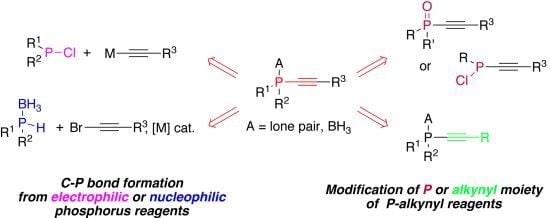
Stoichiometric and Catalytic Synthesis of Alkynylphosphines
Abstract
:1. Introduction

2. Nucleophilic Substitution at P-atom of Halophosphines
2.1. Synthesis of Secondary Alkynylphosphines

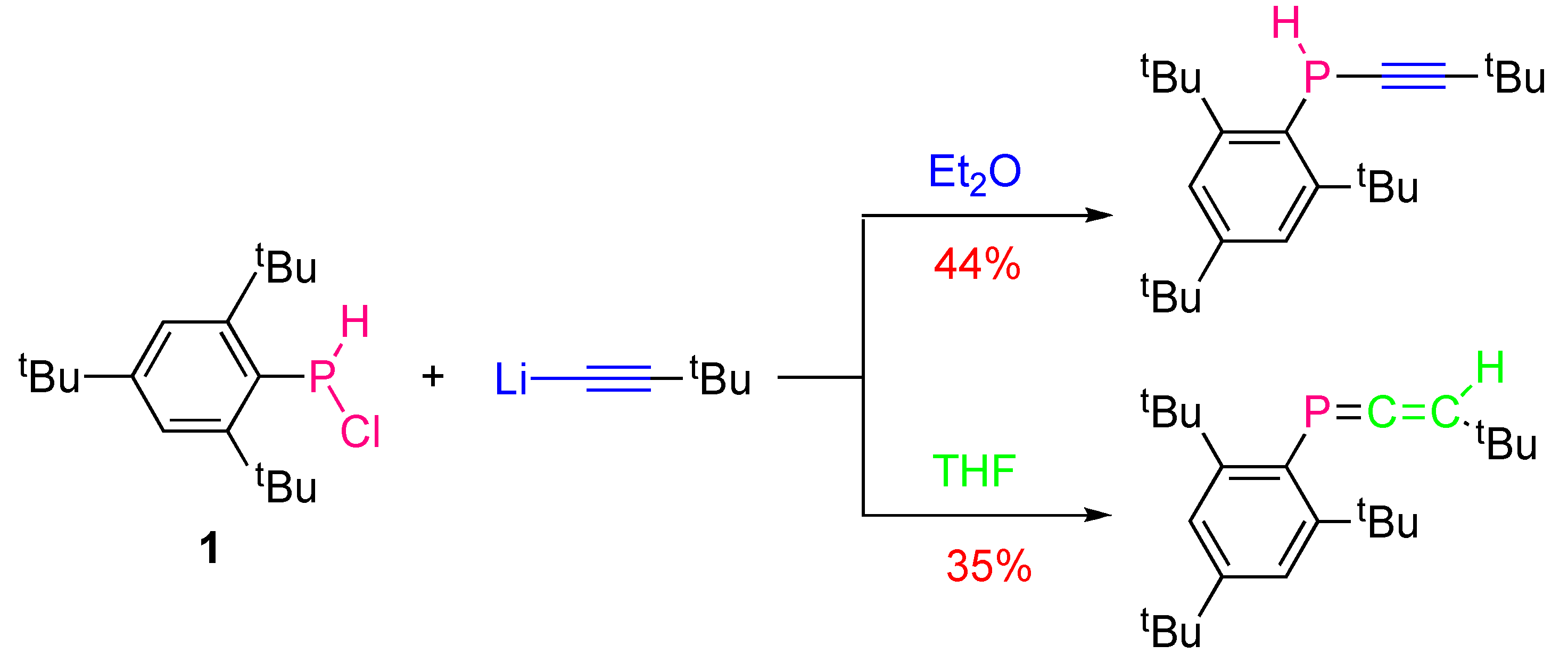
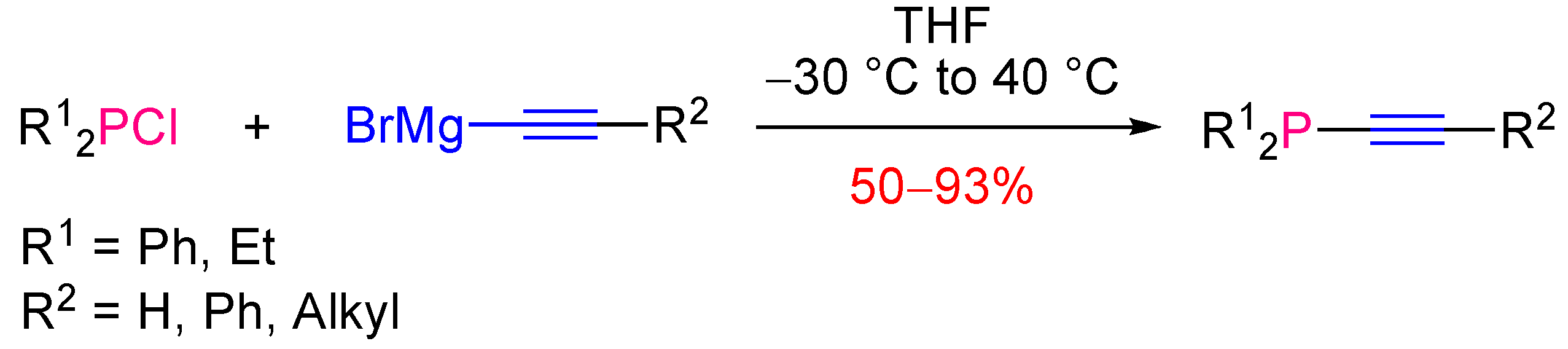
2.2. Synthesis of Tertiary Alkynylphosphines
2.2.1. Version with Achiral Reagents
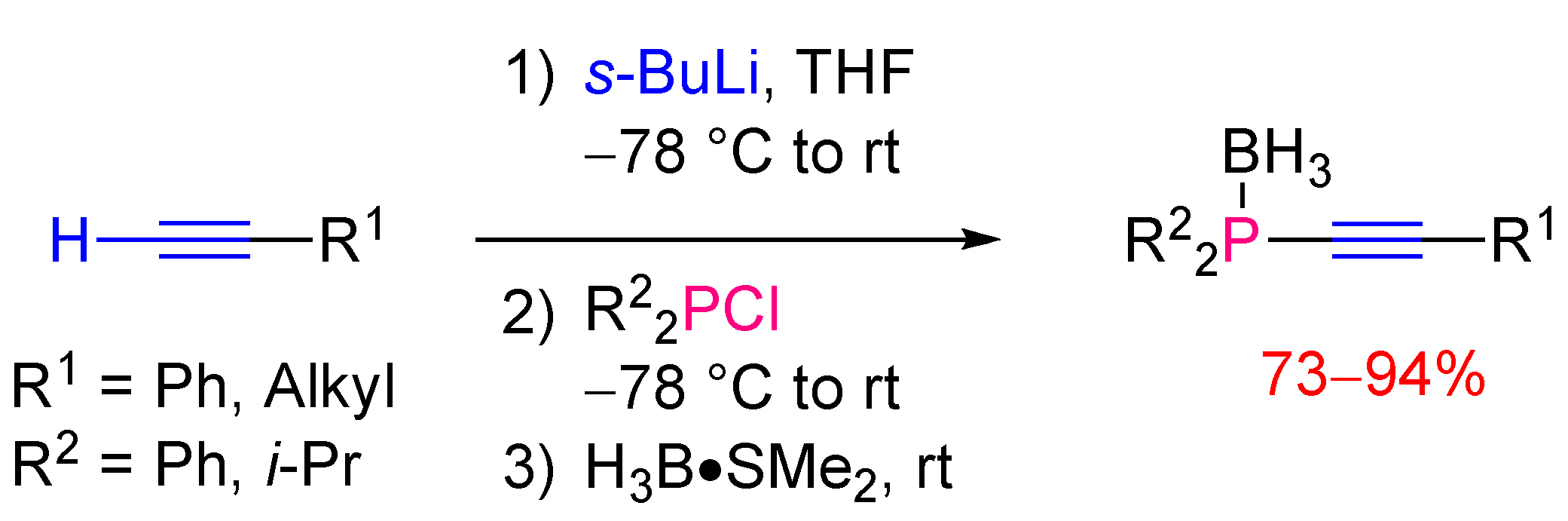
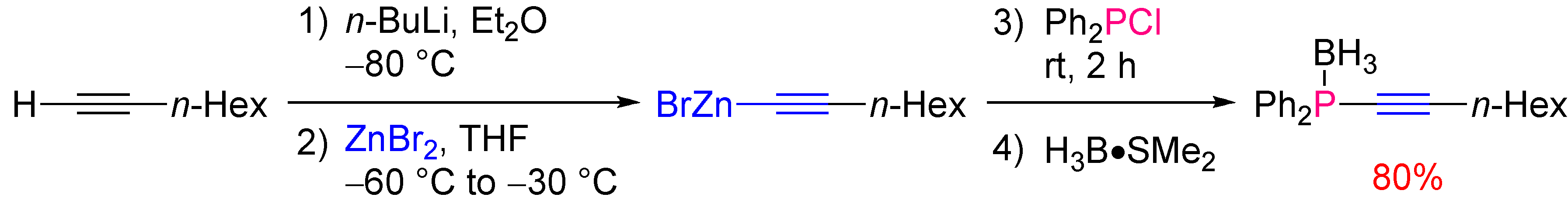

2.2.2. Asymmetric Version
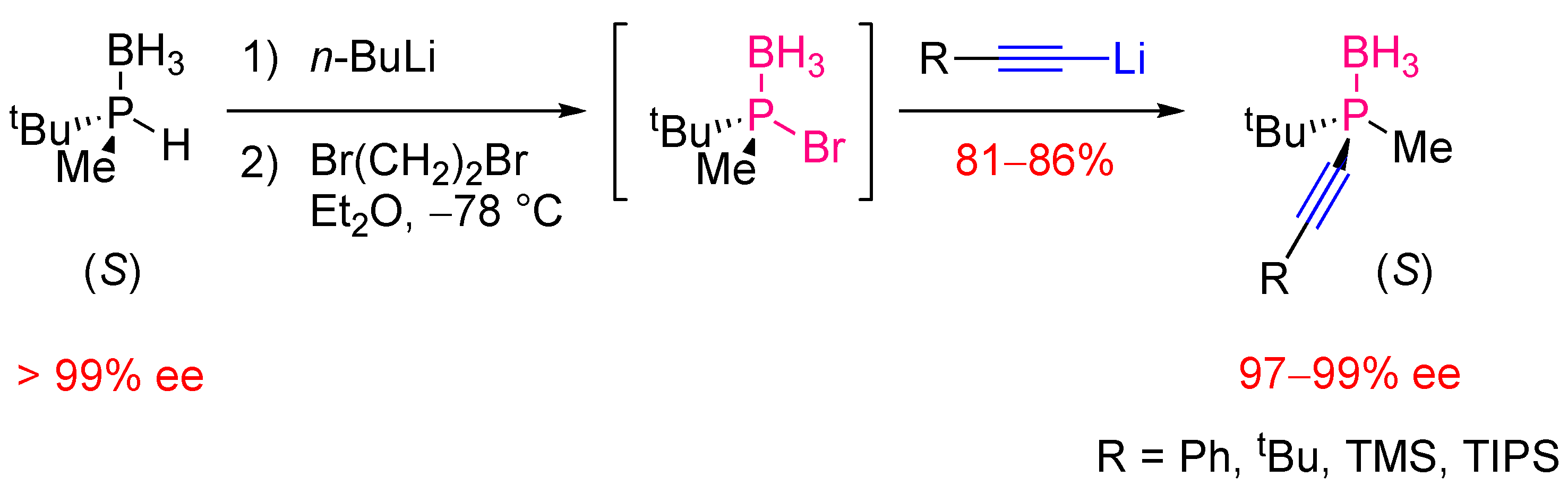
3. Catalytic C-P bond Forming Reactions
3.1. Electrophilic Phosphorus Reagent
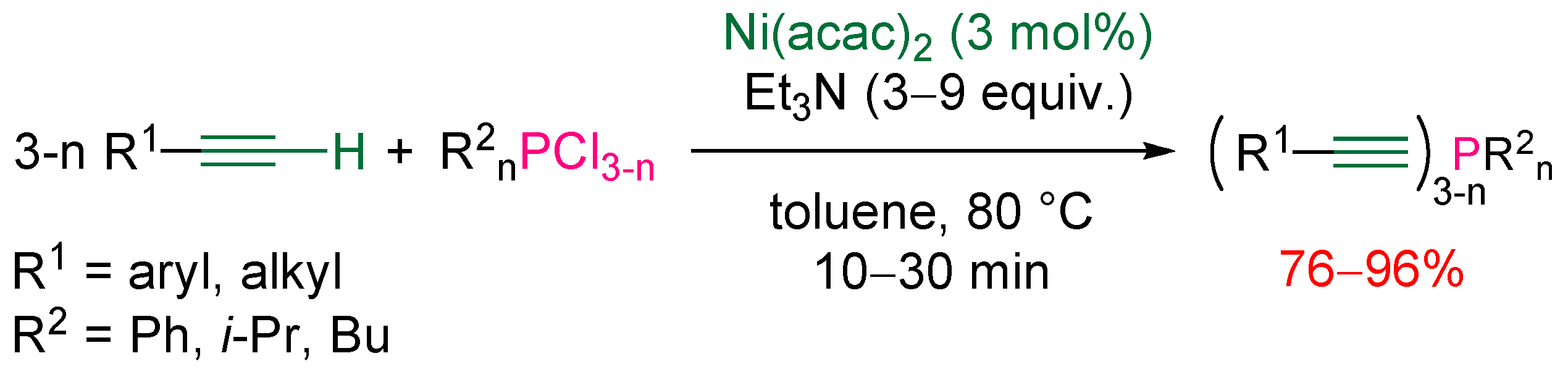
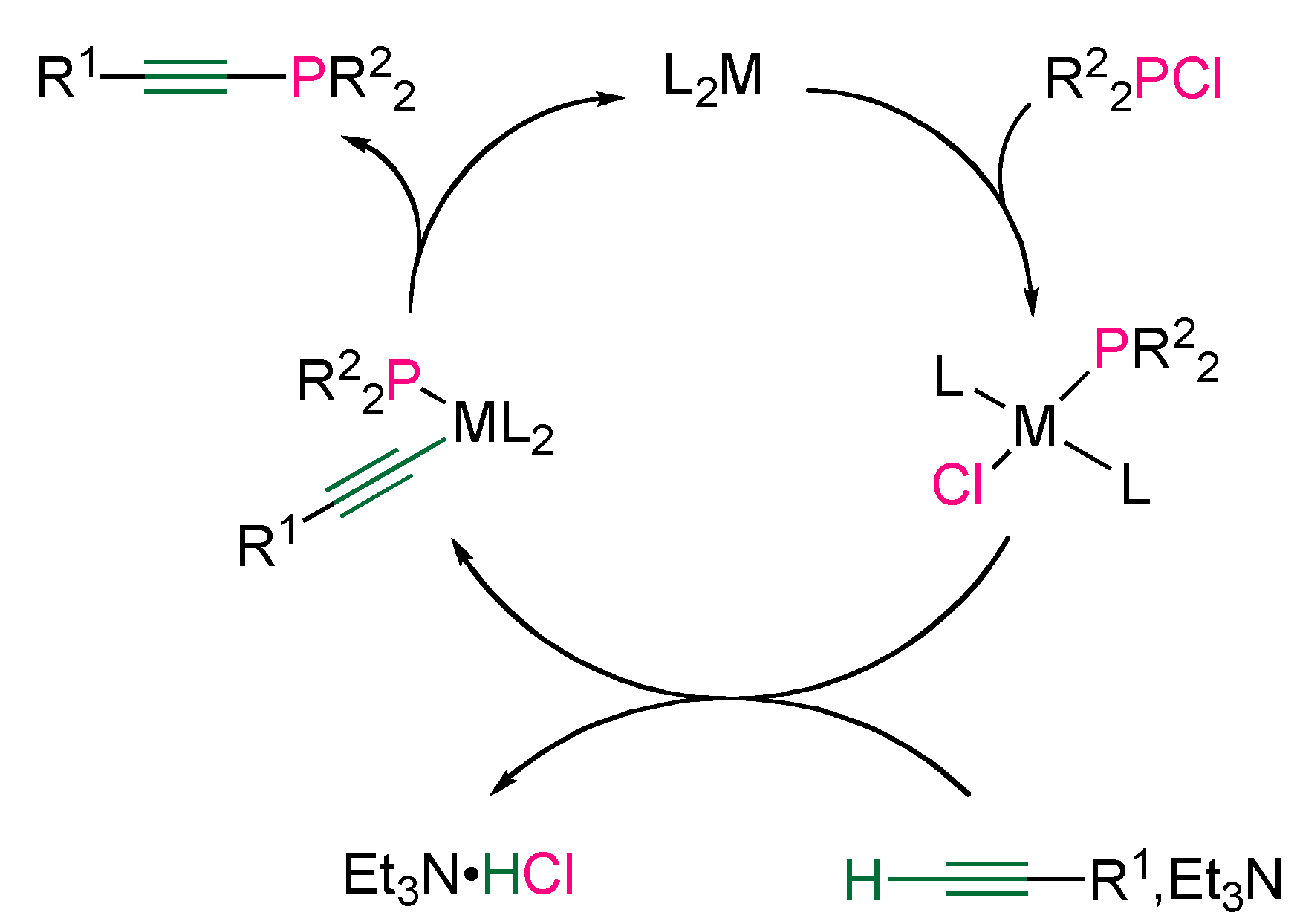

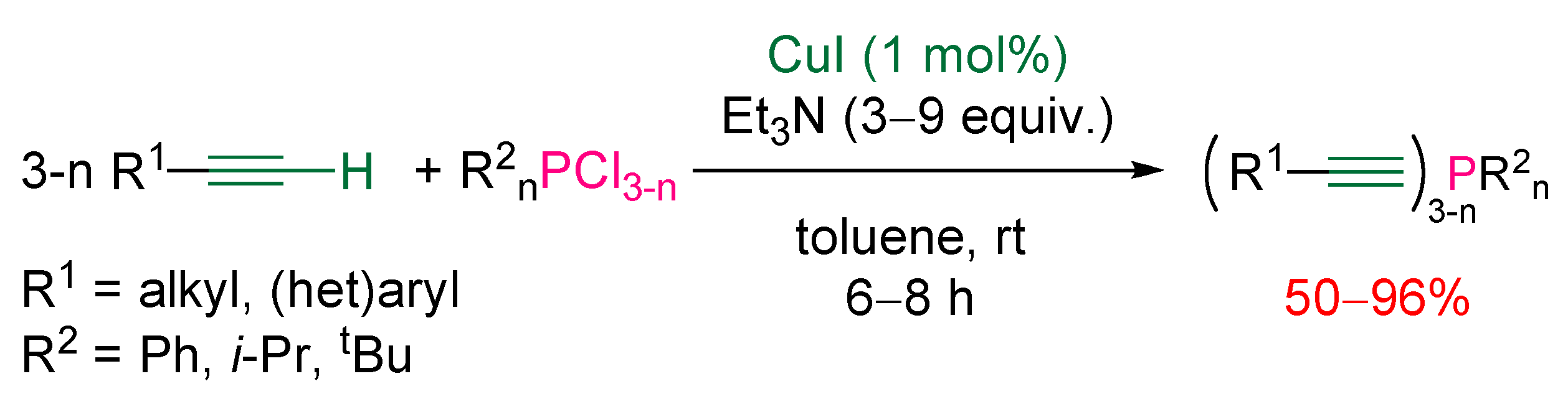

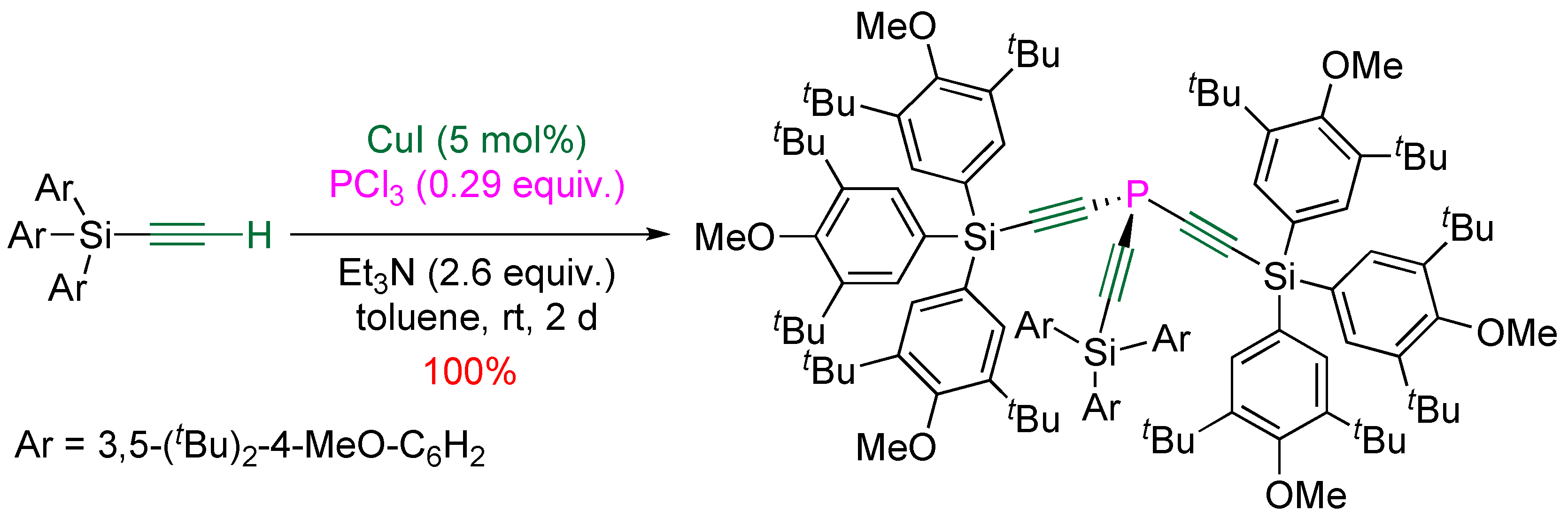
3.2. Nucleophilic Phosphorus Reagent
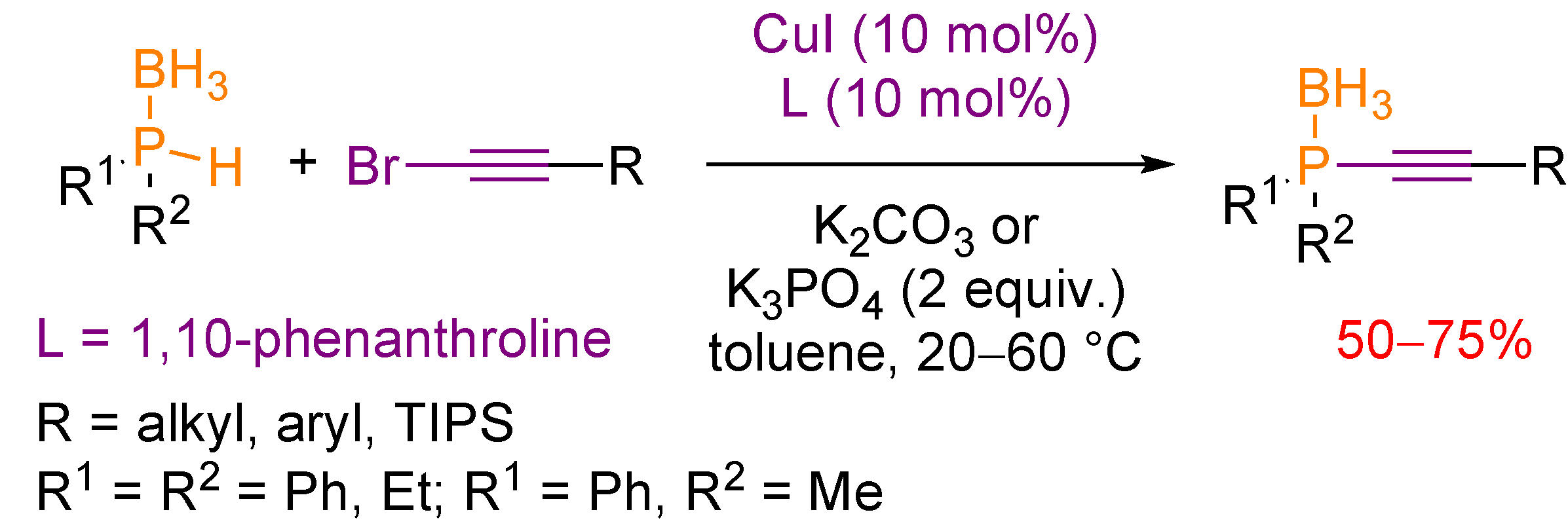
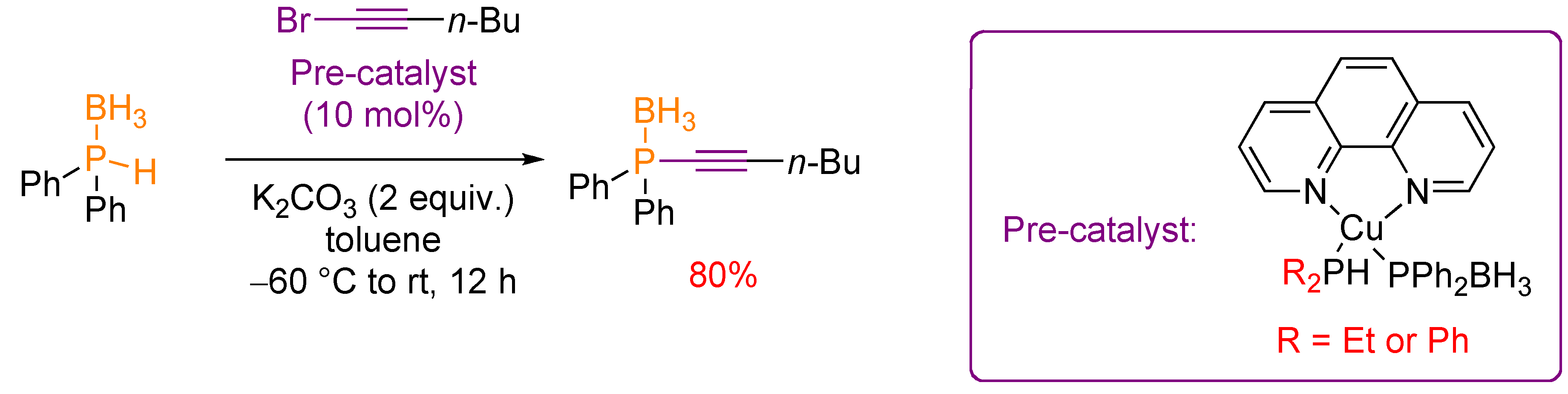
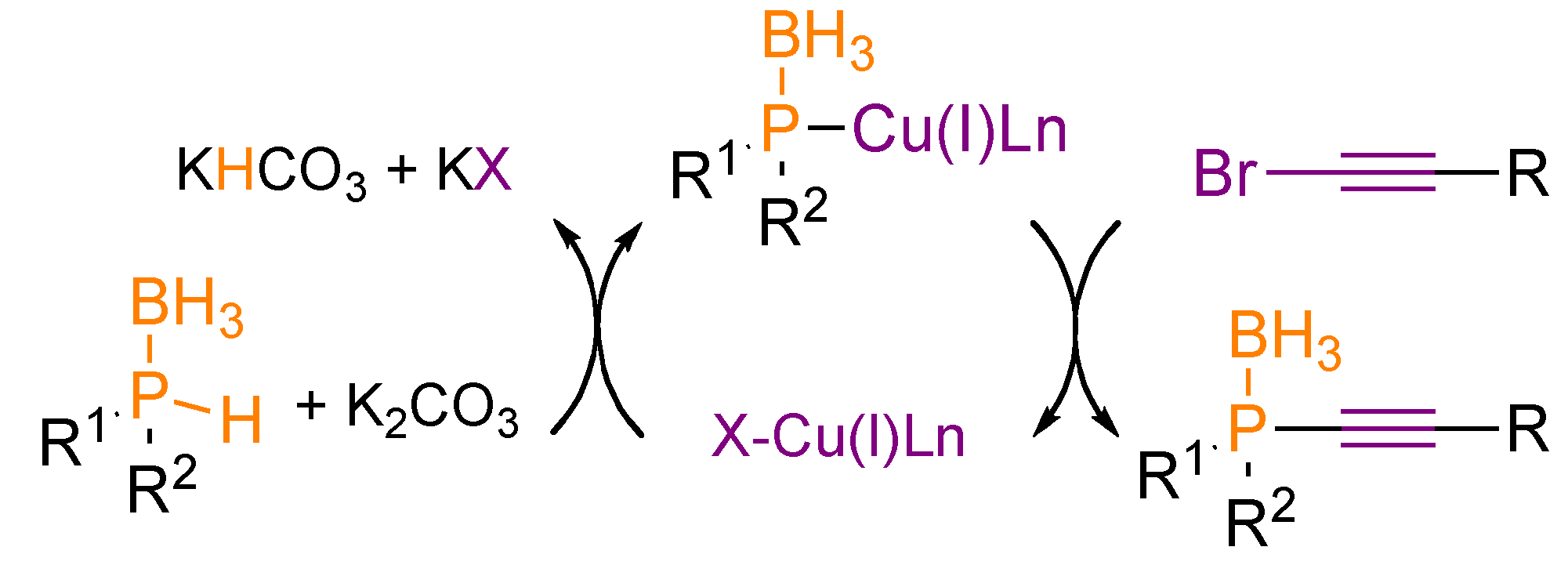
4. Transformation of Alkynylphosphorus Derivatives
4.1. Functionalization of the Triple bond Moiety


4.2. Nucleophilic Substitution at Phosphorus Atom
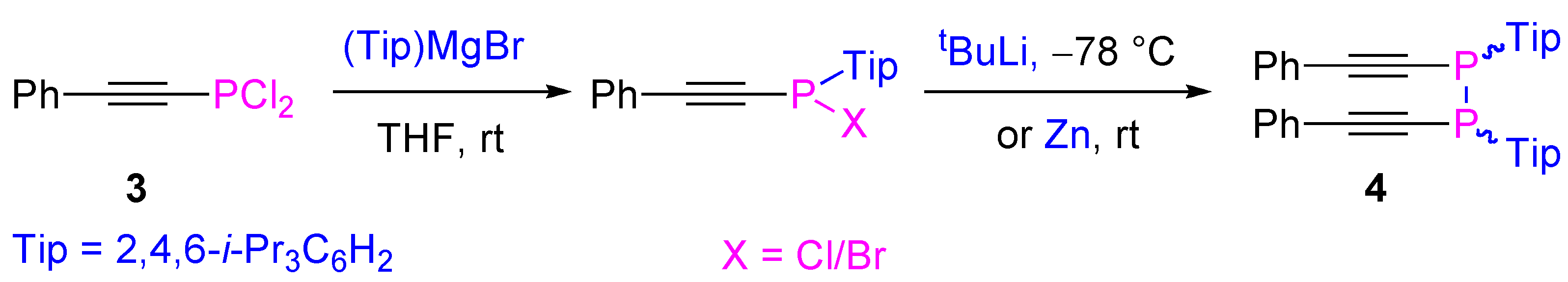
