Isosakuranetin-5-O-rutinoside: A New Flavanone with Antidepressant Activity Isolated from Salvia elegans Vahl.
Abstract
1. Introduction
2. Results and Discussion
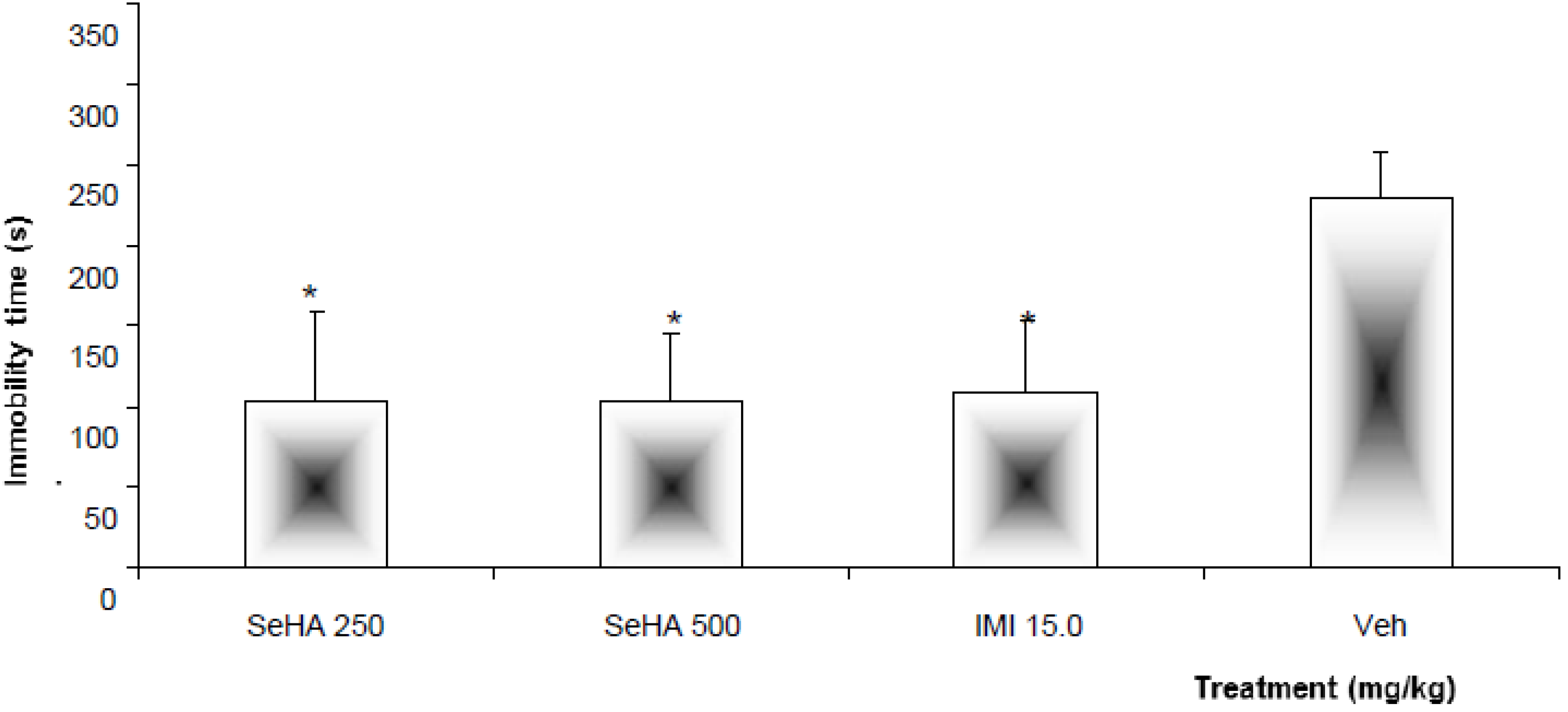
2.1. Evaluation of the Hydroalcoholic Extract
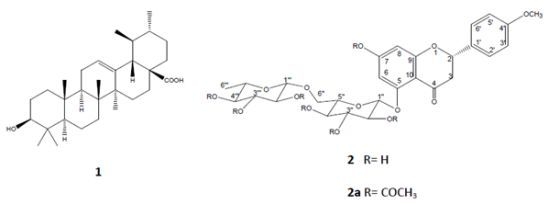
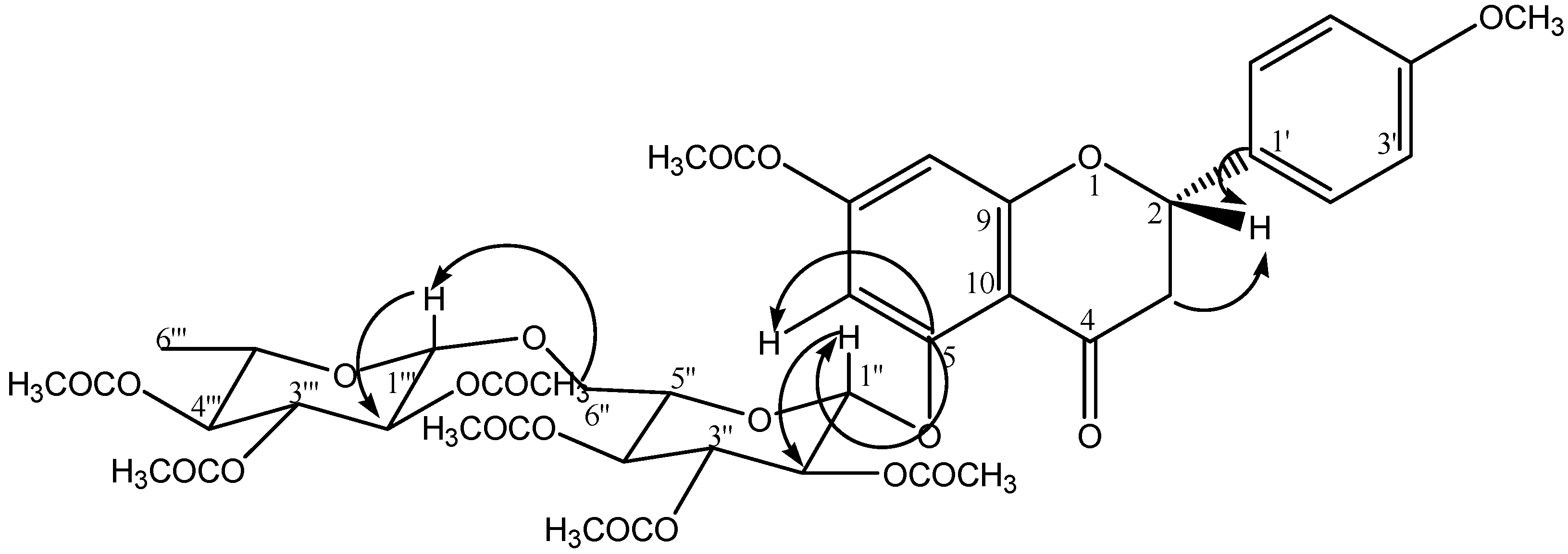
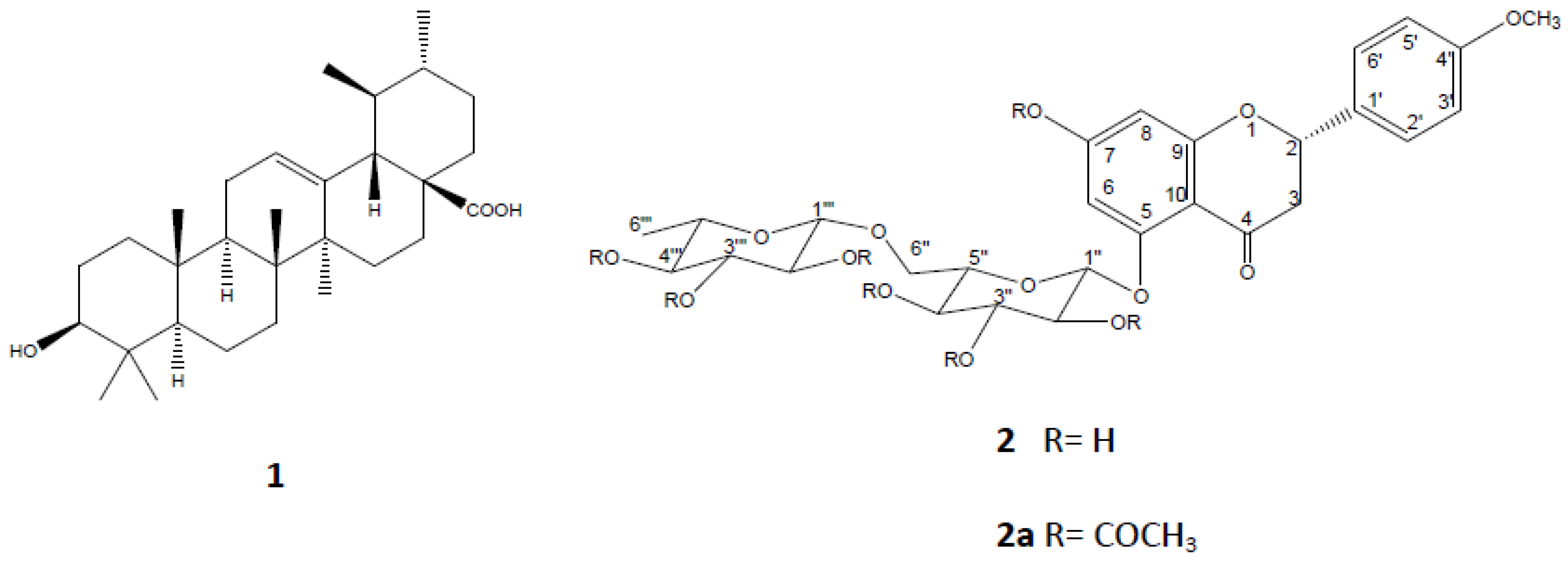
2.2. Chromatographic Separation of the Hydroalcoholic Extract and Isolation and Elucidation of Ursolic Acid (1) and a Flavanone 2
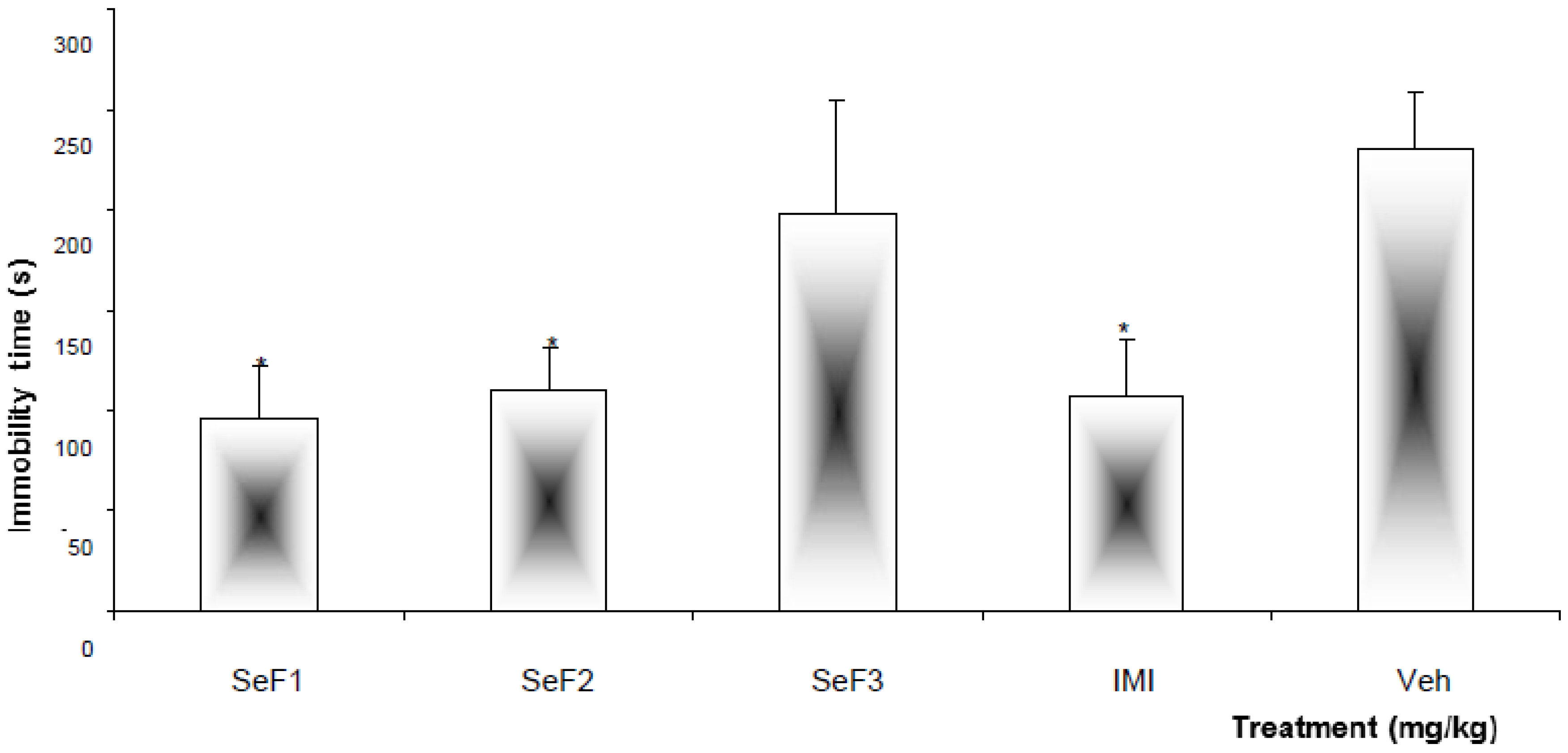
| Treatment | Dose (mg/kg) | Immobility time |
|---|---|---|
| SeF1 | 250 | 96.2 ± 26.1 * |
| F1-1 | 50.0 | 273.1 ± 12.1 |
| F1-2 | 261.7 ± 19.6 | |
| F1-3 | 113.8 ± 23.6 * | |
| UA | 15.0 | 235.5 ± 1.9 |
| 30.0 | 221.2 ± 29.6 | |
| SeF2 | 250.0 | 110.3 ± 21.0 * |
| F2-1 | 50.0 | 286.5 ± 9.2 |
| F2-2 | 241.8 ± 7.5 | |
| Isosakuranetin-5- O-rutinoside | 15.0 | 122.0 ± 19.6 * |
| IMI | 15.0 | 107.9 ± 23.4 * |
| Veh (TW 20, 1%) | 100 µL/10 g | 230.2 ± 28.2 |
3. Experimental
3.1. General Experimental Procedures
3.2. Plant Material
3.3. Extraction and Isolation of Ursolic acid (1) and Isosakuranetin-5-O-Rutinoside (2)
3.4. Acetylation of Isosakuranetin-5-O-rutinoside (2)
3.5. Drugs
3.6. Animals and Drug Administration
