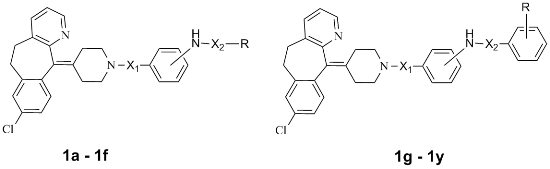
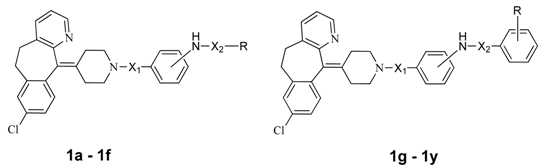
Synthesis and Biological Evaluation of Substituted Desloratadines as Potent Arginine Vasopressin V2 Receptor Antagonists
Abstract
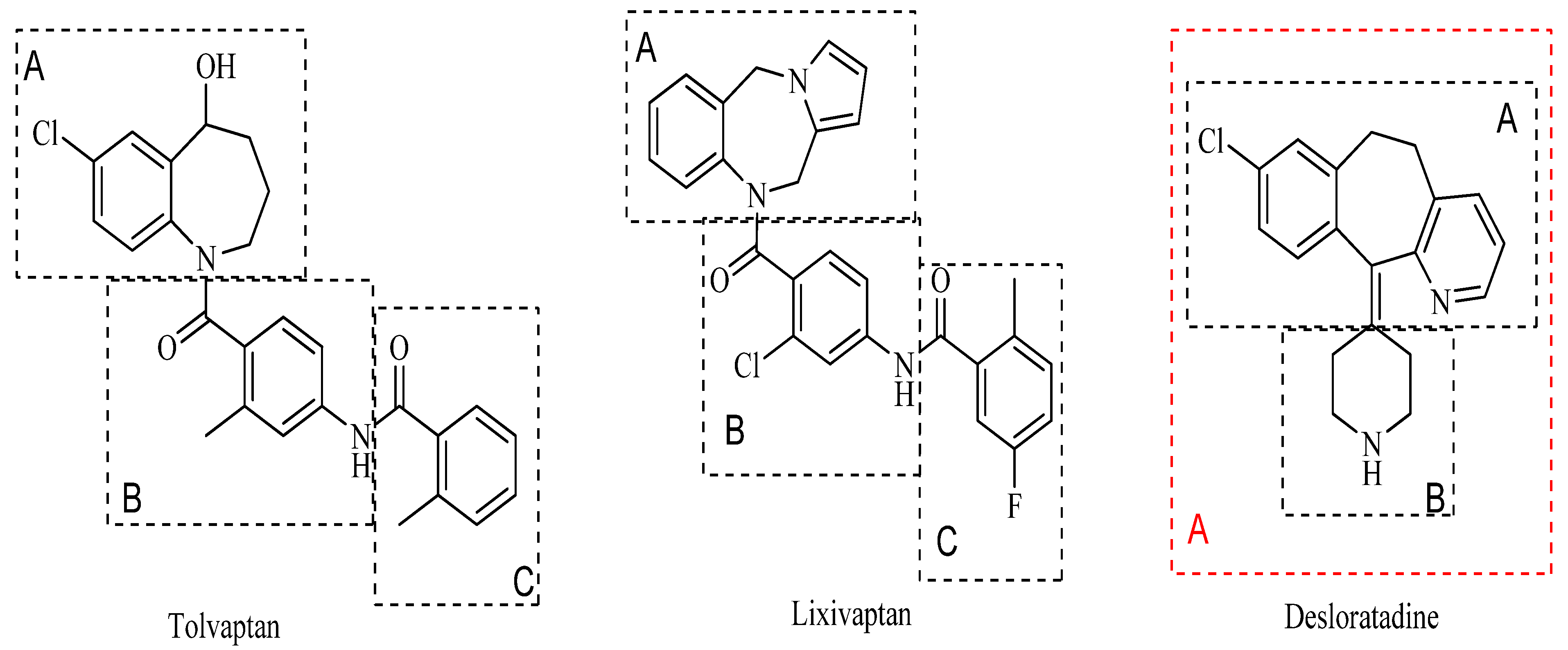
:1. Introduction
| Compound | Structures of Target Compound | Bingding Assy (IC50; nmol/mL) | cAMP Assay (V2, IC50; nmol/mL) | Volume of Urine (mL, 0–20 h) | ||||
|---|---|---|---|---|---|---|---|---|
| X1 | X2 | Position Of -NH2 | R | V2 | V1a | |||
| 1a | CO | CO | p-NH2 | -CH2CH3 | 6.3 | 130 | 26 | 27.5±5.7 |
| 1b | CO | CO | p-NH2 | -CH2CH2CH3 | 47 | >1000 | ||
| 1c | CO | CO | p-NH2 | -CH2CH2CH2Cl | 25 | >1000 | ||
| 1d | CO | SO2 | p-NH2 | -CH2CH3 | 11 | 92 | 160 | 21.4±4.1 |
| 1e | SO2 | CO | m-NH2 | -CH2CH2CH3 | 26 | 176 | ||
| 1f | SO2 | CO | p-NH2 | -CH2CH2CH3 | 40 | 480 | ||
| 1g | CO | CO | p-NH2 | H | 19 | 330 | ||
| 1h | CO | CO | p-NH2 | 2-Me | 23 | 210 | ||
| 1i | CO | CO | p-NH2 | 4-Me | 18 | 220 | ||
| 1j | CO | CO | p-NH2 | 3-Me | 15 | 370 | 53 | 19.3 ± 5.5 |
| 1k | CO | CO | p-NH2 | 2-Cl | 27 | 490 | ||
| 1l | CO | CO | p-NH2 | 3-Cl | 16 | 560 | ||
| 1m | CO | CO | p-NH2 | 2-F | 20 | 170 | ||
| 1n | CO | CO | p-NH2 | 3-OMe | 8.5 | 390 | 380 | 19.9 ± 6.7 |
| 1o | CO | CO | p-NH2 | 3-NO2 | 18 | 550 | ||
| 1p | CO | CO | p-NH2 | 4-NO2 | 52 | >1000 | ||
| 1q | CO | SO2 | p-NH2 | 4-Me | 24 | 720 | ||
| 1r | CO | SO2 | p-NH2 | 2-Cl | 11 | 830 | 220 | 16.1 ± 3.2 |
| 1s | CO | SO2 | p-NH2 | 2,5-DiCl | 9.2 | 320 | 37 | 18.3 ± 4.3 |
| 1t | SO2 | CO | m-NH2 | H | 7.7 | >1000 | 98 | 28.1 ± 5.0 |
| 1u | SO2 | CO | m-NH2 | 3-Cl | 19 | 840 | ||
| 1v | SO2 | CO | m-NH2 | 3- NO2 | 5.5 | 630 | 110 | 11.9 ± 2.7 |
| 1w | SO2 | SO2 | m-NH2 | 4-Me | 30 | 860 | ||
| 1x | SO2 | CO | p-NH2 | 3-Cl | 310 | >1000 | ||
| 1y | SO2 | SO2 | p-NH2 | 4-Me | 170 | 920 | ||
| Control | 6.5 ± 0.5 | |||||||
| tolvaptan | 28.0 ± 6.5 | |||||||
2. Results and Discussion

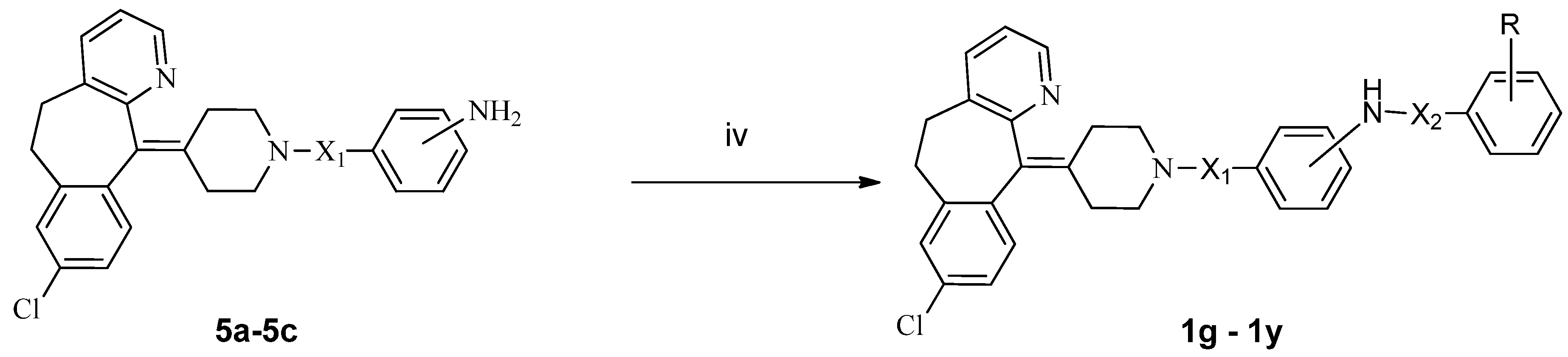
3. Experimental
3.1. General Information
3.2. Synthesis
3.3. Biological Evaluation
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Bowman, B.T.; Rosner, M.H. Lixivaptan-an evidence-based review of its clinical potential in the treatment of hyponatremia. Core Evid. 2013, 8, 47–56. [Google Scholar]
- Kwiatkowska, A.; Lewandowska, M.; Borovickova, L.; Slaninova, J.; Lammek, B.; Prahl, A. Design, synthesis and structure-activity relationship of new arginine vasopressin analogues containing proline derivatives in position 2. Chem. Biol. Drug Des. 2013, 81, 420–428. [Google Scholar]
