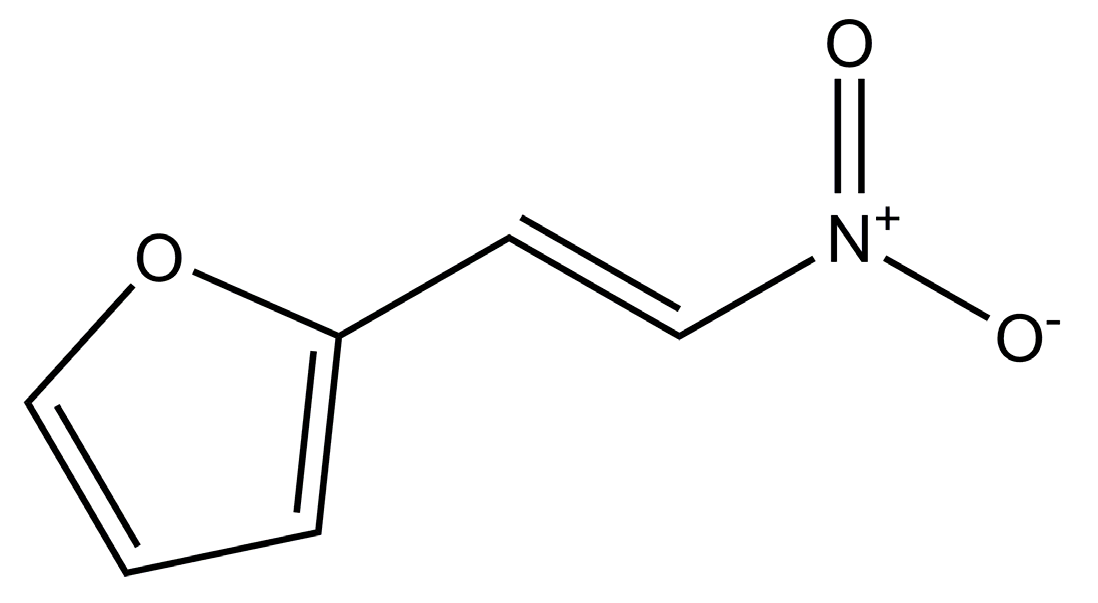
Characterization of the Sublimation and Vapor Pressure of 2-(2-Nitrovinyl) Furan (G-0) Using Thermogravimetric Analysis: Effects of Complexation with Cyclodextrins
Abstract
:1. Introduction
2. Results and Discussion
2.1. Determination of the Purity of G-0 Using DSC
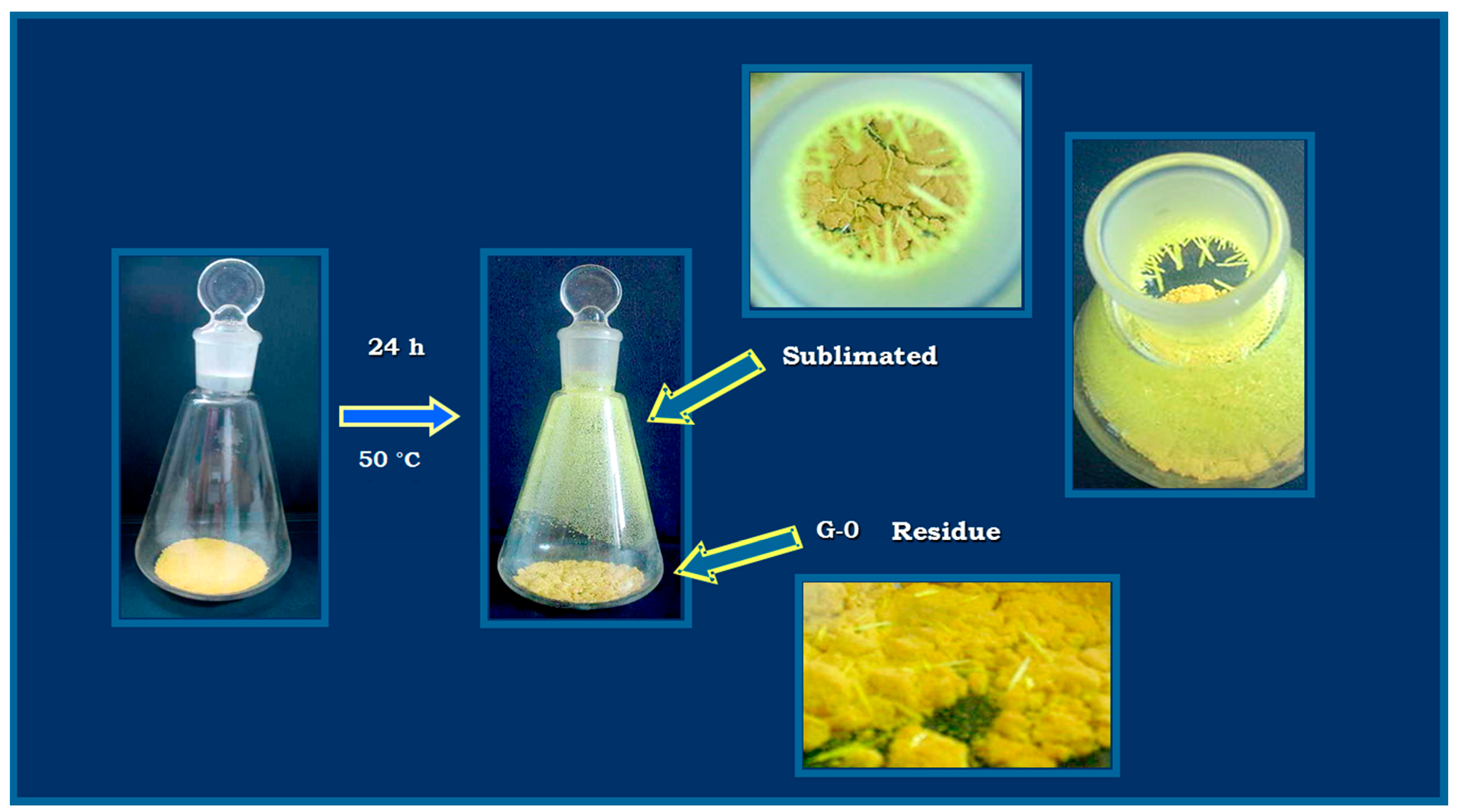
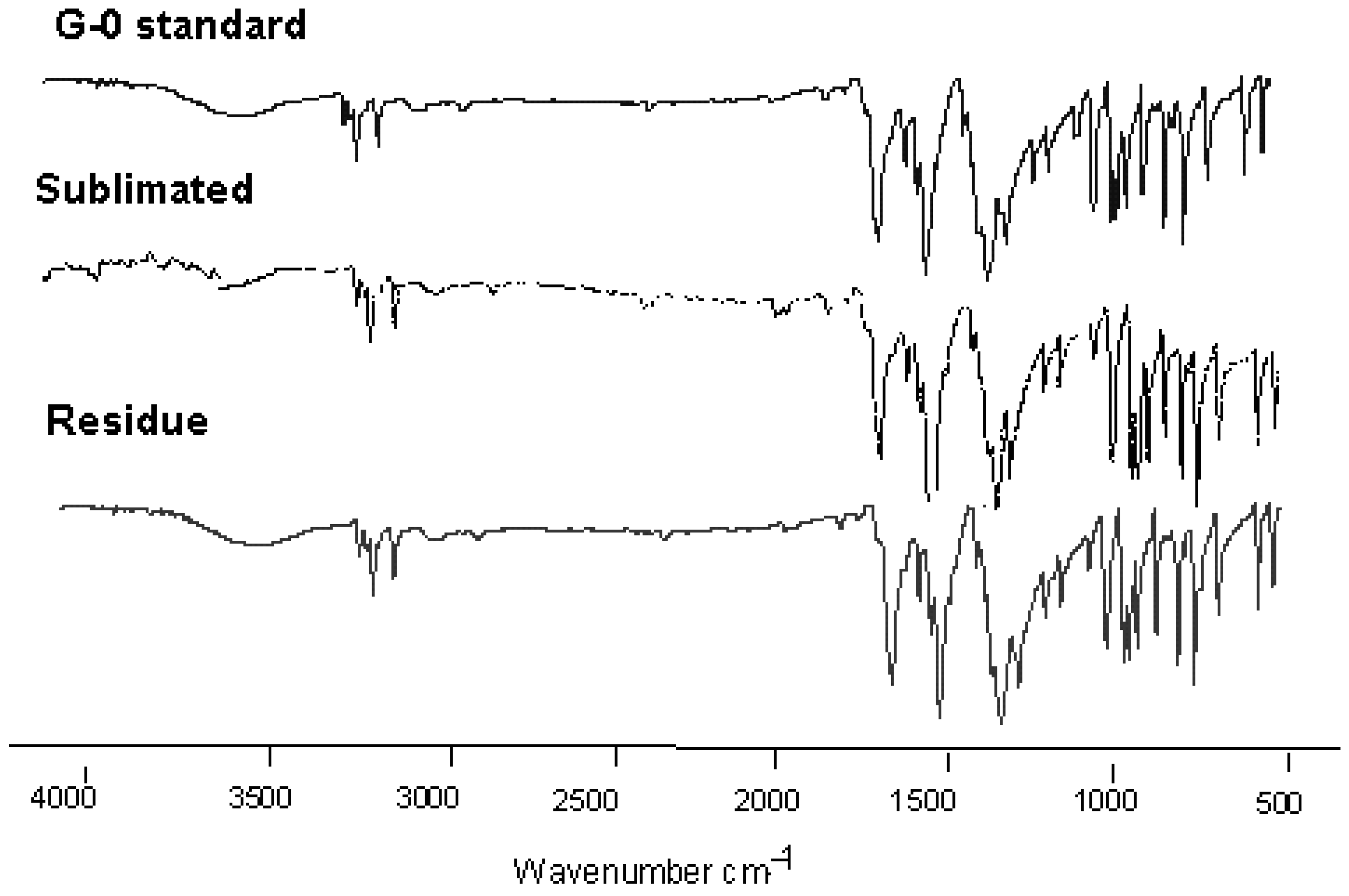
2.2. Characterization of the Sublimated Product
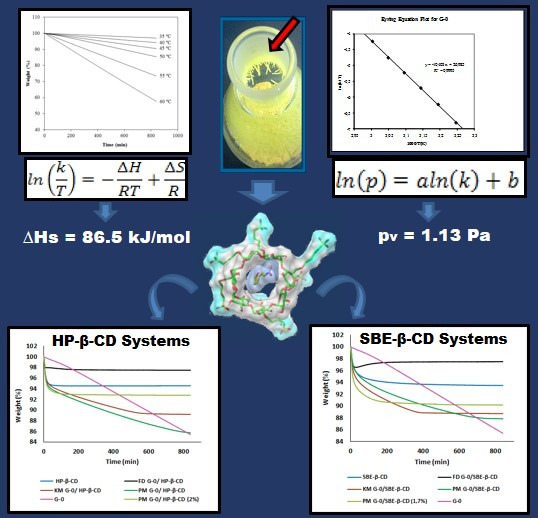
2.3. Sublimation Enthalpy and Vapor Pressure
2.3.1. Sublimation Enthalpy

| Temperature (°C) | Linear Regression Equation | Determination Coefficient (R2) |
|---|---|---|
| 35 | y = −0.0036x + 100.04 | 0.9998 |
| 40 | y = −0.0069x + 99.99 | 1.0000 |
| 45 | y = −0.0114x + 100.06 | 1.0000 |
| 50 | y = −0.0178x + 100.45 | 0.9991 |
| 55 | y = −0.0318x + 100.03 | 1.0000 |
| 60 | y = −0.0507x + 99.91 | 0.9999 |
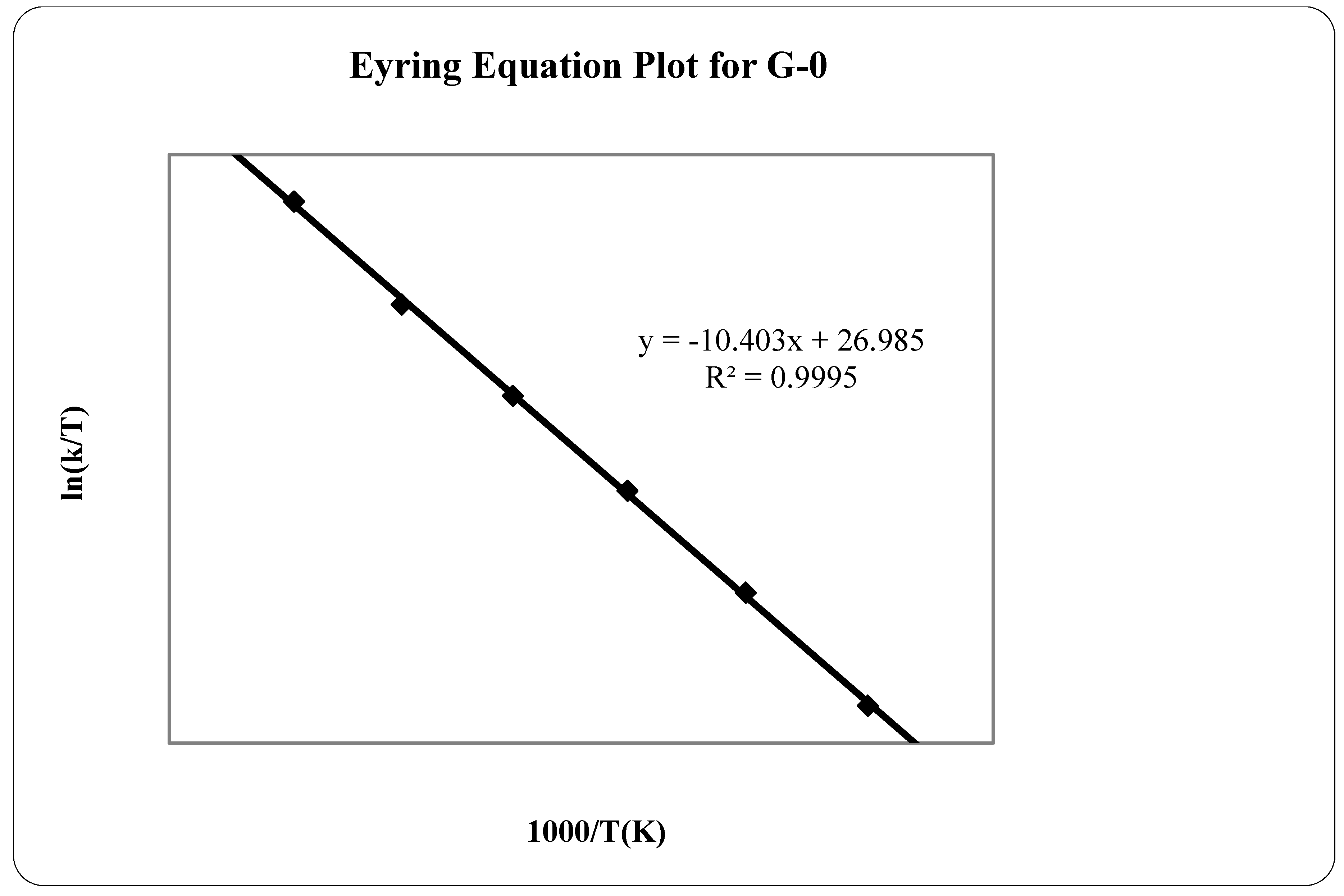
2.3.2. Vapor Pressure Estimation
| Temperature (°C) | Sublimation Rate (µg/min) | Vapor Pressure (Pa) |
|---|---|---|
| 40 | 0.0796 | 0.910 |
| 45 | 0.1507 | 1.546 |
| 50 | 0.2909 | 2.582 |
| 55 | 0.4649 | 4.245 |
| 65 | 1.2250 | 10.985 |
| 70 | 2.0981 | 17.306 |
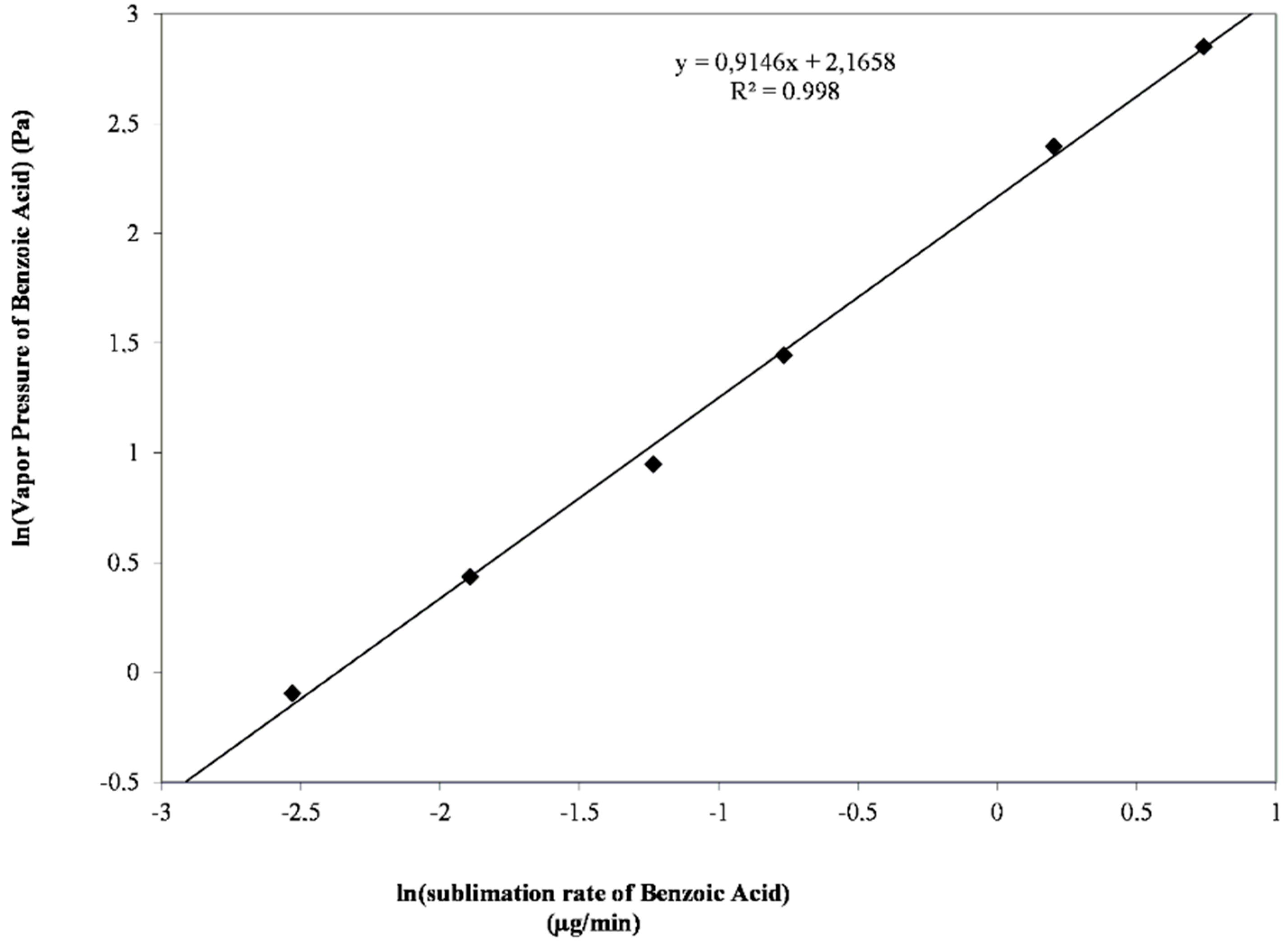
| Temperature (°C) | Sublimation Rate (µg/min) | Calculated Vapor Pressure (Pa) |
|---|---|---|
| 35 | 0.3401 | 3.252 |
| 40 | 0.6146 | 5.588 |
| 45 | 1.0503 | 9.122 |
| 50 | 1.7310 | 14.405 |
| 55 | 2.7983 | 22.352 |
| 60 | 4.8075 | 36.667 |
2.4. Evaluation of the Influence of Complexation with Cyclodextrins on the Sublimation Process
