Synthesis and Antibacterial Activities of Novel 4-Hydroxy-7-hydroxy- and 3-Carboxycoumarin Derivatives
Abstract
:1. Introduction
2. Results and Discussion
| MIC (μg/mL) a | ||||
|---|---|---|---|---|
| Gram (+) bacteria b | Gram (−) bacteria b | |||
| Compds. | B. s. | S. a. | E. c. | P. a. |
| 3a | >256 | >256 | >256 | >256 |
| 3a1 | >256 | >256 | >256 | >256 |
| 3a2 | 128 | >256 | >256 | >256 |
| 3b | 128 | >256 | 128 | >256 |
| 3b1 | 128 | 256 | 256 | >256 |
| 3b2 | >256 | >256 | >256 | >256 |
| 3c | 32 | 32 | >256 | >256 |
| 3c1 | 32 | >256 | >256 | >256 |
| 3c2 | >256 | >256 | >256 | >256 |
| 4a | 128 | >256 | >256 | >256 |
| 4a1 | >256 | >256 | >256 | >256 |
| 4a2 | 64 | >256 | >256 | >256 |
| 4b | 64 | 128 | 256 | >256 |
| 4b1 | >256 | >256 | >256 | >256 |
| 4b2 | 128 | >256 | >256 | >256 |
| 4c | 16 | 16 | 128 | >256 |
| 4c1 | >256 | >256 | >256 | >256 |
| 4c2 | 64 | 32 | >256 | >256 |
| 7d | 64 | 64 | >256 | >256 |
| 7e | >256 | >256 | >256 | >256 |
| 7f | 8 | 8 | >256 | >256 |
| 7g | >256 | >256 | >256 | >256 |
| 7h | >256 | >256 | >256 | >256 |
| 8i | >256 | >256 | >256 | >256 |
| 8j | >256 | >256 | >256 | >256 |
| 10k | >128 | >128 | 128 | >128 |
| 10l | >128 | >128 | >128 | >128 |
| 10m | >128 | >128 | 128 | >128 |
| 10n | >128 | >128 | >128 | >128 |
| 10o | >128 | >128 | >128 | >128 |
| 10p | >128 | >128 | >128 | >128 |
| 10q | >128 | >128 | >128 | >128 |
| 10r | >128 | >128 | >128 | >128 |
| 11k | >128 | >128 | >128 | >128 |
| 14 | >128 | >128 | >128 | >128 |
| 15 | >128 | >128 | >128 | >128 |
| 16 | >128 | >128 | >128 | >128 |
| 17 | >128 | >128 | >128 | >128 |
| 18 | >128 | >128 | >128 | >128 |
