Calculating Distortions of Short DNA Duplexes with Base Pairing Between an Oxidatively Damaged Guanine and a Guanine
Abstract
:1. Introduction
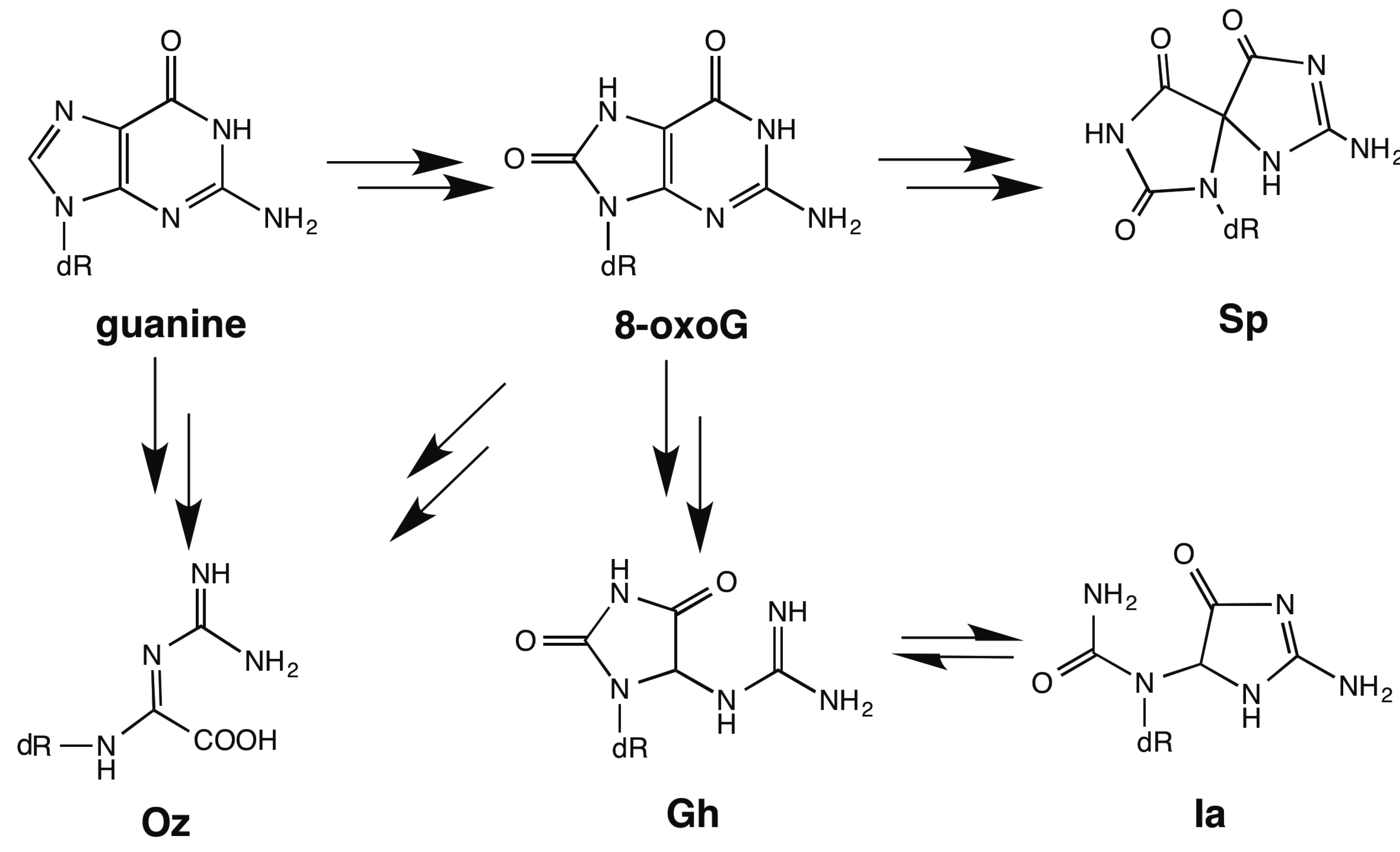
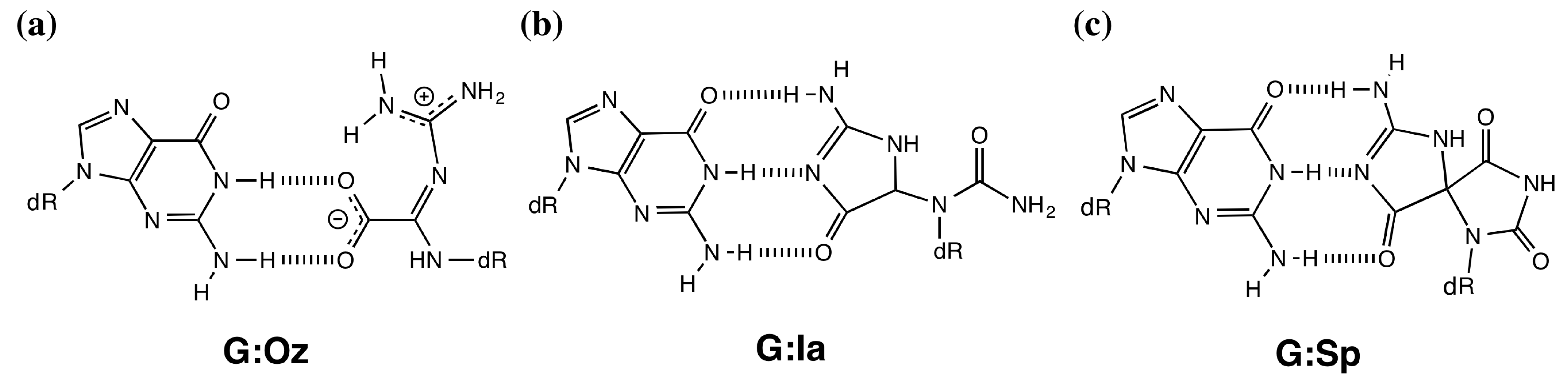
2. Results and Discussion
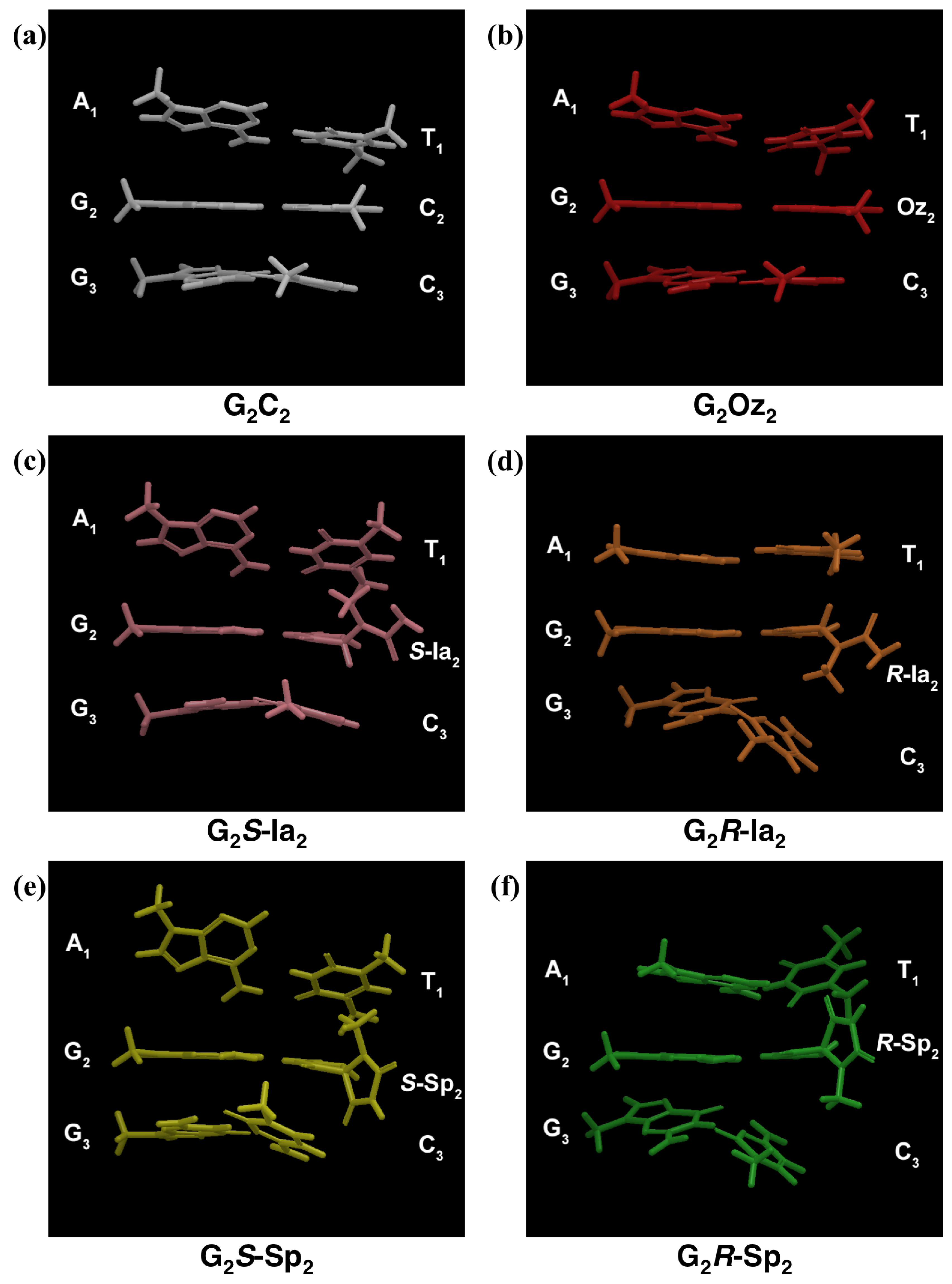
2.1. The Minimized Structures of DNA Duplex Containing an X:G Base Pair
2.2. The Destabilization Energies of the Base Pairs on 5'- and 3'-Side of X
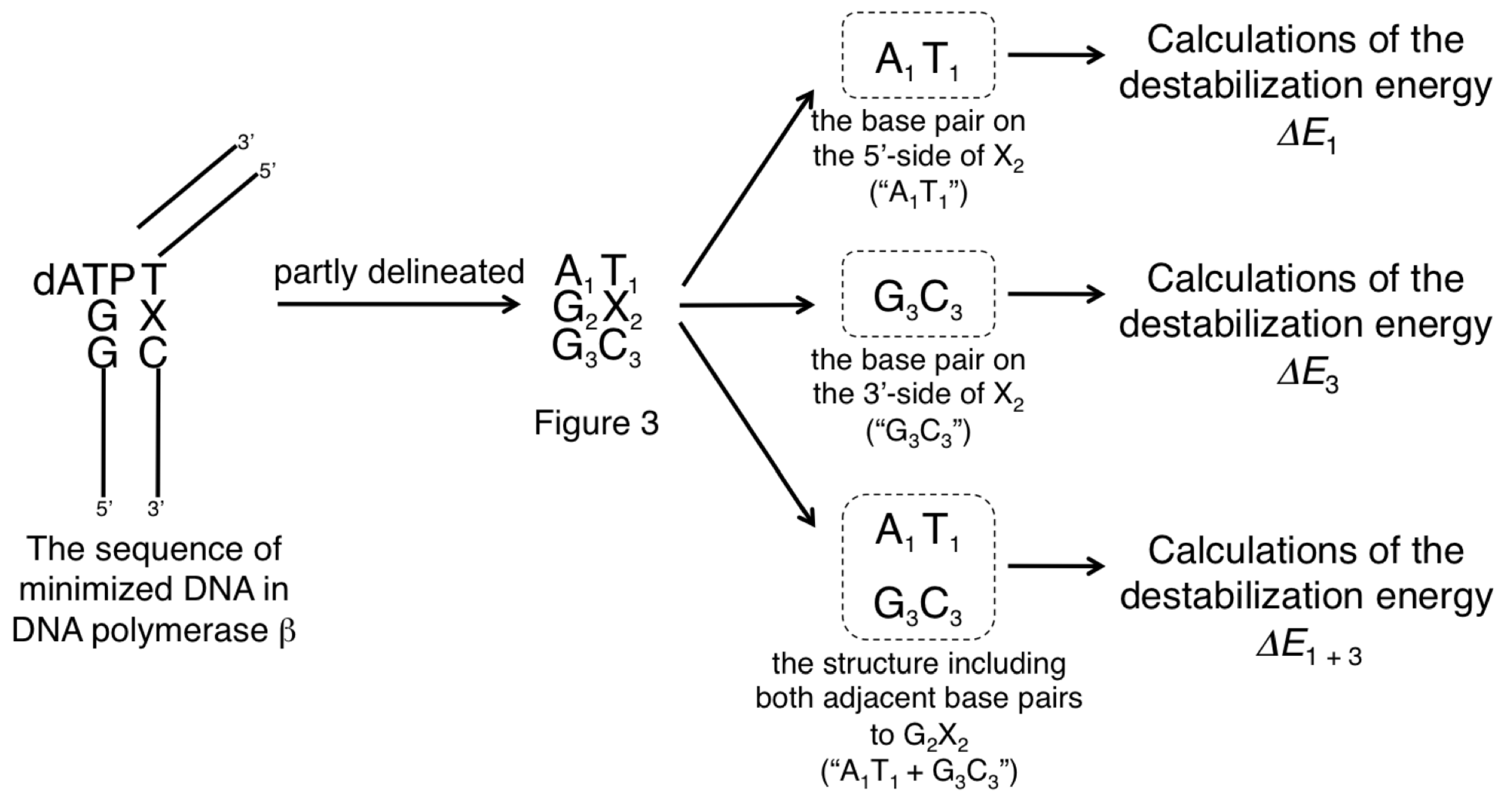
2.2.1. The Destabilization Energies of the Base Pairs on 5'- Side of X
| A1T1 | G3C3 | A1T1 + G3C3 | |||||||
|---|---|---|---|---|---|---|---|---|---|
| X a | ΔE1DFT | ΔE1SCRF | ΔE3DFT | ΔE3SCRF | ΔE1 + 3DFT | ΔE1 + 3SCRF | |||
| Oz | 1.3 | 1.1 | −0.1 | 0.6 | 1.1 | 1.0 | |||
| S-Ia | 1.4 | 1.3 | 2.6 | 1.3 | 4.1 | 4.5 | |||
| R-Ia | 0.5 | 0.6 | 4.0 | 3.9 | 4.8 | 4.6 | |||
| S-Sp | 2.1 | 2.1 | 2.7 | 2.8 | 4.8 | 5.3 | |||
| R-Sp | 12.6 | 12.4 | 5.3 | 4.5 | 18.3 | 18.3 | |||
2.2.2. The Destabilization Energies of the Base Pairs on the 3'-Side of X
