Membrane Interactions of Phytochemicals as Their Molecular Mechanism Applicable to the Discovery of Drug Leads from Plants
Abstract
:1. Introduction
2. Membrane-Interactive Drugs
2.1. Anesthetics and Anesthesia-Related Drugs
2.2. Adrenergic and Cholinergic Drugs
2.3. Non-Steroidal Anti-Inflammatory Drugs and Analgesics
2.4. Antitumor and Antiproliferative Drugs
2.5. Antimicrobial Drugs and Antibiotics
2.6. Antioxidant and Lipid Peroxidation-Inhibitory Drugs
2.7. Antiplatelet and Platelet Aggregation-Inhibitory Drugs
3. Phytochemicals with Membrane Interactivity
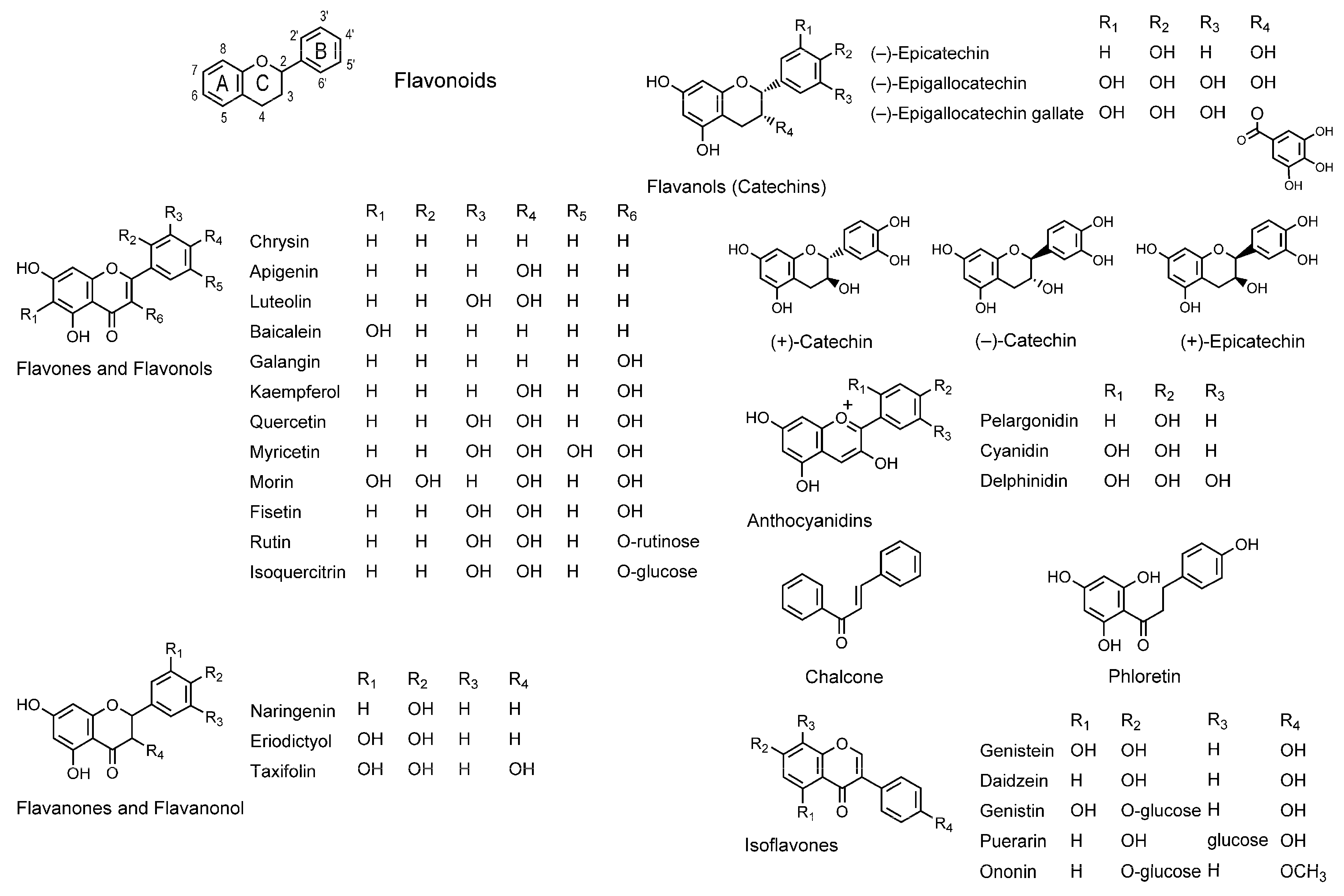
3.1. Flavonoids
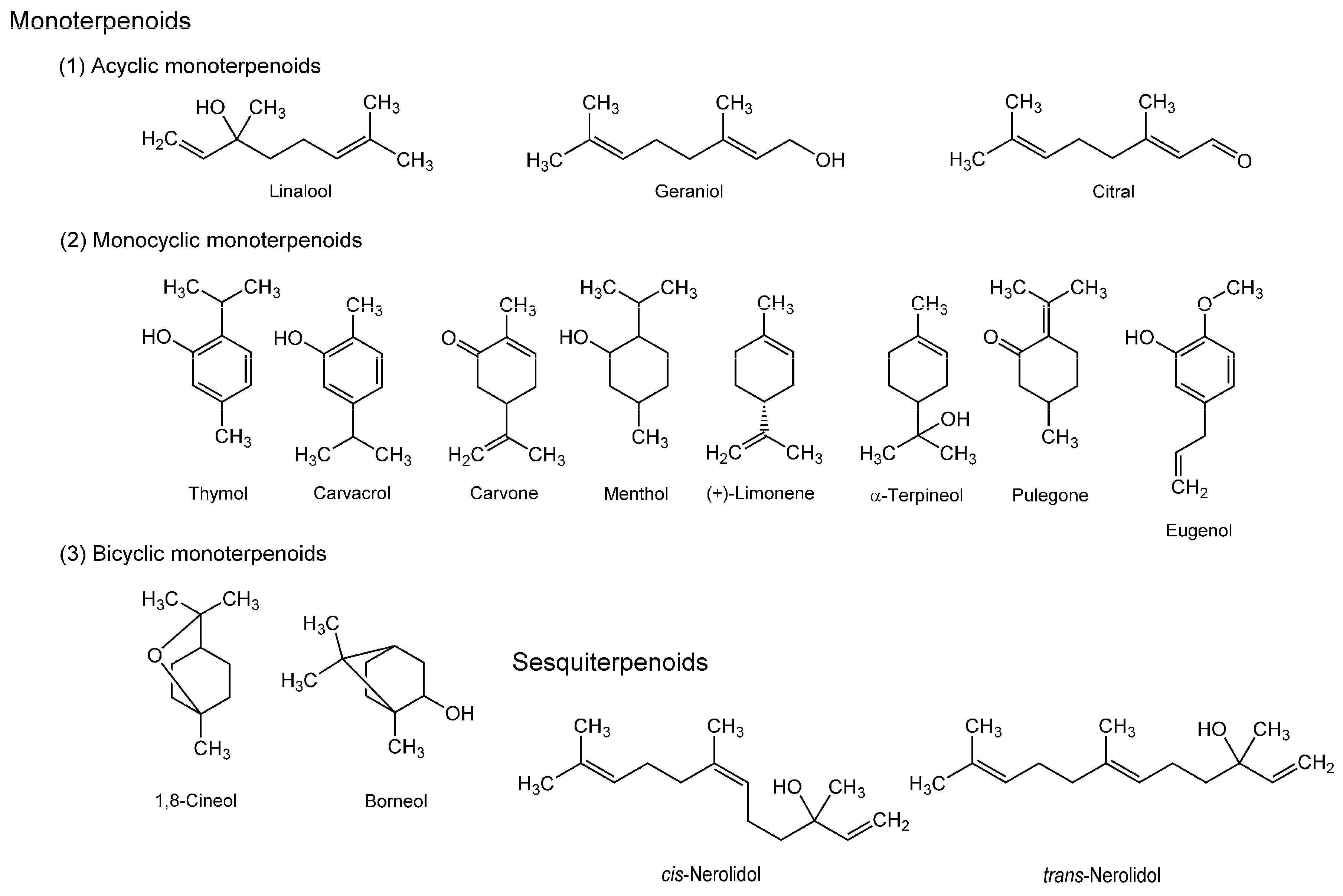
3.2. Terpenoids
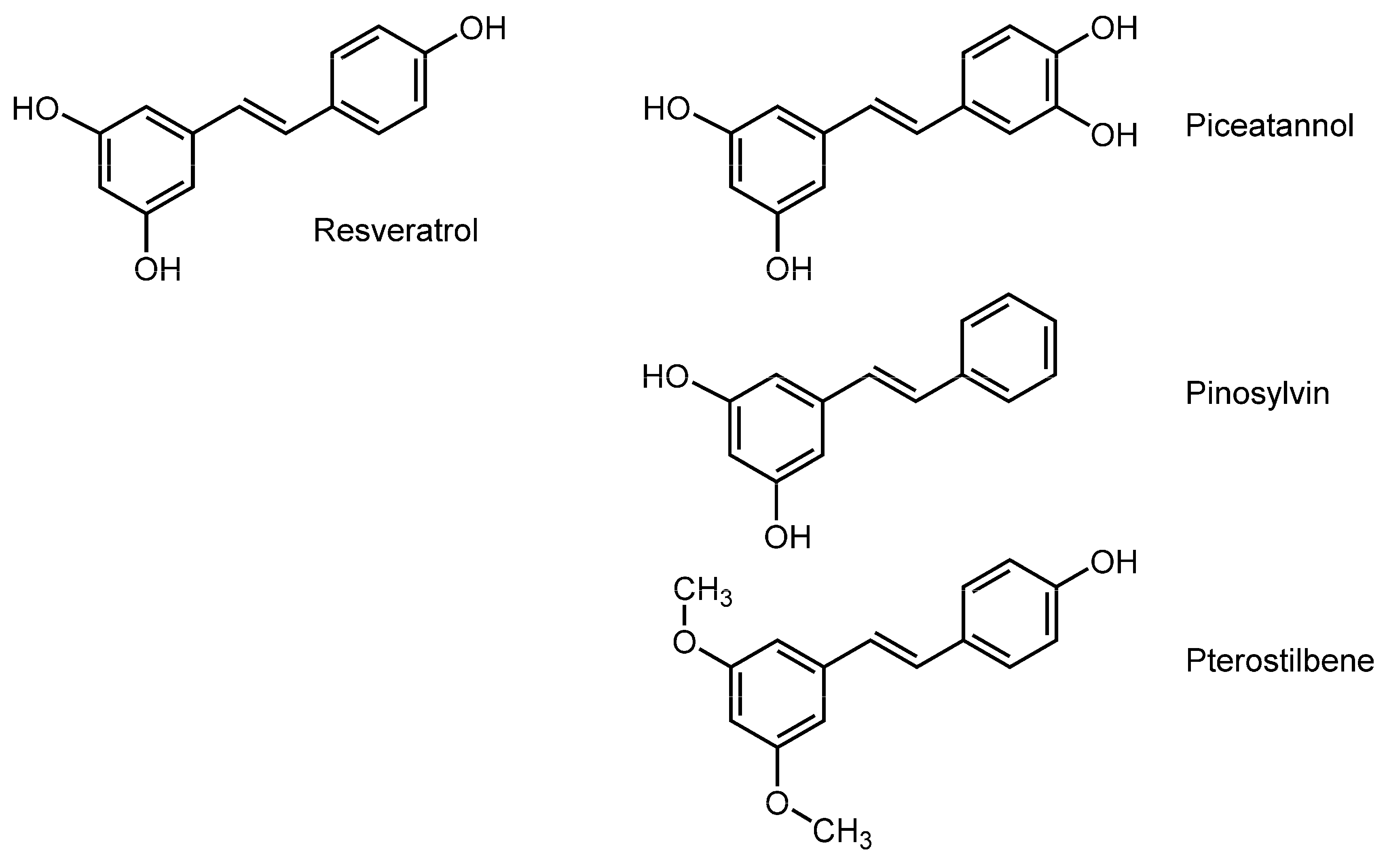
3.3. Stilbenoids
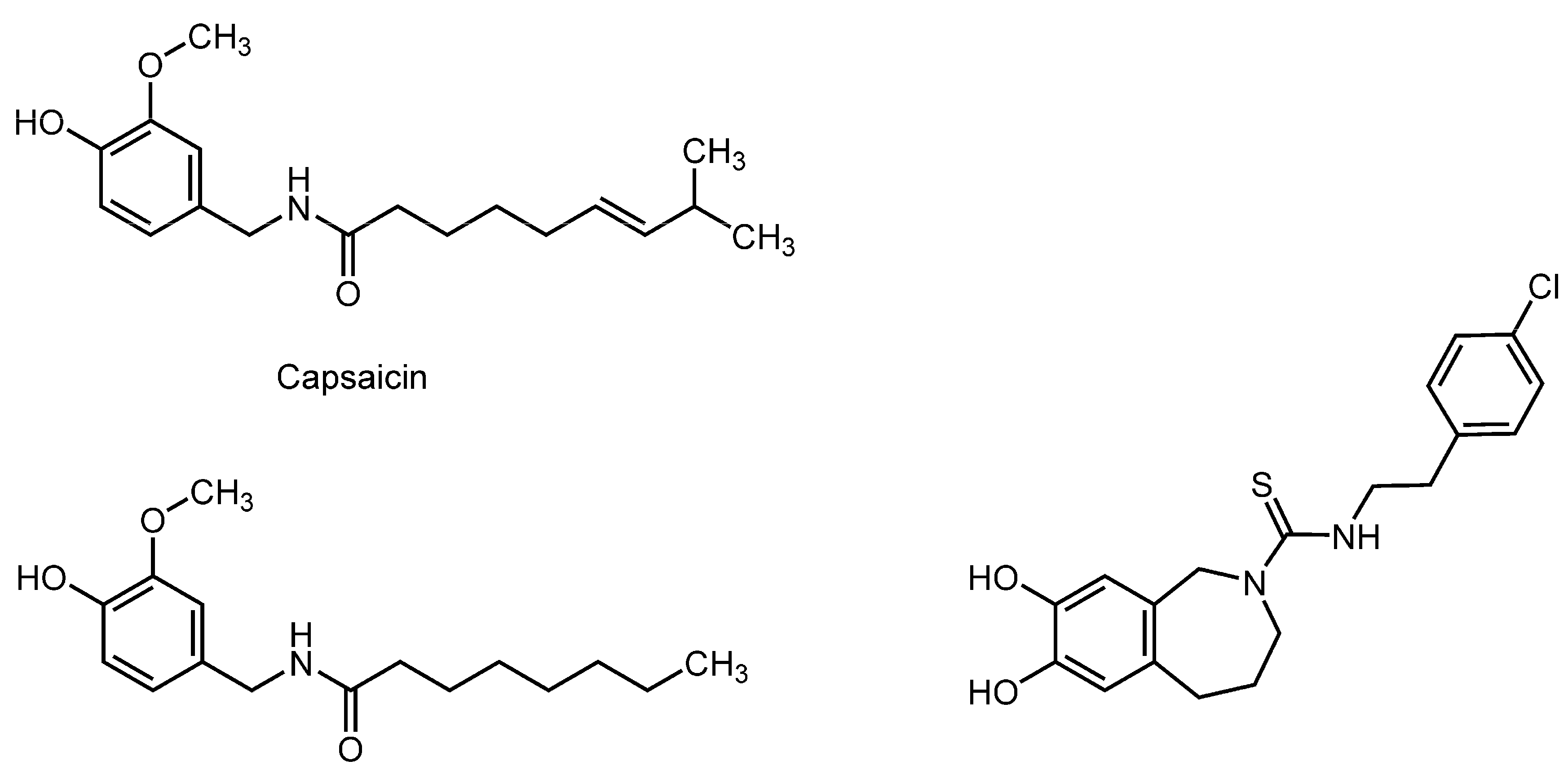
3.4. Capsaicinoids
3.5. Phloroglucinols and Naphthodianthrones
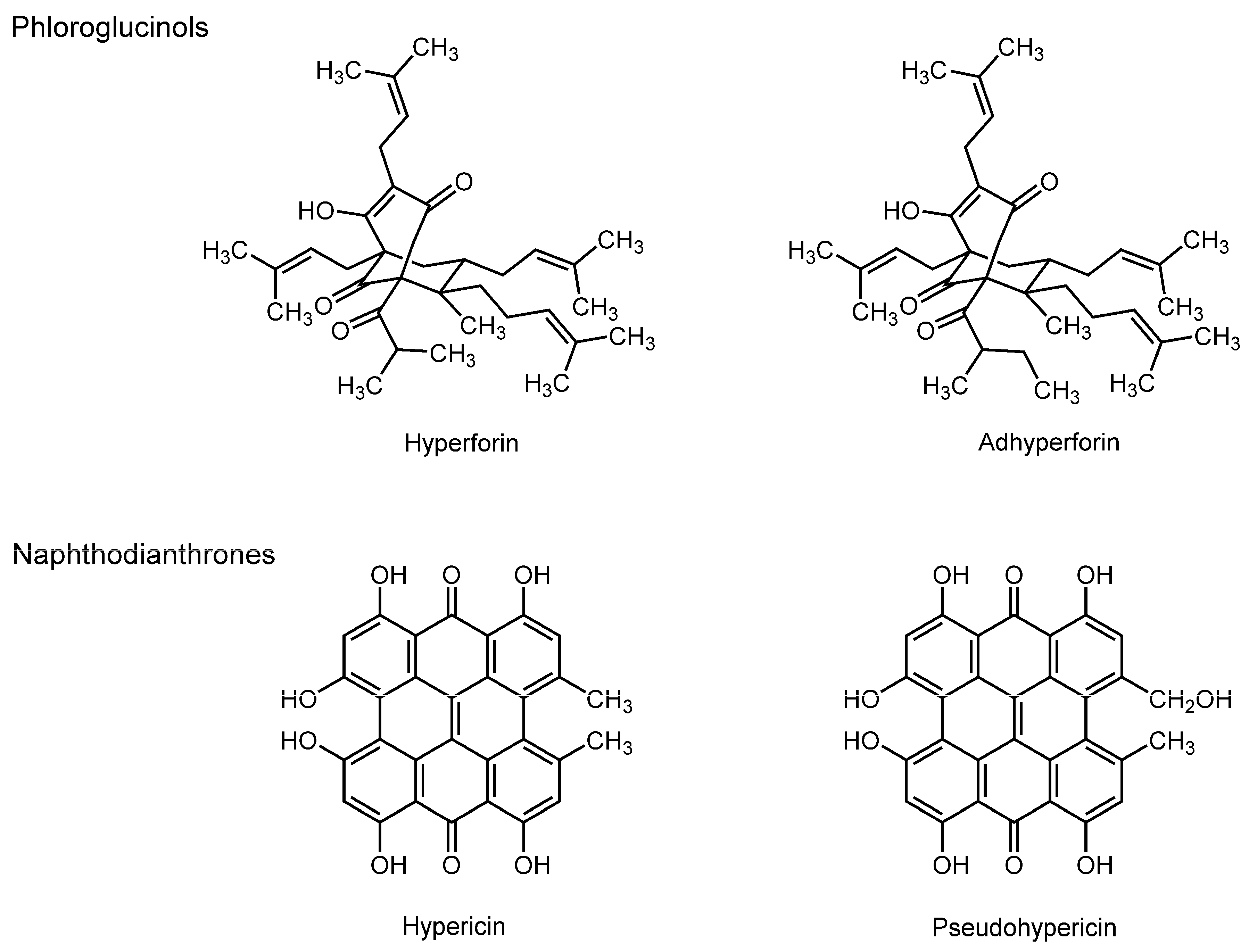
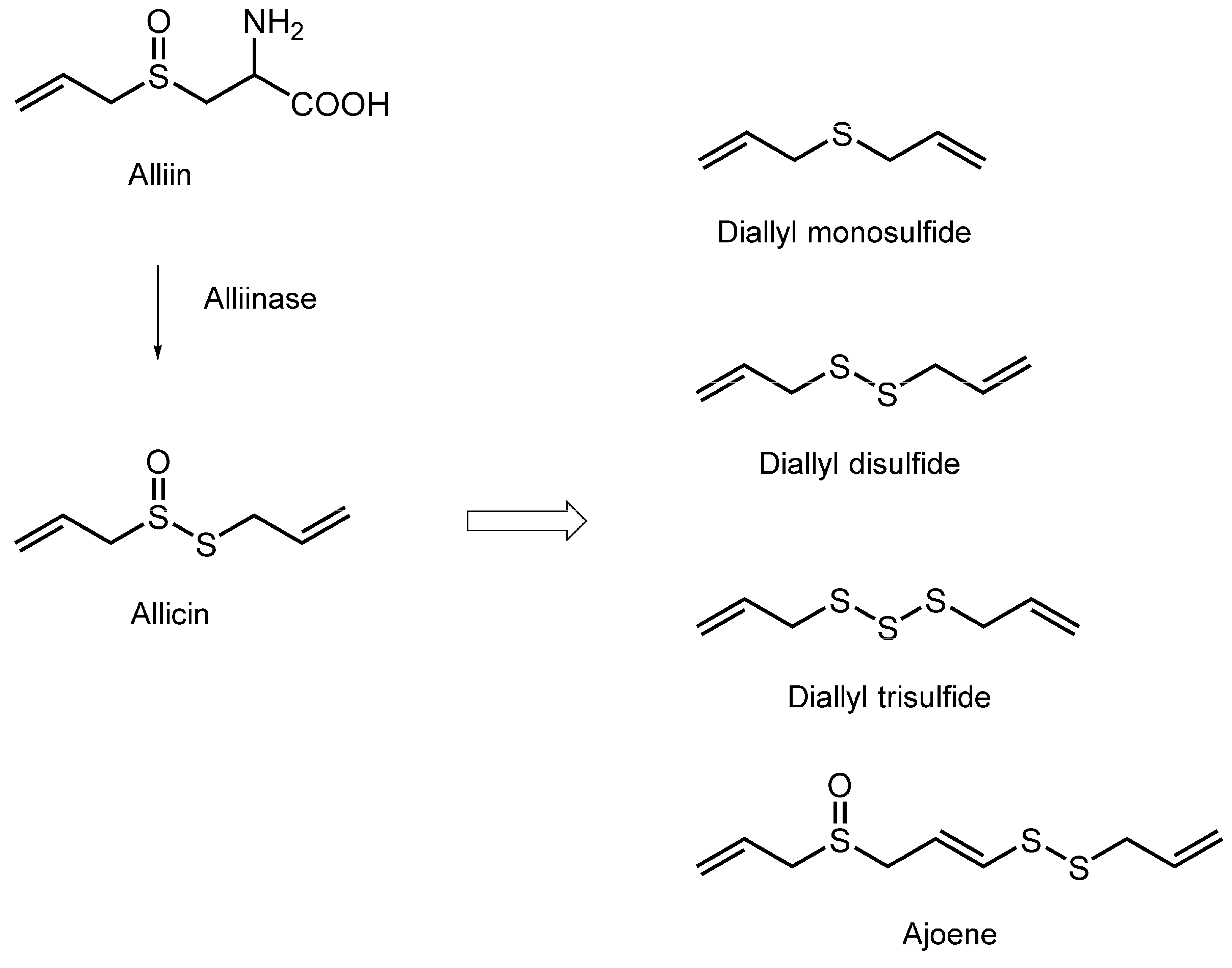
3.6. Organosulfur Compounds
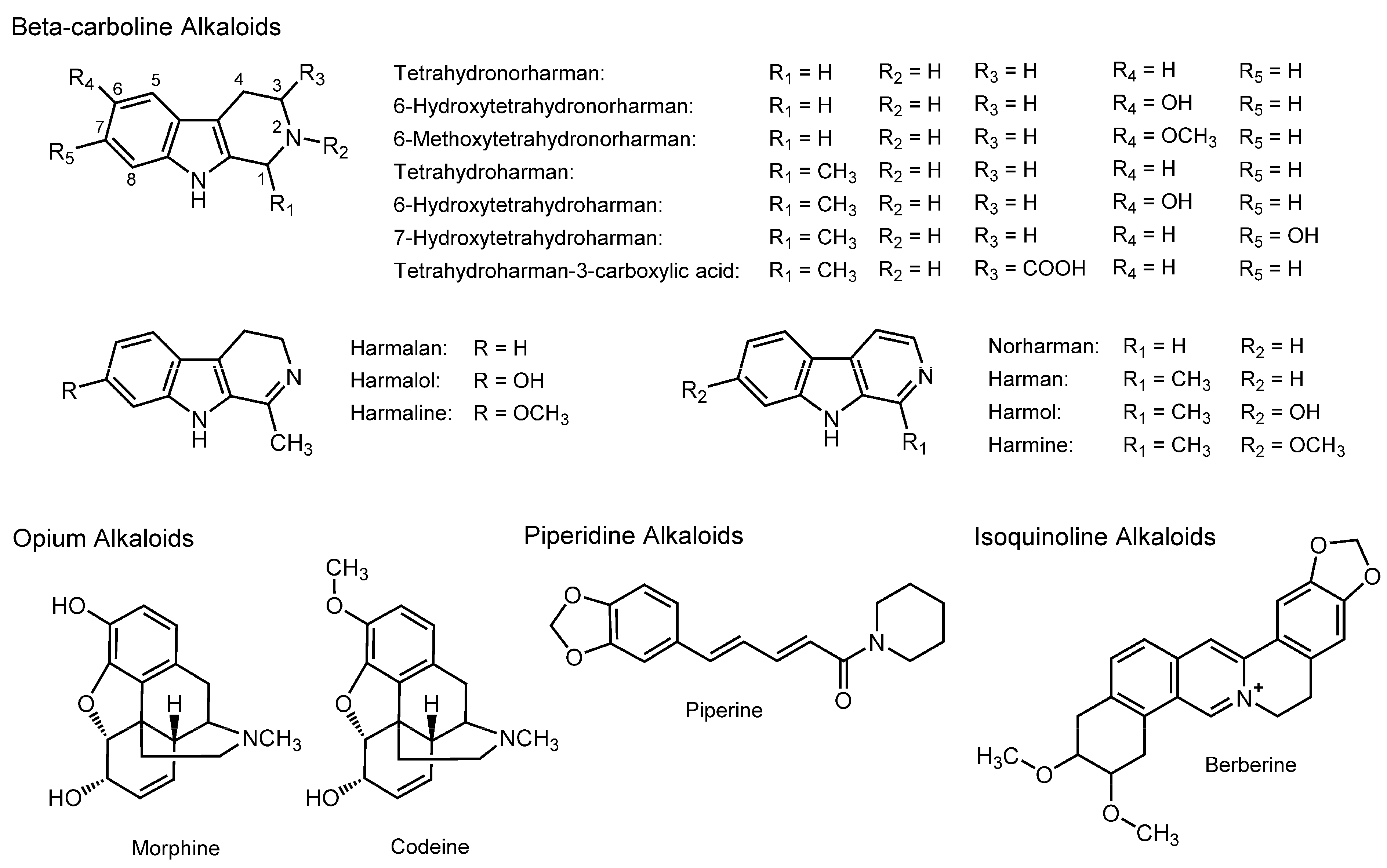
3.7. Alkaloids
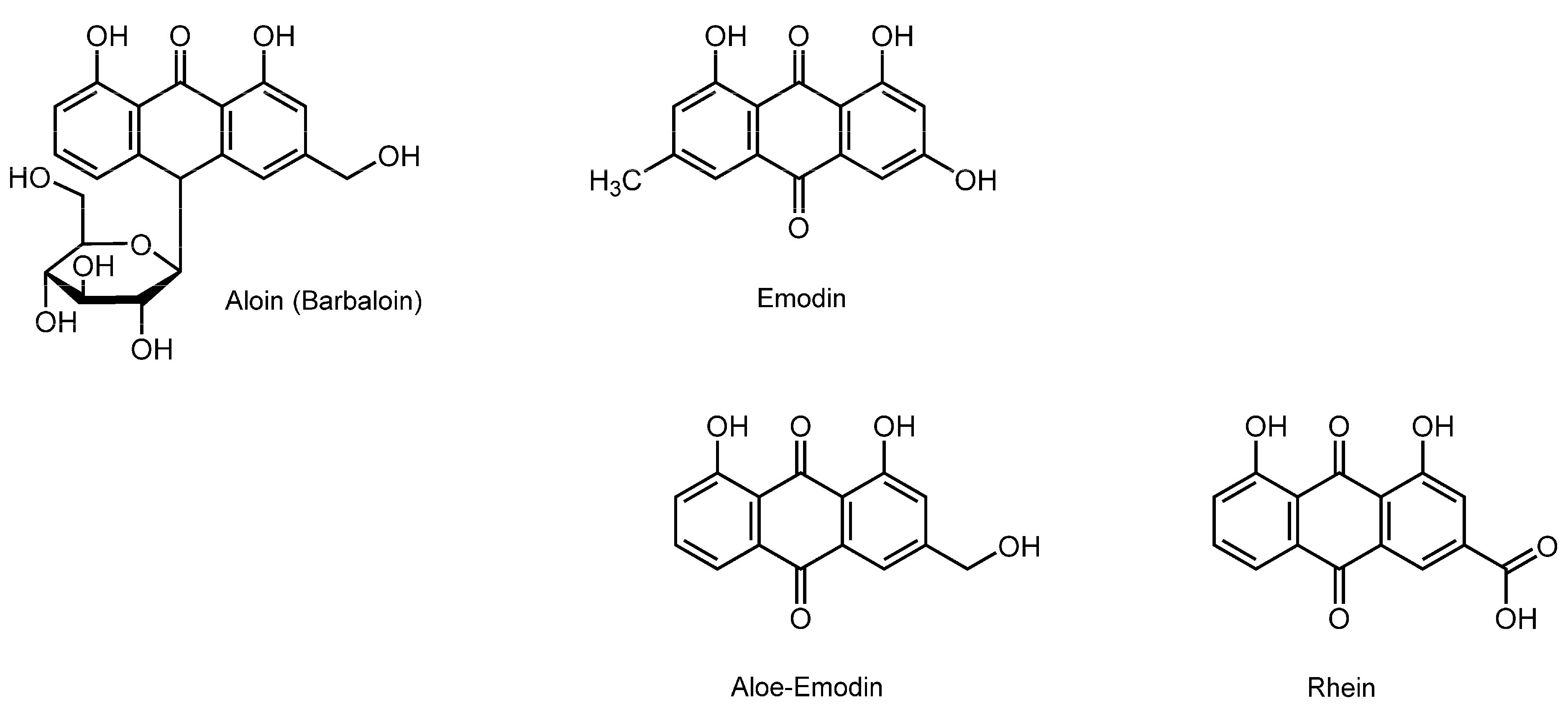
3.8. Anthraquinonoids
3.9. Ginsenosides
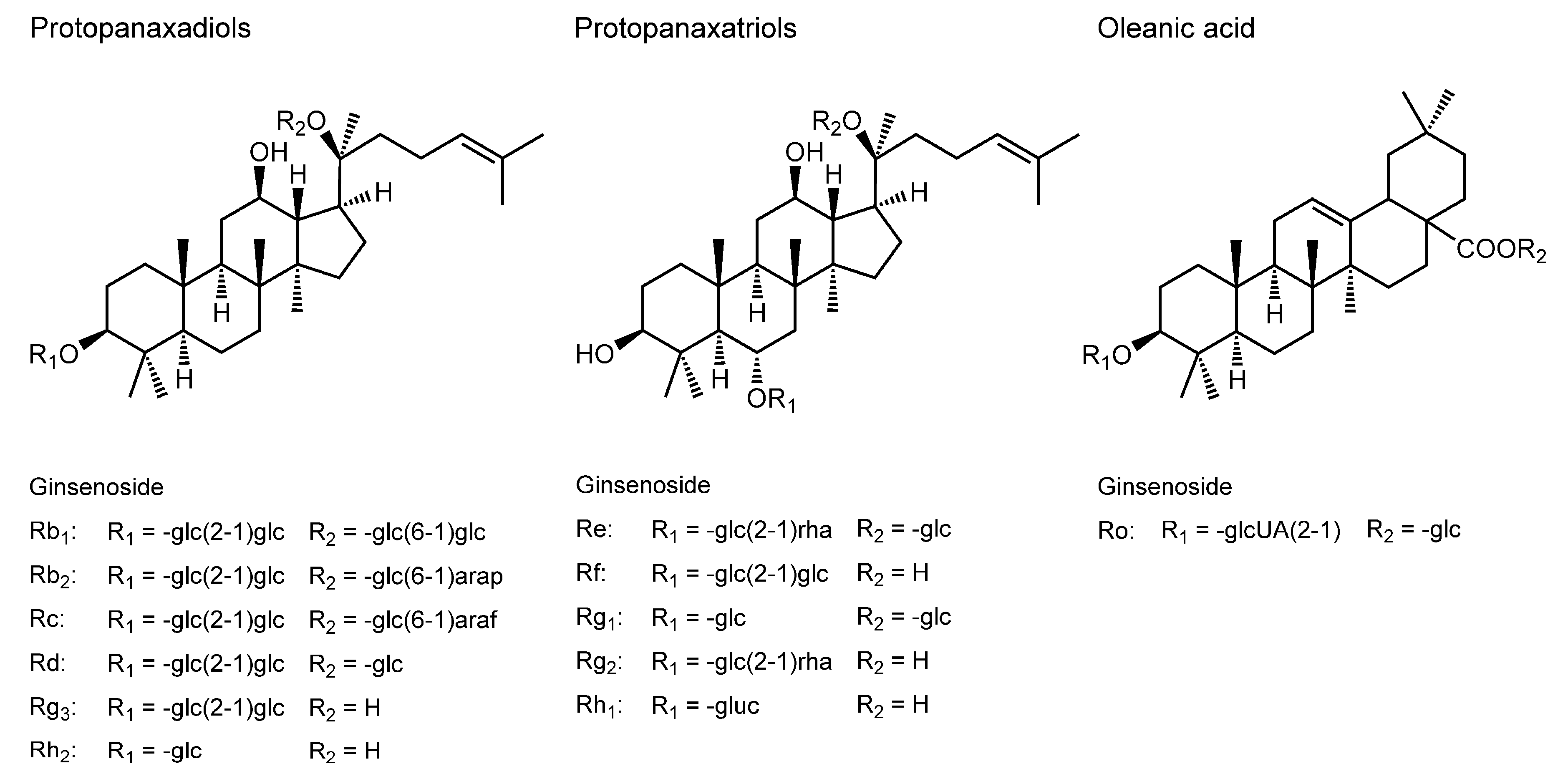
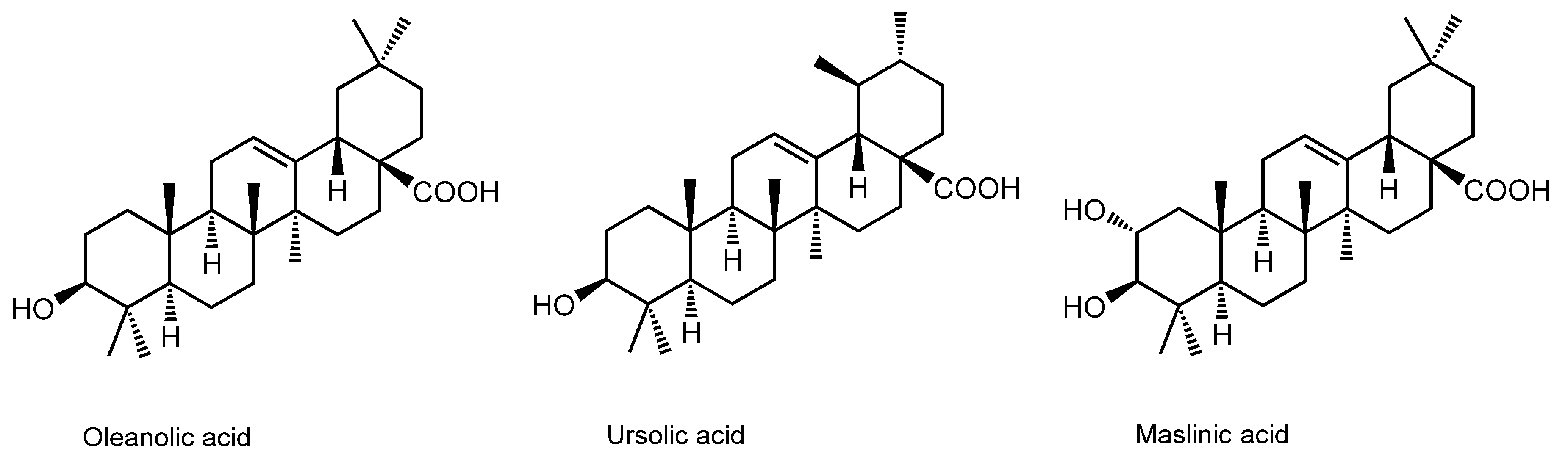
3.10. Pentacyclic Triterpene Acids
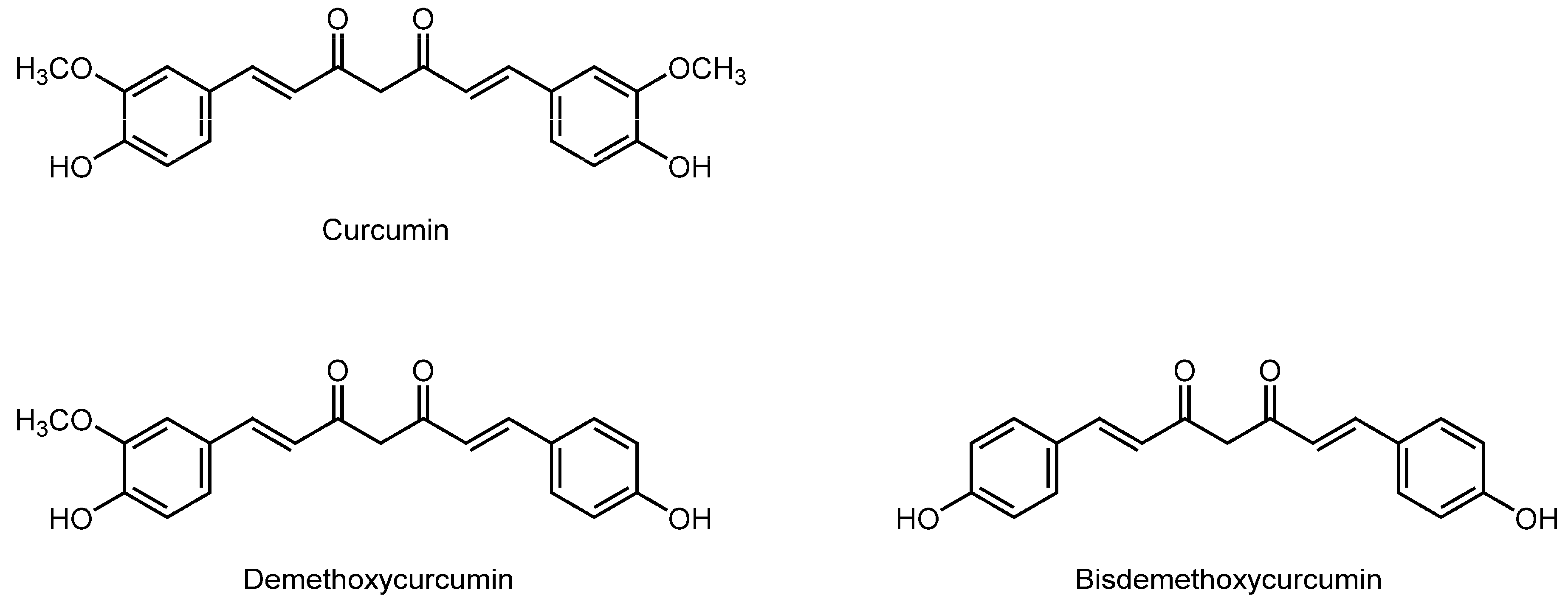
3.11. Curcuminoids
3.12. Bioavailability of Membrane-Interactive Phytochemicals
4. Membrane Interaction Applicable to the Discovery of Phytochemical Drug Leads
5. Conclusions
| Phytochemicals or Plants | Used Membranes | Experimental Methods | Membrane Effects | References |
|---|---|---|---|---|
| Flavonoids | ||||
| Naringenin; Rutin | SLPC unilamellar vesicles | FA with 6-AS, 12-AS and 16-AP | Decrease the fluidity | [72] |
| Apigenin; Luteolin; Kaempferol; Quercetin; Myricetin; Naringenin; Eriodictyol; Taxifolin; Pelargonidin; Cyanidin; Delphinidin; (–)-Epicatechin; (–)-Epigallocatechin; (–)-Epigallocatechin-3-gallate; Phloretin | POPC/POPE/POPS/cholesterol unilamellar vesicles | FP with 2-AS, 6-AS, 9-AS, 12-AS and 16-AP | Decrease the fluidity | [79] |
| Kaempferol; Quercetin; Chrysin; Baicalein; Luteolin | DPPC unilamellar vesicles; DPPG unilamellar vesicles | FP with DPH | Decrease the fluidity | [80] |
| Galangin; Quercetin; Kaempferol; Chrysin; Baicalein; Fisetin; Luteolin; Apigenin; Myricetin; Morin; Rutin; Isoquercitrin | POPC/POPE/SOPS/cholesterol unilamellar vesicles | FP with 2-AS, 6-AS, 9-AS, 12-AS and 16-AP | Decrease the fluidity | [82] |
| Quercetin; (–)-Epigallocatechin-3-gallate | Human U937 monocyte membranes; Jurkat T lymphoblast membranes | FA with TMA-DPH | Decrease the fluidity | [83] |
| (–)-Epicatechin; (–)-Epigallocatechin; (–)-Epigallocatechin-3-gallate; (–)-Epicatechin-3-gallate | DPPC liposomes; DOPC liposomes | FP with PNA and ANS | Decrease the fluidity | [86] |
| (–)-Epicatechin; (+)-Epicatechin; (–)-Catechin; (+)-Catechin | DPPC liposomes; DOPC liposomes; DOPC/cholesterol liposomes | FP with PNA and ANS | Decrease the fluidity | [87] |
| Isoflavonoids | ||||
| Genistein | SLPC unilamellar vesicles | FA with 6-AS, 12-AS and 16-AP | Decrease the fluidity | [72] |
| Puerarin; Ononin; Daidzein; Genistin | DPPC unilamellar vesicles; DPPG unilamellar vesicles | FP with DPH | Increase the fluidity | [80] |
| Genistein | Erythrocyte membranes | EPR | Decrease the fluidity | [81] |
| Daidzein | Erythrocyte membranes | EPR | Increase the fluidity | [81] |
| Terpenoids | ||||
| Geraniol | Candida albicans cell membranes; DPPC multilamellar vesicles | FA with 2-AS, 6-AS, 9-AS, 12-AS, DPH and TMA-DPH | Increase the fluidity from the membrane surface to the membrane interior | [90] |
| Carvacrol | Langmuir monolayer membranes of bacterial phospholipids | Surface pressure-area (π-A) and surface potential-area (Δψ-A) isotherms | Increase the fluidity | [91] |
| Thymol; Eugenol; Carvacrol | DPPC multilamellar vesicles | FA with DPH and TMA-DPH | Increase the fluidity | [93] |
| Nerolidol; Menthol; Pulegone; Carvone; (+)-Limonene; α-Terpineol; 1,8-Cineol | Mouse fibroblast membranes; Human erythrocyte membranes | EPR | Increase the fluidity | [94] |
| Terpenoids | ||||
| Linalool; 1,8-Cineol; α-Terpineol | Staphylococcus aureus membranes; Escherichia coli membranes | Scanning electron microscopy | Alter the permeability and function | [95] |
| Citral | Soybean phospholipid liposomes | FP with DPH | Decrease the fluidity | [96] |
| Borneol | DPPC liposomes | Coarse-grained molecular dynamics simulations | Increase the fluidity | [97] |
| Stilbenoids | ||||
| Resveratrol | DPPC liposomes; POPC liposomes; POPC/cholesterol liposomes | FP with DPH and TMA-DPH | Decrease the fluidity | [101] |
| Resveratrol | Lipid bilayer liposomal membranes | NMR spectroscopy | Decrease the fluidity | [102] |
| Resveratrol | DPPC unilamellar vesicles | FA with TMA-DPH | Increase the fluidity | [103] |
| Resveratrol; Piceatannol | DPPC liposomes; DMPC liposomes | DSC; ESR; Fluorescence spectroscopy | Alter the phospholipid phase transition | [104] |
| Capsaicinoids | ||||
| Capsaicin | Non-neuronal plasma membranes | FP with DPH and TMA-DPH | Increase the fluidity | [106] |
| Capsaicin; N-Vanillylnonanamide | Platelet mimetic membranes; Bacterial cell mimetic membranes | FP with PNA, DPH and TMA-DPH | Increase the fluidity at ~50 μM Decrease the fluidity at 100–500 μM | [107] |
| Maintaining rats on diets containing 0.015% capsaicin for eight weeks | Rat erythrocyte membranes | ESR; FA with DPH | Increase the fluidity | [108] |
| Maintaining rats on diets containing 3.0% chili pepper or 0.01% capsaicin for eight weeks | Rat intestinal brush-border membranes | FP with DPH | Increase the fluidity | [109] |
| Phloroglucinols | ||||
| Hyperforin | Guinea pig crude brain membranes | FA with DPH and TMA-DPH | Increase the fluidity of the hydrocarbon cores | [113] |
| Hyperforin | Guinea pig crude brain membranes | FA with DPH and TMA-DPH | Decrease the fluidity of the hydrophilic regions | [113] |
| Oral administration of St. John’s wort extract or hyperforin to murine | Murine brain neuronal membranes | FA with DPH and TMA-DPH | Decrease the fluidity | [114] |
