Synthesis, Characterization, Crystal Structure and Antimicrobial Activity of Copper(II) Complexes with the Schiff Base Derived from 2-Hydroxy-4-Methoxybenzaldehyde
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemistry
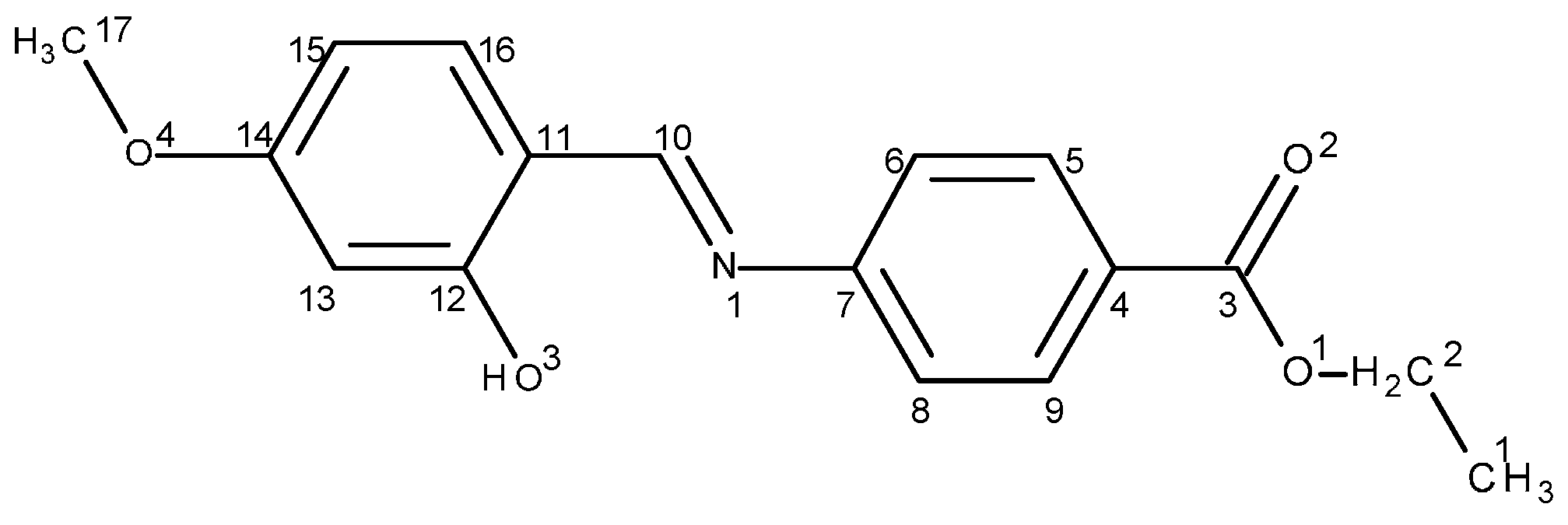

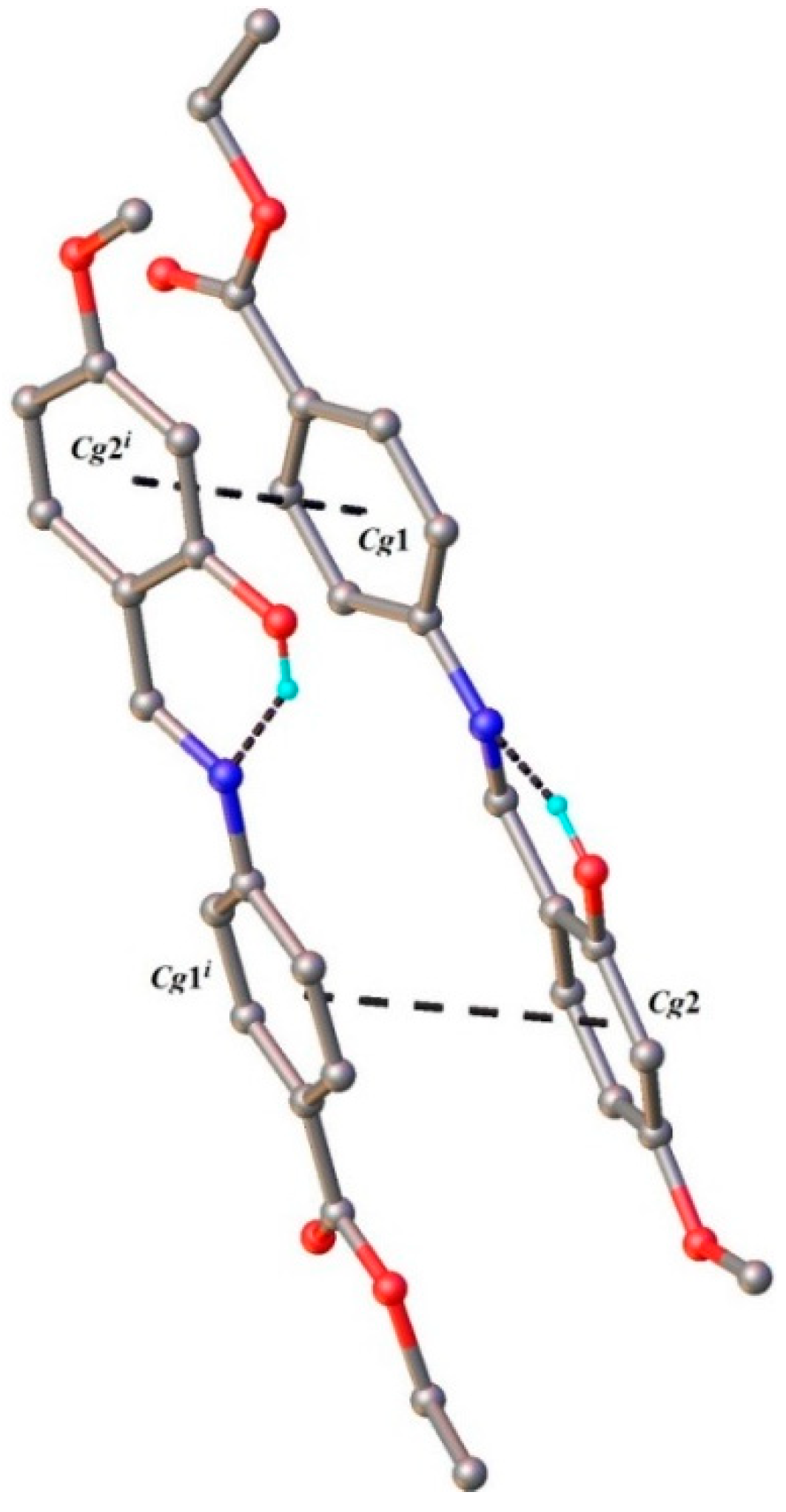
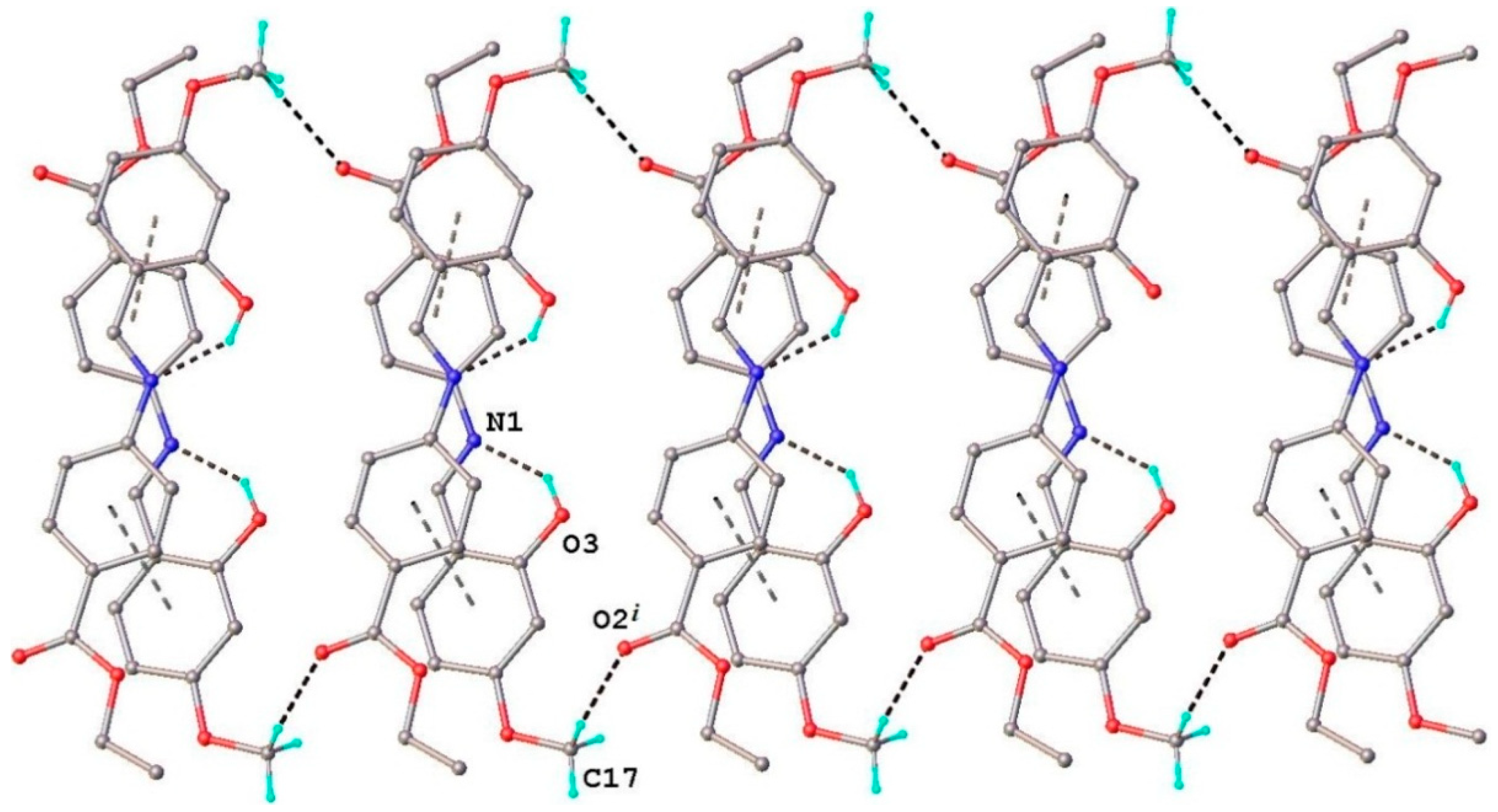
2.1.1. Structural characterization of (HL)

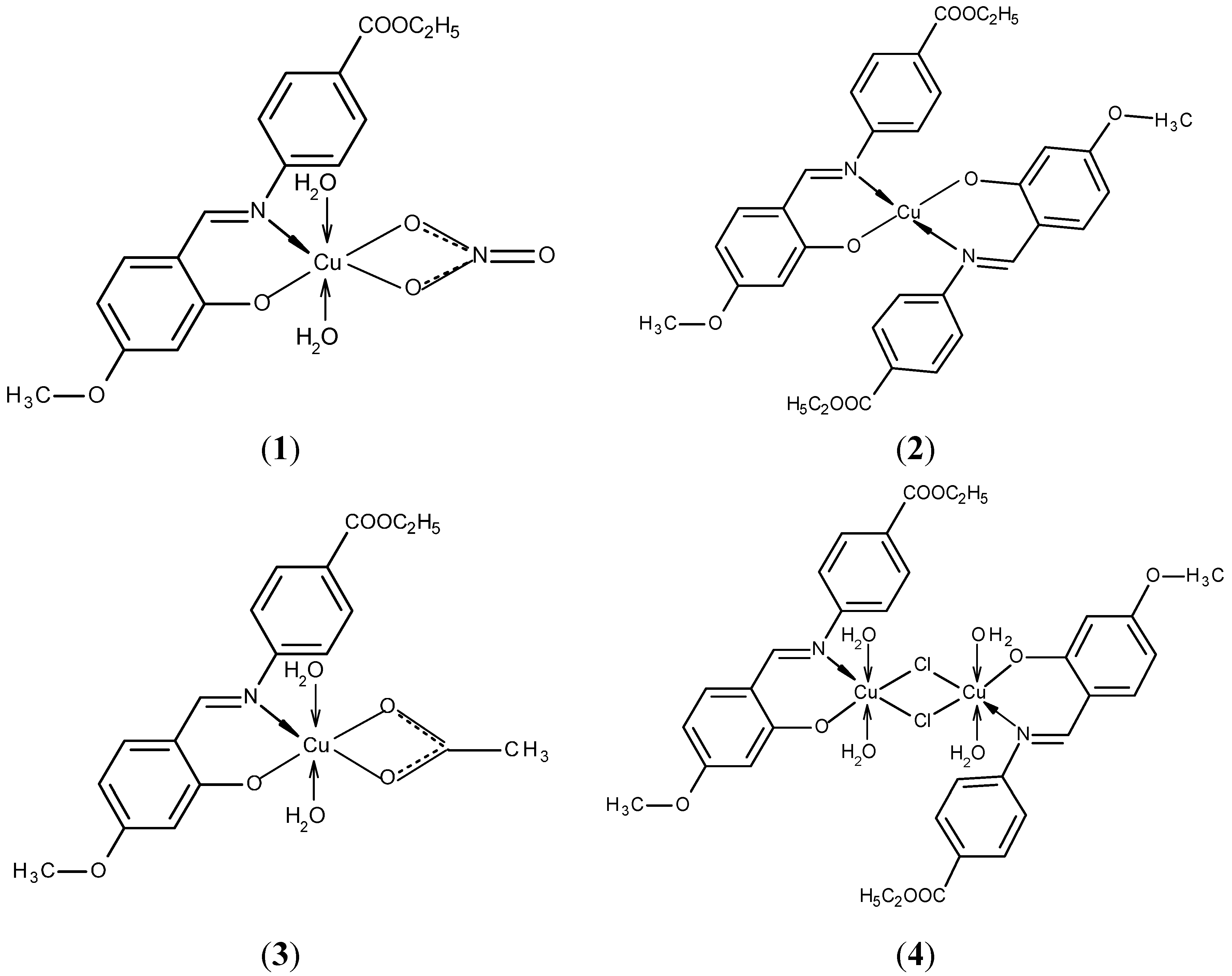
2.1.2. Structural Characterization of [Cu2(L2S)2(ClO4)(H2O)]ClO4·H2O (6)
| Bond Lengths (Å) for HL | Bond Angles (°) for HL | ||
|---|---|---|---|
| O1-C2 | 1.445(2) | C3-O1-C2 | 116.8(2) |
| O1-C3 | 1.343(2) | O1-C2-C1 | 107.4(2) |
| O2C3 | 1.210(2) | O1-C3-C4 | 112.4(2) |
| O3-C12 | 1.349(2) | O2-C3-O1 | 122.7(2) |
| O4-C14 | 1.363(2) | O2-C3-C4 | 124.9(2) |
| O4-C17 | 1.425(2) | C6-C7-N1 | 116.7(2) |
| N1-C7 | 1.411(2) | C8-C7-N1 | 125.0(2) |
| N1-C10 | 1.284(2) | C10-N1-C7 | 122.0(2) |
| C12-C13 | 1.386(2) | N1-C10-C11 | 122.3(2) |
| C13-C14 | 1.382(2) | O3-C12-C11 | 121.1(2) |
| O3-C12-C13 | 118.3(2) | ||
| O4-C14-C13 | 124.7(2) | ||
| O4-C14-C15 | 114.5(2) | ||
| C14-O4-C17 | 117.6(2) | ||
| Bond Lengths (Å) for 6 | |||
| Cu1-S1 | 2.406(2) | Cl1-O17 | 1.363(7) |
| Cu1-O9 | 1.952(4) | Cl1-O20 | 1.374(6) |
| Cu1-O5 | 1.941(4) | Cl1-O18 | 1.375(7) |
| Cu1-O1 | 2.244(4) | Cl1-O19 | 1.42(1) |
| Cu1-N3 | 1.957(5) | O5-C23 | 1.324(7) |
| Cu2-S2 | 2.404(2) | C6-O1 | 1.282(7) |
| Cu2-O9 | 1.947(4) | N2-C25 | 1.317(8) |
| Cu2-O5 | 1.965(4) | N2-C26 | 1.409(8) |
| Cu2-N2 | 1.956(5) | N3-C43 | 1.423(9) |
| Cu2-O13 | 2.323(4) | O13-C57 | 1.292(7) |
| Cu2-O1w | 2.356(6) | S1-C7 | 1.769(6) |
| S2-C41 | 1.786(6) | S1-C24 | 1.788(6) |
| S2-C58 | 1.777(6) | O9-C40 | 1.320(7) |
| Bond Angles (°) for 6 | |||
| O9-Cu1-S1 | 161.5(1) | O5-Cu2-O1W | 92.1(2) |
| O9-Cu1-O1 | 99.5(2) | N2-Cu2-S2 | 105.4(2) |
| O9-Cu1-N3 | 92.1(2) | N2-Cu2-O5 | 92.4(2) |
| O5-Cu1-S1 | 84.1(1) | N2-Cu2-O13 | 87.9(2) |
| O5-Cu1-O9 | 77.6(2) | N2-Cu2-O1W | 95.9(2) |
| O5-Cu1-O1 | 91.9(2) | O13-Cu2-S2 | 76.8(1) |
| O5-Cu1-N3 | 169.7(2) | O13-Cu2-O1W | 169.4(2) |
| O1-Cu1-S1 | 78.1(1) | O1W-Cu2-S2 | 92.6(2) |
| N3-Cu1-S1 | 106.1(2) | Cu2-O9-Cu1 | 101.9(2) |
| N3-Cu1-O1 | 88.8(2) | Cu1-O5-Cu2 | 101.7(2) |
| O9-Cu2-S2 | 84.6(1) | O17-Cl1-O20 | 108.8(5) |
| O9-Cu2-O5 | 77.2(2) | O17-Cl1-O18 | 113.2(6) |
| O9-Cu2-N2 | 168.8(2) | O17-Cl1-O19 | 104.4(8) |
| O9-Cu2-O13 | 89.4(2) | O20-Cl1-O18 | 114.7(4) |
| O9-Cu2-O1W | 88.7(2) | O20-Cl1-O19 | 107.7(6) |
| O5-Cu2-S2 | 161.0(1) | O18-Cl1-O19 | 107.3(6) |
| O5-Cu2-O13 | 97.6(2) | ||
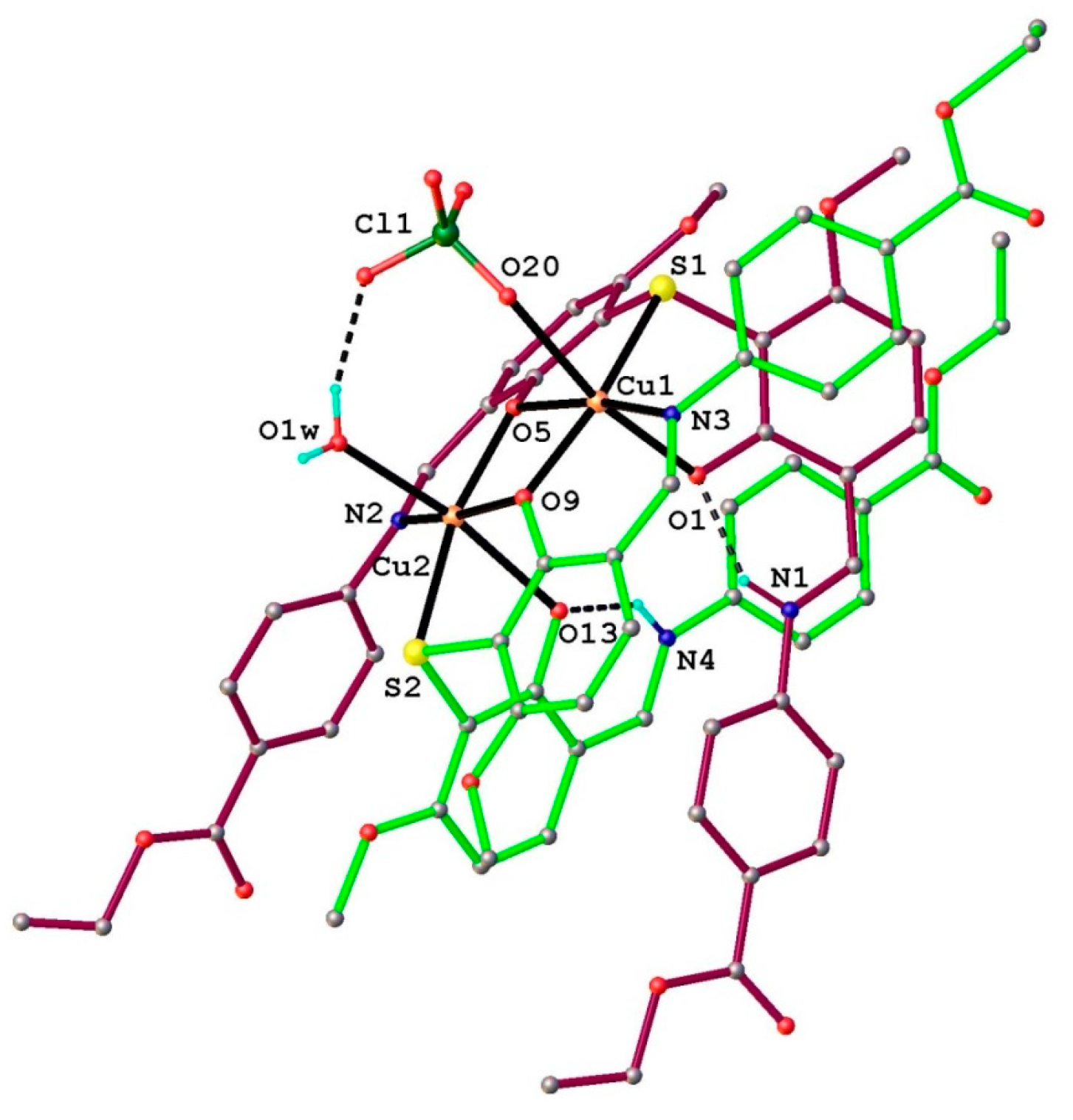
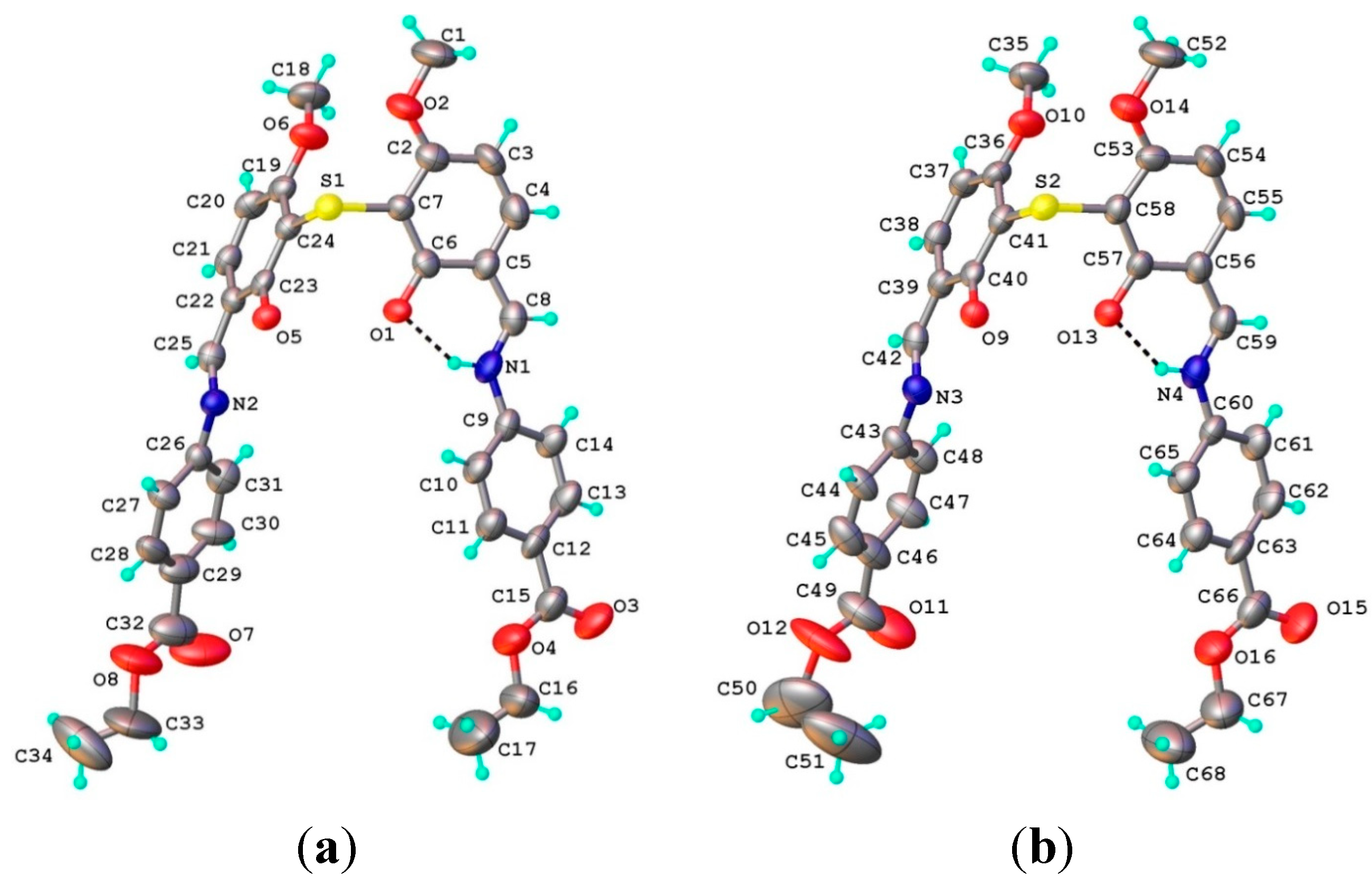
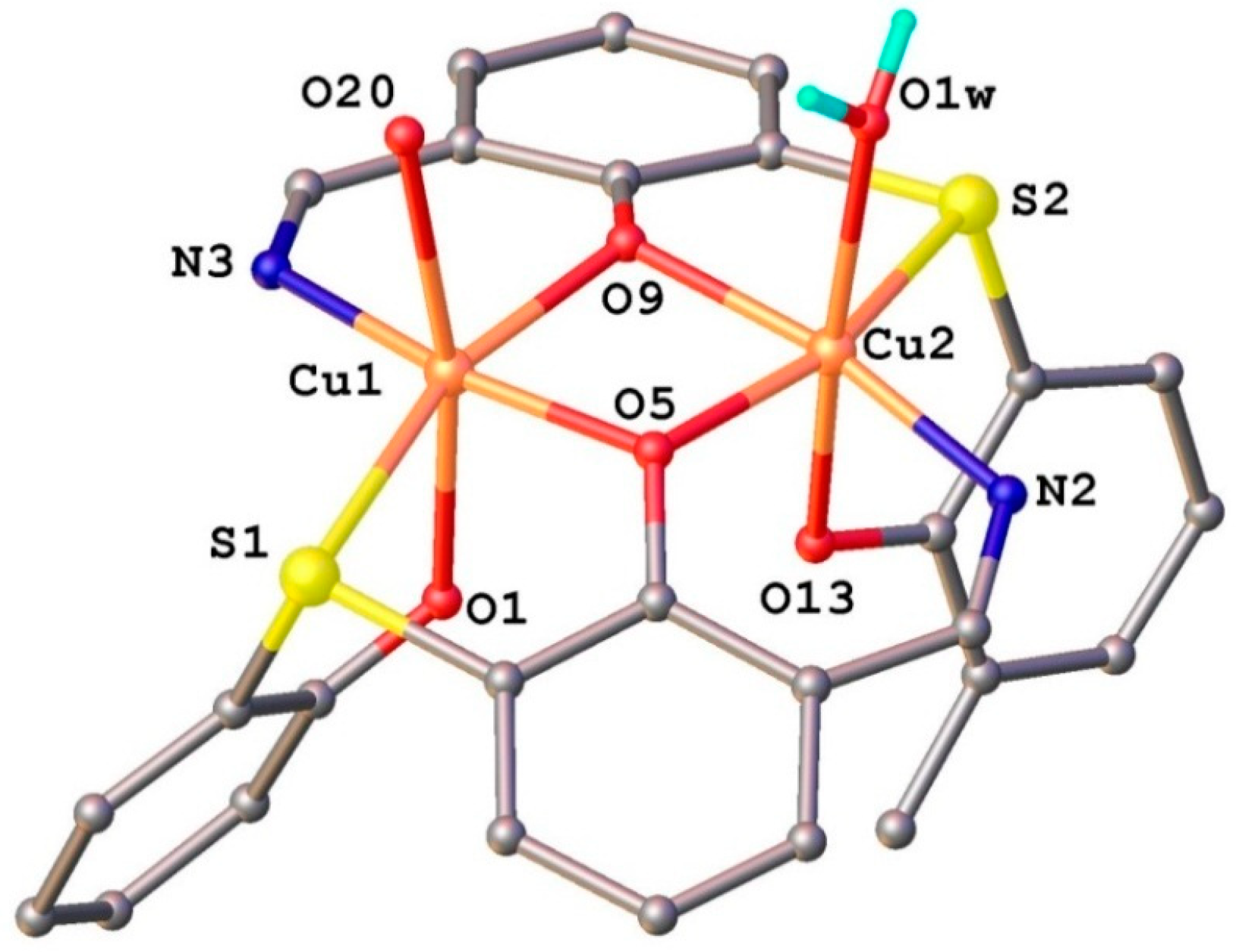
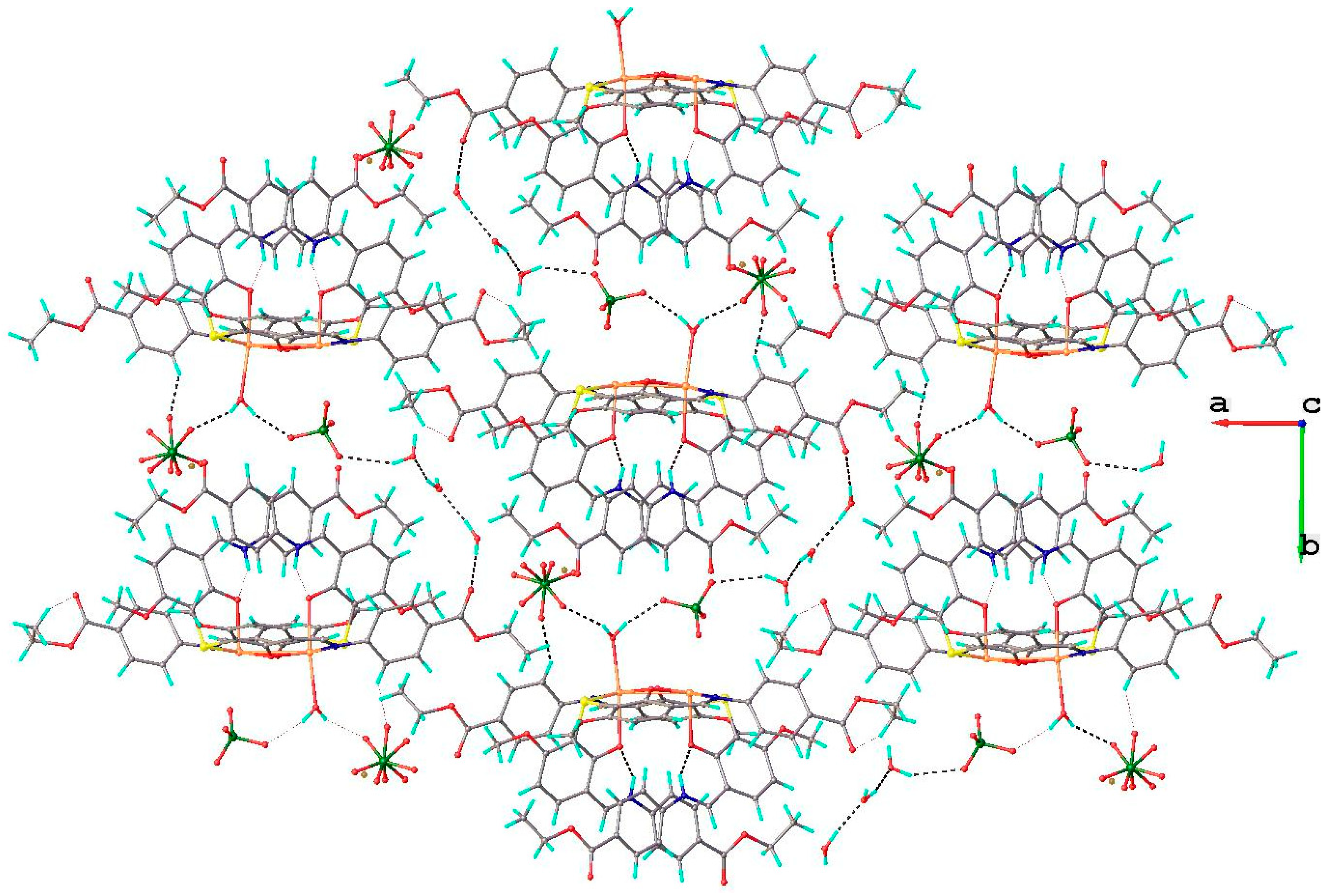
2.1.3. Infrared Spectra and Coordination Mode
2.1.4. Electronic Spectra and Magnetic Studies
| Metal Complex | Transitions d–d (cm−1) | µeff (BM) | Geometry | ||
|---|---|---|---|---|---|