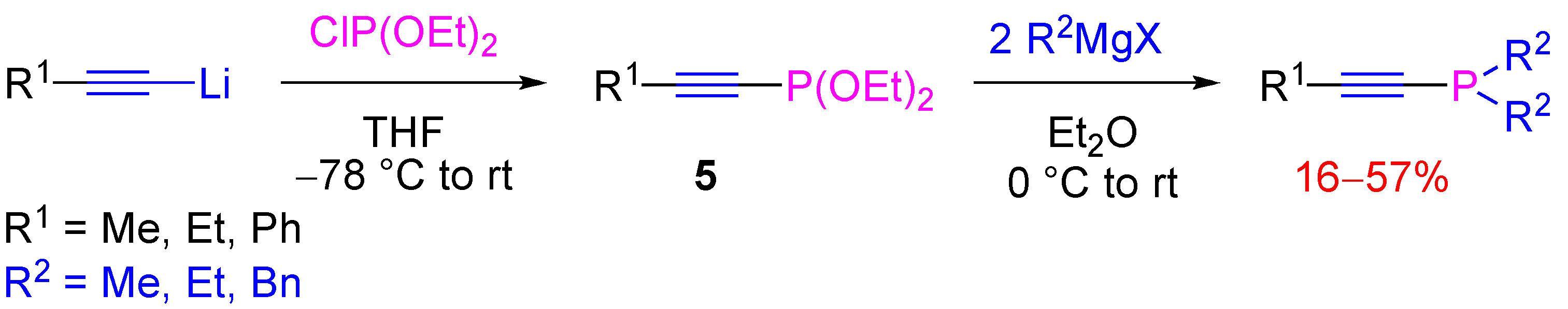
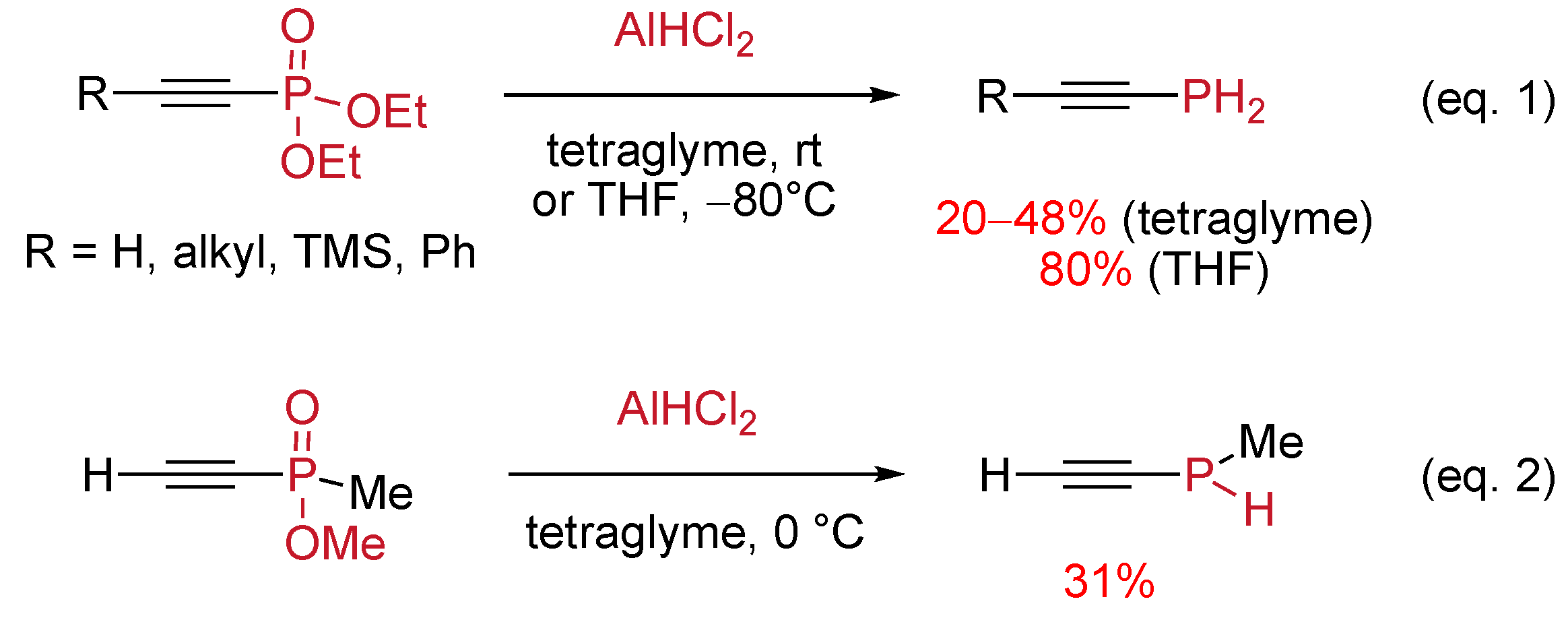
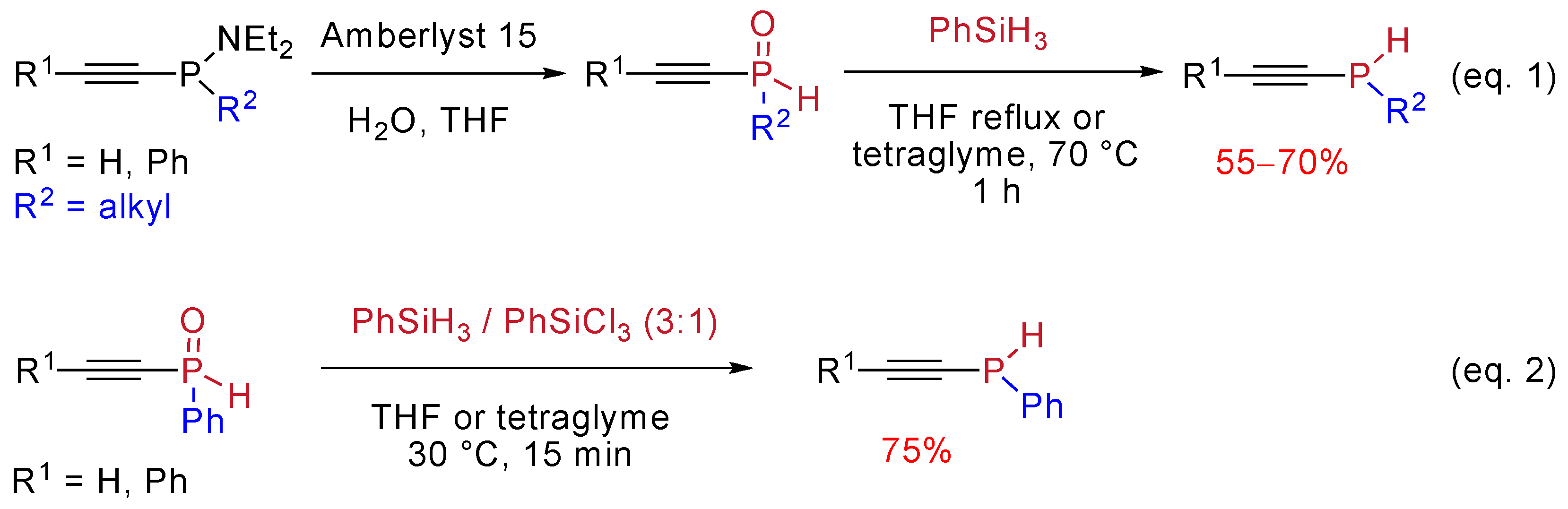
4.3 Reduction of the Phosphoryl Moiety
5. Conclusions
Acknowledgments
References and Notes
- Börner, A. Phosphorus Ligands in Asymmetric Catalysis; Wiley-VCH: Weinheim, Germany, 2008. [Google Scholar]
- Diederich, F.; Stang, P.; Tykwinski, R.R. Acetylene Chemistry; Wiley-VCH: Weinheim, Germany, 2004. [Google Scholar]
- Weymiens, W.; Slootweg, J.C.; Lammertsma, K. Phosphine Acetylenic Macrocycles and Cages: Synthesis and Reactivity. In Phosphorus Compounds Advanced Tools in Catalysis and Material Science, 1st ed; Peruzzini, M., Gonsalvi, L., Eds.; Springer: New York, NY, USA, 2011; Volume 37, pp. 21–55. [Google Scholar]
- Ito, H.; Ohmiya, H.; Sawamura, M. Construction of methylenecycloheptane frameworks through 7-exo-dig cyclisation of acetylenic silyl enol ethers catalyzed by triethynylphosphine-gold complex. Org. Lett. 2010, 12, 4380–4383. [Google Scholar]
- Imamoto, T.; Saitoh, Y.; Koide, A.; Ogura, T.; Yoshida, K. Synthesis and enantioselectivity of P-chiral phosphine ligands with alkynyl groups. Angew. Chem. Int. Ed. 2007, 46, 8636–8639. [Google Scholar] [CrossRef]
