Abstract
The ionic liquid 1-butyl-3-methylimidazolium methylselenite, [bmim][SeO2(OCH3)], was successfully used as solvent in the catalyst-free preparation of 3-arylselenylindoles by the reaction of indole with ArSeCl at room temperature. The products were obtained selectively in good yields without the need of any additive and the solvent was easily reused for several cycles with good results.
1. Introduction
Functionalized indoles, such as 3-arylthioindoles have attracted the attention of researchers in organic synthesis and medicinal chemistry due their potent pharmacological activities, including the inhibition of breast cancer cells [1] and of 5-lipoxygenase, which may increase the antitumor activity of the drug celecoxib [2], and in the treatment of heart disease [3] and HIV [4]. In this sense, there are a range of methods to synthesize this class of compounds, most of them starting from indoles and an electrophilic sulfur reagent [5,6,7,8].
Despite the fact that the usefulness of organoselenium compounds in chemical sciences has already been described in a great number of reviews and books [9,10,11,12,13,14,15] the synthesis of 3-arylselenylindoles and their potential bioactivity have not been extensively studied. These compounds can be obtained by electrophilic cyclization of 2-alkynylanilines with arylselenyl chlorides [16,17] or iodides [18], by the annulation of 2-(gem-dibromo(chloro)vinyl)-N-methylsulfonylanilines with diselenides [19] or by the cyclization of 2-styrylacetanilides using N-phenylselenosuccinimide [20].
The direct selenylation of the easily available indole core, which is a more direct route to 3-arylselenylindoles, was even less explored, as for example in the indoline dehydrogenation using phenylseleninic anhydride, (PhSeO)2O [21,22,23] or phenylseleninic acid, PhSeOH [24] (in these works 3-phenylselenylindole was a side product) and in the reaction of 2-acylphenylselenocyanates with phenylhydrazine (only one example) [25]. The reactions between indoles and electropilic selenium species catalyzed by I2/FeF3 [26] and p-TsOH [27] were also described. More recently, Silveira and co-workers [28] used the PhSeSePh/TCCA/MgO system to generate PhSeCl in situ, which was reacted with several indoles to give the respective 3-arylselenylindoles in good yields.
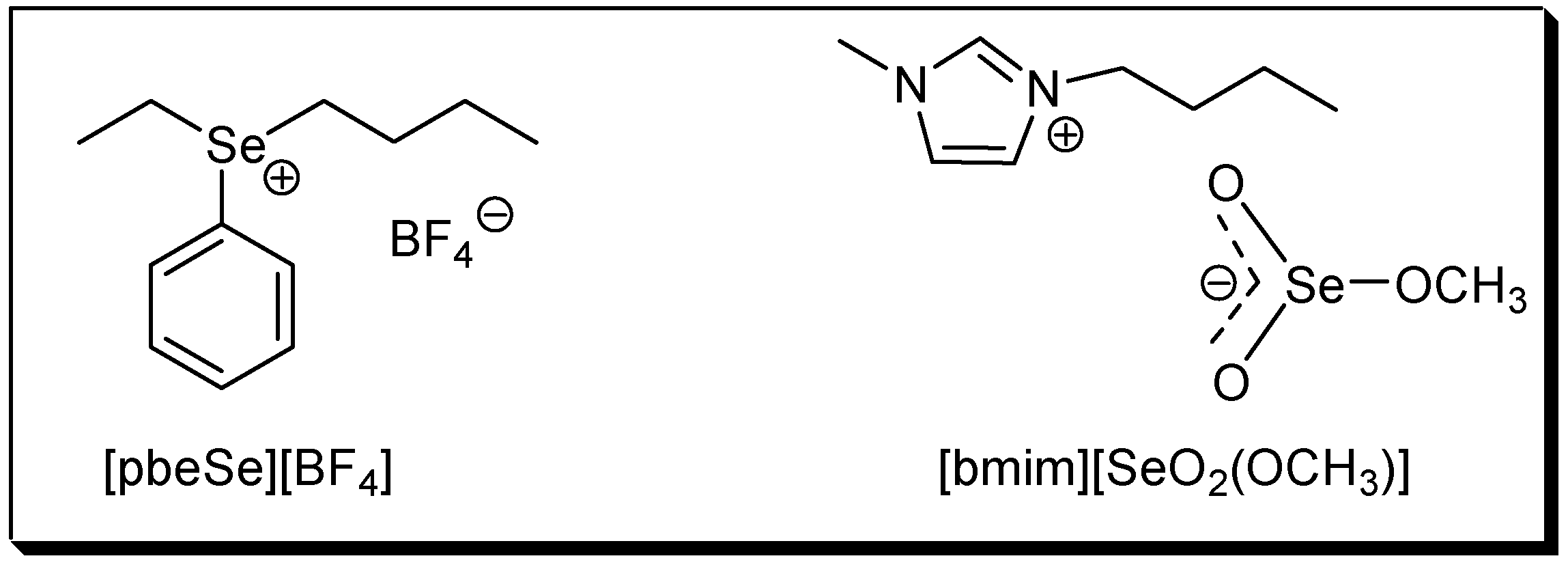
The use of ionic liquids (ILs) as solvent and/or catalyst has attracted much attention in the last years. Because product isolation or catalyst recycling in ILs is very easy and, in some cases, rate accelerations and/or selectivity improvements are also observed, they are regarded as environmentally friendly, green solvents [29,30,31,32,33]. In this context, the use of the new selenium-based ILs phenylbutyl-ethyl selenonium tetrafluoroborate, [pbeSe][BF4] and 1-butyl-3-methylimidazolium methylselenite, [bmim][SeO2(OCH3)] (Figure 1) was recently reported. The selenonium IL was used as an efficient catalyst in several acid-catalyzed reactions [34,35,36], while the selenite IL was employed in the oxidative carbonylation of aniline [37], the base-free oxidation of thiols to disulfides [38] and in the synthesis of vinyl sulfides [39].