2.2.3. The Destabilization Energy of Each Model Duplex was Calculated by Including both Adjacent Base Pairs and Excluding the Central “G2X2” Base Pair
2.3. The Degree of Distortion from DNA Duplex Containing a G:C Base Pair
2.3.1. The Degree of Distortion at the 5'-Side of X
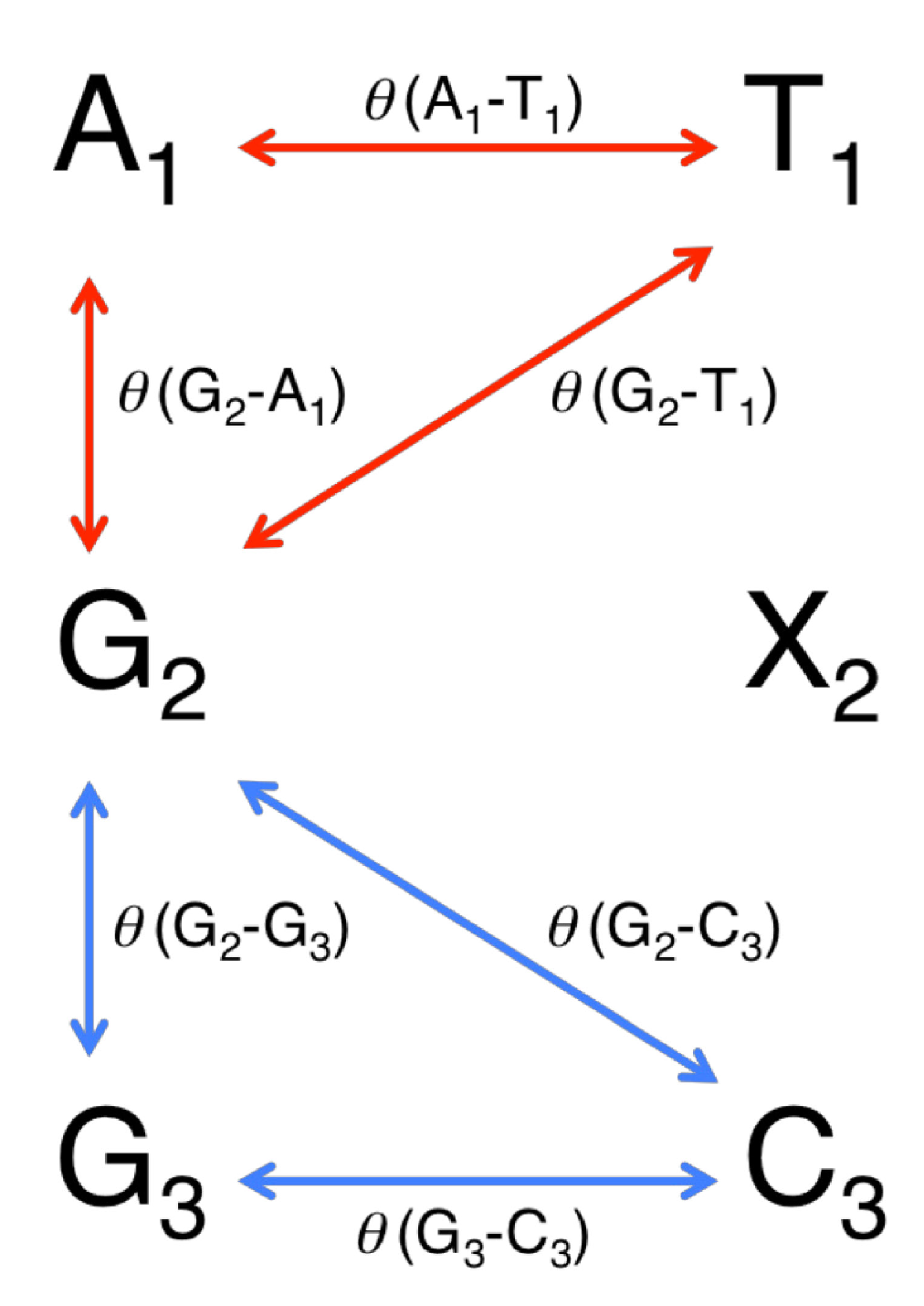
| X a | θ (G2–A1) | θ (G2–T1) | θ (A1–T1) | δ1 |
|---|---|---|---|---|
| C | 25.2° | 12.5° | 13.3° | 51.0° |
| Oz | 19.4° | 18.2° | 18.0° | 55.6° |
| S-Ia | 38.3° | 28.2° | 11.3° | 77.8° |
| R-Ia | 3.3° | 4.0° | 4.0° | 11.3° |
| S-Sp | 56.1° | 29.9° | 26.4° | 112.4° |
| R-Sp | 10.9° | 32.4° | 43.2° | 86.5° |
2.3.2. The Degree of Distortion at the 3'-Side of X
| X a | θ (G2–G3) | θ (G2–C3) | θ (G3–C3) | δ3 |
|---|---|---|---|---|
| C | 9.5° | 8.0° | 17.2° | 34.6° |
| Oz | 11.1° | 3.1° | 11.9° | 26.1° |
| S-Ia | 5.4° | 13.9° | 19.2° | 38.5° |
| R-Ia | 17.9° | 42.6° | 49.2° | 109.7° |
| S-Sp | 5.5° | 25.7° | 23.2° | 54.4° |
| R-Sp | 22.9° | 36.2° | 43.3° | 102.5° |
2.3.3. The Total Degree of Distortion at the 5'-Side and 3'-Side of X
| X a | δ1 + δ3 |
|---|---|
| C | 85.6° |
| Oz | 81.7° |
| S-Ia | 116.3° |
| R-Ia | 121.1° |
| S-Sp | 166.8° |
