Supercritical Fluid Extraction of Plant Flavors and Fragrances
Abstract
:1. Introduction
1.1. Definition and Properties of Supercritical Fluids
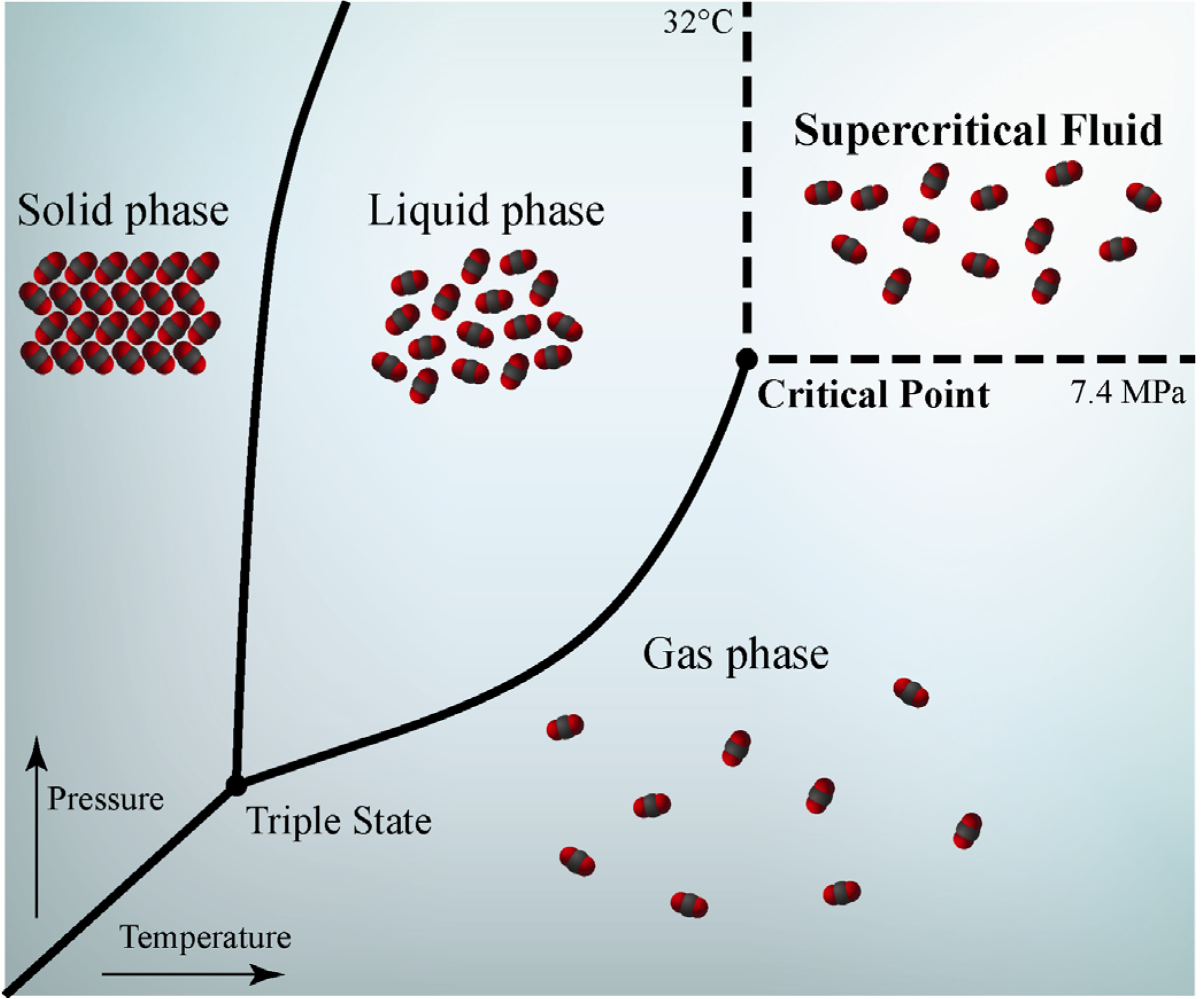
1.2. Supercritical Fluids for Agronomic and Industrial Applications
| Solvent | Critical Temperature °C | Critical Pressure MPa |
|---|---|---|
| Water | 374.0 | 22.1 |
| Methanol | −34.4 | 8.0 |
| Carbon dioxide | 31.2 | 7.3 |
| Ethane | 32.4 | 4.8 |
| Nitrous oxide | 36.7 | 7.1 |
| Propane | 96.6 | 4.2 |
1.3. Other Supercritical Fluids
1.4. Supercritical Fluid Extraction of Flavors and Fragrances
2. Supercritical Fluid Extraction of Flavors
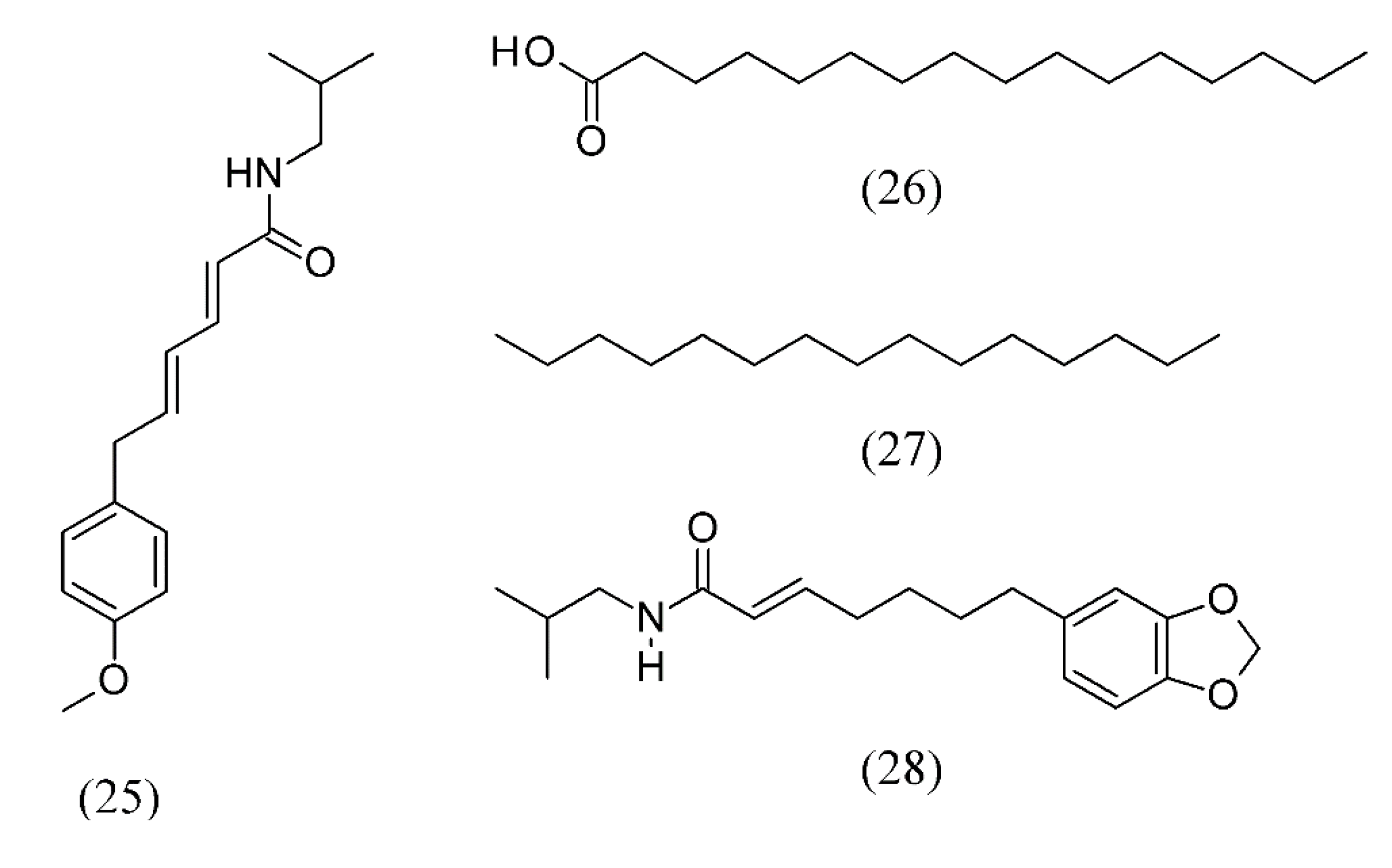
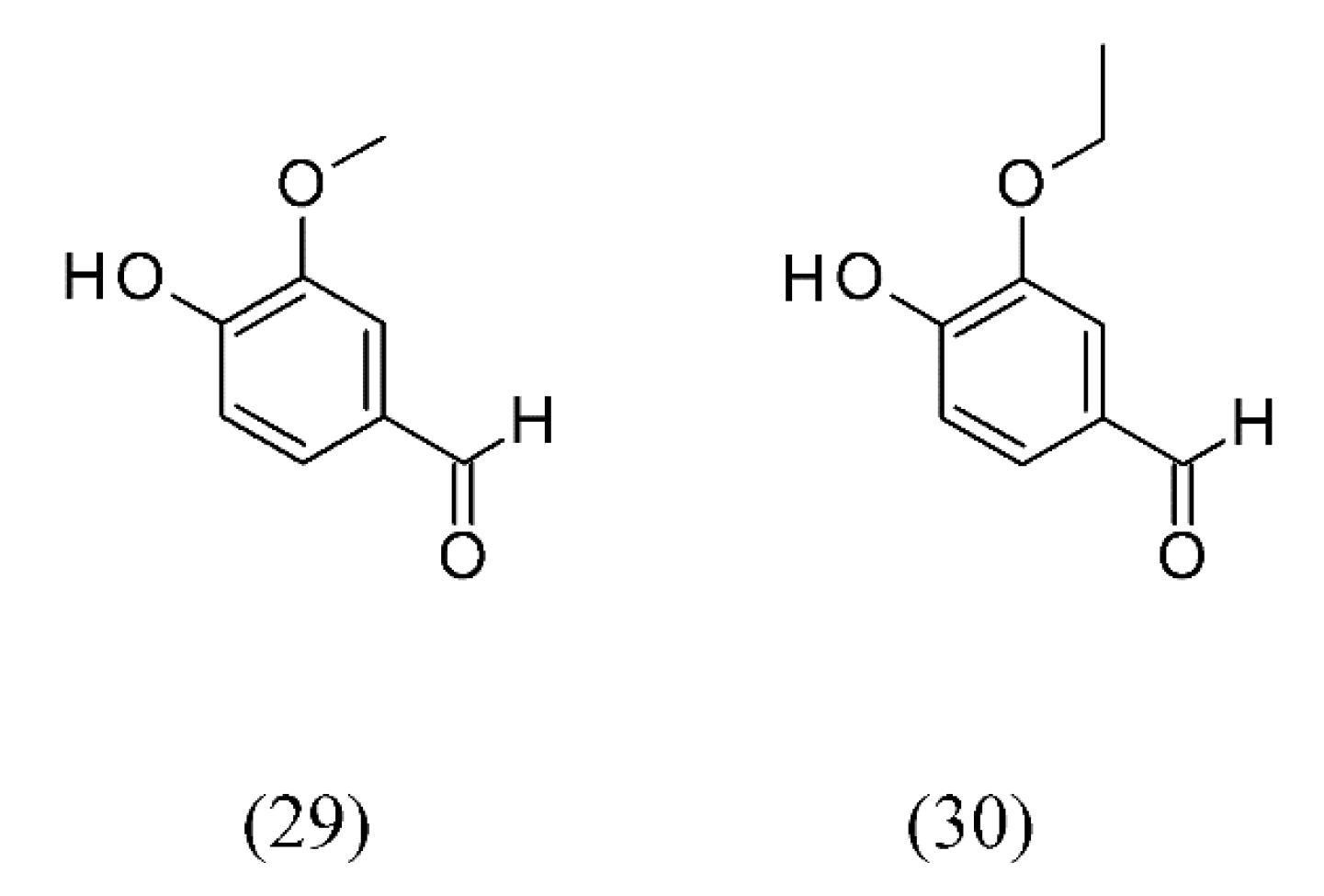
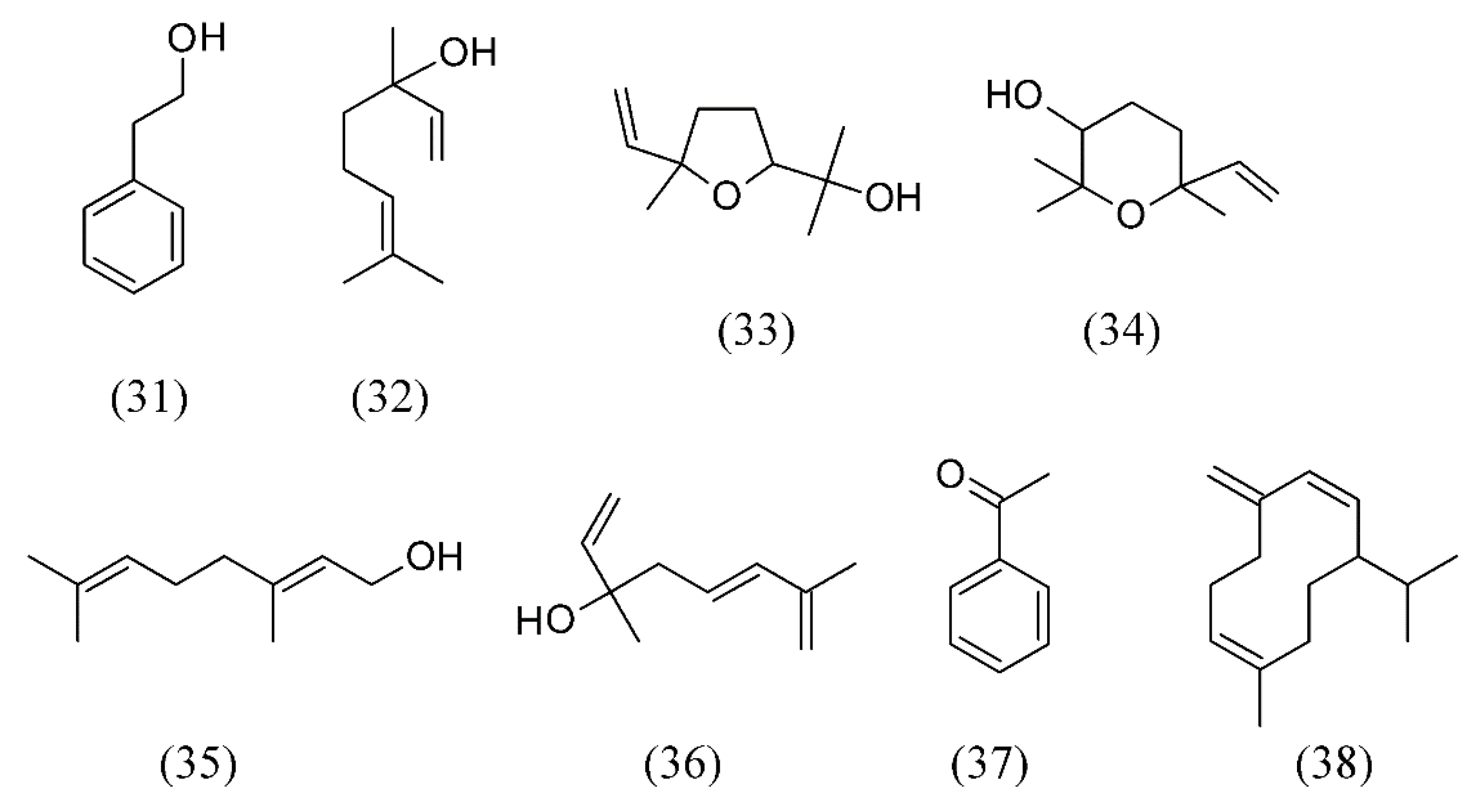
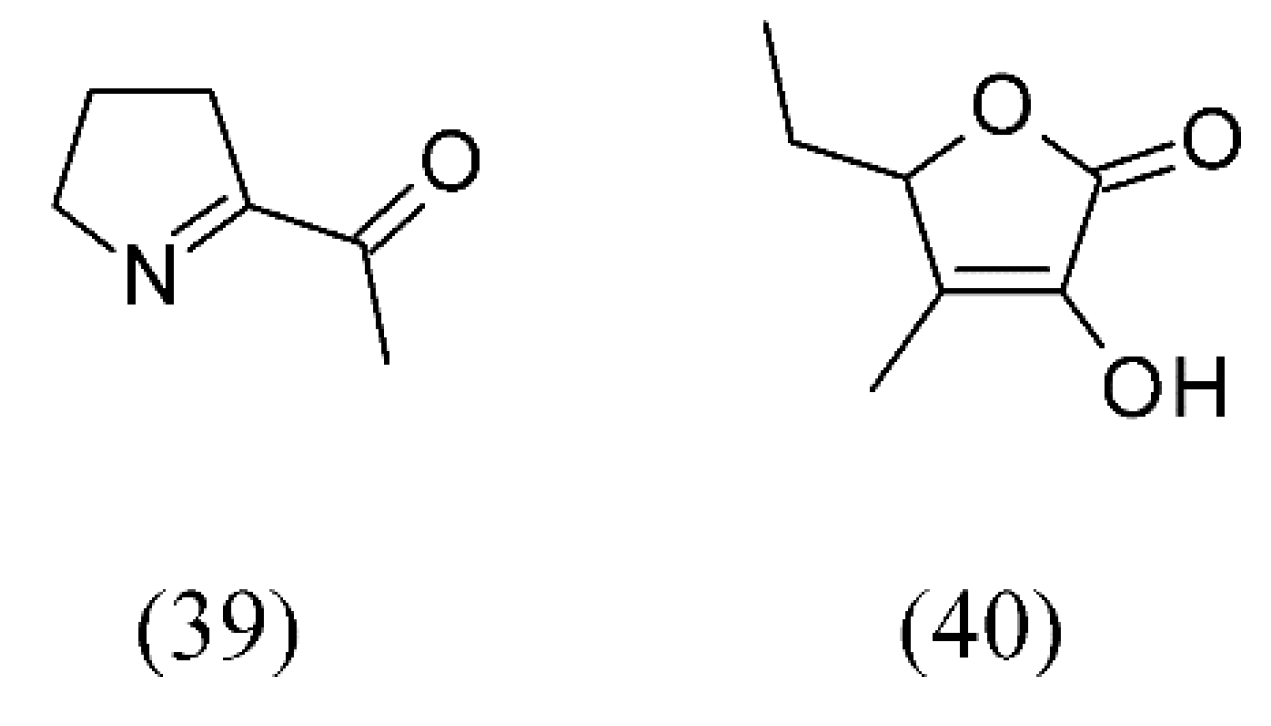
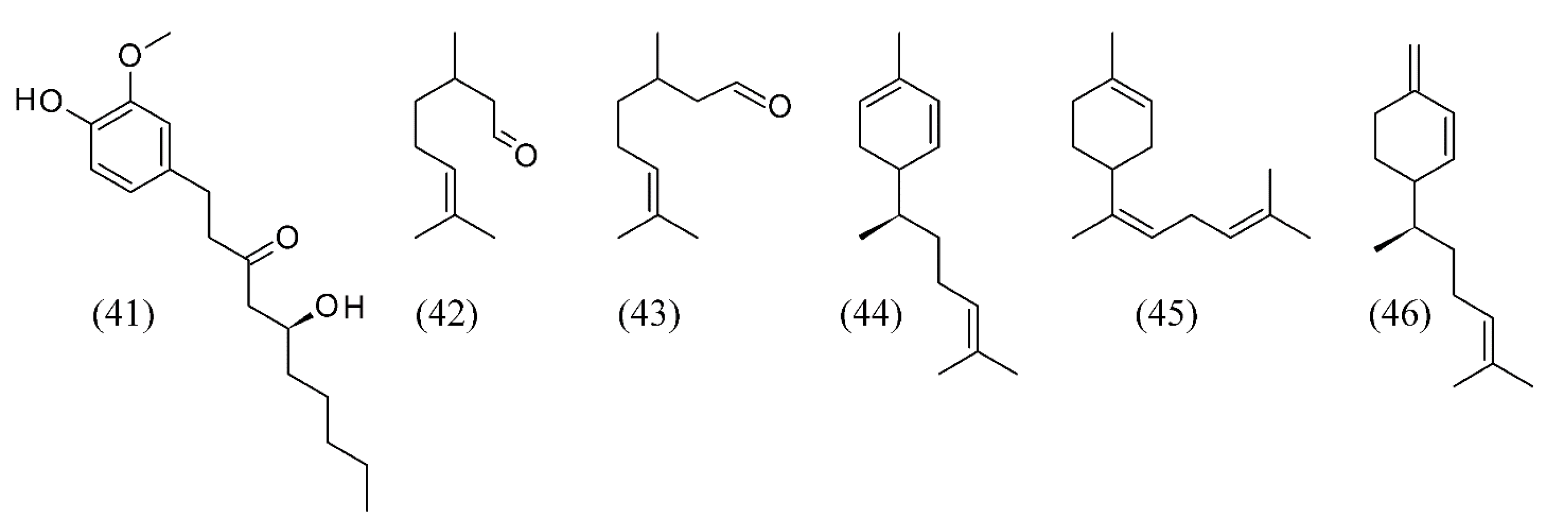
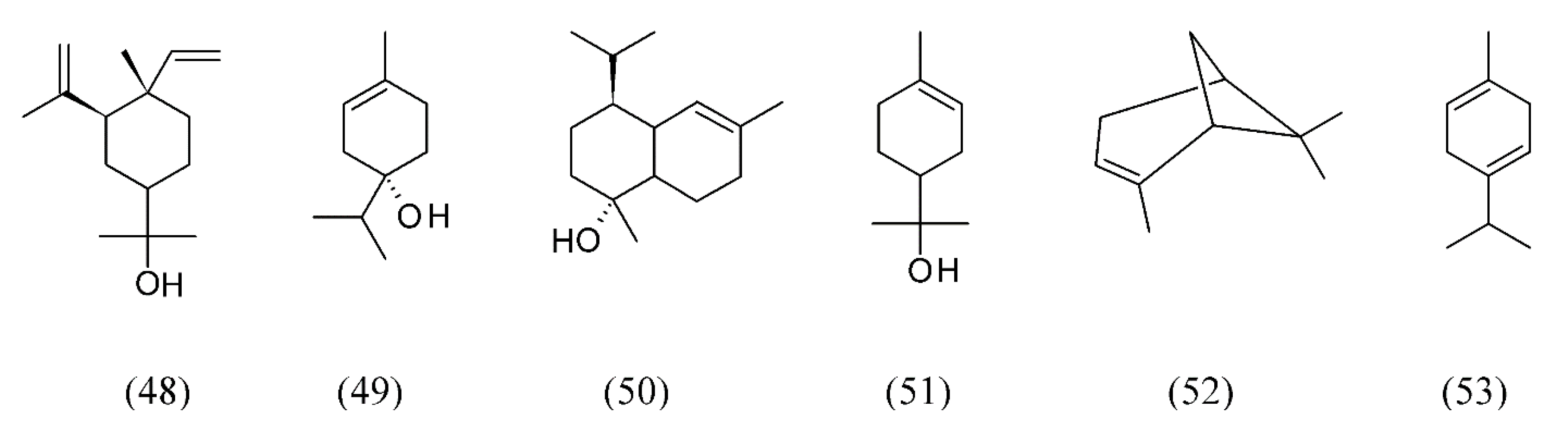
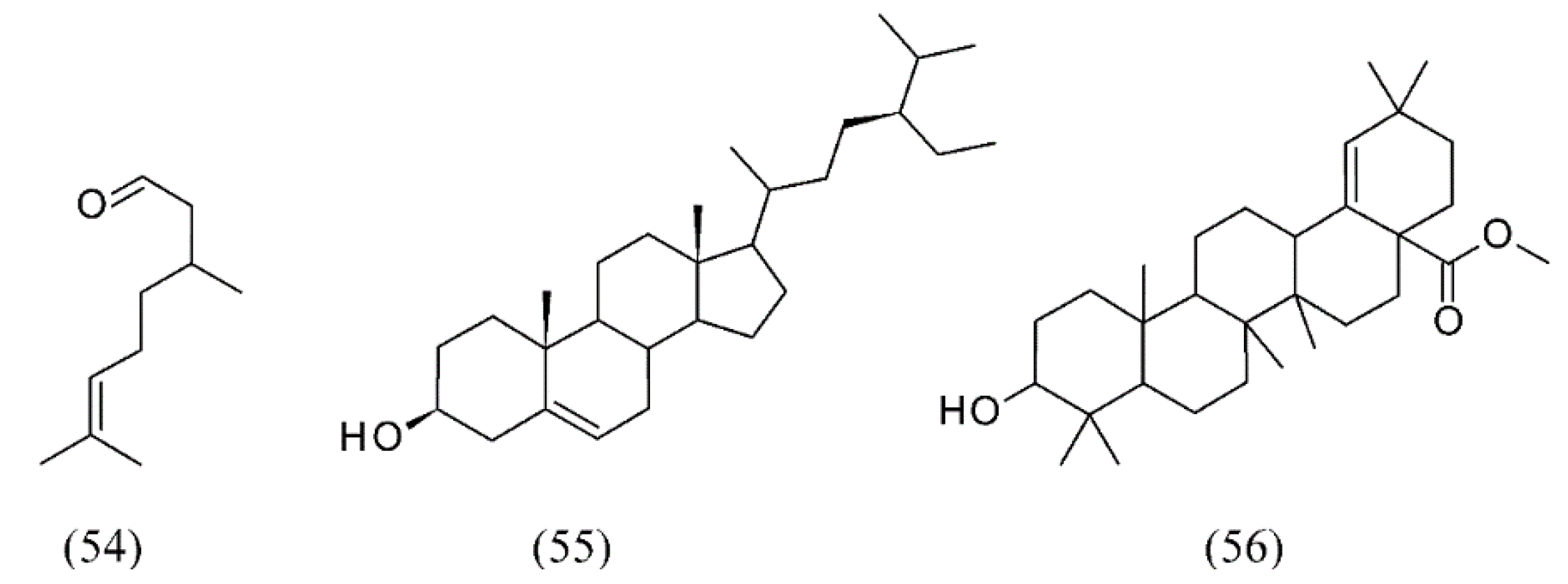
3. Supercritical Fluid Extraction of Fragrances
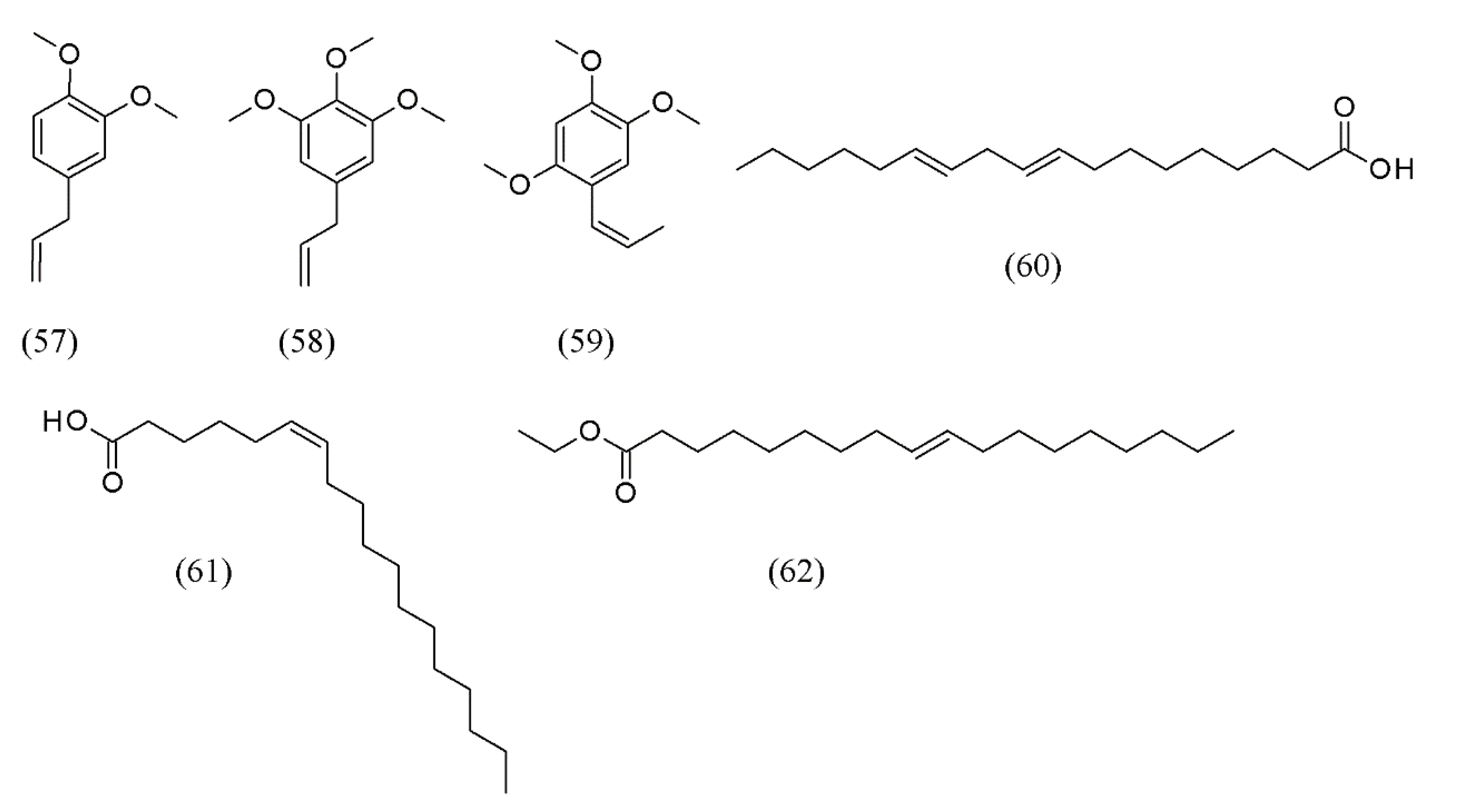
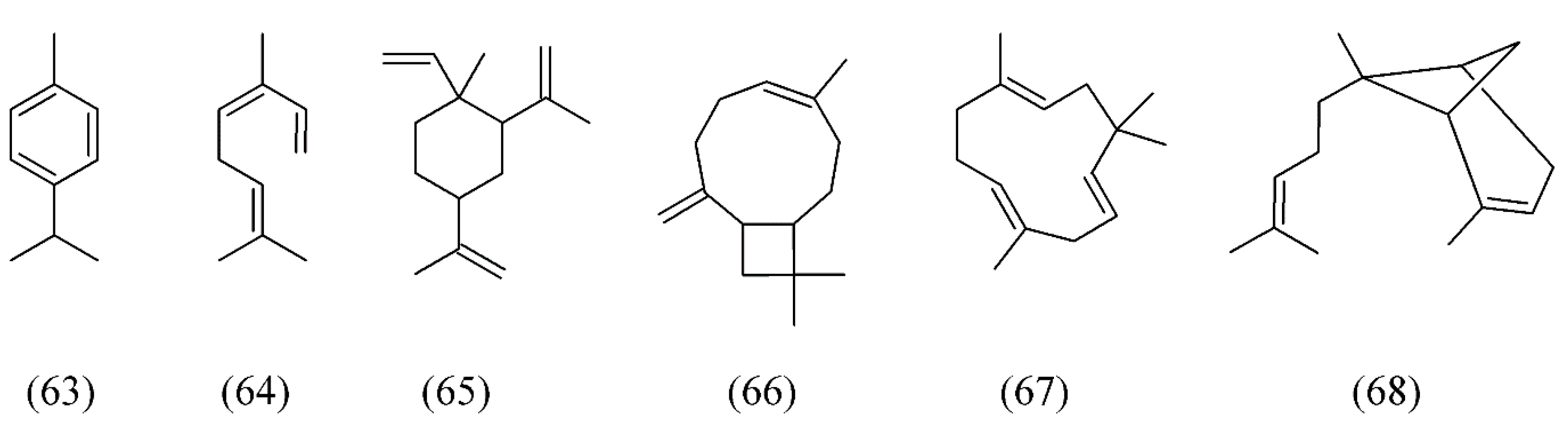
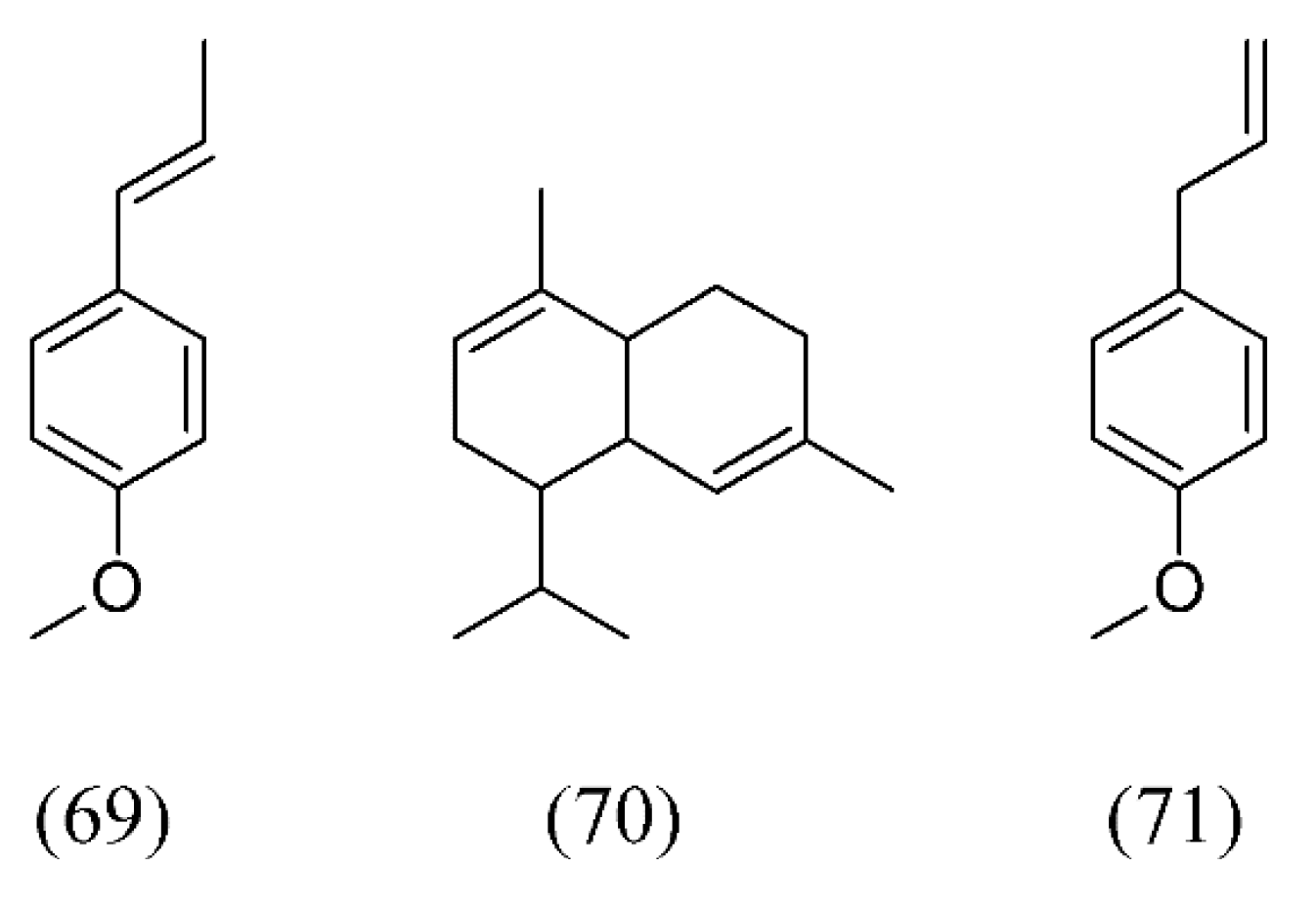
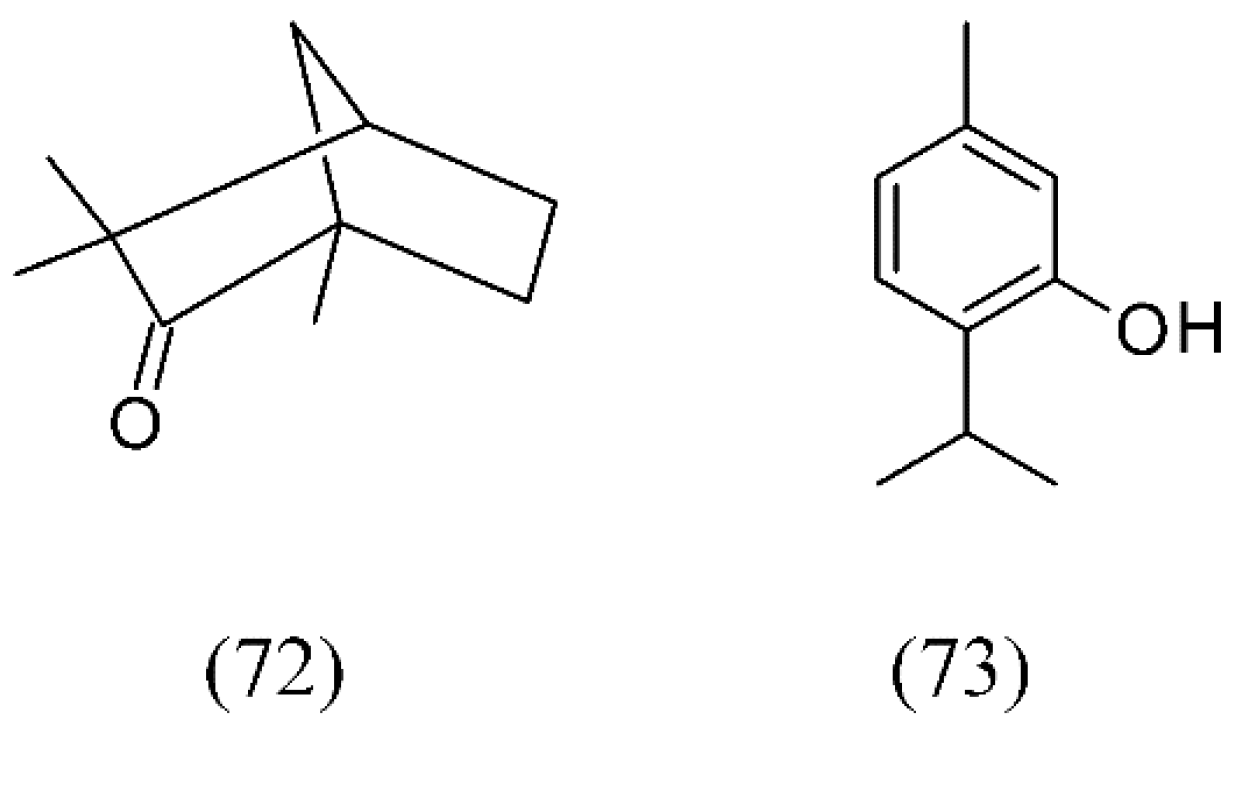
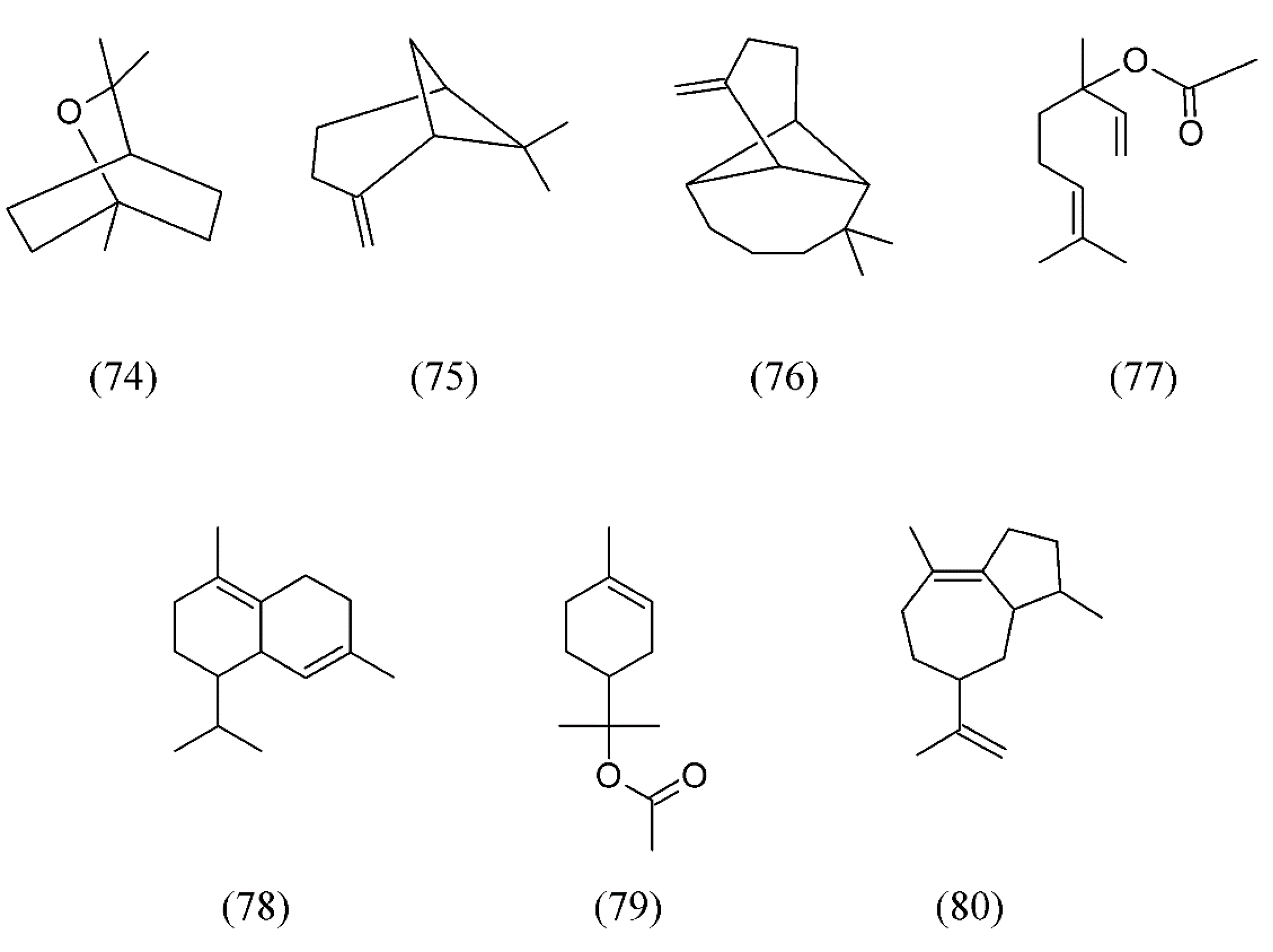
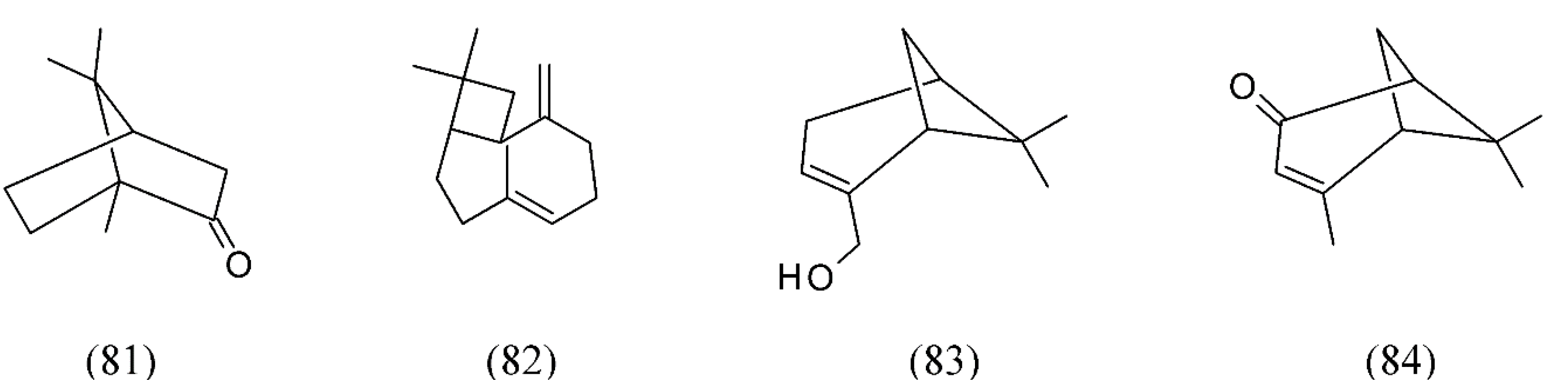
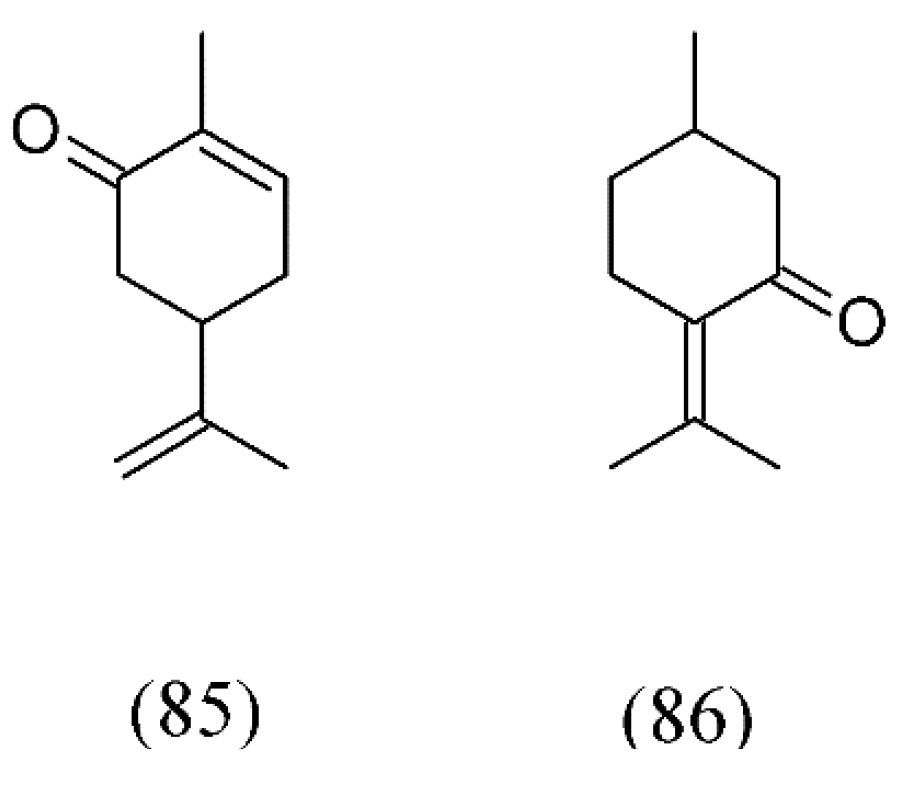
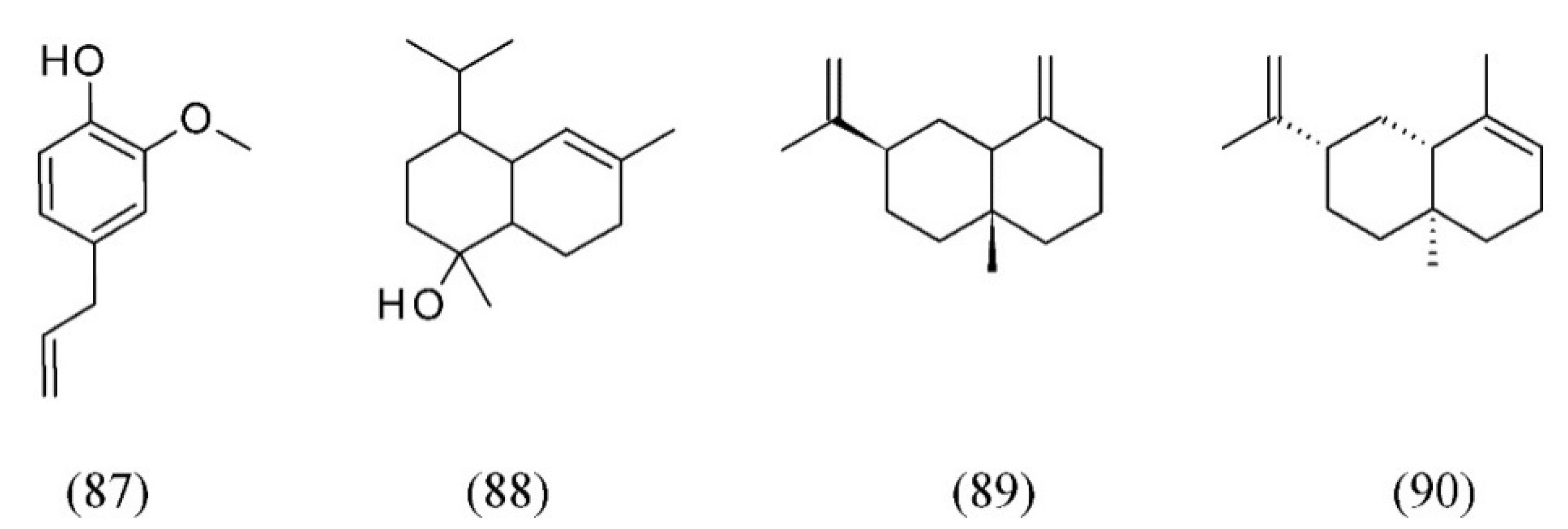
| Plant species | Raw material | Main compound extracted or process | Co-solvent | Ref. |
|---|---|---|---|---|
| Flavors | ||||
| n/a | cheddar and parmesan cheese | defatted cheese | None | [45] |
| n/a | cigarettes | volatile and semivolatile compounds | None | [66] |
| n/a | distilled alcoholic beverages | aromatic components | None | [67] |
| n/a | flavored sugars | vanillin (29) and ethyl vanillin (30) extraction | None | [57] |
| n/a | whey protein isolate | volatile removal | None | [71] |
| Allium cepa | onion flavor | essential oil with a high-sulphur content | Ethanol | [49,50] |
| Allium sativum | homogenized garlic | thiosulfinates, zwiebelanes, and bissulfine | None | [51,52] |
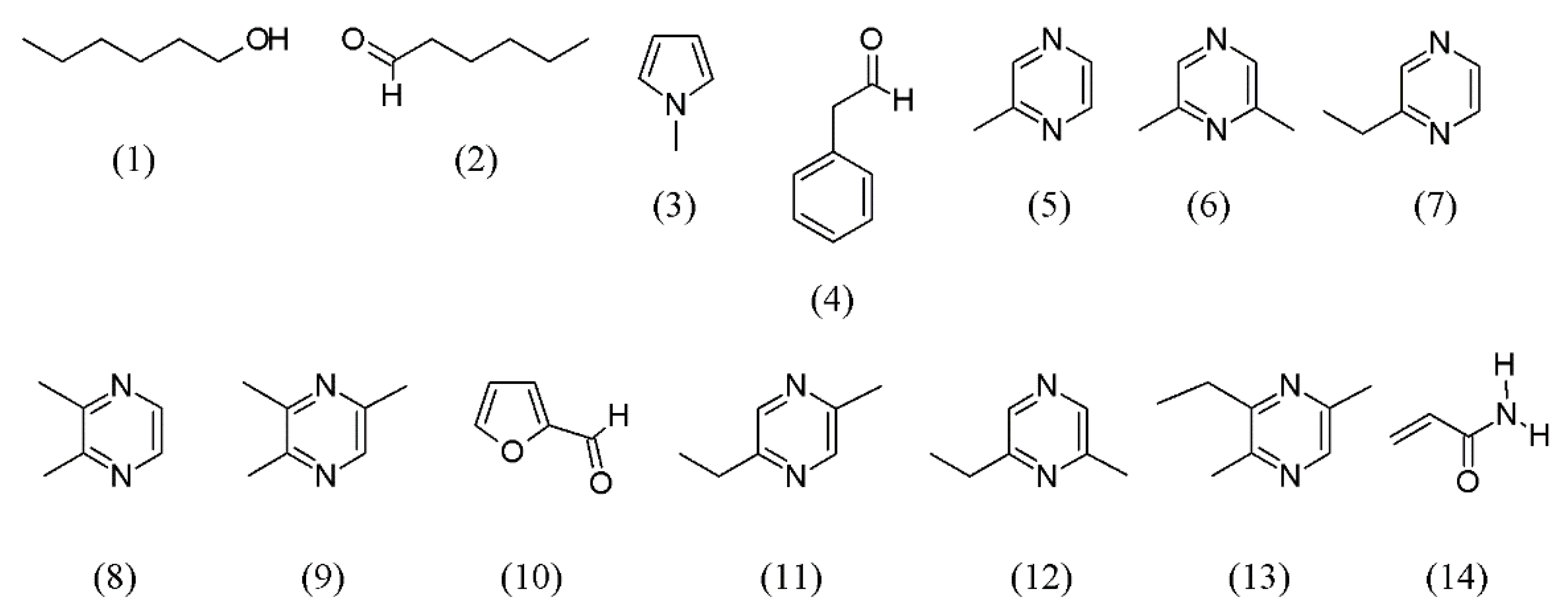
| Arachis hypogaea | roasted peanuts | hexanol (1), hexanal (2), methylpyrrole (3), phenyl acetaldehyde (4), methylpyrazine (5), 2,6-dimethylpyrazine (6), ethylpyrazine (7), 2,3-dimethylpyrazine (8), 2,3,5-trimethylpyrazine (9), 2-furancarboxaldehyde (10), 2-ethyl-5-methyl- (11) and 2-ethyl-6-methylpyrazine (12), and 3-ethyl-2,5-dimethyl-pyrazine (13) | None | [39] |
| Brassica napus | cold-press rapeseed cake | tocopherols and carotenoids | None | [70] |
| Camellia sinensis | tea flowers | phenylethanol (31), linalool (32), (E)-linalool oxide furanoid (33), epoxy linalool (34), geraniol (35), hotrienol (36), acetophenone (37) and germacrene D (38) | None | [62] |
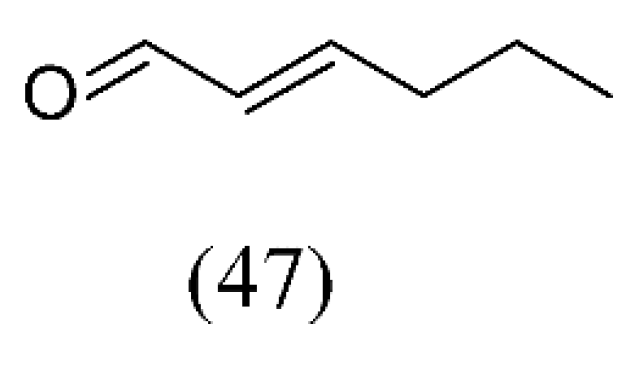
| Camellia sinensis | decaffeinated green teas | hexanal (2), (E)-2-hexenal (47) | None | [69] |
| Citrus sinensis | orange oil | terpene hydrocarbons | None | [58] |
| Coffea arabica | coffee | acrylamide removal | Ethanol | [44] |
| Cuminum cyminum | ground cumin | volatile oil | [47] | |
| Curcuma longa | turmeric roots | curcuminoids | Ethanol | [48] |
| Humulus lupulus | hops | humulones and lupulones | None | [59,60,61] |
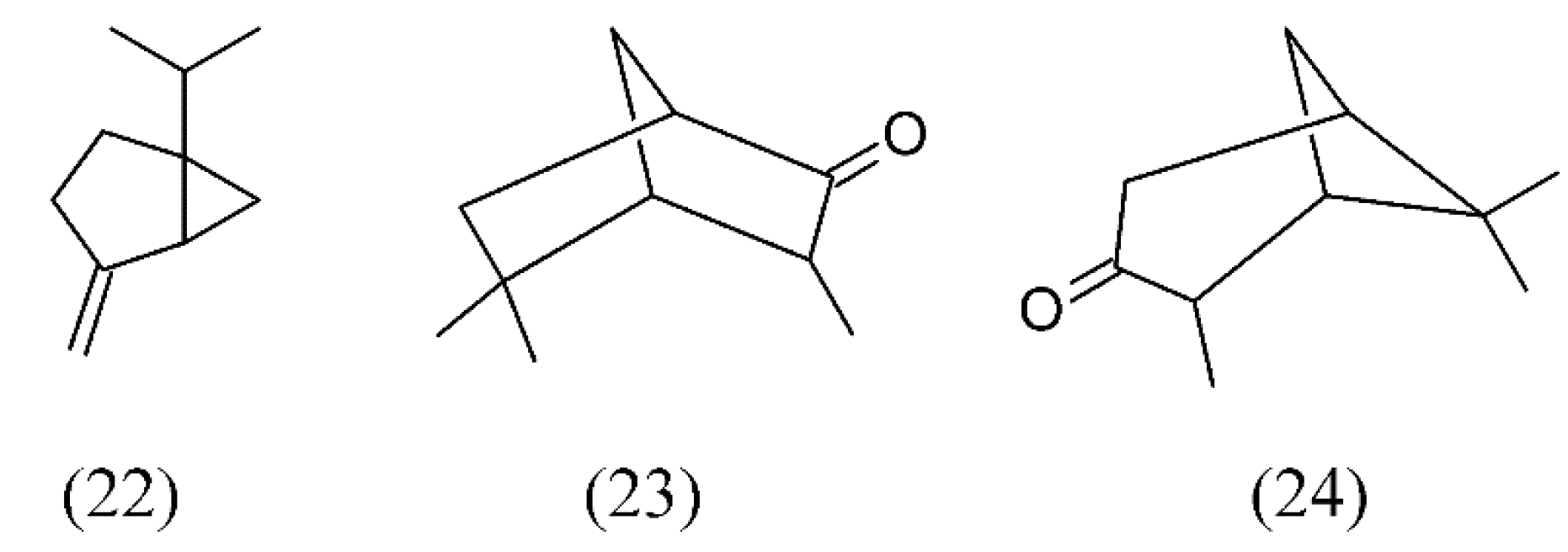
| Hyssopus officinalis | hyssop | sabinene (22), iso-pinocamphone (23) and pinocamphone (24) | Methanol | [53] |
| Mucuna aterrima, M. cinerium, M. deeringiana | linoleic acid (omega-6) | None | [41] | |
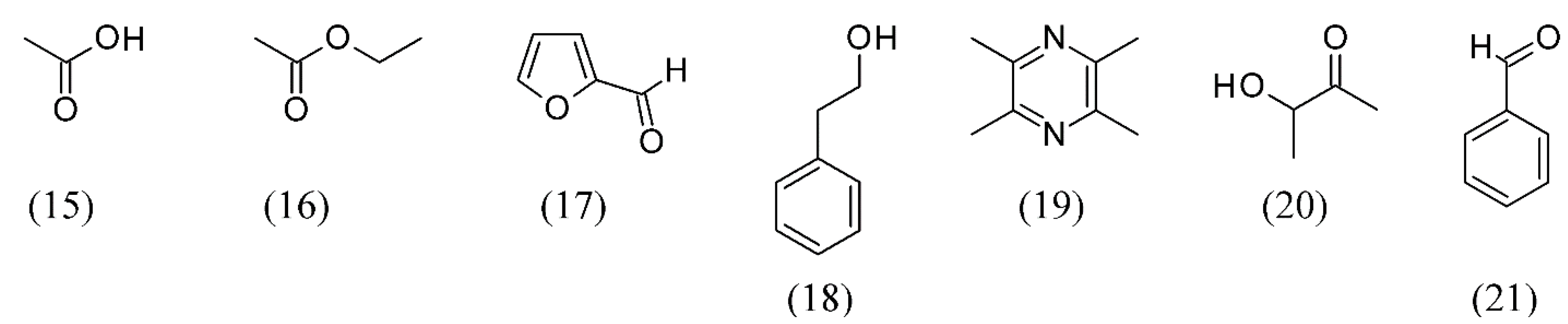
| Oryza sativa | aromatic vinegar from sticky rice | acetic acid (15), ethyl acetate (16), furfural (17), phenethyl alcohol (18), tetramethyl-pyrazine (19), 3-hydroxy-2-butanone (20) and benzaldehyde (21) | [46] | |
| Pandanus amaryllifolius | pandan leaves | 2-acetyl-1-pyrroline (ACPY) (39) and 3-methyl-2(5H)-furanone (40) | None | [63] |
| Piper nigrum | black pepper | essential oil | None | [54,55] |
| Piper piscatorum | pepper | piperovatine (25), followed by palmitic acid (26), pentadecane (27) and pipercallosidine (28) | Ethanol, methanol | [56] |
| Zingiber officinale | ginger | gingerol (41), neral (42), geranial (43), zingiberene (44), α-bisabolene (45) and β-sesquiphellandrene (46) | None | [64,65] |
| Fragrances | ||||
| Abies koreana | needle leaves | elemol (48), terpinen-4-ol (49), sabinene (22), 10(15)-cadinen-4-ol (50), α-terpineol (51), α-pinene (52) and γ-terpinene (53) | None | [73] |
| Bidens tripartita | three-lobe beggarticks | α-pinene (52), p-cymene (63), (E)-β-ocimene (64), β-elemene (65), iso-caryophyllene (66), α-caryophyllene (67), and α-bergamotene (68) | None | [78] |
| Boswellia carterii | frankincense | octyl acetate | None | [73] |
| Cannabis sativa var indica | marihuana | cannabinoids | Ethanol | [80] |
| Croton zehntneri | (E)-anethole (69), α-muurolene (70), methyl chavicol or estragole (71) and germacrene D (38) | None | [81] | |
| Eucalyptus citriodora | leaves | citronellal (54) | None | [75] |
| Eucalyptus globulus | barks | triterpenic acids | Ethanol | [74] |
| Eucalyptus grandis x globulus | inner and outer barks | β-sitosterol (55), methyl morolate (56) | None | [76] |
| Foeniculum vulgare | fennel seeds | (E)-anethole (69), estragole (71), and fenchone (72) | None | [103] |
| Laurus nobilis | bay laurel | (E)-β-ocimene (64), 1,8-cineole (74), α-pinene (52), β-pinene (75), β-longipinene (76), linalyl acetate (77), δ-cadinene (78), α-terpinyl acetate (79) and α-bulnesene (80) | None | [82] |
| Lavandula angustifolia | lavender | 1,8-cineole (74), camphor (81), linalool (32), linalyl acetate (77), fenchone (72), camphor (81) and β-caryophyllene (82) | None | [83,84,85] |
| Lavandula hybrida | lavandin | 1,8-cineole (74), linalool (32), linalyl acetate (77), and camphor (81) | None | [87] |
| Lavandula viridis | lavender | camphor (81), myrtenol (83), verbenone (84) | None | [88] |
| Mentha spicata | spearmint | carvone (85), 1,8-cineole (74), pulegone (86) | None | [89,90] |
| Ocimum basilicum | sweet basil | 1,8-cineole (74), linalool (32), eugenol (87), germacrene D (38), T-cadinol (88) | Water | [91,92] |
| Ocimum gratissimum | clove basil | eugenol (87), β-selinene (89), 1,8-cineole (74), β-caryo-phyllene (82), α-selinene (90) | None | [93] |
| Peumus boldus | boldo | volatile oils | None | [94] |
| Polygala senega, Acorus Tatarinowii | mixture of herbs | methyl eugenol (57), 1,2,3-trimethoxy-5-(2-propenyl)-benzene (58), β-asarone (59), (Z,Z)-9,12-octadecadienoic acid (60), (Z)-6-octadecenoic acid (61), and ethyl oleate (62) | None | [77] |
| Salvia desoleana | sardinian island sage | cuticular waxes and volatile oil | None | [100] |
