2.1. The Local Concentration of Sakuranetin is Estimated to be Superior in Infected Regions in a Resistant Rice Line than in a Susceptible Line
Sakuranetin is a rice flavonoid PA with a flavanone structure, and its increase by blast fungus infection has been reported by Kodama
et al. [
10] and Dillon
et al. [
11]. They used pre-wounded rice leaves for fungal inoculation. To evaluate the net increase in the PA levels caused by fungus infection, experiments should be done under natural infection conditions without any wound effect. Then, we spray-inoculated intact rice seedlings grown in soil with a spore suspension of blast fungus race 003, and the 4th leaves were subjected to sakuranetin quantification. In this experimental system, typical susceptible and resistant responses of host rice plants to the fungal type were observed [
9,
13]; blast fungus grows vigorously in the susceptible rice line Nipponbare (N), and susceptible type enlarged light brown lesions (ELs) become visible at 4 days post inoculation (dpi). On the other hand, in resistant rice line IL7, which was generated by introduction of a rice resistance gene
Pii to the fungus into N and back-crossings [
14], fungal growth is severely restricted during the early infection period [
9,
13] accompanying hypersensitive reaction (HR), which is a typical resistance response of host plants to restrict invasive pathogens to infected regions. HR generally accompanies formation of HR lesions (HRLs), which are very small size and dark brown and become visible at 2 dpi, increasing the number at 3 dpi [
9].
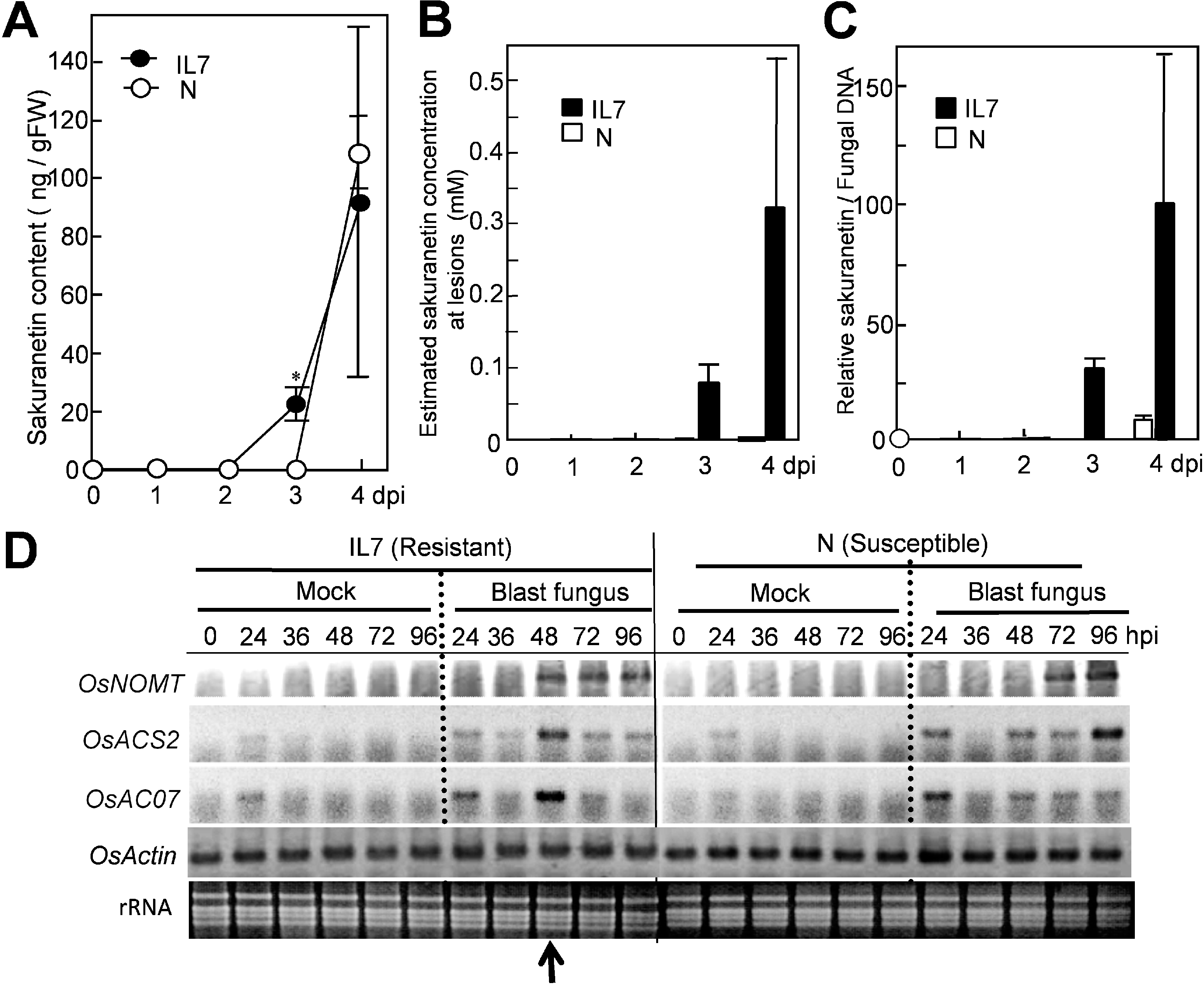
After spray-inoculation with a spore suspension, only a small amount of sakuranetin at a background level was found at 0–2 dpi in both susceptible N and resistant IL7 rice leaves (
Figure 1A). An increase in sakuranetin was first found in IL7 at 3 dpi, and was estimated to be 23 ng per g fresh leaf (closed circles), and the level increased to 92 ng at 4 dpi. In the N leaves, the increase was not detected at 3 dpi, and 108 ng was found at 4 dpi (open circles). The majority of PAs have been reported to accumulate locally at fungus-induced lesions [
15,
16], and the area occupied by HRLs at 3 dpi was roughly estimated to about 0.1% of the whole IL7 leaf area; around 50 HRLs with about 0.01 mm
2 each were formed in the leaf of 500 mm
2 [
9,
17] indicating that about 1,000-fold higher sakuranetin than the mean level is localized at HRLs. As the mean sakuranetin content in IL7 at 3 dpi was 23 ng per g of fresh leaf, the local concentration at HRLs was 23 μg per g leaf corresponding to near 0.1 mM (
Figure 1). At 4 dpi, the mean PA content was 92 ng per g of IL7 leaf, so the local concentration of HRLs was estimated to be about 0.3 mM. In susceptible N leaves, the area occupied by susceptible type enlarged lesions (ELs) was about 20% of the whole leaf at 4 dpi with accumulation of a large fungus mass [
9,
17]. Mean sakuranetin content at 4 dpi was 108 ng per g of N leaf, and accordingly the local concentration at ELs was estimated to 540 ng per g leaf corresponding to 2 μM (
