Natural Products for the Treatment of Trachoma and Chlamydia trachomatis
Abstract
:1. Introduction
1.1. Neglected Tropical Diseases
1.2. Trachoma and Chlamydia trachomatis
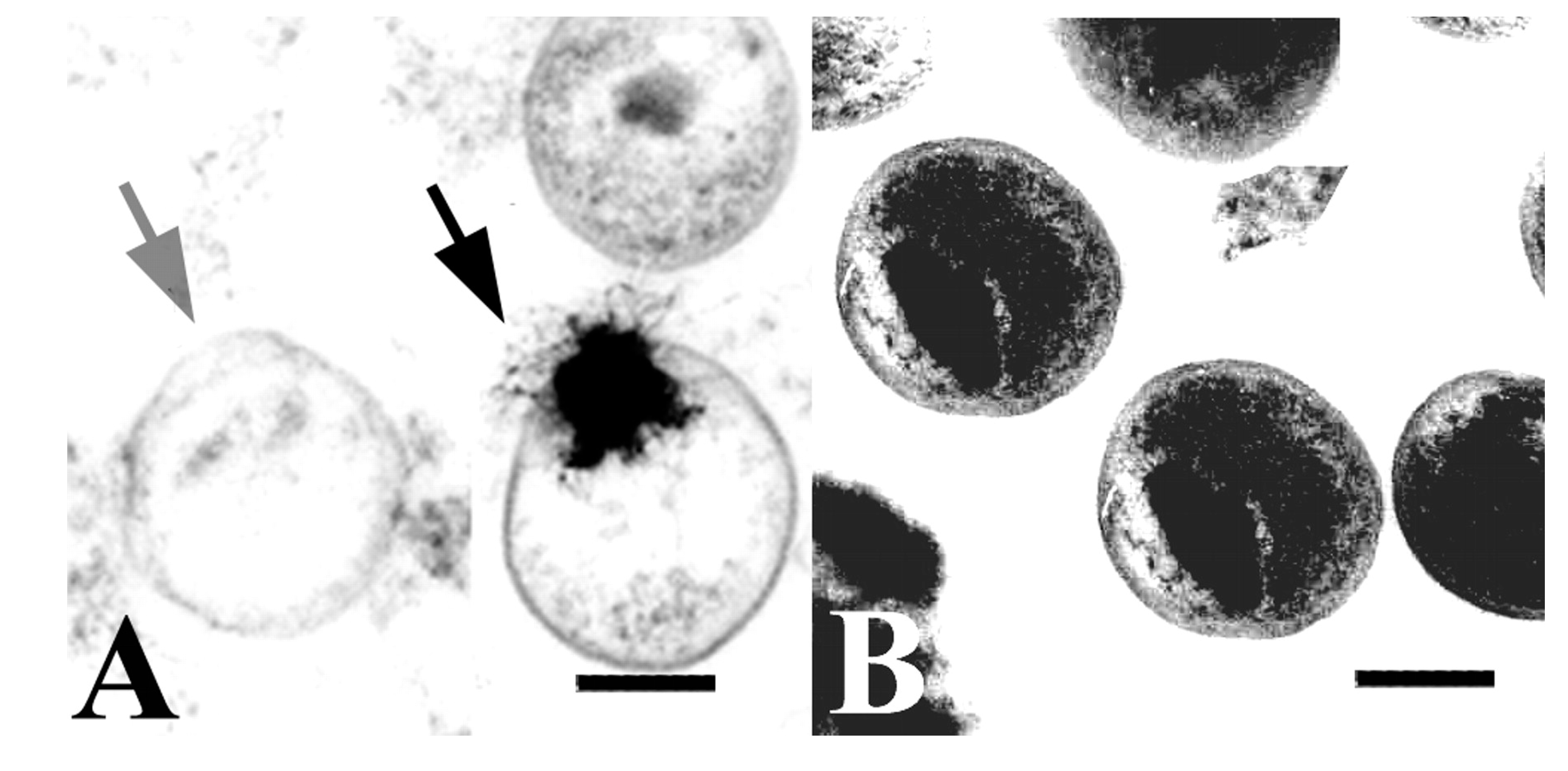
1.2.1. Chlamydial Developmental Cycle
1.2.2. Conventional Chlamydial Treatment
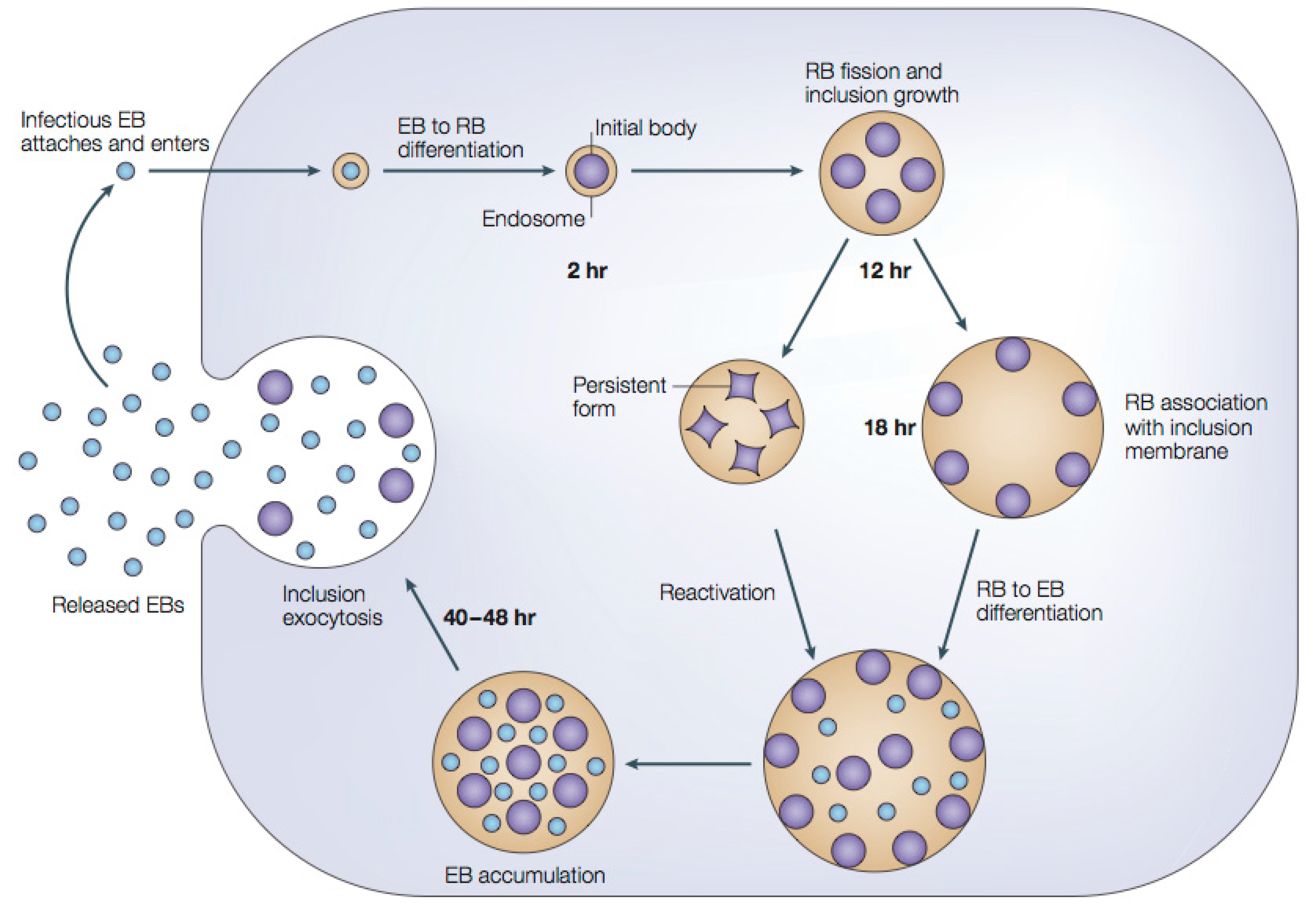
1.3. Drug Resistance and Biopharmaceuticals
2. Ethnobotanical & Traditional Medicine as Drug Leads
2.1. Traditional Medicine
2.2. Modern Herbal Formulations
2.2.1. Praneem Polyherbal Broad-Spectrum Antimicrobial Formulation
2.2.2. BASANT Polyherbal Broad-Spectrum Antimicrobial Formulation
3. Biomedical Phytochemical Groups and Anti-Infective Action
3.1. Phenols/Flavonoids
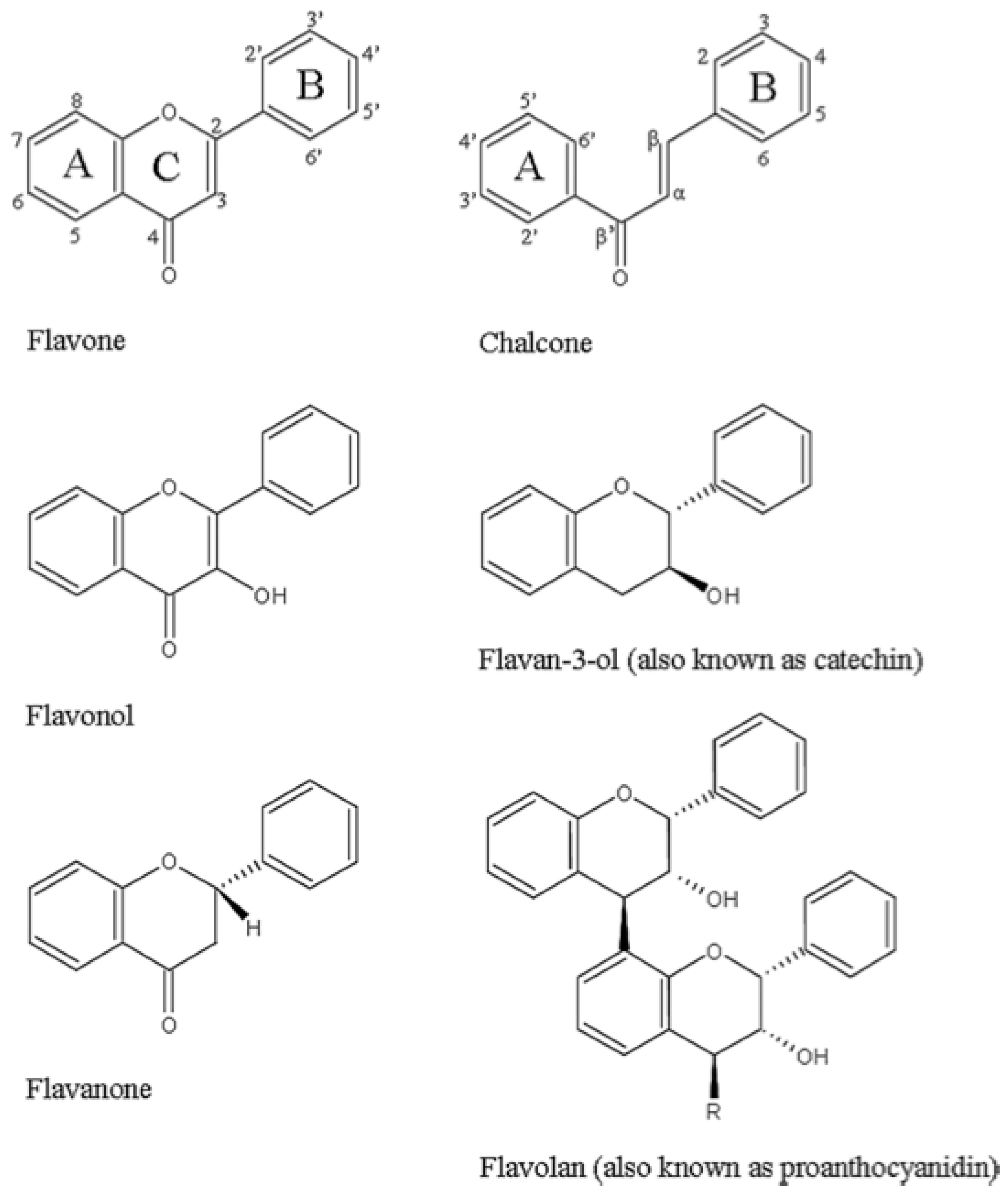
3.1.1. Catechins—Membrane Disruption
3.1.2. Luteolin—Increased Cellular Apoptosis
3.1.3. Baicalin—Improved Immune Detection
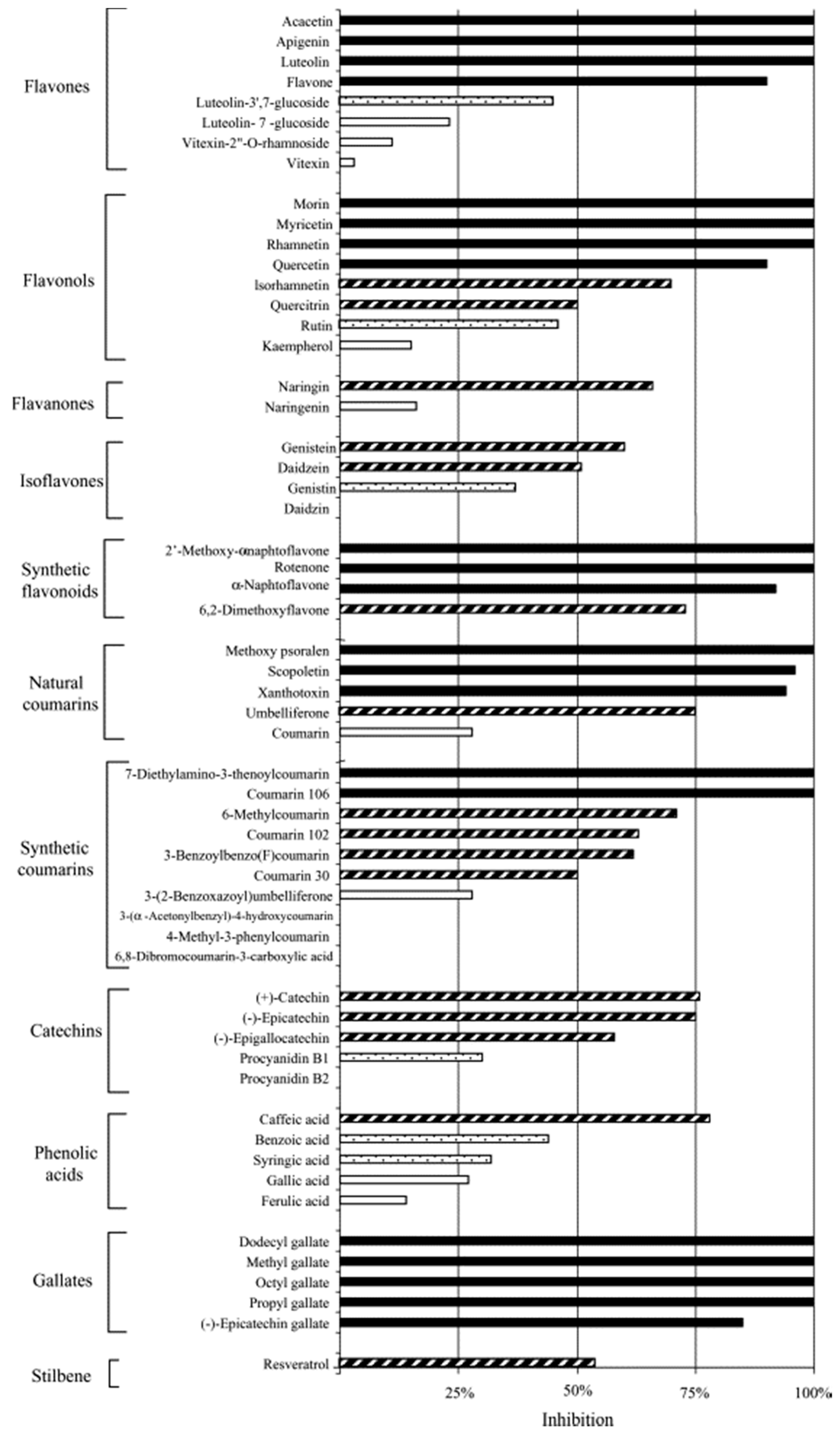
3.1.4. Polyphenols—Antichlamydial Activity and Polyphenol Structure
3.2. Lipids/Fatty Acids
3.2.1. Fatty Acids and Monoglycerides—Membrane Disruption
3.2.2. Synthetic Lipids—Membrane Disruption
3.3. Peptides
Cecropin Peptides—Membrane Disruption
4. Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Daumerie, D.; Lorenzo, S. Sustaining the Drive to Overcome the Global Impact of Neglected Tropical Diseases: Second WHO Report on Neglected Tropical Diseases; Crompton, D.W.T., Ed.; World Health Organization: Geneva, Switzerland, 2013; pp. 1–138. [Google Scholar]
- Hotez, P.J.; Yamey, G. The evolving scope of PLoS Neglected Tropical Diseases. PLoS Negl. Trop. Dis. 2009, 3, e379. [Google Scholar]
- Hotez, P.J.; Molyneux, D.H.; Fenwick, A.; Kumaresan, J.; Sachs, S.E.; Sachs, J.D.; Savioli, L. Control of neglected tropical diseases. N. Engl. J. Med. 2007, 357, 1018–1027. [Google Scholar] [CrossRef] [PubMed]
- Alvesalo, J.; Vuorela, H.; Tammela, P.; Leinonen, M.; Saikku, P.; Vuorela, P. Inhibitory effect of dietary phenolic compounds on Chlamydia pneumoniae in cell cultures. Biochem. Pharmacol. 2006, 71, 735–741. [Google Scholar] [CrossRef] [PubMed]
