Studies on the Low-Temp Oxidation of Coal Containing Organic Sulfur and the Corresponding Model Compounds
Abstract
:1. Introduction
2. The Result and Discussion
2.1. The Result of Experiments with Organic Sulfur Model Compounds
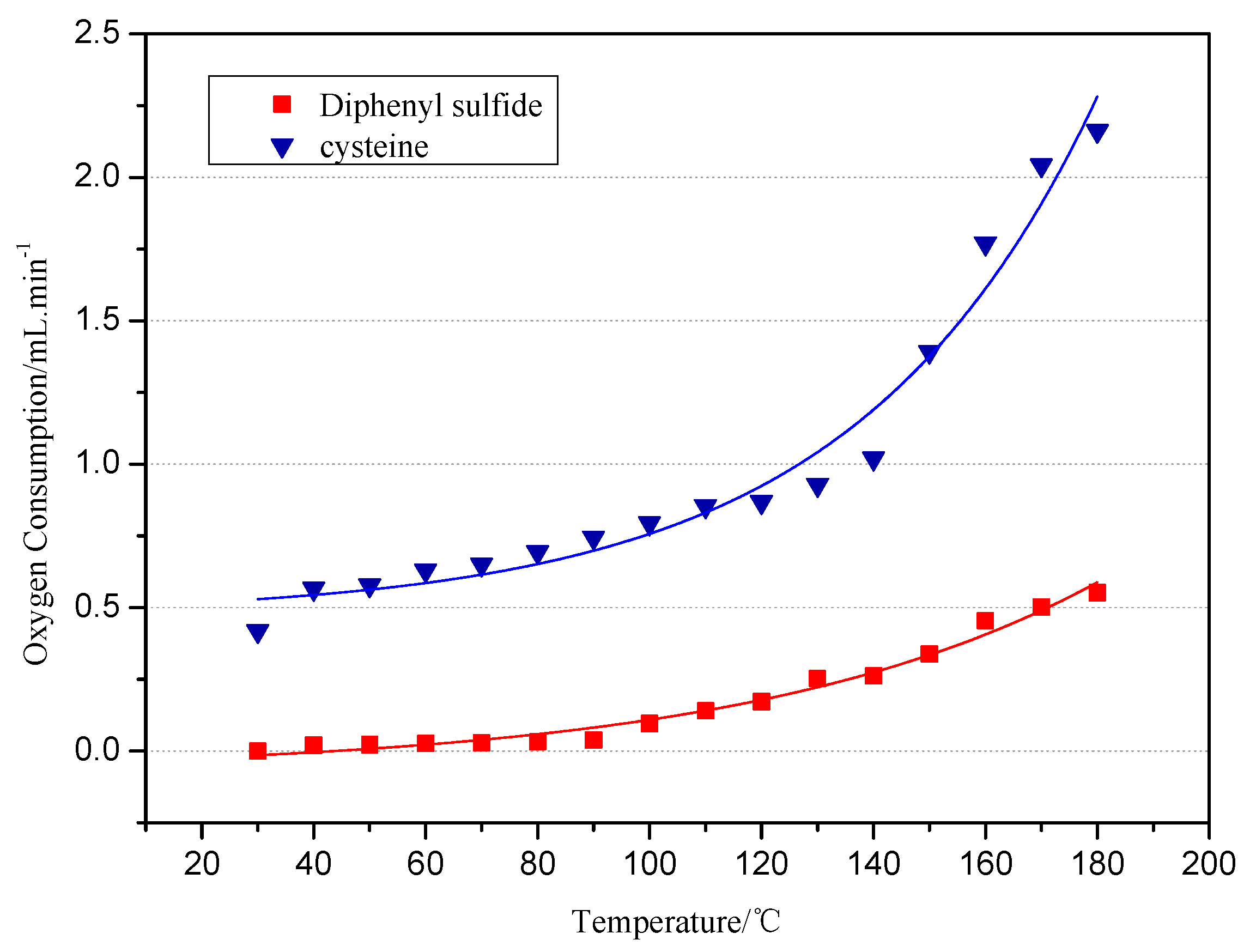
2.1.1. The Oxygen Consumption of Model Compounds during Low-Temp Oxidation
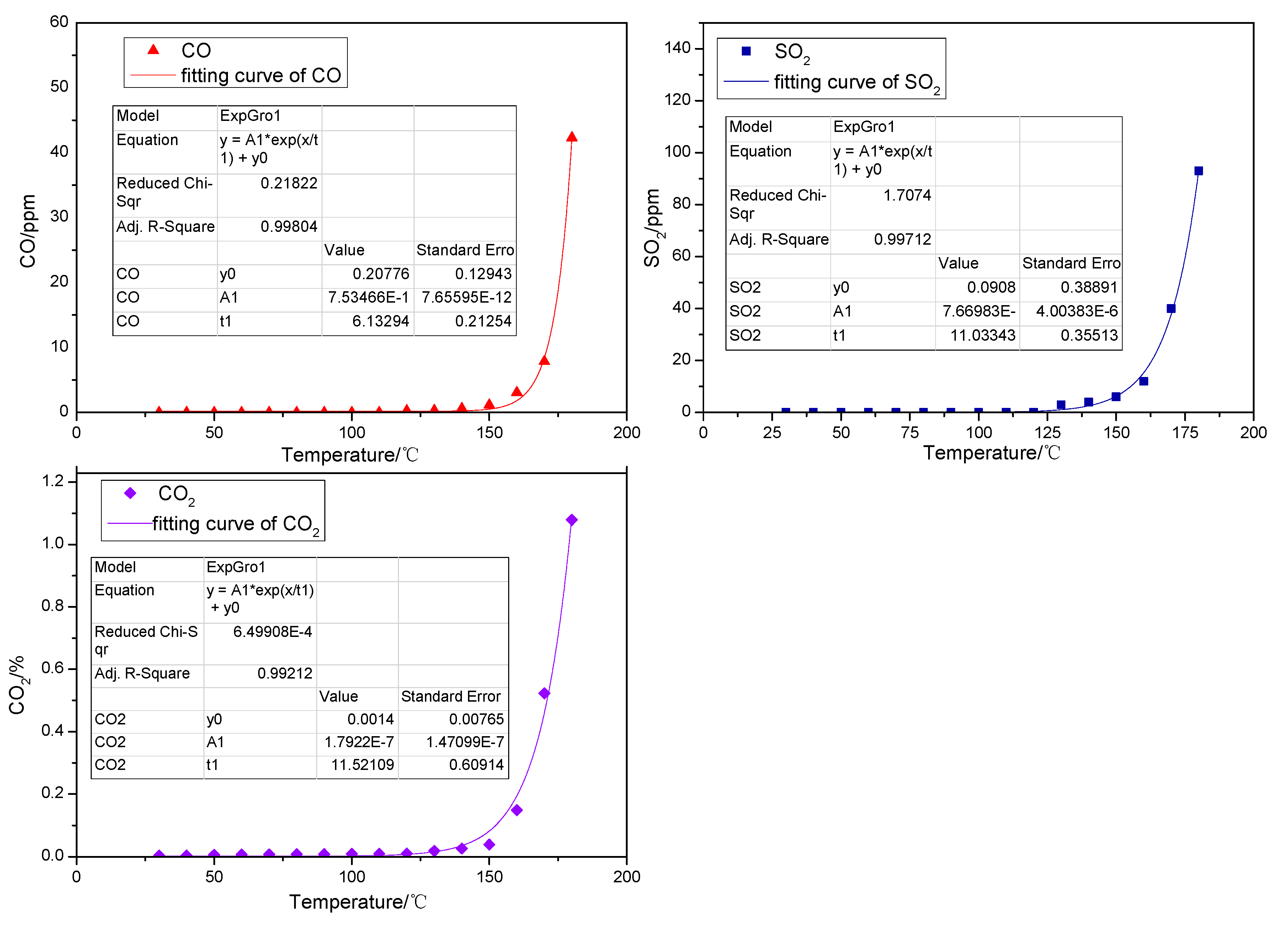
2.1.2. Gaseous Products Generated in the Low-Temp Oxidation Process of Organic Sulfur Model Compounds
2.1.3. GC/MS Analysis Results of Organic Sulfur Model Compounds
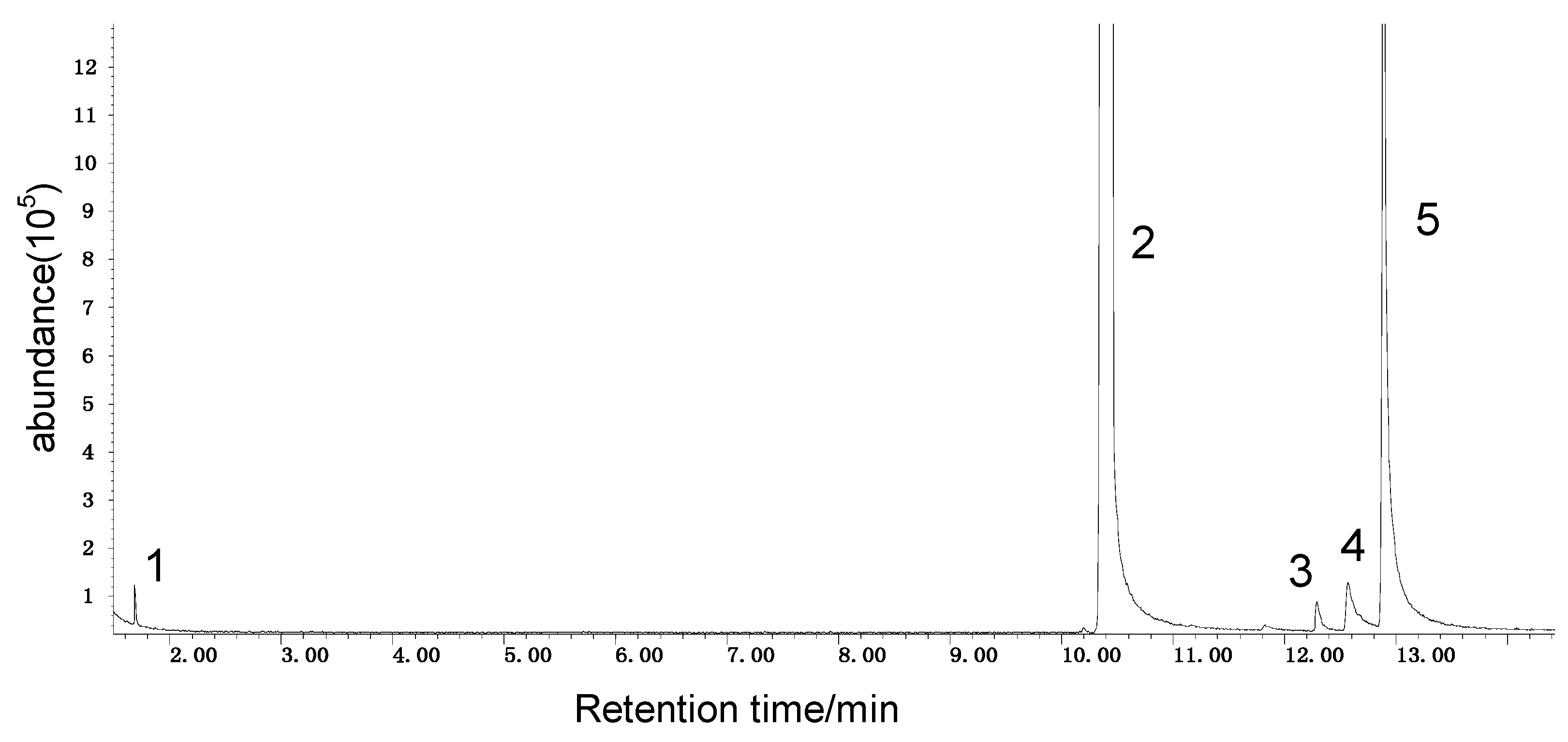
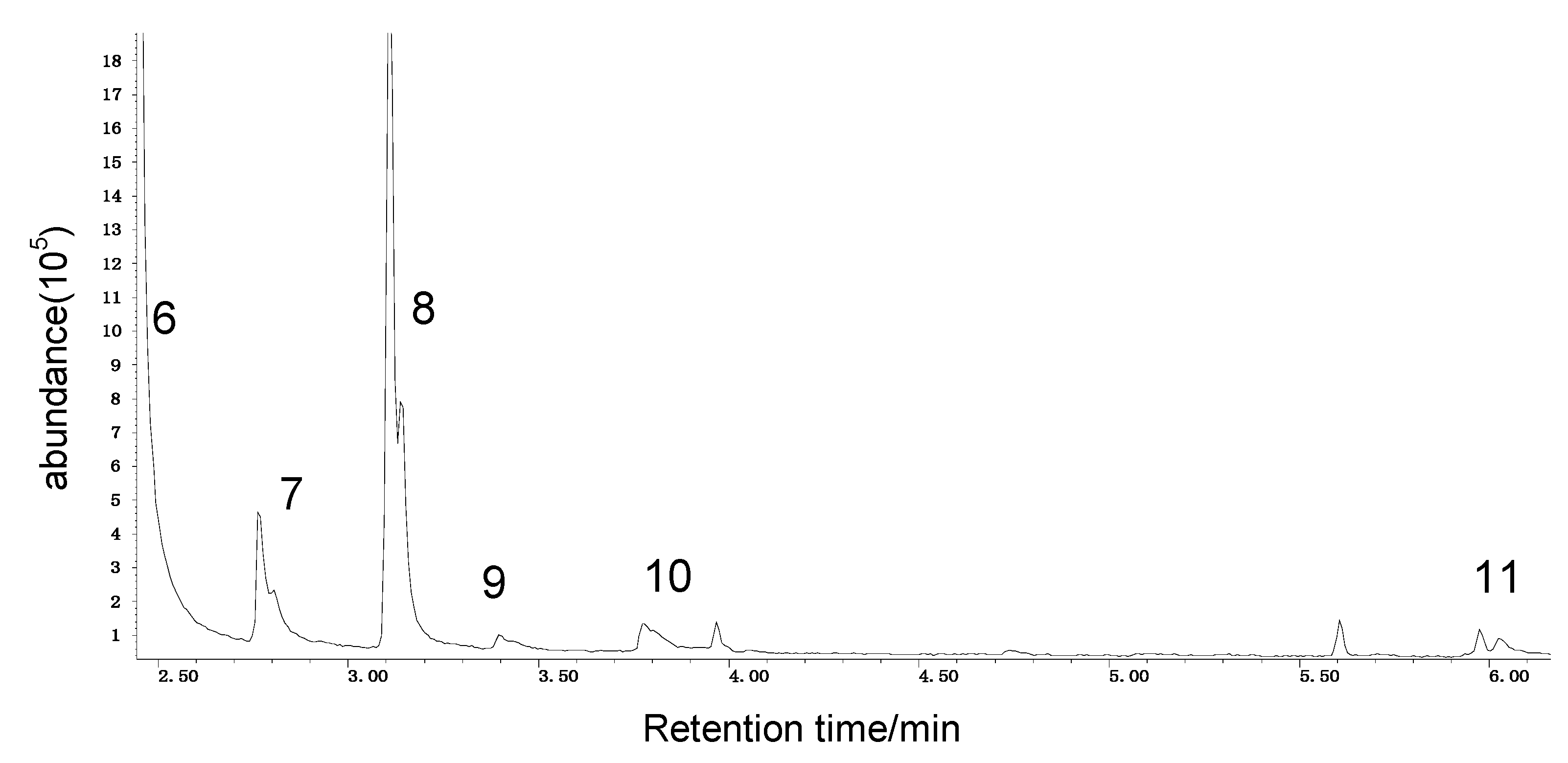
| Peak Number | Chemical Name | Structural Formula | Peak Number | Chemical Name | Structural Formula |
|---|---|---|---|---|---|
| 1 | benzene |  | 7 | diacetone alcohol | 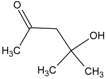 |