| [Cu(L)(NO3)(H2O)2] (1) | 2B1g→2A1g2 10,500 | 2B1g→2B2g 13,420 | 2B1g→2Eg 19,400 | 1.98 | Octahedral distorted |
| [Cu(L)2] (2) | 2B2→2E 11,100 | 2B2→2B1(2A1) 13,100 | - | 1.87 | Pseudo-tetrahedral |
| [Cu(L)(OAc)] (3) | 2B2→2E 10,980 | 2B2→2B1(2A1) 13,500 | - | 1.92 | Pseudo-tetrahedral |
| [Cu2 (L)2Cl2(H2O)4] (4) | 2B1g→2A1g 11,000 | 2B1g→2B2g 12,820 | 2B1g→2Eg 16 940 | 1.07 | Octahedral distorted |
| [Cu(L)(ClO4)(H2O)] (5) | 2B2→2E 9850 | 2B2→2B1(2A1) 12,200 | - | 1.90 | Pseudo-tetrahedral |
2.1.5. Mass Spectra
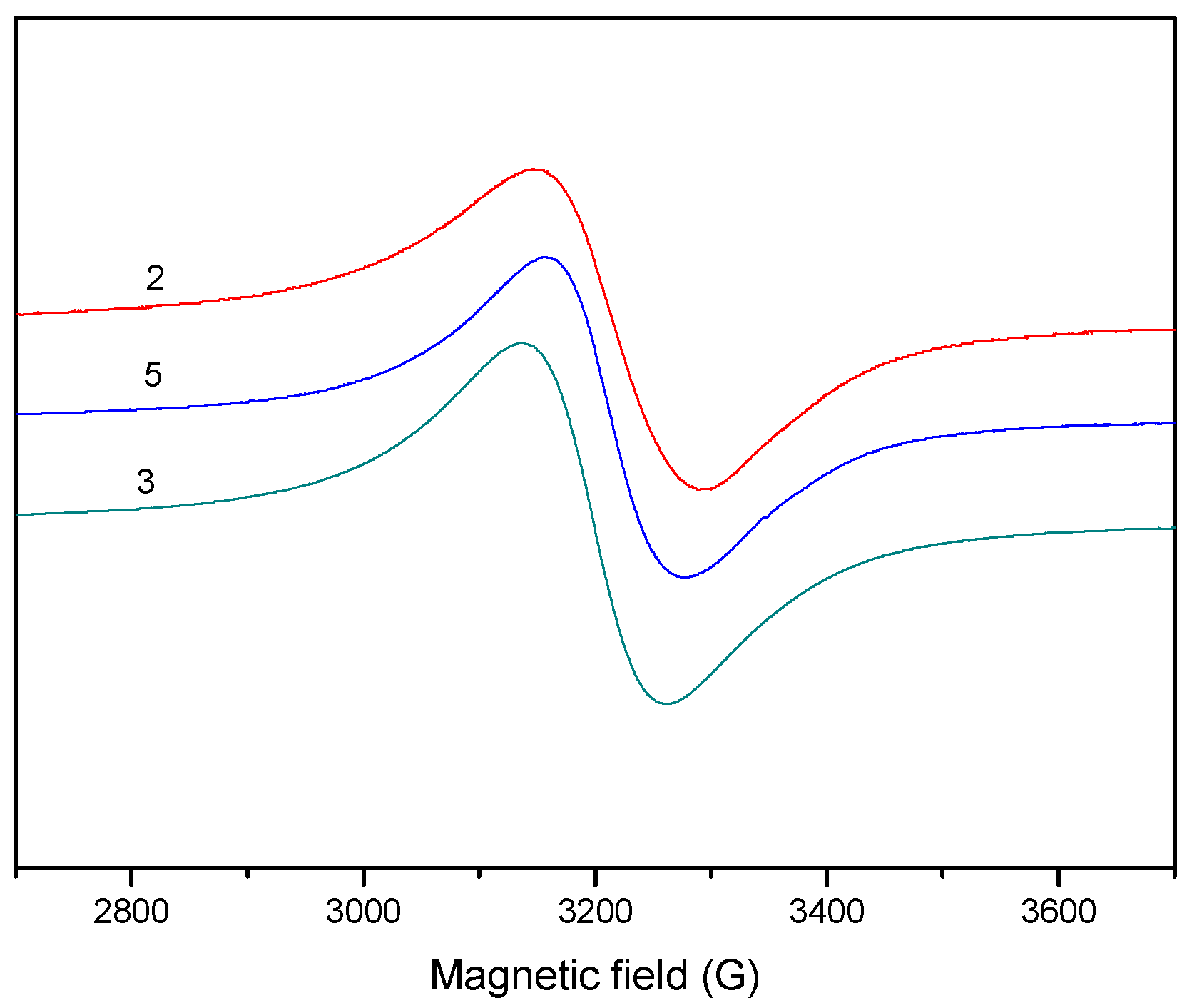
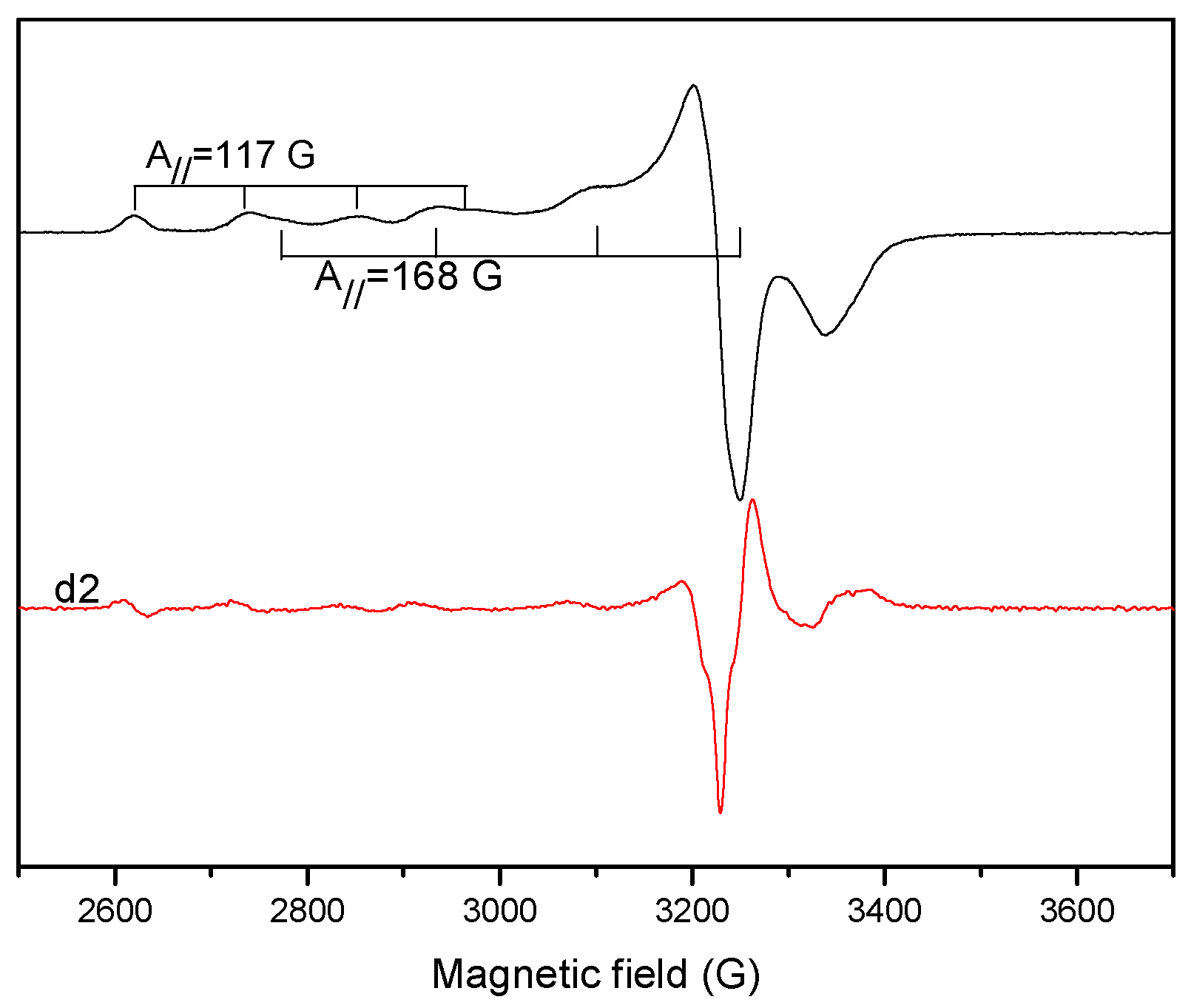
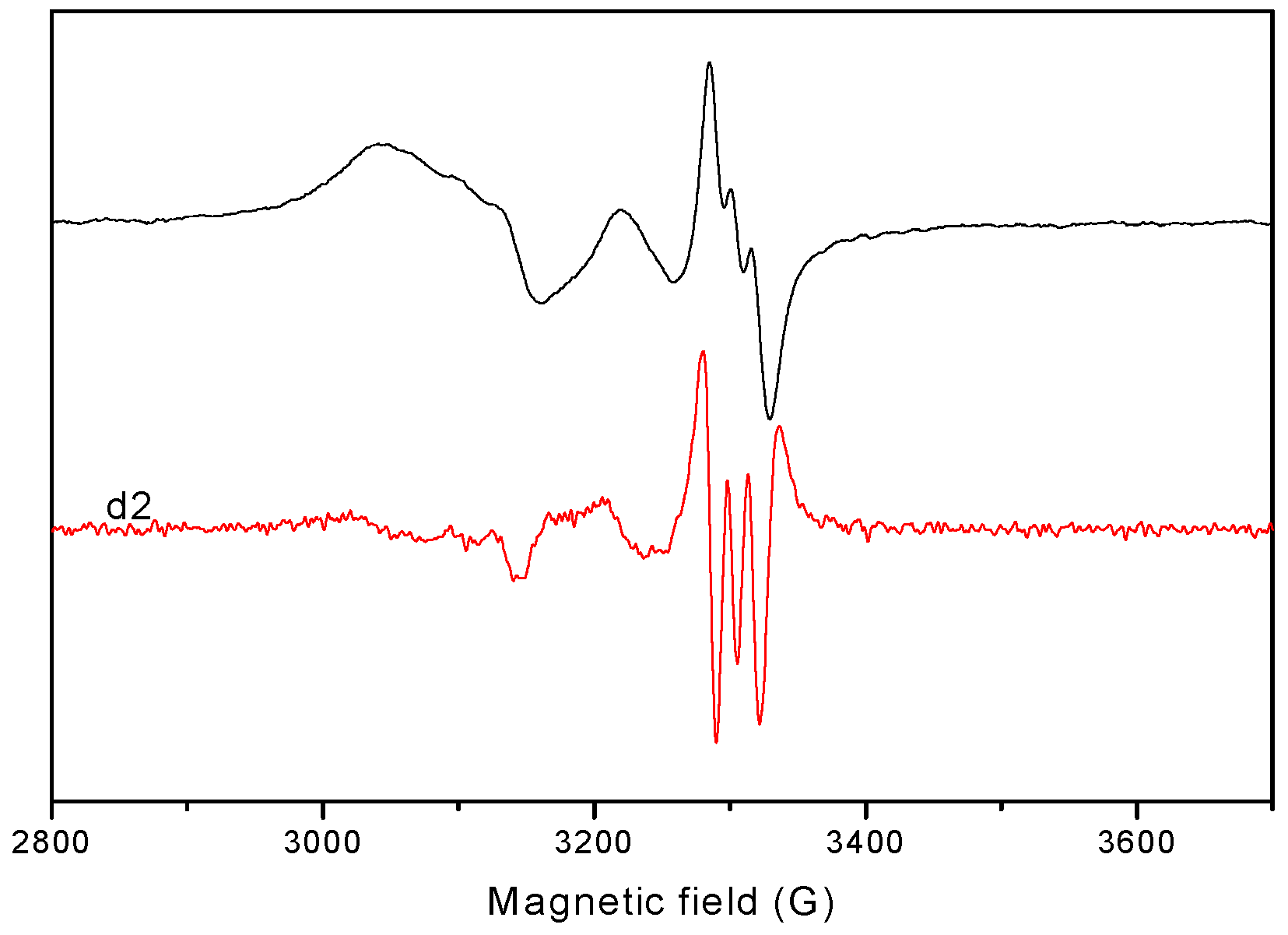
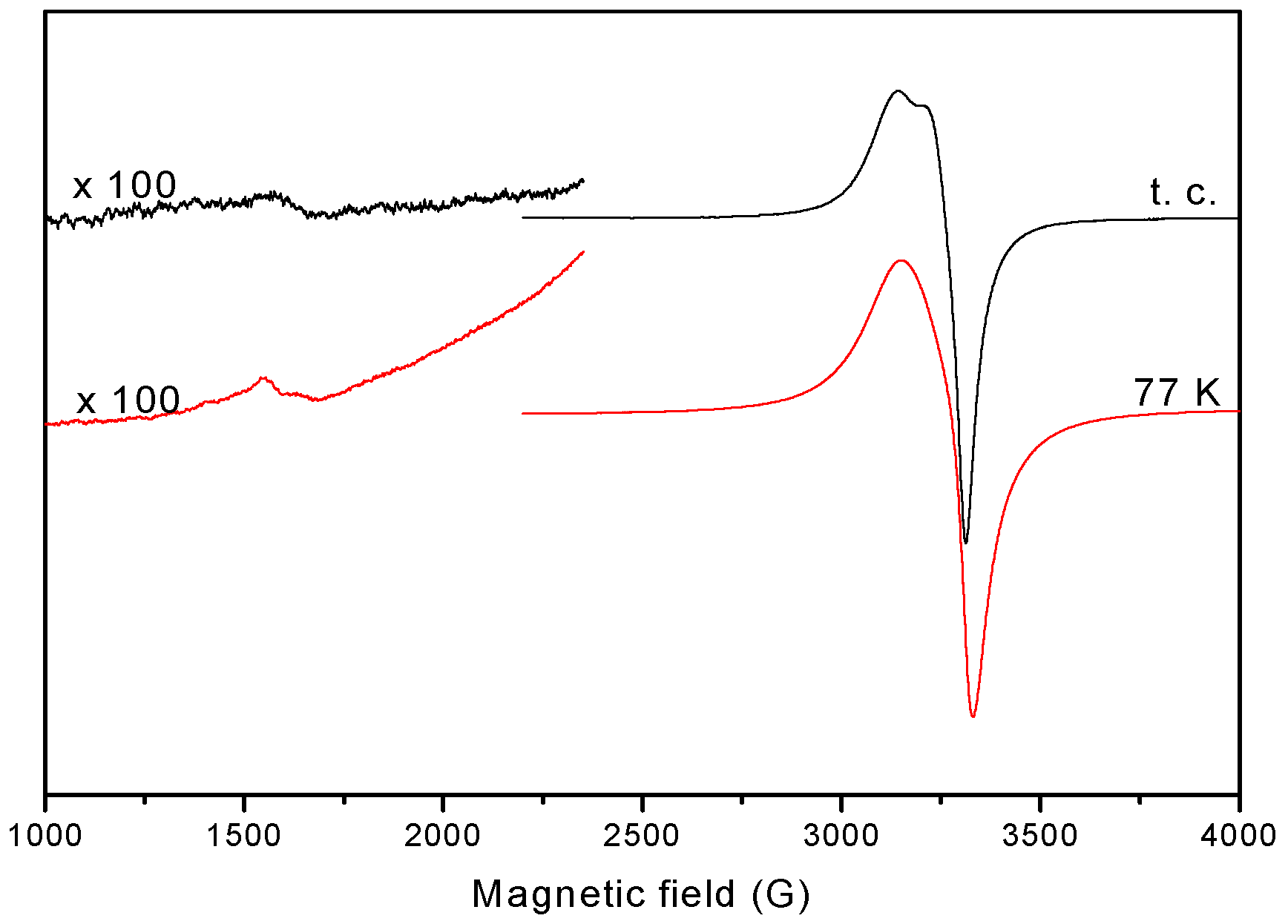
2.1.6. EPR Spectra
| 1 | 2 | 3 | 4 | 5 | |
|---|---|---|---|---|---|
| Polycrystalline (298K) | |||||
| gǁ | 2.230 | - | - | 2.132 | - |
| g⊥ | 2.060 | - | - | 2.059 | - |
| giso | - | 2.097 | 2.102 | - | 2.127 |
| DMSO (77 K) | |||||
| gǁ | 2.400 | 2.290 | 2.289 | 2.239 | 2.282 |
| g⊥ | 2.078 | 2.075 | 2.065 | 2.055 | 2.060 |
| Aǁ | 117.0 | 162.0 | 150.2 | 170.0 | 170.0 |
| α2 | 0.802 | 0.800 | 0.779 | 0.772 | 0.817 |
| β2 | 0.997 | 0.948 | 0.990 | 0.990 | 0.887 |
| δ2 | 0.862 | 0.958 | 0.922 | 0.950 | 0.801 |
| Kǁ | 0.800 | 0.755 | 0.762 | 0.770 | 0.718 |
| K⊥ | 0.692 | 0.760 | 0.715 | 0.735 | 0.658 |
2.2. Biological Activity
| Compounds | E. coli (G−) | S. enteritidis (G−) | S. aureus (G+) | Enterococcus (G+) | C. albicans | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | |
| C17H17NO4 (HL4) | > 10.0 | >10.0 | >10.0 | >10.0 | >10.0 | >10.0 | >10.0 | >10.0 | >10.0 | >10.0 |
| [Cu(L)(NO3)(H2O)2] (1) | >10.0 | >10.0 | >10.0 | >10.0 | 0.5 | >10.0 | 0.5 | >10.0 | 0.5 | >10.0 |
| [Cu(L)2] (2) | >10.0 | >10.0 | >10.0 | >10.0 | 0.5 | >10.0 | 0.5 | >10.0 | 0.12 | >10.0 |
| [Cu(L)(OAc)] (3) | >10.0 | >10.0 | >10.0 | >10.0 | >10.0 | >10.0 | 0.5 | >10.0 | >10.0 | >10.0 |
| [Cu2 (L)2Cl2(H2O)4] (4) | >10.0 | >10.0 | >10.0 | >10.0 | 0.5 | >10.0 | >10.0 | >10.0 | >10.0 | >10.0 |
| [Cu(L)(ClO4)(H2O)] (5) | 0.5 | >10.0 | >10.0 | >10.0 | 0.5 | >10.0 | 0.5 | >10.0 | 0.12 | >10.0 |
| Furacillinum | 0.018 | 0.037 | 0.009 | 0.009 | 0.009 | 0.009 | 0.037 | 0.037 | - | - |
| Nystatine | - | - | - | - | - | - | - | - | 0.08 | 0.08 |
3. Experimental
3.1. General Information
| Compound | HL | 6 |
|---|---|---|
| Empirical formula | C17H17NO4 | C68H66Cl2Cu2N4O26S2 |
| Formula weight | 299.32 | 1617.35 |
| Temperature/K | 200 | 293 |
| Crystal system | monoclinic | monoclinic |
| Space group | C2/c | P21/c |
| a/Å | 15.4089(7) | 16.7582(11) |
| b/Å | 6.4308(3) | 26.2186(13) |
| c/Å | 29.8710(16) | 17.9269(12) |
| α/° | 90.00 | 90.00 |
| β/° | 95.916(4) | 107.710(8) |
| γ/° | 90.00 | 90.00 |
| V/Å3 | 2944.2(2) | 7503.4(8) |
| Z | 8 | 4 |
| Dcalc/mg/mm3 | 1.351 | 1.432 |
| μ/mm−1 | 0.097 | 0.774 |
| Crystal size/mm3 | 0.2 × 0.1 × 0.1 | 0.35 × 0.35 × 0.1 |
| θmin, θ max (°) | 6.22 to 49.42 | 3.92 to 46.52 |
| Reflections collected | 4619 | 30514 |
