Synthesis and Antimicrobial Activity of Some New 1,3,4-Thiadiazole Derivatives
Abstract
:1. Introduction
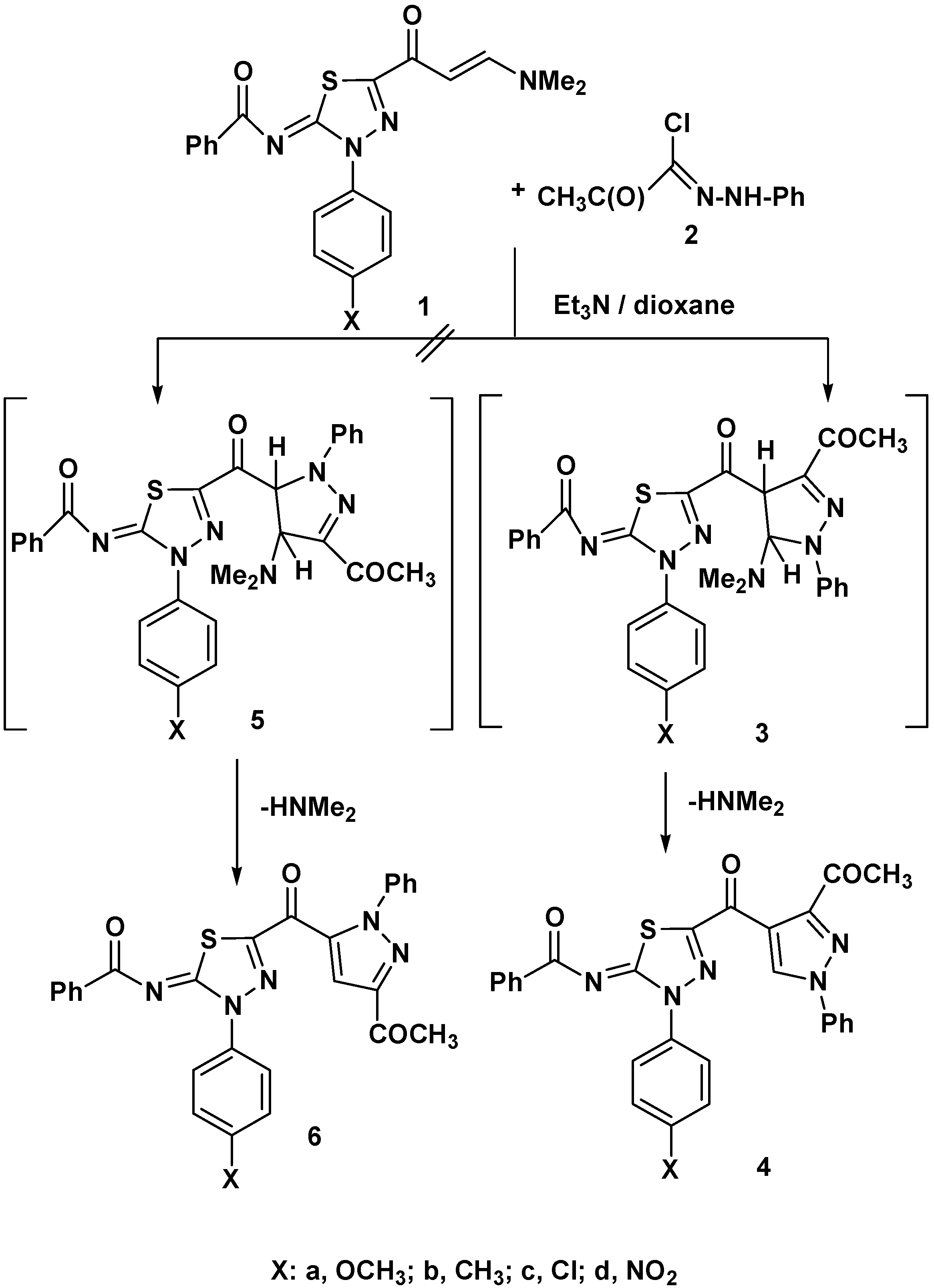
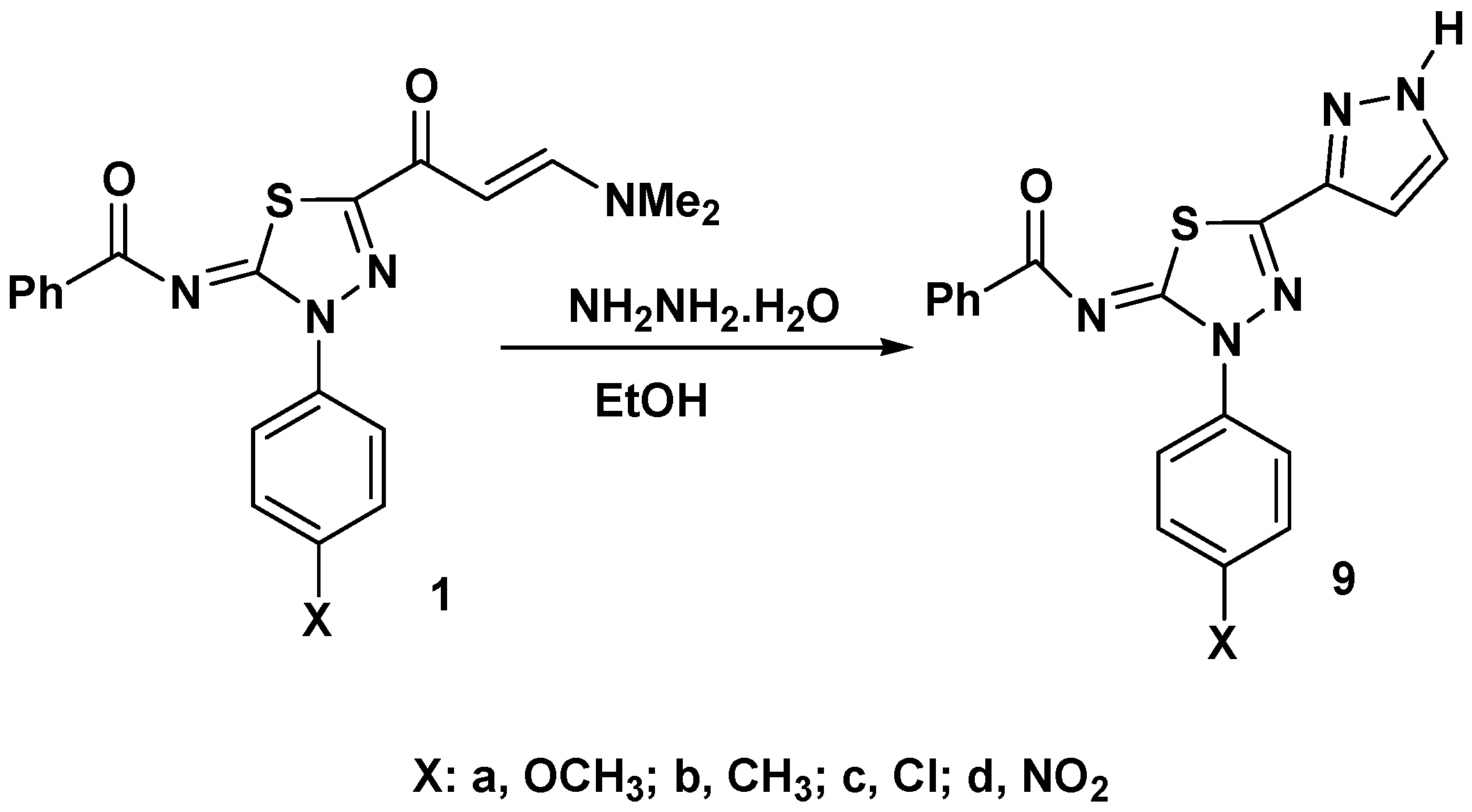
2. Results and Discussion

2.1. Biological Screening
2.1.1. Antimicrobial Activity
2.1.2. Antimicrobial Activity Screening and Structure Activity Relationship
| Compound No. | Fungi | Gram positive bacteria | Gram negative bacteria | |||||
|---|---|---|---|---|---|---|---|---|
| A. fumigatus | P. italicum | C. albicans | G. candidum | S. aureus | B. subtilis | P. aeruginosa | E. coli | |
| 4a | 15.6 (±0.19) | 16.3 (±0.24) | N.A. | 19.8 (±0.38) | 17.9 (±0.27) | 20.2 (±0.14) | N.A. | 10.9 (±0.29) |
| 4b | 19.6 (±0.13) | 20.3 (±0.22) | N.A. | 22.4 (±0.14) | 22.8 (±0.25) | 25.8 (±0.31) | N.A. | 17.6 ±0.25 |
| 4c | 10.3 (±0.12) | N.A. | N.A. | N.A. | 12.0 (±0.21) | 12.3 (±024) | N.A. | 8.4 (±0.12) |
| 4d | N.A. | N.A. | N.A. | N.A. | 13.6 (±0.17) | 15.4 (±0.33) | N.A. | N.A. |
| 7a | 13.9 (±0.25) | 16.7 (±0.19) | N.A. | 19.8 (±0.35) | 11.7 (±0.14) | 14.6 (±0.67) | N.A. | N.A. |
| 7b | N.A | N.A. | N.A. | N.A | N.A | N.A | N.A. | N.A. |
| 7c | 18.5 (±0.15) | 19.2 (±0.11) | N.A. | 20.9 (±0.26) | 18.3 (±0.19) | 19.2 (±0.21) | N.A. | 12.2 (±0.13) |
| 7d | 14.8 (±0.13) | 12.6 (±0.21) | N.A. | 16.8 (±0.22) | 15.0 (±0.18) | 11.2 (±0.12) | N.A. | 11.3 (±0.31) |
| 8a | 14.3 (±0.21) | 15.2 (±0.23) | N.A. | 11.7 (±0.15) | 15.8 (±0.31) | 18.6 (±0.21) | N.A. | N.A. |
| 8b | 20.4 (±0.19) | 21.6 (±0.12) | N.A. | 25.8 (±0.37) | 23.9 (±0.27) | 26.7 (±0.14) | N.A. | 19.8 (±0.10) |
| 8c | 16.9 (±0.13) | 17.8 (±0.24) | N.A. | 21.4 (±0.17) | 19.4 (±0.27) | 20.4 (±0.14) | N.A. | 10.6 (±0.31) |
| 9a | 20.3 (±0.31) | N.A. | N.A. | 22.6 (±0.22) | 23.7 (±0.31) | 25.9 (±0.22) | N.A. | 15.9 (±0.38) |
| 9b | 21.6 (±0.22) | 20.8 (±0.12) | N.A. | 26.2 (±025) | 22.0 (±0.23) | 21.4 (±012) | N.A. | 18.9 (±0.26) |
