Quercitrin, an Inhibitor of Sortase A, Interferes with the Adhesion of Staphylococcal aureus
Abstract
:1. Introduction
2. Results and Discussion
2.1. QEN Blocks the Thioesterification Process of Sortase A Catalysis

| Compounds | ||
|---|---|---|
| Quercitrin | Vitexin | Isoliquiritin |
| Liquiritigenin | Tetrandrine | Honokiol |
| Cyrtopterinetin | Fisetin | Eleutheroside A |
| Chrysin | Daidzin | Quinine |
| Formononetin | Emodin | Sodium houttufonate |
| Tectorigenin | Psoralen | Dryocrassin |
| Viola yedoensis extract | Andrographolide | Wogonoside |
| Prunella vulgaris leaf extract | Artemisinin | Radix platycodi extract |
| Forsythin | Kaempferol | Berberine |
2.2. QEN Inhibits Adherence of S. aureus to Cell-Matrix Proteins
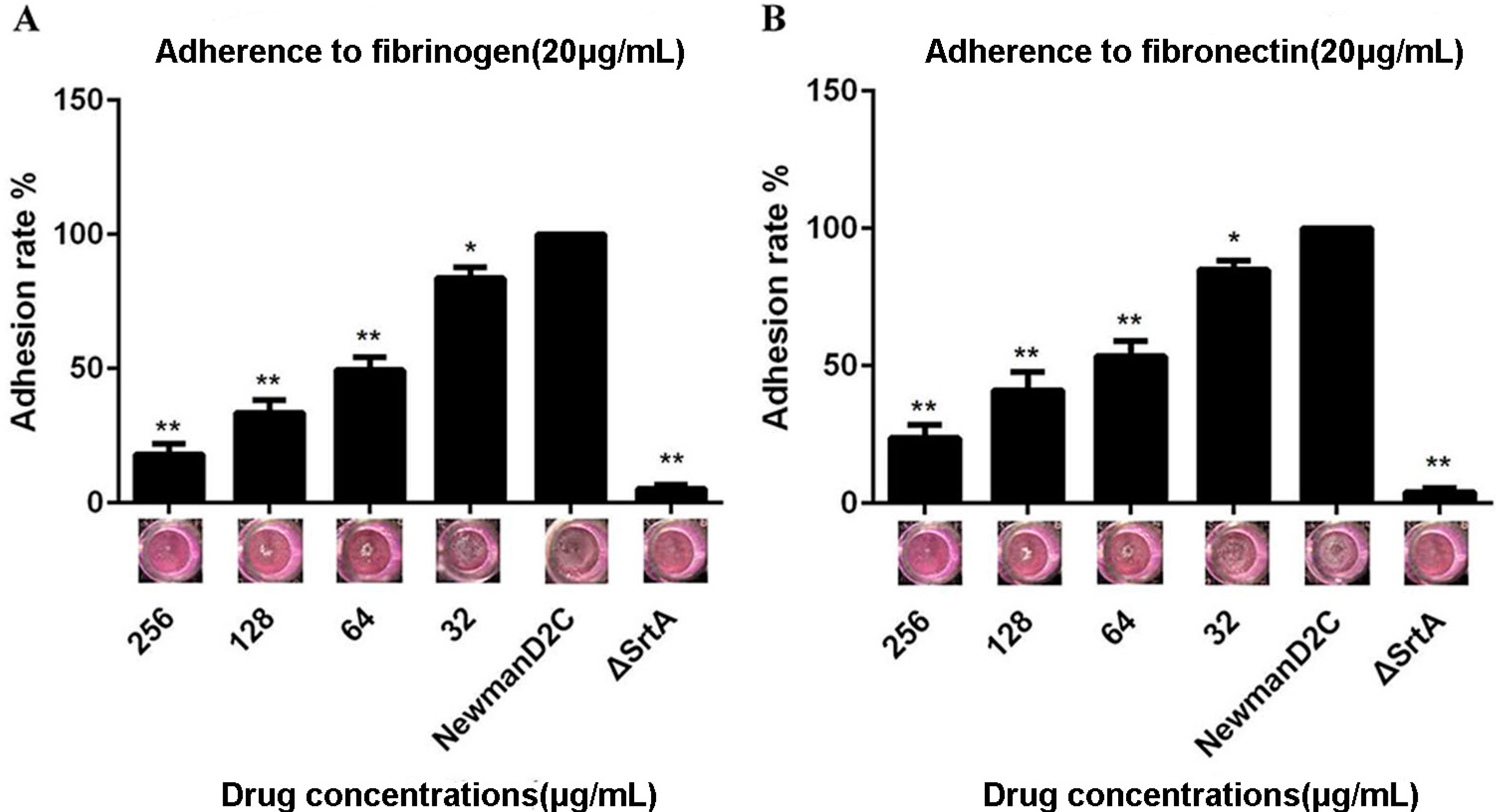
2.3. Determination of the Binding Mode of SrtA with QEN
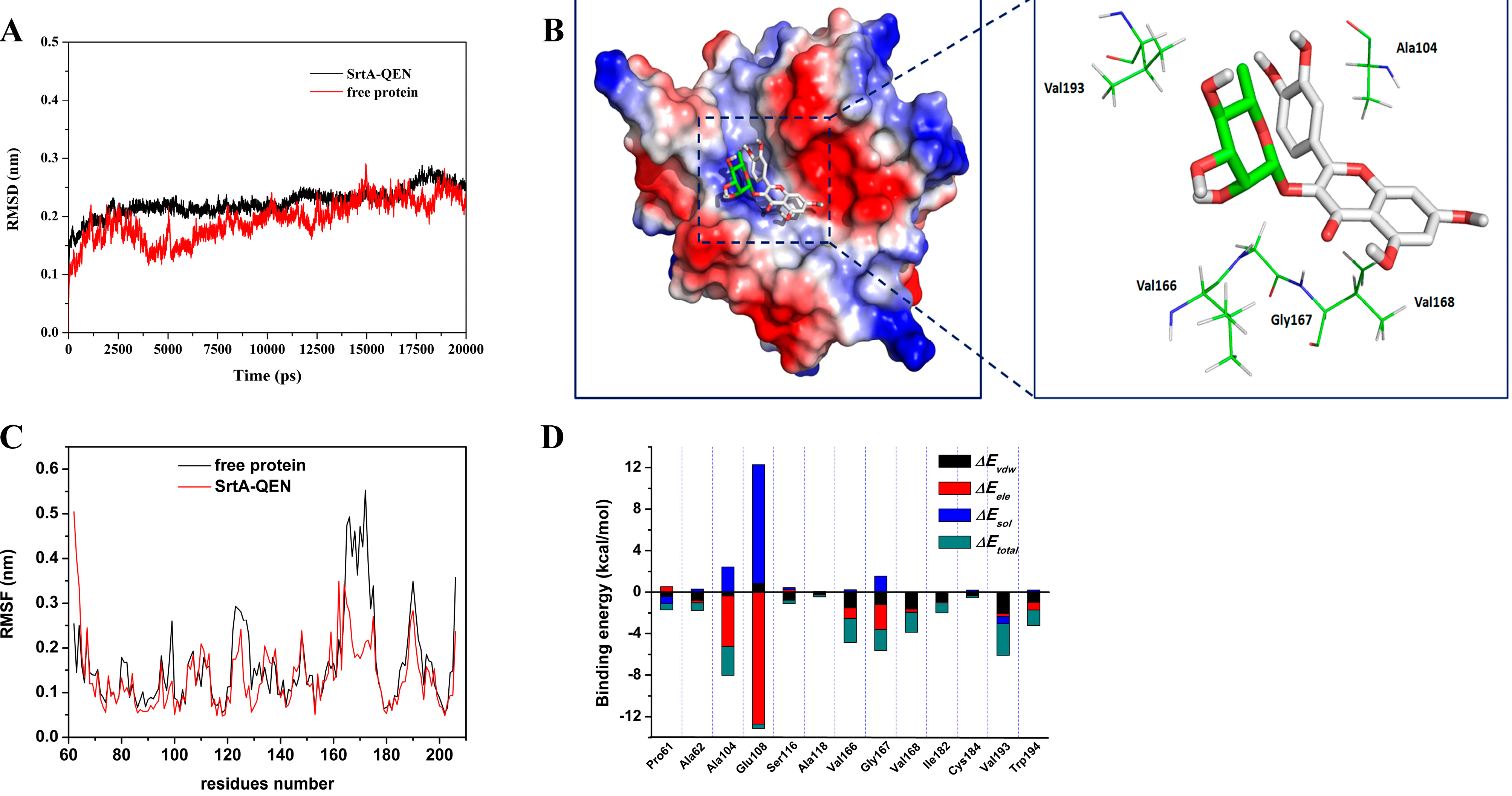
2.4. Identification of the Binding Site in the SrtA-QEN Complex
| TΔS (kcal/mol) | ΔGbind (kcal/mol) | Binding Constants KA (1 × 105) L·mol−1 | n | |
|---|---|---|---|---|
| WT-QEN | 5.9 ± 1.8 | −11.7 ± 1.4 | 6.9 ± 1.3 | 0.9991 |
| G167A-QEN | 6.2 ± 1.6 | −5.6 ± 1.9 | 3.7 ± 0.7 | 1.0044 |
| V193A-QEN | 6.1 ± 1.5 | −7.4 ± 1.3 | 4.2 ± 1.1 | 0.9996 |
2.5. Discussion
3. Experimental Section
3.1. Bacterial Strains, Plasmids and Reagents
| Strain or Plasmid | Relevant Genotype | Source or Reference |
|---|---|---|
| Strains | ||
| S. aureus | ||
| Newman D2C | Wild-type SrtA positive; nonhemolytic; coagulase Negative | ATCC25904 |
| ΔSrtA | srtA::Emr; isogenic mutant of Newman D2C | |
| E. coli | ||
| BL21 | Expression strain, F− ompT hsdS(rB− mB−) gal dcm (DE3) | Invitrogen |
| Plasmids | ||
| pGEX-6P-1 | Expression vector | Amersham |
| pGSrtAΔN59 | pGEX-6P-1 with srtA gene | This study |
| G167A | pGSrtAΔN59 derivative, for the substitution of Gly167 with alanine | This study |