| Independent reflections | 2404 [Rint = 0.0212] | 10,753 [Rint = 0.0590] |
| Data/restraints/parameters | 2404/0/201 | 10,753/29/942 |
| GOF c | 1.010 | 1.028 |
| R1 a (I > 2σ(I)) | 0.0434 | 0.0791 |
| wR2 b (all data) | 0.1020 | 0.2287 |
| Largest diff. peak/hole/e Å‒3 | 0.11/−0.15 | 0.72/−0.87 |
3.2. Chemistry
3.2.1. Synthesis of the Schiff Base Ethyl 4-[(E)-(2-hydroxy-4-methoxyphenyl)methyleneamino] benzoate (HL)
3.2.2. General Procedure for the Preparation of the Metal Complexes 1–6
3.3. Antibacterial Activity
3.4. Antifungal Bioassay
4. Conclusions
Supplementary Materials
Acknowledgments
Appendix
Author Contributions
Conflicts of Interest
References
- Rawal, R.K.; Tripathi, R.; Kati, S.B.; Pannecouque, C.; de Clercq, E. Design, synthesis, and evaluation of 2-aryl-3-heteroaryl-1, 3-thiazolidin-4-ones as anti-HIV agents. Bioorg. Med. Chem. 2007, 15, 1725–1731. [Google Scholar] [CrossRef] [PubMed]
- Giovine, A.; Muraglia, M.; Florio, M.A.; Rosato, A.; Corbo, F.; Franchini, C.; Musio, B.; Degennaro, L.; Luisi, R. Synthesis of functionalized arylaziridines as potential antimicrobial agents. Molecules 2014, 19, 11505–11519. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.H.; Keck, J.G.; Lien, E.J.; Lai, M.M.C. Design, synthesis, testing and quantitative structure-activity relationship analysis of substituted salicylaldehyde Schiff bases of 1-amino-3hydroxyguanidine tosylate as new antiviral agents against coronavirus. J. Med. Chem. 1990, 33, 608–614. [Google Scholar] [CrossRef] [PubMed]
- Jarrahpour, A.; Motamedifar, M.; Pakshir, K.; Hadi, N.; Zarei, M. Synthesis of novel azo Schiff bases and their antibacterial and antifungal activities. Molecules 2004, 9, 815–824. [Google Scholar] [CrossRef] [PubMed]
- Omar, M.M.; Mohamed, G.G.; Ibrahim, A.A. Spectroscopic characterization of metal complexes of novel Schiff base. Synthesis, thermal and biological activity studies. Spectrochim. Acta A 2009, 73, 358–369. [Google Scholar] [CrossRef]
- Ceyhana, G.; Urus, S.; Demirtas, I.; Elmastas, M.; Tumera, M. Antioxidant, electrochemical, thermal, antimicrobial and alkane oxidation properties of tridentate Schiff base ligands and their metal complexes. Spectrochim. Acta A 2011, 81, 184–198. [Google Scholar] [CrossRef]
- Mladenova, R.; Ignatova, M.; Manolova, N.; Petrova, T.; Rashkov, I. Preparation, characterization and biological activity of Schiff base compounds derived from 8-hydroxyquinoline-2-carboxaldehyde and Jeffamines ED. Eur. Polym. J. 2002, 38, 989–999. [Google Scholar] [CrossRef]
- Huang, G.S.; Liang, Y.M.; Wu, X.L.; Liu, W.M.; Ma, Y.X. Some ferrocenyl Schiff bases with nonlinear optical properties. Organomet. Chem. 2003, 17, 706–710. [Google Scholar] [CrossRef]
