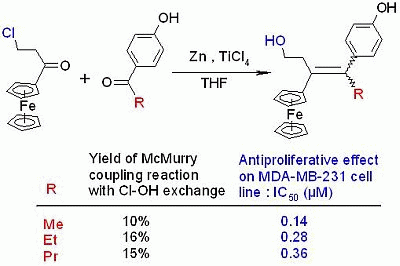
Atypical McMurry Cross-Coupling Reactions Leading to a New Series of Potent Antiproliferative Compounds Bearing the Key [Ferrocenyl-Ene-Phenol] Motif
Abstract
:1. Introduction
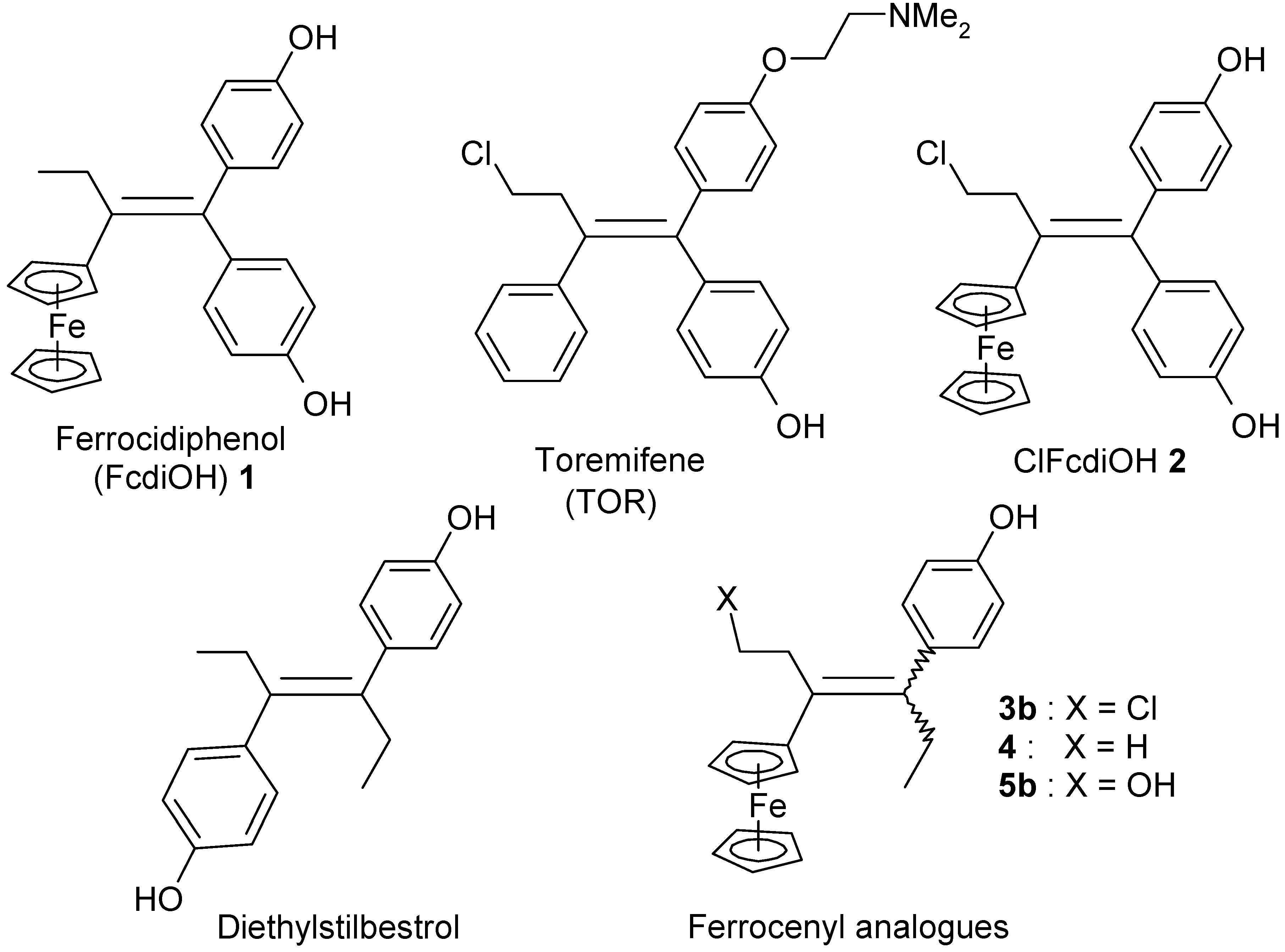
2. Results and Discussion
2.1. Synthetic Aspects


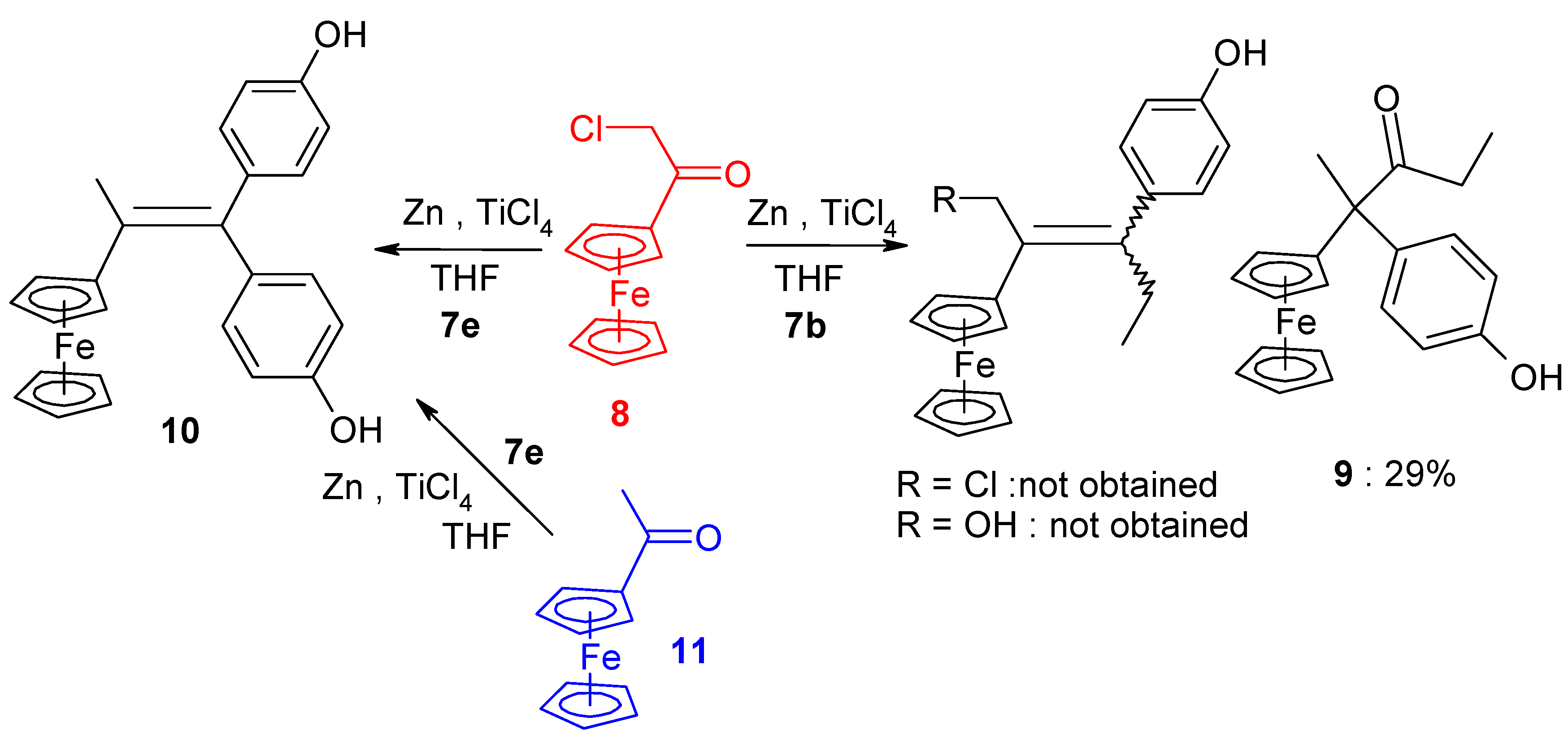
| Entry | Ketone 1 | Ketone 2 | Product (Yield) | Comments |
|---|---|---|---|---|
| 1 | FcC(=O)CH2Cl | p-HO-C6H4-C(=O)Et | 9 (29%) | Rearrangement with exchange of Cl → H |
| 2 | FcC(=O)CH2Cl | (p-HO-C6H4)2C=O | 10 (49%) | Alkene with exchange Cl → H |
| 3 | FcC(=O)Me | (p-HO-C6H4)2C=O | 10 (90%) | Alkene |
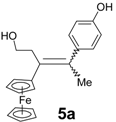
| 4 | FcC(=O)(CH2)2Cl | p-HO-C6H4-C(=O)Me | 5a (10%) | Alkene with exchange Cl → OH |
| 5 | FcC(=O)(CH2)2Cl | p-HO-C6H4-C(=O)Et | 5b (16%) | Alkene with exchange Cl → OH |
| 6 | FcC(=O)(CH2)2Cl | p-HO-C6H4-C(=O)Pr | 5c (15%) | Alkene with exchange Cl → OH |
| 7 | FcC(=O)(CH2)2Cl | p-HO-C6H4-C(=O)H | ----- | Complex inseparable mixture |
| 8 | FcC(=O)(CH2)2Cl | (p-HO-C6H4)2C=O | 2 (47%) | Alkene, no exchange, published work [26] |
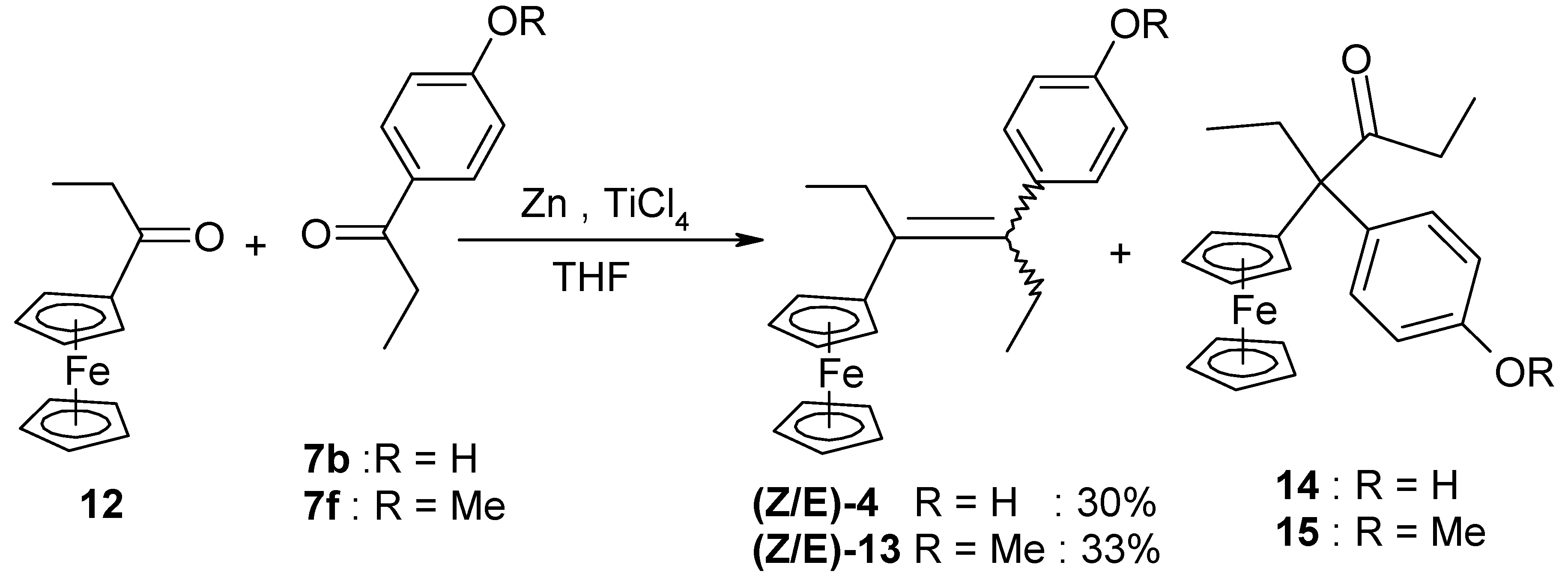
| 9 | FcC(=O)Et | p-HO-C6H4-C(=O)Et | 4 (30%) | Alkene, and also isolation of a rearrangement product, 14 |
| 10 | FcC(=O)Et | p-MeO-C6H4-C(=O)Et | 13 (33%) | Alkene |
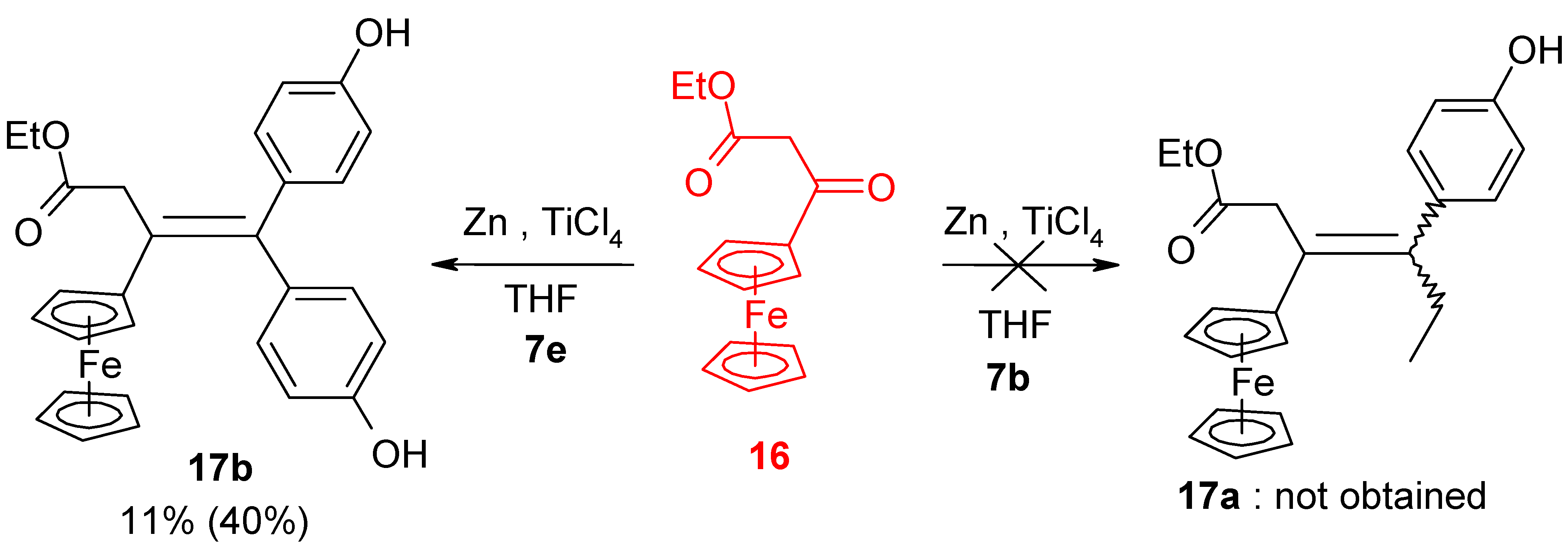
| 11 | FcC(=O)CH2CO2Et | p-HO-C6H4-C(=O)Et | ----- | Homocoupling (DES) |
| 12 | FcC(=O)CH2CO2Et | (p-HO-C6H4)2C=O | 17b (11%) | Alkene |
| 13 | FcC(=O)CH2CO2Et | (p-HO-C6H4)2C=O | 17b (40%) | Alkene when using 2 eq of (p-HOC6H4)2C=O |
2.2. Mechanistic Considerations
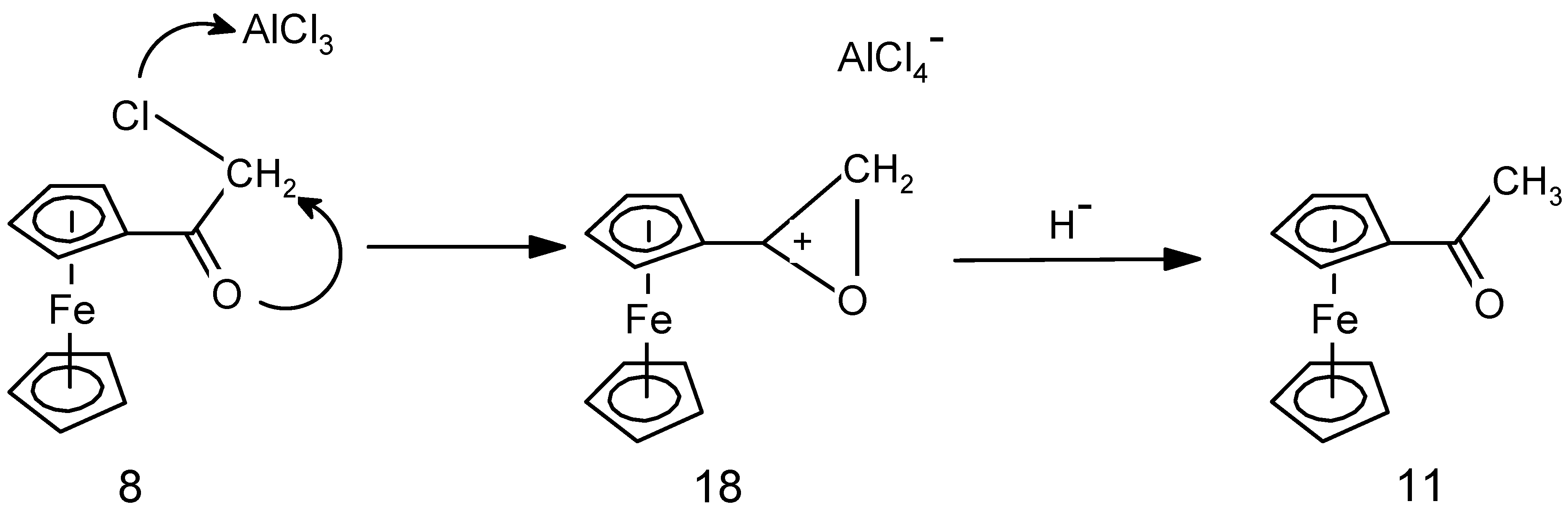