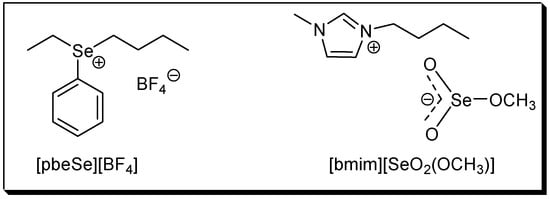
Figure 1.
Structures of the selenium-based ionic liquids.
Figure 1.
Structures of the selenium-based ionic liquids.
To the best of our knowledge, the preparation of 3-arylselenylindoles directly from indoles and electrophilic selenium species under acid-free conditions was not described. In this context, and due our ongoing interest in new applications for selenium-based ionic liquids and selenium-containing compounds, we decide to investigate the use of [bmim][SeO2(OCH3)] as solvent for the general, catalyst-free arylselenation of indoles to prepare 3-arylselenylindoles (Scheme 1).
2. Results and Discussion
Our initial efforts were made towards the determination of the optimum conditions to perform the reaction. Thus, we chose indole (1a) and phenylselenyl chloride (2a) to establish the best conditions for the arylselenylation reaction (Table 1).
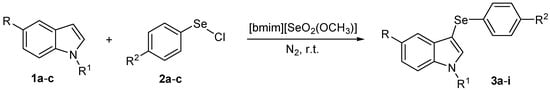
Scheme 1.
Synthesis of 3-arylselenylindoles using [bimim][SeO2(OCH3)] as solvent.
Scheme 1.
Synthesis of 3-arylselenylindoles using [bimim][SeO2(OCH3)] as solvent.
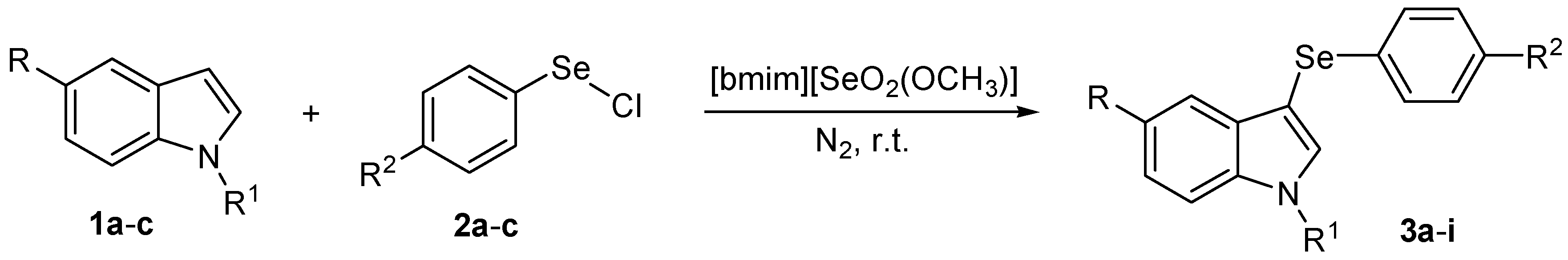
We examined the effect of temperature, use of nitrogen atmosphere and different ionic liquids, such as [bmin][PF6], [bmin][BF4], [bmim][SeO2(OCH3)] and [pbeSe][BF4]. Except for [bmim][PF6] (Table 1, entry 3), the desired selenylated indole 3a was isolated in all the tested conditions and the best yields were observed using [bmim][SeO2(OCH3)] as solvent (78 and 79% yields, entries 1 and 6). In contrast, the selenonium ionic liquid [pbeSe][BF4] was not stable under the reaction conditions, affording a lower yield of 3a among a mixture of diorganyl selenides resulting from the IL (Table 1, entry 2). We also verified the influence of the electrophilic selenium species in the reaction. It was found that when PhSeBr was used the yield decreased to 57% (Table 1, entry 5), while N-phenylselenyl phthalimide (PSP) afforded 3-phenylselenylindole 3a in a similar yield to PhSeCl (79%, entry 6). Since PhSeCl is cheaper than PSP, it is advantageous to use the former reagent instead PSP. It was also observed that when an open atmosphere is used or the mixture is heated, the yield of 3a decreases. In the second case, decomposition of the IL was observed, with darkening of the solution. Thus, the best reaction conditions were defined as stirring a solution of indole 3a (1.0 mmol) and phenylselenyl chloride 2a (1.0 mmol) in [bmim][SeO2(OCH3)] (1.5 mL) at room temperature under a N2 atmosphere for 3 hours.