| 19 | >128 | >128 | >128 | >128 |
| 20 | >128 | >128 | >128 | >128 |
| 21 | >128 | >128 | >128 | >128 |
| PenicillinG c | 8 | 0.0625 | - | - |
| Amikin | - | - | 1 | 0.125 |
2.1. Chemistry
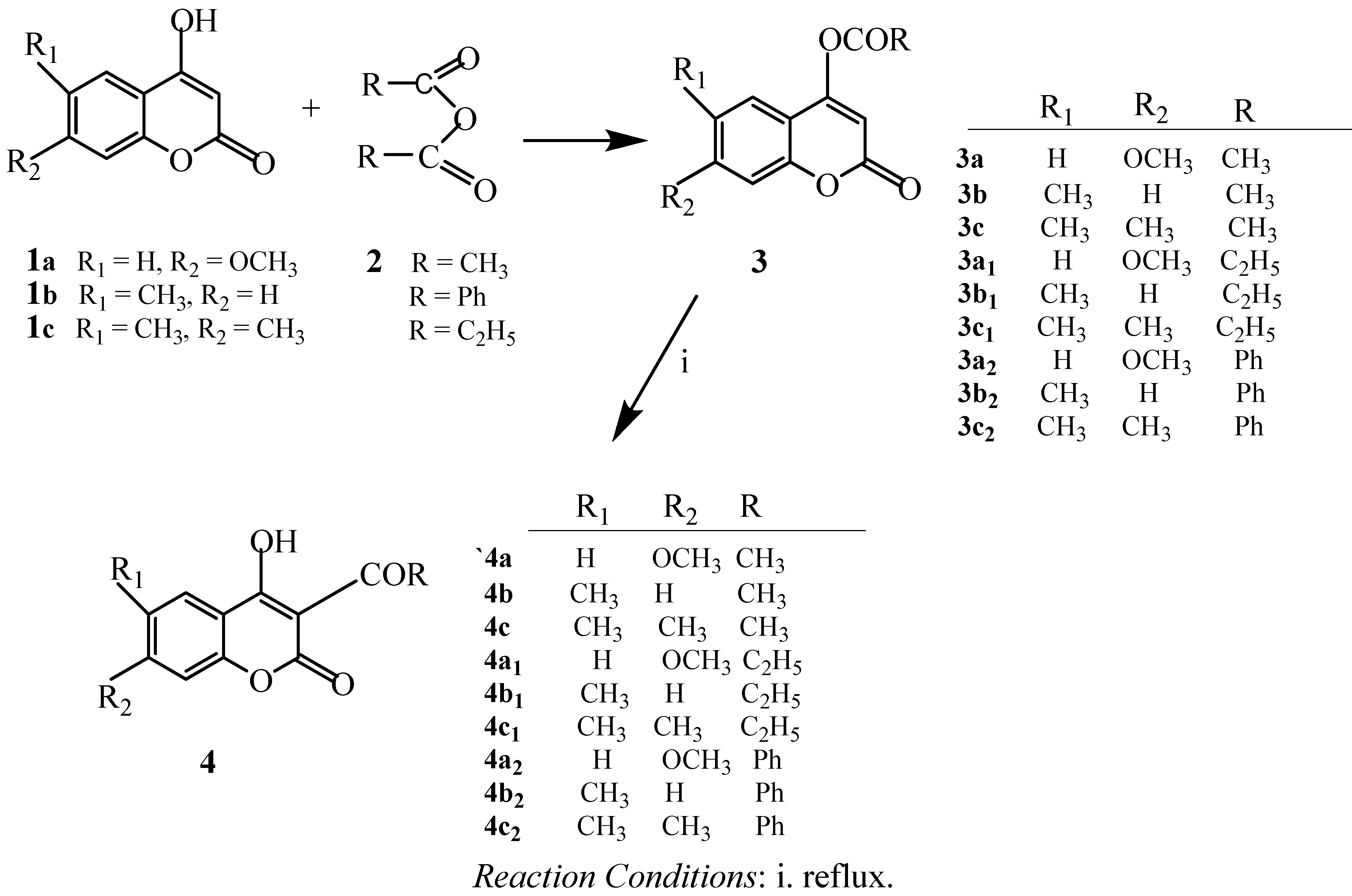
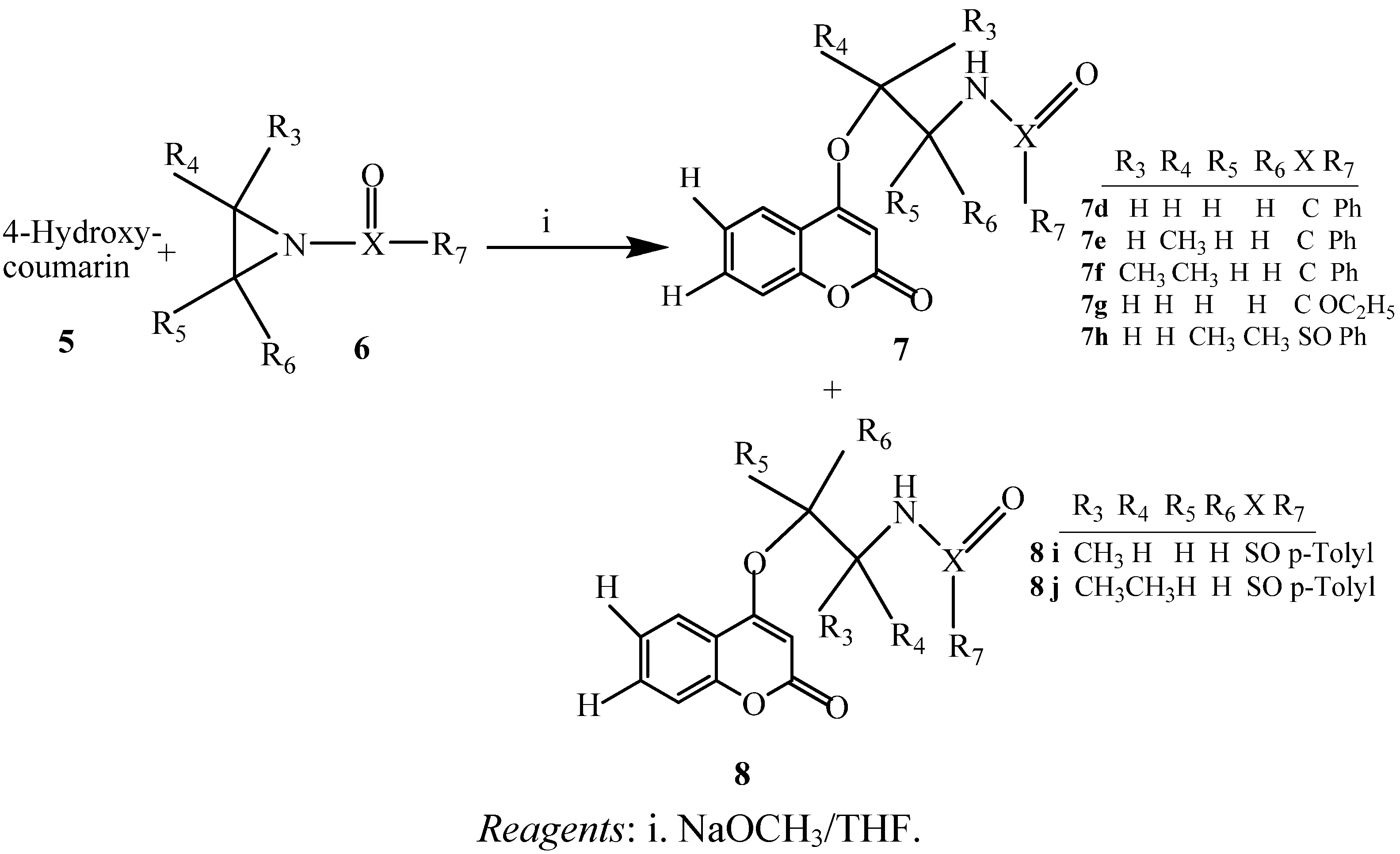
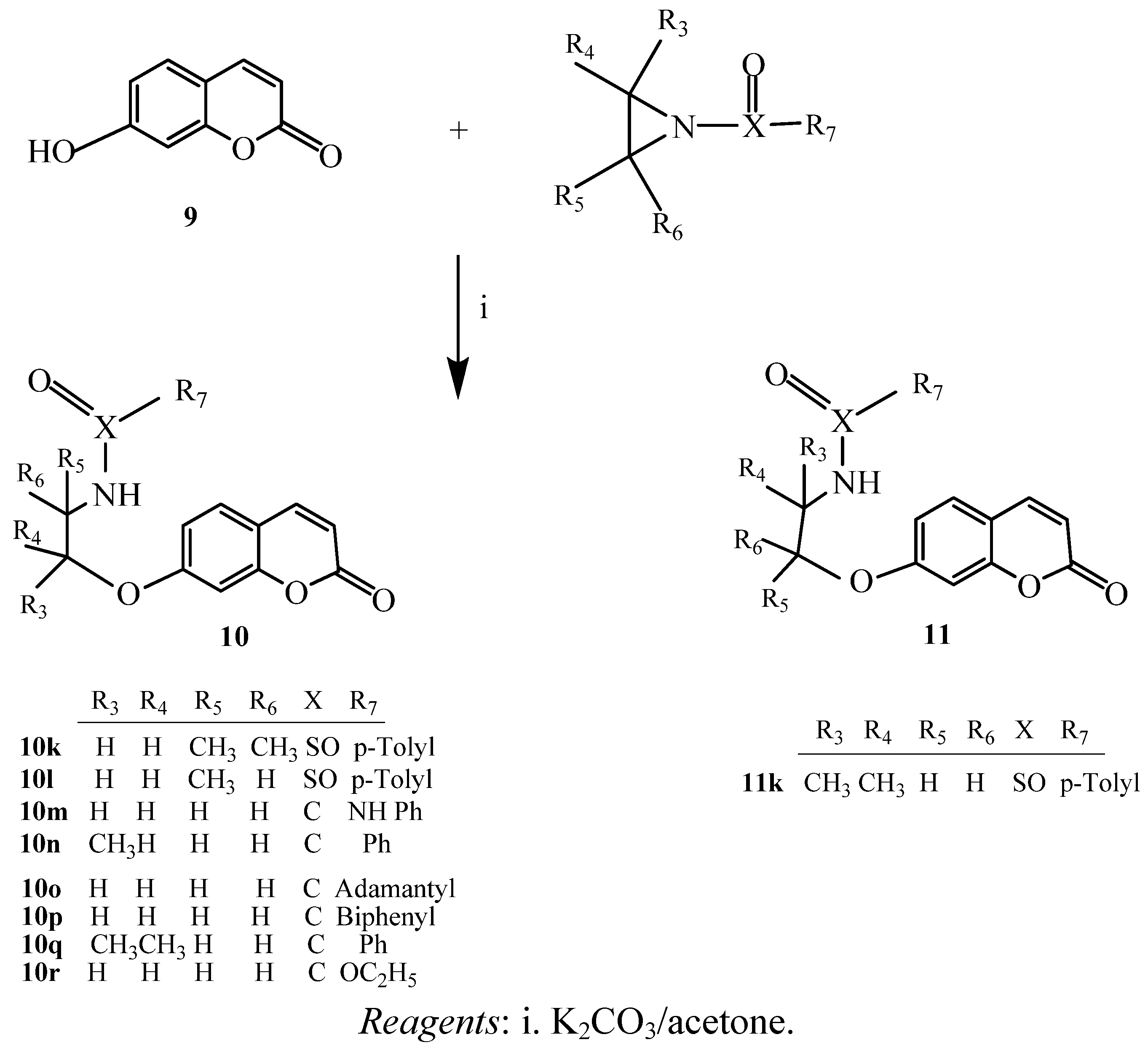
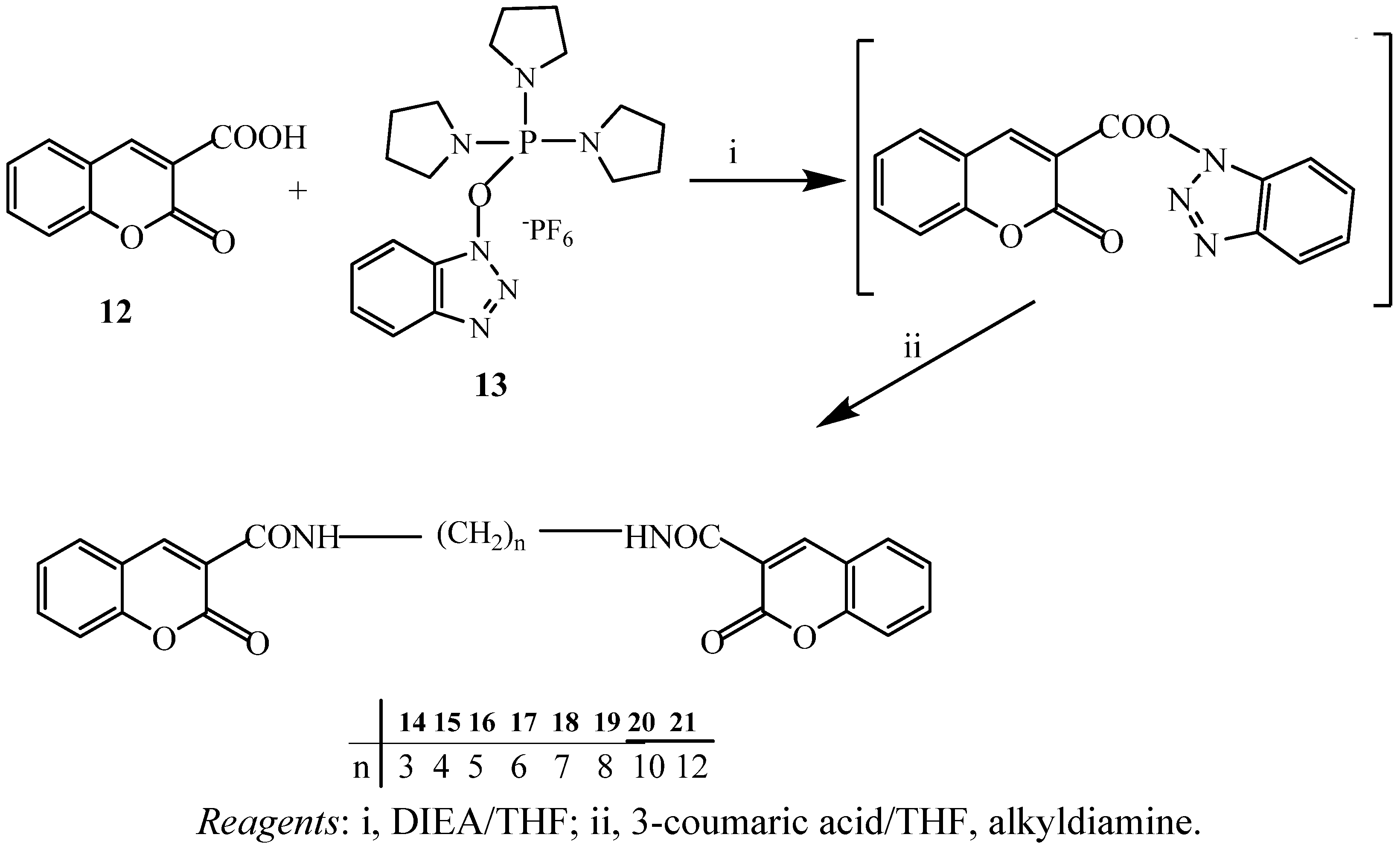
2.2. Antibacterial Activity Test
3. Experimental
3.1. General
3.2. Preparation of Acylcoumarins
3.3. Preparation of 4-hydroxy-, and 7-hydroxycoumarin Derivatives via Ring Opening of Activated Aziridines
3.4. Preparation of Coumarin Amide Dimers
4. Conclusions
Acknowledgments
References
- Dastan, D.; Salehi, P.; Gohari, A.R.; Zimmermann, S.; Kaiser, M.; Hamburger, M.; Khavasi, H.R.; Ebrahimi, S.N. Disesquiterpene and sesquiterpene coumarins from Ferula pseudalliacea, and determination of their absolute configurations. Phytochemistry 2012, 78, 170–178. [Google Scholar] [CrossRef]
- Verma, P.; Pal, H. Intriguing H-aggregate and H-dimer formation of coumarin-481 dye in aqueous solution as evidenced from photophysical studies. J. Phys. Chem. A 2012, 116, 4473–4484. [Google Scholar] [CrossRef]
- Stoianov, S.V.; Robinson, H.D. Two-Photon Activated Two-Photon Fluorescence and Binding of Azidocoumarin in a Gelatin Matrix. J. Fluoresc. 2012, 22, 1291–1300. [Google Scholar] [CrossRef]
- Manevich, Y.; Held, K.D.; Biaglow, J.E. Coumarin-3-carboxylic acid as a detector for hydroxyl radicals generated chemically and by gamma radiation. Radiat. Res. 1997, 148, 580–591. [Google Scholar] [CrossRef]