- Kwiatkowska, A.; Sobolewski, D.; Prahl, A.; Borovickova, L.; Slaninova, J.; Lammek, B. Arginine vasopressin and its analogues-the influence of position 2 modification with 3,3-diphenylalanine enantiomers. Highly potent V-2 agonists. Eur. J. Med. Chem. 2009, 44, 2862–2867. [Google Scholar] [CrossRef]
- Wong, L.L.; Verbalis, J.G. Vasopressin V2 receptor antagonists. Cardiovasc. Res. 2001, 51, 391–402. [Google Scholar] [CrossRef]
- Ohtake, Y.; Naito, A.; Hasegawa, H.; Kawano, K.; Morizono, D.; Taniguchi, M.; Tanaka, Y.; Matsukawa, H.; Naito, K.; Oguma, T.; et al. Novel vasopressin V2 receptor-selective antagonists, pyrrolo[2,1-a]quinoxaline and pyrrolo[2,1-c][1,4]benzodiazepine derivatives. Bioorg. Med. Chem. 1999, 7, 1247–1254. [Google Scholar] [CrossRef]
- Ghali, J.K.; Orlandi, C.; Abraham, W.T.; Investigators, C.-L.S. The efficacy and safety of lixivaptan in outpatients with heart failure and volume overload: Results of a multicentre, randomized, double-blind, placebo-controlled, parallel-group study. Eur. J. Heart Fail. 2012, 14, 642–651. [Google Scholar] [CrossRef]
- Pang, P.S.; Gheorghiade, M.; Dihu, J.; Swedberg, K.; Khan, S.; Maggioni, A.P.; Grinfeld, L.; Zannad, F.; Burnett, J.C.; Ouyang, J.; et al. Effects of tolvaptan on physician-assessed symptoms and signs in patients hospitalized with acute heart failure syndromes: Analysis from the efficacy of vasopressin antagonism in heart failure outcome study with tolvaptan (EVEREST) trials. Am. Heart J. 2011, 161, 1067–1072. [Google Scholar] [CrossRef]
- Gheorghiade, M.; Pang, P.S.; O’Connor, C.M.; Prasad, K.; McMurray, J.; Teerlink, J.R.; Fiuzat, M.; Sabbah, H.; Komajda, M. Clinical development of pharmacologic agents for acute heart failure syndromes: A proposal for a mechanistic translational phase. Am. Heart J. 2011, 161, 224–232. [Google Scholar] [CrossRef]
- Veeraveedu, P.T.; Palaniyandi, S.S.; Yamaguchi, K.; Komai, Y.; Thandavarayan, R.A.; Sukumaran, V.; Watanabe, K. Arginine vasopressin receptor antagonists (vaptans): Pharmacological tools and potential therapeutic agents. Drug Discov. Today 2010, 15, 826–841. [Google Scholar] [CrossRef]