Table 1.
Optimization studies for preparation of 3-(phenylselenyl)-1H-indole a. 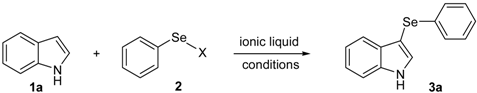

| entry | ionic liquid | X | time (h) | yield (%) b |
|---|---|---|---|---|
| 1 | [bmim][SeO2(OCH3)] | Cl | 3 | 78 |
| 2 | [pbeSe][BF4] | Cl | 3 | 39c |
| 3 | [bmim][PF6] | Cl | 24 | NRd |
| 4 | [bmim][BF4] | Cl | 3 | 28 |
| 5 | [bmim][SeO2(OCH3)] | Br | 3 | 57 |
| 6 | [bmim][SeO2(OCH3)] | 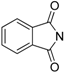 | 3 | 79 |
a The reaction was performed using indole (1a, 1.0 mmol) and electrophilic selenium species (1.0 mmol) in ionic liquid (1.5 mL) at room temperature and under a N2 atmosphere. b Isolated yields. c Decomposition of the IL was observed. d No reaction.
With these optimized conditions in hands, a detailed study was performed with different indoles and arylselenyl chlorides, showing the generality of the method (Table 2).

Table 2.
Synthesis of 3-organylselanylindoles a. 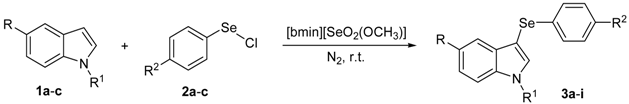

| Entry | Indole 1a–c | 2a–c | Product 3a–i | Time (h) | Yield (%) b |
|---|---|---|---|---|---|
| 1 |  | 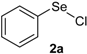 | 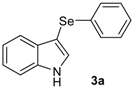 | 3 | 78 (74)c |
| 2 | 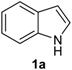 | 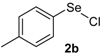 | 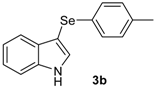 | 2 | 73 |
| 3 | 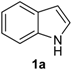 | 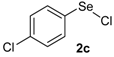 | 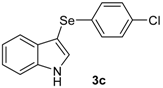 | 3 | 55 |
| 4 | 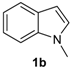 | 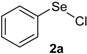 | 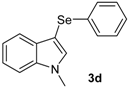 | 3 | 68 |
| 5 | 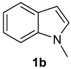 | 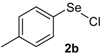 | 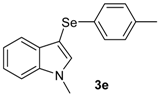 | 2 | 65 |
| 6 | 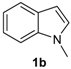 | 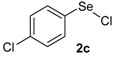 | 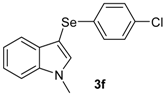 | 3 | 53 |
| 7 | 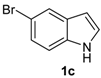 | 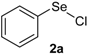 | 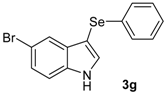 | 3 | 74 |
| 8 | 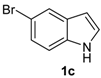 | 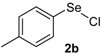 | 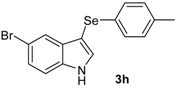 | 2 | 65 |
| 9 | 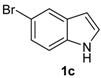 | 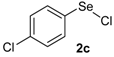 | 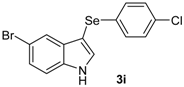 | 3 | 62 |
a The reaction was performed using indole (1, 1.0 mmol) and electrophilic selenium species (2, 1.0 mmol) in ionic liquid (1.5 mL) at room temperature and under N2 atmosphere. b Isolated yields. c Reaction performed in a 10 mmol scale and using 6 mL of IL.
