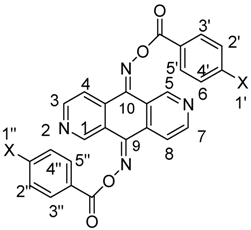
Oxime Esters of 2,6-Diazaanthracene-9,10-dione and 4,5-Diazafluoren-9-one as Photo-induced DNA-Cleaving Agents
Abstract
:1. Introduction
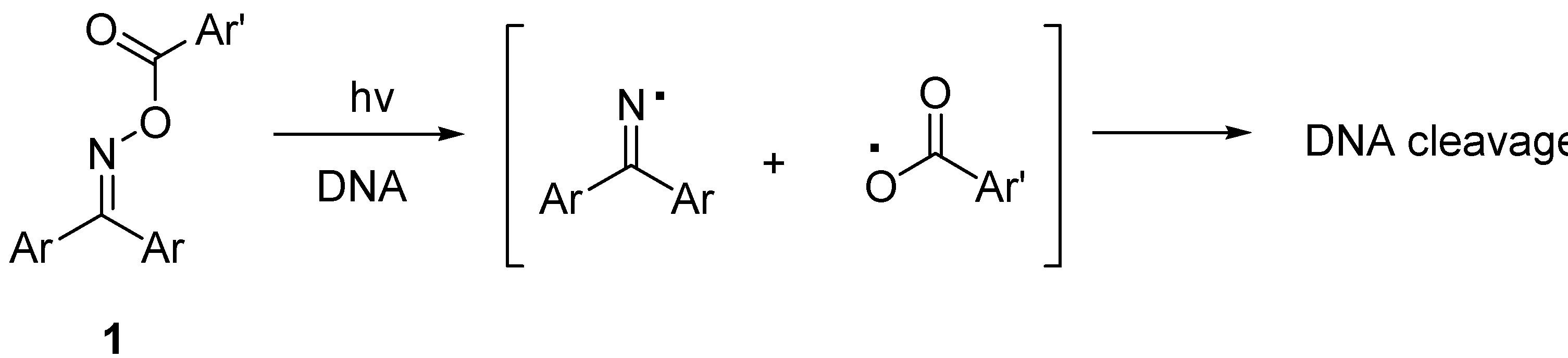
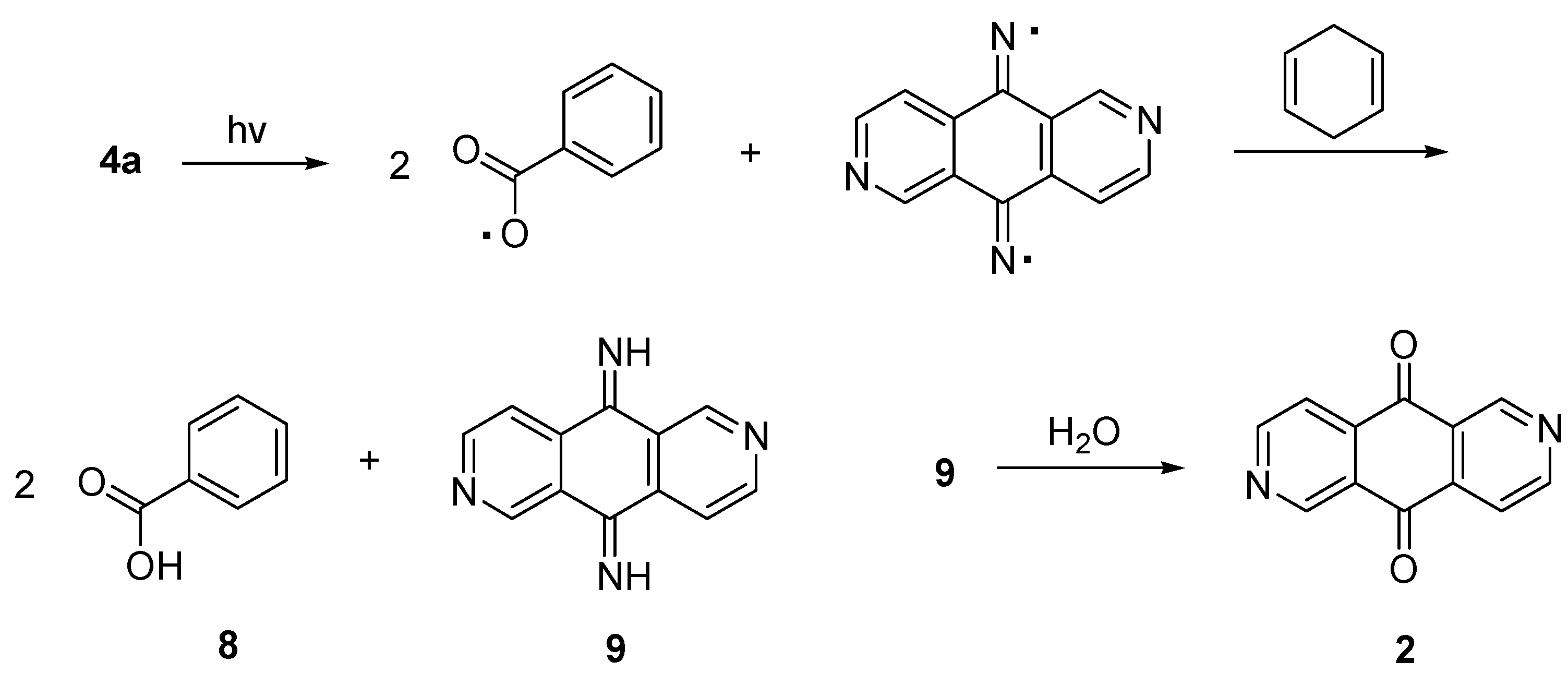
2. Results and Discussion
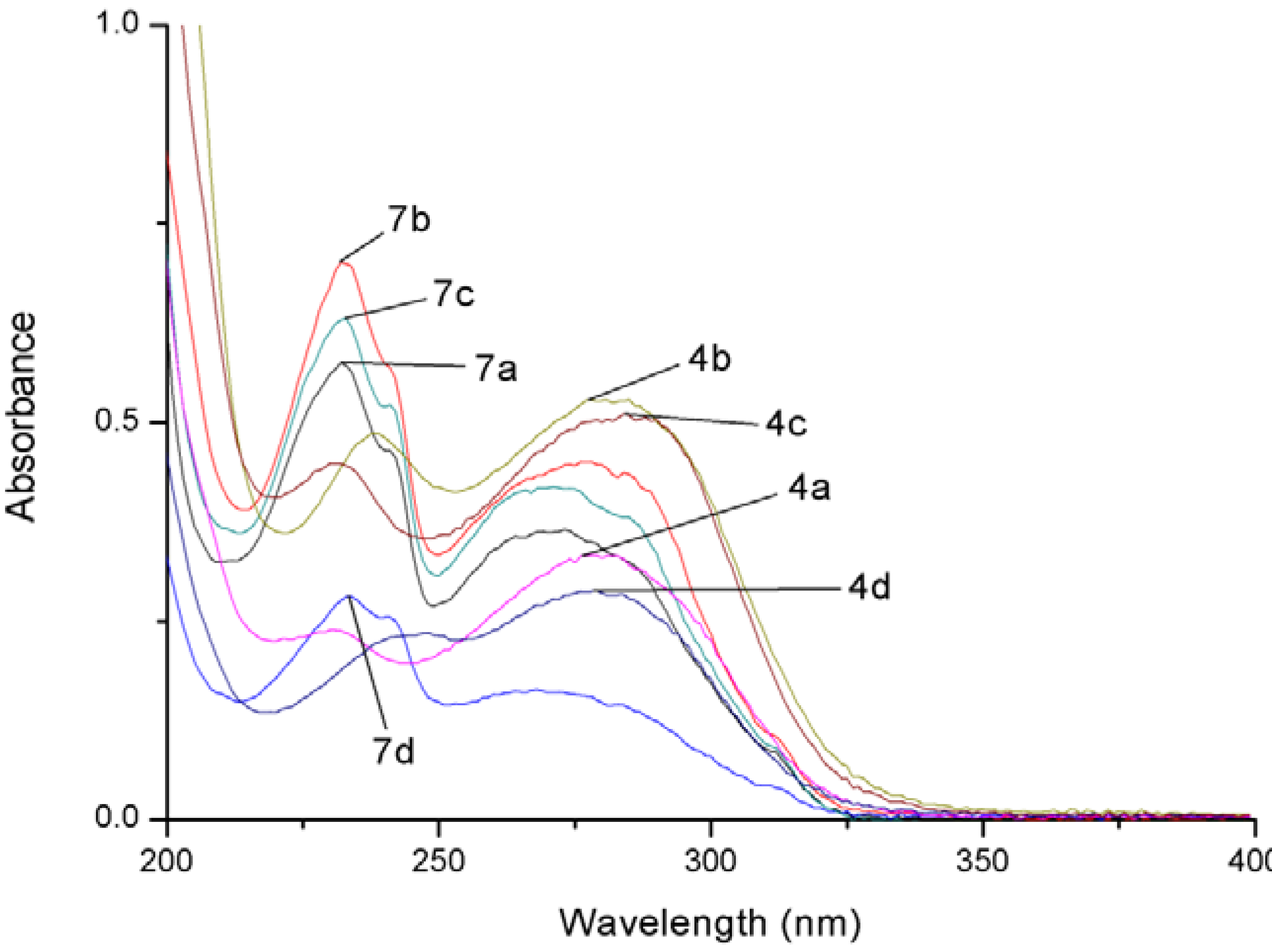
| Compound | λmax (ε) |
|---|---|
| 4a | 230 (23888), 280 (32944) |
| 4b | 238 (48714), 277 (52709) |
| 4c | 230 (44888), 283 (50593) |
| 4d | 248 (23504), 279 (28855) |
| 7a | 233 (57149), 273 (36500) |
| 7b | 232 (70174), 276 (45050) |
| 7c | 233 (63071), 271 (41946) |