Figure 1B, N: open circles). These results suggest that the net sakuranetin concentration at fungus-induced lesions was estimated to be about 160-fold higher in IL7 than N at 4 dpi. The sakuranetin contents quantified here were converted into relative values based on fungal DNA contents in infected leaves [
9], demonstrating that those were much higher in IL7 than in N (
Figure 1C).
Figure 1.
Increase in sakuranetin and transcript of
NOMT in resistant IL7 and susceptible N rice lines after inoculation with blast fungus. (
A) Sakuranetin content in leaves; (
B) Estimated sakuranetin concentration at lesions; (
C) Relative content of sakuranetin/fungal DNA; (
D) RT-PCR analysis on a sakuranetin biosynthesis gene
OsNOMT for naringenin 7
-O-methyltransferase and HR-inducible control genes
OsACS2 and
OsACO7 in rice leaves after mock- or blast-inoculation according to Iwai
et al. [
13]. Arrow indicates the time of HR lesion visualization. Asterisks indicate a significant difference between inoculated N and IL7 leaves at 3 dpi (Student’s paired
t test:
p < 0.01). gFW: g fresh leaf weight. dpi: Day post inoculation. hpi: Hour post inoculation. Data are means ± standard deviation (SD) from independent three samples for
Figure 1A–C.
Figure 1.
Increase in sakuranetin and transcript of
NOMT in resistant IL7 and susceptible N rice lines after inoculation with blast fungus. (
A) Sakuranetin content in leaves; (
B) Estimated sakuranetin concentration at lesions; (
C) Relative content of sakuranetin/fungal DNA; (
D) RT-PCR analysis on a sakuranetin biosynthesis gene
OsNOMT for naringenin 7
-O-methyltransferase and HR-inducible control genes
OsACS2 and
OsACO7 in rice leaves after mock- or blast-inoculation according to Iwai
et al. [
13]. Arrow indicates the time of HR lesion visualization. Asterisks indicate a significant difference between inoculated N and IL7 leaves at 3 dpi (Student’s paired
t test:
p < 0.01). gFW: g fresh leaf weight. dpi: Day post inoculation. hpi: Hour post inoculation. Data are means ± standard deviation (SD) from independent three samples for
Figure 1A–C.
The sakuranetin contents in the resistant rice line detected here were considerably lower than the levels mentioned in previous reports; 100 μg per g leaf at 2 dpi by Kodama
et al. [
10] and 15.8 μg per g leaf at 3 dpi by Dillon
et al. [
11]. As indicated by Tamogami
et al. [
12], treatment with the wound hormone jasmonic acid elicited 40-fold more sakuranetin in rice leaves in comparison with water-treated control. Then, one of the possible reasons for the different results could come from the wound-inducible nature of sakuranetin. Actually, in our experimental system, sakuranetin was detected in a very small amount in healthy rice leaves, and increased about 30-fold by cutting leaves at 48 h. We spray-inoculated a fungal spore suspension and inoculated rice seedlings were carefully grown without wounding before sakuranetin quantification. However, in the previous papers, the inoculation methods involved severe wounding procedures. Another reason for the different results could come from the different experimental system used, such as rice lines, fungus race, incubation conditions and quantification methods.