| Sample | Water Content (%) a |
|---|---|
| SBE-β-CD | 6.38 ± 0.87 |
| HP-β-CD | 6.40 ± 0.02 |
| FD G-0/SBE-β-CD | 8.33 ± 0.36 |
| FD G-0/HP-β-CD | 5.81 ± 0.27 |
2.4.1. HP-β-CD Inclusion Complexes
| System | Total Weight Loss (%) | Weight Loss 1 (%) | Weight Loss 2 (%) | Slope 1 (%/min) | Slope 2 (%/min) |
|---|---|---|---|---|---|
| HP-β-CD | 5.77 ± 0.46 | 5.35 ± 0.35 a | 0.42 ± 0.06 b | −0.20 ± 0.03 a | −0.14 × 10−2 ± 0.18 × 10−3 b |
| PM G-0/HP-β-CD (2%) | 7.47 ± 0.35 | 6.22 ± 0.28 a | 1.19 ± 0.15 b | −0.20 ± 0.66 × 10−2 a | −0.55 × 10−2 ± 1.01 × 10−3 b |
| PM G-0/HP-β-CD (8.8%) | 13.88 ± 0.36 | 4.86 ± 0.28 a | 9.01 ± 0.08 e | −0.29 a | −1.1 × 10−2 ± 0.12 × 10−3 e |
| KM G-0/HP-β-CD | 10.88 ± 0.21 | 5.65 ± 0.67 a | 5.03 ± 0.36 c | −0.19 a | −0.95 × 10−2 ± 0.56 × 10−3 c |
| FD G-0/HP-β-CD | 4.36 ± 1.58 | 3.85 ± 1.55 a | 0.41 ± 0.04 b | −0.55 a | −0.23 × 10−2 ± 0.48 × 10−4 b |
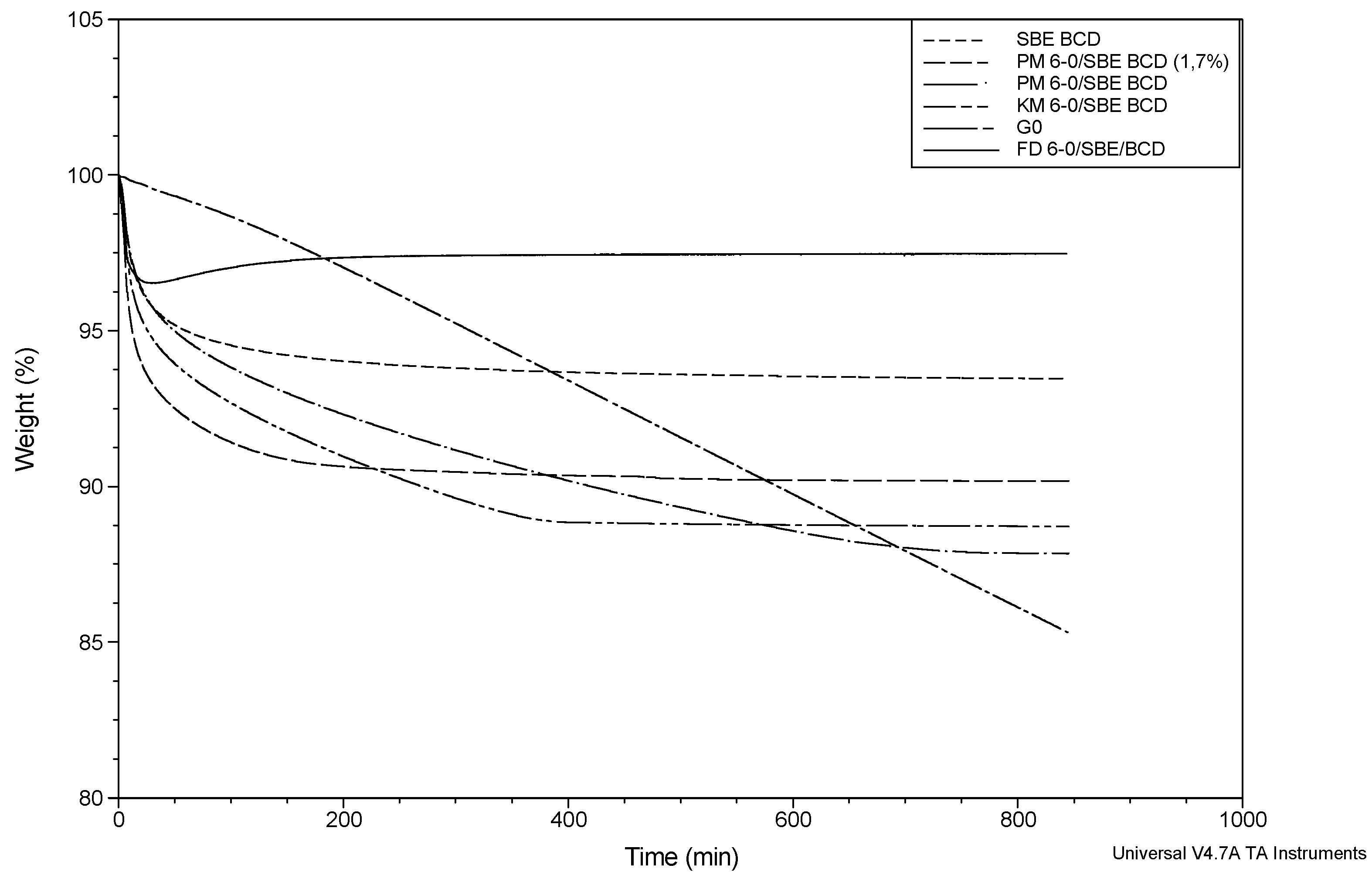
| SBE-β-CD | 6.92 ± 0.55 | 4.61 ± 0.54 a | 2.09 ± 0.02 b | −0.22 ± 0.28 × 10−1 a | −0.70 × 10−2 ± 0.30 × 10−4 b |
| PM G-0/SBE-β-CD (1.7%) | 9.67 ± 0.22 | 6.60 ± 0.14 a | 2.89 ± 0.08 b | −0.21 ± 0.21 × 10−3 a | −1.37 × 10−2 ± 0.54 × 10−3 b |
| PM G-0/SBE-β-CD (7%) | 12.18 ± 0.06 | 5.16 ± 0.20 a | 6.90 ± 0.08 d | −0.17 ± 0.41 × 10−1 a | −1.61 × 10−2 ± 0.37 × 10−2 d |
| KM G-0/SBE-β-CD | 11.85 ± 0.80 | 6.88 ± 1.17 a | 4.83 ± 0.36 c | −0.26 ± 0.98 × 10−1 a | −1.54 × 10−2 ± 0.12 × 10−2 c |
| FD G-0/SBE-β-CD | 5.84 ± 2.40 | 5.46 ± 2.87 a | 0.33 ± 0.13 b | −0.28 ± 0.12 a | −0.13 × 10−2 ± 0.02 × 10−2 b |