3.7. Forced Swimming Test (FST)
3.8. Data Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Conflicts of Interest
References
- Gaffrey, MS.; Luby, JL.; Barch, DM. Towards the study of functional brain development in depression: an interactive specialization approach. Neurobiol. Disease 2013, 52, 38–48. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Available online: http://www.who.int/mediacentre/news/notes/2012/mental_health_day_20121009/en/ (accessed 9, October, 2012). [Green Version]
- Nash, J.; Nutt, D. Antidepressants. Psychiatry 2007, 6, 289–294. [Google Scholar] [CrossRef]
- Moussavi, S.; Chatterji, S.; Verdes, E.; Tandon, A.; Patel, V.; Ustun, B. Depression, chronic diseases, and decrements in health: Results from the World Health Surveys. Lancet 2007, 370, 851–858. [Google Scholar] [CrossRef]
- Rosua, J.L.; Blanca, G. Revisión del género Salvia L. (Lamiaceae) en el mediterráneo occidental: La sección de Salvia. Acta Bot. Malacit. 1986, 11, 227–272. [Google Scholar]
- Rodríguez-Hahn, L.; Cárdenas, J. Comparative chemotaxonomy in Labiatae. Curr. Top. Phytochem. 1999, 2, 91–102. [Google Scholar]
- Foster, S.; Tyler, VE. Tyler’s Honest Herbal: A sensible guide to the use of herbs and related remedies, 4th ed.; The Haworth Press: Binghamton, NY, USA, 2000; pp. 327–329 and 414–415. [Google Scholar]