| Salvia officinalis | sage | mono-, sesqui- and diterpenes | None | [95,96,97,98,99] |
| Thymus vulgaris | thyme leaves | thymol (73) and p-cymene (63) | None | [103] |
| Thymus vulgaris | thyme | p-cymene (63), γ-terpinene (53), linalool (32), thymol (73), carvacrol (91), thymoquinone (92), carvacrol methyl ether (93), camphor (81), cis-linalool oxide (94) | None | [101,102,103] |
4. Biological Effect of Supercritical Fluid Extracts
4.1. Antifungal Activity
4.2. Insecticidal and Acaricidal Activity
4.3. Antibacterial and Cytotoxic Activity
| Bacterial species | Plant species | Reference |
|---|---|---|
| Acinetobacter baumannii | Ramulus cinnamomi | [135] |
| Bacillus cereus | Allium sativum | [140] |
| Azadirachta indica | [134] | |
| Citrus hystrix | [134] | |
| Cordia verbenacea | [138] | |
| Daucus carrota | [133] | |
| Origanum majorana | [120,121,122] | |
| Phyllanthus emblica | [136,137] | |
| Satureja montana | [139] | |
| White grape seeds | [117] | |
| Bacillus subtilis | Azadirachta indica | [134] |
| Citrus hystrix | [134] | |
| Daucus carrota | [133] | |
| Laurus nobilis | [131] | |
| Phyllanthus emblica | [136,137] | |
| Rosmarinus officinalis | [126,129] | |
| Satureja montana | [139] | |
| Chromobacterium violaceum | Anemopsis californica | [130] |
| Citrobacter freundii | White grape seeds | [117] |
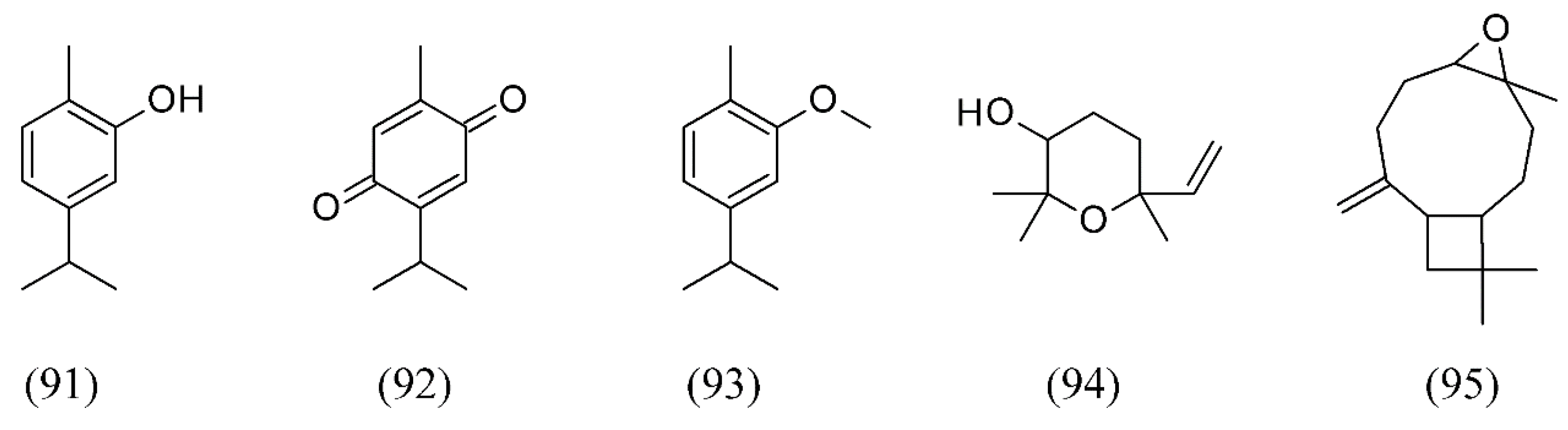
| Corynebacterium pseudotuberculosis | Sideritis scardica | [141] |
| Enterobacter aerogenes | Anemopsis californica | [130] |
| Enterobacter cloacae | Anemopsis californica | [130] |
| White grape seeds | [117] | |
| Enterococcus faecalis | Daucus carrota | [133] |
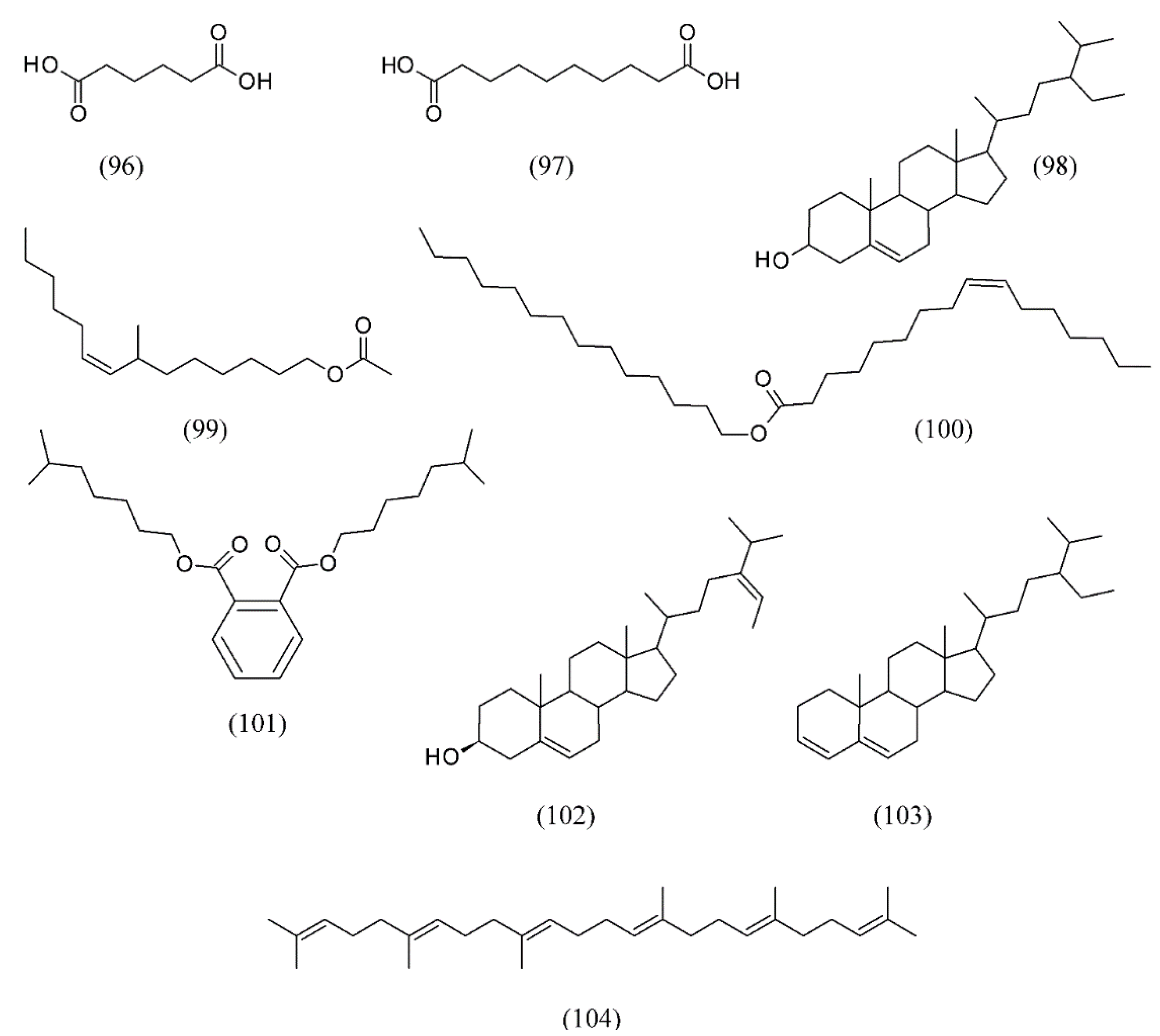
| Satureja montana | [139] | |
| Sideritis scardica | [141] | |
| Escherichia coli | Allium sativum | [140] |
| Cordia verbenacea | [138] | |
| Daucus carrota | [133] | |
| Laurus nobilis | [131] | |
| Origanum majorana | [120,121,122] | |
| Rosmarinus officinalis | [126,129] | |
| Santolina insularis | [118] | |
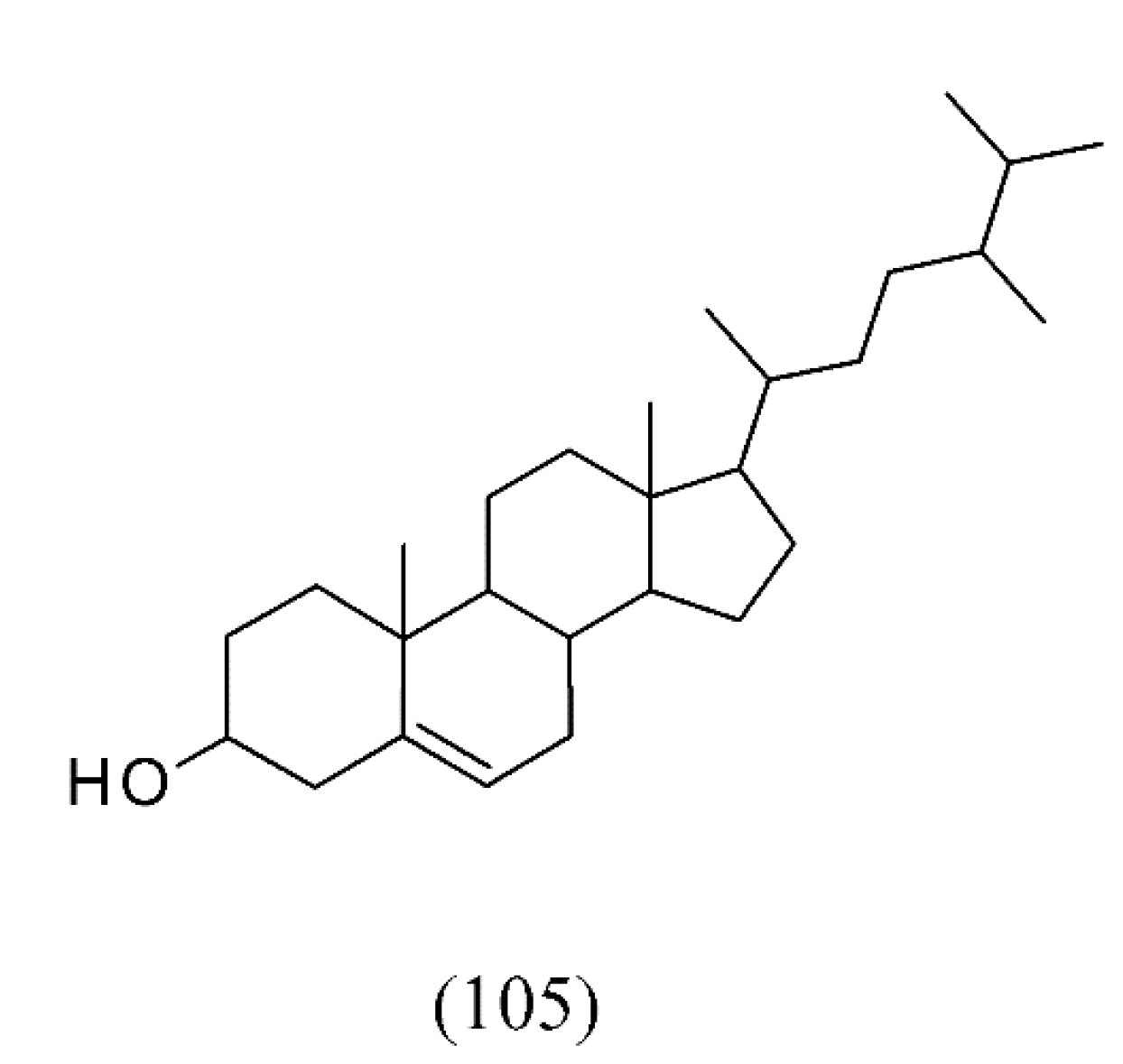
| Satureja montana | [139] | |
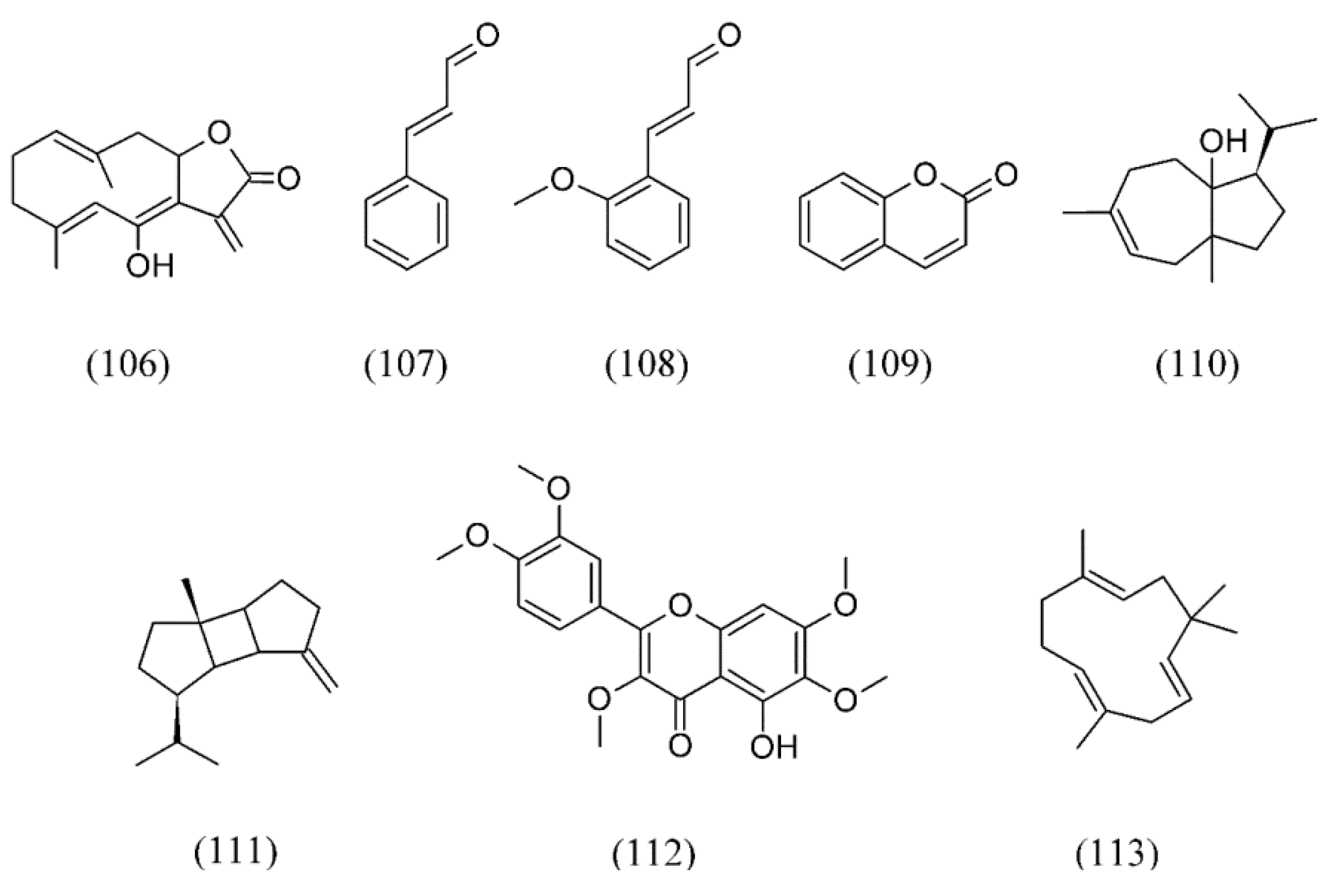
| Sideritis scardica | [141] | |
| White grape seeds | [117] | |
| Haemophilus sp. | Sideritis scardica | [141] |
| Klebsiella pneumoniae | Anemopsis californica | [130] |
| Sideritis scardica | [141] | |
| Listeria monocytogenes | Daucus carrota | [133] |
| Origanum majorana | [120,121,122] | |
| Satureja montana | [139] | |
| Moraxella catarrhalis | Sideritis scardica | [141] |
| Neissera subflava | Anemopsis californica | [130] |
| Pasteurella multocida | Sideritis scardica | [141] |
| Pseudomonas acnes | Azadirachta indica | [134] |
| Citrus hystrix | [134] | |
| Pseudomonas aeruginosa | Cordia verbenacea | [138] |
| Daucus carrota | [133] | |
| Laurus nobilis | [131] | |
| Ramulus cinnamomi | [135] | |
| Rosmarinus officinalis | [126,129] | |
| Satureja montana | [139] | |
| Sideritis scardica | [141] | |
| Pseudomonas aurantiaca | Allium sativum | [140] |
| Pseudomonas fluorescens | Origanum majorana | [120,121,122] |
| Rhodococcus equi | Daucus carrota | [133] |
| Salmonella enteritidis | Daucus carrota | [133] |
| Satureja montana | [139] | |
| Salmonella typhimurium | Anemopsis californica | [130] |
| Origanum majorana | [120,121,122] | |
| Shigella flexneri | Anemopsis californica | [130] |
| Staphylococcus aureus | Anemopsis californica | [130] |
| Azadirachta indica | [134] | |
| Citrus hystrix | [134] | |
| Cordia verbenacea | [138] | |
| Daucus carrota | [133] | |
| Laurus nobilis | [131] | |
| Phyllanthus emblica | [136,137] | |
| Ramulus cinnamomi | [135] | |
| Rosmarinus officinalis | [126,129] | |
| Santolina insularis | [118] | |
| Satureja montana | [139] | |
| Sideritis scardica | [141] | |
| White grape seeds | [117] | |
| Staphylococcus coagulans niger | White grape seeds | [117] |
| Staphylococcus epidermidis | Azadirachta indica | [134] |
| Streptococcus canis | Sideritis scardica | [141] |
| Streptococcus pneumoniae | Anemopsis californica | [130] |
| Streptococcus pyogenes | Sideritis scardica | [141] |
4.4. Antioxidant Activity
5. Process Considerations
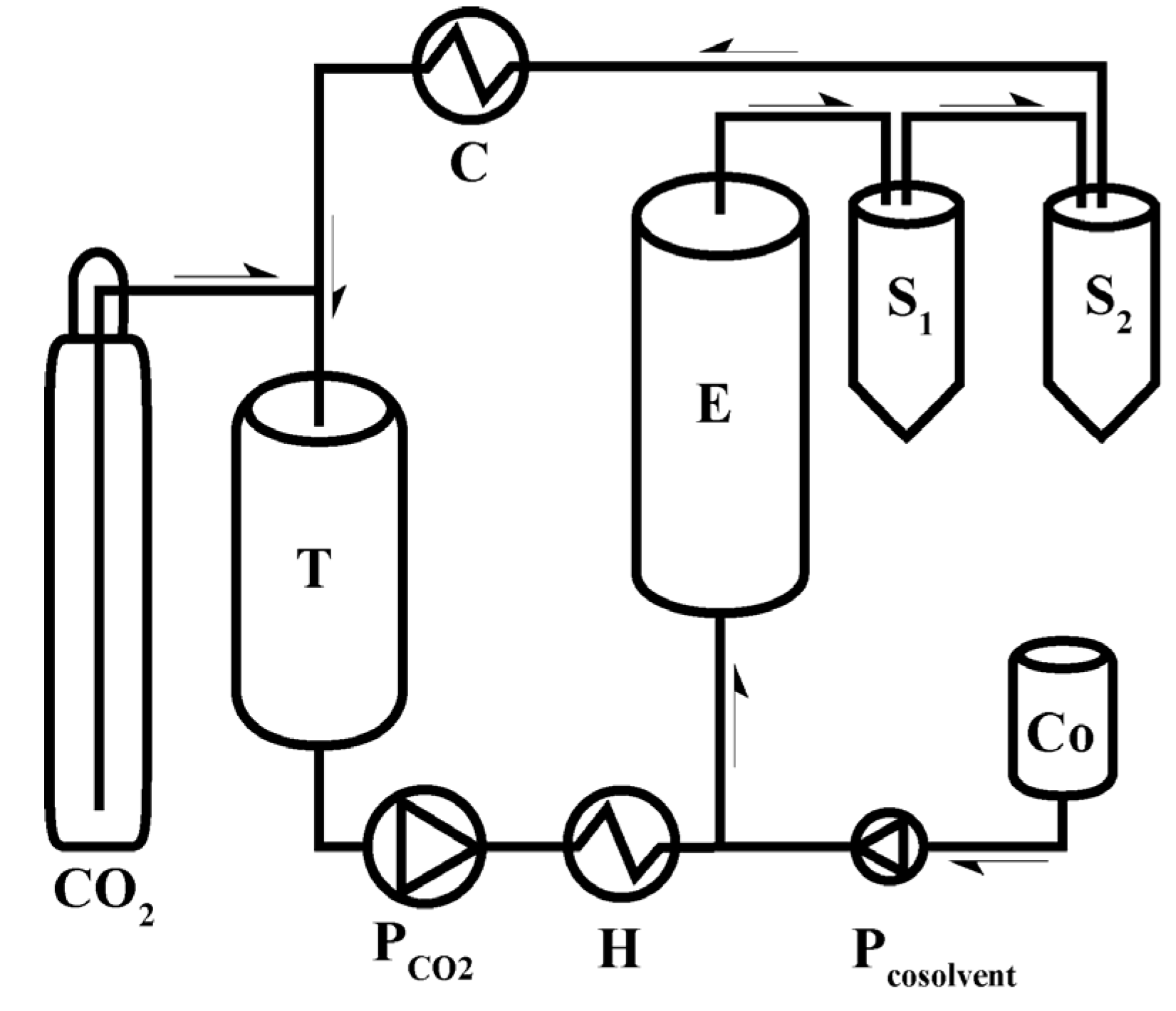
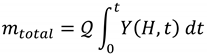
- The solubility and mass transfer of target compounds in the SC CO2. The practical analyses shall verify if SFE is the suitable technique for the extraction of the target compounds [11];
- Evaluation of extract quality. Pressure and temperature can seriously influence the composition of the final extracts;
- Pressure drop effect;
- Process optimization to obtain the best ratio between yield and quantity of solvent amount and time of extraction;
- Scale up optimization
6. Conclusions
Acknowledgments
Conflicts of Interest
References
- Reverchon, E. Supercritical fluid extraction and fractionation of essential oils and related products. J. Supercrit. Fluids 1997, 10, 1–37. [Google Scholar] [CrossRef]
- Herrero, M.; Castro-Puyana, M.; Mendiola, J.A.; Ibanez, E. Compressed fluids for the extraction of bioactive compounds. Trends Anal. Chem. 2013, 43, 67–83. [Google Scholar] [CrossRef]
- Girotra, P.; Singh, S.K.; Nagpal, K. Supercritical fluid technology: a promising approach in pharmaceutical research. Pharm. Dev. Technol. 2013, 18, 22–38. [Google Scholar] [CrossRef]
- Diaz-Maroto, M.C.; Perez-Coello, M.S.; Cabezudo, M.D. Supercritical carbon dioxide extraction of volatiles from spices - Comparison with simultaneous distillation-extraction. J. Chromatogr. A 2002, 947, 23–29. [Google Scholar] [CrossRef]
- Diaz-Reinoso, B.; Moure, A.; Dominguez, H.; Parajo, J.C. Supercritical CO2 extraction and purification of compounds with antioxidant activity. J. Agric. Food Chem. 2006, 54, 2441–2469. [Google Scholar] [CrossRef]
- Herrero, M.; Cifuentes, A.; Ibanez, E. Sub- and supercritical fluid extraction of functional ingredients from different natural sources: Plants, food-by-products, algae and microalgae—A review. Food Chem. 2006, 98, 136–148. [Google Scholar] [CrossRef]
- Reverchon, E.; de Marco, I. Supercritical fluid extraction and fractionation of natural matter. J. Supercrit. Fluids 2006, 38, 146–166. [Google Scholar] [CrossRef]
- Reid, R.C.; Prausnitz, J.M.; Poling, B.E. The Properties of Gases and Liquids; McGraw-Hill: New York, NY, USA, 1987. [Google Scholar]
- Raynie, D.E. Warning concerning the use of nitrous-oxide in supercritical-fluid extractions. Anal. Chem. 1993, 65, 3127–3128. [Google Scholar] [CrossRef]
- Pourmortazavi, S.M.; Hajimirsadeghi, S.S. Supercritical fluid extraction in plant essential and volatile oil analysis. J. Chromatogr. A 2007, 1163, 2–24. [Google Scholar]