| 2 | diphenyl sulfane |  | 8 | 5-hexen-2-one |  |
| 3 | diphenyl disulfide |  | 9 | 4-methoxy-4-methyl-2-pentanone | 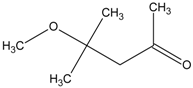 |
| 4 | diphenyl sulfoxide | 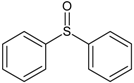 | 10 | 4-mercapto-4-methyl-2-pentanone | 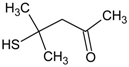 |
| 5 | diphenyl sulfone |  | 11 | 3,3,5,5-tetramethyl-1,2,4-trithiolane |  |
| 6 | mesityl oxide |  | _ | ________ | _____________ |
2.1.4. The Low-Temp Oxidation of Model Compounds Containing Organic Sulfur
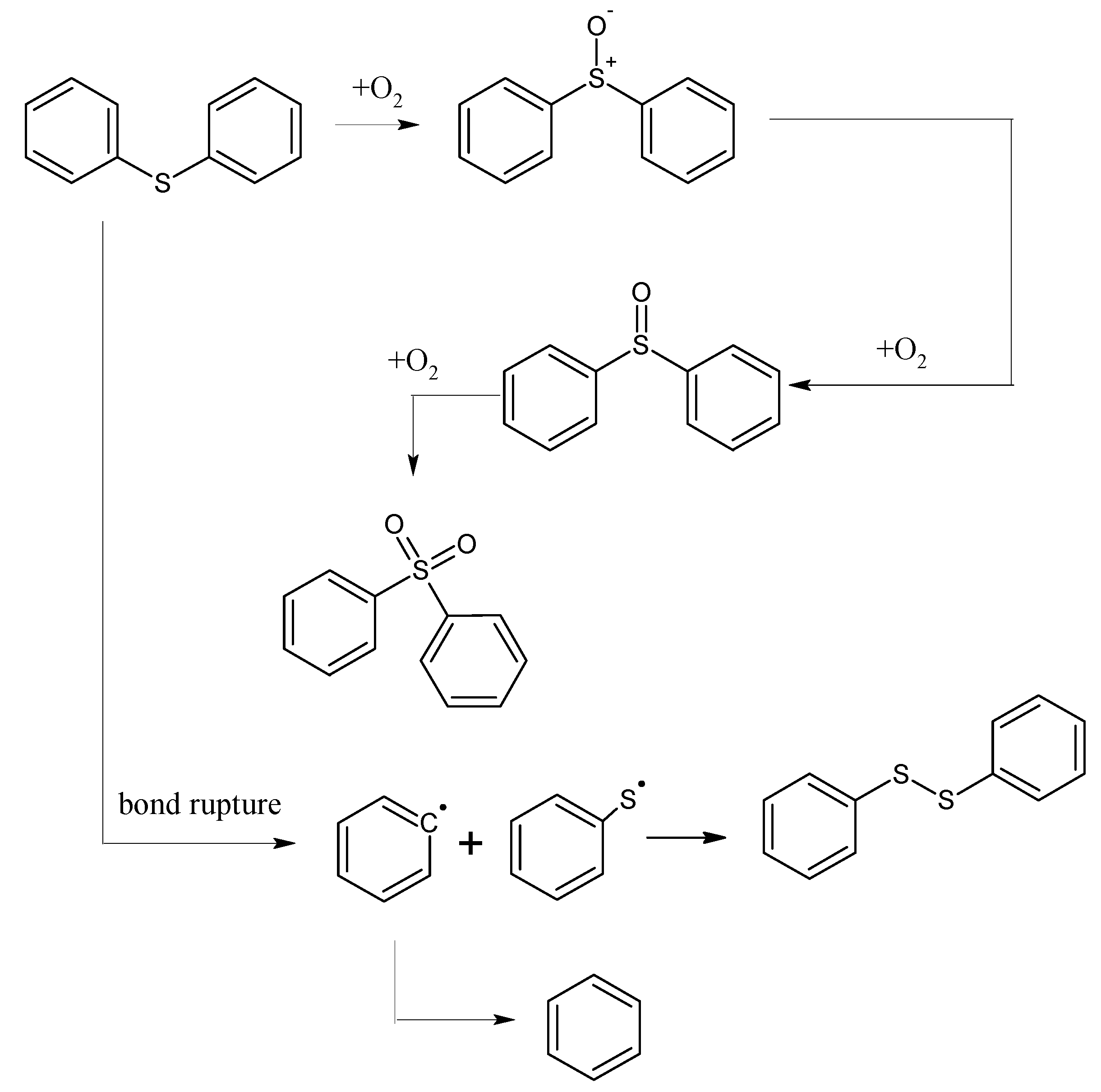
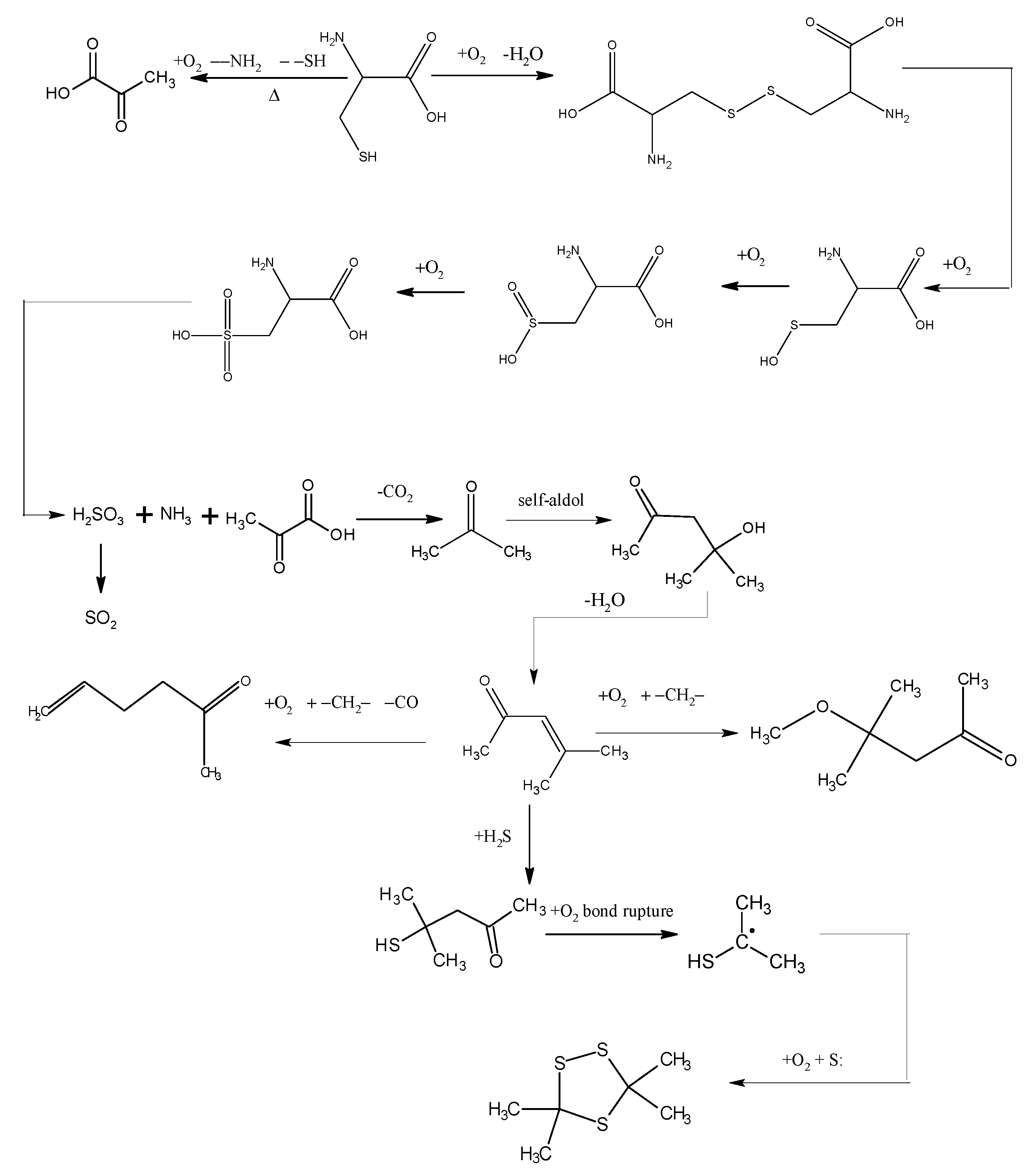
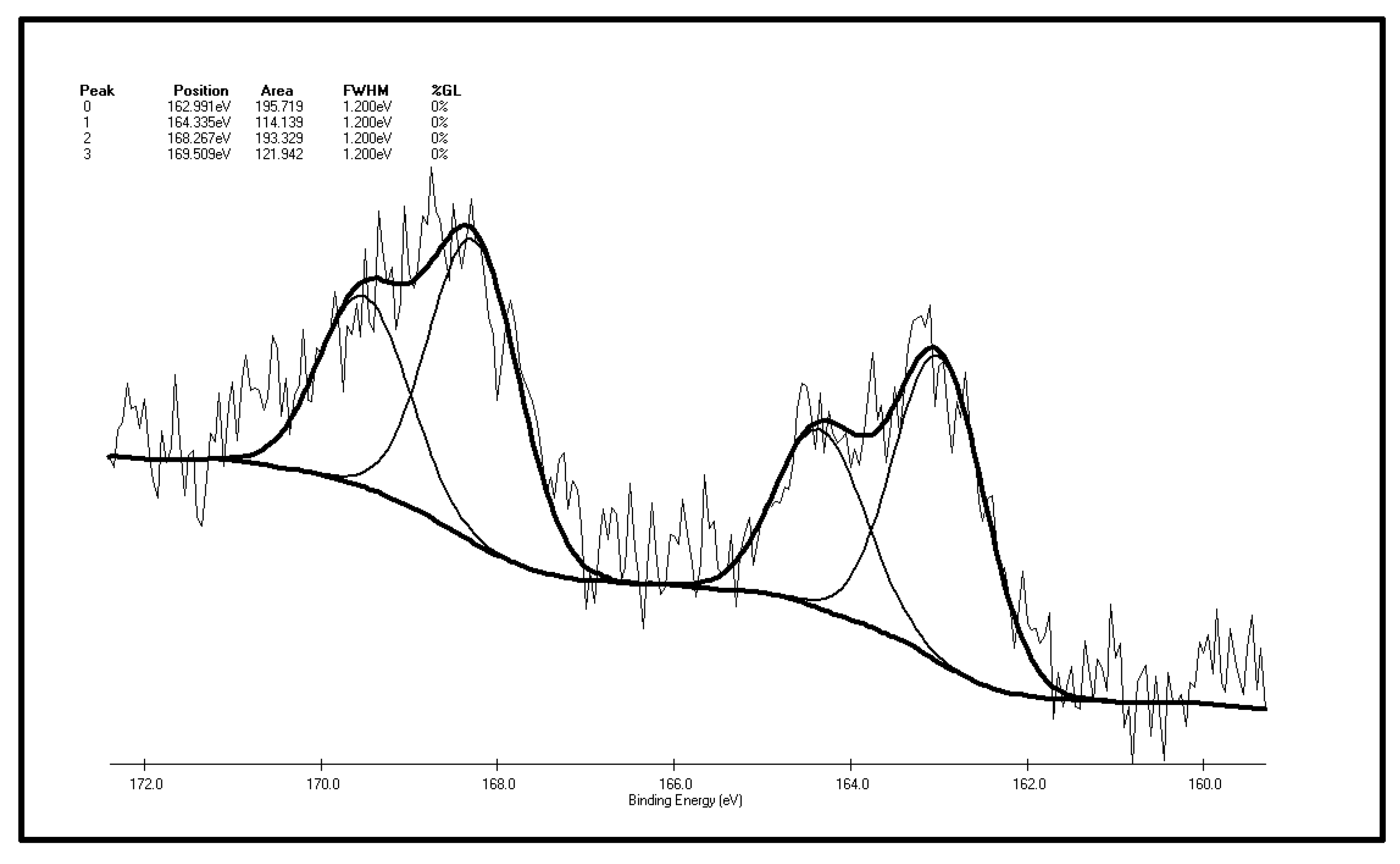
2.2. The XPS Results Before and After the Low-Temp Oxidation of Coal Samples
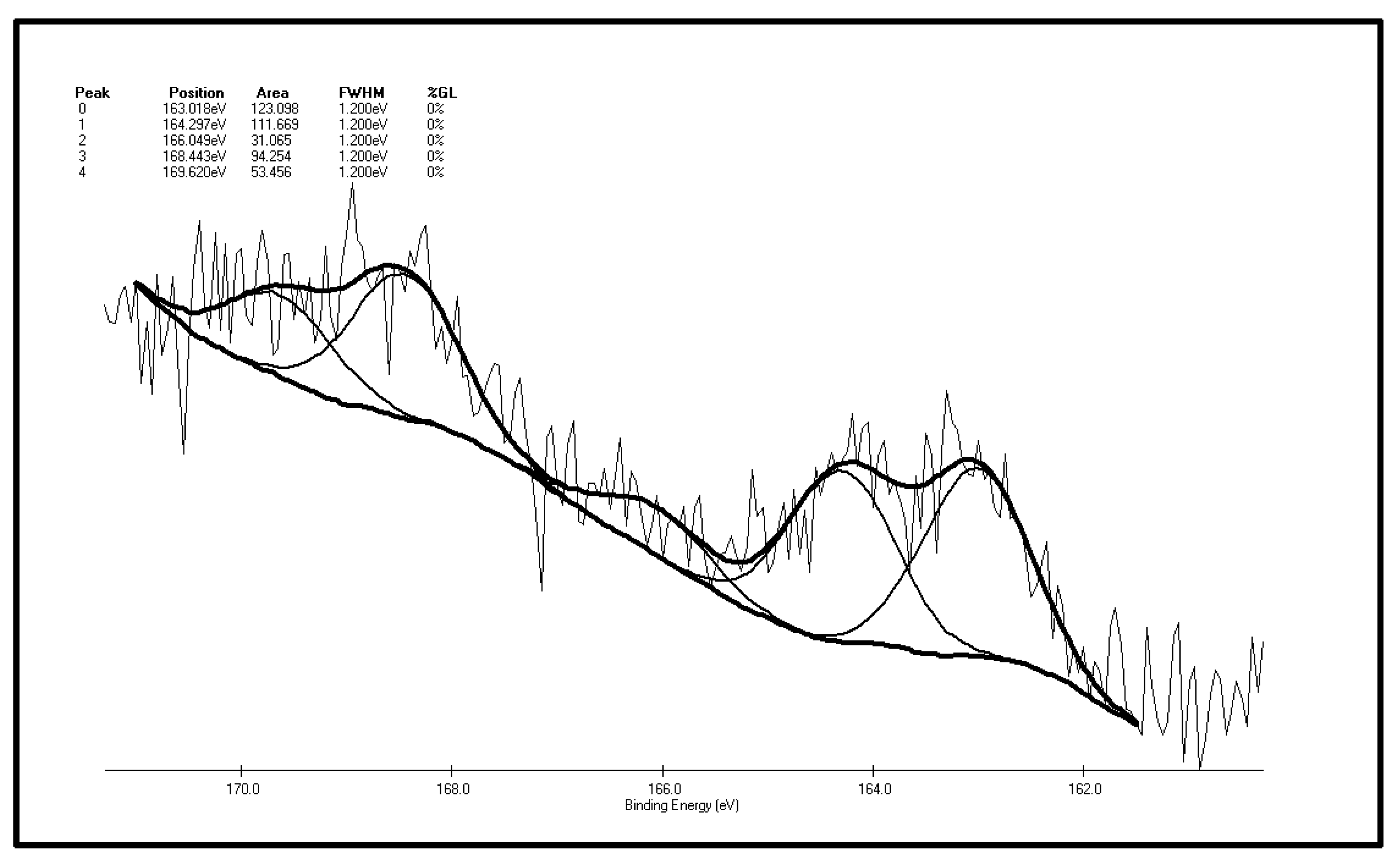
| Peak | Sulfur Form | Postion | Area | FWHM (Ev) | %GL (%) | W (%) |
|---|---|---|---|---|---|---|
| 0 | Pyritic sulfur + sulphide | 163.018 | 123.098 | 1.2 | 0 | 29.77 |
