Identification and Determination of Aconitum Alkaloids in Aconitum Herbs and Xiaohuoluo Pill Using UPLC-ESI-MS
Abstract
:1. Introduction
2. Results and Discussion
2.1. LC-MS Fingerprints
2.1.1. UPLC-ESI-MS Fingerprint Analysis of Aconitum Herbs and Processed Products
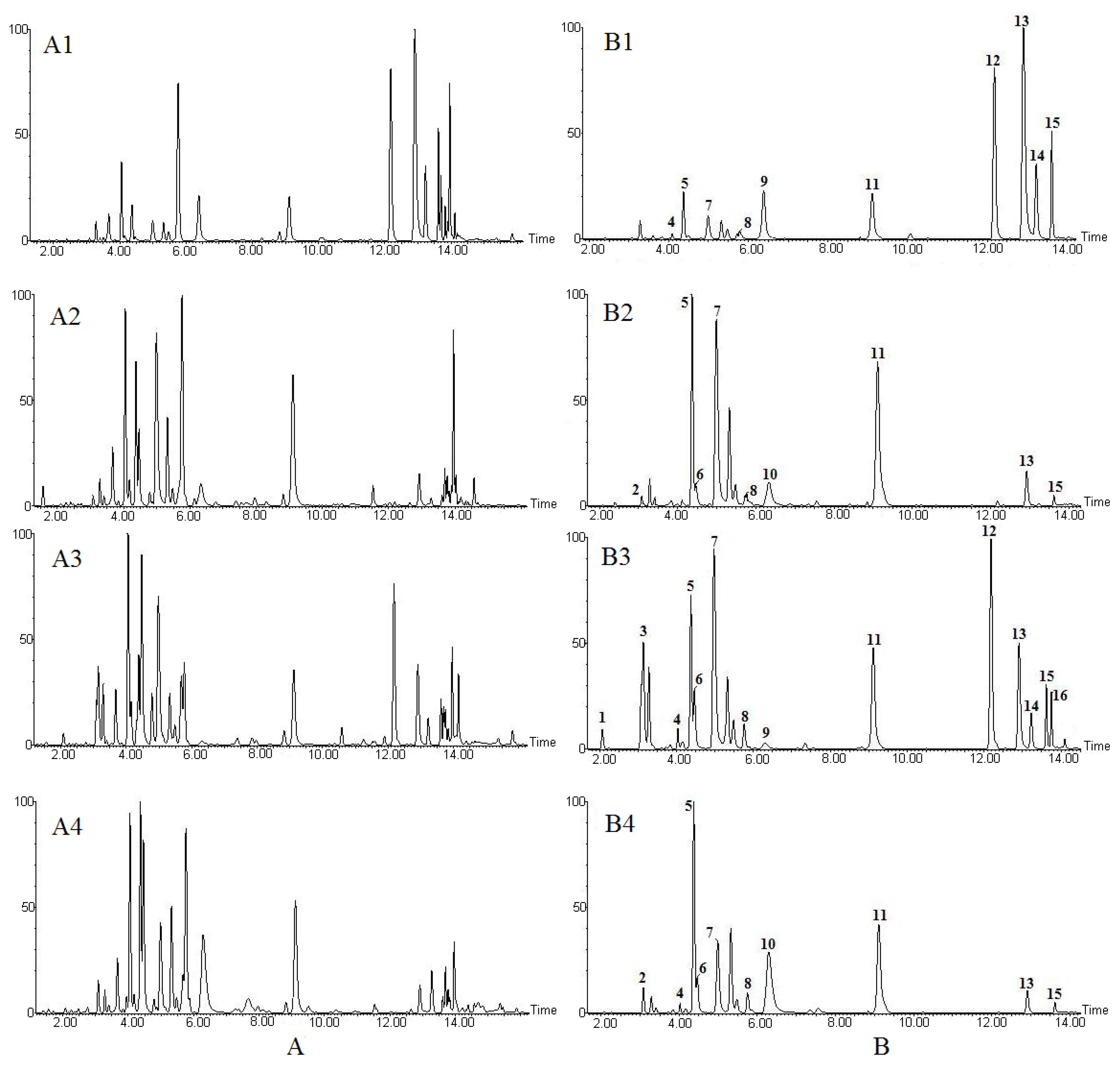
| Peak No. | tR (min) | m/z | Molecular Formula | Identification | VIP |
|---|---|---|---|---|---|
| 1 | 2.07 | 408.4 | C23H37NO6 | cammaconine b | 3.91 |
| 2 | 3.08 | 424.4 | C23H37NO6 | senbusine A b | 5.55 |
| 3 | 3.14 | 378.4 | C22H35NO4 | aconosine b | 7.62 |
| 4 | 4.04 | 454.5 | C24H39NO7 | delcisine b | 6.43 |
| 5 | 4.37 | 408.4 | C23H37NO6 | isotalatizidine b | 7.37 |
| 6 | 4.45 | 422.5 | C24H39NO5 | talatizamine b | 9.46 |
| 7 | 5.00 | 590.7 | C31H43NO10 | benzoylmesaconitine a | 9.29 |
| 8 | 5.76 | 438.5 | C24H39NO6 | neoline b | 8.21 |
| 9 | 6.34 | 360.4 | C22H33NO3 | lepenine b | 5.68 |
| 10 | 6.38 | 574.7 | C31H43NO9 | benzoylhypaconitine a | 6.63 |
| 11 | 9.04 | 358.3 | C22H31NO3 | songorine b | 5.33 |