- Pereira, C.G.; Meireles, M. Supercritical fluid extraction of bioactive compounds: Fundamentals, applications and economic perspectives. Food Bioproc. Technol. 2010, 3, 340–372. [Google Scholar] [CrossRef]
- Ong, E.S.; Cheong, J.S.H.; Goh, D. Pressurized hot water extraction of bioactive or marker compounds in botanicals and medicinal plant materials. J. Chromatogr. A 2006, 1112, 92–102. [Google Scholar]
- Leal, P.F.; Maia, N.B.; Carmello, Q.A.; Catharino, R.R.; Eberlin, M.N.; Meireles, M. Sweet basil (Ocimum basilicum) extracts obtained by supercritical fluid extraction (SFE): Global yields, chemical composition, antioxidant activity, and estimation of the cost of manufacturing. Food Bioproc. Technol. 2008, 1, 326–338. [Google Scholar]
- Gamiz-Gracia, L.; de Castro, M.D.L. Continuous subcritical water extraction of medicinal plant essential oil: comparison with conventional techniques. Talanta 2000, 51, 1179–1185. [Google Scholar] [CrossRef]
- Bicchi, C.; Rubiolo, P.; Frattini, C.; Sandra, P.; David, F. Off-line supercritical fluid extraction and capillary gas-chromatography of pyrrolizidine alkaloids in senecio species. J. Nat. Prod. 1991, 54, 941–945. [Google Scholar] [CrossRef]
- Mohamed, R.S.; Saldana, M.D.A.; Mazzafera, P.; Zetzl, C.; Brunner, G. Extraction of caffeine, theobromine, and cocoa butter from Brazilian cocoa beans using supercritical CO2 and ethane. Ind. Eng. Chem. Res. 2002, 41, 6751–6758. [Google Scholar] [CrossRef]
- Catchpole, O.J.; Grey, J.B.; Perry, N.B.; Burgess, E.J.; Redmond, W.A.; Porter, N.G. Extraction of chill, black pepper, and ginger with near-critical CO2, propane, and dimethyl ether: Analysis of the extracts by quantitative nuclear magnetic resonance. J. Agric. Food Chem. 2003, 51, 4853–4860. [Google Scholar] [CrossRef]
- Illes, V.; Daood, H.G.; Perneczki, S.; Szokonya, L.; Then, M. Extraction of coriander seed oil by CO2 and propane at super- and subcritical conditions. J. Supercrit. Fluids 2000, 17, 177–186. [Google Scholar]
- Hamdan, S.; Daood, H.G.; Toth-Markus, M.; Illes, V. Extraction of cardamom oil by supercritical carbon dioxide and sub-critical propane. J. Supercrit. Fluids 2008, 44, 25–30. [Google Scholar] [CrossRef]
- Simandi, B.; Deak, A.; Ronyai, E.; Gao, Y.X.; Veress, T.; Lemberkovics, E.; Then, M. Supercritical carbon dioxide extraction and fractionation of fennel oil. J. Agric. Food Chem. 1999, 47, 1635–1640. [Google Scholar] [CrossRef]
- Modey, W.K.; Mulholland, D.A.; Raynor, M.W. Analytical supercritical fluid extraction of natural products. Phytochem. Anal. 1996, 7, 1–15. [Google Scholar] [CrossRef]
- Sovilj, M.N.; Nikolovski, B.G.; Spasojevic, M.D. Critical review of supercritical fluid extraction of selected spice plant materials. Maced. J. Chem. Chem. Eng. 2011, 30, 197–220. [Google Scholar]
- Marongiu, B.; Piras, A.; Pani, F.; Porcedda, S.; Ballero, M. Extraction, separation and isolation of essential oils from natural matrices by supercritical CO2. Flav. Fragr. J. 2003, 18, 505–509. [Google Scholar] [CrossRef]
- Marriott, R.J. Greener chemistry preparation of traditional flavour extracts and molecules. Agro Food Ind. Hi-Tech 2010, 21, 46–48. [Google Scholar]
- Simandi, B.; Kery, A.; Lemberkovics, E.; Oszagyan, M.; Ronyai, E.; Mathe, I.; Fekete, J.; Hethelyi, E. Supercritical fluid extraction of medicinal plants. High Press. Chem. Eng. 1996, 12, 357–362. [Google Scholar]
- Fornari, T.; Vicente, G.; Vazquez, E.; Garcia-Risco, M.R.; Reglero, G. Isolation of essential oil from different plants and herbs by supercritical fluid extraction. J. Chromatogr. A 2012, 1250, 34–48. [Google Scholar]
- Jarvis, A.P.; Morgan, D. Isolation of plant products by supercritical-fluid extraction. Phytochem. Anal. 1997, 8, 217–222. [Google Scholar] [CrossRef]
- Sovova, H. Steps of supercritical fluid extraction of natural products and their characteristic times. J. Supercrit. Fluids 2012, 66, 73–79. [Google Scholar] [CrossRef]
- Sovova, H. Modeling the supercritical fluid extraction of essential oils from plant materials. J. Chromatogr. A 2012, 1250, 27–33. [Google Scholar]
- Coelho, J.P.; Cristino, A.F.; Matos, P.G.; Rauter, A.P.; Nobre, B.P.; Mendes, R.L.; Barroso, J.G.; Mainar, A.; Urieta, J.S.; Fareleira, J.M.; Sovova, H.; Palavra, A.F. Extraction of Volatile Oil from Aromatic Plants with Supercritical Carbon Dioxide: Experiments and Modeling. Molecules 2012, 17, 10550–10573. [Google Scholar] [CrossRef]
- Sovova, H.; Stateva, R.P. Supercritical fluid extraction from vegetable materials. Rev. Chem. Eng. 2011, 27, 79–156. [Google Scholar]
- Hawthorne, S.B.; Krieger, M.S.; Miller, D.J. Analysis of flavor and fragrance compounds using supercritical fluid extraction coupled with gas-chromatography. Anal. Chem. 1988, 60, 472–477. [Google Scholar] [CrossRef]
- Fornari, T.; Ruiz-Rodriguez, A.; Vicente, G.; Vazquez, E.; Garcia-Risco, M.R.; Reglero, G. Kinetic study of the supercritical CO2 extraction of different plants from Lamiaceae family. J. Supercrit. Fluids 2012, 64, 1–8. [Google Scholar] [CrossRef]
- del Valle, J.M.; De La Fuente, J.C. Supercritical CO2 extraction of oilseeds: Review of kinetic and equilibrium models. Crit. Rev. Food Sci. Nutr. 2006, 46, 131–160. [Google Scholar] [CrossRef]
- Oliveira, E.L.; Silvestre, A.J.; Silva, C.M. Review of kinetic models for supercritical fluid extraction. Chem. Eng. Res. Design 2011, 89, 1104–1117. [Google Scholar] [CrossRef]
- Khosravi-Darani, K. Research activities on supercritical fluid science in food biotechnology. Crit. Rev. Food Sci. Nutr. 2010, 50, 479–488. [Google Scholar] [CrossRef]
- Malaman, F.S.; Moraes, L.A.; West, C.; Ferreira, N.J.; Oliveira, A.L. Supercritical fluid extracts from the Brazilian cherry (Eugenia uniflora L.): Relationship between the extracted compounds and the characteristic flavour intensity of the fruit. Food Chem. 2011, 124, 85–92. [Google Scholar] [CrossRef]
- Krukonis, V.J. Supercritical fluid extraction in flavor applications. In Characterization and Measurement of Flavor Compounds; American Chemical Society: Washington, DC, USA, 1985; pp. 154–175. [Google Scholar]
- Leunissen, M.; Davidson, V.J.; Kakuda, Y. Analysis of volatile flavor components in roasted peanuts using supercritical fluid extraction and gas chromatography mass spectrometry. J. Agric. Food Chem. 1996, 44, 2694–2699. [Google Scholar] [CrossRef]
- Lasekan, O.; Abbas, K. Analysis of volatile flavour compounds and acrylamide in roasted Malaysian tropical almond (Terminalia catappa) nuts using supercritical fluid extraction. Food Chem. Toxicol. 2010, 48, 2212–2216. [Google Scholar] [CrossRef]