- Steinegger, E.; Hänsel, R. Lehrbuch der Pharmakognosie und Phytopharmazie, 4th ed.; Springer-Verlag: Berlin-Heidelberg, Germany, 1988; pp. 343–345. [Google Scholar]
- Aguilar, A.; Camacho, J.R.; Chino, S.; Vázquez, P.; López, P. Herbario Medicinal del Instituto Mexicano del Seguro Social; Información. Etnobotánica: Distrito Federal, Mexico, 1994; p. 107. [Google Scholar]
- Topҫu, G. Bioactive triterpenoids from Salvia species. J. Nat. Prod. 2006, 69, 482–487. [Google Scholar] [CrossRef]
- Pereda-Miranda, R.; Delgado, G. Flavonoids from Salvia nicolsoniana. J. Nat. Prod. 1986, 49, 1160–1161. [Google Scholar] [CrossRef]
- Esquivel, B.; Vergara, F.; Matus, W.; Hernández-Ortega, S.; Ramírez-Apán, MT. Abietane diterpenoids from the roots of some Mexican Salvia species (Labiatae). Chemical diversity, phytogeographical significance and cytotoxic activity. Chem. Biodivers. 2005, 2, 738–747. [Google Scholar] [CrossRef]
- Jaime-Vasconcelos, M.A.; Frontana-Uribe, B.A.; Morales-Serna, J.A.; Salmón, M.; Cárdenas, J. Structure of salvioccidentalin, a diterpenoid with a rearranged neo-clerodane skeleton from Salvia occidentalis. Molecules 2011, 16, 9109–9115. [Google Scholar]