| 7d | 234 (28102), 268 (16377) |
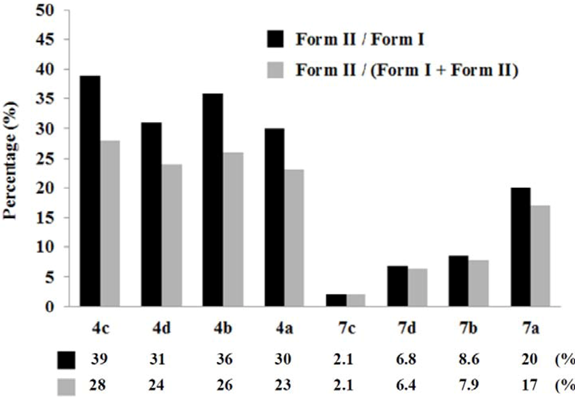

3. Experimental
3.1. General
3.2. General Procedure for the Preparation of Oximes 3 and 6
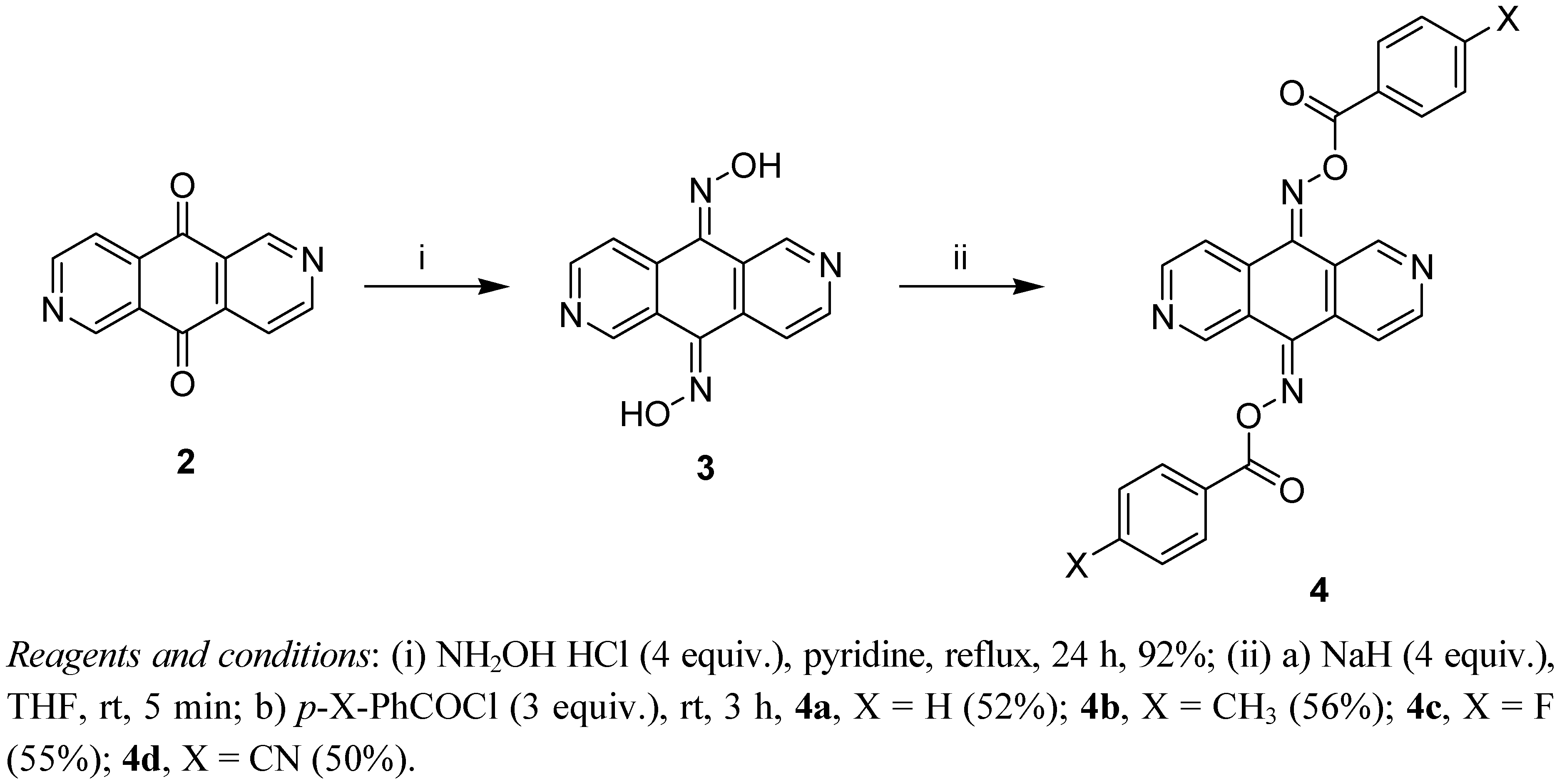
3.2.1. 2,6-Diazaanthracene-9,10-dioxime (3)
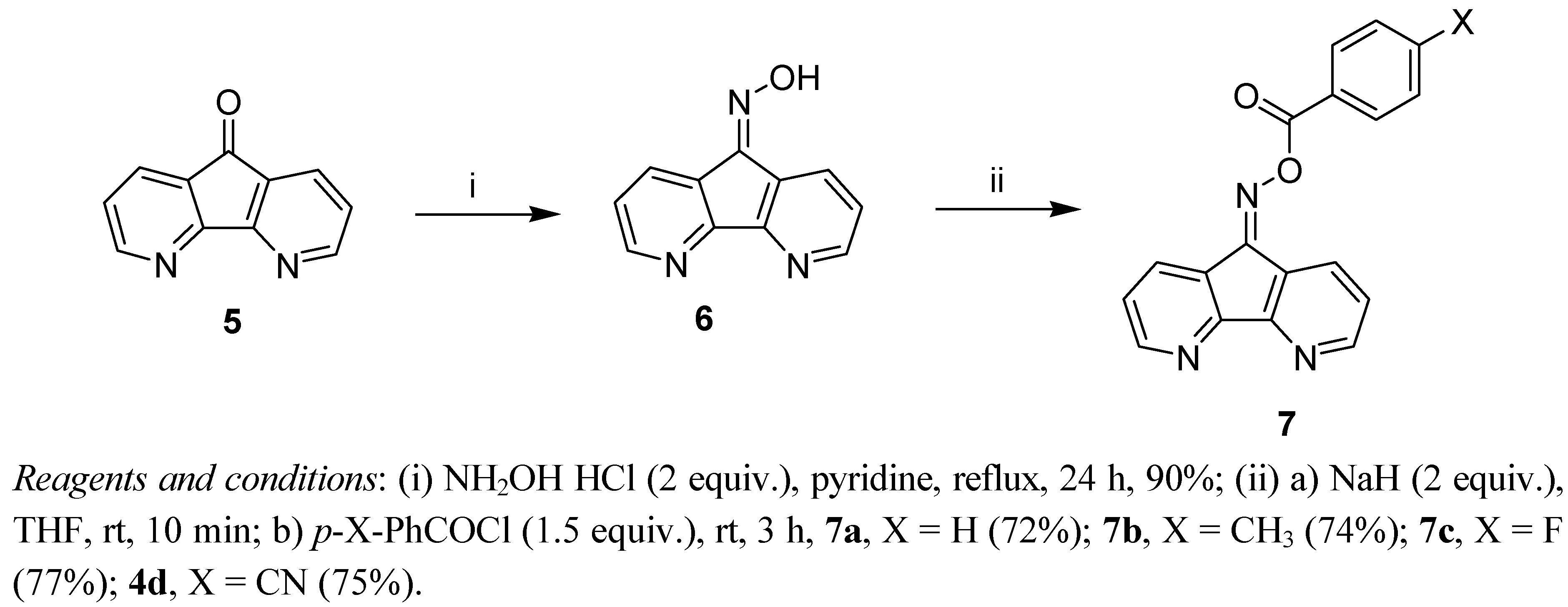
3.2.2. 4,5-Diazafluoren-9-oxime (6)
3.3. General Procedure for the Prepartion of Oxime Esters 4 and 7
3.3.1. 2,6-Diazaanthracene-9,10-dione bis-(O-benzoyloxime) (4a)
3.3.2. 2,6-Diazaanthracene-9,10-dione bis-[O-(4-methylbenzoyl)oxime] (4b)
3.3.3. 2,6-Diazaanthracene-9,10-dione bis-[O-(4-fluorobenzoyl)oxime] (4c)