Next, induced expression of a sakuranetin biosynthesis gene upon HR was studied by RT-PCR according to Iwai
et al. [
13]. Expression of the
OsNOMT gene for naringenin 7
-O-methyltransferase which is a key enzyme in the biosynthesis of sakuranetin (Shimizu
et al. [
18]) was analyzed with that of HR inducible positive control genes,
OsACS2 and
OsACO7 for ethylene biosynthesis [
13]. The transcripts for
OsACS2 and
OsACO7 were transiently accumulated in blast inoculated IL7 at 48 h post inoculation (hpi) at which HRLs became to be visible but not in inoculated N (
Figure 1D). Transcript of
OsNOMT was found in IL7 but not in N at 48 hpi. Expression time of
OsNOMT in IL7 was 48–96 hpi in IL7, while it was at 72–96 hpi in N, suggesting induction of
OsNOMT preceded sakuranetin accumulation in both lines.
2.2. Inhibition of Fungal Growth by Sakuranetin in Vitro and in Planta
The inhibitory activity of sakuranetin to mycelium growth was studied in vitro using a solid medium. Four blast mycelium plugs (about 10 mm2 each) were inoculated on potato dextrose agar (PDA) containing sakuranetin at various concentrations, and fungal growth was analyzed measuring the diameter of the mycelium colony after appropriate incubation.
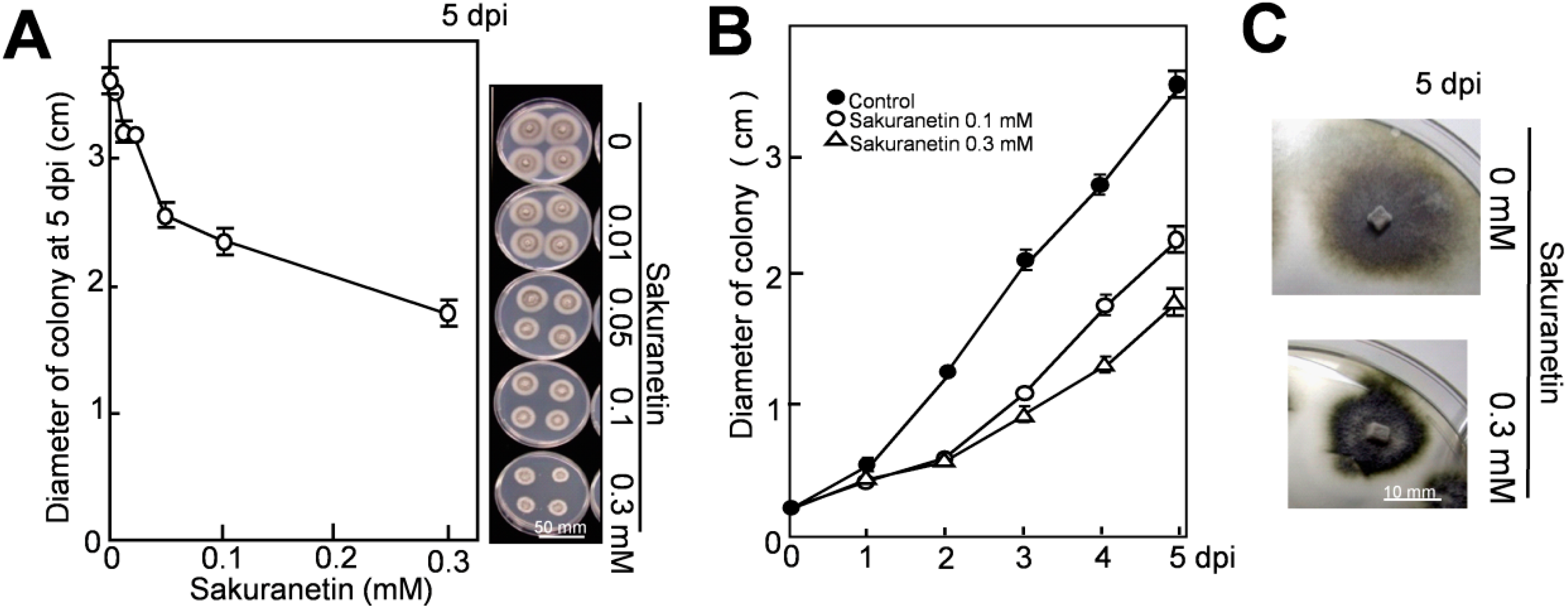
As shown in
Figure 2A, sakuranetin inhibited fungal mycelium growth in a concentration-dependent manner on PDA. On the right side, the phenotypes of mycelium colonies at 5 dpi are shown. A time-course experiment indicates that growth of mycelium colony in diameter at 2 dpi was inhibited about 50% by sakuranetin at both 0.1 and 0.3 mM (
Figure 2B). The inhibition rate by 0.3 mM sakuranetin became slightly higher than by 0.1 mM thereafter, and 51 and 36% of the growth was inhibited by 0.3 and 0.1 mM at 5 dpi, respectively. The level of antifungal activity on PDA was similar to that on potato sucrose agar (PSA), which contains sucrose instead of glucose as the naturally occurring sugar source in plants. The phenotype of the mycelium colony was altered in the presence of sakuranetin. The colony looked like more condensed and risen on both PDA and PSA containing 0.3 mM sakuranetin, and an example on PSA at 5 dpi is shown in
Figure 2C.