From the results listed in Table 2, it can be seen that [bmim][SeO2(OCH3)] was a good reaction medium to afford a wide range of 3-arylselenylindoles. A possible role of the IL here could be a neutralizing effect in the HCl released in the reaction.
The presence of electron-donor and electron-withdrawing groups on the selenium species did not affect substantially the yields or the reaction time. Thus, indole (1a) reacted with p-tolylselenyl chloride (2b) under our conditions to afford 3-p-tolylselenylindole (3b) in 73% yield after 3 h (Table 2, entry 2). Analogously, 4-chlorophenylselenyl chloride (2c) afforded the respective 3-arylselenylindole (3c) in 55% yield after 3 h after reaction with 1a (entry 3). We also extended our protocol to N-methyl-1H-indole (1b), which afforded the 3-arylselenylindoles 3d–f after reaction with 2a–c in slightly lower yields than those obtained for the parent compound 3a (Table 2, entries 4–6). Similarly, the functionalized 5-bromo-1H-indole (3c) reacted at room temperature with the arylselenyl chlorides 2a–c in presence of [bmim][SeO2(OCH3)] to selectively afford the desired 5-bromo-3-(arylselanyl)-1H-indoles 3g–i in good yields after short reaction times (Table 2, entries 7–9).
Additionally, a reuse study of the ionic liquid was carried out for the reaction of 1a with 2a to obtain 3a. After stirring at r.t. during 3 h, the reaction mixture was diluted with ether (3 × 5.0 mL). The upper organic phase was washed with water, the solvent evaporated and the product was isolated. The remaining [bmim][SeO2(OCH3)] was directly reused for further reactions, simple by adding more reagents 1a and 2a. It was observed that as the IL was being reused, it gradually darkened, from an initial light yellow to orange and then to red and finally brown, which may be indicative of its decomposition. It was observed that a good level of efficiency was maintained up until the fourth cycle and it dropped in the fifth and sixth cycles (Table 3).

Table 3.
Reuse of [bmim][SeO2(OCH3)]. 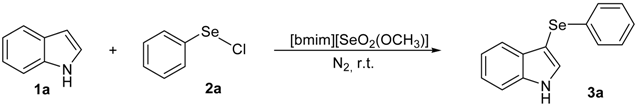
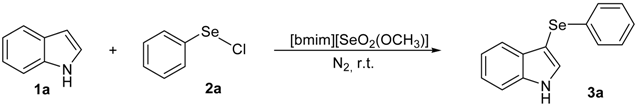
| Cycle | Time (h) | Yield of 3a (%) a |
|---|---|---|
| 1 | 4 | 77 |
| 2 | 4 | 73 |
| 3 | 4 | 75 |
| 4 | 4 | 70 |
| 5 | 24 | 16 |
| 6 | 24 | trace |
a Yields are given for isolated products.
3. Experimental
3.1. General
Nuclear magnetic resonance spectra (1H- and 13C-NMR) were obtained at 200 and 400 MHz on Bruker DPX spectrometers. Spectra were recorded in CDCl3 solutions. Chemical shifts are reported in ppm, referenced to tetramethylsilane (TMS) as the external reference. Data are reported as follows: chemical shift (δ), multiplicity, coupling constant (J) in Hertz and integrated intensity. Carbon-13 nuclear magnetic resonance spectra (13C-NMR) were obtained at 50 and 100 MHz on Bruker DPX spectrometers. Spectra were recorded in CDCl3 solutions. Chemical shifts are reported in ppm, referenced to the solvent peak of CDCl3. Mass spectra (MS) were measured on a Shimadzu GCMS-QP2010 mass spectrometer. Column chromatography was performed using Merck Silica Gel (230–400 mesh). Thin layer chromatography (TLC) was performed using Merck Silica Gel GF254, 0.25 mm thickness. For visualization, TLC plates were either placed under ultraviolet light, or stained with iodine vapor, or acidic vanillin. All solvents were used as purchased unless otherwise noted. p-Tolylselenyl chloride (2b) and 4-chlorophenylselenyl chloride (2c) [40] and the ionic liquids [bmim][SeO2(CH3)] [37] and [pbeSe][BF4] [34] were synthesized as described in the literature.