- Joseph, S.J.; Didelot, X.; Rothschild, J.; de Vries, H.J.; Morré, S.A.; Read, T.D.; Dean, D. Population genomics of Chlamydia trachomatis: Insights on drift, selection, recombination, and population structure. Mol. Biol. Evol. 2012, 29, 3933–3946. [Google Scholar] [CrossRef] [PubMed]
- Darougar, S.; Jones, B.R. Trachoma. Br. Med. Bull. 1983, 39, 117–122. [Google Scholar] [PubMed]
- Mandeel, Q.A.; Al-Laith, A.A.A. Ethnomycological aspects of the desert truffle among native Bahraini and non-Bahraini peoples of the Kingdom of Bahrain. J. Ethnopharmacol. 2007, 110, 118–129. [Google Scholar] [CrossRef] [PubMed]
- Hao, H.; Aixia, Y.; Nancai, Y.; Wen, S. Baicalin suppresses expression of Chlamydia protease-like activity factor in Hep-2 cells infected by Chlamydia trachomatis. Fitoterapia 2009, 80, 448–452. [Google Scholar] [CrossRef] [PubMed]
- Bhengraj, A.R.; Dar, S.A.; Talwar, G.P.; Mittal, A. Potential of a novel polyherbal formulation BASANT for prevention of Chlamydia trachomatis infection. Int. J. Antimicrob. Agents 2008, 32, 84–88. [Google Scholar] [CrossRef] [PubMed]
- Goh, B.T.; Forster, G.E. Sexually transmitted diseases in children: Chlamydial oculo-genital infection. Genitourin. Med. 1993, 69, 213. [Google Scholar]
- Bergsson, G.; Arnfinnsson, J.; Karlsson, S.M.; Steingrímsson, Ó.; Thormar, H. In vitro inactivation of Chlamydia trachomatis by fatty acids and monoglycerides. Antimicrob. Agents Chemother. 1998, 42, 2290–2294. [Google Scholar]
- Ballweber, L.M.; Jaynes, J.E.; Stamm, W.E.; Lampe, M.F. In vitro microbicidal activities of cecropin peptides D2A21 and D4E1 and gel formulations containing 0.1% to 2% D2A21 against Chlamydia trachomatis. Antimicrob. Agents Chemother. 2002, 46, 34–41. [Google Scholar] [CrossRef]
- Weström, L.; Wölner-Hanssen, P. Pathogenesis of pelvic inflammatory disease. Genitourin. Med. 1993, 69, 9. [Google Scholar]
- The University of Chicago Pediatrics Clerkship. Chlamydia of the Newborn. Available online: https://pedclerk.bsd.uchicago.edu/page/chlamydia-newborn (accessed on 15 December 2014).
- Yamazaki, T.; Inoue, M.; Sasaki, N.; Hagiwara, T.; Kishimoto, T.; Shiga, S.; Ogawa, M.; Hara, Y.; Matsumoto, T. In vitro inhibitory effects of tea polyphenols on the proliferation of Chlamydia trachomatis and Chlamydia pneumoniae. Jpn. J. Infect. Dis. 2003, 56, 143–145. [Google Scholar] [PubMed]