- Walsh, O.M.; Meegan, M.J.; Prendergast, R.M.; Nakib, T.A. Synthesis of 3-acetoxyazetidin-2-ones and 3-hydroxyazetidin-2-ones with antifugal and antifungal and antibacterial activity. Eur. J. Med. Chem. 1996, 31, 989–1000. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, Z. Synthesis, crystal structure, antioxidation and DNA binding properties of binuclear Ho(III) complexes of Schiff-base ligands derived from 8-hydroxyquinoline-2-carboxyaldehyde and four aroylhydrazines. J. Organomet. Chem. 2009, 694, 3091–3101. [Google Scholar] [CrossRef]
- Santra, B.K.; Reddy, P.A.N.; Neelakanta, G.; Mahadevan, S.; Nethaji, M.; Chakravarty, A.R. Oxidative cleavage of DNA by a dipyridoquinoxaline copper(II) complex in the presence of ascorbic acid. J. Inorg. Biochem. 2002, 89, 191–196. [Google Scholar] [CrossRef] [PubMed]
- Aslantaş, M.; Kendi, E.; Demir, N.; Şabik, A.E.; Tümer, M.; Kertmen, M. Synthesis, spectroscopic, structural characterization, electrochemical and antimicrobial activity studies of the Schiff base ligand and its transition metal complexes. Spectrochim. Acta A 2009, 74, 617–624. [Google Scholar] [CrossRef]
- Chandra, S.; Kumar, A. Electronic, EPR and magnetic studies of Co(II), Ni(II) and Cu(II) complexes with thiosemicarbazone (L1) and semicarbazone (L2) derived from pyrole-2-carboxyaldehyde. Spectrochim. Acta A 2007, 67, 697–701. [Google Scholar] [CrossRef]
- Das, A.; Trousdale, M.D.; Ren, S.; Lien, E.J. Inhibition of herpes simplex virus type 1 and adenovirus type 5 by heterocyclic Schiff bases of aminohydroxyguanidine tosylate. Antivir. Res. 1999, 44, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Shanker, K.; Rohini, R.; Reddy, P.M.; Ho, Y.P.; Ravinder, V. Ru(II) complexes of N4 and N2O2 macrocyclic Schiff base ligands: Their antibacterial and antifungal studies. Spectrochim. Acta A 2009, 73, 205–211. [Google Scholar] [CrossRef]
- Kureshy, R.I.; Khan, N.H.; Abdi, S.H.R.; Patel, S.T.; Iyer, P. Chiral Ru(II) Schiff base complexes catalysed enantioselective eposidation of styrene derivatives using iodosyl benzene as oxidant II. J. Mol. Catal. 1999, 150, 175–183. [Google Scholar] [CrossRef]
- Aoyama, Y.; Kujisawa, J.T.; Walanawe, T.; Toi, A.; Ogashi, H. Catalytic reactions of metalloporphyrins. 1. Catalytic modification of borane reduction of ketone with rhodium(III) porphyrin as catalyst. J. Am. Chem. Soc. 1986, 108, 943–947. [Google Scholar] [CrossRef]
- Kelly, T.R.; Whiting, A.; Chandrakumar, N.S. A rationally designed, chiral Lewis acid for the asymmetric induction of some Diels-Alder reactions. J. Am. Chem. Soc. 1986, 108, 3510–3512. [Google Scholar] [CrossRef]
- Sengupta, P.; Ghosh, S.; Mak, T.C.W. A new route for the synthesis of bis(pyridine dicarboxylato)bis(triphenylphosphine) complexes of ruthenium(II) and X-ray structural characterisation of the biologically active trans-[Ru(PPh3)2(L1H)2] (L1H2=pyridine 2,3-dicarboxylic acid). Polyhedron 2001, 20, 975–980. [Google Scholar] [CrossRef]
- Ibrahim, O.B.; Mohamed, M.A.; Refat, M.S. Nano Sized Schiff Base Complexes with Mn(II), Co(II), Cu(II), Ni(II) and Zn(II) Metals: Synthesis, Spectroscopic and Medicinal Studies. Can. Chem. Trans. 2014, 2, 108–121. [Google Scholar]