| 12 | 12.18 | 632.7 | C33H45NO11 | mesaconitine a | 9.12 |
| 13 | 12.91 | 616.8 | C33H45NO10 | hypaconitine a | 8.8 |
| 14 | 13.22 | 646.8 | C34H47NO11 | aconitine a | 5.59 |
| 15 | 13.61 | 630.8 | C34H47NO10 | deoxyaconitine b | 7.94 |
| 16 | 13.75 | 402.4 | C24H35NO4 | lucidusculine b | 3.59 |
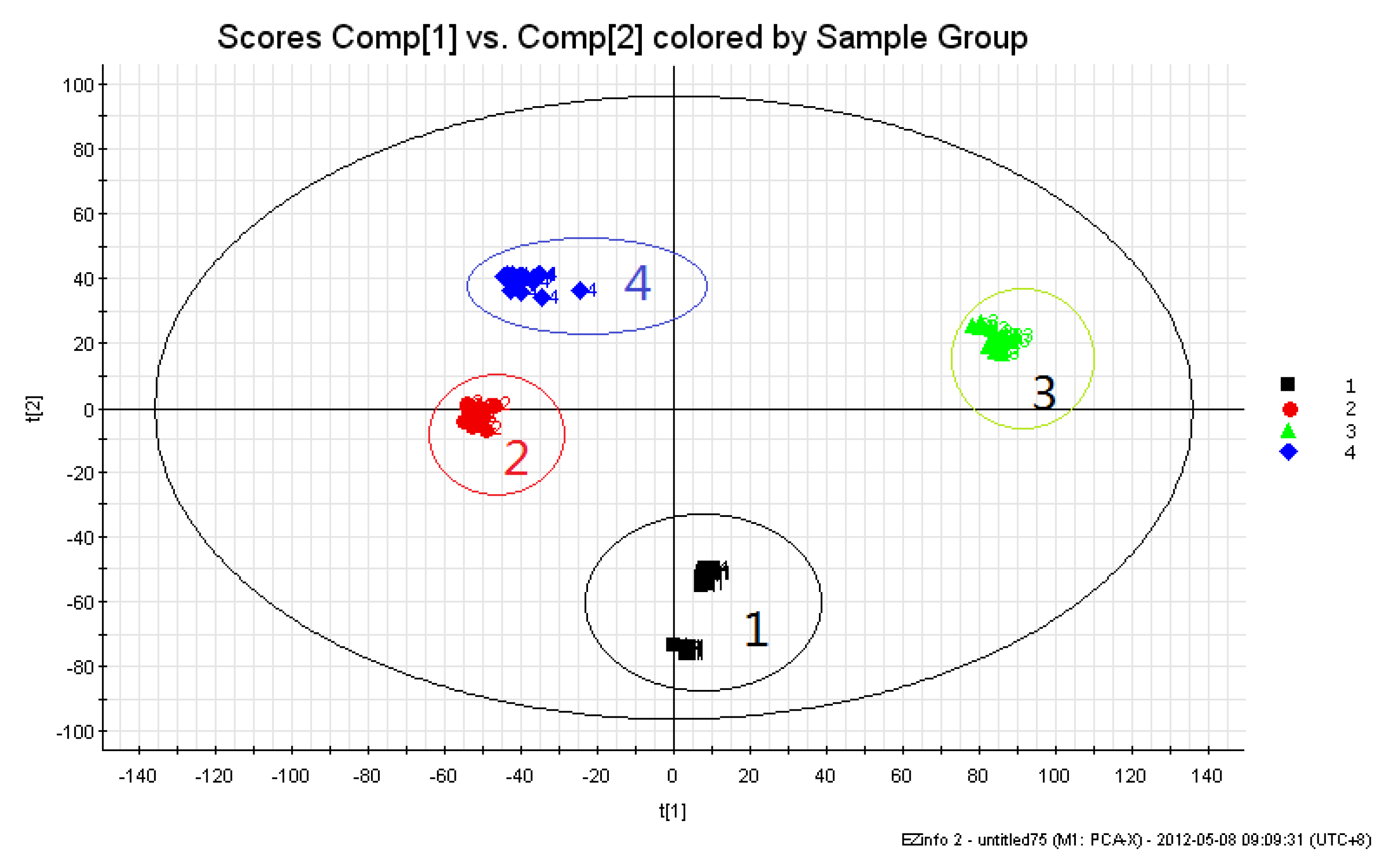
2.1.2. Changes in Chemical Components of Aconitum Herbs and Processed Products
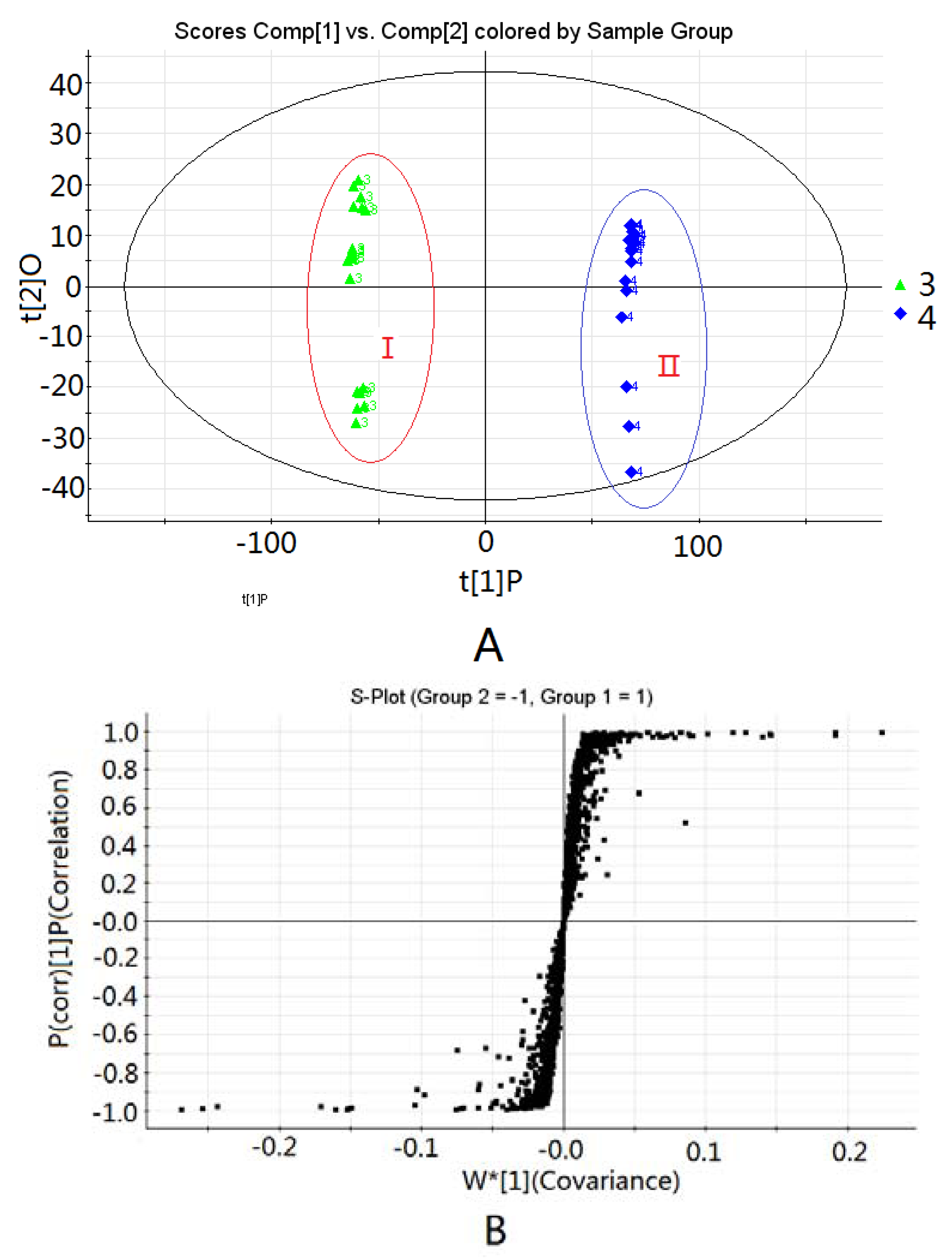
| Peak No. | tR (min) | Identification | VIP |
|---|---|---|---|
| 13 | 12.91 | hypaconitine | 13.70 |
| 12 | 12.18 | mesaconitine | 12.93 |
| 15 | 13.61 | deoxyaconitine | 12.43 |
| 7 | 5.00 | benzoylmesaconitine | 11.41 |
| 4 | 4.04 | delcisine | 9.75 |
| 5 | 4.37 | isotalatizidine | 9.75 |
| 14 | 13.22 | aconitine | 8.71 |
| 9 | 6.34 | lepenine | 7.74 |
2.1.3. Chemical Fingerprint of Xiaohuoluo Pill
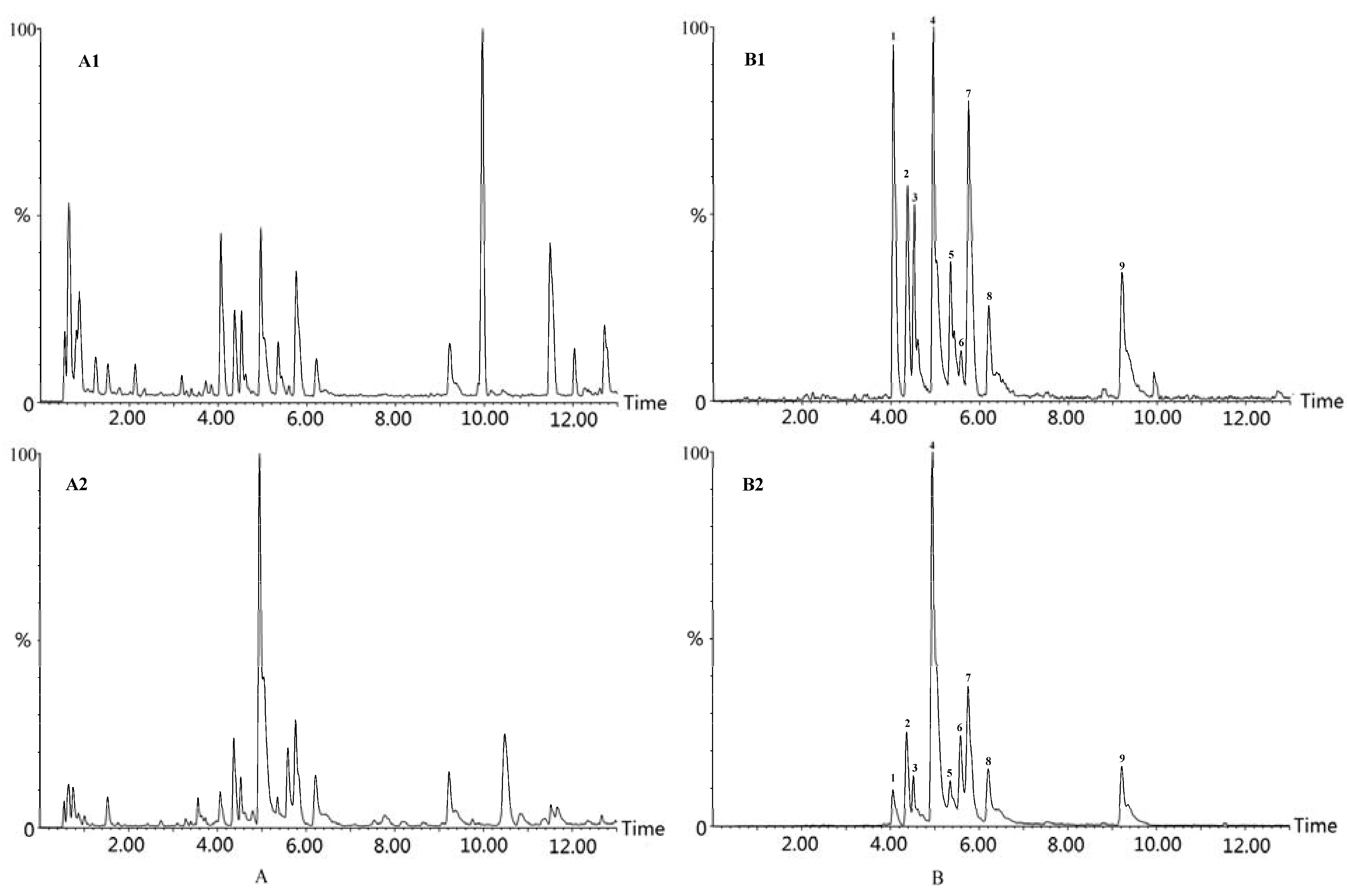
2.2. Quantitative Analysis of Xiaohuoluo Pill
2.2.1. Optimization of UPLC-MS Conditions
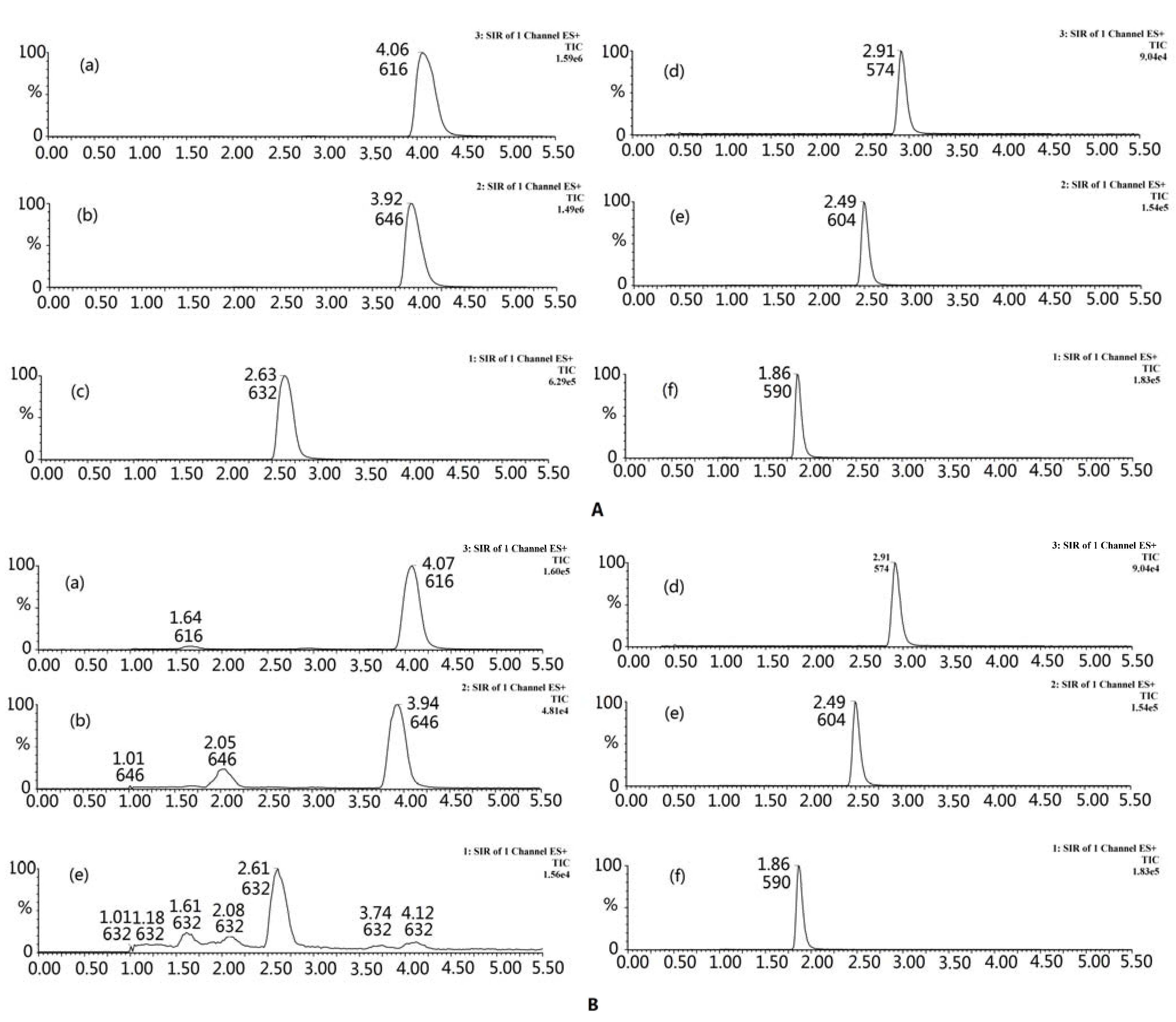