3.3.4. 2,6-Diazaanthracene-9,10-dione bis-[O-(4-cyanobenzoyl)oxime] (4d)
| 4a (X = H) | 4b (X = CH3) | 4c (X = F) | 4d (X = CN) | |
| H-1, H-5 | 9.84 (s) | 9.83 (s) | 9.79 (s) | 9.76 (s) |
| H-3, H-7 | 8.94 (d, 5.2) | 8.92 (d, 5.1) | 8.94 (d, 5.1) | 8.97 (d, 5.2) |
| H-4, H-8 | 8.34 (d, 5.2) | 8.32 (d, 5.1) | 8.33 (dd, 5.1, 0.6) | 8.32 (d, 5.2) |
| H-3′, H-5′, H-3′′, H-5′′ | 8.15 (dd, 7.2, 1.2) | 8.04 (d, 8.1) | 8.21–8.15 (m) | 8.25 (d, 8.1) |
| H-1′, H-1′′ | 7.69 (br t, 7.3) | 2.47 (s) | – | – |
| H-2′, H-4′, H-2′′, H-4′′ | 7.57 (br t, 7.3) | 7.36 (d, 8.1) | 7.29–7.21 (m) | 7.88 (d, 8.1) |
3.3.5. 4,5-Diazafluoren-9-one O-9-benzoyloxime (7a)
3.3.6. 4,5-Diazafluoren-9-one O-9-(4-methylbenzoyl)oxime (7b)
3.3.7. 4,5-Diazafluoren-9-one O-9-(4-fluorobenzoyl)oxime (7c)
3.3.8. 4,5-Diazafluoren-9-one O-9-(4-cyanobenzoyl)oxime (7d)
| 7a (X = H) | 7b (X = CH3) | 7c (X = F) | 7d (X = CN) | |
| H-2, H-7 | 8.80 (dd, 4.8, 0.9) | 8.83–8.78 (m) | 8.83–8.78 (m) | 8.85–8.81 (m) |
| H-5 | 8.56 (dd, 7.8, 1.2) | 8.58 (dd, 7.6, 1.3) | 8.52 (dd, 7.6, 1.0) | 8.47 (dd, 6.6, 1.0) |
| H-4 | 8.35 (dd, 7.5, 0.9) | 8.36 (dd, 7.8, 1.2) | 8.35 (dd, 7.5, 0.9) | 8.36 (dd, 6.3, 1.0) |
