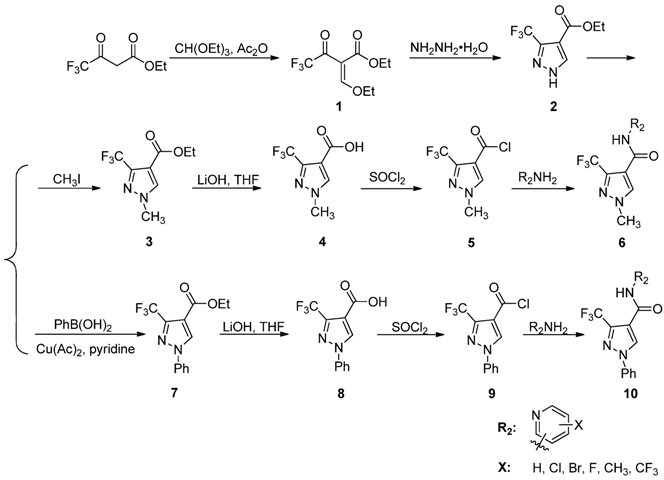
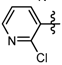
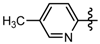
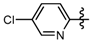
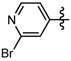
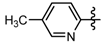
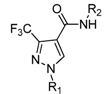
3.7. Preparation of the Target Compounds 6 and 10
To a solution of 2-amino-5-(trifluoromethyl) pyridine (163 mg, 1 mmol) and 80% sodium hydride (45 mg, 1.5 mmol) in anhydrous THF (10 mL), compound 5 (214 mg, 1 mmol) was added slowly, while the temperature was controlled under 10 °C, then reacted at ambient temperature for 10 h. The solvent was distilled off, and the residue was dissolved in ethyl acetate, washed with water and brine, dried over by anhydrous sodium sulfate. After the solvent was removed under reduced pressure, the residue was purified by column chromatography on silica gel (ethyl acetate/petroleum ether 1:15) to afford the target product, N-(5-(trifluoromethyl)-pyridin-2-yl)-1-methyl-3-(trifluoro-methyl)-1H-pyrazole-4-carboxamide (6a). The following compounds 6b–l and 10a–k was prepared from the corresponding starting materials in a similar manner to that described for 6a. The physical and spectral data for compounds 6b–l and 10a–k are listed below.
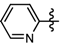
1-Methyl-3-(trifluoromethyl)-N-(5-(trifluoromethyl)pyridin-2-yl)-1H-pyrazole-4-carboxamide (6a): White solid, yield 86.4%, m.p. 128~130 °C. 1H-NMR (DMSO-d6) δ: 11.19 (s, 1H, NH), 8.76 (s, 1H, pyrazole H), 8.73 (1H, pyridine H), 8.34 (d, J = 8.6 Hz, 1H, pyridine H), 8.20 (d, J = 8.6 Hz, 1H, pyridine H), 3.97 (s, 3H, N-CH3); 13C-NMR (DMSO-d6): δ 160.03, 155.60, 145.82, 136.42, 136.07, 123.3, 122.40, 121.25, 120.27, 115.68, 114.21, 40.51; IR (KBr): ν 3305.9, 3121.8, 1681.9, 1591.1, 1542.3, 1525.6, 1051.2, 848.6, 752.5 cm−1; MS(ESI): m/z 339 [M+H]+; Anal. Calc. for C12H8F6N4O: C, 42.62; H, 2.38; N, 16.57. Found: C, 42.22; H, 2.05; N, 16.11.
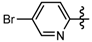
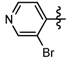
1-Methyl-N-(5-bromopyridin-2-yl)-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide (6b): White solid, yield 74.2%, m.p. 170~173 °C. 1H-NMR (DMSO-d6) δ: 10.91 (s, 1H, NH), 8.68 (s, 1H, pyrazole H), 8.49 (s, 1H, pyridine H), 8.12 (d, J = 9.15 Hz, 1H, pyridine H), 8.03 (d, J = 8.6 Hz, 1H, pyridine H), 3.94 (s, 3H, N-CH3); 13C-NMR (DMSO-d6): δ 159.67, 151.45, 149.03, 141.18, 135.81, 122.44, 120.30, 116.39, 115.94, 114.38, 40.48; IR (KBr): ν 3429.4, 3128.5, 3261.6, 1674.2, 1543.0, 1521.1, 1496.7, 1053.1, 835.1 cm−1; MS(ESI) m/z: 349 [M+H]+; Anal. Calc. for C11H8BrF3N4O: C, 37.84; H, 2.31; N, 16.05. Found: C, 37.38; H, 1.99; N, 15.75.