- Xing, Z.; Fu, Y.; Zhou, J.; Zhu, C.; Cheng, Y. Coumarin-based chiral fluorescence sensor incorporating a thiourea unit for highly enantioselective recognition of N-Boc-protected proline. Org. Biomol. Chem. 2012, 10, 4024–4028. [Google Scholar]
- Barooah, N.; Mohanty, J.; Pal, H.; Bhasikuttan, A.C. Non-covalent interactions of coumarin dyes with cucurbit[7]uril macrocycle: modulation of ICT to TICT state conversion. Org. Biomol. Chem. 2012, 10, 5055–5062. [Google Scholar] [CrossRef]
- Chung, J.W.; Lee, K.; Neikirk, C.; Nelson, C.M.; Priestley, R.D. Photoresponsive coumarin-stabilized polymeric nanoparticles as a detectable drug carrier. Small 2012, 8, 1693–1700. [Google Scholar] [CrossRef]
- Li, H.; Xiao, Y.; Niu, J.; Chen, X.; Ping, Q. Preparation of a cationic nanoemulsome for intratumoral drug delivery and its enhancing effect on cellular uptake in vitro. J. Nanosci. Nanotechnol. 2011, 11, 8547–8555. [Google Scholar] [CrossRef]
- Dahl, O.E. New oral antithrombotics: Focus on dabigatran, an oral, reversible direct thrombin inhibitor for the prevention and treatment of venous and arterial thromboembolic disorders. Vasc. Health Risk Manag. 2012, 8, 45–57. [Google Scholar]
- Penning-Van Beest, F.J.; Koerselman, J.; Herings, R.M. Risk of major bleeding during concomitant use of antibiotic drugs and coumarin anticoagulants. J. Thromb. Haemost. 2008, 6, 284–290. [Google Scholar]
- Kontogiorgis, C.; Hadjipavlou-Litina, D. Biological evaluation of several coumarin derivatives designed possible anti-inflammatory/antioxidant agents. J. Enzyme Inhib. Med. Chem. 2003, 18, 63–69. [Google Scholar] [CrossRef]