| V193A | pGSrtAΔN59 derivative, for the substitution of Val192 with alanine | This study |
3.2. Preparation of Recombinant SrtAΔN59 and Its Mutant
| Primer Name | Oligonucleotide (5–3) ᵅ |
|---|---|
| PsrtA59F | GCGGGATCCCCGGAATTCCAAGCTAAACCTCAAATTCC |
| PsrtA59R | CCGCTCGAGTTATTTGACTTCTGTAGCTACAA |
| G167A–F | AGCCAACAGATGTAGCAGTTCTAGAT |
| G167A–R | AGAACGCTACATCTGTTGGCTTAACATCTC |
| V193A–F | TGAAAAGACAGGCGCTTGGGAAAAAC |
| V193A–R | TTCCCAGCGCCTGTCTTTTCATTGTAATCAT |
3.3. Sortase Activity Inhibition Assay
3.4. Determination of Minimum Inhibitory Concentration (MIC) and Growth Curves
3.5. Fibrinogen-Binding and Fibronectin-Binding Assays
3.6. Computational Chemistry
3.7. Binding Affinity Determination of QEN with WT-SrtAΔN59, G167A and V193A
3.8. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wertheim, H.F.; Melles, D.C.; Vos, M.C.; van Leeuwen, W.; van Belkum, A.; Verbrugh, H.A.; Nouwen, J.L. The role of nasal carriage in Staphylococcus aureus infections. Lancet Infect. Dis. 2005, 5, 751–762. [Google Scholar] [CrossRef] [PubMed]
- Lowy, F.D. Staphylococcus aureus infections. N. Engl. J. Med. 1998, 339, 520–532. [Google Scholar] [CrossRef] [PubMed]
- Oh, K.B.; Oh, M.N.; Kim, J.G.; Shin, D.S.; Shin, J. Inhibition of sortase-mediated Staphylococcus aureus adhesion to fibronectin via fibronectin-binding protein by sortase inhibitors. Appl. Microbiol. Biotechnol. 2006, 70, 102–106. [Google Scholar] [CrossRef] [PubMed]
- Ilangovan, U.; Ton-That, H.; Iwahara, J.; Schneewind, O.; Clubb, R.T. Structure of sortase, the transpeptidase that anchors proteins to the cell wall of Staphylococcus aureus. Proc. Natl. Acad. Sci. USA 2001, 98, 6056–6061. [Google Scholar] [CrossRef] [PubMed]