- Brunham, R.C.; Rey-Ladino, J. Immunology of Chlamydia infection: Implications for a Chlamydia trachomatis vaccine. Nat. Rev. Immunol. 2005, 5, 149–161. [Google Scholar] [CrossRef] [PubMed]
- Lampe, M.F.; Ballweber, L.M.; Isaacs, C.E.; Patton, D.L.; Stamm, W.E. Killing of Chlamydia trachomatis by novel antimicrobial lipids adapted from compounds in human breast milk. Antimicrob. Agents Chemother. 1998, 42, 1239–1244. [Google Scholar] [PubMed]
- Vuorela, P.; Leinonen, M.; Saikku, P.; Tammela, P.; Rauha, J.P.; Wennberg, T.; Vuorela, H. Natural products in the process of finding new drug candidates. Curr. Med. Chem. 2004, 11, 1375–1389. [Google Scholar] [CrossRef] [PubMed]
- Bailey, R.L.; Arullendran, P.; Mabey, D.C.W.; Whittle, H.C. Randomised controlled trial of single-dose azithromycin in treatment of trachoma. Lancet 1993, 342, 453–456. [Google Scholar] [CrossRef] [PubMed]
- Solomon, A.W.; Akudibillah, J.; Abugri, P.; Hagan, M.; Foster, A.; Bailey, R.L.; Mabey, D.C. Pilot study of the use of community volunteers to distribute azithromycin for trachoma control in Ghana. Bull. World Health Organ. 2001, 79, 8–14. [Google Scholar] [PubMed]
- Chait, R.; Vetsigian, K.; Kishony, R. What counters antibiotic resistance in nature? Nat. Chem. Biol. 2012, 8, 2–5. [Google Scholar] [CrossRef]
- Davies, J.; Davies, D. Origins and evolution of antibiotic resistance. Microbiol. Mol. Biol. Rev. 2010, 74, 417–433. [Google Scholar] [CrossRef] [PubMed]
- D’Costa, V.M.; King, C.E.; Kalan, L.; Morar, M.; Sung, W.W.; Schwarz, C.; Froese, D.; Zazula, G.; Calmels, F.; Debruyne, R.; et al. Antibiotic resistance is ancient. Nature 2011, 477, 457–461. [Google Scholar]
- D’Costa, V.M.; McGrann, K.M.; Hughes, D.W.; Wright, G.D. Sampling the antibiotic resistome. Science 2006, 311, 374–377. [Google Scholar] [CrossRef] [PubMed]
- Keshavjee, S.; Farmer, P.E. Tuberculosis, drug resistance, and the history of modern medicine. N. Engl. J. Med. 2012, 367, 931–936. [Google Scholar] [CrossRef] [PubMed]