| Naphthodianthrones | ||||
| Hypericin | DMPC liposomes | Microspectrofluorimetry | Decrease the fluidity | [115] |
| Organosulfur Compounds | ||||
| Diallyl trisulfide; Diallyl disulfide | Tumor cell mimetic membranes; Platelet mimetic membranes | FP with DPH, TMA-DPH, 2-AS, 6-AS, 9-AS, 12-AS and 16-AP | Decrease the fluidity by preferentially acting on the hydrocarbon cores | [117] |
| Diallyl trisulfide; Diallyl disulfide; Diallyl monosulfide | Candida cell mimetic membranes containing ergosterol | FP with DPH, TMA-DPH, 2-AS, 6-AS, 9-AS, 12-AS and 16-AP | Decrease the fluidity | [117] |
| Ajoene | Phospholipid/cholesterol unilamellar vesicles | ESR | Increase the fluidity of the hydrocarbon chains | [118] |
| Ajoene | Platelet plasma membranes; Artificial lipid membranes | ESR | Decrease the microviscosity | [119] |
| Beta-carboline Alkaloids | ||||
| Tetrahydroharman | Platelet mimetic membranes | FP with PNA and ANS | Increase the fluidity at antiplatelet concentrations | [124] |
| Tetrahydroharman; Tetrahydronorharman | DPPC liposomes | FP with PNA and ANS | Increase the fluidity at high micromolar concentrations | [125] |
| Tetrahydroharman | POPC/cholesterol liposomes | FP with PNA, ANS, DPH and TMA-DPH | Decrease the fluidity at low nanomolar concentrations Increase the fluidity at high micromolar concentrations | [126,127,128] |
| Opium Alkaloids | ||||
| Morphine | Rat erythrocyte membranes | Fourier transformed infrared spectroscopy; Fluorescence depolarization | Decrease the fluidity | [45] |
| Morphine; Codeine | DPPC unilamellar vesicles | DSC; EPR | Decrease the mobility of the polar head groups | [129] |
| Oral administration of piperine (5–20 mg/kg body weight) to rats for 5–15 min | Rat intestinal brush-border membranes | Fluorospectroscopy with pyrene | Increase the fluidity | [130] |
| Piperine (2–50 μM) | Brush-border membrane vesicles from rat jejunum | Fluorospectroscopy with pyrene | Increase the fluidity | [130] |
| Maintaining rats on diets containing 0.02% piperine for eight weeks | Rat intestinal brush-border membranes | FP with DPH | Increase the fluidity | [109] |
| Anthraquinonoids | ||||
| Aloin; Emodin | DMPC unilamellar vesicles; DMPG unilamellar vesicles | DSC; ESR | Perturb the membrane property and structure | [132,133] |
| Ginsenosides | ||||
| Ginsenoside Rb2; Rc; Rd; Re; Rf; Rg1; Rg2; Rh2 | HeLa cell membranes | Two-photon fluorescence microscopy; Generalized polarization imaging | Increase the fluidity | [134] |
| Oral administration of ginsenoside Re (5–20 mg/kg body weight) to rats for seven days | Rat brain mitochondrial membranes | FP with DPH | Increase the fluidity | [135] |
| Ginsenoside Rg3 (20 μM) | Human fibroblast carcinoma cell membranes | FA with DPH and TMA-DPH | Decrease the fluidity | [136] |
| Pentacyclic Triterpene Acids | ||||
| Oleanolic acid; Ursolic acid | DPPC liposomes | FP with DPH | Increase the fluidity in crystalline state Decrease the fluidity in liquid-crystalline state | [138] |
| Curcuminoids | ||||
| Curcumin (100–150 μM) | Human erythrocyte membranes | ESR | Increase the fluidity | [140] |
| Maintaining experimental hypercholesterolemic rats on diets containing 0.2% curcumin for eight weeks | Rat erythrocyte membranes | ESR; FA with DPH | Increase the fluidity | [108] |
| Curcumin | DOPC unilamellar vesicles | X-ray diffraction | Thin the lipid bilayer membranes and weaken their elasticity | [143] |
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Lee, K.H. Discovery and development of natural product-derived chemotherapeutic agents based on a medicinal chemistry approach. J. Nat. Prod. 2010, 73, 500–516. [Google Scholar]
- Tapsell, L.C.; Hemphill, I.; Cobiac, L.; Patch, C.S.; Sullivan, D.R.; Fenech, M.; Roodenrys, S.; Keogh, J.B.; Clifton, P.M.; Williams, P.G.; et al. Health benefits of herbs and spices: The past, the present, the future. Med. J. Aust. 2006, 185, S4–S24. [Google Scholar] [PubMed]
- Kopeć, W.; Telenius, J.; Khandelia, H. Molecular dynamics simulations of the interactions of medicinal plant extracts and drugs with lipid bilayer membranes. FEBS J. 2013, 280, 2785–2805. [Google Scholar] [PubMed]
- Lúcio, M.; Lima, J.L.; Reis, S. Drug-membrane interactions: Significance for medicinal chemistry. Curr. Med. Chem. 2010, 17, 1795–1809. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, H.; Mizogami, M. Interaction of local anesthetics with biomembranes consisting of phospholipids and cholesterol: Mechanistic and clinical implications for anesthetic and cardiotoxic effects. Anesthesiol. Res. Pract. 2013. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, H. Structure-specific membrane-fluidizing effect of propofol. Clin. Exp. Pharmacol. Physiol. 2001, 28, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Bahri, M.A.; Heyne, B.J.; Hans, P.; Seret, A.E.; Mouithys-Mickalad, A.A.; Hoebeke, M.D. Quantification of lipid bilayer effective microviscosity and fluidity effect induced by propofol. Biophys. Chem. 2005, 114, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Hauet, N.; Artzner, F.; Boucher, F.; Grabielle-Madelmont, C.; Cloutier, I.; Keller, G.; Lesieur, P.; Durand, D.; Paternostre, M. Interaction between artificial membranes and enflurane, a general volatile anesthetic: DPPC-enflurane interaction. Biophys. J. 2003, 84, 3123–3137. [Google Scholar] [CrossRef]
- Booker, R.D.; Sun, A.K. Biophysical changes induced by xenon on phospholipid bilayers. Biochim. Biophys. Acta 2013, 1828, 1347–1356. [Google Scholar] [CrossRef] [PubMed]
- Harris, R.A.; Schroeder, F. Effects of barbiturates and ethanol on the physical properties of brain membranes. J. Pharmacol. Exp. Ther. 1982, 223, 424–431. [Google Scholar] [PubMed]
- Mennini, T.; Ceci, A.; Caccia, S.; Garattini, S.; Masturzo, P.; Salmona, M. Diazepam increases membrane fluidity of rat hippocampus synaptosomes. FEBS Lett. 1984, 173, 255–258. [Google Scholar] [CrossRef]