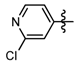
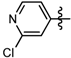
1-Methyl-N-(2-chloropyridin-4-yl)-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide (6c): White solid, yield 45.1%, m.p. 129~131 °C. 1H-NMR (DMSO-d6) δ:10.00 (s, 1H, NH), 8.59 (s, 1H, pyrazole H), 8.29 (1H, pyridine H), 8.07 (s, 1H, pyridine H), 7.49 (1H, pyridine H), 3.97 (s, 3H, N-CH3); 13C-NMR (DMSO-d6): δ 159.43, 147.06, 145.98, 136.80, 135.43, 132.02, 124.01, 122.40, 120.27, 115.79, 40.51; IR (KBr): ν 3284.7, 3121.1, 1660.7, 1657.7, 1548.8, 1541.1, 1521.8, 1456.2, 1496.7, 1078.21, 1051.1, 800 cm−1; MS(ESI): m/z 305 [M+H]+; Anal. Calc. for C11H8ClF3N4O: C, 43.37; H, 2.65; N, 18.39. Found: C, 43.11; H, 2.34; N, 17.93.
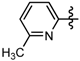
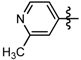
1-Methyl-N-(6-methylpyridin-2-yl)-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide (6d): Light yellow solid, yield 66.3%, m.p. 133~136 °C. 1H-NMR (DMSO-d6) δ: 10.61(s, 1H, NH), 8.66 (s, 1H, pyrazole H), 8.19 (m, 1H, pyridine H), 8.02 (d, J = 8.0 Hz, 1H, pyridine H), 7.63 (d, J = 8.0 Hz, 1H, pyridine H), 3.96 (s, 3H, N-CH3); 2.26 (s, 3H, CH3); 13C-NMR (DMSO-d6): δ 159.41, 150.27, 148.19, 139.05, 135.52, 129.21, 122.51, 120.36, 116.33, 114.32, 40.49, 17.80; IR (KBr): ν 3365.7, 3130.4, 1678.0, 1591.2, 1541.1, 1533.4, 1330.8, 1282.6, 869.9 cm−1; MS(ESI): m/z 285 [M+H]+; Anal. Calc. for C12H11F3N4O: C, 50.71; H, 3.90; N, 19.71. Found: C, 50.53; H, 3.65; N, 19.48.
1-Methyl-N-(pyridin-2-yl)-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide (6e): White solid, yield 75.6%, m.p. 159~162 °C. 1H-NMR (DMSO-d6) δ: 10.70 (s, 1H, NH), 8.68 (s, H, pyrazole H), 8.36 (d, J = 3.5 Hz, 1H, pyridine H), 8.13 (d, J = 8.6 Hz, 1H, pyridine H), 7.81 (t, J = 7.7 Hz, 1H, pyridine H), 7.15 (t, J = 6.3 Hz, 1H, pyridine H), 3.96 (s, 3H, CH3); 13C-NMR (DMSO-d6): δ 159.62, 152.46, 148.47, 140.13, 138.73, 135.65, 120.29, 116.25, 115.81, 114.78, 40.54; IR(KBr): ν 3312.1, 3226.9, 1683.8, 1579.7, 1541.1, 1527.6, 1506.4, 1055.0, 823.6, 790.8, 754.1 cm−1; MS(ESI): m/z 271 [M+H]+; Anal. Calc. for C11H9F3N4O: C, 48.89; H, 3.36; N, 20.73. Found: C, 48.44; H, 3.08; N, 20.22.