- Matsuhisa, A.; Taniguchi, N.; Koshio, H.; Yatsu, T.; Tanaka, A. Nonpeptide arginine vasopressin antagonists for both V1A and V2 Receptors: Synthesis and pharmacological properties of 4'-(1,4,5,6-tetrahydroimidazo[4,5-d][1]benzoazepine-6-carbonyl)benzanilide derivatives and 4'-(5,6-dihydro-4h-thiazolo[5,4-d][1]benzoazepine-6-carbonyl)benzanilide derivatives. Chem. Pharm. Bull. 2000, 48, 21–31. [Google Scholar] [CrossRef]
- Otsuka, F.; Ogura, T.; Yamauchi, T.; Oishi, T.; Hashimoto, M.; Mimura, Y.; Makino, H. Effects of OPC-216268, a vasopressin V1-receptor antagonist, on expression of growth factors from glomeruli in spontaneously hypertensive rats. Regul. Pept. 1997, 72, 87–95. [Google Scholar] [CrossRef]
- Masuda, S.; Hattori, A.; Matsumoto, H.; Miyazawa, S.; Natori, Y.; Mizutani, S.; Tsujimoto, M. Involvement of the V-2 receptor in vasopressin-stimulated translocation of placental leucine aminopeptidase/oxytocinase in renal cells. Eur. J. Biochem. 2003, 270, 1988–1994. [Google Scholar] [CrossRef]
- Blair, J.E.A.; Pang, P.S.; Schrier, R.W.; Metra, M.; Traver, B.; Cook, T.; Campia, U.; Ambrosy, A.; Burnett, J.C.; Grinfeld, L.; et al. Changes in renal function during hospitalization and soon after discharge in patients admitted for worsening heart failure in the placebo group of the EVEREST trial. Eur. Heart J. 2011, 32, 2563–2572. [Google Scholar] [CrossRef]
- Sakaida, I.; Yamashita, S.; Kobayashi, T.; Komatsu, M.; Sakai, T.; Komorizono, Y.; Okada, M.; Okita, K.; Grp, A.D.A.S. Efficacy and safety of a 14-day administration of tolvaptan in the treatment of patients with ascites in hepatic oedema. J. Int. Med. Res. 2013, 41, 835–847. [Google Scholar] [CrossRef]
- Walcott, B.P.; Kahle, K.T.; Simard, J.M. Novel treatment targets for cerebral edema. Neurotherapeutics 2012, 9, 65–72. [Google Scholar] [CrossRef]
- Lee, J.-Y.; Huo, T.-I.; Wang, S.-S.; Huang, H.-C.; Lee, F.-Y.; Lin, H.-C.; Chuang, C.-L.; Lee, S.-D. Diabetes diminishes the portal-systemic collateral vascular response to vasopressin via vasopressin receptor and g(alpha) proteins regulations in cirrhotic rats. PLoS One 2013, 8, e67703. [Google Scholar]
- Guyader, D.; Patat, A.; Ellis-Grosse, E.J.; Orczyk, G.P. Pharmacodynamic effects of a nonpeptide antidiuretic hormone V2 antagonist in cirrhotic patients with ascites. Hepatology 2002, 36, 1197–1205. [Google Scholar] [CrossRef]
- Kazory, A. Hyponatremia in heart failure: Revisiting pathophysiology and therapeutic strategies. Clin. Cardiol. 2010, 33, 322–329. [Google Scholar] [CrossRef]