- Kondoh, A.; Yorimitsu, H.; Oshima, K. 1-Alkynylphosphines and their derivatives as key starting materials in creating new phosphines. Chem. Asian J. 2010, 5, 398–409. [Google Scholar] [CrossRef]
- D’yachkova, S.G.; Nikitin, M.V.; Beskrylaya, E.A.; Arbuzova, S.N.; Kashik, T.V.; Gusarova, N.K.; Trofimov, B.A. Reaction of alkylthio(chloro)acetylenes with bis(2-phenylethyl)phosphine. Russ. J. Gen. Chem. 1999, 69, 767–770. [Google Scholar]
- Pietrusiewicz, K.M.; Stankevic, M. 1-Phosphorus-functionalized alk-1-ynes. In Science of Synthesis: Houben-Weyl Methods of Molecular Transformations; de Meijere, A., Ed.; Thieme: Stuttgart, Germany, 2005; Volume 24, pp. 1073–1085. [Google Scholar]
- Märkl, G.; Reitinger, S. 3H-Phosphaallen—Alkinyl-1H-Phosphan—Tautomere. Tetrahedron Lett. 1988, 29, 463–466. [Google Scholar] [CrossRef]
- Voskuil, W.; Arens, J.F. Chemistry of acetylenic ethers. LXII. Tertiary phosphines with an acetylene-phosphorus bond. Recl. Trav. Chim. Pays-Bas 1962, 81, 993–1008. [Google Scholar]
- Charrier, C.; Chodkiewicz, W.; Cadiot, P. Préparations, propriétés chimiques et spectrographiques de phosphines α-acétyléniques. Bull. Soc. Chim. Fr. 1966, 1002–1011. [Google Scholar]