- Wake, G.; Court, J.; Pickering, A.; Lewis, R.; Wilkins, R.; Perry, E. CNS acetylcholine receptor activity in European medicinal plants traditionally used to improve failing memory. J. Ethnopharmacol. 2000, 69, 105–114. [Google Scholar] [CrossRef]
- Herrera-Ruiz, M.; García-Beltrán, Y.; Mora, S.; Díaz-Véliz, G.; Viana, GS.; Tortoriello, J.; Ramírez, G. Antidepressant and anxiolytic effects of hydroalcoholic extract from Salvia elegans. J. Ethnopharmacol. 2006, 107, 53–58. [Google Scholar] [CrossRef]
- Mora, S.; Millán, R.; Lungenstrass, H.; Díaz-Véliz, G.; Morán, JA.; Herrera-Ruiz, M.; Tortoriello, J. The hydroalcoholic extract of Salvia elegans induces anxiolytic- and antidepressant-like effects in rats. J. Ethnopharmacol. 2006, 106, 76–81. [Google Scholar] [CrossRef]
- Jiménez-Ferrer, E.; Hernández, F.H.; González-Cortazar, M.; Tortoriello, J.; Herrera-Ruiz, M. Antihypertensive activity of Salvia elegans Vahl. (Lamiaceae): ACE inhibition and angiotensin II antagonism. J. Ethnopharmacol. 2010, 130, 340–346. [Google Scholar] [CrossRef]
- Marquina, S.; García, Y.; Álvarez, L.; Tortoriello, J. 3-Acetoxy-7-methoxyflavone, a novel flavonoid from anxiolytic extract of Salvia elegans (Lamiaceae). Nat. Prod. Commun. 2008, 3, 185–188. [Google Scholar]