| H-3′, H-5′ | 8.19 (d, 7.5) | 8.09 (d, 8.1) | 8.22 (dd, 8.7, 5.4) | 8.31 (d, 8.4) |
| H-1′ | 7.72 (dd, 7.5, 7.5) | 2.50 (s) | – | – |
| H-2′, H-4′ | 7.60 (dd, 7.8, 7.2) | 8.09 (d, 8.1) | 7.44–7.36 (m) | 7.92 (d, 8.4), |
| H-3, H-6 | 7.43–7.35 (m) | 7.43–7.36 (m) | 7.29 (t, 8.4) | 7.44–7.38 (m) |
3.4. General Procedures for DNA-Cleavage by Use of Oxime Esters
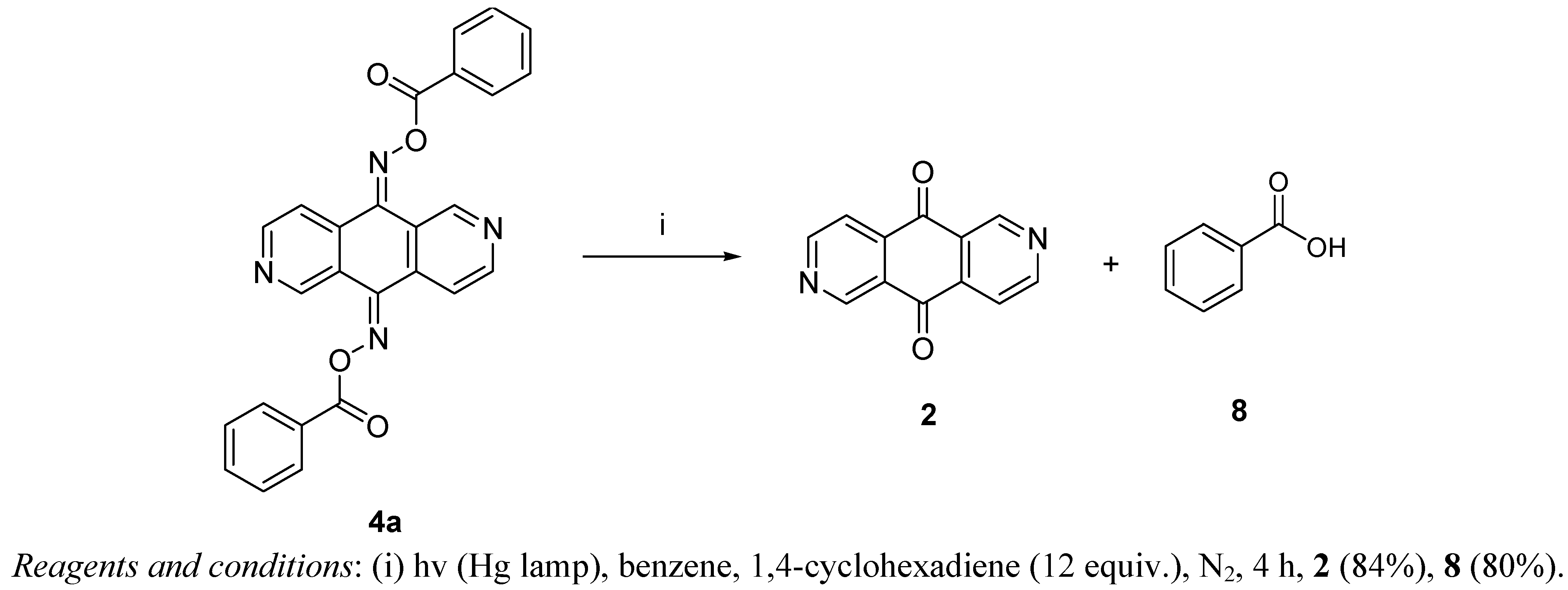
3.5. Photolysis of 2,6-Diazaanthracene-9,10-dione bis-(O-benzoyloxime) (4a)
4. Conclusions
Acknowledgements
- Sample Availability: Not available.