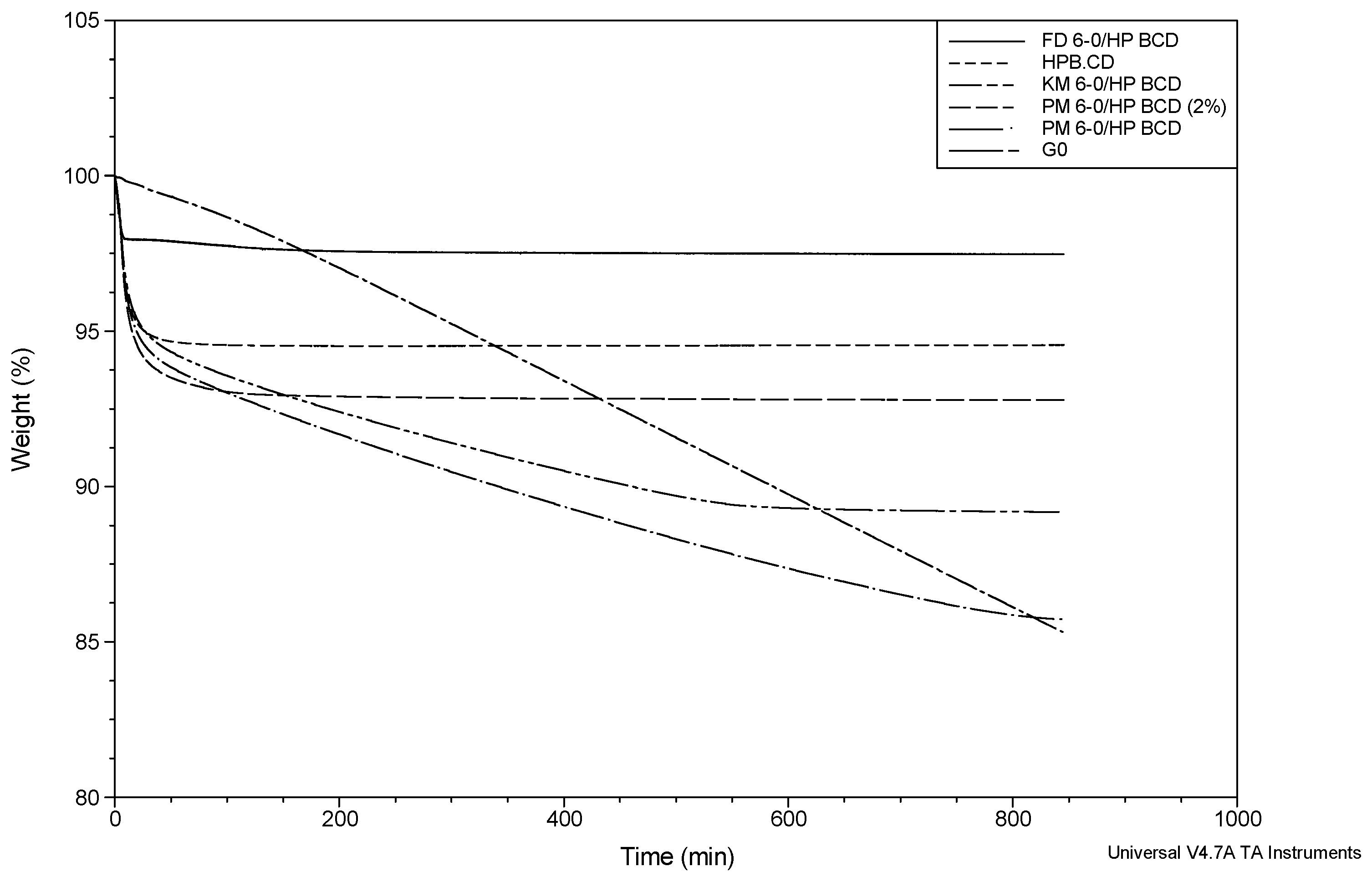
2.4.2. SBE-β-CD Inclusion Complexes
3. Experimental Section
3.1. Materials and Reagents
3.2. Determination of the Purity of G-0 Using DSC
3.3. Characterization of the Sublimated Product
3.3.1. Fourier-Transformed-Infrared Spectroscopy
3.3.2. Thin Layer Chromatography
3.4. Determination of Sublimation Enthalpy and Vapor Pressure of G-0
3.5. Evaluation of the Influence of Complexation with Cyclodextrins on the Sublimation Process
3.5.1. Karl Fischer Titration for Water Content Determination of the Cyclodextrin Complexes and Pure Cyclodextrins
3.5.2. Isothermal TGA of the Cyclodextrin Complexes
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Olazábal, E.; Castañedo, N.; Gaitán, T.; Rodríguez, Z.; González, O.; Serrano, H.; Díaz, M.I.; Calvo, A.M. Method of Obtaining 2-(2-nitrovinyl) Furan and the Use Thereof as a Coccidiostatic. U.S. Patent Application Number 20050043551, 24 September 2003. [Google Scholar]
- Castañedo, N.; Sinfontes, R.S.; Monzote, F.L.; López, H.Y.; Montalvo, A.M.; Infante, B.J.; Olazábal, E. Pharmaceutical Compositions Containing Nitrovinylfuran Derivatives for the Treatment of Leishmaniasis and Trypanosomasis. U.S. Patent Application Number 2009042978, 18 September 2006. [Google Scholar]
- Alabi, K.A.; Hassan, G.F. Determination of minimum inhibitory concentrations of 2-(2-nitrovinyl) furan. Afr. Microbiol. Res. 2014, 2, 028–032. [Google Scholar]
- Borroto, J.I.; Creus, A.; Marcos, R. Genotoxic evaluation of the furylethylene derivative 2-furyl-1-nitroethene in cultured human lymphocytes. Mutat. Res. Gen. Toxicol. Environ. 2001, 497, 177–184. [Google Scholar] [CrossRef]