| R-Sp | 189.0° |
2.3.4. The Difference Between the Degree of Distortion for Oxidatively Damaged Guanine and that for C
3. Experimental
3.1. Molecular Modeling
3.2. ab Initio Calculations
–E(“A1T1” of G:X complex (X = Oz, S-Ia, R-Ia, S-Sp or R-Sp))
–E(“G3C3” of G:X complex (X = Oz, S-Ia, R-Ia, S-Sp or R-Sp))
–E(“A1T1+G3C3” of G:X complex (X = Oz, S-Ia, R-Ia, S-Sp or R-Sp))
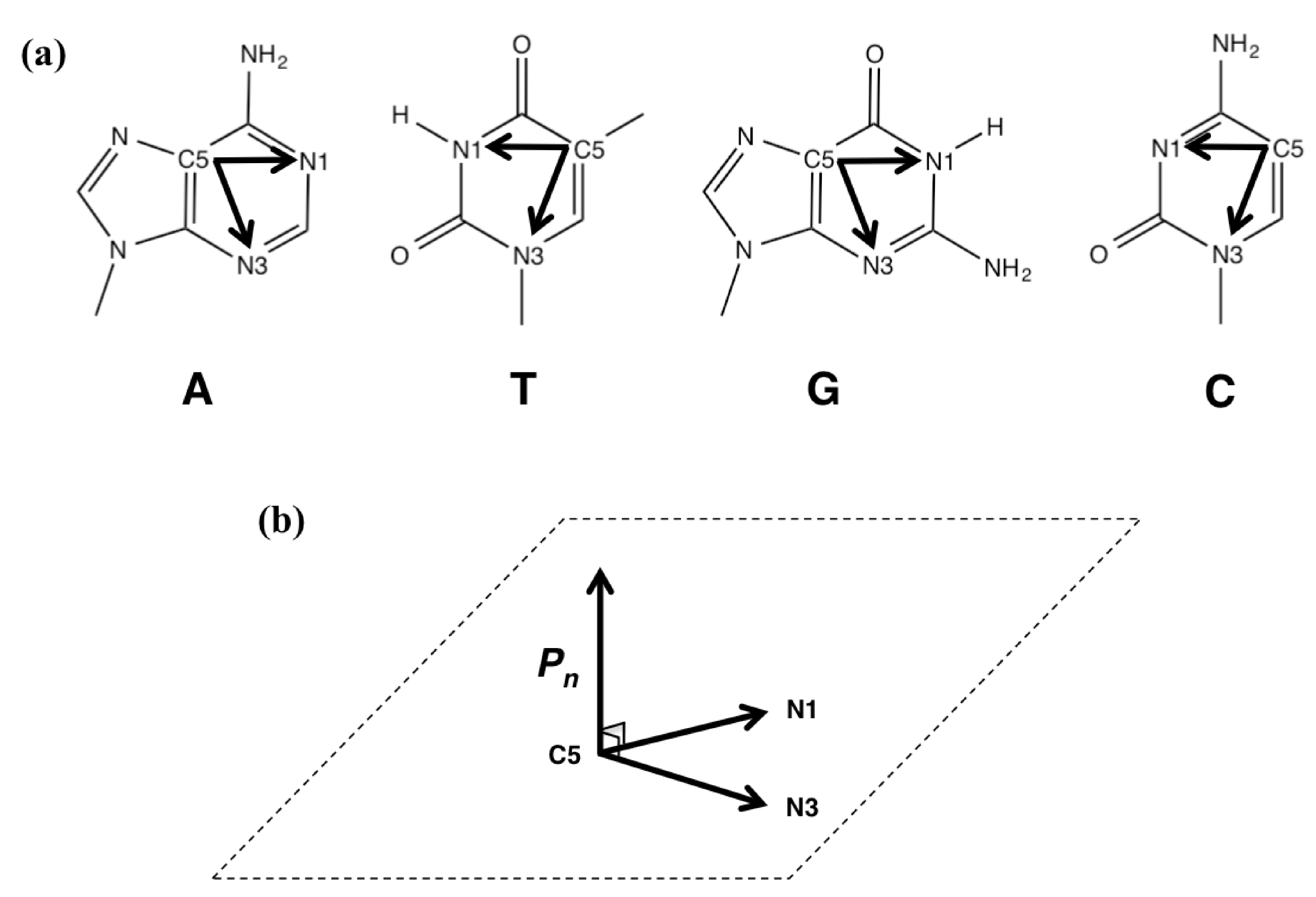
3.3. Calculating the Degree of Distortion
= (xN1– xC5, yN1 – yC5, zN1– zC5) × (xN3– xC5, yN3– yC5, zN3– zC5)
= ((yN1– yC5)•(zN3– zC5)−(zN1– zC5)•(yN3– yC5), (zN1– zC5)•(xN3– xC5) – (xN1–xC5)•(zN3– zC5),
(xN1– xC5)•(yN3– yC5) − (yN1– yC5)•(xN3– xC5))
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kino, K.; Sugiyama, H. UVR-induced G-C to C-G transversions from oxidative DNA damage. Mutat. Res. 2005, 571, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Maehira, F.; Miyagi, I.; Asato, T.; Eguchi, Y.; Takei, H.; Nakatsuki, K.; Fukuoka, M.; Zaha, F. Alterations of protein kinase C,8-hydroxydeoxyguanosine, and K-ras oncogene in rat lungs exposed to passive smoking. Clin. Chim. Acta 1999, 289, 133–144. [Google Scholar] [CrossRef] [PubMed]
