Strong and Nonspecific Synergistic Antibacterial Efficiency of Antibiotics Combined with Silver Nanoparticles at Very Low Concentrations Showing No Cytotoxic Effect
Abstract
:1. Introduction
2. Results and Discussion
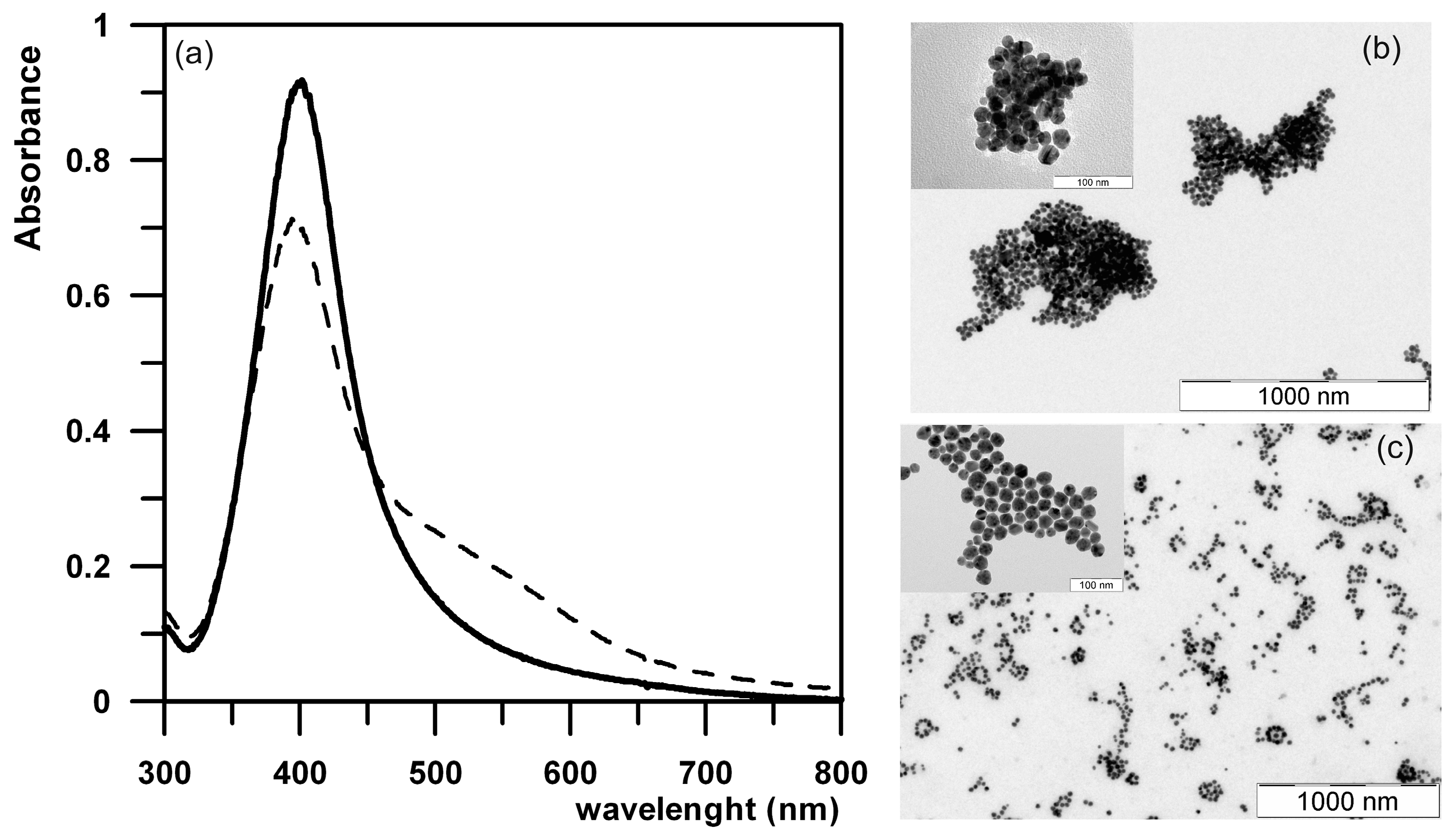
2.1. Synthesis of Silver Nanoparticles

2.2. Synergistic Effect of Antibiotics and Silver Nanoparticles
| Inhibition of Cell Wall Synthesis | Inhibition of Protein Synthesis | Inhibition of Nucleic Acid Synthesis | Alteration in Cytoplasmic Membrane |
|---|---|---|---|
| Ampicillin (AMP) a | Gentamicin (GEN) f | Oxolinic acid (OXO) k | Colistin (COL) m |
| Ampicillin/sulbactam (AMS) a | Tetracycline (TET) g | Ofloxacin (OFL) k | |
| Piperacillin (PIP) a | Amikacin (AMI) f | Ciprofloxacin (CIP) k | |
| Piperacillin/tazobactam (PPT) a | Chloramphenicol (CMP) h | Co-trimoxazole (COT) l | |
| Penicillin (PEN) a | Erythromycin (ERY) i | ||
| Oxacillin (OXA) a | Clindamycin (CLI) j | ||
| Cefazolin (CZL) b | |||
| Cefuroxime (CRX) b | |||
| Cefoxitin (CXT) b | |||
| Cefepime (CPM) b | |||
| Cefoperazone (CPR) b | |||
| Ceftazidime (CTZ) b | |||
| Meropenem (MER) c | |||
| Teicoplanin (TEI) d | |||
| Vancomycin (VAN) d | |||
| Aztreonam (AZT) e |
| AMP | AMS | CZL | CRX | CXT | GEN | COT | COL | OXO | OFL | TET | AZT | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ATB + 0 mg/L AgNPs | - | 8 | 2 | 1 | 1 | 0.5 | 4 | 0.5 | 0.5 | 0.03 | 4 | 0.03 |
| ATB + 0.6 mg/L AgNPs | - | 8 | 2 | 1 | 1 | 0.03 | 2 | 0.125 | 0.5 | 0.015 | 4 | 0.03 |