2.2.2. Method Validation: Linearity, Limits of Detection, and Quantification
| Analyte | Regressive equation a | r | Linear range (ng·mL−1) | LLOD a (ng·mL−1) | LLOQ b (ng·mL−1) |
|---|---|---|---|---|---|
| MA | y = 135.01x + 309.64 | 0.9993 | 1.41–501.00 | 0.45 | 1.41 |
| AC | y = 54.337x – 21.315 | 0.9999 | 1.20–396.80 | 0.39 | 1.20 |
| HA | y =148.34x – 130.21 | 0.9999 | 1.92–533.18 | 0.65 | 1.92 |
| BMA | y = 6385.8x + 1390.3 | 0.9984 | 1004.00–20080.00 | 0.82 | 4.28 |
| BAC | y = 15.025x + 314.98 | 0.9989 | 1.99–2490.00 | 0.54 | 1.99 |
| BHA | y = 17.273x + 129.08 | 0.9993 | 2.02–1518.00 | 0.51 | 2.02 |
2.2.3. Validation: Precision, Repeatability, Stability, and Recovery
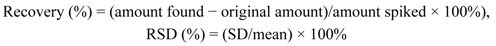
| Compound | Precision RSD (%) (n = 6) | Reproducibility RSD (%) (n = 6) | Stability RSD (%) (n = 6) | Recovery (%) (n = 9)Mean ± RSD (%) |
|---|---|---|---|---|
| MA | 2.40 | 2.26 | 1.76 | 99.8 ± 2.49 |
| AC | 2.36 | 3.78 | 1.42 | 100.9 ± 2.65 |
| HA | 2.82 | 1.79 | 1.11 | 101.7 ± 1.94 |
| BMA | 3.11 | 2.56 | 1.44 | 100.3 ± 1.06 |