- Borroto, J.I.; Pérez, G.; Creus, A.; Marcos, R. Comparative genotoxic evaluation of 2-furylethylenes and 5-nitrofurans by using the comet assay in TK6 cells. Mutagenesis 2005, 20, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Borroto, J.I.; Creus, A.; Marcos, R.; Molla, R.; Zapatero, J. The Mutagenic Potential of the Furylethylene Derivative 2-Furyl-1-nitroethene in the Mouse Bone Marrow Micronucleus Test. Toxicol. Sci. 2003, 72, 359–362. [Google Scholar] [CrossRef] [PubMed]
- Verevkin, S.P. Termochemistry of nitro compounds. Experimental standard enthalpies of formation and improved group-additivity values. Thermochim. Acta 1997, 307, 17–25. [Google Scholar] [CrossRef]
- Knudsen, M. Die Molekularströmung der Gase durch Offnungen und die Effusion. Ann. Phys. 1909, 333, 999–1016. [Google Scholar] [CrossRef]
- Wiedemann, H.G. Applications of Thermogravimetry for Vapor Pressure Determination. Thermochim. Acta 1972, 3, 355–366. [Google Scholar] [CrossRef]
- Goodrum, J.M.; Siesel, E.M. Thermogravimetric analysis for boiling points and vapor pressure. J. Therm. Anal. 1996, 46, 1251–1258. [Google Scholar] [CrossRef]
- Emmenger, F.; Piccand, M. Vapour Pressure Measurements with a Thermobalance. J. Therm. Anal. Calorim. 1999, 57, 235–240. [Google Scholar] [CrossRef]
- Gückel, W.; Kästel, R.; Kröhl, T.; Parg, A. Methods for determining the vapor pressure of active ingredients used in crop protection. Part IV. An improved thermogravimetric determination based on evaporation rate. Pestic. Sci. 1995, 45, 27–31. [Google Scholar] [CrossRef]