- Green, D.R.; Reed, J.C. Mitochondria and apoptosis. Science 1998, 281, 1309–1312. [Google Scholar] [CrossRef] [PubMed]
- Easmon, J.; Pürstinger, G.; Heinisch, G.; Roth, T.; Fiebig, H.H.; Holzer, W.; Jäger, W.; Jenny, M.; Hofmann, J. Synthesis, cytotoxicity, and antitumor activity of copper(II) and iron(II) complexes of (4)N-azabicyclo[3.2.2]nonane thiosemicarbazones derived from acyl diazines. J. Med. Chem. 2001, 44, 2164–2171. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Dhar, D.N.; Saxena, P.N. Applications of metal complexes of Schiff bases—A review. J. Sci. Ind. Res. 2009, 68, 181–187. [Google Scholar]
- Morrow, M.E.; Berry, C.W. Antimicrobial properties of topical anesthetic liquids containing lidocaine or benzocaine. Anesth. Prog. 1988, 35, 9–13. [Google Scholar] [PubMed]
- Pongratz, Z. Über neue pharmakologisch wirksame Amide und Ester der Nicotinsäure. Monatsh. Chem. 1957, 88, 330–335. [Google Scholar] [CrossRef]
- Perrin, D.D.; Armarego, W.L.; Perrin, D.R. Purification of Laboratory Chemicals, 2nd ed.; Pergamon: New York, NY, USA, 1990; pp. 140–142. [Google Scholar]
- Geary, W.J. The use of conductivity measurements in organic solvents for the characterization of coordination compounds. Coord. Chem. Rev. 1971, 7, 81–115. [Google Scholar] [CrossRef]
- Rosu, T.; Pahontu, E.; Maxim, C.; Georgescu, R.; Stanica, N.; Gulea, A. Some new Cu(II) complexes containing an ON donor Schiff base: Synthesis, characterization and antibacterial activity. Polyhedron 2011, 30, 154–162. [Google Scholar] [CrossRef]
- Rosu, T.; Pasculescu, S.; Lazar, V.; Chifiriuc, C.; Cernat, R. Copper(II) Complexes with Ligands Derived from 4-Amino-2,3-dimethyl-1-phenyl-3-pyrazoline-5-one. Molecules 2006, 11, 904–914. [Google Scholar] [CrossRef] [PubMed]
- Rosu, T.; Gulea, A.; Nicolae, A.; Georgescu, R. Complexes of 3d(n) metal ions with thiosemicarbazones: Synthesis and antimicrobial activity. Molecules 2007, 12, 782–796. [Google Scholar] [CrossRef] [PubMed]
- Rosu, T.; Negoiu, M.; Pasculescu, S.; Pahontu, E.; Poirier, D.; Gulea, A. Metal-based biologically active agents: Synthesis, characterization, antibacterial and antileukemia activity evaluation of Cu(II), V(IV) and Ni(II) complexes with antipyrine-derived compounds. Eur. J. Med. Chem. 2010, 45, 774–781. [Google Scholar] [CrossRef] [PubMed]
- Rosu, T.; Pahontu, E.; Maxim, C.; Georgescu, R.; Stanica, N.; Almajan, G.L.; Gulea, A. Synthesis, characterization and antibacterial activity of some new complexes of Cu(II), Ni(II), VO(II), Mn(II) with Schiff base derived from 4-amino-2,3-dimethyl-1-phenyl-3-pyrazolin-5-one. Polyhedron 2010, 29, 757–766. [Google Scholar] [CrossRef]
- Raphael, P.F.; Manoj, E.; Prathapachandra Kurup, M.R. Copper(II) complexes of N(4)-substituted thiosemicarbazones derived from pyridine-2-carbaldehyde: Crystal structure of a binuclear complex. Polyhedron 2007, 26, 818–828. [Google Scholar] [CrossRef]