| Amphotericin B | 23.7 (±0.10) | 21.9 (±0.12) | 19.8 (±0.20) | 28.7 (±0.22) | N.A. | N.A. | N.A. | N.A. |
| Ampicillin | N.A. | N.A. | N.A. | N.A. | 27.4 (±0.18) | 32.4 (±0.10) | N.A. | N.A. |
| Gentamicin | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | 17.3 (±0.15) | 22.3 (±0.18) |
2.1.3. Minimum Inhibitory Concentration (MIC)
| CompoundNo. | Fungi | Gram positive bacteria | Gram negative bacteria | |||||
|---|---|---|---|---|---|---|---|---|
| A. fumigatus | P. italicum | C. albicans | G. candidum | S. aureus | B. subtilis | P. aeruginosa | E. coli | |
| 4a | 125 | 125 | N.A. | 31.25 | 62.5 | 15.63 | N.A. | 500 |
| 4c | 31.3 | 15.6 | N.A. | 3.9 | 1.95 | 0.12 | N.A. | 62.5 |
| 7c | 31.3 | 15.6 | N.A. | 7.81 | 62.5 | 31.3 | N.A. | 500 |
| 8b | 15.6 | 7.8 | N.A. | 0.12 | 1.95 | 0.06 | N.A. | 31.3 |
| 8c | 125 | 62.5 | N.A. | 7.81 | 15.6 | 7.8 | N.A. | 500 |
| 9a | 14.1 | N.A. | N.A. | 0.11 | 1.75 | 0.12 | N.A. | 26.5 |
| 9b | 0.9 | 15.3 | N.A. | 0.08 | 1.95 | 8.5 | N.A. | 500 |
| Amphotericin B | 0.49 | 1.95 | 15.63 | 0.015 | N.A. | N.A. | N.A. | N.A. |
| Ampicillin | N.A. | N.A. | N.A. | N.A. | 0.02 | 0.007 | N.A. | N.A. |
| Gentamicin | N.A. | N.A. | N.A. | N.A. | N.A. | N.A. | 62.5 | 0.98 |
3. Experimental
3.1. General
3.2. Reaction of Enaminones 1a–d with Hydrazonoyl Chloride 2
3.3. Reaction of Enaminones 1a–d with Heterocyclic Amines
3.4. Reaction of Enaminones 1a–d with Hydrazine Hydrate
3.5. Microbiological Studies
3.5.1. Agar Diffusion Well Method to Determine the Antimicrobial Activity
3.5.2. Minimal Inhibitory Concentration (MIC) Measurement
4. Conclusions
References
- Svete, J. Utilisation of chiral enaminones and azomethine imines in the synthesis of functionalised pyrazoles. ARKIVOC 2006, vii, 35–56. [Google Scholar]
- Abushanab, F.A.; Sherif, S.M.; Mousa, S.A.S. Dimethylformamide dimethyl acetal as a building block in heterocyclic synthesis. J. Heterocycl. Chem. 2009, 45, 801–827. [Google Scholar]