| BAC | 3.07 | 3.07 | 1.05 | 99.7 ± 1.32 |
| BHA | 2.89 | 2.50 | 3.09 | 98.1 ± 1.08 |
2.2.4. Sample Analysis
| Analytes/Contents (μg·g−1) | ||||||
|---|---|---|---|---|---|---|
| Samples | MA | AC | HA | BMA | BAC | BHA |
| S-01 | 0.31 ± 0.01 | 7.55 ± 0.16 | 8.05 ± 0.10 | 295.20 ± 6.20 | 88.37 ± 2.16 | 45.45 ± 0.55 |
| S-02 | N.D. a | 0.18 ± 0.003 | 0.67 ± 0.01 | 327.18 ± 7.71 | 62.10 ± 0.53 | 54.75 ± 2.03 |
| S-03 | 0.60 ± 0.02 | 0.78 ± 0.01 | 5.22 ± 0.12 | 343.38 ± 1.03 | 27.46 ± 0.21 | 52.02 ± 0.23 |
| S-04 | 0.02 ± 0.002 | 0.07 ± 0.01 | 0.13 ± 0.01 | 28.52 ± 2.32 | 3.57 ± 0.29 | 3.11 ± 0.24 |
| S-05 | 0.24 ± 0.004 | 2.15 ± 0.02 | 2.16 ± 0.02 | 55.71 ± 0.82 | 11.12 ± 0.13 | 9.49 ± 0.01 |
| S-06 | 0.11 ± 0.003 | 0.13 ± 0.01 | 0.35 ± 0.01 | 14.71 ± 0.80 | 1.28 ± 0.05 | 1.34 ± 0.08 |
| S-07 | 0.18 ± 0.003 | 0.80 ± 0.004 | 0.96 ± 0.01 | 117.80 ± 8.21 | 13.31 ± 1.09 | 11.61 ± 0.60 |
| S-08 | 0.19 ± 0.002 | 0.84 ± 0.01 | 0.70 ± 0.01 | 137.56 ± 4.35 | 16.47 ± 0.40 | 14.34 ± 0.44 |