- Liu, Z.Q.; Yu, W.; Liu, Z.L. Antioxidative and prooxidative effects of coumarin derivatives on free radical initiated and photosensitized peroxidation of human low-density lipoprotein. Chem. Phys. Lipids 1999, 103, 125–135. [Google Scholar] [CrossRef]
- Hamdi, N.; Passarelli, V.; Romerosa, A. Synthesis, spectroscopy and electrochemistry of new 4-(4-acetyl-5-substituted-4,5-dihydro-1,3,4-oxadiazol-2-yl)methoxy)-2H-chromen-2-ones as a novel class of potential antibacterial and antioxidant derivatives. C. R. Chim. 2011, 14, 548–555. [Google Scholar] [CrossRef]
- Hamdi, N.; Bouabdallah, F.; Romerosa, A. Expedious synthesis for α, β unsaturated coumarin derivatives using boran chelates: A novel class of potential antibacterial and antioxidant agents. C. R. Chim. 2010, 13, 1261–1268. [Google Scholar] [CrossRef]
- Hamdi, N.; Fischmeister, C.; Puerta, M.C.; Valerga, P. A rapid access to new coumarinyl chalcone and substituted chromen[4,3-c] pyrazol-4(1H)-ones and their antibacterial and DPPH radical scavenging activities. Med. Chem. Res. 2011, 20, 522–530. [Google Scholar] [CrossRef]
- Finn, G.J.; Kenealy, E.; Creaven, B.S.; Egan, D.A. In vitro cytotoxic potential and mechanism of action of selected coumarins, using human renal cell lines. Cancer Lett. 2002, 183, 61–68. [Google Scholar] [CrossRef]
- Jimenez-Orozco, F.A.; Lopez-Gonzalez, J.S.; Nieto-Rodriguez, A. Decrease of cyclin D1 in the human lung adenocarcinoma cell line A-427 by 7-hydroxycoumarin. Lung Cancer 2001, 34, 185–194. [Google Scholar] [CrossRef]
- Weber, U.S.; Steffen, B.; Siegers, C.P. Antitumor-activities of coumarin, 7-hydroxy-coumarin and its glucuronide in several human tumor cell lines. Res. Commun. Mol. Pathol. Pharmacol. 1998, 99, 193–206. [Google Scholar]