| 1 | Thiophene | 164.297 | 111.669 | 1.2 | 0 | 27.00 |
| 2 | Sulfoxide | 166.049 | 31.065 | 1.2 | 0 | 7.51 |
| 3 | Sulphone | 168.443 | 94.254 | 1.2 | 0 | 22.79 |
| 4 | Sulfate | 169.620 | 53.456 | 1.2 | 0 | 12.93 |
| Peak | Sulfur Form | Postion | Area | FWHM (Ev) | %GL (%) | W (%) |
|---|---|---|---|---|---|---|
| 0 | Pyritic sulfur + sulphide | 162.991 | 195.719 | 1.2 | 0 | 31.31 |
| 1 | Thiophene | 164.335 | 114.139 | 1.2 | 0 | 18.26 |
| 2 | Sulphone | 168.267 | 193.329 | 1.2 | 0 | 30.93 |
| 3 | Sulfate | 169.509 | 121.942 | 1.2 | 0 | 19.51 |
3. Experimental Section
3.1. Low-Temp Oxidation Experimental Facilities

3.2. Preparation of Laboratory Samples
| Model Compound | Structural Formula | Physical and Chemical Properties |
|---|---|---|
| Diphenyl Sulfide |  | Molecular weight 186.27, colorless or light yellow liquid, malodorous |