- Shawali, A.S.; Farghaly, T.A.; Al-Dahshoury, A.R. Synthesis, reactions and antitumor activity of new β-aminovinyl 3- pyrazolyl ketones. ARKIVOC 2009, xiv, 88–99. [Google Scholar]
- Palmieri, G.; Cimarelli, C. Chemo- and stereoselective reduction of enaminones for the preparation of biologically active compounds. ARKIVOC 2006, vi, 104–126. [Google Scholar]
- Al-Mousawi, S.; John, E.; Al-Kandery, N. Studies with enaminones: Synthesis and chemical reactivity of 2-(4-dimethylamino-2-oxobut-3-enyl)-isoindole-1,3-dione and of 4-(4-dimethyl-amino-2-oxobut-3-enyloxy)-2H-phthalazin-1-one. J. Heterocycl. Chem. 2004, 41, 381–385. [Google Scholar] [CrossRef]
- Li, Z.; Wang, X.; Da, Y. Synthesis of 2-(5-(2-chlorophenyl)-2-furoylamino)-5-aryloxymethyl-1,3,4-thiadiazoles under microwave irradiation. Synth. Commun. 2001, 31, 1829–1836. [Google Scholar] [CrossRef]
- Supuran, C.T.; Briganti, F.; Tilli, S.; Chegwidden, W.R.; Scozzafava, A. Carbonic anhydrase inhibitors: Sulfonamides as antitumor agents. Bioorg. Med. Chem. 2001, 9, 703–714. [Google Scholar] [CrossRef]
- Liu, X.; Shi, Y.; Ma, Y.; Zhang, C.; Dong, W.; Pan, L.; Wang, B.; Li, Z. Synthesis, antifungal activities and 3D-QSAR study of N-(5-substituted-1,3,4-thiadiazol-2-yl)cyclopropane-carboxamides. Eur. J. Med. Chem. 2009, 44, 2782–2786. [Google Scholar] [CrossRef]
- Deminbas, N.; Karaoglu, S.A.; Demirbas, A.; Sancak, K. Synthesis and antimicrobial activities of some new 1-(5-phenylamino-[1,3,4]thiadiazol-2-yl)methyl-5-oxo-[1,2,4]triazole and 1-(4-phenyl-5-thioxo-[1,2,4]triazol-3-yl)methyl-5-oxo- [1,2,4]triazole derivatives. Eur. J. Med. Chem. 2004, 39, 793–804. [Google Scholar] [CrossRef]
- Holla, B.S.; Poorjary, K.N.; Rao, B.S.; Shivananda, M.K. New bis-aminomercaptotriazoles and bis-triazolothiadiazoles as possible anticancer agents. Eur. J. Med. Chem. 2002, 37, 511–517. [Google Scholar] [CrossRef]