- Singh, R.K.; Lange, T.S.; Kim, K.K.; Brard, L. A coumarin derivatives (RKS262) inhibits cell-cycle progression, causes pro-apoptotic signaling and cytotoxicity in ovarian cancer cells. Invest. New Drugs 2011, 29, 63–72. [Google Scholar]
- Sabry, N.M.; Mohamed, H.M.; Khattab, E.S.; Motlaq, S.S.; El-Agrody, A.M. Synthesis of 4H-chromene, coumarin, 12H-chromeno[2,3-d]pyrimidine derivatives and some of their antimicrobial and cytotoxicity activitie. Eur. J. Med. Chem. 2011, 46, 765–772. [Google Scholar]
- Musa, M.A.; Badisa, V.L.; Latinwo, L.M.; Cooperwood, J.; Sinclari, A.; Abdullah, A. Cytotoxic activity of new acetoxycoummarin derivatives in cancer cell lines. Anticancer Res. 2011, 31, 2017–2022. [Google Scholar]
- Bronikowska, J.; Szliszka, E.; Jaworska, D.; Czuba, Z.P.; Krol, W. The Coumarin Psoralidin Enhances Anticancer Effect of Tumor Necrosis Factor-Related Apoptosis-Inducing Ligand (TRAIL). Molecules 2012, 17, 6449–6464. [Google Scholar] [CrossRef]
- Roma, G.; di Braccio, M.; Grossi, G.; Piras, D.; Leoncini, G.; Bruzzese, D.; Signorello, M.G.; Fossa, P.; Mosti, L. Synthesis and in vitro antiplatelet activity of new 4-(1-piperazinyl)coumarin derivatives. Human platelet phosphodiesterase 3 inhibitory properties of the two most effective compounds described and molecular modeling study on their interactions with phosphodiesterase 3A catalytic site. J. Med. Chem. 2007, 12, 2886–2895. [Google Scholar]
- Dekić, B.R.; Radulović, N.S.; Dekić, V.S.; Vukićević, R.D.; Palić, R.M. Synthesis and antimicrobial activity of new 4-heteroarylamino coumarin derivatives containing nitrogen and sulfur as heteroatoms. Molecules 2010, 15, 2246–2256. [Google Scholar] [CrossRef]