- Cossart, P.; Jonquieres, R. Sortase, a universal target for therapeutic agents against gram-positive bacteria? Proc. Natl. Acad. Sci. USA 2000, 97, 5013–5015. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, I.M.; Mazmanian, S.K.; Schneewind, O.; Verdrengh, M.; Bremell, T.; Tarkowski, A. On the role of Staphylococcus aureus sortase and sortase-catalyzed surface protein anchoring in murine septic arthritis. J. Infect. Dis. 2002, 185, 1417–1424. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.G.; Kim, H.K.; Burts, M.L.; Krausz, T.; Schneewind, O.; Missiakas, D.M. Genetic requirements for Staphylococcus aureus abscess formation and persistence in host tissues. FASEB J. 2009, 23, 3393–3404. [Google Scholar] [CrossRef] [PubMed]
- McAdow, M.; Kim, H.K.; Dedent, A.C.; Hendrickx, A.P.; Schneewind, O.; Missiakas, D.M. Preventing Staphylococcus aureus sepsis through the inhibition of its agglutination in blood. PLoS Pathogens 2011, 7, e1002307. [Google Scholar] [CrossRef] [PubMed]
- Bolken, T.C.; Franke, C.A.; Zeller, G.O.; Hruby, D.E. Identification of an intragenic integration site for foreign gene expression in recombinant Streptococcus gordonii strains. Appl. Microbiol. Biotechnol. 2001, 55, 192–197. [Google Scholar] [CrossRef] [PubMed]
- Chan, A.H.; Wereszczynski, J.; Amer, B.R.; Yi, S.W.; Jung, M.E.; McCammon, J.A.; Clubb, R.T. Discovery of Staphylococcus aureus sortase A inhibitors using virtual screening and the relaxed complex scheme. Chem. Biol. Drug Des. 2013, 82, 418–428. [Google Scholar] [CrossRef] [PubMed]
- Zhulenkovs, D.; Rudevica, Z.; Jaudzems, K.; Turks, M.; Leonchiks, A. Discovery and structure-activity relationship studies of irreversible benzisothiazolinone-based inhibitors against Staphylococcus aureus sortase A transpeptidase. Bioorganic Med. Chem. 2014, 22, 5988–6003. [Google Scholar] [CrossRef]