- Khandar, A.A.; Hosseini-Yazdi, S.A. Synthesis, X-ray crystal structure, and solution properties of nickel(II) complexes of new 16-membered mixed-donor macrocyclic Schiff base ligand incorporating a pendant alcohol function. Polyhedron 2003, 22, 1481–1487. [Google Scholar] [CrossRef]
- Vicente, M.; Bastida, R.; Lodeiro, C.; Macias, A.; Parola, A.J.; Valencia, L.; Spey, S.E. Metal complexes with a new N4O3 amine pendant-armed macrocyclic ligand: Synthesis, characterization, crystal structures, and fluorescence studies. Inorg. Chem. 2003, 42, 6768–6779. [Google Scholar] [CrossRef] [PubMed]
- Lodeiro, C.; Bastida, R.; Bertolo, E.; Macias, A.; Rodriguez, A. Coordination chemistry of copper(II) with oxaaza macrocyclic ligands: Crystal structure of a dinuclear tetramer copper(II) complex. Polyhedron 2003, 22, 1701–1710. [Google Scholar] [CrossRef]
- Kannappan, R.; Tanase, S.; Mutikainen, I.; Turpeinen, U.; Reedijk, J. Low-spin iron(III) Schiff-base complexes with symmetric hexadentate ligands: Synthesis, crystal structure, spectroscopic and magnetic properties. Polyhedron 2006, 25, 1646–1654. [Google Scholar] [CrossRef]
- Patel, R.N.; Gundla, V.L.N.; Patel, D.K. Synthesis, structure and properties of some copper(II) complexes containing an ONO donor Schiff base and substituted imidazole ligands. Polyhedron 2008, 27, 1054–1060. [Google Scholar] [CrossRef]
- Nakamoto, K. Infrared Spectra of Inorganic and Coordination Compounds; Wiley and Sons: New York, NY, USA, 1986; pp. 212, 232, 248, 251, 256. [Google Scholar]
- Nakamoto, K. Infrared and Raman Spectra of Inorganic and Coordination Compounds, 5th ed.; Wiley-Interscience: New York, NY, USA, 1997; p. 86. [Google Scholar]
- Belicchi-Ferrari, M.; Bisceglie, F.; Cavalieri, C.; Pelosi, G.; Tarasconi, P. Bis(triphenylphosphine)4-fluorobenzaldehyde thiosemicarbazone copper(I): Forcing chelation through oxoanions. Polyhedron 2007, 26, 3774–3782. [Google Scholar] [CrossRef]
- Bosnich, B. An interpretation of the circular dichroism and electronic spectra of salicylaldimine complexes of square-coplanar diamagnetic nickel(II). J. Am. Chem. Soc. 1968, 90, 627–632. [Google Scholar] [CrossRef]
- Dowing, R.S.; Urbach, F.L. Circular dichroism of square-planar, tetradentate Schiff base chelates of copper(II). J. Am. Chem. Soc. 1969, 91, 5977–5983. [Google Scholar] [CrossRef]
- Lever, A.P.B. Inorganic Electronic Spectroscopy, 2nd ed.; Elsevier Science: New York, NY, USA, 1984. [Google Scholar]
- John, R.P.; Sreekanth, A.; Rajakannan, V.; Ajith, T.A.; Kurup, M.R.P. New copper(II) complexes of 2-hydroxyacetophenone N(4)-substituted thiosemicarbazones and polypyridyl co-ligands: Structural, electrochemical and antimicrobial studies. Polyhedron 2004, 23, 2549–2559. [Google Scholar] [CrossRef]
- Wilkinson, G.; Gillard, D.R.; McCleverty, A.J. Comprehensive Coordination Chemistry; Pergamon Press: New York, NY, USA, 1987; pp. 3–5. [Google Scholar]