- Wagner, H.; Ulrich-Merzenich, G. Synergy research: Approaching a new generation of phytopharmaceuticals. Phytomedicine 2009, 16, 97–110. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.K.; Kumar, S. Perspective on plant products as antimicrobial agents: A review. Pharmacologia 2013, 4, 469–480. [Google Scholar] [CrossRef]
- Semenya, S.S.; Potgieter, M.J.; Erasmus, L.J.C. Bapedi phytomedicine and their use in the treatment of sexually transmitted infections in Limpopo Province, South Africa. Afr. J. Pharm. Pharmacol. 2013, 7, 250–262. [Google Scholar] [CrossRef]
- Vermani, K.; Garg, S. Herbal medicines for sexually transmitted diseases and AIDS. J. Ethnopharmacol. 2002, 80, 49–66. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.W.; Hao, X.; Chen, K. Biological screening of natural products and drug innovation in China. Philos. Trans. R. Soc. B Biol. Sci. 2007, 362, 1093–1105. [Google Scholar] [CrossRef]
- Al-Marzooky, M.A. Truffles in eye disease. Proc. Int. Islam. Med. 1981, 1, 353–357. [Google Scholar]
- Chellal, A.; Lukasova, E. Evidence for antibiotics in two Algerien truffles Terfezia and Tirmania. Pharmazie 1995, 50, 228–229. [Google Scholar] [PubMed]
- Janakat, S.; Al‐Fakhiri, S.; Sallal, A.K. A promising peptide antibiotic from Terfezia claveryi aqueous extract against Staphylococcus aureus in vitro. Phytother. Res. 2004, 18, 810–813. [Google Scholar] [CrossRef] [PubMed]
- Janakat, S.M.; Al-Fakhiri, S.M.; Sallal, A.K.J. Evaluation of antibacterial activity of aqueous and methanolic extracts of the truffle Terfezia claveryi against Pseudomonas aeruginosa. Saudi Med. J. 2005, 26, 952–955. [Google Scholar] [PubMed]
- Li, R.W.; Myers, S.P.; Leach, D.N.; Lin, G.D.; Leach, G. A cross-cultural study: Anti-inflammatory activity of Australian and Chinese plants. J. Ethnopharmacol. 2003, 85, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Adedapo, A.A.; Jimoh, F.O.; Koduru, S.; Afolayan, A.J.; Masika, P.J. Antibacterial and antioxidant properties of the methanol extracts of the leaves and stems of Calpurnia aurea. BMC Complement. Altern. Med. 2008, 8, 53. [Google Scholar] [PubMed]
- Rani, M.S.; Pippalla, R.S.; Mohan, K. Dodonaea viscosa Linn.—An overview. Asian J. Pharm. Res. Health Care 2009, 1, 97–112. [Google Scholar]