- Yueh-Hsiung, K.; Shu-Mei, L.; Jeng-Shiow, L. Constituents of the whole herb of Clinoponium laxiflorum. J. Chin. Chem. Soc. 2000, 47, 241–246. [Google Scholar]
- Wagner, H.; Hörhammer, L.; Aurnhammer, G.; Farkas, L. Strukturaufklärung und synthese des didymins, eines isosakuranetin-7-β-rutinosids aus Monarda didyma L. Chem. Ber. 1968, 101, 445–449. [Google Scholar] [CrossRef]
- Doganca, S.; Ulubelen, A.; Tuzlaci, E. Flavanones from Cyclotrichium niveum. Phytochemistry 1989, 28, 3561–3562. [Google Scholar] [CrossRef]
- González, M.; Zamilpa, A.; Marquina, S.; Navarro, V.; Álvarez, L. Antimycotic spriostanol saponins from Solanum hispidum leaves and their structure-activity relationships. J. Nat. Prod. 2004, 67, 938–941. [Google Scholar] [CrossRef]
- Porsolt, R.D.; Bertin, A.; Jalfre, M. Behavioral despair in mice: A primary screening test for antidepressants. Arch. Int. Pharmacodyn. Ther. 1977, 229, 327–336. [Google Scholar]
- Sample Availability: Samples of the compounds 1, 2 and 2a are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