References and Notes
- Nielsen, P.E. Chemical and photochemical probing of DNA complexes. J. Mol. Recognit. 1990, 3, 1–25. [Google Scholar] [CrossRef]
- Nucleic Acid Targeted Drug Design; Prost, C.L.; Perun, T.J. (Eds.) Dekker: New York, NY, USA, 1992.
- Kochevar, I.E.; Dunn, D.A. Photosensitized reactions of DNA: Cleavage and addition. Bioorg. Photochem. 1990, 1, 273–315. [Google Scholar]
- Armitage, B. Photocleavage of nucleic acids. Chem. Rev. 1998, 98, 1171–1200. [Google Scholar] [CrossRef]
- Molecular Biology of the Gene, 5th; Watson, J.D.; Baker, T.A.; Bell, S.P.; Gann, A.; Levine, M.; Losick, R. (Eds.) Cold Spring Harbor Laboratory Press: Plainview, NY, USA, 2004.
- Pogozelski, W.K.; Tullius, T.D. Oxidative strand scission of nucleic acids: Routes initiated by hydrogen abstraction from the sugar moiety. Chem. Rev. 1998, 98, 1089–1107. [Google Scholar] [CrossRef]
- Yang, W.-Y.; Roy, S.; Phrathep, B.; Rengert, Z.; Kenworthy, R.; Zorio, D.A.R.; Alabugin, I.V. Engineering pH-gated transitions for selective and efficient double-strand DNA photocleavage in hypoxic tumors. J. Med. Chem. 2011, 54, 8501–8516. [Google Scholar] [CrossRef]
- Zheng, K.-W.; Zhang, D.; Zhang, L.-X.; Hao, Y.-H.; Zhou, X.; Tan, Z. Dissecting the strand folding orientation and formation of G-quadruplexes in single- and double-stranded nucleic acids by ligand-induced photocleavagefootprinting. J. Am. Chem. Soc. 2011, 133, 1475–1483. [Google Scholar] [CrossRef]
- Naik, H.R.P.; Naik, H.S.B.; Ravikumar, T.R.; Naik, T.R.R.; Aravinda, T.; Lamani, D.S.; Naika, H.R. 2-Thieno/selenopyrano[2,3-b]quinolines: microwave-induced one-pot synthesis, DNA binding, and photocleavage studie. Phosphor. Sulfur Silicon 2009, 184, 2583–2593. [Google Scholar] [CrossRef]
- Roy, M.; Bhowmick, T.; Santhanagopal, R.; Ramakumar, S.; Chakravarty, A.R. Photo-induced double-strand DNA and site-specific protein cleavage activity of L-histidine (μ-oxo)diiron(III) complexes of heterocyclic bas. Dalton Trans. 2009, 4671–4682. [Google Scholar]
- Hwu, J.R.; Huang, J.J.-T.; Tsai, F.-Y.; Tsay, S.-C.; Hsu, M.-H.; Hwang, K.-C.; Horng, J.-C.; Ho, J.A.; Lin, C.-C. Photochemical activities of N-nitrosocarboxamides and sulfoximides and their application to DNA cleavage. Chem. Eur. J. 2009, 15, 8742–8750. [Google Scholar] [CrossRef]
- Kovalenko, S.V.; Alabugin, I.V. Lysine-enediyne conjugates as photochemically triggered DNA double-strand cleavage agents. Chem. Commun. 2005, 1444–1446. [Google Scholar] [CrossRef]
