A Novel Synthesis of the Oxazolidinone Antithrombotic Agent Rivaroxaban
Abstract
:1. Introduction
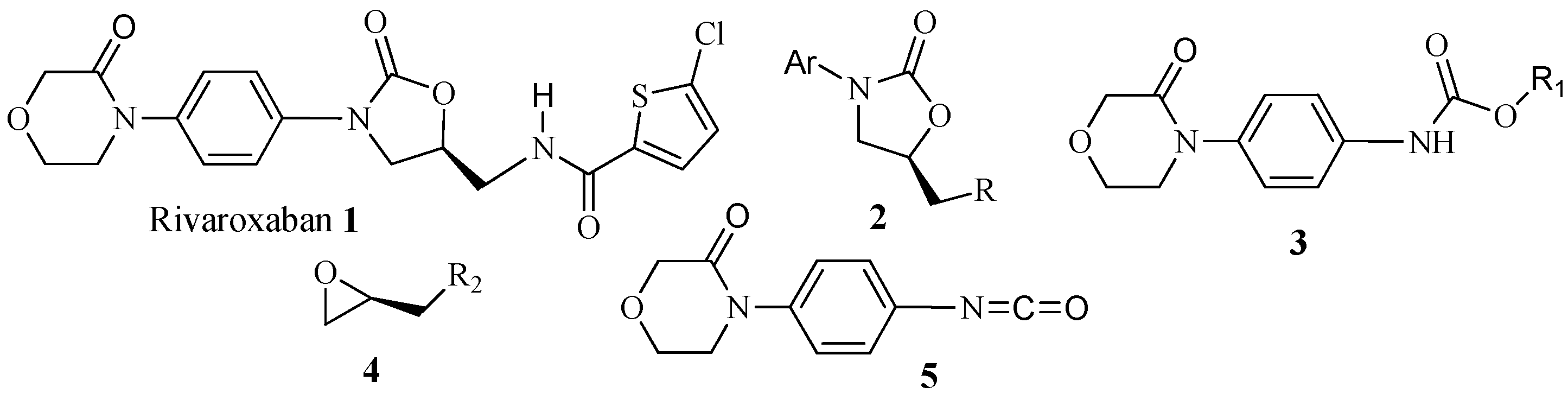
2. Results and Discussion

3. Experimental Section
General Information
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Furie, B.; Furie, B.C. Mechanisms of thrombus formation. N. Engl. J. Med. 2008, 359, 938–949. [Google Scholar] [CrossRef] [PubMed]
- Mederski, W.; Cezanne, B.; Amsterdam, C.V.; Buhring, K.; Dorsch, D.; Gleitz, J.; Marz, J.; Tasklakidis, C. Chlorothiophene carboxamides as P1 surrogates of inhibitors of blood coagulation factor Xa. Bioorg. Med. Chem. Lett. 2004, 14, 5817–5822. [Google Scholar] [CrossRef] [PubMed]
- Ansell, J. Factor Xa or thrombin: Is factor Xa a better target? J. Thromb. Haemost. 2007, 5, 60–64. [Google Scholar] [CrossRef]
- Bauersachs, R.; Berkowitz, S.D.; Brenner, B.; Buller, H.R.; Decousus, H.; Gallus, A.S.; Lensing, A.W.; Misselwitz, F.; Prins, M.H.; Raskob, G.E.; et al. Oral rivaroxaban for symptomatic venous thromboembolism. N. Engl. J. Med. 2010, 363, 2499–2510. [Google Scholar] [CrossRef] [PubMed]
- Brickner, S.J.; Hutchinson, D.K.; Barbachyn, M.R.; Manninen, P.R.; Ulanowicz, D.A.; Garmon, S.A.; Grega, K.C.; Hendges, S.K.; Toops, D.S.; Ford, C.W.; et al. Synthesis and Antibacterial Activity of U-100592 and U-100766, Two Oxazolidinone Antibacterial Agents for the Potential Treatment of Multidrug-Resistant Gram-Positive Bacterial Infections. J. Med. Chem. 1996, 39, 673–679. [Google Scholar] [CrossRef] [PubMed]
- Thomas, C.R. A Process for the Preparation of Rivaroxaban. DE 10300111, 6 May 2004. [Google Scholar]
- Prabhudas, B.; Gamini, W. Processes for the Preparation of Rivaroxaban and Intermediates Thereof. PCT WO 2010124385, 11 April 2010. [Google Scholar]
- Zhang, X.; Chen, W. An Efficient and Highly Regioselective Cycloaddition of Aryl Isocyanates with Oxiranes Promoted by MgI2 Etherate. Chem. Lett. 2010, 39, 527–529. [Google Scholar] [CrossRef]
- Li, C.; Liu, Y.; Zhang, Y.; Zhang, X. An approach to the anticoagulant agent rivaroxaban via an isocyanate-oxirane cycloaddition promoted by MgI2 etherate. J. Chem. Res. 2011, 35, 400–401. [Google Scholar] [CrossRef]
- Sturm, H.; Souza, D.D.; Knepper, K. Method for the Preparation of Rivaroxaban. PCT WO 2011098501, 18 August 2011. [Google Scholar]
- Klapars, A.; Antilla, J.C.; Huang, X.; Buchwald, S.L. A general and efficient copper catalyst for the amidation of aryl halides and the N-arylation of nitrogen heterocycles. J. Am. Chem. Soc. 2001, 123, 7727–7729. [Google Scholar] [CrossRef] [PubMed]
- Klapars, A.; Huang, X.; Buchwald, S.L. A general and efficient copper catalyst for the amidation of aryl halides. J. Am. Chem. Soc. 2002, 124, 7421–7428. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.K.; Kim, D.H.; Park, J.N. Copper-catalyzed N-arylation of aryl iodides with benzamides or nitrogen heterocycles in the Presence of ethylenediamine. Synlett 2002, 427–430. [Google Scholar] [CrossRef]
- Mallesham, B.; Rajesh, B.M.; Reddy, P.R.; Srinivas, D.; Trehan, S. Highly efficient CuI-catalyzed coupling of aryl bromides with oxazolidinones using buchwald’s protocol: A short route to linezolid and toloxatone. Org. Lett. 2003, 5, 963–965. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, I. Ueber Phenylirungen bei Gegenwart von Kupfer als Katalystor. Ber. Dtsch. Chem. Ges. 2006, 39, 1691–1692. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 1, 6–12 are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yuan, J.; Liu, K.; Li, L.; Yuan, Y.; Liu, X.; Li, Y. A Novel Synthesis of the Oxazolidinone Antithrombotic Agent Rivaroxaban. Molecules 2014, 19, 14999-15004. https://doi.org/10.3390/molecules190914999
Yuan J, Liu K, Li L, Yuan Y, Liu X, Li Y. A Novel Synthesis of the Oxazolidinone Antithrombotic Agent Rivaroxaban. Molecules. 2014; 19(9):14999-15004. https://doi.org/10.3390/molecules190914999
Chicago/Turabian StyleYuan, Jianyong, Kai Liu, Lun Li, Yong Yuan, Xuelei Liu, and Yanwu Li. 2014. "A Novel Synthesis of the Oxazolidinone Antithrombotic Agent Rivaroxaban" Molecules 19, no. 9: 14999-15004. https://doi.org/10.3390/molecules190914999
APA StyleYuan, J., Liu, K., Li, L., Yuan, Y., Liu, X., & Li, Y. (2014). A Novel Synthesis of the Oxazolidinone Antithrombotic Agent Rivaroxaban. Molecules, 19(9), 14999-15004. https://doi.org/10.3390/molecules190914999