- Ortial, S.; Montchamp, J.-L. Synthesis of Z-alkenyl phosphorus compounds through hydroalumination and carbocupration of alkynyl precursors. Org. Lett. 2011, 13, 3134–3137. [Google Scholar] [CrossRef]
- Sladkov, A.M.; Ukhin, L.Y.; Korshak, V.V. Reaction of copper acetylides with halogen compounds. Izv. Akad. Nauk SSR Ser. Khim. 1963, 2213–2215. [Google Scholar]
- Hartmann, H. Exchange reactions of some ethynyl-, diethynyl-, and butadiynyltin compounds. Justus Liebigs Ann. Chem. 1968, 714, 1–7. [Google Scholar] [CrossRef]
- Gol’ding, I.R.; Sladkov, A.M. Silver acetylenides in ethynylation reactions. Izv. Akad. Nauk SSR Ser. Khim. 1972, 529–530. [Google Scholar]
- Langer, F.; Püntener, K.; Stürmer, R.; Knochel, P. Preparation of polyfunctional phosphines using zinc organometallics. Tetrahedron: Asymmetry 1997, 8, 715–738. [Google Scholar]
- Bharathi, P.; Periasamy, M. Direct metalation of 1-alkynes using TiCl4/Et3N and the reactions of the organotitanium intermediates with electrophiles. Organometallics 2000, 19, 5511–5513. [Google Scholar] [CrossRef]