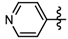
1-Methyl-N-(pyridin-4-yl)-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide (6f): White solid, yield 59.7%, m.p. 205~208 °C. 1H-NMR (DMSO-d6) δ:10.46 (s, 1H, NH), 8.57(s, H, pyrazole H), 8.46 (d, J = 1.7 Hz, 1H, pyridine H), 8.45 (d, J = 1.2 Hz, 1H, pyridine H), 7.66 (d, J = 1.2 Hz, 1H, pyridine H), 7.65 (d, J = 1.7 Hz, 1H, pyridine H), 3.99 (s, 3H, CH3); 13C-NMR (DMSO-d6): δ 159.94, 150.93, 146.11, 139.96, 135.37, 122.39, 116.31, 114.12, 40.53; IR (KBr): ν 3044.1, 3226.9, 1697.3, 1583.5, 1570.0, 1541.1, 1003.8, 835.1, 775.3 cm−1; MS(ESI): m/z 271 [M+H]+; Anal. Calc. for C11H9F3N4O: C, 48.89; H, 3.36; N, 20.73. Found: C, 48.56; H, 3.01; N, 20.39.
1-Methyl-N-(4-methylpyridin-2-yl)-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide (6g): Light yellow solid, yield 47.8%, m.p. 138~141 °C, 1H-NMR (DMSO-d6) δ: 10.61 (s, 1H, NH), 8.41 (s, 1H, pyrazole H), 8.19 (s, 1H, pyridine H), 7.71 (d, J = 5.2 Hz, 1H, pyridine H), 7.17 (d, J = 5.2 Hz, 1H, pyridine H), 3.89 (s, 3H, N-CH3); 2.30 (s, 3H, CH3); 13C-NMR (DMSO-d6): δ 160.04, 152.82, 150.68, 140.73, 136.64, 124.80, 119.95, 116.56, 115.15, 113.91, 40.49, 21.12; IR (KBr): ν 3363.8, 3112.5, 1670.3, 1575.8, 1541.1, 1506.4, 1456.2, 1055.0, 873.7, 825.5, 752.4 cm−1; MS(ESI): m/z 285 [M+H]+; Anal. Calc. for C12H11F3N4O: C, 50.71; H, 3.90; N, 19.71. Found: C, 50.27; H, 3.66; N, 19.54.
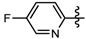
1-Methyl-N-(5-fluoropyridin-2-yl)-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide (6h): Yellow solid, yield 82.4%, m.p. 135~137 °C. 1H-NMR (DMSO-d6) δ: 10.83 (s, 1H, NH), 8.65 (s, H, pyrazole H), 8.37(d, 1H, pyridine H), 8.16 (1H, pyridine H), 7.76 (1H, pyridine H), 3.96 (s, 3H, CH3); 13C-NMR (DMSO-d6): δ 160.07, 151.35, 147.66, 140.10, 137.88, 125.26, 120.23, 119.17, 118.52, 114.73, 40.50; IR (KBr): ν 3.454.5, 3136.2, 1670.3, 1541.1, 1521.8, 1489.0, 1392.6, 1055.0, 840.1, 771.2 cm−1; MS(ESI): m/z 289 [M+H]+; Anal. Calc. for C11H8F4N4O: C, 45.84; H, 2.80; N, 19.44. Found: C, 45.56; H, 2.63; N, 19.14.
1-Methyl-N-(2-chloropyridin-3-yl)-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide (6i): Light yellow solid, yield 33.3%, m.p. 107~109 °C. 1H-NMR (DMSO-d6) δ: 10.25 (s, 1H, NH), 8.48 (s, 1H, pyrazole H), 8.34 (d, J = 7.5 Hz,1H, pyridine H), 8.03 (d, J = 8.0 Hz, 1H, pyridine ), 7.59 (q, J = 4.0 Hz, 1H, pyridine H), 3.89 (s, 3H, CH3); 13C-NMR (DMSO-d6): δ 163.57, 150.55, 149.23, 140.76, 136.72, 133.98, 125.19, 121.98, 119.84, 115.68, 40.51; IR (KBr): ν 3415.5, 3127.1, 1691.5, 1585.4, 1541.1, 1514.1, 1490.9, 1037.7, 815.8, 763.8, 732.9 cm−1; MS(ESI): m/z 305 [M+H]+; Anal. Calc. for C11H8ClF3N4O: C, 43.37; H, 2.65; N, 18.39. Found: C, 42.88; H, 2.14; N, 18.25.