| Cysteine | 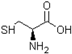 | Molecular weight 121.15, colorless crystals |

3.3. Experimental Procedures
3.4. XPS Experimental Samples and Handling
| Proximate Analysis (wt % as Received) | Ultimate Analysis (wt % Daf) | |||||||
|---|---|---|---|---|---|---|---|---|
| Mad | Aad | Vdaf | FCd | St.d | Odaf | Cdaf | Hdaf | Ndaf |
| 1.39 | 16.61 | 40.17 | 49.89 | 2.39 | 7.11 | 83.11 | 5.47 | 1.43 |
| Forms of Sulfur (wt % db) | |||
|---|---|---|---|
| Content | Pyritic | Sulfate | Organic |
| Absolute (wt %) | 1.07 | 0.03 | 1.29 |
| Relative (wt %) | 44.77 | 1.26 | 53.97 |
4. Conclusions
- (1)
- From 30 °C to 80 °C, the adsorption between diphenyl sulfide and oxygen is mainly physical, at which time both the oxygen consumption and the change in the oxygen concentration are small. However, after 80 °C, chemical adsorption and reactions begin to take place and oxygen consumption increases, then diphenyl sulfoxide is generated in the reaction and finally oxidized to diphenyl sulphone. This process is the main reaction mechanism in low-temp oxidation of the model compound diphenyl sulfide. Besides, some free radicals emerge.
- (2)
- The reducibility of the sulphydryl group in cysteine determines that cysteine can be oxidized to cystine, a kind of disulfide, in low-temperature air. This reaction, which is also a common biochemical reaction, can take place under mild conditions. Cystine, a disulfide, continues to be oxidized in air to sulfenic acid, sulfinic acid and sulfonic acid, among which sulfonic acid generates pyruvic acid, sulfurous acid and ammonia through desulfonation and deamination. Then the sulfurous acid is decomposed into SO2 at a temperature of 130 °C. Besides, C–S sulphydryl bond cleavage can also occur, leading to the generation of H2S gas, while S–S bond and C–S bond cleavage in disulfides may also produce sulfur free radicals.
- (3)
- There are five fitting S 2p peaks on the surface of XF original coal samples. The major inorganic sulfur forms are pyrite and sulfate and the major forms of organic sulfur are thiophene and sulfone. It also contains traces of sulfoxide and sulfide. In accordance with the low-temp oxidation mechanism of the above two model compounds, it can be inferred that there is none or little mercaptan and thiophenol in the coal. Differences exist between the XPS results and chemical analysis results, the reason for which is that the pyrite in the coal particle surface is easily oxidized to sulfate, leading to a much higher content of in the coal particle surface than the bulk phase content of coal samples.
