Mixed Biopolymer Systems Based on Starch
Abstract
:1. Introduction
2. Starch–Starch/Starch–Flour Systems
| Type of starch mixture | Experiment | Observation | Ref. |
|---|---|---|---|
| β-glucans–starch mixtures | Four β-glucan preparations, i.e., curdlan (CL), oat (OG), barley (BG) and yeast (YG) β-glucans, were compared for their effects on the gelatinization and retrogradation of rice starch (RS). | ● The addition of any of these β-glucans significantly increased the peak, breakdown, final, and setback viscosities of RS, whereas the pasting temperatures were significantly decreased by OG or CL addition, but were unaffected by BG or YG addition. ● β-glucans had a negligible effect on the onset (To), peak (Tp), and conclusion (Tc) temperatures but slightly decreased the gelatinization enthalpy (  H1) of RS. Storage of all the gels at 4 °C resulted in a marked decrease in the To, Tp, Tc, and melting enthalpy ( H1) of RS. Storage of all the gels at 4 °C resulted in a marked decrease in the To, Tp, Tc, and melting enthalpy (  H2) values. H2) values. | [11] |
| Milk protein–polysaccharide mixtures | Concentration of whey protein concentrate (WPC) at 1.0% and the pH 7.0 were mixed with commercial polysaccharides (PS) in concentration of 0.0–1.0%. Interactions between WPC and PS in the aqueous phase were evaluated. | ● The results revealed differences in the molecular dynamics of mixed systems. ● The nature of the interactions between WPC and PS depended on the PS type, its relative concentration in the aqueous phase and also on the two WPC fractions. ● Whey protein concentrate/sodium alginate (WPC/SA) mixed systems were distinguished by a tendency to protein aggregation in the aqueous phase and their segregation into separated microdomains. ● WPC/λ–carrageenan (WPC/λ–C) mixed systems showed high degree of attractive interactions over the whole range of concentrations. | [12] |
| Hydrocolloid–flour mixtures in batter systems | The functionalities of hydrocolloid–flour mixtures in terms of the thermal properties of their resulting batter systems were investigated. Gelatinization temperature (TG), total enthalpies of gelatinization (ΔHG), glass transition temperature (Tg), melting peak temperature (Tm), and total melting enthalpies (ΔHm) and the effects of different thermal processes such as cooking-freezing-thawing (CFT) and freezing-cooking (FC) on thermal properties of the various batter systems were determined. | ● The different thermal