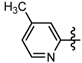
1-Methyl-N-(5-methylpyridin-2-yl)-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide (6j): Yellow solid, yield 43.9%, m.p. 135~138 °C. 1H-NMR (DMSO-d6) δ: 10.34 (s, 1H, NH), 8.80 (s, 1H, pyrazole H), 8.53 (s, 1H, pyridine H), 8.29 (d, J = 7.2 Hz, 1H, pyridine H), 8.11 (d, J = 7.2 Hz, 1H, pyridine H), 3.99 (s, 3H, N-CH3); 2.26 (s, 3H, CH3); 13C-NMR (DMSO-d6): δ 159.55, 151.23, 145.11, 141.89, 139.82, 135.11, 127.36, 124.18, 116.48, 115.94, 40.50, 24.37; IR (KBr): ν 3545.1, 3105.3, 1653.0, 1589.3, 1506.4, 1496.7, 1473.6, 1039.6, 891.1, 808.7, 789.9. cm−1; MS(ESI): m/z 285 [M+H]+; Anal. Calc. for C12H11F3N4O: C, 50.71; H, 3.90; N, 19.71. Found: C, 50.43; H, 3.77; N, 19.62.
1-Methyl-N-(5-chloropyridin-2-yl)-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide (6k): White solid, yield 30.1%, m.p. 138~140 °C, 1H-NMR (DMSO-d6) δ: 10.14 (s, 1H, NH), 8.41 (s, 1H, pyrazole H), 8.15 (d, J = 9.2 Hz, 1H, pyridine H), 8.10 (s, 1H, pyridine H), 7.94 (d, J = 7.5 Hz, 1H, pyridine H), 4.03 (s, 3H, NCH3); 13C-NMR (DMSO-d6): δ 160.28, 150.99, 146.92, 139.30, 138.52, 126.18, 121.25, 119.95, 119.10, 115.84, 40.52; IR (KBr): ν 3282.8, 2964.5, 1701.2, 1575.8, 1525.6, 1456.2, 1292.8, 1155.3, 1006.8, 837.1, 736.8 cm−1; MS(ESI): m/z 305 [M+H]+; Anal. Calc. for C11H8ClF3N4O: C, 43.37; H, 2.65; N, 18.39. Found: C, 43.02; H, 2.18; N, 18.12.
1-Methyl-N-(2-bromopyridin-4-yl)-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide (6l): White solid, yield 56.4%, m.p. 174~176 °C. 1H-NMR (DMSO-d6) δ: 10.67 (s, 1H, NH), 8.57 (s, 1H, pyrazole H), 8.29 (d, J = 5.8 Hz, 1H, pyridine H), 7.84 (s, 1H, pyridine H), 7.59 (d, J = 5.2 Hz, 1H, pyridine H), 3.99 (s, 3H, CH3); 13C-NMR (DMSO-d6): δ 160.05, 151.48, 150.95, 148.69, 135.55, 122.32, 120.17, 115.85, 113.53, 113.40, 40.50; IR (KBr): ν 3330.5, 3115.0, 1699.2, 1587.4, 1541.1, 1489.0, 1049.2, 842.1, 772.1 cm−1; MS(ESI): m/z 349 [M+H]+; Anal. Calc. for C11H8BrF3N4O: C, 37.84; H, 2.31; N, 16.05. Found: C, 37.39; H, 2.11; N, 15.87.
N-(Pyridin-4-yl)-1-phenyl-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide (10a): White solid, yield 80.5%, m.p. 244~245 °C. 1H-NMR (DMSO-d6) δ: 10.63 (s, 1H, NH), 9.35 (s, 1H, pyrazole H), 8.50 (d, J = 6.3 Hz, 2H, pyridine H), 7.89 (d, J = 8.1 Hz, 2H, benzene H), 7.69 (d, J = 6.3 Hz, 2H, pyridine H), 7.64 (t, J = 8.1 Hz, 2H, benzene H), 7.51 (t, J = 7.5 Hz, 1H, benzene H); 13C-NMR (DMSO-d6): δ 159.72, 151.04, 145.94, 141.81, 138.76, 136.44, 132.74, 130.54, 129.04, 120.10, 117.99, 114.14; IR (KBr): ν 3419.7, 3290.5, 2978.0, 1716.6, 1683.8, 1624.0, 1543.0, 1508.3, 1400.2, 1284.5 cm−1; MS(ESI): m/z 333 [M+H]+; Anal. Calc. for C16H11F3N4O: C, 57.83; H, 3.34; N, 16.86. Found: C, 57.65; H, 3.12; N, 16.54.