| S-09 | 0.19 ± 0.002 | 0.83 ± 0.01 | 0.99 ± 0.01 | 129.54 ± 1.43 | 15.33 ± 0.01 | 13.36 ± 0.01 |
3. Experimental
3.1. Chemicals and Reagents
3.2. Materials
| Name | No. | Source | Batch No. |
|---|---|---|---|
| Concentrated pill | S-01 | Foci, Lanzhou | 10B5 |
| S-02 | Taibao, Lanzhou | 62101204 | |
| S-03 | Dalu, Shandong | 110101 | |
| Honey pill | S-04 | Wohua, Shandong | 110302 |
| S-05 | Shiyitang, Harbin | 1101147 | |
| S-06 | Tongrentang, Beijing | 0013070 | |
| S-07 | Hongjitang, Jinan | 1011001 | |
| S-08 | Hongjitang, Jinan | 1104001 | |
| S-09 | Hongjitang, Jinan | 1104002 |
3.3. UPLC-ESI-MS Conditions
3.3.1. UPLC-MS Qualitative Conditions
3.3.2. UPLC-MS Quantitative Conditions
3.4. Preparation of Standard and Quality Control Samples
3.5. Preparation of Sample Solution
3.5.1. Xiaohuoluo Pill
3.5.2. Aconitum Herbs and Processed Products
4. Conclusions
Acknowledgments
References
- The State Pharmacopoeia Committee of People’s Republic of China, Pharmacopoeia of People’s Republic of China; Chemical Industry Press: Beijing, China, 2010; 1, pp. 36–38, 220–221, 509.