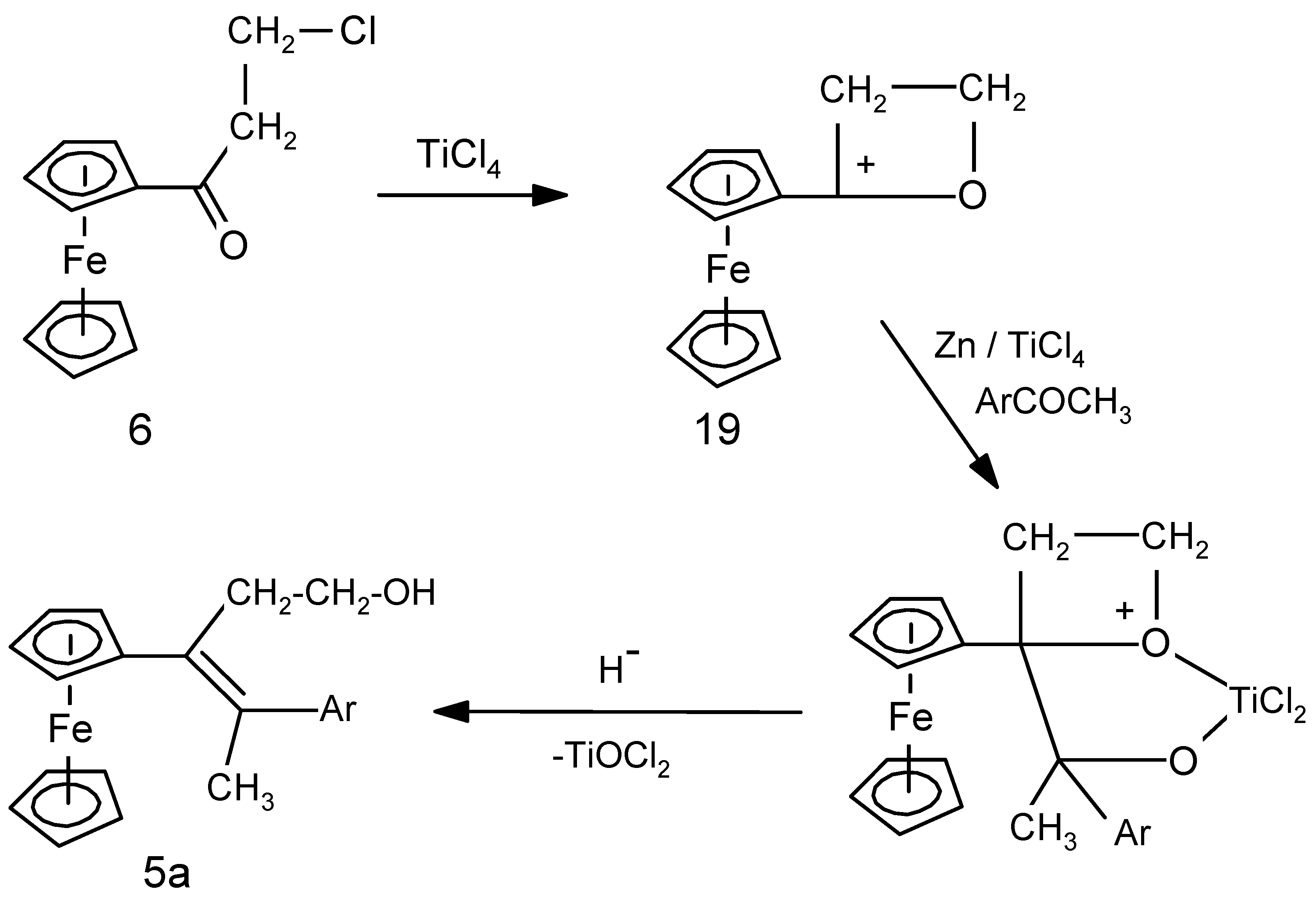
2.3. Biological Results
| Compound | IC50 (μM) | Compound | IC50 (μM) |
|---|---|---|---|
 | 0.60 ± 0.06 a | 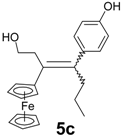 | 0.36 ± 0.04 |
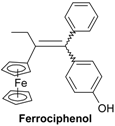
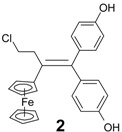 | 1.00 ± 0.01 b | 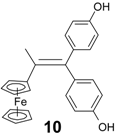 | 1.09 ± 0.22 |
 | 1.13 ± 0.13 c | 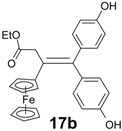 | 1.16 ± 0.10 |
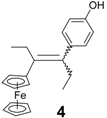 | 1.14 ± 0.05 | 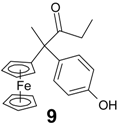 | 3.13 ± 0.07 |
 | 0.14 ± 0.01 |  | 3.5 ± 0.02 d |
 | 0.28 ± 0.01 |
3. Experimental Section
3.1. General Information
3.2. Chemistry
General Procedure for McMurry Coupling Reactions
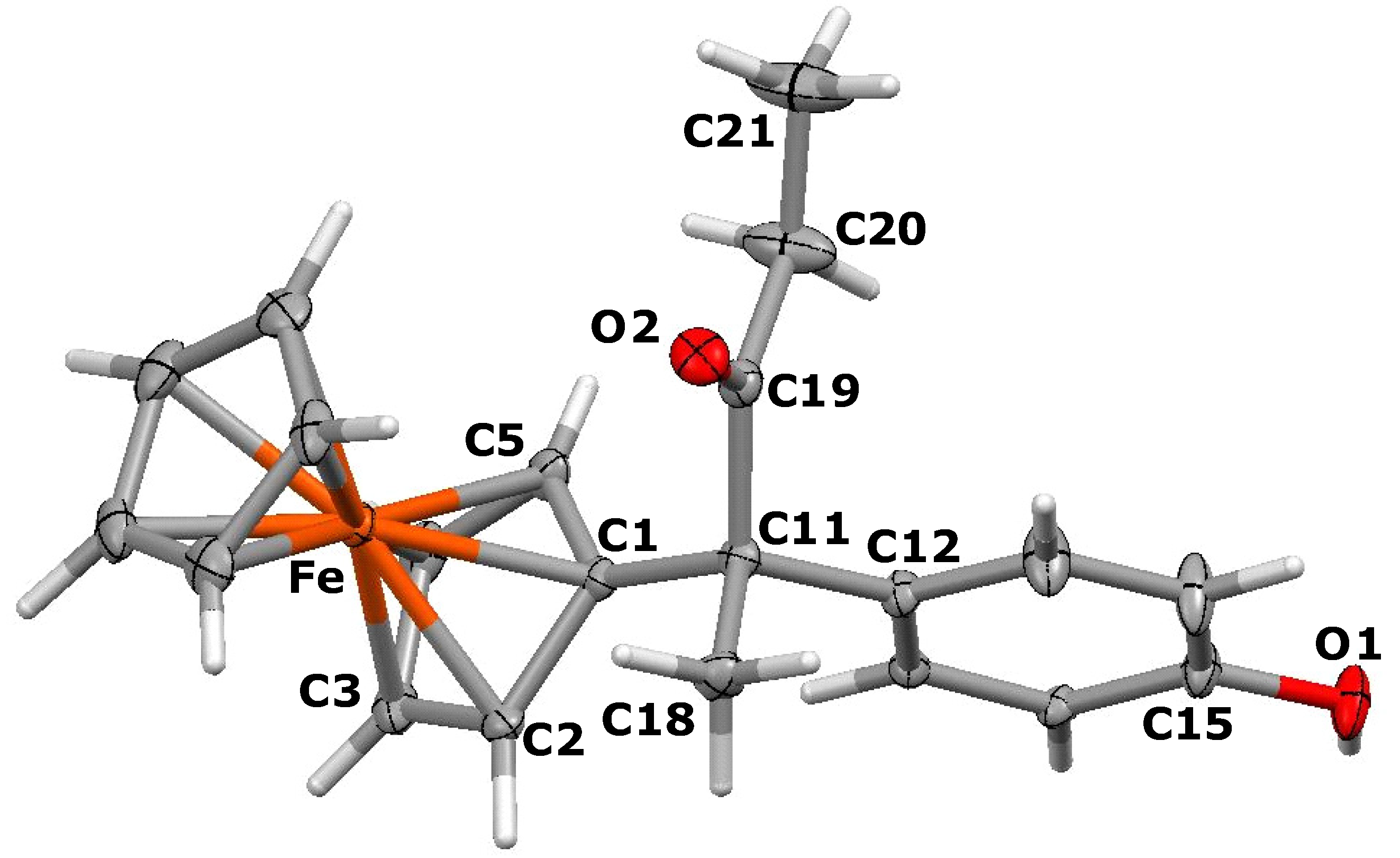
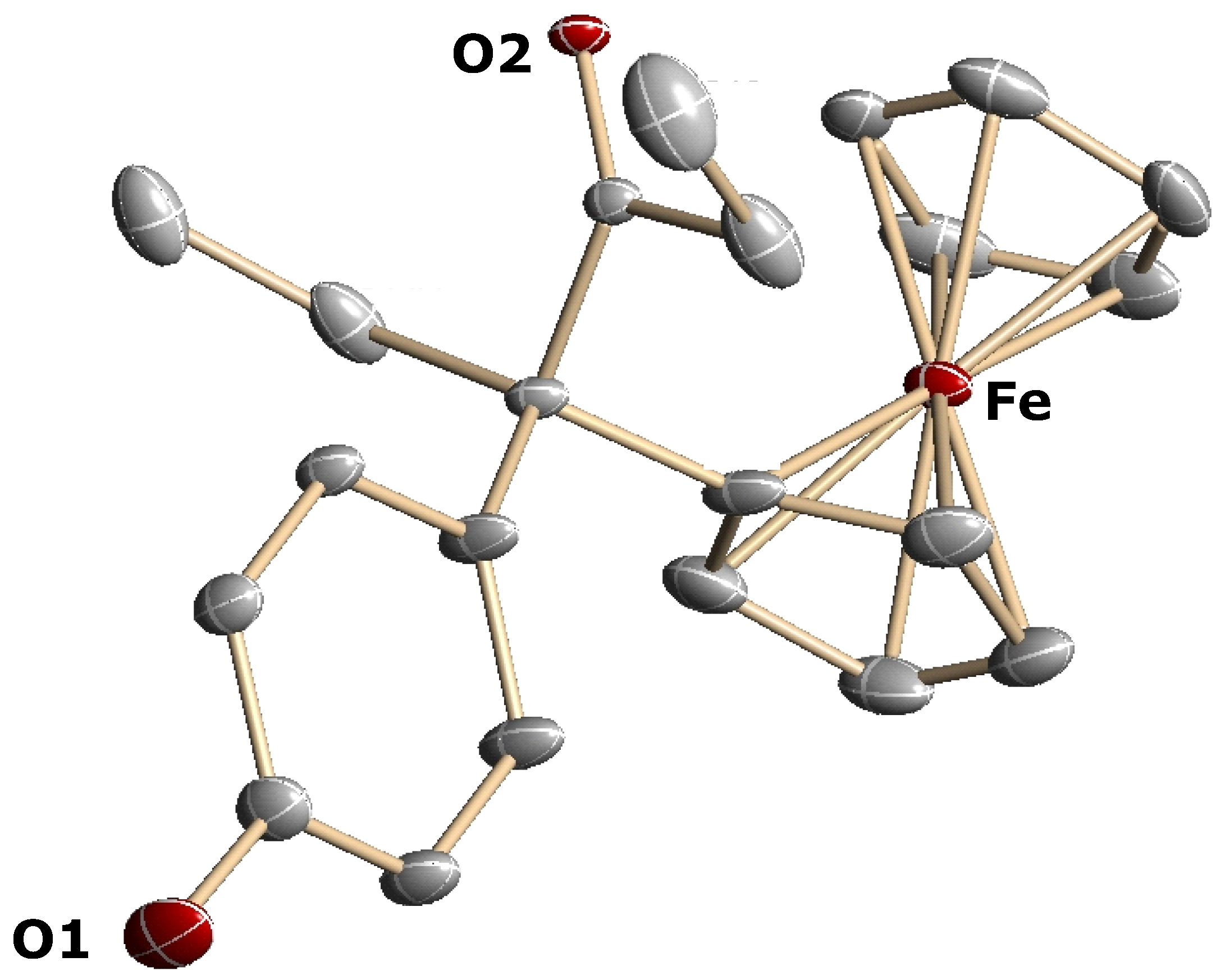
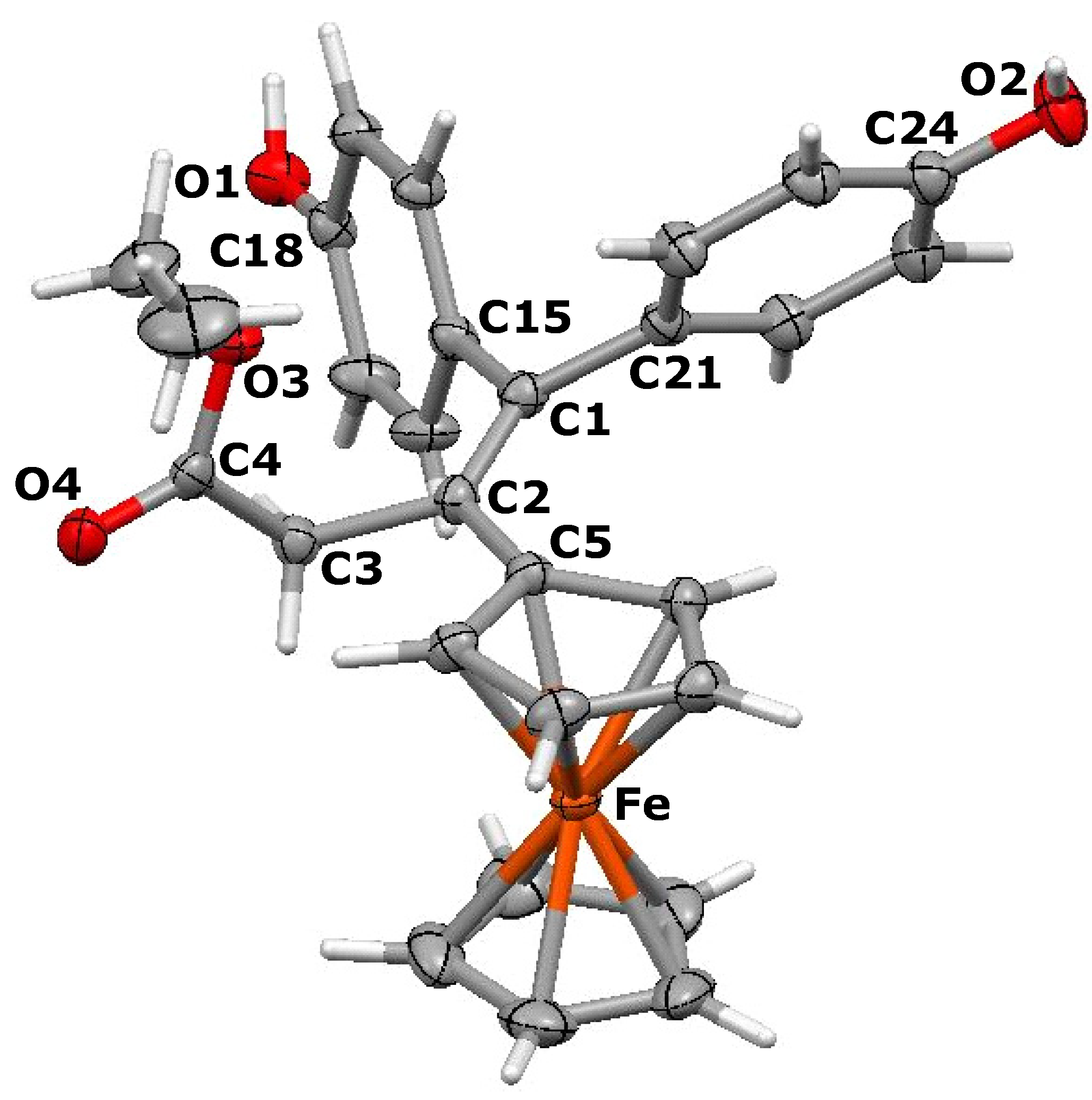
3.3. X-ray Crystallography
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- Jaouen, G.; Top, S. The Ferrocifen family as Potent and Selective Antitumor Compounds: Mechanisms of Action. In Advances in Organometallic Chemistry and Catalysis; Pombeiro, A.J.L., Ed.; Wiley: Hoboken, NJ, USA, 2014; pp. 563–580. [Google Scholar]
- Barry, N.P.E.; Sadler, P.J. Exploration of the medical periodic table: Towards new targets. Chem. Commun. 2013, 49, 5106–5131. [Google Scholar] [CrossRef]
- Bertrand, B.; Casini, A. A golden future in medicinal inorganic chemistry: The promise of anticancer gold organometallic compounds. Dalton Trans. 2014, 43, 4209–4219. [Google Scholar] [CrossRef]
- Braga, S.S.; Silva, A.M.S. A new age for iron: Antitumoral ferrocenes. Organometallics 2013, 32, 5626–5639. [Google Scholar] [CrossRef]