- Kahlon, A.K.; Negi, A.S.; Kumari, R.; Srivastava, K.K.; Kumar, S.; Darokar, M.P.; Sharma, A. Identification of 1-chloro-2-formyl indenes and tetralenes as novel antistaphylococcal agents exhibiting sortase A inhibition. Appl. Microbiol. Biotechnol. 2014, 98, 2041–2051. [Google Scholar] [CrossRef] [PubMed]
- Uddin, R.; Lodhi, M.U.; Ul-Haq, Z. Combined pharmacophore and 3D-QSAR study on a series of Staphylococcus aureus Sortase A inhibitors. Chem. Biol. Drug Des. 2012, 80, 300–314. [Google Scholar] [CrossRef] [PubMed]
- Suree, N.; Liew, C.K.; Villareal, V.A.; Thieu, W.; Fadeev, E.A.; Clemens, J.J.; Jung, M.E.; Clubb, R.T. The structure of the Staphylococcus aureus sortase-substrate complex reveals how the universally conserved LPXTG sorting signal is recognized. J. Biol. Chem. 2009, 284, 24465–24477. [Google Scholar] [CrossRef] [PubMed]
- Ton-That, H.; Mazmanian, S.K.; Faull, K.F.; Schneewind, O. Anchoring of surface proteins to the cell wall of Staphylococcus aureus—Sortase catalyzed in vitro transpeptidation reaction using LPXTG peptide and NH2-Gly(3) substrates. J. Biol. Chem. 2000, 275, 9876–9881. [Google Scholar] [CrossRef] [PubMed]
- Ton-That, H.; Liu, G.; Mazmanian, S.K.; Faull, K.F.; Schneewind, O. Purification and characterization of sortase, the transpeptidase that cleaves surface proteins of Staphylococcus aureus at the LPXTG motif. Proc. Natl. Acad. Sci. USA 1999, 96, 12424–12429. [Google Scholar] [CrossRef] [PubMed]
- Escaich, S. Antivirulence as a new antibacterial approach for chemotherapy. Curr. Opin. Chem. Biol. 2008, 12, 400–408. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Qiu, J.; Tan, W.; Zhang, Y.; Wang, H.; Zhou, X.; Liu, S.; Feng, H.; Li, W.; Niu, X.; et al. Fisetin inhibits Listeria monocytogenes virulence by interfering with the oligomerization of listeriolysin O. J. Infect. Dis. 2014; pii: jiu520. [Google Scholar]