Referring to the report by Hasegawa
et al. on the diterpenoid PA momilactone A [
9], the antifungal activity of sakuranetin on blast fungus was higher than that of momilactone A under similar experimental conditions. For example, momilactone A inhibited 12% and 17% of the mycelium growth on PDA at 0.1 and 0.3 mM at 4 dpi respectively [
9], while sakuranetin inhibited 40% and 55% of it at 0.1 and 0.3 mM, respectively (
Figure 2B).
Figure 2.
Inhibition of blast mycelium growth by sakuranetin on solid medium. (
A) Size of blast mycelium colony on PDA containing sakuranetin at 5 dpi; (
B) Time course analysis on the spread of blast mycelium colony on PDA containing sakuranetin at 0.1 or 0.3 Mm; (
C) Phenotype of the mycelium colony with or without 0.3 mM sakuranetin at 5 dpi. Data are means ± SD from four independent samples. Scale bar in
Figure 2A indicates 50 mm, and that in
Figure 2C 10 mm.
Figure 2.
Inhibition of blast mycelium growth by sakuranetin on solid medium. (
A) Size of blast mycelium colony on PDA containing sakuranetin at 5 dpi; (
B) Time course analysis on the spread of blast mycelium colony on PDA containing sakuranetin at 0.1 or 0.3 Mm; (
C) Phenotype of the mycelium colony with or without 0.3 mM sakuranetin at 5 dpi. Data are means ± SD from four independent samples. Scale bar in
Figure 2A indicates 50 mm, and that in
Figure 2C 10 mm.
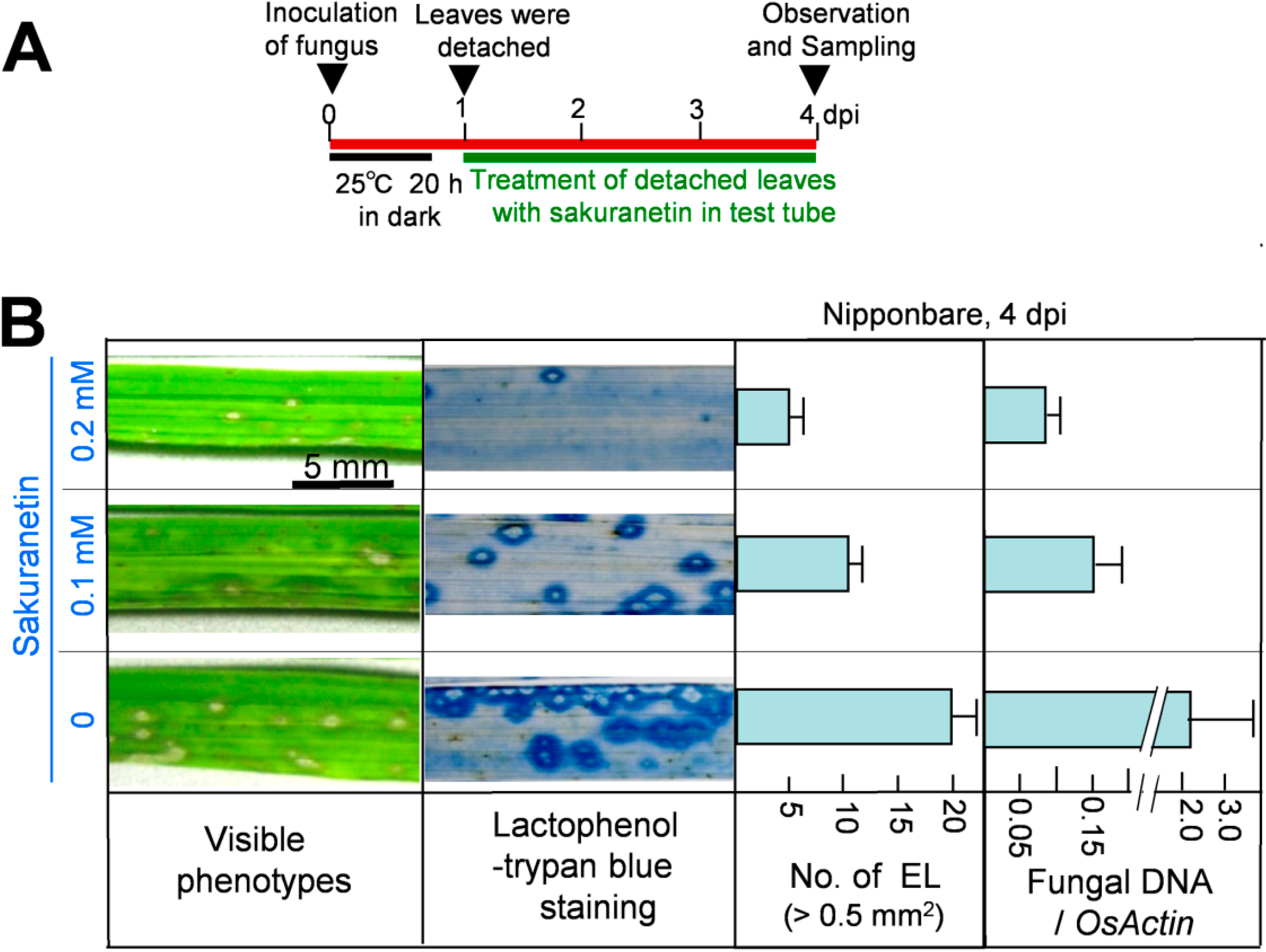
Next, the antifungal activity of sakuranetin was analyzed
in planta. After spray inoculation with a blast spore suspension, ten 4th leaves from intact susceptible N seedlings were detached at 1 dpi, and the leaf bases were put into a glass tube containing 10 mL of chemical solution, and incubated according to the illustrated method in
Figure 3A and in Seo
et al. [
17]. The visible phenotype of inoculated 4th leaves at 4 dpi is shown in the leftmost column of
Figure 3B. Compared with the visible phenotype, spread of fungus was more clearly observed after lactophenol-trypan blue staining of mycelium (second column). Sakuranetin treatment at 0.1 and 0.2 mM reduced the number of susceptible type ELs, whose area was more than 0.5 mm
2, to 50% and 25% respectively (third column) and fungal DNA content to 7% and 5% respectively (most right column) at 4 dpi. When the antifungal activity of sakuranetin
in planta was compared with that of momilactone A described by Hasegawa
et al. [