- Hillard, E.A.; Jaouen, G. Bioorganometallics: Future trends in drug discovery, analytical chemistry and catalysis. Organometallics 2011, 30, 20–27. [Google Scholar] [CrossRef]
- Hillard, E.A.; Vessières, A.; Jaouen, G. Topics in organometallic chemistry. In In Medicinal Organometallic Chemistry; Jaouen, G., Metzler-Nolte, N, Eds.; Springer-Verlag: Heidelberg, Germany, 2010; volume 32, p. 81. [Google Scholar]
- Ornelas, C. Application of ferrocene and its derivatives in cancer research. New J. Chem. 2011, 35, 1973–1985. [Google Scholar] [CrossRef]
- Top, S.; Vessières, A.; Leclercq, G.; Quivy, J.; Tang, J.; Vaissermann, J.; Huché, M.; Jaouen, G. Synthesis, biochemical properties and molecular modeling studies of organometallic specific estrogen receptor modulators (SERMs), the ferrocifens and hydroxyferrocifens: Evidence for an antiproliferative effect of hydroxyferrocifens on both hormone-dependent and hormone-independent breast cancer cell lines. Chem. Eur. J. 2003, 9, 5223–5236. [Google Scholar]
- Top, S.; Tang, J.; Vessières, A.; Carrez, D.; Provot, C.; Jaouen, G. Ferrocenyl hydroxytamoxifen: A prototype for a new range of estradiol receptor site-directed cytotoxics. Chem. Commun. 1996, 8, 955–956. [Google Scholar]
- Vessières, A.; Top, S.; Pigeon, P.; Hillard, E.A.; Boubeker, L.; Spera, D.; Jaouen, G. Modification of the estrogenic properties of diphenols by the incorporation of ferrocene. Generation of anti-proliferative effects in vivo. J. Med. Chem. 2005, 48, 3937–3940. [Google Scholar]
- O’Regan, R.M.; Cisneros, A.; England, G.M.; MacGregor, J.I.; Muenzner, H.D.; Assikis, V.J.; Bilimoria, M.M.; Piette, M.; Dragan, Y.P.; Pitot, H.C.; et al. Effects of the antiestrogens tamoxifen, toremifene, and ICI 182,780 on endometrial cancer growth. J. Natl. Cancer Inst. 1998, 90, 1552–1558. [Google Scholar]