| ATB + 1.25 mg/L AgNPs | - | 8 | 2 | 1 | 1 | 0.06 | 2 | 0.125 | 0.5 | 0.015 | 2 | 0.03 |
| ATB + 2.5 mg/L AgNPs | 0.03 | 0.03 | 0.0019 | 0.00097 | 0.00097 | 0.00048 | 2 | 0.00097 | 0.5 | 0.00024 | 1 | 0.0078 |
| ATB + 5 mg/L AgNPs | 0.00097 | 0.00097 | 0.00097 | 0.00097 | 0.00097 | 0.00048 | 0.0019 | 0.00048 | 0.00097 | 0.00024 | 0.00048 | 0.0019 |
| PIP | PPT | AZT | MER | CTZ | CPR | CPM | GEN | AMI | COL | OFL | CIP | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ATB + 0 mg/L AgNPs | 4 | 4 | 4 | 1 | 1 | 4 | 2 | 0.25 | 1 | 0.5 | 1 | 0.125 |
| ATB + 0.6 mg/L AgNPs | 4 | 4 | 4 | 1 | 1 | 4 | 1 | 0.03 | 0.25 | 0.125 | 1 | 0.03 |
| ATB + 1.25 mg/L AgNPs | 2 | 4 | 4 | 0.5 | 1 | 4 | 1 | 0.015 | 0.25 | 0.06 | 1 | 0.03 |
| ATB + 2.5 mg/L AgNPs | 0.015 | 0.0039 | 0.00097 | 0.0019 | 0.00024 | 0.00048 | 0.00024 | 0.00048 | 0.00048 | 0.00048 | 0.00024 | 0.00024 |
| ATB + 5 mg/L AgNPs | 0.0078 | 0.0039 | 0.00097 | 0.0019 | 0.00024 | 0.00048 | 0.00024 | 0.00048 | 0.00048 | 0.00048 | 0.00024 | 0.00024 |
| PEN | OXA | AMS | CMP | TET | COT | ERY | CLI | CIP | GEN | TEI | VAN | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ATB + 0 mg/L AgNPs | 0.125 | 0.25 | 0.5 | 4 | 0.25 | 1 | 0.125 | 0.125 | 0.25 | 0.25 | 0.25 | 1 |
| ATB + 0.6 mg/L AgNPs | 0.015 | 0.25 | 0.125 | 4 | 0.015 | 0.03 | 0.06 | 0.015 | 0.03 | 0.25 | 0.03 | 0.03 |
| ATB + 1.25 mg/L AgNPs | 0.015 | 0.03 | 0.06 | 2 | 0.015 | 0.0078 | 0.06 | 0.015 | 0.015 | 0.06 | 0.015 | 0.00097 |