9], sakuranetin exhibited higher antifungal activity than momilactone A. For example, the number of ELs in blast-inoculated N leaves was decreased to the 25% of control by 0.2 mM sakuranetin (
Figure 3B), and that was decreased to the 43% by 0.2 mM momilactone A [
9]. Fungal DNA content in infected leaves was decreased to the 4% by 0.2 mM sakuranetin (
Figure 3B), and that was decreased to the 39% by 0.2 mM momilactone A [
9]. These results indicate sakuranetin has a considerable antifungal activity
in vitro and
in vivo, which is higher than that of momilactone A. The calculated concentration of sakuranetin at HRLs in IL7 seems to be effective to restrict fungal growth, while that at ELs was insufficient in susceptible N (
Figure 1B), indicating a possible contribution of sakuranetin for fungal resistance upon HR in IL7.
2.3. Decrease of Sakuranetin in Blast Mycelium Cultures
Detoxification of PA would be a way for the fungus to survive in the invaded host plant. Possible detoxification of sakuranetin by the fungus was examined using both solid and liquid mycelium cultures. Discs 20 mm in diameter of 15-day-old blast mycelium layer on PDA were prepared, and put upside down on new PDA containing 0.3 mM sakuranetin according to the method described by Hasegawa
et al. [
9]. After incubation for the indicated time periods, the agar discs beneath the mycelium discs were prepared and subjected to sakuranetin quantification after treatment with 70% methanol for sakuranetin extraction.
Figure 3.
Inhibition of blast fungus growth by sakuranetin in rice leaves at 4 dpi. (A) Experimental protocol; (B) Leftmost column: visible lesion phenotype on blast-inoculated Nipponbare leaves treated with sakuranetin; second column: phenotype after staining mycelium in inoculated leaves with lactophenol-trypan blue; third column: number of ELs larger than 0.5 mm2 in a leaf. Rightmost column: fungal DNA content/transcript of OsActin. Data are means ± SE from ten independent samples for ELs and three samples for fungal DNA, respectively.
Figure 3.
Inhibition of blast fungus growth by sakuranetin in rice leaves at 4 dpi. (A) Experimental protocol; (B) Leftmost column: visible lesion phenotype on blast-inoculated Nipponbare leaves treated with sakuranetin; second column: phenotype after staining mycelium in inoculated leaves with lactophenol-trypan blue; third column: number of ELs larger than 0.5 mm2 in a leaf. Rightmost column: fungal DNA content/transcript of OsActin. Data are means ± SE from ten independent samples for ELs and three samples for fungal DNA, respectively.