- (4)
- There are four fitting S 2p peaks according to XPS analysis of the oxidized XF sample. The main forms of inorganic sulfur on the surface are still pyrite and sulfate. The major organic sulfur component is sulfone, followed by thiophene sulfur. Sulfoxide is not detected on the coal surface. By comparing the XPS peak parameters of sulfur S 2p before and after oxidation, it can be found that sulfide and pyrite sulfur increased slightly, thiophenic sulfur is reduced, there is no sulfoxide detected on the surface of the coal after oxidation, yet the content of sulfone increases, and the sulfate content also increases too. This phenomenon shows that the form of organic sulfur in the coal particle surface changes after low-temp oxidation. To be more specific, sulfide sulfur is oxidized to sulfoxide, and then this sulfoxide is further oxidized to sulfone. The sulfoxide oxidization step to sulfone can be easily carried out. Sulfur peak intensity and area of the oxidized sample are higher than in the original one, which means there is a sulfur element enrichment on the surface of coal during oxidation.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lin, M.; Huggins, F.; Huffman, G. A correlative investigation of the effects of oxidation on the minerals, macerals and technological properties of coal. Am. Chem. Soc. Fuel Chem. Div. 1983, 4, 1196–1202. [Google Scholar]
- Yang, Y.; Li, Z.; Tang, Y.; Liu, Z.; Ji, H. Fine coal covering for preventing spontaneous combustion of coal pile. Nat. Hazards 2014, 74, 603–622. [Google Scholar] [CrossRef]
- Yang, Y.; Li, Z.; Tang, Y.; Gu, F.; Ji, H.; Liu, Z. Effects of low molecular weight compounds in coal on the characteristics of its spontaneous combustion. Can. J. Chem. Eng. 2015, 93, 648–657. [Google Scholar] [CrossRef]
- He, J.; Song, S.; Sun, Z.; Xu, W. The distribution of sulfur in coal and the developing direction for desulphurization in China. Met. Ore Dress. Abroad 1999, 5, 30–33. [Google Scholar]
- Calkins, W. The chemical forms of sulfur in coal: A review. Fuel 1994, 94, 475–48. [Google Scholar] [CrossRef]
- Borah, D.; Baruah, M.K.; Haque, I. Oxidation of high sulphur coal. Part 1. Desulphurisation and evidence of the formation of oxidised organic sulphur species. Fuel 2001, 2001, 501–507. [Google Scholar] [CrossRef]
- Attar, A. Chemistry, thermodynamics and kinetics of reactions of sulphur in coal-gas reactions: A review. Fuel 1978, 57, 201–212. [Google Scholar] [CrossRef]
- Huffman, G.; Huggins, F.; Dunmyre, G.; Pignocco, A.; Lin, M. Comparative sensitivity of various analytical techniques to the low-temperature oxidation of coal. Fuel 1985, 64, 849–856. [Google Scholar] [CrossRef]
- Huggins, F.E.; Huffman, G.P.; Lin, M.C. Observations on low-temperature oxidation of minerals in bituminous coals. Int. J. Coal Geol. 1983, 3, 157–182. [Google Scholar] [CrossRef]
- Nordstorm, D.K. Aqueous pyrite oxidation and the consequent formation of secondary iron minerals. In Acid Sulfate Weathering; Kittrick, J.A., Fanning, D.S., Hossner, L.R., Eds.; Soil Science Society of America, Inc.: Madison, WI, USA, 1982; Volume 18, pp. 37–56. [Google Scholar]
- Winmill, T. Atmospheric oxidation of iron pyrites. Trans. Inst. Min. Eng. 1951, 4, 500–509. [Google Scholar]
- Hu, J.Z.; Finkleman, R. Distribution and forming cause of sulphur in Chinese coals. Coal Convers. 2005, 28, 5–10. [Google Scholar]
- Li, R. The distribution of sulfur in coal in China. Clean Coal Technol. 1998, 1, 44–48. [Google Scholar]
- Chen, P.; Chen, W. Study on the Distribution Characteristics of Sulphur in Chinese Coals. Fuel 1986, 9, 1305–1309. [Google Scholar] [CrossRef]
- Li, W.; Tang, Y. Sulfur isotopic composition of superhigh-organic-sulfur coals from the Chenxi coalfield, southern China. Int. J. Coal Geol. 2014, 127, 3–13. [Google Scholar] [CrossRef]
- Davidson, R.M. Quantifying organic sulfur in coal: A review. Fuel 1994, 73, 988–1005. [Google Scholar] [CrossRef]
- Kozlowsk, M.; Pietrzak, R.; Wachowska, H.; Yperman, J. AP-TPR study of sulphur in coals subjected to mild oxidation. Part 1. Demineralised coals. Fuel 2002, 81, 2397–2405. [Google Scholar] [CrossRef]
- Boudou, J.P.; Boulègue, J.; Maléchaux, L.; Nip, M.; de Leeuw, J.W.; Boon, J.J. Identification of some sulphur species in a high organic sulphur coal. Fuel 1987, 66, 1558–1569. [Google Scholar] [CrossRef]
- Marinov, S.P.; Tyuliev, G.; Stefanova, M.; Carleer, R.; Yperman, J. Low rank coals sulphur functionality study by AP-TPR/TPO coupled with MS and potentiometric detection and by XPS. Fuel Process. Technol. 2004, 85, 267–277. [Google Scholar] [CrossRef]
- Calkins, W. Investigation of organic sulfur-containing structures in coal by flash pyrolysis experiments. Energy Fuels 1987, 1, 59–64. [Google Scholar] [CrossRef]
- Huffman, G.P.; Mitra, S.; Huggins, F.E.; Shah, N.; Vaidya, S.; Lu, F. Quantitative analysis of all major forms of sulfur in coal by X-ray absorption fine structure spectroscopy. Energy Fuels 1991, 5, 574–581. [Google Scholar] [CrossRef]
- Pietrzak, R.; Grzybek, T.; Wachowska, H. XPS study of pyrite-free coals subjected to different oxidizing agents. Fuel 2007, 86, 2616–2624. [Google Scholar] [CrossRef]
- Grzybek, T.; Pietrzak, R.; Wachowska, H. X-ray photoelectron spectroscopy study of oxidized coals with different sulphur content. Fuel Process Technol. 2002, 77–78, 1–7. [Google Scholar] [CrossRef]
- Perry, D.L.; Grint, A. Application of XPS to coal characterization. Fuel 1983, 62, 1024–1033. [Google Scholar] [CrossRef]