- Kita, Y.; Tto, M. Nuclear ribosomal ITS sequences and phylogeny in East Asian Aconitum subgenus Aconitum (Ranunculaceae), with special reference to extensive polymorphism in individual plants. Plant. Syst. Evol. 2000, 225, 1–13. [Google Scholar] [CrossRef]
- Wang, F.P. Chemical of Alkaloids; Chemical Industry Publishing: Beijing, China, 2008. [Google Scholar]
- Chen, Y.; Chu, Y.L.; Chu, J.H. Alkaloids of the Chinese drugs, Aconitum Spp-IX. Alkaloids from Chuan-wu and Fu-zi, Aconitum Carmichaelidebx. Acta Phram. Sin. 1956, 12, 435–439. [Google Scholar]
- Li, Z.B.; Lu, G.H.; Chen, D.L.; Wang, F.P. Chemical study on the alkaloids of “Cao Wu”. Nat. Prod. Res. Dev. 1997, 1, 9–14. [Google Scholar]
- Yun-Choi, H.S.; Park, K.M.; Chang, K.C.; Lee, D.H. Antithrombotic Effects of YS-49 and YS-51-1-Naphthylmethyl Analogs of Hi genamine. Thromb. Res. 2001, 104, 249–255. [Google Scholar] [CrossRef]
- Shim, S.H.; Kim, J.S.; Son, K.H.; Bae, K.H.; Kang, S.S. Alkaloids from the Roots of Aconitum pseudo-lave var. erectum. J. Nat. Prod. 2006, 69, 400–402. [Google Scholar]
- Barbosa-Filho, J.M.; Piuvezam, M.R.; Moura, D.M.; Sliva, M.S. Anti-inflammatory activity of alkaloids: A twenty-century review. Rev. Bras. Farmacogn. 2006, 16, 109. [Google Scholar] [CrossRef]
- Li, H.Y.; Huang, C.Y.; Ye, Z.J. Experimental study on anti-inflammation and analgesia effect from Aconitum carmichaeli Debx. Chin. J. Curr. Prac. Med. 2006, 6, 27–29. [Google Scholar]
- Zeng, J.; Luo, X. Study on tumor-inhibitory effect of Chuan Wu aqueous extract. J. Sichuan U. 2007, 16, 1345–1348. [Google Scholar]
- De lnés, C.; Reina, M.; Gavín, J.A.; González-Coloma, A. In vitro cytotoxicity of norditerpenoid alkaloids. Z. Natllrforsch C. 2006, 61, 11–18. [Google Scholar]
- Xu, T.H.; Zhao, H.F.; X, Y.J.; Xie, S.X. Cardiotonic constituents of Aconitum carmichaeli. Chin. Tradit. Herb. Drugs 2004, 9, 964. [Google Scholar]
- Liu, W.H.; Zhou, Y.P.; Zeng, G.W. Effects of DL-Demethyloclaurine on experiment heart failure. Acta Phram. Sin. 1988, 2, 81–83. [Google Scholar]
- Kimura, I.; Makino, M.; Honda, R.; Ma, J.; Kimura, M. Expression of major histocompatibility complex in mouse peritoneal macrophages increasingly depends on plasma corticosterone levels: Stimulation by aconitine. Biol. Pharm. Bull. 1995, 18, 1504–1508. [Google Scholar]
- Yang, Q.E. Cytology of 12 species in Aconitum L. and of 8 species in Delphinium L. of tribe Delphineae (Ranunculaceae) from China. Acta Phram. Sin. 2001, 39, 502–514. [Google Scholar]
- Zhang, Y.Y.; Zhang, Y.; Lin, R.C.; Sun, W.J. An expeditious HPLC method to distinguish Aconitum Kusnezoffi from related species. Fitoterapia 2009, 80, 333–338. [Google Scholar] [CrossRef]
- Chen, H.C.; Lee, C.Y.; Liau, B.C.; Lee, M.R.; Jong, T.T.; Chiang, S.T. Determination of aconitine-type alkaloids as makers in fuzi (Aconitum carmichaeli) by LC/ (+) ESI /MS3. J. Pharm. Biomed. Anal. 2008, 48, 1105–1111. [Google Scholar]
- Zhang, F.; Tang, M.H.; Chen, L.J.; Li, R.; Wang, X.H.; Duan, J.G.; Zhao, X.; Wei, Y.Q. Simultaneous quantitation of aconitine, mesaconitine, hypaconitine, benzolyaconitine, benzoylmesaconitine and benzoylhypaconitine in human plasma by liquid chromatography-tandem mass spectrometry and phramacokinetics evaluation of “SHEN-FU” injectable powder. J. Chromatogr. B 2008, 873, 173–179. [Google Scholar] [CrossRef]
- Lin, C.C.; Chan, T.Y.; Deng, J.F. Clinical features and management of herb-induced aconitine poisoning. Ann. Emerg. Med. 2004, 43, 574–579. [Google Scholar]
- Zhang, H.S. Aconite toxicity and poisoning management. Adverse Drug Reac. J. 2005, 2, 114–115. [Google Scholar]