3.2. General Synthesis Procedure
To a mixture of indole 1 (1.0 mmol) in [bmim][SeO2(OCH3)] (1.5 mL) under a N2 atmosphere, organylselenyl chloride 2 (1.0 mmol) was added at room temperature and the mixture was stirred for the time indicated in Table 2. The progress of the reaction was monitored by TLC. After the reaction was complete, the product was extracted by successive washings with ether (3 × 5 mL). The upper organic phase was washed with water, dried over MgSO4, and concentrated under vacuum. The residue was purified by column chromatography on silica gel using ethyl acetate/hexanes as the eluent. All the compounds were characterized and the corresponding spectral data are listed below:
3-(Phenylselenyl)-1H-indole (3a) [26]: Yield: 0.213 g (78%). 1H-NMR (CDCl3, 400 MHz): δ = 8.35 (br s, 1H), 7.64 (d, J = 7.9, 1H), 7.39–7.43 (m, 2H), 7.22–7.25 (m, 4H), 7.08–7.14 (m, 3H). 13C-NMR (100 MHz, CDCl3): δ = 136.4, 133.8, 131.2, 129.9, 128.9, 128.7, 125.6, 122.9, 120.8, 120.3, 111.3, 98.2. MS: m/z (rel. int.) 273 (6.0), 193 (100.0), 117 (5.3), 77 (21.0).
3-(p-Tolylselenyl)-1H-indole (3b): Yield: 0.210 g (73%). 1H-NMR (CDCl3, 200 MHz): δ = 8.18 (s, 1H), 7.63 (d, J = 7.6, 1H), 7.14–7.32 (m, 7H), 6.91 (d, J = 7.6, 1H), 2.20 (s, 3H). 13C-NMR (50 MHz, CDCl3): δ = 136.5, 135.6, 133.4, 130.8, 130.5, 129.7, 129.4, 122.8, 120.8, 120.4, 111.3, 99.0, 20.8. MS: m/z (rel. int.) 287 (2.7), 207 (100.0), 117 (4.9), 77 (14.4).
3-(4-Chlorophenylselenyl)-1H-indole (3c): Yield: 0.169 g (55%). 1H-NMR (CDCl3, 200 MHz): δ = 8.39 (br s, 1H), 7.59 (d, J = 7.7 Hz, 1H), 7.38–7.42 (m, 2H), 7.03-7.29 (m, 6H). 13C-NMR (50 MHz, CDCl3): δ = 136.6, 132.1, 131.8, 130.3, 129.9, 129.0, 123.2, 121.1, 120.3, 111.4, 98.4. MS: m/z (rel. int.) 307 (4.9), 227 (100.0), 116 (12.5), 77 (14.1).
1-Methyl-3-(phenylselenyl)-1H-indole (3d) [27]: Yield: 0.195 g (68%). 1H-NMR (CDCl3, 200 MHz): δ= 7.63 (d, J = 7.8, 1H), 7.07–7.35 (m, 9H), 3.74–3,80 (s, 3H). 13C-NMR (50 MHz, CDCl3): δ = 137.6, 135.5, 134.2, 130.8, 129.3, 128.9, 125.6, 122.4, 120.5, 120.4, 109.5, 96.4, 33.0. MS: m/z (rel. int.) 287 (8.0), 207 (100.0), 130 (18.7), 77 (11.2).
1-Methyl-3-(p-tolylselenyl)-1H-indole (3e): Yield: 0.196 g (65%). 1H-NMR (CDCl3, 200 MHz): δ = 7,61 (d, J = 7.9, 1H), 7.35–6.87 (m, 8H), 3.79 (s, 3H), 2.22 (s, 3H). 13C-NMR (50 MHz, CDCl3): δ = 137.6, 135.4, 135.2, 130.8, 130.2, 129.7, 129.3, 122.4, 120.6, 120.3, 109.4, 97.0, 32.8, 20.8. MS: m/z (rel. int.) 301 (5.4), 221 (100.0), 130 (17.1).
3-(4-Chlorophenylselenyl)-1-methyl-1H-indole (3f): Yield: 0.170 g (53%). 1H-NMR (CDCl3, 200 MHz): δ = 7.59 (d, J = 7.7, 1H), 7.03–7.39 (m, 8H), 3.83 (s, 3H). 13C-NMR (50 MHz, CDCl3): δ = 137.5, 135.6, 132.5, 131.5, 130.4, 129.9, 128.9, 122.6, 120.5, 120.3, 109.6, 91,7, 33.0. MS: m/z (rel. int.) 321 (8.7), 241 (100.0), 130.0 (26.7), 77 (11.5).
5-Bromo-3-(phenylselenyl)-1H-indole (3g) [26]: Yield: 0.260 g (74%). 1H-NMR (CDCl3, 400 MHz): δ= 8.38 (br s, 1H), 7.74 (s, 1H), 7.38 (d, J = 2.2, 1H), 7.09–7.33 (m, 8H). 13C-NMR (100 MHz, CDCl3): δ = 135.0, 133.3, 132.4, 131.8, 129.0, 128.7, 125.9, 125.8, 122.9, 114.3, 112.9, 97.8. MS: m/z (rel. int.) 351 (15.2), 271 (100.0), 192 (73.8), 116 (11.3), 77 (42.3).