- Krause, N.; Seebach, D. Chemistry of acetylenic titanium compounds. Chem. Ber. 1987, 120, 1845–1851. [Google Scholar] [CrossRef]
- Beletskaya, I.P.; Afanasiev, V.V.; Kazankova, M.A.; Efimova, I.V. New approach to phosphinoalkynes based on Pd- and Ni-catalyzed cross-coupling of terminal alkynes with chlorophosphanes. Org. Lett. 2003, 5, 4309–4311. [Google Scholar] [CrossRef]
- Kondoh, A.; Yorimitsu, H.; Oshima, K. Copper-catalyzed anti-hydrophosphination reaction of 1-alkynylphosphines with diphenylphosphine providing (Z)-1,2-diphosphino-1-alkenes. J. Am. Chem. Soc. 2007, 129, 4099–4104. [Google Scholar]
- Afanasiev, V.V.; Beletskaya, I.P.; Kazankova, M.A.; Efimova, I.V.; Antipin, M.U. A convenient and direct route to phosphinoalkynes via copper-catalyzed cross-coupling of terminal alkynes with chlorophosphanes. Synthesis 2003, 2835–2838. [Google Scholar]
- Ochida, A.; Ito, H.; Sawamura, M. Using triethynylphosphine ligands bearing bulky end caps to create a holey catalytic environment: Application to gold(I)-catalyzed alkyne cyclizations. J. Am. Chem. Soc. 2006, 128, 16486–16487. [Google Scholar] [CrossRef]