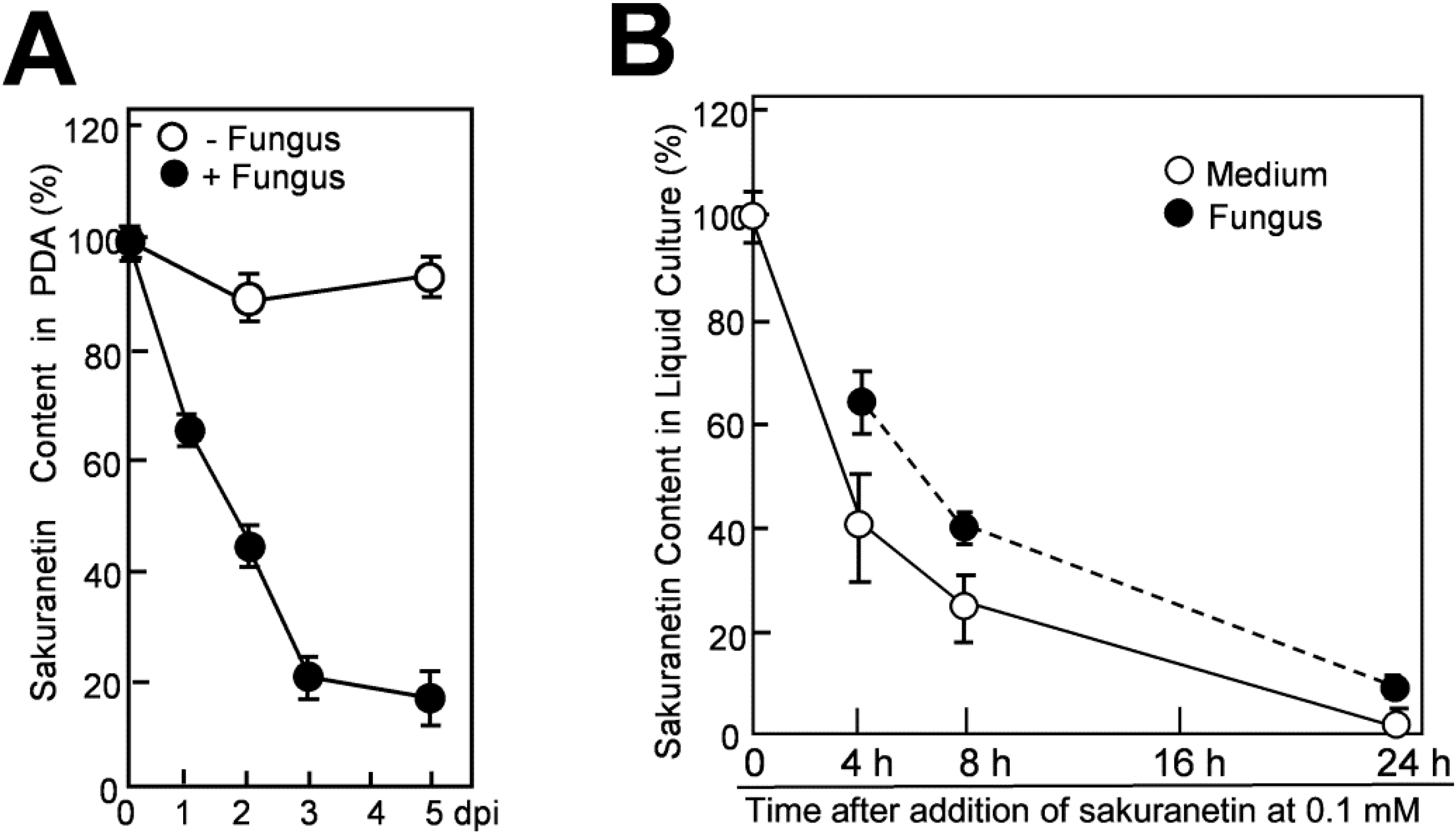
Sakuranetin in the agar medium decreased with time after being overlaid by the mycelium layer. Recovered sakuranetin levels at 1, 2, 3 and 5 dpi decreased to 65%, 45%, 20% and 17% of the initial level, respectively (
Figure 4A, closed circles), while no clear decrease was seen after mock-inoculation (open circles). Compared with the data of momilactone A under similar experimental conditions, the rate of decrease of sakuranetin was considerably slower than that of momilactone A; recovered momilactone levels at 1, 2 and 3 dpi were 53%, 3% and 0% of the initial level, respectively [
9].
Next, decrease and possible conversion of sakuranetin were analyzed in liquid mycelium culture. The mycelium culture of blast fungus was prepared as described by Hasegawa
et al. [
9]. After addition of sakuranetin at 0.1 mM, 1 mL of culture containing 2 mg equivalent of fungal protein was collected at appropriate time intervals, and fractionated into the supernatant and the precipitated hyphae, designated as medium fraction and fungal mass fraction, respectively. Sakuranetin was then extracted from each fraction with 70% methanol and subjected to quantification. After addition of sakuranetin, about the 60% was transferred to the fungal mass fraction and about the 40% was retained in the medium fraction at 4 h (
Figure 4B). The sakuranetin content gradually decreased thereafter in both fractions, and the contents in fungal mass and the medium became 25% and 40% of the initial level, respectively, at 8 h, retaining 65% as total content. At 24 h, it was decreased to 10% in fungal mass and 2% in the medium (
Figure 4B).
Figure 4.
Decrease of sakuranetin in the blast mycelium culture. (A) Decrease of sakuranetin content in solid medium PDA after being overlaid by blast mycelium layer (+Fungus) or mock-layer (−Fungus). At time 0, 0.3 mM sakuranetin was present in the agar medium (100%), and sakuranetin in PDA was quantified thereafter; (B) Decrease in sakuranetin in liquid mycelium culture. At time 0, 0.1 mM sakuranetin was present in the PD liquid mycelium culture medium (100%). After incubation for the indicated time period, sakuranetin in the medium fraction and fungus mass fraction was quantified separately. Data are means ± SD from three independent samples.
Figure 4.