| ATB + 2.5 mg/L AgNPs | 0.00006 | 0.00048 | 0.00048 | 0.00048 | 0.00012 | 0.0019 | 0.00012 | 0.00012 | 0.00012 | 0.00048 | 0.00097 | 0.00048 |
2.3. Cytotoxicity of Silver Antibiotics, Nanoparticles and Their Combinations
| AgNPs/ATB | Corresponding to MIC (mg/L) | Below MIC (mg/L) |
|---|---|---|
| Ag NPs | 7.5 | 2.5 |
| AMS | 8 | 0.003 |
| CZL | 2 | 0.0019 |
| MER | 1 | 0.0019 |
| CMP | 4 | 0.00048 |
| GEN | 0.5 | - |
| VAN | 1 | - |
| TET | 4 | - |
| CIP | 0.25 | - |
| COT | 4 | - |
| COL | 0.5 | - |
| AMS | CZL | MER | CMP | GEN | VAN | TET | CIP | COT | COL | |
|---|---|---|---|---|---|---|---|---|---|---|
| ATB + 0 mg/L AgNPs | 160 | 200 | 250 | 200 | 250 | 200 | 200 | 120 | 120 | 120 |
| ATB + 7.5 mg/L AgNPs | 100 | 130 | 180 | 150 | 180 | 150 | 180 | 100 | 100 | 100 |
| ATB + 15 mg/L AgNPs | 80 | 90 | 130 | 100 | 150 | 100 | 150 | 90 | 90 | 80 |
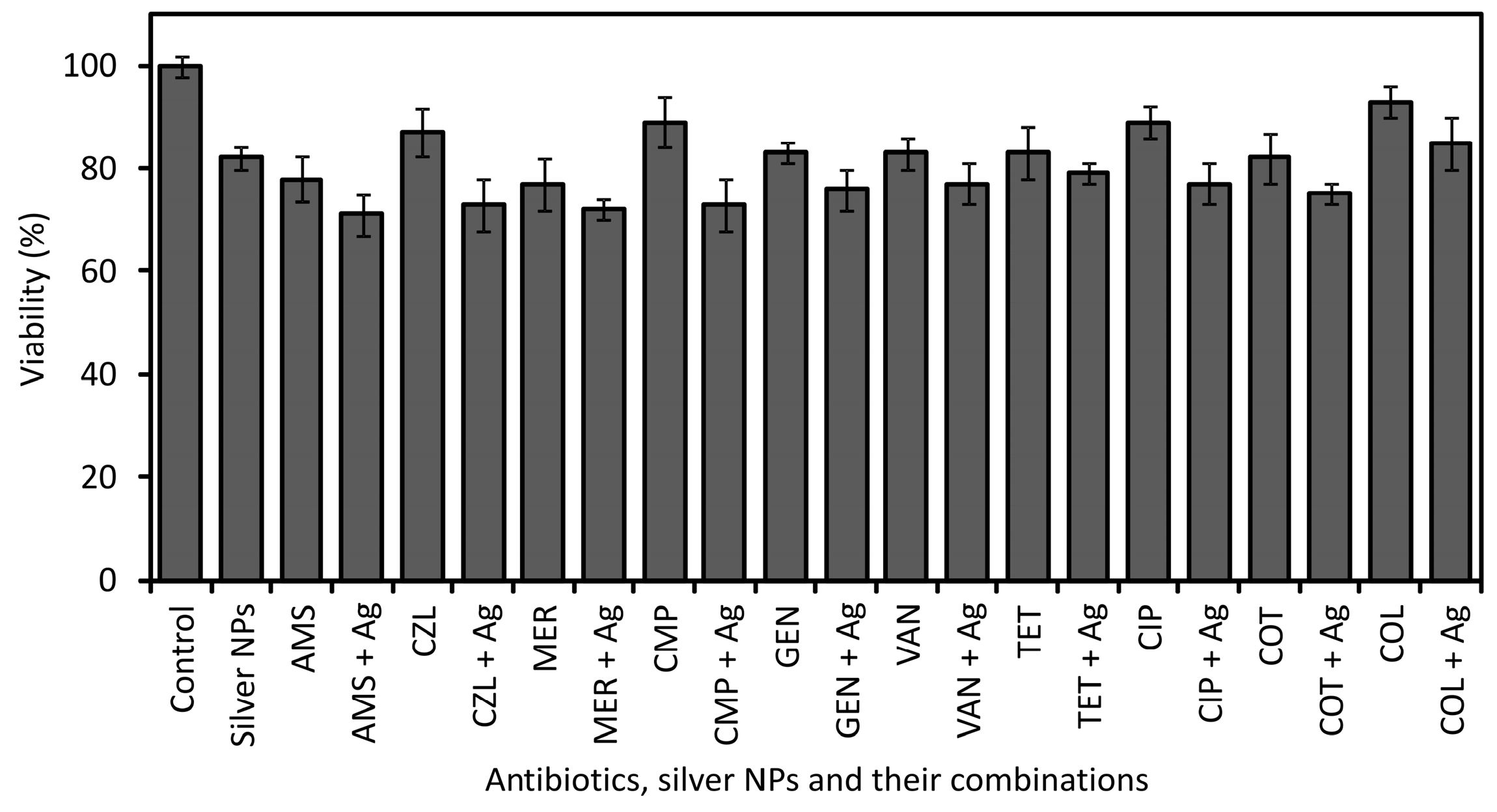
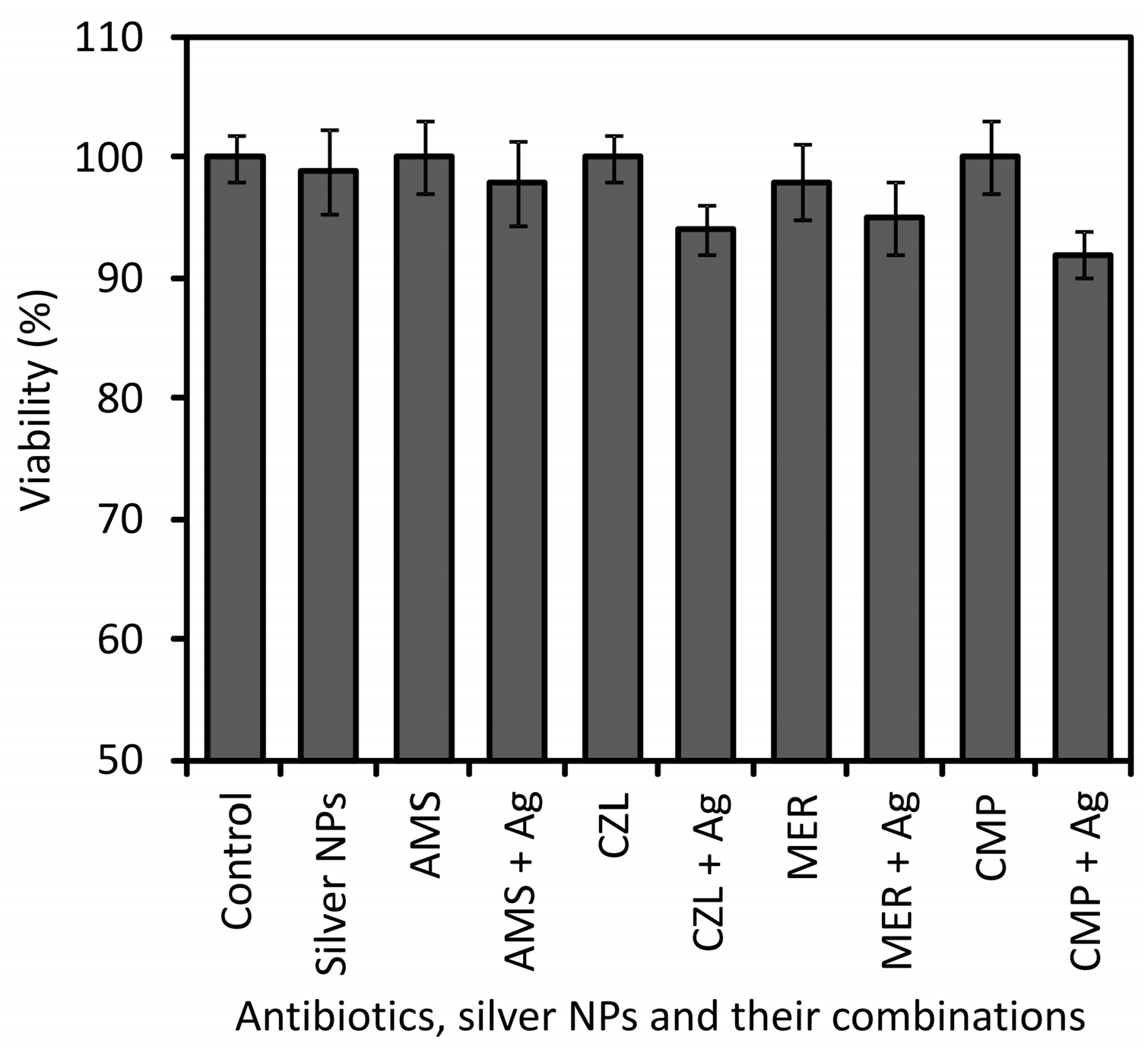
2.4. Discussion
3. Experimental Section
3.1. Chemicals and Biological Materials
3.2. Synthesis and Characterization of Silver Nanoparticles
3.3. Determination of the Synergistic Effect of Antibiotics and Silver Nanoparticles
3.4. Cytotoxicity Evaluation of Antibiotics, Silver Nanoparticles and Their Combinations
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Walsh, T.R.; Toleman, M.A. The emergence of pan-resistant Gram-negative pathogens merits a rapid global political response. J. Antimicrob. Chemother. 