- Dasta, J.F.; Chiong, J.R.; Christian, R.; Friend, K.; Lingohr-Smith, M.; Lin, J.; Cassidy, I.B. Update on tolvaptan for the treatment of hyponatremia. Expert Rev. Pharmacoecon. Outcomes Res. 2012, 12, 399–410. [Google Scholar] [CrossRef]
- Bettari, L.; Fiuzat, M.; Felker, G.M.; O’Connor, C.M. Significance of hyponatremia in heart failure. Heart Fail. Rev. 2012, 17, 17–26. [Google Scholar] [CrossRef]
- Aronson, D.; Verbalis, J.G.; Mueller, M.; Krum, H.; Investigators, D. Short- and long-term treatment of dilutional hyponatraemia with satavaptan, a selective arginine vasopressin V-2-receptor antagonist: The DILIPO study. Eur. J. Heart Fail. 2011, 13, 327–336. [Google Scholar] [CrossRef]
- Gross, P. Treatment of hyponatremia. Intern. Med. 2008, 47, 885–891. [Google Scholar] [CrossRef]
- Artom, N.; Oddo, S.; Pende, A.; Ottonello, L.; Giusti, M.; Dallegri, F. Syndrome of inappropriate antidiuretic hormone secretion and Ibuprofen, a rare association to be considered: Role of tolvaptan. Case Rep. Endocrinol. 2013, 2013, 818259–818259. [Google Scholar]
- Rotas, G.; Kimbaris, A.; Varvounis, G. Synthesis of a novel pyrrolo[1,2-c][1.3]benzodiazepine analogue of VPA-985. Tetrahedron 2011, 67, 7805–7810. [Google Scholar] [CrossRef]
- Crombie, A.L.; Antrilli, T.M.; Campbell, B.A.; Crandall, D.L.; Failli, A.A.; He, Y.; Kern, J.C.; Moore, W.J.; Nogle, L.M.; Trybulski, E.J. Synthesis and evaluation of azabicyclo 3.2.1 octane derivatives as potent mixed vasopressin antagonists. Bioorg. Med. Chem. Lett. 2010, 20, 3742–3745. [Google Scholar] [CrossRef]
- Ponthieux, S.; Cabot, J.; Mouillac, B.; Seyer, R.; Barberis, C.; Carnazzi, E. Design of benzophenone-containing photoactivatable linear vasopressin antagonists: Pharmacological and photoreactive properties. J. Med. Chem. 2005, 48, 3379–3388. [Google Scholar] [CrossRef]
- Kakefuda, A.; Suzuki, T.; Tobe, T.; Tsukada, J.; Tahara, A.; Sakamoto, S.; Tsukamoto, S. Synthesis and pharmacological evaluation of 5-(4-biphenyl)-3-methyl-4-phenyl-1,2,4-triazole derivatives as a novel class of selective antagonists for the human vasopressin V-1A receptor. J. Med. Chem. 2002, 45, 2589–2598. [Google Scholar] [CrossRef]
- Carnazzi, E.; Aumelas, A.; Mouillac, B.; Breton, C.; Guillou, L.; Barberis, C.; Seyer, R. Design, synthesis and pharmacological characterization of a potent radioiodinated and photoactivatable peptidic oxytocin antagonist. J. Med. Chem. 2001, 44, 3022–3030. [Google Scholar] [CrossRef]