González-Cortazar, M.; Maldonado-Abarca, A.M.; Jiménez-Ferrer, E.; Marquina, S.; Ventura-Zapata, E.; Zamilpa, A.; Tortoriello, J.; Herrera-Ruiz, M. Isosakuranetin-5-O-rutinoside: A New Flavanone with Antidepressant Activity Isolated from Salvia elegans Vahl. Molecules 2013, 18, 13260-13270. https://doi.org/10.3390/molecules181113260
González-Cortazar M, Maldonado-Abarca AM, Jiménez-Ferrer E, Marquina S, Ventura-Zapata E, Zamilpa A, Tortoriello J, Herrera-Ruiz M. Isosakuranetin-5-O-rutinoside: A New Flavanone with Antidepressant Activity Isolated from Salvia elegans Vahl. Molecules. 2013; 18(11):13260-13270. https://doi.org/10.3390/molecules181113260
Chicago/Turabian StyleGonzález-Cortazar, Manáses, Ana María Maldonado-Abarca, Enrique Jiménez-Ferrer, Silvia Marquina, Elsa Ventura-Zapata, Alejandro Zamilpa, Jaime Tortoriello, and Maribel Herrera-Ruiz. 2013. "Isosakuranetin-5-O-rutinoside: A New Flavanone with Antidepressant Activity Isolated from Salvia elegans Vahl." Molecules 18, no. 11: 13260-13270. https://doi.org/10.3390/molecules181113260
APA StyleGonzález-Cortazar, M., Maldonado-Abarca, A. M., Jiménez-Ferrer, E., Marquina, S., Ventura-Zapata, E., Zamilpa, A., Tortoriello, J., & Herrera-Ruiz, M. (2013). Isosakuranetin-5-O-rutinoside: A New Flavanone with Antidepressant Activity Isolated from Salvia elegans Vahl. Molecules, 18(11), 13260-13270. https://doi.org/10.3390/molecules181113260