2012, 67, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Theuretzbacher, U. Accelerating resistance, inadequate antibacterial drug pipelines and international responses. Int. J. Antimicrob. Agents 2012, 39, 295–299. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, T.M.; Levy, S.B. The impact of antibiotic use on resistance development and persistence. Drug Resist. Update 2000, 3, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Mokaddas, E.; Rotimi, V.O.; Sanyal, S.C. In vitro activity of piperacillin/tazobactam versus other broad-spectrum antibiotics against nosocomial gram-negative pathogens isolated from burn patients. J. Chemother. 1998, 10, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Retsema, J.A.; English, A.R.; Girard, A.; Lynch, J.E.; Anderson, M.; Brennan, L.; Cimochowski, C.; Faiella, J.; Norcia, W.; Sawyer, P. Sulbactam ampicillin—in vitro spectrum, potency, and activity in models of acute infection. Rev. Infect. Dis. 1986, 8, S528–S534. [Google Scholar] [CrossRef] [PubMed]
- Todd, P.A.; Benfield, P. Amoxicillin clavulanic acid—An update of its antibacterial activity, pharmacokinetic properties and therapeutic use. Drugs 1990, 39, 264–307. [Google Scholar] [CrossRef] [PubMed]
- Antimicrobial Resistance Surveillance in Europe 2013. Annual Report of the European Antimicrobial Resistance Surveillance Network (Ears-Net). 978-92-9193-603-8. Stockholm, 2014. Avalaible online: http://www.ecdc.europa.eu/en/publications/Publications/antimicrobial-resistance-surveillance-europe-2013.pdf (accessed on 17 November 2014).