- Elder, J.P. Sublimation measurements of pharmaceutical compounds by isothermal thermogravimetry. J. Therm. Anal. 1997, 49, 897–905. [Google Scholar] [CrossRef]
- Xie, M.; Ziemba, T.M.; Maurin, M.B. Sublimation Characterization and Vapor Pressure Estimation of an HIV Nonnucleoside Reverse Transcriptase Inhibitor Using Thermogravimetric Analysis. AAPS PharmSciTech 2003, 4, 2. [Google Scholar] [CrossRef] [PubMed]
- Price, D.M.; Bashir, S.; Derrick, P.R. Sublimation properties of x, y-dihydroxybenzoic acid isomers as model matrix assisted laser desorption ionisation (MALDI) matrices. Thermochim. Acta 1999, 327, 167–171. [Google Scholar] [CrossRef]
- Pankajavalli, P.; Ananthasivan, K.; Anthonysamy, S.; Vasudeva, P.R. Vapour pressure and standard enthalpy of sublimation of KBF4 by a TG based transpiration technique. J. Nucl. Mater. 2005, 345, 96–100. [Google Scholar] [CrossRef]
- Guillory, J.K.; Poust, R.I. Chemical Kinetics and Drug Stability. In Modern Pharmaceutics, 4th ed.; Banker, G.S., Rhodes, C.T., Eds.; Marcel Dekker, Inc.: New York, NY, USA, 2002; pp. 154–155. [Google Scholar]
- Pikal, M.J.; Lukes, A.L.; Ellis, L.F. Vapor pressure of nitroglycerin in sublingual molded tablets: Implications for stability. J. Pharm. Sci. 2006, 65, 1278–1284. [Google Scholar] [CrossRef]
- Centkowska, K.; Sznitowska, M. Comparison of sublingual tablets with nitroglycerin complexed with β-cyclodextrin or titrated with crosspovidone—Technological approach. Acta Pol. Pharm. Drug Res. 2008, 65, 585–589. [Google Scholar]