- Theodorakis, E.A.; Wilcoxen, K.M. N-Aroyloxy-2-thiopyridones as efficient oxygen-radical generators: novel time-controlled DNA photocleaving reagents. Chem. Commun. 1996, 1927–1928. [Google Scholar] [CrossRef]
- Hwu, J.R.; Tsay, S.-C.; Hong, S.C.; Leu, Y.-J.; Liu, C.-F.; Chou, S.-S.P. Oxime esters of anthraquinone as photo-induced DNA-cleaving agents for single- and double-strand scissions. Tetrahedron Lett. 2003, 44, 2957–2960. [Google Scholar] [CrossRef]
- Hwu, J.R.; Yang, J.-R.; Tsay, S.-C.; Hsu, M.-H.; Chen, Y.-C.; Chou, S.-S.P. Photo-induced DNA cleavage by (heterocyclo)carbonyl oxime esters of anthraquinone. Tetrahedron Lett. 2008, 49, 3312–3315. [Google Scholar]
- Surkau, G.; Boehm, K.J.; Mueller, K.; Prinz, H. Synthesis, antiproliferative activity and inhibition of tubulin polymerization by anthracenone-based oxime derivatives. Eur. J. Med. Chem. 2010, 45, 3354–3364. [Google Scholar] [CrossRef]
- Chowdhury, N.; Dutta, S.; Nishitha, B.; Dasgupta, S.; Singh, N.D.P. N,O-Diacyl-4-benzoyl-N-phenylhydroxylamines as photoinduced DNA cleaving agents. Bioorg. Med. Chem. Lett. 2010, 20, 5414–5417. [Google Scholar] [CrossRef]
- Ikbal, M.; Jana, A.; Singh, N.D.P.; Banerjee, R.; Dhara, D. Photoacid generators (PAGs) based on N-acyl-N-phenylhydroxylamines for carboxylic and sulfonic acids. Tetrahedron 2011, 67, 3733–3742. [Google Scholar] [CrossRef]
- Bolitt, V.; Mioskowski, C.; Reddy, S.P.; Flack, J.R. A convenient synthesis of pyrido[3,4-g]isoquinoline via ortho-directed metallation/dimerization. Synthesis 1988, 388–389. [Google Scholar]
- Wong, K.T.; Chen, R.T.; Fang, F.C.; Wu, C.C.; Lin, Y.T. 4,5-Diazafluorene-incorporated ter(9,9-diarylfluorene): a novel molecular doping strategy for improving the electron injection property of a highly efficient OLED blue emitter. Org. Lett. 2005, 7, 1979–1982. [Google Scholar] [CrossRef]
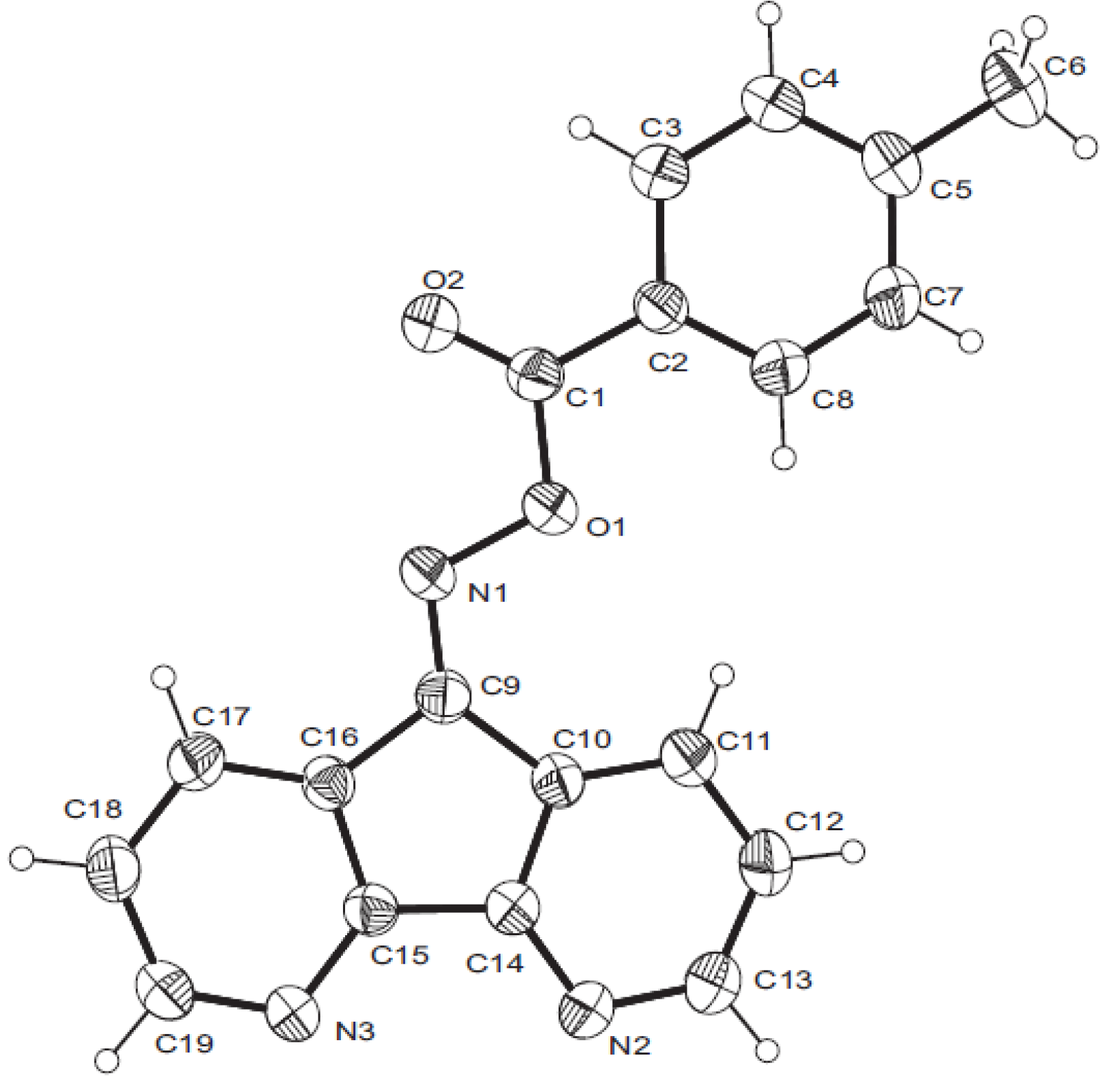
- Crystal data for 7b: C19H13N3O2, M = 315.32, bond precision: C–C = 0.0041 Å, cell: a = 7.3611(2), b = 11.2055(3), c = 18.5898(6) Å, β = 90.643(1), T = 293 K, V = 1533.28(8) Å3, space group P 21/c, Dx = 1.366 g cm−3, Z = 4, μ = 0.091 mm−1, F(000) = 656.0. CCDC 836168 contains the supplementary crystallographic data for 7b. These data can be obtained free of charge via www.ccdc.cam.ac.uk/conts/retrieving.html (or from the CCDC, 12 Union Road, Cambridge CB2 1EZ, UK; fax: +44 1223 336033; e-mail: deposit@ccdc.cam.ac.uk).