- Garcia, V.A.d.S.; Cabral, V.F.; Zanoelo, É.F.; da Silva, C.; Filho, L.C. Extraction of Mucuna seed oil using supercritical carbon dioxide to increase the concentration of l-Dopa in the defatted meal. J. Supercrit. Fluids 2012, 69, 75–81. [Google Scholar] [CrossRef]
- Zhou, M.X.; Robards, K.; Glennie-Holmes, M.; Helliwell, S. Analysis of volatile compounds and their contribution to flavor in cereals. J. Agric. Food Chem. 1999, 47, 3941–3953. [Google Scholar] [CrossRef]
- Sarrazin, C.; Le Quere, J.L.; Gretsch, C.; Liardon, R. Representativeness of coffee aroma extracts: A comparison of different extraction methods. Food Chem. 2000, 70, 99–106. [Google Scholar] [CrossRef]
- Banchero, M.; Pellegrino, G.; Manna, L. Supercritical fluid extraction as a potential mitigation strategy for the reduction of acrylamide level in coffee. J. Food Eng. 2013, 115, 292–297. [Google Scholar] [CrossRef]
- Yee, J.L.; Khalil, H.; Jimenez-Flores, R. Flavor partition and fat reduction in cheese by supercritical fluid extraction: processing variables. Lait 2007, 87, 269–285. [Google Scholar] [CrossRef]
- Lu, Z.M.; Xu, W.; Yu, N.H.; Zhou, T.; Li, G.Q.; Shi, J.S.; Xu, Z.H. Recovery of aroma compounds from Zhenjiang aromatic vinegar by supercritical fluid extraction. Int. J. Food Sci. Technol. 2011, 46, 1508–1514. [Google Scholar] [CrossRef]
- Heikes, D.L.; Scott, B.; Gorzovalitis, N.A. Quantitation of volatile oils in ground cumin by supercritical fluid extraction and gas chromatography with flame ionization detection. J. AOAC Int. 2001, 84, 1130–1134. [Google Scholar]
- Braga, M.E.; Angela, M.; Meireles, A. Accelerated solvent extraction and fractioned extraction to obtain the Curcuma longa volatile oil and oleoresin. J. Food Proc. Eng. 2007, 30, 501–521. [Google Scholar] [CrossRef]
- Simandi, B.; Sass-Kiss, A.; Czukor, B.; Deak, A.; Prechl, A.; Csordas, A.; Sawinsky, J. Pilot-scale extraction and fractional separation of onion oleoresin using supercritical carbon dioxide. J. Food Eng. 2000, 46, 183–188. [Google Scholar] [CrossRef]
- Dron, A.; Guyer, D.E.; Gage, D.A.; Lira, C.T. Yield and quality of onion flavor oil obtained by supercritical fluid extraction and other methods. J. Food Proc. Eng. 1997, 20, 107–124. [Google Scholar] [CrossRef]
- Calvey, E.M.; Matusik, J.E.; White, K.D.; DeOrazio, R.; Sha, D.Y.; Block, E. Allium chemistry: Supercritical fluid extraction and LC-APCI-MS of thiosulfinates and related compounds from homogenates of garlic, onion, and ramp. Identification in garlic and ramp and synthesis of 1-propanesulfinothioic acid S-allyl ester. J. Agric. Food Chem. 1997, 45, 4406–4413. [Google Scholar] [CrossRef]
- Calvey, E.M.; Block, E. Supercritical fluid extraction of Allium species. Spices Flav. Chem. Antiox. Prop. 1997, 660, 113–124. [Google Scholar] [CrossRef]
- Kazazi, H.; Rezaei, K.; Ghotb-Sharif, S.J.; Emam-Djomeh, Z.; Yamini, Y. Supercriticial fluid extraction of flavors and fragrances from Hyssopus officinalis L. cultivated in Iran. Food Chem. 2007, 105, 805–811. [Google Scholar] [CrossRef]
- Perakis, C.; Louli, V.; Magoulas, K. Supercritical fluid extraction of black pepper oil. J. Food Eng. 2005, 71, 386–393. [Google Scholar] [CrossRef]
- Kumoro, A.C.; Hasan, M.; Singh, H. Extraction of sarawak black pepper essential oil using supercritical carbon dioxide. Arab. J. Sci. Eng. 2010, 35, 7–16. [Google Scholar]
- Pimentel, F.A.; Cardoso, M.d.; Guimaraes, L.G.; Queiroz, F.; Barbosa, L.C.; Morais, A.R.; Nelson, D.L.; Andrade, M.A.; Zacaroni, L.M.; Pimentel, S.M. Extracts from the leaves of Piper piscatorum (Trel. Yunc.) obtained by supercritical extraction of with CO2, employing ethanol and methanol as co-solvents. Ind. Crops Prod. 2013, 43, 490–495. [Google Scholar] [CrossRef]
- Anklam, E.; Muller, A. Supercritical-fluid extraction of vanillin and ethyl vanillin from European commercially available vanilla sugars. Deut. Lebens. Rund. 1993, 89, 344–346. [Google Scholar]
- Shen, Z.P.; Mishra, V.; Imison, B.; Palmer, M.; Fairclough, R. Use of adsorbent and supercritical carbon dioxide to concentrate flavor compounds from orange oil. J. Agric. Food Chem. 2002, 50, 154–160. [Google Scholar] [CrossRef]
- Verschuere, M.; Sandra, P.; David, F. Fractionation by SFE and microcolumn analysis of the essential oil and the bitter principles of Hops. J. Chromatogr. Sci. 1992, 30, 388–391. [Google Scholar] [CrossRef]
- Van Opstaele, F.; Goiris, K.; De Rouck, G.; Aerts, G.; De Cooman, L. Production of novel varietal hop aromas by supercritical fluid extraction of hop pellets-Part 1: Preparation of single variety total hop essential oils and polar hop essences. J. Supercrit. Fluids 2012, 69, 45–56. [Google Scholar] [CrossRef]
- Van Opstaele, F.; Goiris, K.; De Rouck, G.; Aerts, G.; de Cooman, L. Production of novel varietal hop aromas by supercritical fluid extraction of hop pellets-Part 2: Preparation of single variety floral, citrus, and spicy hop oil essences by density programmed supercritical fluid extraction. J. Supercrit. Fluids 2012, 71, 147–161. [Google Scholar] [CrossRef]
- Joshi, R.; Poonam; Saini, R.; Guleria, S.; Babu, G.D.; Kumari, M.; Gulati, A. Characterization of volatile components of tea flowers (Camellia sinensis) growing in Kangra by GC/MS. Nat. Prod. Comm. 2011, 6, 1155–1158. [Google Scholar]
- Laohakunjit, N.; Noomhorm, A. Supercritical carbon dioxide extraction of 2-acetyl-1-pyrroline and volatile components from panclan leaves. Flav. Fragr. J. 2004, 19, 251–259. [Google Scholar] [CrossRef]
- Bartley, J.P.; Jacobs, A.L. Effects of drying on flavour compounds in Australian-grown ginger (Zingiber officinale). J. Sci. Food Agric. 2000, 80, 209–215. [Google Scholar] [CrossRef]
- Bartley, J.P.; Foley, P. Supercritical-fluid extraction of Australian-grown ginger (Zingiber officinale). J. Sci. Food Agric. 1994, 66, 365–371. [Google Scholar] [CrossRef]
- Xu, Z.G.; Zheng, L. Comparison of volatile and semivolatile compounds from commercial cigarette by supercritical fluid extraction and simultaneous distillation extraction. J. Zhejiang Univ. Sci. 2004, 5, 1528–1532. [Google Scholar] [CrossRef]
- Senorans, F.J.; Ruiz-Rodriguez, A.; Ibanez, E.; Tabera, J.; Reglero, G. Countercurrent supercritical fluid extraction and fractionation of alcoholic beverages. J.Agric.Food Chem. 2001, 49, 1895–1899. [Google Scholar] [CrossRef]
- Ruiz-Rodriguez, A.; Fornari, T.; Jaime, L.; Vazquez, E.; Amador, B.; ntonio Nieto, J.; Yuste, M.; Mercader, M.; Reglero, G. Supercritical CO2 extraction applied toward the production of a functional beverage from wine. J. Supercrit. Fluids 2012, 61, 92–100. [Google Scholar] [CrossRef]