5-Bromo-3-(p-tolylselenyl)-1H-indole (3h) [28]: Yield: 0.237 g (65%). 1H-NMR (CDCl3, 200 MHz): δ = 8.42 (br s, 1H), 7.77 (s, 1H), 7.42 (d, J = 2.3, 1H), 7.12–7.30 (m, 4H), 6.95 (d, J = 8.0, 2H), 2.36 (s, 3H). 13C-NMR (50 MHz, CDCl3): δ = 135.8, 135.0, 132.1, 131.8, 129.9, 129.3, 129.2, 125.8, 122.9, 114.3, 112.8, 98.4, 20.9. MS: m/z (rel. int.) 365 (18.3), 285 (100.0), 194 (12.7), 91 (42.2).
5-Bromo-3-(4-chlorophenylselenyl)-1H-indole (3i): Yield: 0.239 g (62%). 1H-NMR (CDCl3, 400 MHz): δ = 8.47 (br s, 1H), 7.71 (d, J = 1.7, 1H), 7.41 (d, J = 2.5, 1H), 7.25–7.33 (m, 2H), 7.06–7.12 (m, 4H). 13C-NMR (100 MHz, CDCl3): δ = 135.0, 132.4, 131.8, 131.6, 131.5, 130.0, 129.1, 126.0, 122.7, 114.4, 114.4, 112.9. MS: m/z (rel. int.) 385 (5.6), 307 (75.0), 191 (72.1), 115 (46.8), 75 (100.0).
4. Conclusions
In summary, we present here the first report on the use of the ionic liquid [bmim][SeO2(OCH3)] in the selective synthesis of 3-arylselenylindoles. The products were obtained in good yields at room temperature in a relatively short time without the need of any additive. Moreover, the IL could be reused directly for up to four cycles with good performance.
Acknowledgments
The authors are grateful to FAPERGS and CNPq (PRONEX 10/0005-1, PRONEM 11/2026-4, PqG 11/0719-3 and PqG 11/0881-2), CAPES and FINEP for their financial support.
References and Notes
- De Martino, G.; Edler, M.C.; la Regina, R.; Coluccia, A.; Barbera, M.C.; Barrow, D.; Nicholson, R.I.; Chiosis, G.; Brancale, A.; Hamel, E.; et al. New arylthioindoles: Potent Inhibitors of tubulin polymerization. 2. Structure−activity relationships and molecular modeling studies. J. Med. Chem. 2006, 49, 947–954. [Google Scholar] [CrossRef]
- Cianchi, F.; Cortesini, C.; Magnelli, L.; Fanti, E.; Papucci, L.; Schiavone, N.; Messerini, L.; Vannacci, A.; Capaccioli, S.; Perna, F.; et al. Inhibition of 5-lipoxygenase by MK886 augments the antitumor activity of celecoxib in human colon cancer cells. Mol. Cancer Ther. 2006, 5, 2716–2726. [Google Scholar] [CrossRef]
- Funk, C.D. Leukotriene modifiers as potential therapeutics for cardiovascular disease. Nat. Rev. Drug Discov. 2005, 4, 664–672. [Google Scholar] [CrossRef]
- Ragno, R.; Coluccia, A.; La Regina, G.; De Martino, G.; Piscitelli, F.; Lavecchia, A.; Novellino, E.; Bergamini, A.; Ciaprini, C.; Sinistro, A.; et al. Design, Molecular modeling, Synthesis, And anti-HIV-1 activity of new indolyl aryl sulfones. Novel derivatives of the indole-2-carboxamide. J. Med. Chem. 2006, 49, 3172–3184. [Google Scholar] [CrossRef]
- Tudge, M.; Tamiya, M.; Savarin, C.; Humphrey, G.R. Development of a novel, highly efficient halide-catalyzed sulfenylation of indoles. Org. Lett. 2006, 8, 565–568. [Google Scholar] [CrossRef]
- Pezzella, A.; Palma, A.; Iadonisi, A.; Napolitano, A.; d’Ischia, M. The first entry to 5,6-dihydroxy-3-mercaptoindole, 5-hydroxy-3-mercaptoindole and their 2-carbomethoxy derivatives by a mild thiocyanation/reduction methodology. Tetrahedron Lett. 2007, 48, 3883–3886. [Google Scholar] [CrossRef]
- Yadav, J.S.; Reddy, B.V. S.; Krishna, A.D.; Reddy, C.S.; Narsaiah, A.V. Ferric(III) chloride-promoted electrophilic thiocyanation of aromatic and heteroaromatic compounds. Synthesis 2005, 961–964. [Google Scholar]