- Farghaly, T.A.; Abdallah, M.A.; Muhammad, Z.A. Synthesis and evaluation of the anti-microbial activity of new heterocycles containing the 1,3,4-thiadiazole moiety. Molecules 2011, 16, 10420–10432. [Google Scholar] [CrossRef]
- Farghaly, T.A.; Abdalla, M.M. Synthesis, tautomerism, antimicrobial, anti-HCV, anti-SSPE, antioxidant and antitumor activities of arylazobenzosuberones. Bioorg. Med. Chem. 2009, 17, 8012–8019. [Google Scholar] [CrossRef]
- Abdel Hafez, N.A.; Farghaly, T.A.; Al-Omar, M.A.; Abdalla, M.M. Synthesis of bioactive polyheterocyclic ring systems as 5α-reductase inhibitors. Eur. J. Med. Chem. 2010, 45, 4838–4844. [Google Scholar] [CrossRef]
- Riyadh, S.M.; Farghaly, T.A.; Abdallah, M.A.; Abdalla, M.M.; Abd El-Aziz, M.R. New pyrazoles incorporating pyrazolylpyrazole moiety: Synthesis, anti-HCV and antitumor activity. Eur. J. Med. Chem. 2010, 45, 1042–1050. [Google Scholar] [CrossRef]
- Farghaly, T.A.; Abdel Hafez, N.A.; Ragab, E.A.; Awad, H.M.; Abdalla, M.M. Synthesis, anti-HCV, antioxidant, and peroxynitrite inhibitory activity of fused benzosuberone derivatives. Eur. J. Med. Chem. 2010, 45, 492–500. [Google Scholar] [CrossRef]
- Farghaly, T.A.; Gomha, S.M.; Abbas, E.M.; Abdalla, M.M. Hydrazonoyl halides as precursors for new fused heterocycles of 5α-reductase inhibitors. Archiv der Pharmazie 2012, 345, 117–122. [Google Scholar] [CrossRef]
- Negri, G.; Kascheres, C.; Kascheres, A.J. Recent development in preparation reactivity and biological activity of enaminoketones and enaminothiones and their utilization to prepare heterocyclic compounds. J. Heterocycl. Chem. 2004, 41, 461–491. [Google Scholar] [CrossRef]
- Shawali, A.S.; Farghaly, T.A.; Al-Dahshoury, A.R. Efficient synthesis of functionalized 3-(hetaryl)pyrazoles. ARKIVOC 2010, ix, 19–30. [Google Scholar]