- Jerabek, H.; Pabst, G.; Rappolt, M.; Stockner, T. Membrane-mediated effect on ion channels induced by the anesthetic drug ketamine. J. Am. Chem. Soc. 2010, 132, 7990–7997. [Google Scholar] [CrossRef] [PubMed]
- Søgaard, R.; Werge, T.M.; Bertelsen, C.; Lundbye, C.; Madsen, K.L.; Nielsen, C.H.; Lundbæk, J.A. GABAA receptor function is regulated by lipid bilayer elasticity. Biochemistry 2006, 45, 13118–13129. [Google Scholar] [CrossRef] [PubMed]
- Mizogami, M.; Tsuchiya, H.; Harada, J. Membrane effects of ropivacaine compared with those of bupivacaine and mepivacaine. Fundam. Clin. Pharmacol. 2002, 16, 325–330. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, H.; Mizogami, M. Membrane interactivity of charged local anesthetic derivative and stereoselectivity in membrane interaction of local anesthetic enantiomers. Local Reg. Anesth. 2008, 1, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Lorite, G.S.; Nobre, T.M.; Zaniquelli, M.E.; de Paula, E.; Cotta, M.A. Dibucaine effects on structural and elastic properties of lipid bilayers. Biophys. Chem. 2009, 13, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Yi, Z.; Nagao, M.; Bossev, D.P. Effect of charged lidocaine on static and dynamic properties of model bio-membranes. Biophys. Chem. 2012, 160, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Lundbæk, J.A. Lipid bilayer-mediated regulation of ion channel function by amphiphilic drugs. J. Gen. Physiol. 2008, 131, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Leifert, W.R.; McMurchie, E.J.; Saint, D.A. Inhibition of cardiac sodium currents in adult rat myocytes by n-3 polyunsaturated fatty acids. J. Physiol. 1999, 520 Pt 3, 671–679. [Google Scholar] [CrossRef] [PubMed]
- Hahin, R.; Larsen, J.; Gasser, K. Predictions of the EC50 for action potential block for aliphatic solutes. J. Membr. Biol. 2008, 221, 73–85. [Google Scholar] [CrossRef] [PubMed]
- Lundbæk, J.A.; Birn, P.; Tape, S.E.; Toombes, G.E.; Søgaard, R.; Koeppe, R.E., II; Gruner, S.M.; Hansen, A.J.; Andersen, O.S. Capsaicin regulates voltage-dependent sodium channels by altering lipid bilayer elasticity. Mol. Pharmacol. 2005, 68, 680–689. [Google Scholar] [PubMed]
- Frangopol, P.T.; Mihăilescu, D. Interactions of some local anesthetics and alcohols with membranes. Colloids Surf. B Biointerfaces 2001, 22, 3–22. [Google Scholar] [CrossRef]
- Tsuchiya, H.; Mizogami, M. R(+)-, Rac-, and S(–)-bupivacaine stereostructure-specifically interact with membrane lipids at cardiotoxically relevant concentrations. Anesth. Analg. 2012, 114, 310–312. [Google Scholar] [CrossRef] [PubMed]
- Cantor, R.S. Receptor desensitization by neurotransmitters in membranes: Are neurotransmitters the endogenous anesthetics? Biochemistry 2003, 42, 11891–11897. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, D.; Cuenoud, B.; Krämer, S.D. Lipid membrane interactions of indacaterol and salmeterol: Do they influence their pharmacological properties? Eur. J. Pharm. Sci. 2009, 38, 533–547. [Google Scholar] [CrossRef] [PubMed]
- Butler, S.; Wang, R.; Wunder, S.L.; Cheng, H.Y.; Randall, C.S. Perturbing effects of carvedilol on a model membrane system: Role of lipophilicity and chemical structure. Biophys. Chem. 2006, 119, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Mizogami, M.; Takakura, K.; Tsuchiya, H. The interactivities with lipid membranes differentially characterize selective and nonselective β1-blockers. Eur. J. Anaesthesiol. 2010, 27, 829–834. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, H.; Mizogami, M. Characteristic interactivity of landiolol, an ultra-short-acting highly selective β1-blocker, with biomimetic membranes: Comparisons with β1-selective esmolol and non-selective propranolol and alprenolol. Front. Pharmacol. 2013. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.Y.; Randall, C.S.; Holl, W.W.; Constantinides, P.P.; Yue, T.L.; Feuerstein, G.Z. Carvedilol-liposome interaction: Evidence for strong association with the hydrophobic region of the lipid bilayers. Biochim. Biophys. Acta 1996, 1284, 20–28. [Google Scholar] [CrossRef]
- Manevich, E.M.; Köiv, A.; Järv, J.; Molotkovsky, J.G.; Bergelson, L.D. Binding to specific ligands to muscarinic receptors alters the fluidity of membrane fragments from rat brain. A fluorescence polarization study with lipid-specific probes. FEBS. Lett. 1988, 236, 43–46. [Google Scholar] [CrossRef]
- Tang, C.; Castoldi, A.F.; Costa, L.G. Effects of the muscarinic agonist oxotremorine on membrane fluidity in rat lymphocytes. Biochem. Mol. Biol. Int. 1993, 29, 1047–1054. [Google Scholar] [PubMed]
- Lichtenberger, L.M.; Zhou, Y.; Jayaraman, V.; Doyen, J.R.; O’Neil, R.G.; Dial, E.J.; Volk, D.E.; Gorenstein, D.G.; Boggara, M.B.; Krishnamoorti, R. Insight into NSAID-induced membrane alterations, pathogenesis and therapeutics: Characterization of interaction of NSAIDs with phosphatidylcholine. Biochim. Biophys. Acta 2012, 1821, 994–1002. [Google Scholar] [CrossRef] [PubMed]
- Rand Doyen, J.; Yucer, N.; Lichtenberger, L.M.; Kulmacz, R.J. Phospholipid actions on PGHS-1 and -2 cyclooxygenase kinetics. Prostaglandins Other Lipid Mediat. 2008, 85, 134–143. [Google Scholar] [CrossRef] [PubMed]
- Alsop, R.J.; Barrett, M.A.; Zheng, S.; Dies, H.; Rheinstädter, M.C. Acetylsalicylic acid (ASA) increases the solubility of cholesterol when incorporated in lipid membranes. Soft Matter. 2014, 10, 4275–4286. [Google Scholar] [CrossRef] [PubMed]
- Alsop, R.J.; Toppozini, L.; Marquardt, D.; Kučerka, N.; Harroun, T.A.; Rheinstädter, M.C. Aspirin inhibits formation of cholesterol rafts in fluid lipid membranes. Biochim. Biophys. Acta 2015, 1848, 805–812. [Google Scholar] [CrossRef] [PubMed]
- Manrique-Moreno, M.; Garidel, P.; Suwalsky, M.; Howe, J.; Brandenburg, K. The membrane-activity of Ibuprofen, Diclofenac, and Naproxen: A physic-chemical study with lecithin phospholipids. Biochim. Biophys. Acta 2009, 1788, 1296–1303. [Google Scholar] [CrossRef] [PubMed]