Decrease of sakuranetin in the blast mycelium culture. (A) Decrease of sakuranetin content in solid medium PDA after being overlaid by blast mycelium layer (+Fungus) or mock-layer (−Fungus). At time 0, 0.3 mM sakuranetin was present in the agar medium (100%), and sakuranetin in PDA was quantified thereafter; (B) Decrease in sakuranetin in liquid mycelium culture. At time 0, 0.1 mM sakuranetin was present in the PD liquid mycelium culture medium (100%). After incubation for the indicated time period, sakuranetin in the medium fraction and fungus mass fraction was quantified separately. Data are means ± SD from three independent samples.
The decrease in momilactone A using a similar mycelium culture has been reported [
9]. However, the rate of decrease was higher than that of sakuranetin. At 4 h after PA application, 35% of the initially added momilactone A remained [
9], while the 100% of the sakuranetin remained (
Figure 4B). At 8 h, 20% of the momilactone A remained [
9], while 65% of sakuranetin was still detected in the culture. These results indicate that sakuranetin is metabolized more slowly by the fungus than momilactone A in both solid and liquid mycelium media.
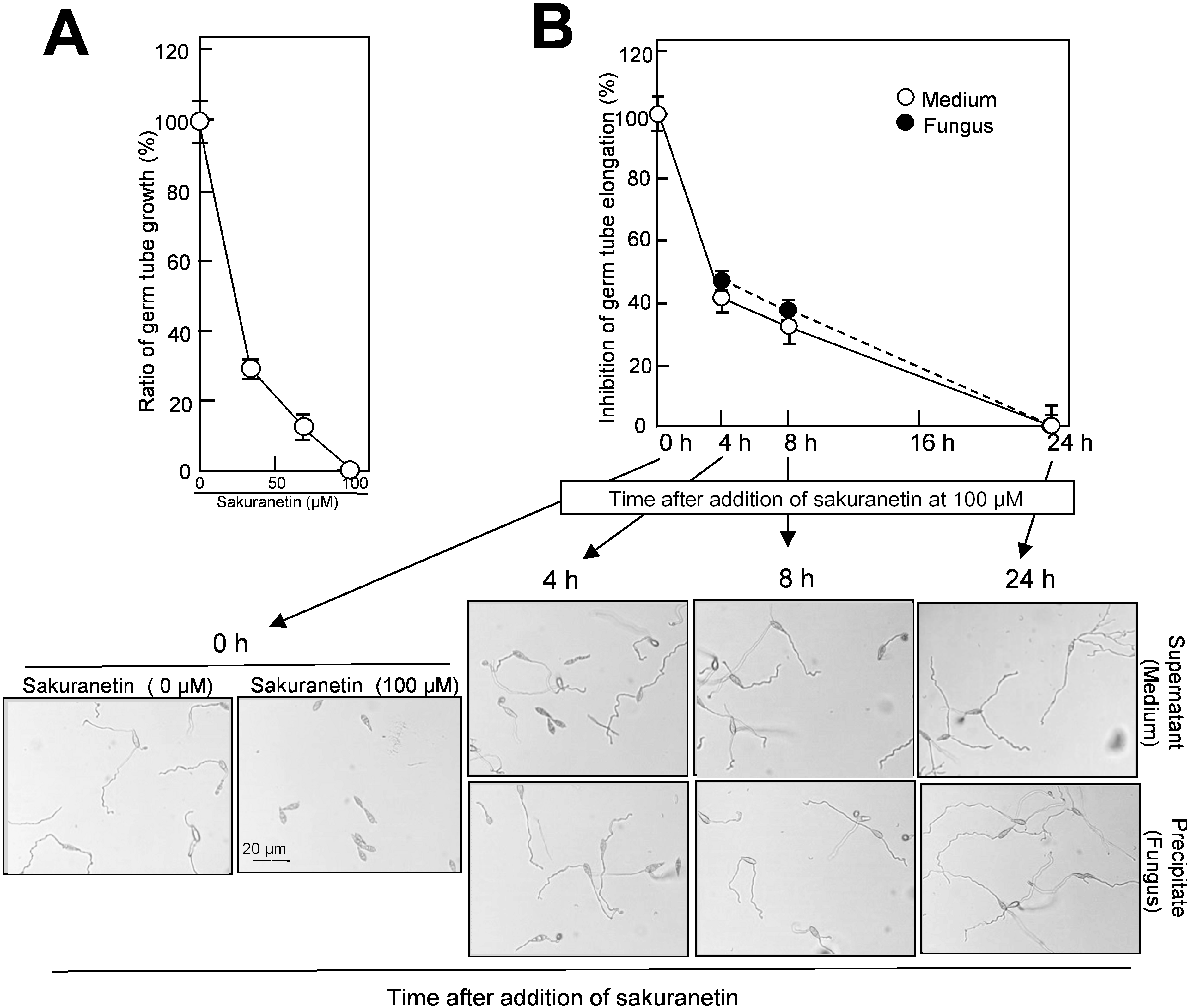
2.4. Detoxification of Sakuranetin in the Culture of Blast Mycelium
Next, we evaluated whether the decrease in sakuranetin from the liquid mycelium culture accompanies the decrease in antifungal activity or not. As the first experiment, a standard antifungal activity test was conducted using authentic sakuranetin according to the method by Hasegawa
et al. [
9]. In the presence of sakuranetin at 34, 67 and 100 μM, germ tube growth from blast spore was inhibited by 72%, 89% and 100% at 24 h, respectively (
Figure 5A). In this system, the sakuranetin concentration required for 50% inhibition of germ tube elongation was estimated to be about 20 μM, which is similar to the concentration reported by Kodama
et al. [
10]. On the other hand, in the presence of momilactone A at 100, 200 and 300 μM, germ tube growth was inhibited 45%, 78% and 96%, respectively [
9]. These results indicate that sakuranetin more effectively inhibits fungal growth than momilactone A in liquid culture.