- Carlin, R.L. Transition Metal Chemistry, 2nd ed.; Marcel Decker: New York, NY, USA, 1965. [Google Scholar]
- Zanello, P.; Tamburini, S.; Vigato, P.A.; Mazzocchin, G.A. Syntheses, structure, and electrochemical characterization of homo- and heterodinuclear copper complexes with compartmental ligands. Coord. Chem. Rev. 1987, 77, 165–273. [Google Scholar] [CrossRef]
- Guerriero, P.; Tamburini, S.; Vigato, P.A. From mononuclear to polynuclear macrocyclic or macroacyclic complexes. Coord. Chem. Rev. 1995, 139, 17–243. [Google Scholar] [CrossRef]
- Yu, J.W.; Tao, R.J.; Zhou, X.Y.; Jui, D.M.; Liao, D. Syntheses and magnetic properties of heterobinuclear complexes with N,N'-bis(3-caboxylsalicylidene)trimethylenediamine. Polyhedron 1994, 13, 951–955. [Google Scholar] [CrossRef]
- Lam, F.; Wang, R.J.; Mak, T.C.W.; Chan, K.S. Synthesis of novel dinickel(II) and nickel(II)-copper(II) bimetallic complexes derived from an acyclic dinucleating Schiff base-pyridine ligand. J. Chem. Soc. Chem. Commun. 1994, 21, 2439–2440. [Google Scholar] [CrossRef]
- Benzekri, A.; Dubourdeaux, P.; Latour, J.M.; Rey, P.; Laugier, J. Binuclear copper(II) complexes of a new sulphur-containing binucleating ligand: Structural and physicochemical properties. J. Chem. Soc. Dalton Trans. 1991, 12, 3359–3365. [Google Scholar] [CrossRef]
- Kettle, S.F.A. Physical Inorganic Chemistry Approach; Oxford University Press: Oxford, UK, 1998. [Google Scholar]
- Anderson, P.W. Antiferromagnetism. Theory of Superexchange Interaction. Phys. Rev. 1950, 79, 350–352. [Google Scholar] [CrossRef]
- Hathaway, B.J.; Billing, D.E. The electronic properties and stereochemistry of mononuclear complexes of the copper(II) ion. Coord. Chem. Rev. 1970, 5, 143–207. [Google Scholar] [CrossRef]
- Bew, M.J.; Hathaway, B.J.; Faraday, R.R. Electronic properties and stereochemistry of the copper(II) ion. Part VII. Mono(diethylenetriamine)copper(II) complexes. J. Chem. Soc. Dalton Trans. 1972, 12, 1229–1237. [Google Scholar] [CrossRef]
- Siegel, I.; Jones, E.P. Electronic bonding of Cu2+ in amorphous and crystalline TeO2: EPR and optical spectra. J. Chem. Phys. 1972, 57, 2364–2371. [Google Scholar] [CrossRef]
- Maki, A.H.; McGarvey, B.R. Electron spin resonance in transition metal chelates. I. Copper (II) bis-acetylacetonate. J. Chem. Phys. 1958, 29, 31. [Google Scholar] [CrossRef]
- Kivelson, D.; Neiman, R. ESR studies on the bonding in copper complexes. J. Chem. Phys. 1961, 35, 149–155. [Google Scholar] [CrossRef]
- Hathaway, B.J. Structure and Bonding; Springer Verlag: Heidelberg, Germany, 1973; p. 60. [Google Scholar]
- Hathaway, B.J. Comprehensive Coordination Chemistry; Wilkinson, G., Gillard, D.R., McCleverty, A.J., Eds.; Pergamon Press: New York, NY, USA, 1987. [Google Scholar]
- Barry, A. Procedures and theoretical considerations for testing antimicrobial agents in agar media. In Antibiotics in Laboratory Medicine, 5th ed.; Lorian, A., Ed.; Williams and Wilkins: Baltimore, MD, USA, 1991. [Google Scholar]