- Abramson, S.B.; Cherksey, B.; Gude, D.; Leszczynska-Piziak, J.; Phillips, M.R.; Blau, L.; Weissmann, G. Nonsteroidal anti-inflammatory drugs exert differential effects on neutrophil function and plasma membrane viscosity. Studies in human neutrophils and liposomes. Inflammation 1990, 14, 11–30. [Google Scholar] [CrossRef] [PubMed]
- Winocour, P.D.; Watala, C.; Perry, D.W.; Kinlough-Rathbone, R.L. Decreased platelet membrane fluidity due to glycation or acetylation of membrane proteins. Thromb. Haemost. 1992, 68, 577–582. [Google Scholar] [PubMed]
- Song, Y.; Guallar, V.; Baker, N.A. Molecular dynamics simulations of salicylate effects on the micro- and mesoscopic properties of a dipalmitoylphosphatidylcholine bilayer. Biochemistry 2005, 44, 13425–13438. [Google Scholar] [CrossRef] [PubMed]
- Khandelia, H.; Witzke, S.; Mouritsen, O.G. Interaction of salicylate and a terpenoid plant extract with model membranes: Reconciling experiments and simulations. Biophys. J. 2010, 99, 3887–3894. [Google Scholar] [CrossRef] [PubMed]
- Lúcio, M.; Ferreira, H.; Lima, J.L.; Reis, S. Interactions between oxicams and membrane bilayers: An explanation for their different COX selectivity. Med. Chem. 2006, 2, 447–456. [Google Scholar] [CrossRef] [PubMed]
- Tavolari, S.; Munarini, A.; Storci, G.; Laufer, S.; Chieco, P.; Guarnieri, T. The decrease of cell membrane fluidity by the non-steroidal anti-inflammatory drug licofelone inhibits epidermal growth factor receptor signalling and triggers apoptosis in HCA-7 colon cancer cells. Cancer Lett. 2012, 321, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Sade, A.; Tunçay, S.; Cimen, I.; Severcan, F.; Banerjee, S. Celecoxib reduces fluidity and decreases metastatic potential of colon cancer cell lines irrespective of COX-2 expression. Biosci. Rep. 2012, 32, 35–44. [Google Scholar] [PubMed]
- Nunes, C.; Lopes, D.; Pinheiro, M.; Pereira-Leite, C.; Reis, S. In vitro assessment of NSAIDs-membrane interactions: Significance for pharmacological actions. Pharm. Res. 2013, 30, 2097–2107. [Google Scholar] [CrossRef] [PubMed]
- Nie, X.; Wen, Z.Y.; Yan, Z.Y.; Huang, L.; Sun, D.; Cheng, B. Effects of morphine on rheological properties of rat red blood cells. Clin. Hemorheol. Microcirc. 2000, 22, 189–195. [Google Scholar] [PubMed]
- García, D.A.; Bujons, J.; Vale, C.; Suñol, C. Allosteric positive interaction of thymol with the GABAA receptor in primary cultures of mouse cortical neurons. Neuropharmacology 2006, 50, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, H.; Mizogami, M. Comparative interactions of anesthetic alkylphenols with lipid membranes. Open J. Anesthesiol. 2014, 4, 308–317. [Google Scholar] [CrossRef]
- Daoud, S.S. Cell membranes as targets for anti-cancer drug action. Anticancer Drugs 1992, 3, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Muller, C.P.; Krueger, G.R. Modulation of membrane proteins by vertical phase separation and membrane lipid fluidity. Basis for a new approach to tumor immunotherapy. Anticancer Res. 1986, 6, 1181–1193. [Google Scholar] [PubMed]
- Czapla, K.; Korchowiec, B.; Rogalska, E. Differentiating oxicam nonsteroidal anti-inflammatory drugs in phosphoglyceride monolayers. Langmuir 2010, 26, 3485–3492. [Google Scholar] [CrossRef] [PubMed]
- Baritaki, S.; Apostolakis, S.; Kanellou, P.; Dimanche-Boitrel, M.T.; Spandidos, D.A.; Bonavida, B. Reversal of tumor resistance to apoptotic stimuli by alteration of membrane fluidity: Therapeutic implications. Adv. Cancer Res. 2007, 98, 149–190. [Google Scholar] [PubMed]
- Dimanche-Boitrel, M.T.; Meurette, O.; Rebillard, A.; Lacour, S. Role of early plasma membrane events in chemotherapy-induced cell death. Drug Resist. Updat. 2005, 8, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Rebillard, A.; Lagadic-Gossmann, D.; Dimanche-Boitrel, M.T. Cisplatin cytotoxicity: DNA and plasma membrane targets. Curr. Med. Chem. 2008, 15, 2656–2663. [Google Scholar] [CrossRef] [PubMed]
- Marutaka, M.; Iwagaki, H.; Suguri, T.; Tanaka, N.; Orita, K. Alterations of membrane fluidity in K562 cells exposed to the anticancer drug adriamycin. Res. Commun. Mol. Pathol. Pharmacol. 1994, 85, 163–170. [Google Scholar] [PubMed]
- Marczak, A.; Kowalczyk, A.; Wrzesień-Kuś, A.; Robak, T.; Jóźwiak, Z. Interaction of doxorubicin and idarubicin with red blood cells from acute myeloid leukemia patients. Cell Biol. Int. 2006, 30, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Mandlekar, S.; Kong, A.N. Mechanisms of tamoxifen-induced apoptosis. Apoptosis 2001, 6, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Cronier, F.; Patenaude, A.; C-Gaudreault, R.; Auger, M. Membrane composition modulates the interaction between a new class of antineoplastic agents deriving from aromatic 2-chloroethylureas and lipid bilayers: A solid-state NMR study. Chem. Phys. Lipids 2007, 146, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Grunicke, H.; Grünewald, K.; Helliger, W.; Scheidl, F.; Wolff-Schreiner, E.; Puschendorf, B. Inhibition of tumor growth by an alkylation of the plasma membrane. Adv. Enzyme Regul. 1983, 21, 21–30. [Google Scholar] [CrossRef]
- Bernsdorff, C.; Reszka, R.; Winter, R. Interaction of the anticancer agent Taxol (paclitaxel) with phospholipid bilayers. J. Biomed. Mater. Res. 1999, 46, 141–149. [Google Scholar] [CrossRef]
- Houari, A.; di Martino, P. Effect of chlorhexidine and benzalkonium chloride on bacterial biofilm formation. Lett. Appl. Microbiol. 2007, 45, 652–656. [Google Scholar] [CrossRef] [PubMed]
- Takasaki, A.; Hashida, T.; Kato, K.; Moriyama, T.; Nishihara, T. Action of a quaternary ammonium disinfectant on cell membrane of Staphylococcus aureus. Jpn. J. Toxicol. Environ. Health 1994, 40, 520–526. [Google Scholar] [CrossRef]
- Bensikaddour, H.; Snoussi, K.; Lins, L.; van Bambeke, F.; Tulkens, P.M.; Brasseur, R.; Goormaghtigh, E.; Mingeot-Leclercq, M.P. Interactions of ciprofloxacin with DPPC and DPPG: Fluorescence anisotropy, ATR-FTIR and 31P NMR spectroscopies and conformational analysis. Biochim. Biophys. Acta 2008, 1778, 2535–2543. [Google Scholar] [CrossRef] [PubMed]