- Buschmann, H.J.; Schollmeyer, E. Applications of cyclodextrins in cosmetic products: A review. J. Cosmet. Sci. 2002, 53, 185–191. [Google Scholar]
- Bergamasco, R.C.; Zanin, G.M.; de Moraes, F.F. Sulfluramid volatility reduction by beta-cyclodextrin. J. Agric. Food Chem. 2005, 53, 1139–1143. [Google Scholar]
- Szente, L.; Szejtli, J. Cyclodextrin complex of a volatile insecticide (DDVP). Acta Chim. Acad. Sci. Hung. 1981, 107, 195–202. [Google Scholar]
- Szente, L. Stable, Controlled—Release Organophosphorous Pesticides Entrapped in β-Cyclodextrin. J. Therm. Anal. 1998, 51, 957–963. [Google Scholar]
- Fourmentin, S.; Landy, D.; Leyssens, G.; Surpateanu, G. Reduction of the volatility of volatile organic compounds with cyclodextrins: Application to their capture. Ann. Pharm. Fr. 2001, 59, 369–373. [Google Scholar]
- Food and Drug Administration. USFDA Environmental Assessment Technical Guide; National Press Office: Rockville, MD, USA, 1987; No. 10.03. [Google Scholar]
- Ruz, V.; Froeyen, M.; Busson, R.; González, M.M.; Baudemprez, L.; van den Mooter, G. Characterization and molecular modeling of the inclusion complexes of 2-(2-nitrovinyl) furan (G-0) with cyclodextrins. Int. J. Pharm. 2012, 439, 275–285. [Google Scholar]
- De Rojas, M. Estudio de Los Productos de Degradación del 2-(2-nitrovinil) Furano (G-0). Ph.D. Thesis, Universidad de Cádiz, Cádiz, España, 2006. [Google Scholar]
- Wright, S.F.; Phang, P.; Dollimore, D.; Alexander, K.S. An overview of calibration materials used in thermal analysis-benzoic acid. Termochim. Acta 2002, 392, 251–257. [Google Scholar]