- Lee, S.; Park, M.; Kim, K.; Kim, Y. Effect of supercritical carbon dioxide decaffeination on volatile components of green teas. J. Food Sci. 2007, 72, S497–S502. [Google Scholar] [CrossRef]
- Uquiche, E.; Romero, V.; Ortiz, J.; del Valle, J. Extraction of oil and minor lipids from cold-press rapeseed cake with supercritical CO2. Braz. J. Chem. Eng. 2012, 29, 585–597. [Google Scholar] [CrossRef]
- Lamsen, M.R.L.; Zhong, Q. Impacts of supercritical extraction on GC/MS profiles of volatiles in whey protein isolate sampled by solid-phase microextraction. J. Food Proc. Preserv. 2011, 35, 869–883. [Google Scholar] [CrossRef]
- Cabaleiro, N.; de la Calle, I.; Bendicho, C.; Lavilla, I. Current trends in liquid-liquid and solid-liquid extraction for cosmetic analysis: a review. Anal. Meth. 2013, 5, 323–340. [Google Scholar] [CrossRef]
- Kim, K.; Bu, Y.; Jeong, S.; Lim, J.; Kwon, Y.; Cha, D.S.; Kim, J.; Jeon, S.; Eun, J.; Jeon, H. Memory-enhancing effect of a supercritical carbon dioxide fluid extract of the needles of Abies koreana on scopolamine-induced amnesia in mice. Biosci. Biotechnol. Biochem. 2006, 70, 1821–1826. [Google Scholar] [CrossRef]
- Domingues, R.M.; de Melo, M.M.; Oliveira, E.L.; Neto, C.P.; Silvestre, A.J.; Silva, C.M. Optimization of the supercritical fluid extraction of triterpenic acids from Eucalyptus globulus bark using experimental design. J. Supercrit. Fluids 2013, 74, 105–114. [Google Scholar] [CrossRef]
- Mann, T.S.; Babu, G.D.K.; Guleria, S.; Singh, B. Variation in the volatile oil composition of Eucalyptus citriodora produced by hydrodistillation and supercritical fluid extraction techniques. Nat. Prod. Res. 2013, 27, 675–679. [Google Scholar] [CrossRef]
- Patinha, D.; Domingues, R.; Villaverde, J.; Silva, A.; Silva, C.; Freire, C.; Pascoal Neto, C.; Silvestre, A. Lipophilic extractives from the bark of Eucalyptus grandis x globulus, a rich source of methyl morolate: Selective extraction with supercritical CO2. Ind. Crops Prod. 2013, 43, 340–348. [Google Scholar] [CrossRef]
- Wang, Y.; Chang, L.; Zhao, X.; Meng, X.; Liu, Y. Gas chromatography-mass spectrometry analysis on compounds in volatile oils extracted from Yuan Zhi (Radix Polygalae) and Shi Chang Pu (Acorus Tatarinowii) by supercritical CO2. J. Trad.Chin. Med. 2012, 32, 459–464. [Google Scholar] [CrossRef]
- Kaskoniene, V.; Kaskonas, P.; Maruska, A.; Ragazinskiene, O. Essential oils of Bidens tripartita L. collected during period of 3 years composition variation analysis. Acta Physiol. Plant. 2013, 35, 1171–1178. [Google Scholar] [CrossRef]
- Zhou, J.; Ma, X.M.; Qiu, B.H.; Chen, J.X.; Bian, L.; Pan, L.M. Parameters optimization of supercritical fluid-CO2 extracts of frankincense using response surface methodology and its pharmacodynamics effects. J. Sep. Sci. 2013, 36, 383–390. [Google Scholar] [CrossRef]
- Omar, J.; Olivares, M.; Alzaga, M.; Etxebarria, N. Optimisation and characterisation of marihuana extracts obtained by supercritical fluid extraction and focused ultrasound extraction and retention time locking GC-MS. J. Sep. Sci. 2013, 36, 1397–1404. [Google Scholar] [CrossRef]
- Sousa, E.M.B.D.; Martinez, J.; Chiavone, O.; Rosa, P.T.V.; Domingos, T.; Meireles, M.A.A. Extraction of volatile oil from Croton zehntneri Pax et Hoff with pressurized CO2: Solubility, composition and kinetics. J. Food Eng. 2005, 69, 325–333. [Google Scholar] [CrossRef]
- Marzouki, H.; Piras, A.; Marongiu, B.; Rosa, A.; Dessi, M. Extraction and separation of volatile and fixed oils from berries of Laurus nobilis L. by supercritical CO(2). Molecules 2008, 13, 1702–1711. [Google Scholar] [CrossRef]
- Luu, T.D.; Ngo Duy, A.T.; Le Thi, N.H.; Zhao, J.; Mammucari, R.; Foster, N. Antioxidant activity, yield and chemical composition of lavender essential oil extracted by supercritical CO2. J. Supercrit. Fluids 2012, 70, 27–34. [Google Scholar] [CrossRef]
- Da Porto, C.; Decorti, D.; Kikic, I. Flavour compounds of Lavandula angustifolia L. to use in food manufacturing: Comparison of three different extraction methods. Food Chem. 2009, 112, 1072–1078. [Google Scholar] [CrossRef]
- Ghoreishi, S.M.; Kamali, H.; Ghaziaskar, H.S.; Dadkhah, A.A. Optimization of supercritical extraction of linalyl acetate from lavender via Box-Behnken design. Chem. Eng. Technol. 2012, 35, 1641–1648. [Google Scholar] [CrossRef]
- Chemat, F.; Lucchesi, M.E.; Smadja, J.; Favretto, L.; Colnaghi, G.; Visinoni, F. Microwave accelerated steam distillation of essential oil from lavender: A rapid, clean and environmentally friendly approach. Anal. Chim. Acta 2006, 555, 157–160. [Google Scholar] [CrossRef]
- Kamali, H.; Jalilvand, M.R.; Aminimoghadamfarouj, N. Pressurized fluid extraction of essential oil from Lavandula hybrida using a modified supercritical fluid extractor and a central composite design for optimization. J. Sep. Sci. 2012, 35, 1479–1485. [Google Scholar] [CrossRef]
- Costa, P.; Grosso, C.; Goncalves, S.; Andrade, P.B.; Valentao, P.; Gabriela Bernardo-Gil, M.; Romano, A. Supercritical fluid extraction and hydrodistillation for the recovery of bioactive compounds from Lavandula viridis L'Her. Food Chem. 2012, 135, 112–121. [Google Scholar] [CrossRef]
- Almeida, P.P.; Mezzomo, N.; Ferreira, S.R. Extraction of Mentha spicata L. volatile compounds: Evaluation of process parameters and extract composition. Food Bioproc. Technol. 2012, 5, 548–559. [Google Scholar] [CrossRef]
- Ansari, K.; Goodarznia, I. Optimization of supercritical carbon dioxide extraction of essential oil from spearmint (Mentha spicata L.) leaves by using Taguchi methodology. J. Supercrit. Fluids 2012, 67, 123–130. [Google Scholar] [CrossRef]
- Occhipinti, A.; Capuzzo, A.; Bossi, S.; Milanesi, C.; Maffei, M.E. Comparative analysis of supercritical CO2 extracts and essential oils from an Ocimum basilicum chemotype particularly rich in T-cadinol. J. Essent. Oil Res. 2013. [Google Scholar] [CrossRef]
- Gainar, L.; Vilcu, R.; Mocan, M. Supercritical fluid extraction of basil essential oil. Influence of the process parameters and chemical characterization of extracts. Rev. Roum. Chim. 2002, 47, 29–32. [Google Scholar]
- Leal, P.F.; Chaves, F.; Ming, L.C.; Petenate, A.J.; Meireles, M. Global yields, chemical compositions and antioxidant activities of clove basil (Ocimum gratissimum L.) extracts obtained by supercritical fluid extraction. J. Food Proc. Eng. 2006, 29, 547–559. [Google Scholar] [CrossRef]