- Wu, G.; Liu, Q.; Shen, Y.; Wu, W.; Wu, L. Regioselective thiocyanation of aromatic and heteroaromatic compounds using ammonium thiocyanate and oxone. Tetrahedron Lett. 2005, 46, 5831–5834. [Google Scholar] [CrossRef]
- Devillanova, F.A. Handbook of Chalcogen Chemistry: New Perspectives in S, Se and Te; Royal Society of Chemistry: Cambridge, UK, 2006. [Google Scholar]
- Alberto, E.E.; Braga, A.L. Selenium and Tellurium Chemistry—From Small Molecules to Biomolecules and Materials; Derek, W.J., Risto, L., Eds.; Springer-Verlag: Berlin Heidelberg, Germany, 2011. [Google Scholar]
- Wirth, T. Organoselenium Chemistry: Synthesis and Reactions; Wiley-VCH: Weinheim, Germany, 2011. [Google Scholar]
- Menezes, P.H.; Zeni, G. Vinyl Selenides. In Patai’s Chemistry of Functional Groups; John Wiley & Sons: Oxford, UK, 2011. [Google Scholar]
- Perin, G.; Lenardão, E.J.; Jacob, R.G.; Panatieri, R.B. Synthesis of vinyl selenides. Chem. Rev. 2009, 109, 1277–1301. [Google Scholar] [CrossRef]
- Freudendahl, D.M.; Santoro, S.; Shahzad, S.A.; Santi, C.; Wirth, T. Green chemistry with selenium reagents: Development of efficient catalytic reactions. Angew. Chem. Int. Ed. Engl. 2009, 48, 8409–8411. [Google Scholar] [CrossRef]
- Santi, C.; Santoro, S.; Battistelli, B. Organoselenium compounds as catalysts in nature and laboratory. Curr. Org. Chem. 2010, 14, 2442–2462. [Google Scholar] [CrossRef]
- Chen, Y.; Cho, C.-H; Shi, F.; Larock, R.C. Synthesis of 3-sulfenyl- and 3-selenylindoles by the Pd/Cu-catalyzed coupling of N,N-dialkyl-2-iodoanilines and terminal alkynes, followed by n-Bu4NI-induced electrophilic cyclization. J. Org. Chem. 2009, 74, 6802–6811. [Google Scholar] [CrossRef]
- Chen, Y.; Cho, C.-H.; Shi, F.; Larock, R.C. A novel synthetic route to 3-sulfenyland 3-selenylindoles by n-Bu4NI-induced electrophilic cyclization. Org. Lett. 2009, 11, 173–176. [Google Scholar] [CrossRef]
- Du, H.-A; Tang, R.-Y; Deng, C.-L; Liu, Y.; Li, J.-H; Zhang, X.-G. Iron-facilitated iodine-mediated electrophilic annulation of N,N-dimethyl-2-alkynylanilines with disulfides or diselenides. Adv. Synth. Catal. 2011, 353, 2739–2748. [Google Scholar] [CrossRef]
- Liu, J.; Li, P.; Chen, W.; Wang, L. An efficient synthesis of 2-bromo(chloro)-3-selenyl(sulfenyl)indoles via tandem reactions of 2-(gem-dibromo(chloro)vinyl)anilines with diselenides(disulfides). Chem. Commun. 2012, 48, 10052–10054. [Google Scholar] [CrossRef]
- Izumi, T.; Sugano, M.; Konno, T. Synthesis of indoles via amidoselenation. J. Heterocycl. Chem. 1992, 29, 899–904. [Google Scholar] [CrossRef]
- Barton, D.H.R.; Lusinchi, X; Milliet, P. La transformation d'indolines en indoles et d'autres reactions apparentees. Tetrahedron Lett. 1982, 23, 4949–4952. [Google Scholar]
- Barton, D.H.R.; Lusinchi, X.; Milliet, P. Studies on the reaction of primary and secondary amines with phenylseleninic anhydride and with phenylseleninic acid. Tetrahedron 1985, 41, 4727–4738. [Google Scholar] [CrossRef]
- Danieli, B.; Lesma, G.; Palmisano, G.; Passarella, D.; Silvani, A. Aspidosperma alkaloids via cyclization of secodine intermediate: synthesis of (+/−)-3-oxovincadifformine ethyl ester. Tetrahedron 1994, 50, 6941–6954. [Google Scholar]
- Ninomiya, I.; Kiguchi, T.; Hashimoto, C. An improved procedure for the conversion of indolines into indoles. Tetrahedron Lett. 1985, 26, 4183–4186. [Google Scholar]