- Li, W.R.; Xie, X.B.; Shi, Q.S.; Duan, S.S.; Ouyang, Y.S.; Chen, Y.B. Antibacterial effect of silver nanoparticles on staphylococcus aureus. Biometals 2011, 24, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Li, W.R.; Xie, X.B.; Shi, Q.S.; Zeng, H.Y.; Ou-Yang, Y.S.; Chen, Y.B. Antibacterial activity and mechanism of silver nanoparticles on escherichia coli. Appl. Microbiol. Biot. 2010, 85, 1115–1122. [Google Scholar] [CrossRef] [PubMed]
- Cui, L.; Chen, P.Y.; Chen, S.D.; Yuan, Z.H.; Yu, C.P.; Ren, B.; Zhang, K.S. In situ study of the antibacterial activity and mechanism of action of silver nanoparticles by surface-enhanced raman spectroscopy. Anal. Chem. 2013, 85, 5436–5443. [Google Scholar] [CrossRef] [PubMed]
- Lara, H.H.; Ayala-Nunez, N.V.; Turrent, L.D.I.; Padilla, C.R. Bactericidal effect of silver nanoparticles against multidrug-resistant bacteria. World J. Microb. Biot. 2010, 26, 615–621. [Google Scholar] [CrossRef]
- Lok, C.N.; Ho, C.M.; Chen, R.; He, Q.Y.; Yu, W.Y.; Sun, H.Z.; Tam, P.K.H.; Chiu, J.F.; Che, C.M. Proteomic analysis of the mode of antibacterial action of silver nanoparticles. J. Proteome Res. 2006, 5, 916–924. [Google Scholar] [CrossRef] [PubMed]
- Morones, J.R.; Elechiguerra, J.L.; Camacho, A.; Holt, K.; Kouri, J.B.; Ramirez, J.T.; Yacaman, M.J. The bactericidal effect of silver nanoparticles. Nanotechnology 2005, 16, 2346–2353. [Google Scholar] [CrossRef] [PubMed]
- Sondi, I.; Salopek-Sondi, B. Silver nanoparticles as antimicrobial agent: A case study on e-coli as a model for gram-negative bacteria. J. Colloid Interf. Sci. 2004, 275, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Choi, O.; Hu, Z.Q. Size dependent and reactive oxygen species related nanosilver toxicity to nitrifying bacteria. Environ. Sci. Technol. 2008, 42, 4583–4588. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.S.; Kuk, E.; Yu, K.N.; Kim, J.H.; Park, S.J.; Lee, H.J.; Kim, S.H.; Park, Y.K.; Park, Y.H.; Hwang, C.Y.; et al. Antimicrobial effects of silver nanoparticles. Nanomed. Nanotechnol. 2007, 3, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.Y.; Qu, F.; Xu, H.; Lai, W.H.; Wang, Y.A.; Aguilar, Z.P.; Wei, H. Role of reactive oxygen species in the antibacterial mechanism of silver nanoparticles on escherichia coli o157:H7. Biometals 2012, 25, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Cabiscol, E.; Tamarit, J.; Ros, J. Oxidative stress in bacteria and protein damage by reactive oxygen species. Int. Microbiol. 2000, 3, 3–8. [Google Scholar] [PubMed]
- Kvitek, L.; Panacek, A.; Soukupova, J.; Kolar, M.; Vecerova, R.; Prucek, R.; Holecova, M.; Zboril, R. Effect of surfactants and polymers on stability and antibacterial activity of silver nanoparticles (nps). J. Phys. Chem. C 2008, 112, 5825–5834. [Google Scholar] [CrossRef]
- Panacek, A.; Kolar, M.; Vecerova, R.; Prucek, R.; Soukupova, J.; Krystof, V.; Hamal, P.; Zboril, R.; Kvitek, L. Antifungal activity of silver nanoparticles against candida spp. Biomaterials 2009, 30, 6333–6340. [Google Scholar] [CrossRef] [PubMed]
- Panacek, A.; Kvitek, L.; Prucek, R.; Kolar, M.; Vecerova, R.; Pizurova, N.; Sharma, V.K.; Nevecna, T.; Zboril, R. Silver colloid nanoparticles: Synthesis, characterization, and their antibacterial activity. J. Phys. Chem. B 2006, 110, 16248–16253. [Google Scholar] [CrossRef] [PubMed]
- Shrivastava, S.; Bera, T.; Roy, A.; Singh, G.; Ramachandrarao, P.; Dash, D. Characterization of enhanced antibacterial effects of novel silver nanoparticles. Nanotechnology 2007, 18, 225103. [Google Scholar] [CrossRef]
- Silver, S. Bacterial silver resistance: Molecular biology and uses and misuses of silver compounds. FEMS Microbiol. Rev. 2003, 27, 341–353. [Google Scholar] [CrossRef]
- Silver, S.; Phung, L.T.; Silver, G. Silver as biocides in burn and wound dressings and bacterial resistance to silver compounds. J. Ind. Microbiol. Biotechnol. 2006, 33, 627–634. [Google Scholar] [CrossRef] [PubMed]
- Kremer, A.N.; Hoffmann, H. Subtractive hybridization yields a silver resistance determinant unique to nosocomial pathogens in the enterobacter cloacae complex. J. Clin. Microbiol. 2012, 50, 3249–3257. [Google Scholar] [CrossRef] [PubMed]
- Haefeli, C.; Franklin, C.; Hardy, K. Plasmid-determined silver resistance in pseudomonas-stutzeri isolated from a silver mine. J. Bacteriol. 1984, 158, 389–392. [Google Scholar] [PubMed]
- Gupta, A.; Matsui, K.; Lo, J.F.; Silver, S. Molecular basis for resistance to silver cations in salmonella. Nat. Med. 1999, 5, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Li, X.Z.; Nikaido, H.; Williams, K.E. Silver-resistant mutants of escherichia coli display active efflux of ag+ and are deficient in porins. J. Bacteriol. 1997, 179, 6127–6132. [Google Scholar] [PubMed]
- Nies, D.H. Efflux-mediated heavy metal resistance in prokaryotes. FEMS Microbiol. Rev. 2003, 27, 313–339. [Google Scholar] [CrossRef]