- Uquiche, E.; Huerta, E.; Sandoval, A.; Manuel del Valle, J. Effect of boldo (Peumus boldus M.) pretreatment on kinetics of supercritical CO2 extraction of essential oil. J. Food Sci. 2012, 109, 230–237. [Google Scholar]
- Glisic, S.; Ivanovic, J.; Ristic, M.; Skala, D. Extraction of sage (Salvia officinalis L.) by supercritical CO2: Kinetic data, chemical composition and selectivity of diterpenes. J. Supercrit. Fluids 2010, 52, 62–70. [Google Scholar] [CrossRef]
- Dauksas, E.; Venskutonis, P.R.; Povilaityte, V.; Sivik, B. Rapid screening of antioxidant activity of sage (Salvia officinalis L.) extracts obtained by supercritical carbon dioxide at different extraction conditions. Nahrung Food 2001, 45, 338–341. [Google Scholar] [CrossRef]
- Glisic, S.B.; Ristic, M.; Skala, D.U. The combined extraction of sage (Salvia officinalis L.): Ultrasound followed by supercritical CO2 extraction. Ultrason. Sonochem. 2011, 18, 318–326. [Google Scholar] [CrossRef]
- Reverchon, E.; Taddeo, R.; DellaPorta, G. Extraction of sage oil by supercritical CO2: Influence of some process parameters. J. Supercrit. Fluids 1995, 8, 302–309. [Google Scholar] [CrossRef]
- Aleksovski, S.; Sovova, H. Supercritical CO2 extraction of Salvia officinalis L. J. Supercrit. Fluids 2007, 40, 239–245. [Google Scholar] [CrossRef]
- Marongiu, B.; Porcedda, S.; la Porta, G.; Reverchon, E. Extraction and isolation of Salvia desoleana and Mentha spicata subsp insularis essential oils by supercritical CO2. Flav. Fragr. J. 2001, 16, 384–388. [Google Scholar] [CrossRef]
- Grosso, C.; Figueiredo, A.C.; Burillo, J.; Mainar, A.M.; Urieta, J.S.; Barroso, J.G.; Coelho, J.A.; Palavra, A.M. Composition and antioxidant activity of Thymus vulgaris volatiles: Comparison between supercritical fluid extraction and hydrodistillation. J. Sep. Sci. 2010, 33, 2211–2218. [Google Scholar] [CrossRef]
- Costa, P.; Goncalves, S.; Grosso, C.; Andrade, P.B.; Valentao, P.; Gabriela Bernardo-Gil, M.; Romano, A. Chemical profiling and biological screening of Thymus lotocephalus extracts obtained by supercritical fluid extraction and hydrodistillation. Ind. Crops Prod. 2012, 36, 246–256. [Google Scholar] [CrossRef]
- Diaz-Maroto, M.C.; Diaz-Maroto Hidalgo, I.J.; Sanchez-Palomo, E.; Perez-Coello, M.S. Volatile components and key odorants of fennel (Foeniculum vulgare Mill.) and thyme (Thymus vulgaris L.) oil extracts obtained by simultaneous distillation-extraction and supercritical fluid extraction. J. Agric. Food Chem. 2005, 53, 5385–5389. [Google Scholar] [CrossRef]
- Hanif, M.A.; Bhatti, H.N.; Jamil, M.S.; Anjum, R.S.; Jamil, A.; Khan, M.M. Antibacterial and Antifungal Activities of Essential Oils Extracted from Medicinal Plants Using CO2 Supercritical Fluid Extraction Technology. Asian J. Chem. 2010, 22, 7787–7798. [Google Scholar]
- Marongiu, B.; Piras, A.; Porcedda, S.; Falconieri, D.; Frau, M.; Maxia, A.; Goncalves, M.; Cavaleiro, C.; Salgueiro, L. Antifungal activity and chemical composition of essential oils from Smyrnium olusatrum L. (Apiaceae) from Italy and Portugal. Nat. Prod. Res. 2012, 26, 993–1003. [Google Scholar] [CrossRef]
- Li, D.; Wang, Z.; Zhang, Y. Antifungal activity of extracts by supercritical carbon dioxide extraction from roots of Echinacea angustifolia and analysis of their constituents using gas chromatography-mass spectrometry (GC-MS). J. Med. Plants Res. 2011, 5, 5605–5610. [Google Scholar]
- Du, T.; Shupe, T.F.; Hse, C.Y. Antifungal activities of three supercritical fluid extracted cedar oils. Holzforschung 2011, 65, 277–284. [Google Scholar]
- Wenqiang, G.; Shufen, L.; Ruixiang, Y.; Yanfeng, H. Comparison of composition and antifungal activity of Artemisia argyi Levl. et Vant inflorescence essential oil extracted by hydrodistillation and supercritical carbon dioxide. Nat. Prod. Res. 2006, 20, 992–998. [Google Scholar]
- Bai, X.N.; Cheng, J.; Liang, W.; Ma, L.Q.; Liu, Y.B.; Shi, G.L.; Wang, Y.N. Antifungal activity of extracts by supercritical carbon dioxide extraction from roots of Stellera chamaejasme L. and analysis of their constituents using GC-MS. Inf. Technol. Agric. Eng. 2012, 134, 653–662. [Google Scholar] [CrossRef]
- Pavela, R.; Sajfrtova, M.; Sovova, H.; Barnet, M.; Karban, J. The insecticidal activity of Tanacetum parthenium (L.) Schultz Bip. extracts obtained by supercritical fluid extraction and hydrodistillation. Ind. Crop. Prod. 2010, 31, 449–454. [Google Scholar] [CrossRef]
- Wu, H.; Zhang, G.A.; Zeng, S.; Lin, K.c. Extraction of allyl isothiocyanate from horseradish (Armoracia rusticana) and its fumigant insecticidal activity on four stored-product pests of paddy. Pest Manag. Sci. 2009, 65, 1003–1008. [Google Scholar] [CrossRef]
- Pavela, R.; Sajfrtova, M.; Sovova, H.; Karban, J.; Barnet, M. The Effects of Extracts Obtained by Supercritical Fluid Extraction and Traditional Extraction Techniques on Larvae Leptinotarsa decemlineata SAY. J. Essent. Oil Ress. 2009, 21, 367–373. [Google Scholar] [CrossRef]
- Pavela, R.; Sajfrtova, M.; Sovova, H.; Barnet, M. The insecticidal activity of Satureja hortensis L. extracts obtained by supercritical fluid extraction and traditional extraction techniques. Appl. Entomol. Zool. 2008, 43, 377–382. [Google Scholar] [CrossRef]
- Martin, L.; Julio, L.F.; Burillo, J.; Sanz, J.; Mainar, A.M.; Gonzalez-Coloma, A. Comparative chemistry and insect antifeedant action of traditional (Clevenger and Soxhlet) and supercritical extracts (CO2) of two cultivated wormwood (Artemisia absinthium L.) populations. Ind. Crops Prod. 2011, 34, 1615–1621. [Google Scholar] [CrossRef]
- Liang, W.; Cheng, J.; Ma, L.Q.; Liu, Y.B.; Shi, G.L.; Wang, Y.N. Componential analysis and acaricidal activities of Stellera chamaejasme extracts by supercritical fluid extraction. Inf. Technol. Agric. Eng. 2012, 134, 643–652. [Google Scholar] [CrossRef]
- Misic, D.; Asanin, R.; Ivanovic, J.; Zizovic, I. Investigation of Antibacterial Activity of Supercritical Extracts of Plants, As Well As of Extracts Obtained by Other Technological Processes on Some Bacteria Isolated from Animals. Acta Veter. Beog. 2009, 59, 557–568. [Google Scholar] [CrossRef]
- Palma, M.; Taylor, L.T.; Varela, R.M.; Cutler, S.J.; Cutler, H.G. Fractional extraction of compounds from grape seeds by supercritical fluid extraction and analysis for antimicrobial and agrochemical activities. J. Agric. Food Chem. 1999, 47, 5044–5048. [Google Scholar] [CrossRef]
- Cherchi, G.; Deidda, D.; de Gioannis, B.; Marongiu, B.; Pompei, R.; Porcedda, S. Extraction of Santolina insularis essential oil by supercritical carbon dioxide: influence of some process parameters and biological activity. Flav. Fragr. J. 2001, 16, 35–43. [Google Scholar] [CrossRef]