- Van Bambeke, F.; Mingeot-Leclercq, M.P.; Schanck, A.; Brasseur, R.; Tulkens, P.M. Alterations in membrane permeability induced by aminoglycoside antibiotics: Studies on liposomes and cultured cells. Eur. J. Pharmacol. 1993, 247, 155–168. [Google Scholar] [CrossRef]
- Berquand, A.; Fa, N.; Dufrêne, Y.F.; Mingeot-Leclercq, M.P. Interaction of the macrolide antibiotic azithromycin with lipid bilayers: Effect on membrane organization, fluidity, and permeability. Pharm. Res. 2005, 22, 465–475. [Google Scholar] [CrossRef] [PubMed]
- Tyteca, D.; Schanck, A.; Dufrêne, Y.F.; Deleu, M.; Courtoy, P.J.; Tulkens, P.M.; Mingeot-Leclercq, M.P. The macrolide antibiotic azithromycin interacts with lipids and affects membrane organization and fluidity: Studies on Langmuir-Blodgett monolayers, liposomes and J774 macrophages. J. Membr. Biol. 2003, 192, 203–215. [Google Scholar] [CrossRef] [PubMed]
- Yasui, K.; Masuda, M.; Matsuoka, T.; Yamazaki, M.; Komiyama, A.; Akabane, T.; Murata, K. Miconazole and amphotericin B alter polymorphonuclear leukocyte functions and membrane fluidity in similar fashions. Antimicrob. Agents Chemother. 1988, 32, 1864–1868. [Google Scholar] [CrossRef] [PubMed]
- Virág, E.; Belagyi, J.; Gazdag, Z.; Vágvölgyi, C.; Pesti, M. Direct in vivo interaction of the antibiotic primycin with the plasma membrane of Candida albicans: An EPR study. Biochim. Biophys. Acta 2012, 1818, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Tai, W.Y.; Yang, Y.C.; Lin, H.J.; Huang, C.P.; Cheng, Y.L.; Chen, M.F.; Yen, H.L.; Liau, I. Interplay between structure and fluidity of model lipid membranes under oxidative attack. J. Phys. Chem. B 2010, 114, 15642–15649. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, H. Lipid peroxidation-inhibitory effects of perioperatively used drugs associated with their membrane interactions. Oxid. Antioxid. Med. Sci. 2014, 3, 91–98. [Google Scholar] [CrossRef]
- Lúcio, M.; Ferreira, H.; Lima, J.L.; Reis, S. Use of liposomes to evaluate the role of membrane interactions on antioxidant activity. Anal. Chim. Acta 2007, 597, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, M.E.; García, A.F.; Africa de Madariaga, M.; Sagrista, M.L.; Casadó, F.J.; Mora, M. Interaction of tocopherols and phenolic compounds with membrane lipid components: Evaluation of their antioxidant activity in a liposomal model system. Life Sci. 2003, 72, 2337–2360. [Google Scholar] [CrossRef]
- Arora, A.; Byrem, T.M.; Nair, M.G.; Strasburg, G.M. Modulation of liposomal membrane fluidity by flavonoids and isoflavonoids. Arch. Biochem. Biophys. 2000, 373, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Vlasic, N.; Medow, M.S.; Schwarz, S.M.; Pritchard, K.A., Jr.; Stemerman, M.B. Lipid fluidity modulates platelet aggregation and agglutination in vitro. Life Sci. 1993, 53, 1053–1060. [Google Scholar] [CrossRef]
- Rao, G.H.; White, J.G. Disaggregation and reaggregation of “irreversibly” aggregated platelets: A method for more complete evaluation of anti-platelet drugs. Agents Actions 1985, 16, 425–434. [Google Scholar] [CrossRef] [PubMed]
- Sheu, J.R.; Hung, W.C.; Lee, L.W.; Chang, P.T.; Kan, Y.C.; Yen, M.H. Mechanism involved in the antiplatelet activity of naloxone in human platelets. Biochem. Biophys. Res. Commun. 1997, 231, 12–16. [Google Scholar] [CrossRef] [PubMed]
- Matera, C.; Falzarano, C.; Vacca, C.; Falciani, M.; Rossi, F. Effects of some antineoplastic drugs (vincristine, doxorubicin and epirubicin) on human platelet aggregation. J. Med. 1994, 25, 2–16. [Google Scholar] [CrossRef]
- Chang, Y.; Chen, T.L.; Wu, G.J.; Hsiao, G.; Shen, M.Y.; Lin, K.H.; Chou, D.S.; Lin, C.H.; Sheu, J.R. Mechanisms involved in the antiplatelet activity of ketamine in human platelets. J. Biomed. Sci. 2004, 11, 764–772. [Google Scholar] [CrossRef] [PubMed]
- Sheu, J.R.; Hsiao, G.; Luk, H.N.; Chen, Y.W.; Chen, T.L.; Lee, L.W.; Lin, C.H.; Chou, D.S. Mechanisms involved in the antiplatelet activity of midazolam in human platelets. Anesthesiology 2002, 96, 651–658. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, H. Effects of red wine flavonoid components on biomembranes and cell proliferation. Int. J. Wine Res. 2011, 3, 9–17. [Google Scholar] [CrossRef]
- Wu, T.; He, M.; Zang, X.; Zhou, Y.; Qiu, T.; Pan, S.; Xu, X. A structure-activity relationship study of flavonoids as inhibitors of E. coli by membrane interaction effect. Biochim. Biophys. Acta 2013, 1828, 2751–2756. [Google Scholar] [CrossRef] [PubMed]
- Ajdzanović, V.; Spasojević, I.; Filipović, B.; Sosić-Jurjević, B.; Sekulić, M.; Milosević, V. Effects of genistein and daidzein on erythrocyte membrane fluidity: An electron paramagnetic resonance study. Can. J. Physiol. Pharmacol. 2010, 88, 497–500. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, H. Structure-dependent membrane interaction of flavonoids associated with their bioactivity. Food Chem. 2010, 120, 1089–1096. [Google Scholar] [CrossRef]
- Margina, D.; Ilie, M.; Manda, G.; Neagoe, I.; Mocanu, M.; Ionescu, D.; Gradinaru, D.; Ganea, C. Quercetin and epigallocatechin gallate effects on the cell membranes biophysical properties correlate with their antioxidant potential. Gen. Physiol. Biophys. 2012, 31, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Suarez, J.M.; Giampieri, F.; González-Paramás, A.M.; Damiani, E.; Astolfi, P.; Martinez-Sanchez, G.; Bompadre, S.; Quiles, J.L.; Santos-Buelga, C.; Battino, M. Phenolics from monofloral honeys protect human erythrocyte membranes against oxidative damage. Food Chem. Toxicol. 2012, 50, 1508–1516. [Google Scholar] [CrossRef] [PubMed]
- Selvaraj, S.; Krishnaswamy, S.; Devashya, V.; Sethuraman, S.; Krishnan, U.M. Influence of membrane lipid composition on flavonoid-membrane interactions: Implications on their biological activity. Prog. Lipid Res. 2015, 58, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, H. Effects of green tea catechins on membrane fluidity. Pharmacology 1999, 59, 34–44. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, H. Stereospecificity in membrane effects of catechins. Chem. Biol. Interact. 2001, 134, 41–54. [Google Scholar] [CrossRef]