- Valavaara, R.; Pyrhönen, S.; Heikkinen, M.; Rissanen, P.; Blanco, G.; Thölix, E.; Nordman, E.; Taskinen, P.; Holsti, L.; Hajba, A. Toremifene, a new antiestrogenic compound, for treatment of advanced breast cancer. Phase II study. J. Cancer Clin. Oncol. 1988, 24, 785–790. [Google Scholar] [CrossRef]
- Gundersen, S. Toremifene, a new antiestrogenic compound in the treatment of metastatic mammary cancer. A phase II study. J. Steroid Biochem. 1990, 36, 233–234. [Google Scholar] [CrossRef]
- Modig, H.; Borgström, S.; Nilsson, I.; Westman, G. Phase II clinical study of toremifene in patients with metastatic breast cancer. Preliminary communication. J. Steroid Biochem. 1990, 36, 235–236. [Google Scholar] [CrossRef]
- Milla-Santos, A.; Milla, L.; Rallo, L.; Solano, V. Phase III randomized trial of toremifene vs. tamoxifen in hormonodependant advanced breast cancer. Breast Cancer Res. Treat. 2001, 65, 119–124. [Google Scholar] [CrossRef]
- Baur, J.A.; Pearson, K.J.; Price, N.L.; Jamieson, H.A.; Lerin, C.; Kalra, A.; Prabhu, V.V.; Allard, J.S.; Lopez-Lluch, G.; Lewis, K.; et al. Resveratrol improves health and survival of mice on a high-calorie diet. Nature 2006, 444, 337–342. [Google Scholar]
- Raghow, S.; Hooshdaran, M.Z.; Katiyar, S.; Steiner, M.S. Toremifene prevents prostate cancer in the transgenic adenocarcinoma of mouse prostate model. Cancer Res. 2002, 62, 1370–1376. [Google Scholar]