processes did not significantly affect either TG or ΔHG of batter systems, but they influenced the glass transition behavior and the ΔHm of batter systems. ● The thermal processes also showed different effects on the batter systems containing different hydrocolloids such as methylcellulose (MC), carboxymethyl–cellulose (CMC), and xanthan gum (XG). ● The hydrocolloids shifted TG upwards, depressed Tg, and increased Tm of batters. ● The effect of these hydrocolloids on glass transition temperature was more pronounced in raw samples (FC process) than in cooked samples and increased with increasing levels of CMC and MC used in the formulations. ● Batters with MC showed increased ΔHm for all the thermal processes. | [13] |
| Non-waxy rice starch–hydrocolloid mixtures | The swelling and pasting properties of non-waxy rice starch–hydrocolloid mixtures were investigated using commercial and laboratory-generated hydrocolloids at low concentration of (0–0.1%). | ●Hydrocolloids enhanced the trough and final viscosity of rice starch dispersions. ● They also lowered peak viscosity of rice starch dispersions. ● Total setback viscosity appeared to be not affected by hydrocolloids at low concentration (0.05%). ● The hot and cold paste of the starch–gellan gum mixture exhibited the highest viscosity values. | [14] |
| Rice starch–β-glucan mixtures | Rice starch (RS)-β-glucan (BG) mixture were investigated as functions of mixing ratio and of storage time at concentrations of RS/BG = 6.0/0.0, 5.7/0.3, 5.4/0.6, and 5.0/1.0. | ●An increase in onset (To), peak (Tp), and conclusion (Tc) temperatures and a decrease in gelatinization enthalpy (ΔH1) with increasing BG concentration. ● Storage of the mixed gels at 4 °C resulted in a decrease in To, Tp, Tc, and melting enthalpy (ΔH2). ● The retrogradation ratio (ΔH2/ΔH1) and the phase transition temperature range (Tc−To) of the mixed gels increased with storage time. ● BG addition also slowed the syneresis of the mixed gels. ● The added BG also retarded the development of gel hardness during refrigerated storage of the RS/BG mixed gels. | [15] |
| Rice starch–hydrocolloid mixtures | Dynamic viscoelastic and steady flow properties of the freshly prepared pastes of starch alone and starch–hydrocolloid mixtures; cellulose derivatives and carrageenans were determined after holding at room temperature (~25 °C) for 1 h. | ● Increases in apparent pasting temperatures and peak and final viscosities in the following decreasing order were observed: Methylcellulose > carboxymethyl–cellulose for cellulose derivatives and λ– > í– > κ–carregeenan for carrageenans. ● Slight decreases in peak and final viscosities were observed when hydroxypropyl–methylcellulose was the hydrocolloid. | [16] |