N-(2-Chloropyridin-4-yl)-1-phenyl-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide (10b): White solid, yield 16.5%, m.p. 216~218 °C. 1H-NMR (DMSO-d6) δ: 10.23 (s, 1H, NH), 8.69 (s, 1H, pyrazole H), 8.51 (d, J = 5.8 Hz, 1H, pyridine H), 7.76 (d, J = 5.8 Hz, 1H, pyridine H), 7.58 (d, J = 8.1 Hz, 2H, benzene H), 7.43 (t, J = 7.6 Hz, 2H, benzene H), 7.51 (t, J = 7.5 Hz, 1H, benzene H), 7.36 (s, 1H, pyridine H); 13C-NMR (DMSO-d6): δ 159.35, 159.21, 149.78, 145.69, 138.66, 132.23, 129.97, 129.12, 125.54, 122.14, 120.04, 116.75, 114.03, 110.28; IR (KBr): ν 3444.8, 3383.1, 1701.2, 1653.0, 1577.7, 1490.9, 1356.8, 1298.0, 765.7 cm−1; MS(ESI): m/z 367 [M+H]+; Anal. Calc. for C16H10ClF3N4O: C, 52.40; H, 2.75; N, 15.28. Found: C, 52.22; H, 2.59; N, 15.04.
N-(2-Bromopyridin-4-yl)-1-phenyl-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide (10c): White solid, yield 26.9%, m.p. 235~236 °C. 1H-NMR (DMSO-d6) δ: 10.83 (s, 1H, NH), 9.35 (s, 1H, pyrazole H), 8.35 (d, J = 5.7 Hz, 1H, pyridine H), 7.89 (s, 1H, pyridine H), 7.87 (d, J = 3.4 Hz, 2H, benzene H), 7.65–7,61 (m, 3H, benzene 2H and pyridine H), 7.51 (t, J = 7.2 Hz, 1H, benzene H); 13C-NMR (DMSO-d6): δ 159.86, 151.57, 151.12, 148.51, 143.56, 138.70, 132.98, 130.58, 129.15, 120.15, 117.56, 116.72, 113.56, 113.44; IR (KBr): ν 3365.8, 3111.1, 3066.8, 2922.1, 2850.7, 1705.0, 1579.7, 1543.0, 1494.8, 1386.8, 1298.0, 1043.4, 688.5 cm−1; MS(ESI): m/z411 [M+H]+, 412 [M+2H]+; Anal. Calc. for C16H10BrF3N4O: C, 46.74; H, 2.45; N, 13.63. Found: C, 46.34; H, 2.18; N, 13.47.
N-(Pyridin-2-yl)-1-phenyl-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide (10d): White solid, yield 40.5%, m.p. 149~151 °C. 1H-NMR (DMSO-d6) δ: 10.85 (s, 1H, NH), 9.52 (s, 1H, pyrazole H), 8.23 (d, J = 8.0 Hz, 1H, benzene H), 7.87–7.49 (m, 9 H, pyridine and benzene H); 13C-NMR (DMSO-d6): δ 159.33, 152.36, 148.50, 138.81, 133.44, 132.97, 130.39, 128.82, 120.39, 120.35, 119.96, 118.36, 117.75, 114.64; IR (KBr): ν 3130.4, 3068.7, 2987.7, 2927.7, 1695.4, 1600.9, 1490.9, 1435.0, 1307.7 cm−1; MS(ESI): m/z 333 [M+H]+; Anal. Calc. for C16H11F3N4O: C, 57.83; H, 3.34; N, 16.86. Found: C, 57.66; H, 3.04; N, 16.49.