- Csupor, D.; Wenziq, E.M.; Zupkó, I.; Wölkart, K.; Hohmann, J.; Bauer, R. Qualitative and quantitative analysis of aconitine-type and lipo-type alkaloids of Aconitum carmichaelli roots. J. Chromatogr. A 2009, 1216, 2079–2086. [Google Scholar] [CrossRef]
- Zhang, S.W.; Liu, Y.; Huang, G.Z.; Liu, L. Aconitine alters connexin43 phosphorylation status and [Ca2+] oscillation patterns in cultured neonatal rats. Toxicol. In Vitro 2007, 21, 1476–1485. [Google Scholar] [CrossRef]
- Liu, W.L.; Liu, Z.Q.; Song, F.R.; Liu, S.Y. Specific Conversion of Diester-diterpenoid Aconitum Alkaloids Components into Hydrolysis Monoester-diterpenoid Alkaloids Components and Lipo-alkaloids Components. Chem. J. Chin. U. 2007, 3, 717–720. [Google Scholar]
- Zhou, Y.; Cui, Y.; Liu, Y.; Shen, G. Bioavailability study on xiaohuoluo pills. Chin. J. Chin. Mater. Med. 1995, 20, 159–161. [Google Scholar]
- Liu, J.B.; Zhang, Y.J.; Chen, J.X. Pharmacodynamics study on Xiaohuoluo pills. Chin. Pharm. 2007, 18, 26–28. [Google Scholar]
- Pan, J.Q.; Xiao, L.Y.; Zhang, D.; Han, C. Immunosuppresive, antioxidative, anti-inflammatory and analgesic effect of Xiaohuoluo pills. Chin. J. Clin. Rehabil. 2006, 47, 183–188. [Google Scholar]
- Zhang, J.; Li, J.M.; Jiang, Y. Fast limit test of trace aconitine in Xiaohuoluo Pills. Chin. Trad. Pat. Med. 2009, 10, 1543–1546. [Google Scholar]
- Zhu, Z.Y.; Wang, B.L.; Li, J.S. Quantitative Determination of Mesaconitine, Hypaconitine and Aconitine in Xiaohuoluo Pills by TLC-HPLC. J. Pharm. Anal. 1996, 3, 154–156. [Google Scholar]
- Liu, X.X.; Chao, R.B. Determination of alkaloids in Radix Aconiti Lateralis Preparata by RP-ion-pair HPLC. Acta Phram. Sin. 2006, 4, 365–369. [Google Scholar]
- Liu, F.; Yu, X.H.; Li, F.; Tan, Y.Y.; Qiao, Y.J. Determination of three kinds of diester diterpenoid alkaloids (DDAs) in Aconitum carmichaeli and its processed products by HPLC. Chin. J. Chin. Mater. Med. 2006, 14, 1160–1162. [Google Scholar]
- Huang, Z.F.; Yi, J.H.; Chen, D.A.; Liu, Y.H. HPLC specific chromatogram of Radix Aconiti Praeparata and determination of six esteric alkaloids. J. Pharm. Anal. 2011, 2, 217–221. [Google Scholar]
- Li, H.; Deng, G.H. HPLC fingerprint of Radix Aconiti. Chin. J. Exp. Tradit. Med. Form. 2011, 3, 3–6. [Google Scholar]
- Yang, Z.F.; Liu, J.F.; Zhang, H. Study on the Quality Standards of Aconitum pendulum Busch. Chin. J. Chin. Mater. Med. 2005, 22, 1772–1773. [Google Scholar]
- Yang, X.L.; Wen, Z.H. Study on Quality Standards of Aconitum vilmorinianum Komarov. Chin. J. Ethnomed. and Ethnopharm. 2005, 72, 54–57. [Google Scholar]
- Koji, W.; Hideo, B.; Norio, K. Determination and Quantitative Analysis of aconitum Alkaloids in Plants by Liquid Chromatography Atmospheric Pressure Chemical Ionization Mass Spectrometry. J. Chromatogr. A 1993, 644, 43–48. [Google Scholar] [CrossRef]
- Ohta, H.; Seto, Y.; Tsunoda, N.; Takahashi, Y.; Matsuura, K.; Oqasawara, K. Determination of Aconitum alkaloids in blood and urine samples II. Capillary liquid chromatographic–frit fast atom bombardment mass spectrometric analysis. J. Chromatogr. B Biomed. Sci. Appl. 1998, 714, 215–221. [Google Scholar]
- Feng, H.T.; Li, S.F. Determination of five toxic alkaloids in two common herbal medicines with capillary electrophoresis. J. Chromatogr. A 2002, 973, 243–248. [Google Scholar] [CrossRef]
- Zhou, D.; Pan, G.M. Detection of Aconitine and Mesaconitine and Hypaconitine in Biology Samples by LC/MS/MS. Mod. Sci. Ins. 2005, 4, 64–67. [Google Scholar]
- Qiao, Y.L; Zhang, Y.H.; Zhang, W.; Zhang, J.L. A rapid resolution liquid chromatographic method for fingerprint analysis of raw and processed Caowu (Aconitum kusnezoffii). J. AOAC Int. 2009, 92, 653–662. [Google Scholar]