- Marongiu, B.; Porcedda, S.; Caredda, A.; Piras, A.; Mascia, V.; Cadeddu, A.; Loddo, R. Isolation of Juniperus phoenicea volatiles by supercritical carbon dioxide extraction and bioactivity assays. J. Essent. Oil Res. 2004, 16, 256–261. [Google Scholar] [CrossRef]
- Vagi, E.; Simandi, B.; Suhajda, A.; Hethelyi, E. Essential oil composition and antimicrobial activity of Origanum majorana L. extracts obtained with ethyl alcohol and supercritical carbon dioxide. Food Res. Int. 2005, 38, 51–57. [Google Scholar] [CrossRef]
- Santoyo, S.; Cavero, S.; Jaime, L.; Ibanez, E.; Senorans, F.J.; Reglero, G. Supercritical carbon dioxide extraction of compounds with antimicrobial activity from Origanum vulgare L.: Determination of optimal extraction parameters. J. Food Protect. 2006, 69, 369–375. [Google Scholar]
- Karakaya, S.; El, S.N.; Karagozlu, N.; Sahin, S. Antioxidant and Antimicrobial Activities of Essential Oils Obtained from Oregano (Origanum vulgare ssp hirtum) by Using Different Extraction Methods. J. Med. Food 2011, 14, 645–652. [Google Scholar] [CrossRef]
- Bernardes, W.A.; Lucarini, R.; Tozatti, M.G.; Bocalon Flauzino, L.G.; Souza, M.G.; Turatti, I.C.; Andrade e Silva, M.; Martins, C.H.; da Silva Filho, A.A.; et al. Antibacterial Activity of the Essential Oil from Rosmarinus officinalis and its Major Components against Oral Pathogens. Z. Naturforsch. C 2010, 65, 588–593. [Google Scholar]
- Bozin, B.; Mlmica-Dukic, N.; Samojlik, I.; Jovin, E. Antimicrobial and antioxidant properties of rosemary and sage (Rosmarinus officinalis L. and Salvia officinalis L., Lamiaceae) essential oils. J. Agric. Food Chem. 2007, 55, 7879–7885. [Google Scholar] [CrossRef]
- Okoh, O.; Sadimenko, A.; Afolayan, A. Comparative evaluation of the antibacterial activities of the essential oils of Rosmarinus officinalis L. obtained by hydrodistillation and solvent free microwave extraction methods. Food Chem. 2010, 120, 308–312. [Google Scholar] [CrossRef]
- Genena, A.K.; Hense, H.; Smania Junior, A.; de Souza, S.M. Rosemary (Rosmarinus officinalis)—A study of the composition, antioxidant and antimicrobial activities of extracts obtained with supercritical carbon dioxide. Cienc. Tecnol. Alim. 2008, 28, 463–469. [Google Scholar] [CrossRef]
- Carvalho, R.N.; Moura, L.S.; Rosa, P.T.V.; Meireles, M.A.A. Supercritical fluid extraction from rosemary (Rosmarinus officinalis): Kinetic data, extract's global yield, composition, and antioxidant activity. J. Supercrit. Fluids 2005, 35, 197–204. [Google Scholar] [CrossRef]
- Klancnik, A.; Guzej, B.; Kolar, M.H.; Abramovic, H.; Mozina, S.S. In Vitro Antimicrobial and Antioxidant Activity of Commercial Rosemary Extract Formulations. J. Food Protect. 2009, 72, 1744–1752. [Google Scholar]
- Santoyo, S.; Cavero, S.; Jaime, L.; Ibanez, E.; Senorans, F.J.; Reglero, G. Chemical composition and antimicrobial activity of Rosmarinus officinalis L. essential oil obtained via supercritical fluid extraction. J. Food Protect. 2005, 68, 790–795. [Google Scholar]
- Medina, A.L.; Lucero, M.E.; Holguin, F.O.; Estell, R.E.; Posakony, J.J.; Simon, J.; O’Connell, M.A. Composition and antimicrobial activity of Anemopsis californica leaf oil. J. Agric. Food Chem. 2005, 53, 8694–8698. [Google Scholar] [CrossRef]
- Santoyo, S.; Lloria, R.; Jaime, L.; Ibanez, E.; Senorans, F.J.; Reglero, G. Supercritical fluid extraction of antioxidant and antimicrobial compounds from Laurus nobilis L. Chemical and functional characterization. Eur. Food Res. Technol. 2006, 222, 565–571. [Google Scholar] [CrossRef]
- Yang, C.H.; Yang, C.S.; Hwang, M.L.; Chang, C.C.; Li, R.X.; Chuang, L.Y. Antimicrobial Activity of Various Parts of Cinnamomum cassia Extracted with Different Extraction Methods. J. Food Biochem. 2012, 36, 690–698. [Google Scholar] [CrossRef]
- Glisic, S.B.; Misic, D.R.; Stamenic, M.D.; Zizovic, I.T.; Asanin, R.M.; Skala, D.U. Supercritical carbon dioxide extraction of carrot fruit essential oil: Chemical composition and antimicrobial activity. Food Chem. 2007, 105, 346–352. [Google Scholar] [CrossRef]
- Pyo, D.; Oo, H.H. Supercritical fluid extraction of drug-like materials from selected Myanmar natural plants and their antimicrobial activity. J. Liquid Chromatogr. Rel. Technol. 2007, 30, 377–392. [Google Scholar] [CrossRef]
- Liang, M.T.; Yang, C.H.; Li, S.T.; Yang, C.S.; Chang, H.W.; Liu, C.S.; Cham, T.M.; Chuang, L.Y. Antibacterial and antioxidant properties of Ramulus Cinnamomi using supercritical CO2 extraction. Eur. Food Res. Technol. 2008, 227, 1387–1396. [Google Scholar] [CrossRef]
- Liu, X.; Zhao, M.; Luo, W.; Yang, B.; Jiang, Y. Identification of Volatile Components in Phyllanthus emblica L. and Their Antimicrobial Activity. J. Med. Food 2009, 12, 423–428. [Google Scholar] [CrossRef]
- Liu, X.; Zhao, M.; Wang, J.; Luo, W. Antimicrobial and Antioxidant Activity of Emblica Extracts Obtained by Supercritical Carbon Dioxide Extraction and Methanol Extraction. J. Food Biochem. 2009, 33, 307–330. [Google Scholar] [CrossRef]
- Michielin, E.M.; Salvador, A.A.; Riehl, C.A.; Smania, A., Jr.; Smania, E.F.; Ferreira, S.R. Chemical composition and antibacterial activity of Cordia verbenacea extracts obtained by different methods. Biores. Technol. 2009, 100, 6615–6623. [Google Scholar] [CrossRef]
- Silva, F.V.; Martins, A.; Salta, J.; Neng, N.R.; Nogueira, J.M.; Mira, D.; Gaspar, N.; Justino, J.; Grosso, C.; Urieta, J.S.; Palavra, A.M.; Rauter, A.P. Phytochemical Profile and Anticholinesterase and Antimicrobial Activities of Supercritical versus Conventional Extracts of Satureja montana. J. Agric. Food Chem. 2009, 57, 11557–11563. [Google Scholar] [CrossRef]
- Zalepugin, D.; Tilkunova, N.; Yashin, Y.S.; Chernyshova, I., V; Mishin, V.; Mulyukin, A. Application of supercritical fluid extraction to the development of new potential biocides on the basis of garlic (Allium sativum L.). Russ. J. Phys. Chem. B 2010, 4, 1103–1111. [Google Scholar] [CrossRef]
- Tadic, V.; Bojovic, D.; Arsic, I.; Dordevic, S.; Aksentijevic, K.; Stamenic, M.; Jankovic, S. Chemical and Antimicrobial Evaluation of Supercritical and Conventional Sideritis scardica Griseb., Lamiaceae Extracts. Molecules 2012, 17, 2683–2703. [Google Scholar] [CrossRef]
- Miguel, M.G. Antioxidant and Anti-Inflammatory Activities of Essential Oils: A Short Review. Molecules 2010, 15, 9252–9287. [Google Scholar] [CrossRef]
- Grosso, C.; Oliveira, A.C.; Mainar, A.M.; Urieta, J.S.; Barroso, J.G.; Palavra, A.M.F. Antioxidant Activities of the Supercritical and Conventional Satureja montana Extracts. J. Food Sci. 2009, 74, C713–C717. [Google Scholar] [CrossRef]