| Corm starch–guar gum mixtures | Gelatinization behavior of corn starch was studied in the presence or absence of various guar gum samples with different molecular weights in order to clarify the difference in functions of each guar to starch. | ● Guars with Mw values higher than 12.2 × 105 g/mol shifted the onset of viscosity increase for the system to lower temperatures and increased its peak viscosity upon heating at a relatively low starch concentration. ● The earlier onset of viscosity increase was independent of Mw of guar, while the increase in peak viscosity was dependent on its Mw. ● These guars shifted the onset of viscosity increase for the system upward, on the contrary, at a relatively high starch concentration (e.g., 15%). | [17] |
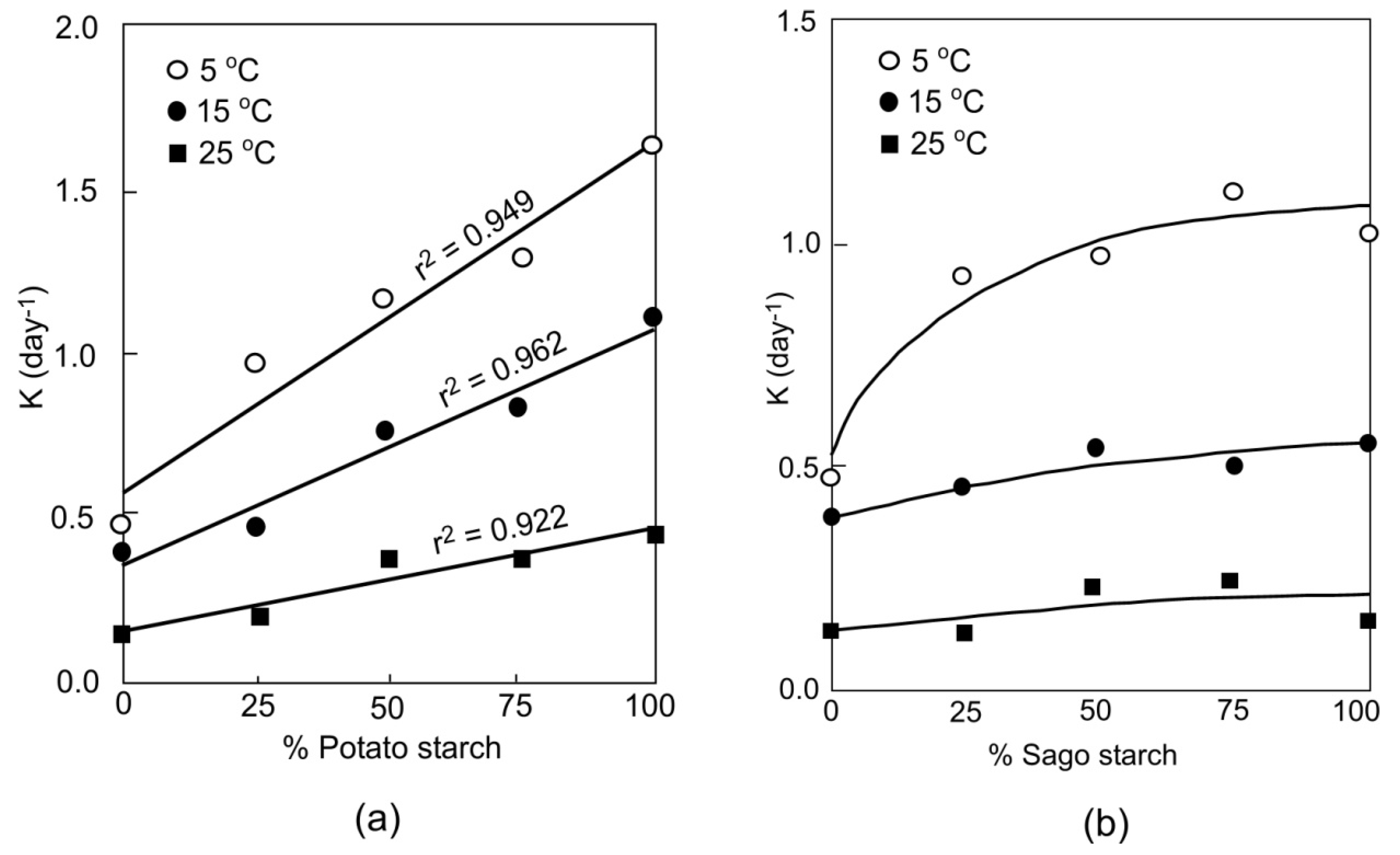
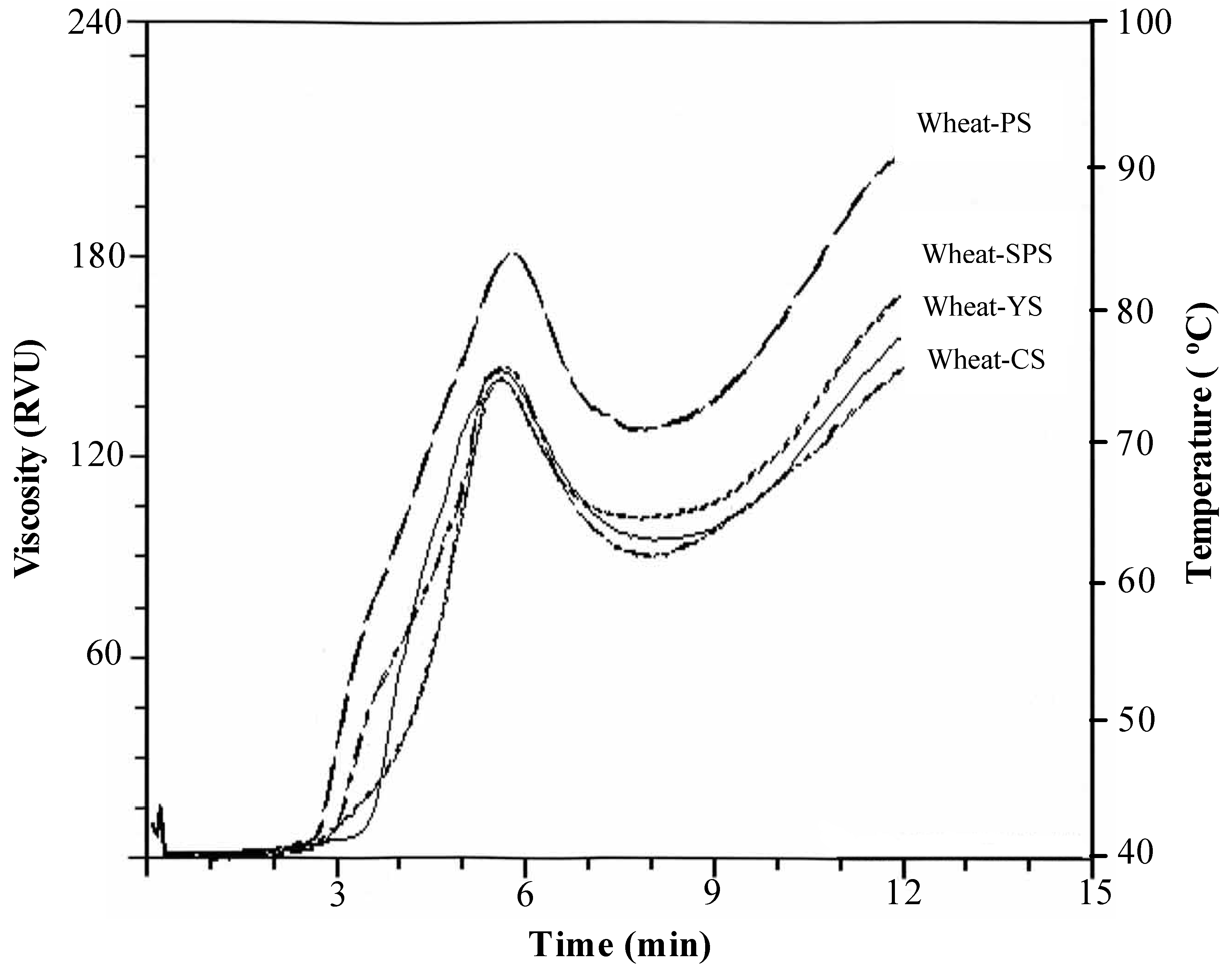
3. Starch–Protein Systems
| Starch/treatment | peak viscosity (RVU) a | final viscosity (RVU) a | Peak temp. (°C) | |||||
|---|---|---|---|---|---|---|---|---|
| ΔPV b | ΔFV b | ΔPT b | ||||||
| waxy corn | 244 | 115 | 80 | |||||
| waxy corn caseinate | 393 | 149 | 208 | 93 | 83 | 3 | ||
| potato | 760 | 261 | 72 | |||||
| potato caseinate | 389 | −371 | 362 | 101 | 95 | 23 | ||
| cassava | 281 | 168 | 88 | |||||
| cassava caseinate | 403 | 122 | 287 | 119 | 91 | 3 | ||
| wheat | 122 | 131 | 95 | |||||
| wheat caseinate | 148 | 26 | 269 | 138 | 95 | 0 | ||
| corn | 141 | 140 | 95 | |||||
| corn caseinate | 206 | 65 | 328 | 188 | 95 | 0 | ||
| rice | 96 | 159 | 95 | |||||
| rice caseinate | 123 | 27 | 251 | 92 | 95 | 0 | ||
| P levels c | ||||||||
| starch | <0.001 | <0.001 | <0.001 | |||||
| caseinate | 0.0308 | <0.001 | <0.001 | |||||
| starch × caseinate | <0.001 | <0.001 | <0.001 | |||||
4. Starch–Hydrocolloid Systems
5. Directions for Future Study
6. Conclusions and Future Outlook
References and Notes
- Biliaderis, C.G. Structures and Phase Transitions of Starch Polymers. In Polysaccharide Association Structures in Food; Walter, R.H., Ed.; Marcel Dekker: New York, NY, USA, 1998; pp. 57–168. [Google Scholar]
- Tolstoguzov, V.B. Some thermodynamic considerations in food formulation. Food Hydrocolloid. 2003, 17, 1–23. [Google Scholar] [CrossRef]