- Tsuchiya, H.; Sato, M.; Kato, H.; Okubo, T.; Juneja, L.R.; Kim, M. Simultaneous determination of catechins in human saliva by high-performance liquid chromatography. J. Chromatogr. B 1998, 703, 253–258. [Google Scholar] [CrossRef]
- Cyboran, S.; Strugała, P.; Włoch, A.; Oszmiański, J.; Kleszczyńska, H. Concentrated green tea supplement: Biological activity and molecular mechanisms. Life Sci. 2015, 126, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Bard, M.; Albrecht, M.R.; Gupta, N.; Guynn, C.J.; Stillwell, W. Geraniol interferes with membrane functions in strains of Candida and Saccharomyces. Lipids 1988, 23, 534–538. [Google Scholar] [CrossRef] [PubMed]
- Nowotarska, S.W.; Nowotarski, K.J.; Friedman, M.; Situ, C. Effect of structure on the interactions between five natural antimicrobial compounds and phospholipids of bacterial cell membrane on model monolayers. Molecules 2014, 19, 7497–7515. [Google Scholar] [CrossRef] [PubMed]
- Reiner, G.N.; Fraceto, L.F.; de Paula, E.; Perillo, M.A.; García, D.A. Effects of gabaergic phenols on phospholipid bilayers as evaluated by 1H-NMR. J. Biomater. Nanobiotechnol. 2013, 4, 28–34. [Google Scholar] [CrossRef]
- Reiner, G.N.; Delgado-Marín, L.; Olguín, N.; Sánchez-Redondo, S.; Sánchez-Borzone, M.; Rodríguez-Farré, E.; Suñol, C.; García, D.A. Gabaergic pharmacological activity of propofol related compounds as possible enhancers of general anesthetics and interaction with membranes. Cell Biochem. Biophys. 2013, 67, 515–525. [Google Scholar] [CrossRef] [PubMed]
- Mendanha, S.A.; Moura, S.S.; Anjos, J.L.; Valadares, M.C.; Alonso, A. Toxicity of terpenes on fibroblast cells compared to their hemolytic potential and increase in erythrocyte membrane fluidity. Toxicol. In Vitro 2013, 27, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Zengin, H.; Baysal, A.H. Antibacterial and antioxidant activity of essential oil terpenes against pathogenic and spoilage-forming bacteria and cell structure-activity relationships evaluated by SEM microscopy. Molecules 2014, 19, 17773–17798. [Google Scholar] [CrossRef] [PubMed]
- Nomura, T.; Kurihara, K. Liposomes as a model for olfactory cells: Changes in membrane potential in response to various odorants. Biochemistry 1987, 26, 6135–6140. [Google Scholar] [CrossRef] [PubMed]
- Yin, Q.; Shi, X.; Ding, H.; Dai, X.; Wan, G.; Qiao, Y. Interactions of borneol with DPPC phospholipid membranes: A molecular dynamics simulation study. Int. J. Mol. Sci. 2014, 15, 20365–20381. [Google Scholar] [PubMed]
- DeFeudis, F.V.; Drieu, K. Ginkgo biloba extract (EGb 761) and CNS functions: Basic studies and clinical applications. Curr. Drug Targets 2000, 1, 25–58. [Google Scholar] [PubMed]
- Marín, L.D.; Sánchez-Borzone, M.; García, D.A. Comparative antioxidant properties of some GABAergic phenols and related compounds, determined for homogeneous and membrane systems. Med. Chem. 2011, 7, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Camargos, H.S.; Moreira, R.A.; Mendanha, S.A.; Fernandes, K.S.; Dorta, M.L.; Alonso, A. Terpenes increase the lipid dynamics in the Leishmania plasma membrane at concentrations similar to their IC50 values. PLoS ONE 2014, 9, e104429. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, H.; Nagayama, M.; Tanaka, T.; Furusawa, M.; Kashimata, M.; Takeuchi, H. Membrane-rigidifying effects of anti-cancer dietary factors. Biofactors 2002, 16, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Selvaraj, S.; Mohan, A.; Narayanan, S.; Sethuraman, S.; Krishnan, U.M. Dose-dependent interaction of trans-resveratrol with biomembranes: Effects on antioxidant property. J. Med. Chem. 2013, 56, 970–981. [Google Scholar] [CrossRef] [PubMed]
- Brittes, J.; Lúcio, M.; Nunes, C.; Lima, J.L.; Reis, S. Effects of resveratrol on membrane biophysical properties: Relevance for its pharmacological effects. Chem. Phys. Lipids 2010, 163, 747–754. [Google Scholar] [CrossRef] [PubMed]
- Wesołowska, O.; Kużdżał, M.; Štrancar, J.; Michalak, K. Interaction of the chemopreventive agent resveratrol and its metabolite, peceatannol, with model membranes. Biochim. Biophys. Acta 2009, 1788, 1851–1860. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Hurdle, J.G.; Lee, R.; Lee, R.; Cushman, M.; Pezzuto, J.M. Evaluation of flavonoid and resveratrol chemical libraries reveals abyssinone II as a promising antibacterial lead. ChemMedChem 2012, 7, 1541–1545. [Google Scholar] [CrossRef] [PubMed]
- Meddings, J.B.; Hogaboam, C.M.; Tran, K.; Reynolds, J.D.; Wallace, J.L. Capsaicin effects on non-neuronal plasma membranes. Biochim. Biophys. Acta 1991, 1070, 43–50. [Google Scholar] [CrossRef]
- Tsuchiya, H. Biphasic membrane effects of capsaicin, an active component in Capsicum species. J. Ethnopharmacol. 2001, 75, 295–299. [Google Scholar] [CrossRef]
- Kempaiah, R.K.; Srinivasan, K. Influence of dietary spices on the fluidity of erythrocytes in hypercholesterolaemic rats. Br. J. Nutr. 2005, 93, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Prakash, U.N.; Srinivasan, K. Beneficial influence of dietary spices on the ultrastructure and fluidity of the intestinal brush border in rats. Br. J. Nutr. 2010, 104, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Binshtok, A.M.; Bean, B.P.; Woolf, C.J. Inhibition of nociceptors by TRPV1-mediated entry of impermeant sodium channel blockers. Nature 2007, 449, 607–610. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, H.; Mizogami, M. Plant components exhibit pharmacological activities and drug interactions by acting on lipid membranes. Pharmacogn. Commun. 2012, 2, 58–71. [Google Scholar]
- Asgarpanah, J. Phytochemistry, pharmacology and medicinal properties of Hypericum perforatum L. Afr. J. Pharm. Pharmacol. 2012, 6, 1387–1394. [Google Scholar] [CrossRef]
- Eckert, G.P.; Müller, W.E. Effects of hyperforin on the fluidity of brain membranes. Pharmacopsychiatry 2001, 34, S22–S25. [Google Scholar] [CrossRef] [PubMed]