- Shibutani, S.; Takeshita, M.; Grollman, A.P. Insertion of specific bases during DNA synthesis past the oxidation-damaged base 8-oxodG. Nature 1991, 349, 431–434. [Google Scholar] [CrossRef] [PubMed]
- Cadet, J.; Berger, M.; Buchko, G.W.; Joshi, P.C.; Raoul, S.; Ravanat, J.-L. 2,2-Diamino-4-[(3,5-di-O-acetyl-2-deoxy-β-d-erythro-pentofuranosyl)amino]-5-(2H)-oxazolone: A novel and predominant radical oxidation product of 3',5'-di-O-acetyl-2'-deoxyguanosine. J. Am. Chem. Soc. 1994, 116, 7403–7404. [Google Scholar] [CrossRef]
- Kino, K.; Sugasawa, K.; Mizuno, T.; Bando, T.; Sugiyama, H.; Akita, M.; Miyazawa, H.; Hanaoka, F. Eukaryotic DNA polymerases α,β and ε incorporate guanine opposite 2,2,4-triamino-5(2H)-oxazolone. ChemBioChem 2009, 10, 2613–2616. [Google Scholar] [CrossRef] [PubMed]
- Duarte, V.; Gasparutto, D.; Jaquinod, M.; Cadet, J. In vitro DNA synthesis opposite oxazolone and repair of this DNA damage using modified oligonucleotides. Nucleic Acids Res. 2000, 28, 1555–1563. [Google Scholar] [CrossRef] [PubMed]