- Grissom, J.W.; Klingberg, D. Aryl radical additions to aldehydes and oxime ethers: the tandem enediyne-radical cyclization. J. Org. Chem. 1993, 58, 6559–6564. [Google Scholar] [CrossRef]
- Sako, M.; Nagai, K.; Maki, Y. Facile generation of hydroxyl radical by photolyis of pyrimido[5,4-g]pteridinetetrone N-oxides in aqueous solution. A new efficient DNA-photocleaving agent. J. Chem. Soc. Chem. Commun. 1993, 750–751. [Google Scholar]
- Sentagne, C.; Meunier, B.; Paillous, N. DNA cleavage photoinduced by new water-soluble zinc porphyrins linked to 9-methoxyellipticine. J. Photochem. Photobiol. B Biol. 1992, 16, 47–59. [Google Scholar] [CrossRef]
- Saito, I.; Takayama, M. Photoactivatable DNA-cleaving amino acids: Highly sequence-selective DNA photocleavage by novel L-lysine derivatives. J. Am. Chem. Soc. 1995, 117, 5590–5591. [Google Scholar] [CrossRef]
- Devasagayam, T.P.A.; Steenken, S.; Obendorf, M.S.W.; Schulz, W.A.; Sies, H. Formation of 8-hydroxy(deoxy)guanosine and generation of strand breaks at guanine residues in DNA by singlet oxygen. Biochemistry 1991, 30, 6283–6289. [Google Scholar] [CrossRef]
- Okada, T.; Kawanisi, M.; Nozaki, H. Photolysis of aromatic oxime benzoates. Bull. Chem. Soc. Jpn. 1969, 42, 2981–2983. [Google Scholar] [CrossRef]
- Ohta, H.; Tokumaru, K. Finding of aromatic substitution bydiphenylmethyleneimino radicals. Bull. Chem. Soc. Jpn. 1975, 48, 2393–2394. [Google Scholar] [CrossRef]
- Sakuragi, H.; Ishikawa, S.-I.; Nishimura, T.; Yoshida, M.; Inamoto, N.; Tokumaru, K. Homolytic aromatic substitution by iminyl radicals. Photolysis of aromatic ketone O-acyloximes in aromatic solvents. Bull. Chem. Soc. Jpn. 1976, 49, 1949–1954. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chou, S.-S.P.; Juan, J.-C.; Tsay, S.-C.; Huang, K.P.; Hwu, J.R. Oxime Esters of 2,6-Diazaanthracene-9,10-dione and 4,5-Diazafluoren-9-one as Photo-induced DNA-Cleaving Agents. Molecules 2012, 17, 3370-3382. https://doi.org/10.3390/molecules17033370
Chou S-SP, Juan J-C, Tsay S-C, Huang KP, Hwu JR. Oxime Esters of 2,6-Diazaanthracene-9,10-dione and 4,5-Diazafluoren-9-one as Photo-induced DNA-Cleaving Agents. Molecules. 2012; 17(3):3370-3382. https://doi.org/10.3390/molecules17033370
Chicago/Turabian StyleChou, Shang-Shing P., Jui-Chi Juan, Shwu-Chen Tsay, Kuei Pin Huang, and Jih Ru Hwu. 2012. "Oxime Esters of 2,6-Diazaanthracene-9,10-dione and 4,5-Diazafluoren-9-one as Photo-induced DNA-Cleaving Agents" Molecules 17, no. 3: 3370-3382. https://doi.org/10.3390/molecules17033370
APA StyleChou, S.-S. P., Juan, J.-C., Tsay, S.-C., Huang, K. P., & Hwu, J. R. (2012). Oxime Esters of 2,6-Diazaanthracene-9,10-dione and 4,5-Diazafluoren-9-one as Photo-induced DNA-Cleaving Agents. Molecules, 17(3), 3370-3382. https://doi.org/10.3390/molecules17033370