- Potara, M.; Jakab, E.; Damert, A.; Popescu, O.; Canpean, V.; Astilean, S. Synergistic antibacterial activity of chitosan-silver nanocomposites on staphylococcus aureus. Nanotechnology 2011, 22, 135101. [Google Scholar] [CrossRef] [PubMed]
- Vertelov, G.K.; Krutyakov, Y.A.; Efremenkova, O.V.; Olenin, A.Y.; Lisichkin, G.V. A versatile synthesis of highly bactericidal myramistin (r) stabilized silver nanoparticles. Nanotechnology 2008, 19. [Google Scholar] [CrossRef] [PubMed]
- Ammons, M.C.B.; Ward, L.S.; James, G.A. Anti-biofilm efficacy of a lactoferrin/xylitol wound hydrogel used in combination with silver wound dressings. Int. Wound. J. 2011, 8, 268–273. [Google Scholar] [CrossRef] [PubMed]
- Ruden, S.; Hilpert, K.; Berditsch, M.; Wadhwani, P.; Ulrich, A.S. Synergistic interaction between silver nanoparticles and membrane-permeabilizing antimicrobial peptides. Antimicrob. Agents Chemother. 2009, 53, 3538–3540. [Google Scholar] [CrossRef] [PubMed]
- Birla, S.S.; Tiwari, V.V.; Gade, A.K.; Ingle, A.P.; Yadav, A.P.; Rai, M.K. Fabrication of silver nanoparticles by phoma glomerata and its combined effect against escherichia coli, pseudomonas aeruginosa and staphylococcus aureus. Lett. Appl. Microbiol. 2009, 48, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Fayaz, A.M.; Balaji, K.; Girilal, M.; Yadav, R.; Kalaichelvan, P.T.; Venketesan, R. Biogenic synthesis of silver nanoparticles and their synergistic effect with antibiotics: A study against gram-positive and gram-negative bacteria. Nanomed. Nanotechnol. 2010, 6, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Patil, S.; Ahire, M.; Kitture, R.; Kale, S.; Pardesi, K.; Cameotra, S.S.; Bellare, J.; Dhavale, D.D.; Jabgunde, A.; et al. Synthesis of silver nanoparticles using dioscorea bulbifera tuber extract and evaluation of its synergistic potential in combination with antimicrobial agents. Int. J. Nanomed. 2012, 7, 483–496. [Google Scholar]
- Muhsin, T.M.; Hachim, A.K. Mycosynthesis and characterization of silver nanoparticles and their activity against some human pathogenic bacteria. World J. Microbiol. Biotechnol. 2014, 30, 2081–2090. [Google Scholar] [CrossRef] [PubMed]
- Naqvi, S.Z.H.; Kiran, U.; Ali, M.I.; Jamal, A.; Hameed, A.; Ahmed, S.; Ali, N. Combined efficacy of biologically synthesized silver nanoparticles and different antibiotics against multidrug-resistant bacteria. Int. J. Nanomed. 2013, 8, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Sathiyanarayanan, G.; Kiran, G.S.; Selvin, J. Synthesis of silver nanoparticles by polysaccharide bioflocculant produced from marine bacillus subtilis msbn17. Colloid. Surf. B 2013, 102, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Shahverdi, A.R.; Fakhimi, A.; Shahverdi, H.R.; Minaian, S. Synthesis and effect of silver nanoparticles on the antibacterial activity of different antibiotics against Staphylococcus aureus and Escherichia coli. Nanomed. Nanotechnol. 2007, 3, 168–171. [Google Scholar] [CrossRef] [PubMed]
- Hwang, I.S.; Hwang, J.H.; Choi, H.; Kim, K.J.; Lee, D.G. Synergistic effects between silver nanoparticles and antibiotics and the mechanisms involved. J. Med. Microbiol. 2012, 61, 1719–1726. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Li, J.; Wu, C.Z.; Wu, Q.S.; Li, J. Synergistic antibacterial effects of beta-lactam antibiotic combined with silver nanoparticles. Nanotechnology 2005, 16, 1912–1917. [Google Scholar] [CrossRef]
- Markowska, K.; Grudniak, A.M.; Krawczyk, K.; Wrobel, I.; Wolska, K.I. Modulation of antibiotic resistance and induction of a stress response in pseudomonas aeruginosa by silver nanoparticles. J. Med. Microbiol. 2014, 63, 849–854. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Wagh, P.; Wadhwani, S.; Gaidhani, S.; Kumbhar, A.; Bellare, J.; Chopade, B.A. Synthesis, optimization, and characterization of silver nanoparticles from acinetobacter calcoaceticus and their enhanced antibacterial activity when combined with antibiotics. Int. J. Nanomed. 2013, 8, 4277–4289. [Google Scholar]
- Brown, A.N.; Smith, K.; Samuels, T.A.; Lu, J.R.; Obare, S.O.; Scott, M.E. Nanoparticles functionalized with ampicillin destroy multiple-antibiotic-resistant isolates of pseudomonas aeruginosa and enterobacter aerogenes and methicillin-resistant staphylococcus aureus. Appl. Environ. Microbiol. 2012, 78, 2768–2774. [Google Scholar] [CrossRef] [PubMed]
- Kvitek, L.; Prucek, R.; Panacek, A.; Novotny, R.; Hrbac, J.; Zboril, R. The influence of complexing agent concentration on particle size in the process of sers active silver colloid synthesis. J. Mater. Chem. 2005, 15, 1099–1105. [Google Scholar] [CrossRef]
- The European Committee on Antimicrobial Susceptibility Testing. Breakpoint Tables for Interpretation of Mics and Zone Diameters. Version 5.0. Avalaible online: http://www.eucast.org/clinical_breakpoints/ (accessed on 1 January 2015).