N-(3-Chloropyridin-4-yl)-1-phenyl-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide (10e): White solid, yield 74.9%, m.p. 146~147 °C. 1H-NMR (DMSO-d6) δ: 10.08 (s, 1H, NH), 9.41 (s, 1H, pyridine H), 8.69 (s, 1H, pyrazole H), 8.51 (d, J = 5.7 Hz, 1H, pyridine H), 7.99 (d, J = 5.7 Hz, 1H, pyridine H), 7.91 (d, J = 8.0 Hz, 2H, benzene H), 7.64 (t, J = 7.8 Hz, 2H, benzene H), 7.51 (t, J = 7.5 Hz, 1H, benzene H); 13C-NMR (DMSO-d6): δ 159.46, 150.34, 149.26, 142.23, 138.76, 133.26, 130.49, 129.04, 123.34, 120.22, 119.04, 118.56, 117.31, 110.23; IR (KBr): ν 3387.0, 3101.5, 3034.0, 1707.0, 1581.6, 1496.7, 1406.1, 1294.2, 763.8 cm−1; MS(ESI): m/z 367 [M+H]+; Anal. Calc. for C16H10ClF3N4O: C, 52.40; H, 2.75; N, 15.28. Found: C, 51.98; H, 2.54; N, 14.96.
N-(3-Bromopyridin-4-yl)-1-phenyl-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide (10f): White solid, yield 69.7%, m.p. 136~138 °C. 1H-NMR (DMSO-d6) δ: 9.98 (s, 1H, NH), 9.39 (s, 1H, pyridine H), 8.80 (s, 1H, pyrazole H), 8.53 (d, J = 5.2 Hz, 1H, pyridine H), 7.90–7.87 (m, 3H, pyridine and benzene H), 7.63 (d, J = 8.0 Hz, 2H, benzene H), 7.64 (t, J = 7.8 Hz, 2H, benzene H), 7.51 (t, J = 6.9 Hz, 1H, benzene H); 13C-NMR (DMSO-d6): δ 159.26, 152.92, 149.69, 143.61, 138.76, 133.09, 130.49, 129.05, 122.21, 120.24, 120.07, 117.37, 114.93, 110.58; IR (KBr): ν 3400.5, 3325.2, 2924.0, 1670.3, 1579.7, 1494.8, 1406.8, 1235.3, 682.8 cm−1; MS(ESI): m/z 411 [M+H]+; Anal. Calc. for C16H10BrF3N4O: C, 46.74; H, 2.45; N, 13.63. Found: C, 46.57; H, 2.33; N, 13.47.
N-(3-Methylpyridin-4-yl)-1-phenyl-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide (10g): White solid, yield 65.3%, m.p. 207~208 °C. 1H-NMR (DMSO-d6) δ: 9.83 (s, 1H, NH), 9.35 (s, 1H, pyridine H), 8. 44 (s, 1H, pyrazole H), 8.38 (d, J = 5.2 Hz, 1H, pyridine H), 7.90 (d, J = 8.0 Hz, 2H, benzene H), 7.73 (d, J = 5.2 Hz, 1H, benzene H), 7.63 (t, J = 6.9 Hz, 2H, benzene H), 7.51 (d, J = 6.3 Hz, 1H, pyridine H), 2.31 (s, 3H, CH3); 13C-NMR (DMSO-d6): δ 159.39, 152.05, 148.40, 143.70, 138.83, 132.86, 130.48, 128.97, 125.91, 120.16, 118.07, 117.78, 114.93, 111.23, 15.33; IR (KBr): ν 3244.2, 3116.9, 3030.1, 1699.2, 1581.6, 1544.9, 1490.9, 1409.9, 1300.0 cm−1; MS(ESI): m/z347 [M+H]+; Anal. Calc. for C17H13F3N4O: C, 58.96; H, 3.78; N, 16.18. Found: C, 58.66; H, 3.59; N, 16.01.
N-(2-Methylpyridin-4-yl)-1-phenyl-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide (10h): White solid, yield 60.9%, m.p. 196~197 °C. 1H-NMR (DMSO-d6) δ: 10.53 (s, 1H, NH), 9.34 (s, 1H, pyridine H), 8.37 (s, 1H, pyrazole H), 7.88 (d, J = 8.0 Hz, 1H, pyridine H), 7.64–7.62 (m, 3H, pyridine and benzene H), 7.50 (d, J = 9.0 Hz, 2H, benzene H), 2.46 (s, 3H, CH3); 13C-NMR (DMSO-d6): δ 159.64, 159.33, 150.27, 146.22, 138.76, 132.67, 130.54, 129.03, 126.12, 122.34, 120.10, 118.04, 113.03, 111.58, 24.84; IR (KBr): ν 3261.6, 3115.0, 3066.8, 1683.8, 1604.7, 1588.4, 1490.9, 1409.9, 1301.9 cm−1; MS(ESI): m/z 347 [M+H]+; Anal. Calc. for C17H13F3N4O: C, 58.96; H, 3.78; N, 16.18. Found: C, 58.67; H, 3.63; N, 15.94.