- Yenesew, A.; Twinomuhwezi, H.; Kiremire, B.T.; Mbugua, M.N.; Gitu, P.M.; Heydenreich, M.; Peter, M.G. 8-Methoxyneorautenol and radical scavenging flavonoids from Erythrina abyssinica. Bull. Chem. Soc. Ethiop. 2009, 23, 205–210. [Google Scholar]
- Waffo, A.K.; Azebaze, G.A.; Nkengfack, A.E.; Fomum, Z.T.; Meyer, M.; Bodo, B.; van Heerden, F.R. Indicanines B and C, two isoflavonoid derivatives from the root bark of Erythrina indica. Phytochemistry 2000, 53, 981–985. [Google Scholar] [CrossRef] [PubMed]
- Adewumi, A.A.J.; Yahaya, H.K.; Aina, V.O.; Olorunmaiye, B.O.; Abdulsalami, M.S. Phytochemical screening and antibacterial activity of Jatropha curcas seeds and stem extractions on some wound pathogens. J. Pharm. Allied Sci. 2014, 10, 1852–1862. [Google Scholar]
- Jaberian, H.; Piri, K.; Nazari, J. Phytochemical composition and in vitro antimicrobial and antioxidant activities of some medicinal plants. Food Chem. 2013, 136, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Talwar, G.P.; Garg, S.; Dhar, V.; Chabra, R.; Ganju, A.; Upadhyay, S.N. Praneem polyherbal cream and pessaries with dual properties of contraception and alleviation of genital infections. Curr. Sci. 1995, 68, 437–440. [Google Scholar]
- Bhengraj, A.R.; Goyal, A.; Talwar, G.P.; Mittal, A. Assessment of antichlamydial effects of a novel polyherbal tablet Basant. Sex. Transm. Infect. 2009, 85, 561–5621. [Google Scholar] [CrossRef] [PubMed]
- Törmäkangas, L.; Vuorela, P.; Saario, E.; Leinonen, M.; Saikku, P.; Vuorela, H. In vivo treatment of acute Chlamydia pneumoniae infection with the flavonoids quercetin and luteolin and an alkyl gallate, octyl gallate, in a mouse model. Biochem. Pharmacol. 2005, 70, 1222–1230. [Google Scholar] [CrossRef] [PubMed]
- Cushnie, T.P.; Lamb, A.J. Recent advances in understanding the antibacterial properties of flavonoids. Int. J. Antimicrob. Agents 2011, 38, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.Z.; Harnly, J.M. A screening method for the identification of glycosylated flavonoids and other phenolic compounds using a standard analytical approach for all plant materials. J. Agric. Food Chem. 2007, 55, 1084–1096. [Google Scholar] [CrossRef] [PubMed]