- Kelemen, S.R.; George, G.N. Direct determination and quantification of sulfur forms in heavy petroleum and coals 1. The X-ray photoelectron spectroscopy (XPS) approach. Fuel 1990, 69, 939–944. [Google Scholar] [CrossRef]
- Rong, G.; Su, K. University of Organic Chemistry; East China University of Science and Technology Press: Shanghai, China, 2000; pp. 307–314. [Google Scholar]
- Capozzi, G.; Modena, G. Oxidation of Thiols, Chemistry of the Thiol Group; Wiley: New York, NY, USA, 1974; pp. 785–839. [Google Scholar]
- March, J. Advanced Organic Chemistry; Wiley Eastern: New Delhi, India, 1994. [Google Scholar]
- Ignasiak, T.M.; Strausz, O.P. Reaction of Athabasca asphaltene with tetralin. Fuel 1978, 57, 617–621. [Google Scholar] [CrossRef]
- Borah, D. Electron transfer process. Part 2. Desulphurization of organic sulphur from feed and mercury-treated coals oxidized in air at 50, 100 and 150 °C. Fuel 2000, 79, 1785–1796. [Google Scholar] [CrossRef]
- Clark, P.D.; Hyne, J.B.; Tyrer, J.D. Some chemistry of organosulphur compound types occurring in heavy oil sands: 2. Influence of pH on the high temperature hydrolysis of tetrahydrothiophene and thiophene. Fuel 1984, 63, 125–128. [Google Scholar] [CrossRef]
- Benson, S.W. Thermochemistry and kinetics of sulfur-containing molecules and radicals. Chem. Rev. 1978, 23–35. [Google Scholar] [CrossRef]
- Dacka, S.W.; Hobdaya, M.D.; Smitha, T.D.; Pilbrowa, J.R. Free-radical involvement in the drying and oxidation of victorian brown coal. Fuel 1984, 63, 39–42. [Google Scholar] [CrossRef]
- Borah, D.; Baruah, M.K. Electron transfer process 1. Removal of organic sulphur from high sulphur Indian coal. Fuel 1999, 78, 1083–1088. [Google Scholar] [CrossRef]
- Gorbaty, M.L.; George, G.N.; Kelemen, S.R. Chemistry of organically bound sulphur forms during the mild oxidation of coal. Fuel 1990, 69, 1065–1067. [Google Scholar] [CrossRef]
- Gorbaty, M.L.; Kelemen, S.R.; George, G.N.; Kwiatek, P.J. Characterization and thermal reactivity of oxidized organic sulphur forms in coals. Fuel 1992, 71, 1255–1264. [Google Scholar] [CrossRef]
- Grzybek, T.; Pietrzak, R.; Wachowska, H. The Comparison of Oxygen and Sulfur Species Formed by Coal Oxidation with O2/Na2CO3 or Peroxyacetic Acid Solution. XPS Studies. Energy Fuels 2004, 18, 804–809. [Google Scholar] [CrossRef]
- Pietrzak, R.; Wachowska, H. Low temperature oxidation of coals of different rank and different sulphur content. Fuel 2003, 2003, 705–713. [Google Scholar] [CrossRef]
- Sönmez, Ö.; Giray, E.S. The influence of process parameters on desulfurization of two Turkish lignites by selective oxidation. Fuel Process. Technol. 2001, 70, 159–169. [Google Scholar] [CrossRef]
- Tang, Y.; Li, Z. Oxidation Experiment of Coal Spontaneous Combustion Model Compounds. Asian J. Chem. 2013, 25, 441–446. [Google Scholar]
- Tang, Y.B.; Li, Z.H.; Yang, Y.L.; Ma, D.J.; Liu, Z.; Ji, H.J. Low-temperature oxidation of anisole and benzyl alcohol by model compounds of coal spontaneous combustion. J. China Coal Soc. 2012, 37, 2048–2052. [Google Scholar]
- Li, Z.H.; Wang, Y.L.; Song, N.; Yang, Y.J. Experiment study of model compound oxidation on spontaneous combustion of coal. J. Hunan Univ. Sci. Technol. 2010, 1, 123–129. [Google Scholar]
- Yan, J.; Yang, J.; Liu, Z. SH Radical: The Key Intermediate in Sulfur Transformation during Thermal Processing of Coal. Environ. Sci.Technol. 2005, 39, 5043–5051. [Google Scholar] [CrossRef] [PubMed]
- Mullens, S.; Yperman, J.; Reggers, G.; Carleer, R.; Buchanan, A.C.; Britt, P.F.; Rutkowski, P.; Gryglewicz, G. A study of the reductive pyrolysis behaviour of sulphur model compounds. J. Anal. Appl. Pyrolysis 2003, 70, 469–491. [Google Scholar] [CrossRef]
- Buckley, A.N.; Riley, K.W.; Wilson, M.A. Heteroatom functionality in a high-sulfur Chinese bituminous coal. Org. Geochem. 1996, 24, 389–392. [Google Scholar] [CrossRef]
- Grzybek, T.; Pietrzak, R.; Wachowska, H. The influence of oxidation with air in comparison to oxygen in sodium carbonate solution on the surface composition of coals of different ranks. Fuel 2006, 85, 1016–1023. [Google Scholar] [CrossRef]
- Merwe, E.M.V.D.; Prinsloo, L.C.; Mathebula, C.L.; Swart, H.C.; Coetsee, E.; Doucet, F.J. Surface and bulk characterization of an ultrafine South African coal fly ash with reference to polymer applications. Appl. Surf. Sci. 2014, 317, 73–83. [Google Scholar] [CrossRef]
- Hao, S.; Wen, J.; Yu, X.; Chu, W. Effect of the surface oxygen groups on methane adsorption on coals. Appl. Surf. Sci. 2013, 264, 433–442. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are not available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, L.; Li, Z.; Li, J.; Zhou, Y.; Yang, Y.; Tang, Y. Studies on the Low-Temp Oxidation of Coal Containing Organic Sulfur and the Corresponding Model Compounds. Molecules 2015, 20, 22241-22256. https://doi.org/10.3390/molecules201219843
Zhang L, Li Z, Li J, Zhou Y, Yang Y, Tang Y. Studies on the Low-Temp Oxidation of Coal Containing Organic Sulfur and the Corresponding Model Compounds. Molecules. 2015; 20(12):22241-22256. https://doi.org/10.3390/molecules201219843
Chicago/Turabian StyleZhang, Lanjun, Zenghua Li, Jinhu Li, Yinbo Zhou, Yongliang Yang, and Yibo Tang. 2015. "Studies on the Low-Temp Oxidation of Coal Containing Organic Sulfur and the Corresponding Model Compounds" Molecules 20, no. 12: 22241-22256. https://doi.org/10.3390/molecules201219843
APA StyleZhang, L., Li, Z., Li, J., Zhou, Y., Yang, Y., & Tang, Y. (2015). Studies on the Low-Temp Oxidation of Coal Containing Organic Sulfur and the Corresponding Model Compounds. Molecules, 20(12), 22241-22256. https://doi.org/10.3390/molecules201219843