- Kino, K.; Ito, N.; Sugasawa, K.; Sugiyama, H.; Hanaoka, F. Translesion synthesis by human DNA polymerase η across oxidative products of guanine. Nucleic Acids Symp. Ser. 2004, 48, 171–172. [Google Scholar] [CrossRef]
- Luo, W.; Muller, J.G.; Rachlin, E.M.; Burrows, C.J. Characterization of spiroiminodihydantoin as a product of one-electron oxidation of 8-oxo-7,8-dihydroguanosine. Org. Lett. 2000, 2, 613–616. [Google Scholar] [CrossRef] [PubMed]
- Kino, K.; Morikawa, M.; Kobayashi, T.; Kobayashi, T.; Komori, R.; Sei, Y.; Miyazawa, H. The oxidation of 8-oxo-7,8-dihydroguanine by iodine. Bioorg. Med. Chem. Lett. 2010, 20, 3818–3820. [Google Scholar] [CrossRef] [PubMed]
- Munk, B.H.; Burrows, C.J.; Schlegel, H.B. An exploration of mechanisms for the transformation of 8-oxoguanine to guanidinohydantoin and spiroiminodihydantoin by density functional theory. J. Am. Chem. Soc. 2008, 130, 5245–5256. [Google Scholar] [CrossRef] [PubMed]
- Ye, Y.; Munk, B.H.; Muller, J.G.; Cogbill, A.; Burrows, C.J.; Schlegel, H.B. Mechanistic aspects of the formation of guanidinohydantoin from spiroiminodihydantoin under acidic conditions. Chem. Res. Toxicol. 2009, 22, 526–535. [Google Scholar] [CrossRef] [PubMed]