- Gao, G.Y.; Lin, S.Y. Thermodynamic investigations of Nitroxoline sublimation by simultaneous DSC-FTIR method and Isothermal TG Analysis. J. Pharm. Sci. 2010, 99, 255–261. [Google Scholar]
- Zhu, G.; Xiao, Z.; Zhou, R.; Zhu, Y. Study of production and pyrolysis characteristics of sweet orange flavor-β-cyclodextrin inclusion complex. Carbohydr. Polym. 2014, 105, 75–80. [Google Scholar]
- Zhu, G.; Xiao, Z.; Zhou, R.; Feng, N. Production of a transparent lavender flavour nanocapsule aqueous solution and pyrolisis characteristics of flavour nanocapsule. J. Food Sci. Technol. 2014. [Google Scholar] [CrossRef]
- He, Z.; Wang, Z.; Zhang, H.; Pan, X.; Su, W.; Liang, D.; Wu, C. Doxycycline and hydroxypropyl-β-cyclodextrin complex in poloxamer thermal sensitive hydrogel for ophthalmic delivery. Acta Pharm. Sin. B 2011, 4, 254–260. [Google Scholar] [CrossRef]
- Whang, H.S.; Tonelli, A. Release characteristics of the non-toxic insect repellant 2-undecanone from its crystalline inclusion compound with α-cyclodextrin. J. Incl. Phenom. Macrocycl. Chem. 2008, 62, 127–134. [Google Scholar] [CrossRef]
- Zhu, G.; Feng, N.; Zuobing, X.; Zhou, R.; Niu, Y. Production and pirolisis characteristics of citral-monochlorotriazinyl-β-cyclodextrin inclusion complex. J Therm. Anal. Calorim. 2015. [Google Scholar] [CrossRef]
- Rodríguez, M.; Valdés, T.; Bravo, L. Analytical Validation of the Methods for Purity Determination for the Bioactives G-1, G-0 and UC-244; Centro de Bioactivos Químicos. Universidad Central “Marta Abreu”: De Las Villas, Cuba, 2015; Unpublished Work. [Google Scholar]