- Zhou, S.S.; Ma, Z.C.; Liang, Q.D.; Wang, Y.G.; Tan, H.L.; Xiao, C.R.; Zhang, B.L.; Gao, Y. UPLC/Q-TOF-MS based chemical profiling approach to evaluate chemical composition of augmentation toxicity in combination of Radix Aconiti and Pinellia Praeparata. Acta Chim. Sin. 2010, 70, 284–290. [Google Scholar]
- Chen, X.G.; Lai, Y.Q.; Cai, Z.W. Simultaneous analysis aconitine and its metabolites by liquid chromatography-electrospray ion trap mass spectrometry. J. Chin. Mass Spectrom. Soc. 2012, 2, 65–73. [Google Scholar]
- Zhang, H.G.; Sun, Y.; Duan, M.Y.; Chen, Y.J.; Zhong, D.F.; Zhang, H.Q. Separation and identification of Aconitum alkaloids and their metabolites in human urine. Toxicon 2005, 46, 500–506. [Google Scholar] [CrossRef]
- Xiang, P.; Shen, M.; Shen, B.H.; Zhuo, X.Y. Simultaneous Screening for 59 Drugs of Abuse in Biological Samples by LC-MS/MS-MRM. J. Chin. Mass Spectrom. Soc. 2006, 4, 237–241. [Google Scholar]
- Trygg, J.; Wold, S. Orthogonal projections to latent structures (O-PLS). J. Chemometr. 2002, 16, 119–128. [Google Scholar] [CrossRef]
- Qi, M.; Xiong, A.Z.; Geng, F.; Yang, L.; Wang, Z.T. A novel strategy for target profiling analysis of bioactive phenylethanoid glycosides in Plantago meidicine plants using ultra-performace liquid chromatography coupled with tandem quadrupole mass spectrometry. J. Sep. Sci. 2012, 35, 1470–1478. [Google Scholar]
- Xiong, A.Z.; Yang, L.; Ji, L.L.; Wang, Z.Y.; Yang, X.J.; Chen, Y.; Wang, X.L.; Wang, C.H.; Wang, Z.T. UPLC-MS based metabolomices study on Senecio scandens and S. vulgaris: An approach for the differentiation of two Senecio herbs with similar morphology but different toxicity. Metabolomics 2012, 8, 614–623. [Google Scholar]
- Wiklund, S.; Johansson, E.; Sjöström, L.; Mellerowicz, E.J.; Edlund, U.; Shochcor, J.P.; Gottfries, J.; Moritz, T.; Trygg, J. Visualization of GC/TOF-MS-based metabolomics data for identification of biochemically interesting compounds using OPLS class models. Anal. Chem. 2008, 80, 115–122. [Google Scholar]
- Xie, G.X.; Ni, Y.; Su, M.M.; Zhang, Y.Y.; Zhao, A.H.; Gao, X.F.; Liu, Z.; Xiao, P.G.; Jia, W. Application of Ultra-performance LC-TOF MS metabolite profiling techniques to the analysis of medicinal Panax herbs. Metabolomics 2008, 4, 248–260. [Google Scholar] [CrossRef]
- Kobayashi, S.; Putri, S.P.; Yamamoto, Y.; Donghyo, K.; Bamba, T.; Fukusaki, E. Gas chromatography-mass spectrometry based metabolic profiling for the identification of discrimination markers of Angelicae Radix and its application to gas cheomatography-flame ionization detector system. J. Biosci. Bioeng. 2012, 114, 232–236. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds from Radix Aconiti (RA), Radix Aconiti Kusnezoffii (RAK), Radix Aconiti Preparata (RAP), Radix Aconiti Kusnezoffii Preparata (RAKP) and Xiaohuoluo pill are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cui, P.; Han, H.; Wang, R.; Yang, L. Identification and Determination of Aconitum Alkaloids in Aconitum Herbs and Xiaohuoluo Pill Using UPLC-ESI-MS. Molecules 2012, 17, 10242-10257. https://doi.org/10.3390/molecules170910242
Cui P, Han H, Wang R, Yang L. Identification and Determination of Aconitum Alkaloids in Aconitum Herbs and Xiaohuoluo Pill Using UPLC-ESI-MS. Molecules. 2012; 17(9):10242-10257. https://doi.org/10.3390/molecules170910242
Chicago/Turabian StyleCui, Ping, Han Han, Rui Wang, and Li Yang. 2012. "Identification and Determination of Aconitum Alkaloids in Aconitum Herbs and Xiaohuoluo Pill Using UPLC-ESI-MS" Molecules 17, no. 9: 10242-10257. https://doi.org/10.3390/molecules170910242
APA StyleCui, P., Han, H., Wang, R., & Yang, L. (2012). Identification and Determination of Aconitum Alkaloids in Aconitum Herbs and Xiaohuoluo Pill Using UPLC-ESI-MS. Molecules, 17(9), 10242-10257. https://doi.org/10.3390/molecules170910242