- Okechukwu, P.E.; Anandha Rhao, M. Rheology of structured polysaccharide food systems: Starch and pectin. In Polysaccharide Association Structures in Food; Walter, R.H., Ed.; Marcel Dekker: New York, NY, USA, 1998; pp. 289–328. [Google Scholar]
- Obani, M.; BeMiller, J.N. Properties of some starch blends. Cereal Chem. 1997, 74, 431–436. [Google Scholar] [CrossRef]
- Piculell, L.; Bergfeldt, K.; Nilsson, S. Factors determining phase behaviour of multi component polymer systems. In Biopolymer Mixtures; Harding, S.E., Hill, S.E., Mitchell, J.R., Eds.; Nottingham NottinghUniversity Press: Nottingham, UK, 1995; pp. 13–35. [Google Scholar]
- Doublier, J.-L.; Garnier, C.; Renard, D.; Sanchez, C. Protein–polysaccharide interactions. Curr. Opin. Colloid Interface Sci. 2000, 5, 202–214. [Google Scholar] [CrossRef]
- de Kruif, C.G.; Tuinier, R. Polysaccharide protein interactions. Food Hydrocolloid. 2001, 15, 555–563. [Google Scholar] [CrossRef]
- Turgeon, S.L.; Beaulieu, M.; Schmitt, C.; Sanchez, C. Protein-polysaccharide interactions: Phase-ordering kinetics, thermodynamic and structural aspects. Curr. Opin. Colloid Interface Sci. 2003, 8, 401–414. [Google Scholar] [CrossRef]
- Tolstoguzov, V.B. Some physic-chemical aspects of protein processing in foods. Multicomponent gels. Food Hydrocolloid. 1995, 9, 317–332. [Google Scholar] [CrossRef]
- Appleqvist, I.A.M.; Debet, M.R.M. Starch-biopolymer interactions—A review. Food Rev. Int. 1997, 13, 163–224. [Google Scholar] [CrossRef]
- Banchathanakij, R.; Suphantharika, M. Effect of different β-glucans on the gelatinisation and retrogradation of rice starch. Food Chem. 2009, 114, 5–14. [Google Scholar] [CrossRef]
- Perez, A.A.; Carlos, R.; Carrara, C.C.; Sánchez, C.C.; Rodríguez, P.J.M.; Santiago, L.G. Interactions between milk whey protein and polysaccharide in solution. Food Chem. 2009, 116, 104–113. [Google Scholar] [CrossRef]
- Xue, J.; Ngadi, M. Effects of methylcellulose, xanthan gum and carboxymethylcellulose on thermal properties of batter systems formulated with different flour combinations. Food Hydrocolloid. 2009, 23, 286–295. [Google Scholar] [CrossRef]
- Song, J.Y.; Kwon, J.Y.; Choi, J.; Kim, Y.C.; Shin, M. Pasting properties of non-waxy rice starch-hydrocolloid mixtures. Starch/Stärke 2006, 58, 223–230. [Google Scholar] [CrossRef]
- Satrapai, S.; Suphantharika, M. Influence of spent brewer’s yeast β-glucan on gelatinization and retrogradation of rice starch. Carbohydr. Polym. 2007, 67, 500–510. [Google Scholar] [CrossRef]