- Ochida, A.; Sawamura, M. Phosphorus ligands with a large cavity: synthesis of triethynylphosphines with bulky end caps and application to the rhodium-catalyzed hydrosilylation of ketones. Chem. Asian J. 2007, 2, 609–618. [Google Scholar] [CrossRef]
- Bernoud, E.; Alayrac, C.; Delacroix, O.; Gaumont, A.-C. Copper-catalyzed synthesis of alkynylphosphine derivatives: unprecedented use of nucleophilic phosphorus compounds. Chem. Commun. 2011, 47, 3239–3241. [Google Scholar] [CrossRef]
- Abdellah, I.; Bernoud, E.; Lohier, J.-F.; Alayrac, C.; Toupet, L.; Lepetit, C.; Gaumont, A.-C. Neutral copper-phosphido-borane complexes: Synthesis, characterization, and use as precatalysts in Csp-P bond formation. Chem. Commun. 2012, 48, 4088–4090. [Google Scholar]
- Huc, V.; Balueva, A.; Sebastian, R.-M.; Caminade, A.-M.; Majoral, J.-P. Synthesis of functionalized mono-, di-, tri- and tetraphosphines: attempted application to prepare hyperbranched polymers and dendrimers built with phosphines at each branching point. Synthesis 2000, 726–730. [Google Scholar]
- Knowles, W.S.; Sabacky, M.J.; Vineyard, B.D.; Weinkauff, D.J. Asymmetric hydrogenation with a complex of rhodium and a chiral bisphosphine. J. Am. Chem. Soc. 1975, 97, 2567–2568. [Google Scholar] [CrossRef]