N-(Pyridin-3-yl)-1-phenyl-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide (10i): White solid, yield 79.8%, m.p. 196~197 °C. 1H-NMR (DMSO-d6) δ: 10.50 (s, 1H, NH), 9.32 (s, 1H, pyridine H), 8.84 (s, 1H, pyrazole H), 8.33 (d, J = 3.5 Hz, 1H, pyridine H), 8.14 (d, J = 8.6 Hz, 1H, pyridine H), 7.88 (d, J = 7.5 Hz, 2H, benzene H), 7.63 (t, J = 8.0 Hz, 2H, benzene H), 7.50 (t, J = 7.5 Hz, 1H, benzene H), 7.42 q, J = 7.5 Hz, 1H, pyridine H); 13C-NMR (DMSO-d6): δ 161.11, 159.32, 145.34, 141.85, 138.79, 135.92, 132.50, 130.55, 129.00, 127.34, 124.30, 122.28, 120.07, 118.18; IR (KBr): ν 3259.7, 3122.7, 3066.8, 1695.4, 1653.0, 1600.9, 1490.9, 1419.6, 1303.8 cm−1; MS(ESI): m/z 333 [M+H]+; Anal. Calc. for C16H11F3N4O: C, 57.83; H, 3.34; N, 16.86. Found: C, 57.75; H, 3.16; N, 16.62.
N-(5-Chloropyridin-2-yl)-1-phenyl-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide (10j): White solid, yield 67.8%, m.p. 101~102 °C. 1H-NMR (DMSO-d6) δ: 10.17 (s, 1H, NH), 9.36 (s, 1H, pyrazole H), 8.33 (d, J = 4.6 Hz, 1H, pyridine H), 8.13 (d, J = 8.1 Hz, 1H, pyridine H), 7.90 (d, J = 8.1 Hz, 2H, benzene H), 7.64 (t, J = 7.7 Hz, 2H, benzene H), 7.54–7.50 (m, 2H, benzene and pyrazole H); 13C-NMR (DMSO-d6): δ 160.97, 159.36, 146.94, 138.57, 135.39, 133.14, 130.49, 130.28, 129.00, 126.14, 120.30, 117.44, 115.71, 113.84; IR (KBr): ν 3421.7, 2926.0, 1705.0, 1653.0, 1575.8, 1506.4, 1386.8, 1298.0, 765.7 cm−1; MS(ESI): m/z 367 [M+H]+; Anal. Calc. for C16H10ClF3N4O: C, 52.40; H, 2.75; N, 15.28. Found: C, 52.03; H, 2.61; N, 14.98.
N-(5-Methylpyridin-2-yl)-1-phenyl-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide (10k): White solid, yield 64.4%, m.p. 182~183 °C. 1H-NMR (DMSO-d6) δ: 11.17 (s, 1H, NH), 9.36 (s, 1H, pyrazole H), 8.33 (d, J = 4.6 Hz, 1H, pyridine H), 8.14 (d, J = 8.1 Hz, 2H, benzene H), 7.90 (d, J = 8.1 Hz, 2H, benzene H), 7.64–7.52 (m, 3H, benzene and pyridine H); 13C-NMR (DMSO-d6): δ 159.32, 147.12, 145.62, 138.80, 136.49, 132.76, 131.89, 130.51, 129.02, 124.07, 120.20, 117.48, 116.13, 112.32, 14.54; IR (KBr): ν 3282.8, 2918.3, 1654.9, 1544.9, 1490.9, 1388.7, 1224.8, 1080.1 cm−1; MS(ESI): m/z 347 [M+H]+; Anal. Calc. for C17H13F3N4O: C, 58.96; H, 3.78; N, 16.18. Found: C, 58.78; H, 3.55; N, 16.02.