- Sample Availability: Samples of the compounds used in this study were available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruz, V.; González, M.M.; Winant, D.; Rodríguez, Z.; Van den Mooter, G. Characterization of the Sublimation and Vapor Pressure of 2-(2-Nitrovinyl) Furan (G-0) Using Thermogravimetric Analysis: Effects of Complexation with Cyclodextrins. Molecules 2015, 20, 15175-15191. https://doi.org/10.3390/molecules200815175
Ruz V, González MM, Winant D, Rodríguez Z, Van den Mooter G. Characterization of the Sublimation and Vapor Pressure of 2-(2-Nitrovinyl) Furan (G-0) Using Thermogravimetric Analysis: Effects of Complexation with Cyclodextrins. Molecules. 2015; 20(8):15175-15191. https://doi.org/10.3390/molecules200815175
Chicago/Turabian StyleRuz, Vivian, Mirtha Mayra González, Danny Winant, Zenaida Rodríguez, and Guy Van den Mooter. 2015. "Characterization of the Sublimation and Vapor Pressure of 2-(2-Nitrovinyl) Furan (G-0) Using Thermogravimetric Analysis: Effects of Complexation with Cyclodextrins" Molecules 20, no. 8: 15175-15191. https://doi.org/10.3390/molecules200815175
APA StyleRuz, V., González, M. M., Winant, D., Rodríguez, Z., & Van den Mooter, G. (2015). Characterization of the Sublimation and Vapor Pressure of 2-(2-Nitrovinyl) Furan (G-0) Using Thermogravimetric Analysis: Effects of Complexation with Cyclodextrins. Molecules, 20(8), 15175-15191. https://doi.org/10.3390/molecules200815175