- Tipsrisukond, N.; Fernando, L.N.; Clarke, A.D. Antioxidant effects of essential oil and oleoresin of black pepper from supercritical carbon dioxide extractions in ground pork. J. Agric. Food Chem. 1998, 46, 4329–4333. [Google Scholar] [CrossRef]
- Topal, U.; Sasaki, M.; Goto, M.; Otles, S. Chemical compositions and antioxidant properties of essential oils from nine species of Turkish plants obtained by supercritical carbon dioxide extraction and steam distillation. Int. J. Food Sci. Nutr. 2008, 59, 619–634. [Google Scholar] [CrossRef]
- Pronyk, C.; Mazza, G. Design and scale-up of pressurized fluid extractors for food and bioproducts. J. Food Eng. 2009, 95, 215–226. [Google Scholar] [CrossRef]
- Braga, M.E.M.; Leal, P.F.; Carvalho, J.E.; Meireles, M.A.A. Comparison of yield, composition, and antioxidant activity of turmeric (Curcuma longa L.) extracts obtained using various techniques. J. Agric. Food Chem. 2003, 51, 6604–6611. [Google Scholar] [CrossRef]
- Moura, L.S.; Carvalho, R.N.; Stefanini, M.B.; Ming, L.C.; Meireles, M.A.A. Supercritical fluid extraction from fennel (Foeniculum vulgare): Global yield, composition and kinetic data. J. Supercrit. Fluids 2005, 35, 212–219. [Google Scholar] [CrossRef]
- Mezzomo, N.; Martinez, J.; Ferreira, S.R.S. Supercritical fluid extraction of peach (Prunus persica) almond oil: Kinetics, mathematical modeling and scale-up. J. Supercrit. Fluids 2009, 51, 10–16. [Google Scholar] [CrossRef]
- Mendez-Santiago, J.; Teja, A.S. Solubility of solids in supercritical fluids: Consistency of data and a new model for cosolvent systems. Ind. Eng. Chem. Res. 2000, 39, 4767–4771. [Google Scholar] [CrossRef]
- Ashour, I.; Almehaideb, R.; Fateen, S.E.; Aly, G. Representation of solid-supercritical fluid phase equilibria using cubic equations of state. Fluid Phase Equil. 2000, 167, 41–61. [Google Scholar] [CrossRef]
- Chafer, A.; Fornari, T.; Berna, A.; Stateva, R.P. Solubility of quercetin in supercritical CO2 plus ethanol as a modifier: measurements and thermodynamic modelling. J. Supercrit. Fluids 2004, 32, 89–96. [Google Scholar] [CrossRef]
- Martinez, J.; Monteiro, A.R.; Rosa, P.T.V.; Marques, M.O.M.; Meireles, M.A.A. Multicomponent model to describe extraction of ginger oleoresin with supercritical carbon dioxide. Ind. Eng. Chem. Res. 2003, 42, 1057–1063. [Google Scholar] [CrossRef]
- Lucas, S.; Calvo, M.P.; Garcia-Serna, J.; Palencia, C.; Cocero, M.J. Two-parameter model for mass transfer processes between solid matrixes and supercritical fluids: Analytical solution. J. Supercrit. Fluids 2007, 41, 257–266. [Google Scholar] [CrossRef]
- Grosso, C.; Coelho, J.P.; Pessoa, F.L.P.; Fareleira, J.M.N.A.; Barroso, J.G.; Urieta, J.S.; Palavra, A.F.; Sovová, H. Mathematical modelling of supercritical CO2 extraction of volatile oils from aromatic plants. Chem. Eng. Sci. 2010, 65, 3579–3590. [Google Scholar] [CrossRef]
- Sousa, E.M.B.D.; Chiavone, O.; Moreno, M.T.; Silva, D.N.; Marques, M.O.M.; Meireles, M.A.A. Experimental results for the extraction of essential oil from Lippia sidoides Cham. using pressurized carbon dioxide. Braz. J. Chem. Eng. 2002, 19, 229–241. [Google Scholar] [CrossRef]
- Pourmortazavi, S.M.; Sefidkon, F.; Hosseini, S.G. Supercritical carbon dioxide extraction of essential oils from Perovskia atriplicifolia Benth. J. Agric. Food Chem. 2003, 51, 5414–5419. [Google Scholar] [CrossRef]
- Baysal, T.; Starmans, D.A.J. Supercritical carbon dioxide extraction of carvone and limonene from caraway seed. J. Supercrit. Fluids 1999, 14, 225–234. [Google Scholar] [CrossRef]
- Braga, M.E.M.; Ehlert, P.A.D.; Ming, L.C.; Meireles, M.A.A. Supercritical fluid extraction from Lippia alba: Global yields, kinetic data, and extract chemical composition. J. Supercrit. Fluids 2005, 34, 149–156. [Google Scholar] [CrossRef]
- Quispe-Condori, S.; Sanchez, D.; Foglio, M.A.; Rosa, P.T.V.; Zetzl, C.; Brunner, G.; Meireles, M.A.A. Global yield isotherms and kinetic of artemisinin extraction from Artemisia annua L leaves using supercritical carbon dioxide. J. Supercrit. Fluids 2005, 36, 40–48. [Google Scholar] [CrossRef]
- Barghamadi, A.; Mehrdad, M.; Sefidkon, F.; Yamini, Y.; Khajeh, M. Comparison of the volatiles of Achillea millefolium L. obtained by supercritical carbon dioxide extraction and hydrodistillation methods. J. Essent. Oil Res. 2009, 21, 259–263. [Google Scholar] [CrossRef]
- Zarena, A.S.; Sachindra, N.M.; Sankar, K.U. Optimisation of ethanol modified supercritical carbon dioxide on the extract yield and antioxidant activity from Garcinia mangostana L. Food Chem. 2012, 130, 203–208. [Google Scholar] [CrossRef]
- Kagliwal, L.D.; Pol, A.S.; Patil, S.C.; Singhal, R.S.; Patravale, V.B. Antioxidant-rich extract from dehydrated seabuckthorn berries by supercritical carbon dioxide extraction. Food Bioproc. Technol. 2012, 5, 2768–2776. [Google Scholar] [CrossRef]
- Pereira, C.G.; Meireles, M. Economic analysis of rosemary, fennel and anise essential oils obtained by supercritical fluid extraction. Flav. Fragr. J. 2007, 22, 407–413. [Google Scholar] [CrossRef]
- Rosa, P.T.V.; Meireles, M.A.A. Rapid estimation of the manufacturing cost of extracts obtained by supercritical fluid extraction. J. Food Eng. 2005, 67, 235–240. [Google Scholar] [CrossRef]
- Del Valle, J.M.; Rivera, O.; Mattea, M.; Ruetsch, L.; Daghero, J.; Flores, A. Supercritical CO2 processing of pretreated rosehip seeds: effect of process scale on oil extraction kinetics. J. Supercrit. Fluids 2004, 31, 159–174. [Google Scholar] [CrossRef]
- Zuknik, M.H.; Norulaini, N.A.N.; Omar, A.K.M. Supercritical carbon dioxide extraction of lycopene: A review. J. Food Eng. 2012, 112, 253–262. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Capuzzo, A.; Maffei, M.E.; Occhipinti, A. Supercritical Fluid Extraction of Plant Flavors and Fragrances. Molecules 2013, 18, 7194-7238. https://doi.org/10.3390/molecules18067194
Capuzzo A, Maffei ME, Occhipinti A. Supercritical Fluid Extraction of Plant Flavors and Fragrances. Molecules. 2013; 18(6):7194-7238. https://doi.org/10.3390/molecules18067194
Chicago/Turabian StyleCapuzzo, Andrea, Massimo E. Maffei, and Andrea Occhipinti. 2013. "Supercritical Fluid Extraction of Plant Flavors and Fragrances" Molecules 18, no. 6: 7194-7238. https://doi.org/10.3390/molecules18067194
APA StyleCapuzzo, A., Maffei, M. E., & Occhipinti, A. (2013). Supercritical Fluid Extraction of Plant Flavors and Fragrances. Molecules, 18(6), 7194-7238. https://doi.org/10.3390/molecules18067194