- Smith, M.R.; Malkowicz, S.B.; Chu, F.; Forrest, J.; Price, D.; Sieber, P.; Barnette, K.G.; Rodriguez, D.; Steiner, M.S. Toremifene increases bone mineral density in men receiving androgen deprivation therapy for prostate cancer: Interim analysis of a multicenter phase 3 clinical study. J. Urol. 2008, 179, 152–155. [Google Scholar]
- Buriez, O.; Heldt, J.M.; Labbé, E.; Vessières, A.; Jaouen, G.; Amatore, C. Reactivity and antiproliferative activity of ferrocenyl-tamoxifen adducts with cyclodextrins against hormone-independent breast-cancer cell lines. Chem.-Eur. J. 2008, 14, 8195–8203. [Google Scholar]
- Van Lipzig, M.M.H.; ter Laak, A.M.; Jongejan, A.; Vermeulen, N.P.E.; Wamelink, M.; Geerke, D.; Meerman, J.H.N. Prediction of ligand binding affinity and orientation of xenoestrogens to the estrogen receptor by molecular dynamics simulations and the linear interaction energy method. J. Med. Chem. 2004, 47, 1018–1030. [Google Scholar] [CrossRef]
- Giusti, R.M.; Iwamoto, K.; Hatch, E.E. Diethylstilbestrol revisited: A review of the long-term health effects. Ann. Intern. Med. 1995, 122, 778–788. [Google Scholar] [CrossRef]
- Schrager, S.; Potter, B.E. Diethylstilbestrol exposure. Am. Fam. Physician 2004, 69, 2395–2400. [Google Scholar]
- Jaouen, G.; Top, S.; Vessières, A.; Sayer, B.G.; Frampton, C.S.; McGlinchey, M. Molecular recognition using bioorganometallic probes: NMR, X-ray crystallographic, and molecular modeling study of the conformations of chromium tricarbonyl derivatives of hexestrol and their relevance to estradiol-receptor binding. J. Organomet. 1992, 11, 4061–4068. [Google Scholar]
- Tondu, S.; Top, S.; Vessières, A.; Jaouen, G. The use of tricarbonyl chromium hexestrol derivatives in the detection of oestradiol receptor sites. J. Chem. Soc. Chem. Commun. 1985, 326–328. [Google Scholar]