- Ames, D.E.; Singh, A.G.; Smyth, W.F. Reaction of 2-acylphenylselenocyanates with hydroxylamine and phenylhydrazine. Tetrahedron 1983, 39, 831–833. [Google Scholar] [CrossRef]
- Fang, X.L.; Tang, R.Y.; Zhong, P.; Li, J.H. Iron-catalyzed sulfenylation of indoles with disulfides promoted by a catalytic amount of iodine. Synthesis 2009, 4183–4189. [Google Scholar]
- Zhao, X.; Yu, Z.; Xu, T.; Wu, P.; Yu, H. Novel Brønsted acid catalyzed three-component alkylations of indoles with N-phenylselenophthalimide and styrenes. Org. Lett. 2007, 9, 5263–5266. [Google Scholar] [CrossRef]
- Silveira, C.C.; Mendes, S.R.; Wolf, L.; Martins, G.M.; von Mühlen, L. Efficient synthesis of 3-selanyl- and 3-sulfanylindoles employing trichloroisocyanuric acid and dichalcogenides. Tetrahedron 2012, 68, 10464–10469. [Google Scholar] [CrossRef]
- Tadesse, H.; Luque, R. Advances on biomass pretreatment using ionic liquids: An overview. Energy Environ. Sci. 2011, 4, 3913–3929. [Google Scholar] [CrossRef]
- Welton, T. Room-temperature ionic liquids. Solvents for synthesis and catalysis. Chem. Rev. 1999, 99, 2071–2083. [Google Scholar] [CrossRef]
- Dupont, J.; Souza, R.F.; Suarez, P.A.Z. Ionic liquid (molten salt) phase organometallic catalysis. Chem. Rev. 2002, 102, 3667–3692. [Google Scholar] [CrossRef]
- Wasserscheid, P.; Welton, P. Ionic Liquids in Synthesis; Wiley-VCH: Weinheim, Germany, 2003. [Google Scholar]
- Martins, M.A.P.; Frizzo, C.P.; Moreira, D.N.; Zanatta, N.; Bonacorso, H.G. Ionic Liquids in Heterocyclic Synthesis. Chem. Rev. 2008, 108, 2015–2050. [Google Scholar] [CrossRef]
- Lenardão, E.J.; Mendes, S.R.; Ferreira, P.C.; Perin, G.; Silveira, C.C.; Jacob, R.G. Selenium- and tellurium-based ionic liquids and their use in the synthesis of octahydroacridines. Tetrahedron Lett. 2006, 47, 7439–7442. [Google Scholar] [CrossRef]
- Lenardão, E.J.; Feijó, J.O.; Thurow, S.; Perin, G.; Jacob, R.G.; Silveira, C.C. Selenonium ionic liquid as efficient catalyst for the Baylis–Hillman reaction. Tetrahedron Lett. 2009, 50, 5215–5217. [Google Scholar] [CrossRef]
- Lenardão, E.J.; Borges, E.L.; Mendes, S.R.; Perin, G.; Jacob, R.G. Selenonium ionic liquid as an efficient catalyst for the synthesis of thioacetals under solvent-free conditions. Tetrahedron Lett. 2008, 49, 1919–1921. [Google Scholar] [CrossRef]
- For the synthesis of [bmim][SeO2(OCH3)] see: Kim, H.S.; Kim, Y.J.; Lee, H.; Park, K.Y.; Lee, C.; Chin, C.S. Ionic Liquids Containing Anionic Selenium Species: Applications for the Oxidative Carbonylation of Aniline. Angew. Chem. Int. Ed. Engl. 2002, 41, 4300–4303. [Google Scholar]
- Thurow, S.; Pereira, V.A.; Martinez, D.M.; Alves, D.; Perin, G.; Jacob, R.G.; Lenardão, E.J. Base-free oxidation of thiols to disulfides using selenium ionic liquid. Tetrahedron Lett. 2011, 52, 640–643. [Google Scholar] [CrossRef]
- Thurow, S.; Ostosi, N.T.; Mendes, S.R.; Jacob, R.G.; Lenardão, E.J. Synthesis of vinyl sulfides under base-free conditions using selenium ionic liquid. Tetrahedron Lett. 2012, 53, 2651–2653. [Google Scholar]
- Nicolaou, K.C.; Petatis, N.A.; Claremon, D.A. N-Phenylselenophthalimide (NPSP) a valuable selenenylating agent. Tetrahedron 1985, 41, 4835–4841. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 3a–i are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