- Behbehani, H.; Ibrahim, H.M. 4-Thiazolidinones in heterocyclic synthesis: synthesis of novel enaminones, azolopyrimidines and 2-arylimino-5-arylidene-4-thiazolidinones. Molecules 2012, 17, 6362–6385. [Google Scholar] [CrossRef]
- Ho, Y.W. 5-(1-Pyrrolyl)-2-phenylthieno[2,3-d]pyrimidine as building block in heterocyclic synthesis. J. Chin. Chem. Soc. 2007, 54, 1075–1085. [Google Scholar]
- Wen, L.-R.; Wang, S.-W.; Li, M.; Yang, H.-Z. Reaction of enaminones with aminopyrazoles: Synthesis, structures and bioactivities of 7-aryl-3-cyano-2-substituted pyrazolo[1,5-a]pyrimidines. Chin. J. Chem. 2005, 23, 1231–1235. [Google Scholar] [CrossRef]
- Smania, J.A.; Monache, F.D.; Smania, E.F.A.; Cuneo, R.S. Antibacterial activity of steroidal compounds isolated from Ganoderma applanatum (Pers.) Pat. (Aphyllophoromycetideae) fruit body. Int. J. Med. Mushrooms 1999, 1, 325–330. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 4, 7, 8 and 9 are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Farghaly, T.A.; Abdallah, M.A.; Aziz, M.R.A. Synthesis and Antimicrobial Activity of Some New 1,3,4-Thiadiazole Derivatives. Molecules 2012, 17, 14625-14636. https://doi.org/10.3390/molecules171214625
Farghaly TA, Abdallah MA, Aziz MRA. Synthesis and Antimicrobial Activity of Some New 1,3,4-Thiadiazole Derivatives. Molecules. 2012; 17(12):14625-14636. https://doi.org/10.3390/molecules171214625
Chicago/Turabian StyleFarghaly, Thoraya A., Magda A. Abdallah, and Mohamed R. Abdel Aziz. 2012. "Synthesis and Antimicrobial Activity of Some New 1,3,4-Thiadiazole Derivatives" Molecules 17, no. 12: 14625-14636. https://doi.org/10.3390/molecules171214625
APA StyleFarghaly, T. A., Abdallah, M. A., & Aziz, M. R. A. (2012). Synthesis and Antimicrobial Activity of Some New 1,3,4-Thiadiazole Derivatives. Molecules, 17(12), 14625-14636. https://doi.org/10.3390/molecules171214625