- Techawipharat, J.; Suphantharika, M.; BeMiller, J.N. Effects of cellulose derivatives and carrageenans on the pasting, paste, and gel properties of rice starches. Carbohydr. Polym. 2008, 73, 417–426. [Google Scholar] [CrossRef]
- Funami, T.; Kataoka, Y.; Omoto, T.; Goto, Y.; Asai, I.; Nishinari, K. Effects of non-ionic polysaccharides on the gelatinization and retrogradation behavior of wheat starch. Food Hydrocolloid. 2005, 19, 1–13. [Google Scholar] [CrossRef]
- Ortega–Ojeda, F.E.; Larsson, H.; Eliasson, A.C. Gel formation in mixtures of hydrophobically modified potato and high amylopectin potato starch. Carbohydr. Polym. 2005, 59, 313–327. [Google Scholar] [CrossRef]
- Teo, C.H.; Seow, C.C. A pulsed NMR method for the study of starch retrogradation. Starch/Stärke 1992, 44, 288–292. [Google Scholar] [CrossRef]
- Karim, A.A.; Norziah, M.H.; Seow, C.C. Methods for the study of starch retrogradation. Food Chem. 2000, 71, 9–36. [Google Scholar] [CrossRef]
- Karim, A.A.; Teo, C.H.; Norziah, M.H.; Seow, C.C. Retrogradation behaviour of binary starch mixtures of rice starch with other types of starches. In Starch: Advance in Structure and Function; Barsby, T.L., Donald, A.M., Frazier, P.J., Eds.; Royal Society of Chemistry: Cambridge, UK, 2001; pp. 59–66. [Google Scholar]
- Yao, Y.; Zhang, J.M.; Ding, X. Retrogradation of starch mixtures containing rice starch. J. Food Sci. 2003, 68, 260–265. [Google Scholar] [CrossRef]
- Ortega–Ojeda, F.E.; Eliasson, A.C. Gelatinization and retrogradation behavior of some starch mixtures. Starch/Stärke 2001, 53, 520–529. [Google Scholar] [CrossRef]
- Zaidul, I.S.M.; Nik Norulaini, N.A.; Mohd Omar, A.K.; Yamauchi, H.; Noda, T. RVA analysis of mixtures of wheat flour and potato, sweet potato, yam, and cassava starches. Carbohydr. Polym. 2007, 69, 784–791. [Google Scholar] [CrossRef]
- Zaidul, I.S.M.; Karim, A.A.; Manan, D.M.A.; Norulaini, N.A.N.; Omar, A.K.M. Gelatinization properties of sago and wheat flour mixtures. ASEAN Food J. 2003, 12, 199–209. [Google Scholar]
- Zaidul, I.S.M.; Yamauchi, H.; Kim, S.J.; Hashimoto, N.; Noda, T. RVA study of mixtures of wheat flour and potato starches with different phosphorus content. Food Chem. 2007, 102, 1105–1111. [Google Scholar] [CrossRef]
- Hermansson, A.M.; Svegmark, K.C. Development in the understanding of starch functionality. Trends Food Sci. Technol. 1996, 7, 345–353. [Google Scholar] [CrossRef]