- Yoshifuji, M.; Ichikawa, Y.; Yamada, N.; Toyota, K. Preparation and X-ray analysis of novel carbonyltungsten(0) complexes of diphosphinidenecyclobutenes. Chem. Commun. 1998, 27–28. [Google Scholar]
- Aguiar, A.M.; Irelan, J.R. S.; Morrow, C.J.; John, J.P.; Prejean, G.W. A convenient, synthetic pathway to dialkyl-1-alkynylphosphines. J. Org. Chem. 1969, 34, 2684–2686. [Google Scholar]
- Keglevich, G.; Gaumont, A.-C.; Denis, J.-M. Selective reductions in the sphere of organophosphorus compounds. Heteroat. Chem. 2001, 12, 161–167. [Google Scholar] [CrossRef]
- Guillemin, J.-C.; Savignac, P.; Denis, J.-M. Primary alkynylphosphines and allenylphosphines. Inorg. Chem. 1991, 30, 2170–2173. [Google Scholar]
- Guillemin, J.-C.; Janati, T.; Denis, J.-M.; Guenot, P.; Savignac, P. Unstabilized 1-phosphaallenes: synthesis and characterization. Tetrahedron Lett. 1994, 35, 245–248. [Google Scholar] [CrossRef]
- Pilard, J.-F.; Baba, G.; Gaumont, A.-C.; Denis, J.-M. Reduction of phosphine oxides, a mild and practical synthesis of secondary P-ethynylphosphines. Synlett 1995, 1168–1170. [Google Scholar]