- Ikigai, H.; Nakae, T.; Hara, Y.; Shimamura, T. Bactericidal catechins damage the lipid bilayer. Biochim. Biophys. Acta BBA Biomembr. 1993, 1147, 132–136. [Google Scholar] [CrossRef]
- Nakayama, M.; Toda, M.; Okubo, S.; Shimamura, T. Inhibition of influenza virus infection by tea. Lett. Appl. Microbiol. 1990, 11, 38–40. [Google Scholar] [CrossRef]
- Nakayama, M.; Suzuki, K.; Toda, M.; Okubo, S.; Hara, Y.; Shimamura, T. Inhibition of the infectivity of influenza virus by tea polyphenols. Antivir. Res. 1993, 21, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Toda, M.; Okubo, S.; Ikigai, H.; Suzuki, T.; Suzuki, Y.; Shimamura, T. The protective activity of tea against infection by Vibrio cholerae O1. J. Appl. Bacteriol. 1991, 70, 109–112. [Google Scholar] [CrossRef] [PubMed]
- Yam, T.S.; Hamilton-Miller, J.M.; Shah, S. The effect of a component of tea (Camellia sinensis) on methicillin resistance, PBP2' synthesis, and beta-lactamase production in Staphylococcus aureus. J. Antimicrob. Chemother. 1998, 42, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Diker, K.S.; Akan, M.; Hascelik, G.; Yurdakök, M. The bactericidal activity of tea against Campylobacter jejuni and Campylobacter coli. Lett. Appl. Microbiol. 1991, 12, 34–35. [Google Scholar] [CrossRef]
- Xagorari, A.; Papapetropoulos, A.; Mauromatis, A.; Economou, M.; Fotsis, T.; Roussos, C. Luteolin inhibits an endotoxin-stimulated phosphorylation cascade and proinflammatory cytokine production in macrophages. J. Pharmacol. Exp. Ther. 2001, 296, 181–187. [Google Scholar] [PubMed]
- Kotanidou, A.; Xagorari, A.; Bagli, E.; Kitsanta, P.; Fotsis, T.; Papapetropoulos, A.; Roussos, C. Luteolin reduces lipopolysaccharide-induced lethal toxicity and expression of proinflammatory molecules in mice. Am. J. Respir. Crit. Care Med. 2002, 165, 818–823. [Google Scholar] [CrossRef] [PubMed]