- Yang, H.; Irudayaraj, J.; Otgonchimeg, S.; Walsh, M. Rheological study of starch and dairy ingredient-based food systems. Food Chem. 2004, 86, 571–578. [Google Scholar] [CrossRef]
- Niwa, E. The chemistry of surimi gelation. In Surimi Technology; Lanier, T.C., Lee, C.M., Eds.; Marcel Dekker: New York, NY, USA, 1992; pp. 389–427. [Google Scholar]
- Bertolini, A.C.; Creamer, L.K.; Eppink, M.; Boland, M. Some rheological properties of sodium caseinate-starch gels. J. Agric. Food Chem. 2005, 53, 2248–2254. [Google Scholar] [CrossRef]
- Wang, B.; Wang, L.; Li, D.; Bhandari, B.; Wu, W.; Shi, J.; Chen, X.D.; Mao, Z. Effects of potato starch addition and cooling rate on rheological characteristics of flaxseed protein concentrate. J. Food Eng. 2009, 91, 392–401. [Google Scholar] [CrossRef]
- Oh, H.E.; Anema, S.; Wong, G.M.; Pinder, D.N.; Hemar, Y. Effect of potato starch addition on the acid gelation of milk. Int. Dairy J. 2007, 17, 808–815. [Google Scholar] [CrossRef]
- Li, J.Y.; Yeh, A.I.; Fan, K.L. Gelation characteristics and morphology of corn starch/soy protein concentrate composites during heating. J. Food Eng. 2007, 78, 1240–1247. [Google Scholar] [CrossRef]
- Ryan, K.J.; Brewer, M.S. In situ examination of starch granule-soy protein and wheat protein interactions. Food Chem. 2007, 104, 619–629. [Google Scholar] [CrossRef]
- Eliasson, A.C. On the surface activity of wheat proteins. In Gluten Proteins; Association of cereal research: Detmold, Germany, 1993; pp. 198–206. [Google Scholar]
- Carballo, J.; Femandez, P.; Barreto, G.; Solas, M.T.; Colmenro, F.J. Morphology and texture of bologna sausage as related to content of fat, starch and egg white. J. Food Sci. 1996, 61, 652–655. [Google Scholar] [CrossRef]
- Chung, K.H.; Lee, C.M. Water binding and ingredient dispersion pattern effect on surimi gel texture. J. Food Sci. 1991, 56, 1263–1266. [Google Scholar] [CrossRef]
- Claus, J.R.; Hunt, M.C. Low-fat, high value-added bologna formulated with texture-modifying ingredients. J. Food Sci. 1991, 56, 643–647. [Google Scholar] [CrossRef]
- Yang, A.; Trout, G.R.; Shay, B.J. Evaluation of carrageenan, isolated soy protein and a modified starch in low fat frankfurters. In Proceedings of the 41st Annual International Congress of Meat Science and Technology, San Antonio, TX, USA, 20–25 August 1995; Elsevier: Amsterdam, The Netherlands, 1995; 2, pp. 435–436. [Google Scholar]
- Pietrasik, Z. Effect of content of protein, fat and modified starch on binding textural characteristics, and colour of comminuted scalded sausages. Meat Sci. 1999, 51, 17–25. [Google Scholar] [CrossRef]
