. Investig. Drugs 2000, 9, 1383–1396. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, Z.-L.; Bai, J.; Zhang, L.-M.; Wu, X.; Zhang, L.; Pei, Y.-H.; Jing, Y.-K.; Hua, H.-M. 2,5-Diketopiperazines from the marine-derived fungus Aspergillus fumigatus YK-7. Chem. Biodivers. 2012, 9, 385–393. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.-C.; Tsunematsu, Y.; Dhingra, S.; Xu, W.; Fukutomi, M.; Chooi, Y.-H.; Cane, D.-E.; Calvo, A.-M.; Watanabe, K.; Tang, Y. Generation of complexity in fungal terpene biosynthesis: Discovery of a multifunctional cytochrome P450 in the fumagillin pathway. J. Am. Chem. Soc. 2014, 13, 4426–4436. [Google Scholar] [CrossRef] [PubMed]
- Nozoe, S.; Kobayashi, H.; Morisaki, N. Isolation of β-trans-bergamotene from Aspergillus fumigatus, a fumagillin producing fungi. Tetrahedron Lett. 1976, 17, 4625–4626. [Google Scholar] [CrossRef]
- Zhang, P.; Bao, B.; Dang, H.-T.; Hong, J.; Lee, H.-J.; Yoo, E.-S.; Bae, K.-S.; Jung, J.-H. Anti-inflammatory sesquiterpenoids from a sponge-derived fungus Acremonium sp. J. Nat. Prod. 2009, 72, 270–275. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, Y.-S.; Niwa, M.; Ron, E.; Snider, B.-B. Synthesis of terpenes containing the bicyclo[3.1.1]heptane ring system by the intramolecular [2 + 2] cycloaddition reaction of vinylketenes with alkenes. Preparation of chrysanthenone, β-pinene, β-cis-bergamotene, β-trans-bergamotene, β-copaene, and β-ylangene and lemnalol. J. Org. Chem. 1987, 52, 1568–1576. [Google Scholar]
- Peng, G.-P.; Tian, G.; Huang, X.-F.; Lou, F.-C. Guaiane-type sesquiterpenoids from Alisma orientalis. Phytochemistry 2003, 63, 877–881. [Google Scholar] [CrossRef]
- Tomoda, H.; Tabata, N.; Yang, D.-J.; Takayanagi, H.; Nishida, H.; Ōmura, S. Pyripyropenes, novel ACAT inhibitors produced by Aspergillus fumigatus III. Structure elucidation of pyripyropenes E to L. J. Antibiot. 1995, 48, 495–503. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.-L.; Mou, Y.; Shan, T.-J.; Li, Y.; Zhou, L.-G.; Wang, M.-G.; Wang, J.-G. Antimicrobial metabolites from the endophytic fungus Pichia guilliermondii isolated from Paris polyphylla var. yunnanensis. Molecules 2010, 15, 7961–7970. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-Y.; Kinoshita, H.; Ihara, F.; Igarashi, Y.; Nihira, T. Identification of novel derivative of helvolic acid from Metarhizium anisopliae grown in medium with insect component. J. Biosci. Bioeng. 2008, 105, 476–480. [Google Scholar] [CrossRef] [PubMed]
- Henry, T.; Iwen, P.-C.; Hinrichs, S.-H. Identification of Aspergillus species using internal transcribed spacer regions 1 and 2. J. Clin. Microbiol. 2000, 38, 1510–1515. [Google Scholar] [PubMed]
- Wang, F.; Hua, H.-M.; Pei, Y.-H.; Chen, D.; Jing, Y.-K. Triterpenoids from the resin of Styrax tonkinensis and their antiproliferative and differentiation effects in human leukemia HL-60 cells. J. Nat. Prod. 2006, 69, 807–810. [Google Scholar] [CrossRef] [PubMed]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Sample Availability: Sample of the compound 5 is available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Li, D.-H.; Li, Z.-L.; Sun, Y.-J.; Hua, H.-M.; Liu, T.; Bai, J. Terpenoids from the Marine-Derived Fungus Aspergillus fumigatus YK-7. Molecules 2016, 21, 31. https://doi.org/10.3390/molecules21010031
Wang Y, Li D-H, Li Z-L, Sun Y-J, Hua H-M, Liu T, Bai J. Terpenoids from the Marine-Derived Fungus Aspergillus fumigatus YK-7. Molecules. 2016; 21(1):31. https://doi.org/10.3390/molecules21010031
Chicago/Turabian StyleWang, Yu, Da-Hong Li, Zhan-Lin Li, Yan-Jun Sun, Hui-Ming Hua, Tao Liu, and Jiao Bai. 2016. "Terpenoids from the Marine-Derived Fungus Aspergillus fumigatus YK-7" Molecules 21, no. 1: 31. https://doi.org/10.3390/molecules21010031
APA StyleWang, Y., Li, D.-H., Li, Z.-L., Sun, Y.-J., Hua, H.-M., Liu, T., & Bai, J. (2016). Terpenoids from the Marine-Derived Fungus Aspergillus fumigatus YK-7. Molecules, 21(1), 31. https://doi.org/10.3390/molecules21010031