- Kondo, K.; Ogawa, H.; Shinohara, T.; Kurimura, M.; Tanada, Y.; Kan, K.; Yamashita, H.; Nakamura, S.; Hirano, T.; Yamamura, Y.; et al. Novel design of nonpeptide AVP V-2 receptor agonists: Structural requirements for an agonist having 1-(4-aminobenzoyl)-2,3,4,5-tetrahydro-1H-1-benzazepine as a template. J. Med. Chem. 2000, 43, 4388–4397. [Google Scholar]
- Albright, J.D.; Reich, M.F.; Delos Santos, E.G.; Dusza, J.P.; Sum, F.W.; Venkatesan, A.M.; Coupet, J.; Chan, P.S.; Ru, X.; Mazandarani, H.; et al. 5-fluoro-2-methyl-N- 4-(5H-pyrrolo 2,1-c 1,4 benzodiazepin-10(11H)-ylca rbonyl)-3-chlorophenyl benzamide (VPA-985): An orally active arginine vasopressin antagonist with selectivity for V-2 receptors. J. Med. Chem. 1998, 41, 2442–2444. [Google Scholar] [CrossRef]
- Mu, S.; Xie, X.-S.; Niu, D.; Zhang, D.-S.; Liu, D.-K.; Liu, C.-X. Synthesis and biological evaluation of novel derivatives of desloratadine. Chin. Chem. Lett. 2013, 24, 531–534. [Google Scholar]
- Geha, R.S.; Meltzer, E.O. Desloratadine: A new, nonsedating, oral antihistamine. J. Allergy Clin. Immunol. 2001, 107, 751–762. [Google Scholar] [CrossRef]
- Xie, X.-S.; Mu, S.; Tan, C.-B.; Zhou, Z.-X.; Liu, D.-K.; Xu, W.-R. Synthesis and diuretic activity of novel amide derivatives of desloratadine. (In Chinese)Chin. J. Synth. Chem. 2014, 22. in press. [Google Scholar]
- Sample Availability: Samples of the compounds 1a–y are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mu, S.; Liu, Y.; Gong, M.; Liu, D.-K.; Liu, C.-X. Synthesis and Biological Evaluation of Substituted Desloratadines as Potent Arginine Vasopressin V2 Receptor Antagonists. Molecules 2014, 19, 2694-2706. https://doi.org/10.3390/molecules19022694
Mu S, Liu Y, Gong M, Liu D-K, Liu C-X. Synthesis and Biological Evaluation of Substituted Desloratadines as Potent Arginine Vasopressin V2 Receptor Antagonists. Molecules. 2014; 19(2):2694-2706. https://doi.org/10.3390/molecules19022694
Chicago/Turabian StyleMu, Shuai, Ying Liu, Min Gong, Deng-Ke Liu, and Chang-Xiao Liu. 2014. "Synthesis and Biological Evaluation of Substituted Desloratadines as Potent Arginine Vasopressin V2 Receptor Antagonists" Molecules 19, no. 2: 2694-2706. https://doi.org/10.3390/molecules19022694
APA StyleMu, S., Liu, Y., Gong, M., Liu, D.-K., & Liu, C.-X. (2014). Synthesis and Biological Evaluation of Substituted Desloratadines as Potent Arginine Vasopressin V2 Receptor Antagonists. Molecules, 19(2), 2694-2706. https://doi.org/10.3390/molecules19022694