- Li, J.Y.; Yeh, A.I. Effects of starch properties on rheological characteristics of starch/meat complexes. J. Food Eng. 2003, 57, 287–294. [Google Scholar] [CrossRef]
- Wu, M.C.; Hamann, D.D.; Lanier, T.C. Rheological and clorimetric investigations of starch-fish protein systems during thermal processing. J. Texture Stud. 2007, 16, 53–74. [Google Scholar]
- Wu, M.C.; Akahane, T.A.; Lanier, T.C.; Hamann, D.D. Thermal transitions of actomyosin and surimi prepared from Atlantic croaker as studied by DSC. J. Food Sci. 1985, 50, 10–13. [Google Scholar]
- Shi, X.; BeMiller, J.N. Effects of food gums on viscosities of starch suspensions during pasting. Carbohydr. Polym. 2002, 50, 7–18. [Google Scholar] [CrossRef]
- Chaisawang, M.; Suphantharika, M. Pasting and rheological properties of native and anionic tapioca starches as modified by guar gum and xanthan gum. Food Hydrocolloid. 2006, 20, 641–649. [Google Scholar] [CrossRef]
- Freitas, R.A.; Gorin, P.A.J.; Neves, J.; Sierakowski, M.R. A rheological description of mixtures of a galactoxyloglucan with high amylose and waxy corn starches. Carbohydr. Polym. 2003, 51, 25–32. [Google Scholar]
- Kim, C.; Yoo, B. Rheological properties of rice starch-xanthan gum mixtures. J. Food Eng. 2006, 75, 120–128. [Google Scholar] [CrossRef]
- Alloncle, M.; Doublier, J.L. Viscoelastic properties of maize starch/hydrocolloid pastes and gels. Food Hydrocolloid. 1991, 5, 455–467. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Elgadir, M.A.; Akanda, M.J.H.; Ferdosh, S.; Mehrnoush, A.; Karim, A.A.; Noda, T.; Sarker, M.Z.I. Mixed Biopolymer Systems Based on Starch. Molecules 2012, 17, 584-597. https://doi.org/10.3390/molecules17010584
Elgadir MA, Akanda MJH, Ferdosh S, Mehrnoush A, Karim AA, Noda T, Sarker MZI. Mixed Biopolymer Systems Based on Starch. Molecules. 2012; 17(1):584-597. https://doi.org/10.3390/molecules17010584
Chicago/Turabian StyleElgadir, M. Abd, Md. Jahurul Haque Akanda, Sahena Ferdosh, Amid Mehrnoush, Alias A. Karim, Takahiro Noda, and Md. Zaidul Islam Sarker. 2012. "Mixed Biopolymer Systems Based on Starch" Molecules 17, no. 1: 584-597. https://doi.org/10.3390/molecules17010584
APA StyleElgadir, M. A., Akanda, M. J. H., Ferdosh, S., Mehrnoush, A., Karim, A. A., Noda, T., & Sarker, M. Z. I. (2012). Mixed Biopolymer Systems Based on Starch. Molecules, 17(1), 584-597. https://doi.org/10.3390/molecules17010584