- Luo, W.; Muller, J.G.; Rachlin, E.M.; Burrows, C.J. Characterization of hydantoin products from one-electron oxidation of 8-oxo-7,8-dihydroguanosine in a nucleoside model. Chem. Res. Toxicol. 2001, 14, 927–938. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.; Kino, K.; Morikawa, M.; Kobayashi, T.; Komori, R.; Miyazawa, H. Calculation of the stabilization energies of oxidatively damaged guanine. Molecules 2012, 17, 6705–6715. [Google Scholar] [CrossRef] [PubMed]
- Kornyushyna, O.; Berges, A.M.; Muller, J.G.; Burrows, C.J. In vitro nucleotide misinsertion opposite the oxidized guanosine lesions spiroiminodihydantoin and guanidinohydantoin and DNA synthesis past the lesions using escherichia coli DNA Polymerase I (Klenow Fragment). Biochemistry 2002, 41, 15304–15314. [Google Scholar] [CrossRef] [PubMed]
- Prakash, S.; Prakash, L. Translesion DNA synthesis in eukaryotes: A one- or two-polymerase affair. Genes Dev. 2002, 16, 1872–1883. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Yan, S.; Patel, D.J.; Geacintov, N.E.; Broyde, S. Relating repair susceptibility of carcinogen-damaged DNA with structural distortion and thermodynamic stability. Nucleic Acids Res. 2002, 30, 3422–3432. [Google Scholar] [CrossRef] [PubMed]
- Sawaya, M.R.; Prasad, R.; Wilson, S.H.; Kraut, J.; Pelletier, H. Crystal structures of human DNA polymerase β complexed with gapped and nicked DNA: Evidence for an induced fit mechanism. Biochemistry 1997, 36, 11205–11215. [Google Scholar] [CrossRef] [PubMed]
- Neeley, W.L.; Delaney, S.; Alekseyev, Y.O.; Jarosz, D.F.; Delaney, J.C.; Walker, G.C.; Essigmann, J.M. DNA polymerase V allows bypass of toxic guanine oxidation products in vivo. J. Biol. Chem. 2007, 282, 12741–12748. [Google Scholar] [CrossRef] [PubMed]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Montgomery, J.A.; Vreven, T., Jr.; Kudin, K.N.; Burant, J.C.; et al. Gaussian 03; Revision C.02; Gaussian, Inc.: Wallingford, CT, USA, 2004. [Google Scholar]