- Krishnaraj, C.; Jagan, E.G.; Rajasekar, S.; Selvakumar, P.; Kalaichelvan, P.T.; Mohan, N. Synthesis of silver nanoparticles using acalypha indica leaf extracts and its antibacterial activity against water borne pathogens. Colloid. Surf. B 2010, 76, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Wang, P.; Sun, H.; Wang, Z. Superhydrophobic Surface with Hierarchical Architecture and Bimetallic Composition for Enhanced Antibacterial Activity. ACS Appl. Mater. Interfaces 2014, 6, 22108–22115. [Google Scholar] [CrossRef] [PubMed]
- Agnihotri, S.; Mukherji, S.; Mukherji, S. Size-controlled silver nanoparticles synthesized over the range 5–100 nm using the same protocol and their antibacterial efficacy. RSC Adv. 2014, 4, 3974–3983. [Google Scholar] [CrossRef]
- El-Zahry, M.R.; Mahmoud, A.; Refaat, I.H.; Mohamed, H.A.; Bohlmann, H.; Lendl, B. Antibacterial effect of various shapes of silver nanoparticles monitored by sers. Talanta 2015, 138, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.; Tak, Y.K.; Song, J.M. Does the antibacterial activity of silver nanoparticles depend on the shape of the nanoparticle? A study of the gram-negative bacterium escherichia coli. Appl. Environ. Microbiol. 2007, 73, 1712–1720. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Bharti, A.; Meena, V.K. Green synthesis of multi-shaped silver nanoparticles: Optical, morphological and antibacterial properties. J. Mater. Sci. Mater. Electron. 2015, 26, 3638–3648. [Google Scholar] [CrossRef]
- Habash, M.B.; Park, A.J.; Vis, E.C.; Harris, R.J.; Khursigara, C.M. Synergy of silver nanoparticles and aztreonam against pseudomonas aeruginosa pao1 biofilms. Antimicrob. Agents Chemother. 2014, 58, 5818–5830. [Google Scholar] [CrossRef] [PubMed]
- Kareem, P.A.; Alsammak, E.G. The Effect of Silver and Zinc Oxide Nanoparticles on Multi Drug Resistance Staphylococcus aureus. Int. J. Adv. Res. 2014, 12, 405–417. [Google Scholar]
- Kora, A.J.; Rastogi, L. Enhancement of antibacterial activity of capped silver nanoparticles in combination with antibiotics, on model gram-negative and gram-positive bacteria. Bioinorg. Chem. Appl. 2013, 2013, 871097. [Google Scholar] [CrossRef] [PubMed]
- Krajewski, S.; Prucek, R.; Panacek, A.; Avci-Adali, M.; Nolte, A.; Straub, A.; Zboril, R.; Wendel, H.P.; Kvitek, L. Hemocompatibility evaluation of different silver nanoparticle concentrations employing a modified chandler-loop in vitro assay on human blood. Acta Biomater. 2013, 9, 7460–7468. [Google Scholar] [CrossRef] [PubMed]
- Kvitek, L.; Vanickova, M.; Panacek, A.; Soukupova, J.; Dittrich, M.; Valentova, E.; Prucek, R.; Bancirova, M.; Milde, D.; Zboril, R. Initial study on the toxicity of silver nanoparticles (nps) against paramecium caudatum. J. Phys. Chem. C 2009, 113, 4296–4300. [Google Scholar] [CrossRef]
- Panacek, A.; Prucek, R.; Safarova, D.; Dittrich, M.; Richtrova, J.; Benickova, K.; Zboril, R.; Kvitek, L. Acute and chronic toxicity effects of silver nanoparticles (nps) on drosophila melanogaster. Environ. Sci. Technol. 2011, 45, 4974–4979. [Google Scholar] [CrossRef] [PubMed]
- Richter, A.P.; Brown, J.S.; Bharti, B.; Wang, A.; Gangwal, S.; Houck, K.; Hubal, E.A.C.; Paunov, V.N.; Stoyanov, S.D.; Velev, O.D. An environmentally benign antimicrobial nanoparticle based on a silver-infused lignin core. Nat. Nanotechnol. 2015, 10, 817–823. [Google Scholar] [CrossRef] [PubMed]
- Munger, M.A.; Radwanski, P.; Hadlock, G.C.; Stoddard, G.; Shaaban, A.; Falconer, J.; Grainger, D.W.; Deering-Rice, C.E. In vivo human time-exposure study of orally dosed commercial silver nanoparticles. Nanomed. Nanotechnol. 2014, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Kim, J.S.; Cho, H.S.; Rha, D.S.; Kim, J.M.; Park, J.D.; Choi, B.S.; Lim, R.; Chang, H.K.; Chung, Y.H.; et al. Twenty-eight-day oral toxicity, genotoxicity, and gender-related tissue distribution of silver nanoparticles in sprague-dawley rats. Inhal. Toxicol. 2008, 20, 575–583. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Song, M.Y.; Park, J.D.; Song, K.S.; Ryu, H.R.; Chung, Y.H.; Chang, H.K.; Lee, J.H.; Oh, K.H.; Kelman, B.J.; et al. Subchronic oral toxicity of silver nanoparticles. Part. Fibre Toxicol. 2010, 7. [Google Scholar] [CrossRef] [PubMed]