- Gruselle, M.; Malezieux, B.; Sokolov, V.I.; Troitskaya, L.L. Enantioselective synthesis of ferrocene analogs of hexestrol and estradiol; recognition towards estradiol receptors. Inorganica. Chim. Acta 1994, 222, 51–61. [Google Scholar] [CrossRef]
- Hillard, E.A.; Vessières, A.; Top, S.; Pigeon, P.; Kowalski, K.; Huché, M.; Jaouen, G. Organometallic diphenols: The importance of the organometallic moiety on the expression of a cytotoxic effect on breast cancer cells. J. Organomet. Chem. 2007, 692, 1315–1326. [Google Scholar] [CrossRef]
- Elecko, P.; Foltinova, P.; Salisova, M.; Solcaniova, E.; Toma, S. Synthesis, proton magnetic resonance spectra, and biological activity of haloacylferrocenes. Chemicke. Zvesti. 1974, 28, 94–99. [Google Scholar]
- Ephritikhine, M. A new look at the McMurry reaction. Chem. Commun. 1998, 23, 2549–2554. [Google Scholar]
- Görmen, M.; Pigeon, P.; Hillard, E.A.; Vessières, A.; Huché, M.; Richard, M.-A.; McGlinchey, M.J.; Top, S.; Jaouen, G. Synthesis and antiproliferative effects of [3]ferrocenophane transposition products and pinacols obtained from McMurry cross-coupling reactions. Organometallics 2012, 31, 5856–5866. [Google Scholar] [CrossRef]
- Tan, Y.L.K.; Pigeon, P.; Hillard, E.A.; Top, S.; Plamont, M.-A.; Vessières, A.; McGlinchey, M.J.; Müller-Bunz, H.; Jaouen, G. Synthesis, oxidation chemistry and cytotoxicity studies on ferrocene derivatives of diethylstilbestrol. Dalton Trans. 2009, 10871–10881. [Google Scholar]
- Schlögl, K.; Egger, H. Syntheses and reactions of ferrocenylacetylenes. Monatsh. Chem. 1963, 94, 376–392. [Google Scholar] [CrossRef]