- National Committee for Clinical Laboratory Standard. NCCLS: Methods for Antimicrobial Dilution and Disk Susceptibility Testing of Infrequently Isolated or Fastidious Bacteria, Approved Guideline; Document M45-A, 26(19); NCCLS: Willanova, PA, USA, 1999. [Google Scholar]
- CrysAlis RED; Version 1.171.36.32; Oxford Diffraction Ltd.: Abingdon, UK, 2003.
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. Olex2: A complete structure solution, refinement and analysis program. J. Appl. Crystallogr. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A short history of SHELXS. Acta Crystallogr. A 2008, 64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Samusi, N.; Prisacari, V.; Tapcov, V.; Buraciov, S. Di(μ-O)di[N-(2-oxo-1-naftali)-N1-α-oxobenzalihidrazincopper] Dihidrate, Which Have Selective Antibacterien Activity. Patent of Invention MD 678 BOPI, 1997. [Google Scholar]
- Samusi, N.M.; Prisacari, V.I.; Tapcov, V.I.; Buraciov, S.A.; Gulea, A.P. Synthesis and antimicrobial activity of the complexes of 3d-metals with substituted salicylaldehyde benzoylhydrazones. Pharm. Chem. J. 2004, 38, 373–377. [Google Scholar] [CrossRef]
- Gulea, A.P.; Samusi, N.M.; Prisacari, V.I.; Tapcov, V.; Buraciov, S.A.; Spinu, S.N.; Begenari, N.P.; Poirier, D.; Roy, J. Synthesis and antimicrobial activity of sulfanylamide-containing copper(II) and nickel(II) salicylidene thiosemicarbazidates. Pharm. Chem. 2007, 41, 596–599. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pahonțu, E.; Ilieș, D.-C.; Shova, S.; Paraschivescu, C.; Badea, M.; Gulea, A.; Roșu, T. Synthesis, Characterization, Crystal Structure and Antimicrobial Activity of Copper(II) Complexes with the Schiff Base Derived from 2-Hydroxy-4-Methoxybenzaldehyde. Molecules 2015, 20, 5771-5792. https://doi.org/10.3390/molecules20045771
Pahonțu E, Ilieș D-C, Shova S, Paraschivescu C, Badea M, Gulea A, Roșu T. Synthesis, Characterization, Crystal Structure and Antimicrobial Activity of Copper(II) Complexes with the Schiff Base Derived from 2-Hydroxy-4-Methoxybenzaldehyde. Molecules. 2015; 20(4):5771-5792. https://doi.org/10.3390/molecules20045771
Chicago/Turabian StylePahonțu, Elena, Diana-Carolina Ilieș, Sergiu Shova, Codruța Paraschivescu, Mihaela Badea, Aurelian Gulea, and Tudor Roșu. 2015. "Synthesis, Characterization, Crystal Structure and Antimicrobial Activity of Copper(II) Complexes with the Schiff Base Derived from 2-Hydroxy-4-Methoxybenzaldehyde" Molecules 20, no. 4: 5771-5792. https://doi.org/10.3390/molecules20045771
APA StylePahonțu, E., Ilieș, D.-C., Shova, S., Paraschivescu, C., Badea, M., Gulea, A., & Roșu, T. (2015). Synthesis, Characterization, Crystal Structure and Antimicrobial Activity of Copper(II) Complexes with the Schiff Base Derived from 2-Hydroxy-4-Methoxybenzaldehyde. Molecules, 20(4), 5771-5792. https://doi.org/10.3390/molecules20045771