- Sample Availability: Not available.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Suzuki, M.; Kino, K.; Morikawa, M.; Kobayashi, T.; Miyazawa, H. Calculating Distortions of Short DNA Duplexes with Base Pairing Between an Oxidatively Damaged Guanine and a Guanine. Molecules 2014, 19, 11030-11044. https://doi.org/10.3390/molecules190811030
Suzuki M, Kino K, Morikawa M, Kobayashi T, Miyazawa H. Calculating Distortions of Short DNA Duplexes with Base Pairing Between an Oxidatively Damaged Guanine and a Guanine. Molecules. 2014; 19(8):11030-11044. https://doi.org/10.3390/molecules190811030
Chicago/Turabian StyleSuzuki, Masayo, Katsuhito Kino, Masayuki Morikawa, Takanobu Kobayashi, and Hiroshi Miyazawa. 2014. "Calculating Distortions of Short DNA Duplexes with Base Pairing Between an Oxidatively Damaged Guanine and a Guanine" Molecules 19, no. 8: 11030-11044. https://doi.org/10.3390/molecules190811030
APA StyleSuzuki, M., Kino, K., Morikawa, M., Kobayashi, T., & Miyazawa, H. (2014). Calculating Distortions of Short DNA Duplexes with Base Pairing Between an Oxidatively Damaged Guanine and a Guanine. Molecules, 19(8), 11030-11044. https://doi.org/10.3390/molecules190811030