- Ortin, Y.; Grealis, J.; Scully, C.; Müller-Bunz, H.; Manning, A.R.; McGlinchey, M.J. McMurry reactions of (η5-acetylcyclopentadienyl)cobalt(η4-tetraphenylcyclo-butadiene) with benzo-phenone: Ketone couplings and a pinacol-pinacolone rearrangement. J. Organomet. Chem. 2004, 689, 4683–4690. [Google Scholar] [CrossRef]
- Baddeley, G. Modern aspects of the Friedel-Crafts reaction. Quart. Rev. 1954, 8, 355–379. [Google Scholar] [CrossRef]
- Hillard, E.A.; Pigeon, P.; Vessières, A.; Amatore, C.; Jaouen, G. The influence of phenolic hydroxy substitution on the electron transfer and anti-cancer properties of compounds based on the 2-ferrocenyl-1-phenyl-but-1-ene motif. Dalton Trans. 2007, 43, 5073–5081. [Google Scholar]
- Hillard, E.A.; Vessières, A.; le Bideau, F.; Plazuk, D.; Spera, D.; Huché, M.; Jaouen, G. A series of unconjugated ferrocenyl phenols: Prospects as anticancer agents. Chem. Med. Chem. 2006, 1, 551–559. [Google Scholar] [CrossRef]
- Heilmann, J.B.; Hillard, E.A.; Plamont, M.-A.; Pigeon, P.; Bolte, M.; Jaouen, G.; Vessières, A. Ferrocenyl compounds possessing protected phenol and thiophenol groups: Synthesis, X-ray structure, and in vitro biological effects against breast cancer. J. Organomet. Chem. 2008, 693, 1716–1722. [Google Scholar] [CrossRef]
- Görmen, M.; Pigeon, P.; Top, S.; Vessières, A.; Plamont, M.-A.; Hillard, E.A.; Jaouen, G. Facile synthesis and strong antiproliferative activity of disubstituted diphenylmethylidenyl-[3]ferrocenophanes on breast and prostate cancer cell lines. Med. Chem. Commun. 2010, 1, 149–151. [Google Scholar] [CrossRef]
- Görmen, M.; Plażuk, D.; Pigeon, P.; Hillard, E.A.; Plamont, M.-A.; Top, S.; Vessières, A.; Jaouen, G. Comparative toxicity of [3]ferrocenophane and ferrocene moieties on breast cancer cells. Tetrahedron Lett. 2010, 51, 118–120. [Google Scholar]