- Cheng, A.C.; Huang, T.C.; Lai, C.S.; Pan, M.H. Induction of apoptosis by luteolin through cleavage of Bcl-2 family in human leukemia HL-60 cells. Eur. J. Pharmacol. 2005, 509, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Fischer, S.F.; Harlander, T.; Vier, J.; Häcker, G. Protection against CD95-induced apoptosis by chlamydial infection at a mitochondrial step. Infect. Immun. 2004, 72, 1107–1115. [Google Scholar] [CrossRef] [PubMed]
- Kuang, Z.; Fu, L.; Wo, H.; Zhang, S. Exploring the Mechanism of Baicalin in Influencing Cells Infected with Chlamydia Pneumoniae by Observing Its Effect on Receptors. J. Guangzhou Univers. Tradit. Chin. Med. 2005, 3, 014. [Google Scholar]
- Liu, Y.D.; Wu, W.; Wang, S.; Kuang, Z.Y.; Li, R.; Huang, Y.S. The influence of Baicalin on the level of Serum TNF-α, IL-6, IL-10 in Diet-induced Hyperlipidemic and CPn infected Mice. Sichuan Zhong Yi 2006, 24, 19–20. [Google Scholar]
- Zhong, G.; Fan, P.; Ji, H.; Dong, F.; Huang, Y. Identification of a chlamydial protease–like activity factor responsible for the degradation of host transcription factors. J. Exp. Med. 2001, 193, 935–942. [Google Scholar] [CrossRef] [PubMed]
- Fan, P.; Dong, F.; Huang, Y.; Zhong, G. Chlamydia pneumoniae secretion of a protease-like activity factor for degrading host cell transcription factors is required for major histocompatibility complex antigen expression. Infect. Immun. 2002, 70, 345–349. [Google Scholar] [CrossRef] [PubMed]
- Stephens, R.S. The cellular paradigm of chlamydial pathogenesis. Trends Microbiol. 2003, 11, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, S.J.; Eckmann, L.; Quayle, A.J.; Shen, L.; Zhang, Y.X.; Anderson, D.J.; Fierer, J.; Stephens, R.S.; Kagnoff, M.F. Secretion of proinflammatory cytokines by epithelial cells in response to Chlamydia infection suggests a central role for epithelial cells in chlamydial pathogenesis. J. Clin. Investig. 1997, 99, 77. [Google Scholar] [PubMed]
- Thormar, H. Antibacterial effects of lipids: Historical review (1881 to 1960). Lipids Essent. Oils 2011, 25. [Google Scholar] [CrossRef]
- Thormar, H.; Isaacs, C.E.; Brown, H.R.; Barshatzky, M.R.; Pessolano, T. Inactivation of enveloped viruses and killing of cells by fatty acids and monoglycerides. Antimicrob. Agents Chemother. 1987, 31, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Isaacs, C.E.; Kashyap, S.; Heird, W.C.; Thormar, H. Antiviral and antibacterial lipids in human milk and infant formula feeds. Arch. Dis. Child. 1990, 65, 861–864. [Google Scholar] [CrossRef] [PubMed]