- Cascioferro, S.; Totsika, M.; Schillaci, D. Sortase A: An ideal target for anti-virulence drug development. Microb. Pathog. 2014, 77, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Macia, M.D.; Rojo-Molinero, E.; Oliver, A. Antimicrobial susceptibility testing in biofilm-growing bacteria. Clin. Microbiol. Infect. 2014, 20, 981–990. [Google Scholar] [CrossRef]
- Wang, J.; Zhou, X.; Liu, S.; Li, G.; Zhang, B.; Deng, X.; Niu, X. Novel inhibitor discovery and the conformational analysis of inhibitors of listeriolysin O via protein-ligand modeling. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Samples Availability: Samples of the compounds screened in this study are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, B.; Chen, F.; Bi, C.; Wang, L.; Zhong, X.; Cai, H.; Deng, X.; Niu, X.; Wang, D. Quercitrin, an Inhibitor of Sortase A, Interferes with the Adhesion of Staphylococcal aureus. Molecules 2015, 20, 6533-6543. https://doi.org/10.3390/molecules20046533
Liu B, Chen F, Bi C, Wang L, Zhong X, Cai H, Deng X, Niu X, Wang D. Quercitrin, an Inhibitor of Sortase A, Interferes with the Adhesion of Staphylococcal aureus. Molecules. 2015; 20(4):6533-6543. https://doi.org/10.3390/molecules20046533
Chicago/Turabian StyleLiu, Bingrun, Fuguang Chen, Chongwei Bi, Lin Wang, Xiaobo Zhong, Hongjun Cai, Xuming Deng, Xiaodi Niu, and Dacheng Wang. 2015. "Quercitrin, an Inhibitor of Sortase A, Interferes with the Adhesion of Staphylococcal aureus" Molecules 20, no. 4: 6533-6543. https://doi.org/10.3390/molecules20046533
APA StyleLiu, B., Chen, F., Bi, C., Wang, L., Zhong, X., Cai, H., Deng, X., Niu, X., & Wang, D. (2015). Quercitrin, an Inhibitor of Sortase A, Interferes with the Adhesion of Staphylococcal aureus. Molecules, 20(4), 6533-6543. https://doi.org/10.3390/molecules20046533