- Chimenti, F.; Bizzarri, B.; Bolasco, A.; Secci, D.; Chimenti, P.; Granese, A.; Carradori, S.; Rivanera, D.; Zicari, A.; Scaltrito, M.M.; et al. Synthesis, selective anti-Helicobacter pylori activity, and cytotoxicity of novel N-substituted-2-oxo-2H-1-benzopyran-3-carboxamides. Bioorg. Med. Chem. Lett. 2010, 20, 4922–4926. [Google Scholar]
- Kulkarni, A.; Patil, S.A.; Badami, P.S. Synthesis, characterization, DNA cleavage and in vitro antimicrobial studies of La(III), Th(IV) and VO(IV) complexes with Schiff bases of coumarin derivatives. Eur. J. Med. Chem. 2009, 44, 2904–2912. [Google Scholar] [CrossRef]
- Zavrsnik, D.; Muratovic, S.; Spirtovic, S.; Softic, D.; Medic-Saric, M. The synthesis and antimicrobial activity of some 4-hydroxycoumarin derivatives. Bosn. J. Basic Med. Sci. 2008, 8, 277–281. [Google Scholar]
- Siddiqui, Z.N.; Musthafa, T.N.M.; Ahmad, A.; Khan, A.U. Synthesis of 4-hydroxycoumarin heteroarylhybrids as potential antimicrobial agents. Arch. Pharm. (Weinheim) 2011, 344, 394–401. [Google Scholar] [CrossRef]
- Basanagouda, M.; Shivashankar, K.; Kulkarni, M.V.; Rasal, V.P.; Patel, H.; Mutha, S.S.; Mohite, A.A. Synthesis and antimicrobial studies on novel sulfonamides containing 4-azidomethyl coumarin. Eur. J. Med. Chem. 2010, 45, 1151–1157. [Google Scholar] [CrossRef]
- Rehman, S.U.; Chohan, Z.H.; Glunaz, F.; Supuran, C.T. In-vitro antibacterial, antifungal and cytotoxic activities of some coumarins and their metal complexes. J. Enzyme Inhib. Med. Chem. 2005, 20, 333–340. [Google Scholar] [CrossRef]