- Gao, Y.; Wang, G.; Chen, L.; Xu, P.; Zhao, Y.; Zhou, Y.; Han, L.-B. Copper-catalyzed aerobic oxidative coupling of terminal alkynes with H-phosphonates leading to alkynylphosphonates. J. Am. Chem. Soc. 2009, 131, 7956–7957. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bernoud, E.; Veillard, R.; Alayrac, C.; Gaumont, A.-C. Stoichiometric and Catalytic Synthesis of Alkynylphosphines. Molecules 2012, 17, 14573-14587. https://doi.org/10.3390/molecules171214573
Bernoud E, Veillard R, Alayrac C, Gaumont A-C. Stoichiometric and Catalytic Synthesis of Alkynylphosphines. Molecules. 2012; 17(12):14573-14587. https://doi.org/10.3390/molecules171214573
Chicago/Turabian StyleBernoud, Elise, Romain Veillard, Carole Alayrac, and Annie-Claude Gaumont. 2012. "Stoichiometric and Catalytic Synthesis of Alkynylphosphines" Molecules 17, no. 12: 14573-14587. https://doi.org/10.3390/molecules171214573
APA StyleBernoud, E., Veillard, R., Alayrac, C., & Gaumont, A.-C. (2012). Stoichiometric and Catalytic Synthesis of Alkynylphosphines. Molecules, 17(12), 14573-14587. https://doi.org/10.3390/molecules171214573