- Loeschner, K.; Hadrup, N.; Qvortrup, K.; Larsen, A.; Gao, X.Y.; Vogel, U.; Mortensen, A.; Lam, H.R.; Larsen, E.H. Distribution of silver in rats following 28 days of repeated oral exposure to silver nanoparticles or silver acetate. Part. Fibre Toxicol. 2011, 8. [Google Scholar] [CrossRef] [PubMed]
- Van der Zande, M.; Vandebriel, R.J.; van Doren, E.; Kramer, E.; Rivera, Z.H.; Serrano-Rojero, C.S.; Gremmer, E.R.; Mast, J.; Peters, R.J.B.; Hollman, P.C.H.; et al. Distribution, elimination, and toxicity of silver nanoparticles and silver ions in rats after 28-day oral exposure. ACS Nano 2012, 6, 7427–7442. [Google Scholar] [CrossRef] [PubMed]
- Hadrup, N.; Loeschner, K.; Mortensen, A.; Sharma, A.K.; Qvortrup, K.; Larsen, E.H.; Lam, H.R. The similar neurotoxic effects of nanoparticulate and ionic silver in vivo and in vitro. Neurotoxicology 2012, 33, 416–423. [Google Scholar] [CrossRef] [PubMed]
- Cha, K.; Hong, H.W.; Choi, Y.G.; Lee, M.J.; Park, J.H.; Chae, H.K.; Ryu, G.; Myung, H. Comparison of acute responses of mice livers to short-term exposure to nano-sized or micro-sized silver particles. Biotechnol. Lett. 2008, 30, 1893–1899. [Google Scholar] [CrossRef] [PubMed]
- Park, E.J.; Bae, E.; Yi, J.; Kim, Y.; Choi, K.; Lee, S.H.; Yoon, J.; Lee, B.C.; Park, K. Repeated-dose toxicity and inflammatory responses in mice by oral administration of silver nanoparticles. Environ. Toxicol. Pharmacol. 2010, 30, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Korani, M.; Rezayat, S.M.; Gilani, K.; Bidgoli, S.A.; Adeli, S. Acute and subchronic dermal toxicity of nanosilver in guinea pig. Int. J. Nanomed. 2011, 6, 855–862. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.L.; Xiong, L.; Wang, S.; Wang, J.Y.; Liu, L.; Li, J.A.; Wan, Z.Y.; Xi, T.F. Influence of silver nanoparticles on neurons and blood-brain barrier via subcutaneous injection in rats. Appl. Surf. Sci. 2008, 255, 502–504. [Google Scholar] [CrossRef]
- Tang, J.L.; Xiong, L.; Wang, S.; Wang, J.Y.; Liu, L.; Li, J.G.; Yuan, F.Q.; Xi, T.F. Distribution, translocation and accumulation of silver nanoparticles in rats. J. Nanosci. Nanotechnol. 2009, 9, 4924–4932. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Panáček, A.; Smékalová, M.; Kilianová, M.; Prucek, R.; Bogdanová, K.; Večeřová, R.; Kolář, M.; Havrdová, M.; Płaza, G.A.; Chojniak, J.; et al. Strong and Nonspecific Synergistic Antibacterial Efficiency of Antibiotics Combined with Silver Nanoparticles at Very Low Concentrations Showing No Cytotoxic Effect. Molecules 2016, 21, 26. https://doi.org/10.3390/molecules21010026
Panáček A, Smékalová M, Kilianová M, Prucek R, Bogdanová K, Večeřová R, Kolář M, Havrdová M, Płaza GA, Chojniak J, et al. Strong and Nonspecific Synergistic Antibacterial Efficiency of Antibiotics Combined with Silver Nanoparticles at Very Low Concentrations Showing No Cytotoxic Effect. Molecules. 2016; 21(1):26. https://doi.org/10.3390/molecules21010026
Chicago/Turabian StylePanáček, Aleš, Monika Smékalová, Martina Kilianová, Robert Prucek, Kateřina Bogdanová, Renata Večeřová, Milan Kolář, Markéta Havrdová, Grażyna Anna Płaza, Joanna Chojniak, and et al. 2016. "Strong and Nonspecific Synergistic Antibacterial Efficiency of Antibiotics Combined with Silver Nanoparticles at Very Low Concentrations Showing No Cytotoxic Effect" Molecules 21, no. 1: 26. https://doi.org/10.3390/molecules21010026
APA StylePanáček, A., Smékalová, M., Kilianová, M., Prucek, R., Bogdanová, K., Večeřová, R., Kolář, M., Havrdová, M., Płaza, G. A., Chojniak, J., Zbořil, R., & Kvítek, L. (2016). Strong and Nonspecific Synergistic Antibacterial Efficiency of Antibiotics Combined with Silver Nanoparticles at Very Low Concentrations Showing No Cytotoxic Effect. Molecules, 21(1), 26. https://doi.org/10.3390/molecules21010026