- Guo, Q.; Liu, M.L.; Feng, L.S.; Lv, K.; Guan, Y.; Guo, H.Y.; Xiao, C.L. Synthesis and in vitro antimycobacterial activity of fluoroquinolone derivatives containing a coumarin moiety. Arch. Pharm. (Weinheim) 2011, 344, 802–809. [Google Scholar] [CrossRef]
- Chiang, C.C.; Cheng, M.J.; Peng, C.F.; Huang, H.Y.; Chen, I.S. A novel dimeric coumarin analog and antimycobacterial constituents from Fatoua pilosa. Chem. Biodivers. 2010, 7, 1728–1736. [Google Scholar] [CrossRef]
- Upadhyay, K.; Bavishi, A.; Thakrar, S.; Radadiya, A.; Vala, H.; Parekh, S.; Bhavsar, D.; Savant, M.; Parmar, M.; Adlakha, P.; et al. Synthesis and biological evaluation of 4-styrylcoumarin derivatives as inhibitors of TNF-α and IL-6 with anti-tubercular activity. Bioorg. Med. Chem. Lett. 2011, 21, 2547–2549. [Google Scholar]
- Kampranis, S.C.; Gormley, N.A.; Tranter, R.; Orphanides, G.; Maxwell, A. Probing of Coumarins and Cyclothialidines to DNA Gyrase. Biochemistry 1999, 38, 1967–1976. [Google Scholar] [CrossRef]
- Wang, H.X.; Ng, T.B. Examination of lectins, polysaccharopeptide, polysaccharide, alkaloid, coumarin and trypsin inhibitors for inhibitory activity against human immunodeficiency virus reverse transcriptase and glycohydrolases. Planta Med. 2001, 67, 669–672. [Google Scholar] [CrossRef]
- Maoa, P.C.; Mouscadet, J.F.; Leh, H.; Auclair, C.; Hsu, L.Y. Chemical modification of coumarin dimmer and HIV-1 integrase inhibitory activity. Chem. Pharm. Bull. 2002, 50, 1634–1637. [Google Scholar] [CrossRef]
- Le, B.G.; Radanyi, C.; Peyrat, J.F.; Brion, J.D.; Alami, M.; Marsaud, V.; Stella, B.; Renoir, J.M. New novobiocin analogues as antiproliferative agents in breast cancer cells and potential inhibitors of heat shock protein90. J. Med. Chem. 2007, 50, 6189–6200. [Google Scholar] [CrossRef]