Figure 5.
Decrease in antifungal activity in the liquid blast mycelium culture after addition of sakuranetin. (
A) Standard experiment on germ tube growth inhibition by sakuranetin. Length of germ tubes grown from blast spore was determined at 24 h after incubation with sakuranetin; (
B) The same lots of samples used in
Figure 4B were employed for the antifungal assay. Data are means ± SD from three independent samples each of which contains 10–20 spores for
Figure 5A, and from 10 to 20 independent samples each of which contains 5–20 spores for
Figure 5B, respectively. The precise method was according to Hasegawa
et al. [
9].
Figure 5.
Decrease in antifungal activity in the liquid blast mycelium culture after addition of sakuranetin. (
A) Standard experiment on germ tube growth inhibition by sakuranetin. Length of germ tubes grown from blast spore was determined at 24 h after incubation with sakuranetin; (
B) The same lots of samples used in
Figure 4B were employed for the antifungal assay. Data are means ± SD from three independent samples each of which contains 10–20 spores for
Figure 5A, and from 10 to 20 independent samples each of which contains 5–20 spores for
Figure 5B, respectively. The precise method was according to Hasegawa
et al. [
9].
The same lots of samples which were used for the fungus-induced sakuranetin decrease experiment in liquid culture (
Figure 4B), were employed for the antifungal activity analysis. Each sample from the medium and fungal mass fractions was treated with 70% methanol for extraction, and subjected to the assay after concentration. The antifungal activity in both fractions decreased with time, and almost all was lost at 24 h (
Figure 5B). A representative photograph used for length determination of elongated germ tube from blast spores is shown at the bottom of
Figure 5B. When the antifungal activity found in the medium fraction at time 0 was designated as 100%, the antifungal activity in the medium was decreased to 40% at 4 h, 32% at 8 h and was nearly zero at 24 h, and the antifungal activity in fungal mass fraction decreased to 46% at 4 h, 38% at 8 h and zero at 24 h (
Figure 5B). The time-course profile of the loss of antifungal activity was similar to that of the decrease in sakuranetin content shown in
Figure 4B, indicating the sakuranetin content in the culture fractions reflects the level of antifungal activity determined by inhibition of germ tube elongation. Thus, the conversion of sakuranetin would accompany its detoxification by blast fungus. The detoxification of sakuranetin may be the second example on fungus-induced conversion of PA from the family Graminaceae. As the first example, Hasegawa
et al. [
9] reported detoxification of momilactone A by blast fungus
in vitro [
9], and Imai
et al. [
19] reported a degradation intermediate of momilactone A. In the mycelium culture after addition of sakuranetin, a small amount of naringenin, which is the precursor of sakuranetin biosynthesis in rice plant, was detected. Quantification and evaluation of naringenin as the metabolite of sakuranetin should be studied in future. Including this subject, isolation and identification of possible metabolites of sakuranetin are in progress.
2.5. Possible Mechanism of Sakuranetin to Restrict Fungus
The results above indicate that superior accumulation of sakuranetin at HRLs in resistant rice contributes to blast resistance. The precise mechanism of how the flavonoid PA sakuranetin inhibits fungal growth is not obvious now. Mizutani
et al. [
20] suggested that a widely used fungicide metominostrobin (SSF-126) is similar in mode of action to CN, which inhibits CN-sensitive respiration in the mitochondrial respiration chain of blast fungus. Although blast fungus then induced CN-resistant respiration to survive, the CN-resistant fungal respiration was inhibited in the presence of synthetic flavone inducing death of the fungus. Seo
et al. [
17] demonstrated that an exogenously supplied synthetic flavone induced blast resistance as well as CN, and further that treatment of CN together with flavone enhanced the resistance
in vitro and
in vivo. If the flavonoid PA sakuranetin could inhibit CN-resistant fungal respiration as the flavone did, the PA and CN may co-operationally contribute to the resistance by inhibition of both respiration mechanisms in the fungus. A study to reveal this possibility is in progress.
Shimizu
et al. [
18] identified the gene for naringenin 7-
O-methyltransferase (NOMT), which confers the last step of sakuranetin biosynthesis from naringenin. To elucidate the importance of sakuranetin for the resistance, experiments on
NOMT gene for gain of function in susceptible rice and loss of function in resistant rice would be valuable.