- X-ray data for 14: The unit cell is orthogonal with a = 9.8552(7) Å, b = 9.7478(8) Å, c = 18.523(2) Å, α = β = γ = 90°, V = 1729.5(2) Å3, and the space group is Pnma (#62).
- Sample Availability: Samples of the compounds 5a–c are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pigeon, P.; Görmen, M.; Kowalski, K.; Müller-Bunz, H.; McGlinchey, M.J.; Top, S.; Jaouen, G. Atypical McMurry Cross-Coupling Reactions Leading to a New Series of Potent Antiproliferative Compounds Bearing the Key [Ferrocenyl-Ene-Phenol] Motif. Molecules 2014, 19, 10350-10369. https://doi.org/10.3390/molecules190710350
Pigeon P, Görmen M, Kowalski K, Müller-Bunz H, McGlinchey MJ, Top S, Jaouen G. Atypical McMurry Cross-Coupling Reactions Leading to a New Series of Potent Antiproliferative Compounds Bearing the Key [Ferrocenyl-Ene-Phenol] Motif. Molecules. 2014; 19(7):10350-10369. https://doi.org/10.3390/molecules190710350
Chicago/Turabian StylePigeon, Pascal, Meral Görmen, Konrad Kowalski, Helge Müller-Bunz, Michael J. McGlinchey, Siden Top, and Gérard Jaouen. 2014. "Atypical McMurry Cross-Coupling Reactions Leading to a New Series of Potent Antiproliferative Compounds Bearing the Key [Ferrocenyl-Ene-Phenol] Motif" Molecules 19, no. 7: 10350-10369. https://doi.org/10.3390/molecules190710350
APA StylePigeon, P., Görmen, M., Kowalski, K., Müller-Bunz, H., McGlinchey, M. J., Top, S., & Jaouen, G. (2014). Atypical McMurry Cross-Coupling Reactions Leading to a New Series of Potent Antiproliferative Compounds Bearing the Key [Ferrocenyl-Ene-Phenol] Motif. Molecules, 19(7), 10350-10369. https://doi.org/10.3390/molecules190710350