- Lin, L.H.; Lee, L.W.; Sheu, S.Y.; Lin, P.Y. Study on the stevioside analogues of steviolbioside, steviol, and isosteviol 19-alkyl amide dimers: synthesis and cytotoxic and antibacterial activity. Chem. Pharm. Bull. 2004, 52, 1117–1122. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 14–21 are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lin, P.-Y.; Yeh, K.-S.; Su, C.-L.; Sheu, S.-Y.; Chen, T.; Ou, K.-L.; Lin, M.-H.; Lee, L.-W. Synthesis and Antibacterial Activities of Novel 4-Hydroxy-7-hydroxy- and 3-Carboxycoumarin Derivatives. Molecules 2012, 17, 10846-10863. https://doi.org/10.3390/molecules170910846
Lin P-Y, Yeh K-S, Su C-L, Sheu S-Y, Chen T, Ou K-L, Lin M-H, Lee L-W. Synthesis and Antibacterial Activities of Novel 4-Hydroxy-7-hydroxy- and 3-Carboxycoumarin Derivatives. Molecules. 2012; 17(9):10846-10863. https://doi.org/10.3390/molecules170910846
Chicago/Turabian StyleLin, Pen-Yuan, Kuang-Sheng Yeh, Chien-Ling Su, Shiow-Yunn Sheu, Tiffany Chen, Keng-Liang Ou, Mei-Hsiang Lin, and Lin-Wen Lee. 2012. "Synthesis and Antibacterial Activities of Novel 4-Hydroxy-7-hydroxy- and 3-Carboxycoumarin Derivatives" Molecules 17, no. 9: 10846-10863. https://doi.org/10.3390/molecules170910846
APA StyleLin, P.-Y., Yeh, K.-S., Su, C.-L., Sheu, S.-Y., Chen, T., Ou, K.-L., Lin, M.-H., & Lee, L.-W. (2012). Synthesis and Antibacterial Activities of Novel 4-Hydroxy-7-hydroxy- and 3-Carboxycoumarin Derivatives. Molecules, 17(9), 10846-10863. https://doi.org/10.3390/molecules170910846