- Eckert, G.P.; Keller, J.H.; Jourdan, C.; Karas, M.; Volmer, D.A.; Schubert-Zsilavecz, M.; Müller, W.E. Hyperforin modifies neuronal membrane properties in vivo. Neurosci. Lett. 2004, 367, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Chaloupka, R.; Obšil, T.; Plášek, J.; Sureau, F. The effect of hypericin and hypocrellin-A on lipid membranes and membrane potential of 3T3 fibroblasts. Biochim. Biophys. Acta 1999, 1418, 39–47. [Google Scholar] [CrossRef]
- Kraus, B.; Wolff, H.; Heilmann, J.; Elstner, E.F. Influence of Hypericum perforatum extract and its single compounds on amyloid-β mediated toxicity in microglial cells. Life Sci. 2007, 81, 884–894. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, H.; Nagayama, M. Garlic allyl derivatives interact with membrane lipids to modify the membrane fluidity. J. Biomed. Sci. 2008, 15, 653–660. [Google Scholar] [CrossRef] [PubMed]
- Debouzy, J.C.; Neumann, J.M.; Hervé, M.; Daveloose, D.; Viret, J.; Apitz-Castro, R. Interaction of antiaggregant molecule ajoene with membranes. An ESR and 1H-, 2H-, 31P-NMR study. Eur. Biophys. J. 1989, 17, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Rendu, F.; Daveloose, D.; Debouzy, J.C.; Bourdeau, N.; Levy-Toledano, S.; Jain, M.K.; Apitz-Castro, R. Ajoene, the antiplatelet compound derived from garlic, specifically inhibits platelet release reaction by affecting the plasma membrane internal microviscosity. Biochem. Pharmacol. 1989, 38, 1321–1328. [Google Scholar] [CrossRef]
- Knowles, L.M.; Milner, J.A. Allyl sulfides modify cell growth. Drug Metabol. Drug Interact. 2000, 17, 81–107. [Google Scholar] [CrossRef] [PubMed]
- Rosenkranz, V.; Wink, M. Alkaloids induce programmed cell death in bloodstream forms of trypanosomes (Trypanosoma b. brucei). Molecules 2008, 13, 2462–2473. [Google Scholar] [CrossRef] [PubMed]
- Moloudizargari, M.; Mikaili, P.; Aghajanshakeri, S.; Asghari, M.H.; Shayegh, J. Pharmacological and therapeutic effects of Peganum harmala and its main alkaloids. Pharmacogn. Rev. 2013, 7, 199–212. [Google Scholar] [CrossRef] [PubMed]
- Cao, R.; Peng, W.; Wang, Z.; Xu, A. β-Carboline alkaloids: Biochemical and pharmacological functions. Curr. Med. Chem. 2007, 14, 479–500. [Google Scholar] [CrossRef] [PubMed]
- Peura, P.; Mackenzie, P.; Koivusaari, U.; Lang, M. Increased fluidity of a model membrane caused by tetrahydro-β-carbolines. Mol. Pharmacol. 1982, 22, 721–724. [Google Scholar] [PubMed]
- Tsuchiya, H.; Ohmoto, S. Comparative effects of β-carbolines on platelet aggregation and lipid membranes. Pharmacol. Rep. 2010, 62, 689–695. [Google Scholar] [CrossRef]
- Tsuchiya, H. Biphasic effects of acetaldehyde-biogenic amine condensation products on membrane fluidity. J. Pharm. Pharmacol. 2001, 53, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, H. Inhibition of membrane effects of general anesthetic propofol by benzodiazepine inverse agonist tetrahydro-β-carboline. Int. J. Pharmacol. 2012, 8, 542–548. [Google Scholar] [CrossRef]
- Tsuchiya, H.; Mizogami, M. Drinking-related tetrahydroharmans counteract the membrane effects of local anesthetic lidocaine. J. Drug Alcohol Res. 2014, 3. [Google Scholar] [CrossRef]
- Budai, M.; Szabó, Z.; Szogyi, M.; Gróf, P. Molecular interactions between DPPC and morphine derivatives: A DSC and ESR study. Int. J. Pharm. 2003, 250, 239–250. [Google Scholar] [CrossRef]
- Khajuria, A.; Thusu, N.; Zutshi, U. Piperine modulates permeability characteristics of intestine by inducing alterations in membrane dynamics: Influence on brush border membrane fluidity, ultrastructure and enzyme kinetics. Phytomedicine 2002, 9, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Gąsiorowska, J.; Wesołowska, O.; Michalak, K. Interaction of plant alkaloid, berberine, with zwitterionic and negatively charged phospholipid bilayers. Curr. Top. Biophys. 2011, 34, 45–51. [Google Scholar] [CrossRef]
- Alves, D.S.; Pérez-Fons, L.; Estepa, A.; Micol, V. Membrane-related effects underlying the biological activity of the anthraquinones emodin and barbaloin. Biochem. Pharmacol. 2004, 68, 549–561. [Google Scholar] [CrossRef] [PubMed]
- Duarte, E.L.; Oliveira, T.R.; Alves, D.S.; Micol, V.; Lamy, M.T. On the interaction of the anthraquinone barbaloin with negatively charged DMPG bilayers. Langmuir 2008, 24, 4041–4049. [Google Scholar] [CrossRef] [PubMed]
- Yi, J.S.; Choo, H.J.; Cho, B.R.; Kim, H.M.; Kim, Y.N.; Ham, Y.M.; Ko, Y.G. Ginsenoside Rh2 induces ligand-independent Fas activation via lipid raft disruption. Biochem. Biophys. Res. Commun. 2009, 385, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.M.; Cao, Y.L.; Dou, D.Q. Protective effect of ginsenoside-Re against cerebral ischemia/reperfusion damage in rats. Biol. Pharm. Bull. 2006, 29, 2502–2505. [Google Scholar] [CrossRef] [PubMed]
- Kwon, H.Y.; Kim, E.H.; Kim, S.W.; Kim, S.N.; Park, J.D.; Rhee, D.K. Selective toxicity of ginsenoside Rg3 on multidrug resistant cells by membrane fluidity modulation. Arch. Pharm. Res. 2008, 31, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Tachikawa, E.; Kudo, K.; Nunokawa, M.; Kashimoto, T.; Takahashi, E.; Kitagawa, S. Characterization of ginseng saponin ginsenoside-Rg3 inhibition of catecholamine secretion in bovine adrenal chromaffin cells. Biochem. Pharmacol. 2001, 62, 943–951. [Google Scholar] [CrossRef]
- Han, S.K.; Ko, Y.I.; Park, S.J.; Jin, I.J.; Kim, Y.M. Oleanolic acid and ursolic acid stabilize liposomal membranes. Lipids 1997, 32, 769–773. [Google Scholar] [CrossRef] [PubMed]
- Broniatowski, M.; Flasiński, M.; Ziȩba, K.; Miśkowiec, P. Interactions of pentacyclic triterpene acids with cardiolipins and related phosphatidylglycerols in model systems. Biochim. Biophys. Acta 2014, 1838, 2530–2538. [Google Scholar] [CrossRef] [PubMed]