- Bergsson, G.; Hilmarsson, H.; Thormar, H. Antibacterial, antiviral and antifungal activities of lipids. Lipids Essent. Oils Antimicrob. Agents 2011, 47–80. [Google Scholar] [CrossRef]
- Zasloff, M. Antimicrobial peptides of multicellular organisms. Nature 2002, 415, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Boman, H.G. Gene-encoded peptide antibiotics and the concept of innate immunity: An update review. Scand. J. Immunol. 1998, 48, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Bechinger, B. Structure and functions of channel-forming peptides: Magainins, cecropins, melittin and alamethicin. J. Membr. Biol. 1997, 156, 197–211. [Google Scholar] [CrossRef] [PubMed]
- Saberwal, G.; Nagaraj, R. Cell-lytic and antibacterial peptides that act by perturbing the barrier function of membranes: Facets of their conformational features, structure-function correlations and membrane-perturbing abilities. Biochim. Biophys. Acta BBA Rev. Biomembr. 1994, 1197, 109–131. [Google Scholar] [CrossRef]
- Hendrich, A.B. Flavonoid‐membrane interactions: Possible consequences for biological effects of some polyphenolic compounds. Acta Pharmacol. Sin. 2006, 27, 27–40. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Potroz, M.G.; Cho, N.-J. Natural Products for the Treatment of Trachoma and Chlamydia trachomatis. Molecules 2015, 20, 4180-4203. https://doi.org/10.3390/molecules20034180
Potroz MG, Cho N-J. Natural Products for the Treatment of Trachoma and Chlamydia trachomatis. Molecules. 2015; 20(3):4180-4203. https://doi.org/10.3390/molecules20034180
Chicago/Turabian StylePotroz, Michael G., and Nam-Joon Cho. 2015. "Natural Products for the Treatment of Trachoma and Chlamydia trachomatis" Molecules 20, no. 3: 4180-4203. https://doi.org/10.3390/molecules20034180
APA StylePotroz, M. G., & Cho, N.-J. (2015). Natural Products for the Treatment of Trachoma and Chlamydia trachomatis. Molecules, 20(3), 4180-4203. https://doi.org/10.3390/molecules20034180