- Jaruga, E.; Sokal, A.; Chrul, S.; Bartosz, G. Apoptosis-independent alterations in membrane dynamics induced by curcumin. Exp. Cell Res. 1998, 245, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Atsumi, T.; Fujisawa, S.; Tonosaki, K. Relationship between intracellular ROS production and membrane mobility in curcumin- and tetrahydrocurcumin-treated human gingival fibroblasts and human submandibular gland carcinoma cells. Oral Dis. 2005, 11, 236–242. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Lee, C.C.; Hung, W.C.; Chen, F.Y.; Lee, M.T.; Huang, H.W. The bound states of amphipathic drugs in lipid bilayers: Study of curcumin. Biophys. J. 2008, 95, 2318–2324. [Google Scholar] [CrossRef] [PubMed]
- Hung, W.C.; Chen, F.Y.; Lee, C.C.; Sun, Y.; Lee, M.T.; Huang, H.W. Membrane-thinning effect of curcumin. Biophys. J. 2008, 94, 4331–4338. [Google Scholar] [CrossRef] [PubMed]
- Bohn, T. Dietary factors affecting polyphenol bioavailability. Nutr. Rev. 2014, 72, 429–452. [Google Scholar] [PubMed]
- Ma, Y.; Zeng, M.; Sun, R.; Hu, M. Disposition of flavonoids impacts their efficacy and safety. Curr. Drug Metab. 2014, 15, 841–864. [Google Scholar] [CrossRef] [PubMed]
- Thilakarathna, S.H.; Rupasinghe, H.P. Flavonoid bioavailability and attempts for bioavailability enhancement. Nutrients 2013, 5, 3367–3387. [Google Scholar] [CrossRef] [PubMed]
- Murota, K.; Terao, J. Antioxidative flavonoid quercetin: Implication of its intestinal absorption and metabolism. Arch. Biochem. Biophys. 2003, 417, 12–17. [Google Scholar] [CrossRef]
- Goldberg, D.M.; Yan, J.; Soleas, G.J. Absorption of three wine-related polyphenols in three different matrices by healthy subjects. Clin. Biochem. 2003, 36, 79–87. [Google Scholar] [CrossRef]
- Oh, H.J.; Shin, K.C.; Oh, D.K. Production of ginsenosides Rg1 and Rh1 by hydrolyzing the outer glycoside at the C-6 position in protopanaxatriol-type ginsenosides using β-glucosidase from Pyrococcus furiosus. Biotechnol. Lett. 2014, 36, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.; Jang, J.Y.; Park, D.; Yon, J.M.; Baek, I.J.; Hwang, B.Y.; Nam, S.Y.; Yun, Y.W.; Kim, K.Y.; Joo, S.S.; et al. Korean red ginseng extract does not cause embryo-fetal death or abnormalities in mice. Birth Defects Res. B Dev. Reprod. Toxicol. 2010, 89, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Fang, X.L. Difference in oral absorption of ginsenoside Rg1 between in vitro and in vivo models. Acta Pharmacol. Sin. 2006, 27, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.K. Pharmacokinetics of ginsenoside Rb1 and its metabolite compound K after oral administration of Korean Red Ginseng extract. J. Ginseng Res. 2013, 37, 451–456. [Google Scholar] [CrossRef] [PubMed]
- Ingólfsson, H.I.; Thakur, P.; Herold, K.F.; Hobart, E.A.; Ramsey, N.B.; Periole, X.; de Jong, D.H.; Zwama, M.; Yilmaz, D.; Hall, K.; et al. Phytochemicals perturb membranes and promiscuously alter protein function. ACS Chem. Biol. 2014, 9, 1788–1798. [Google Scholar] [CrossRef] [PubMed]
- Hendrich, A. Flavonoid-membrane interactions: Possible consequences for biological effects of some polyphenolic compounds. Acta Pharmacol. Sin. 2006, 27, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Erlejman, A.G.; Verstraeten, S.V.; Fraga, C.G.; Oteiza, P.I. The interaction of flavonoids with membranes: Potential determinant of flavonoid antioxidant effects. Free Radic. Res. 2004, 38, 1311–1320. [Google Scholar] [CrossRef] [PubMed]
- Sinha, R.; Srivastava, S.; Joshi, A.; Joshi, U.J.; Govil, G. In-vitro anti-proliferative and anti-oxidant activity of galangin, fisetin and quercetin: Role of localization and intermolecular interaction in model membrane. Eur. J. Med. Chem. 2014, 79, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Phan, H.T.; Yoda, T.; Chahal, B.; Morita, M.; Takagi, M.; Vestergaard, M.C. Structure-dependent interactions of polyphenols with a biomimetic membrane system. Biochim. Biophys. Acta 2014, 1838, 2670–2677. [Google Scholar] [CrossRef] [PubMed]
- Furusawa, M.; Tsuchiya, H.; Nagayama, M.; Tanaka, T.; Nakaya, K.; Iinuma, M. Anti-platelet and membrane-rigidifying flavonoids in brownish scale of onion. J. Health Sci. 2003, 49, 475–480. [Google Scholar] [CrossRef]
- Furusawa, M.; Tsuchiya, H.; Nagayama, M.; Tanaka, T.; Oyama, M.; Ito, T.; Iinuma, M.; Takeuchi, H. Cell growth inhibition by membrane-active components in brownish scale of onion. J. Health Sci. 2006, 52, 578–584. [Google Scholar] [CrossRef]
- Tsuchiya, H.; Tanaka, T.; Nagayama, M.; Oyama, M.; Iinuma, M. Membrane activity-guided isolation of antiproliferative and antiplatelet constituent from Evodiopanax innovans. Nat. Prod. Commun. 2008, 3, 809–814. [Google Scholar]
- Oyedapo, O.O.; Akinpelu, B.A.; Akinwunmi, K.F.; Adeyinka, M.O.; Sipeolu, F.O. Red blood cell membrane stabilizing potentials of extracts of Lantana camara and its fractions. Int. J. Plant Physiol. Biochem. 2010, 2, 46–51. [Google Scholar]
- Raimi, M.M.; Oyedapo, O.O. Bioactivity-guided evaluation of the root extract of Plumbago zeylanica. Int. J. Biol. Chem. Sci. 2009, 3, 672–680. [Google Scholar] [CrossRef]
- Gadamsetty, G.; Maru, S.; Tyagi, A.; Chakravarthula, S.N. Anti-inflammatory, cytotoxic and antioxidant effects of methanolic extracts of Drypetes Sepiaria (Euphorbiaceae). Afr. J. Tradit. Complement. Altern. Med. 2013, 10, 274–282. [Google Scholar] [CrossRef] [PubMed][Green Version]
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsuchiya, H. Membrane Interactions of Phytochemicals as Their Molecular Mechanism Applicable to the Discovery of Drug Leads from Plants. Molecules 2015, 20, 18923-18966. https://doi.org/10.3390/molecules201018923
Tsuchiya H. Membrane Interactions of Phytochemicals as Their Molecular Mechanism Applicable to the Discovery of Drug Leads from Plants. Molecules. 2015; 20(10):18923-18966. https://doi.org/10.3390/molecules201018923
Chicago/Turabian StyleTsuchiya, Hironori. 2015. "Membrane Interactions of Phytochemicals as Their Molecular Mechanism Applicable to the Discovery of Drug Leads from Plants" Molecules 20, no. 10: 18923-18966. https://doi.org/10.3390/molecules201018923
APA StyleTsuchiya, H. (2015). Membrane Interactions of Phytochemicals as Their Molecular Mechanism Applicable to the Discovery of Drug Leads from Plants. Molecules, 20(10), 18923-18966. https://doi.org/10.3390/molecules201018923





