Metabolism Study of Notoginsenoside R1, Ginsenoside Rg1 and Ginsenoside Rb1 of Radix Panax Notoginseng in Zebrafish
Abstract
:1. Introduction
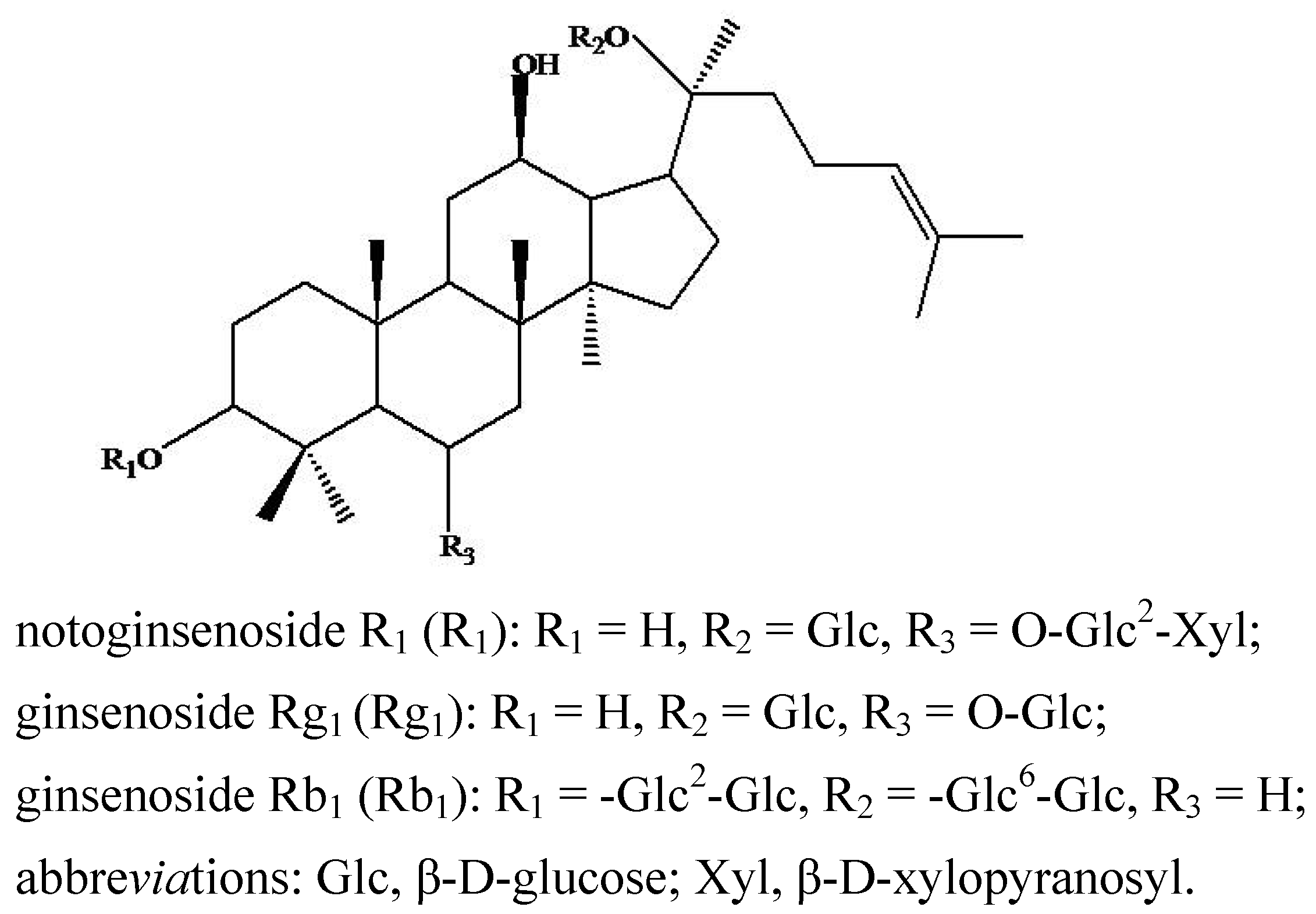
2. Results and Discussion
2.1. Conditions Selected for Zebrafish Metabolism Experiments
2.2. Analysis of Metabolic Components of Notoginsenoside R1, Ginsenoside Rg1 and Ginsenoside Rb1 after Zebrafish Exposure by HPLC-ESI-MS
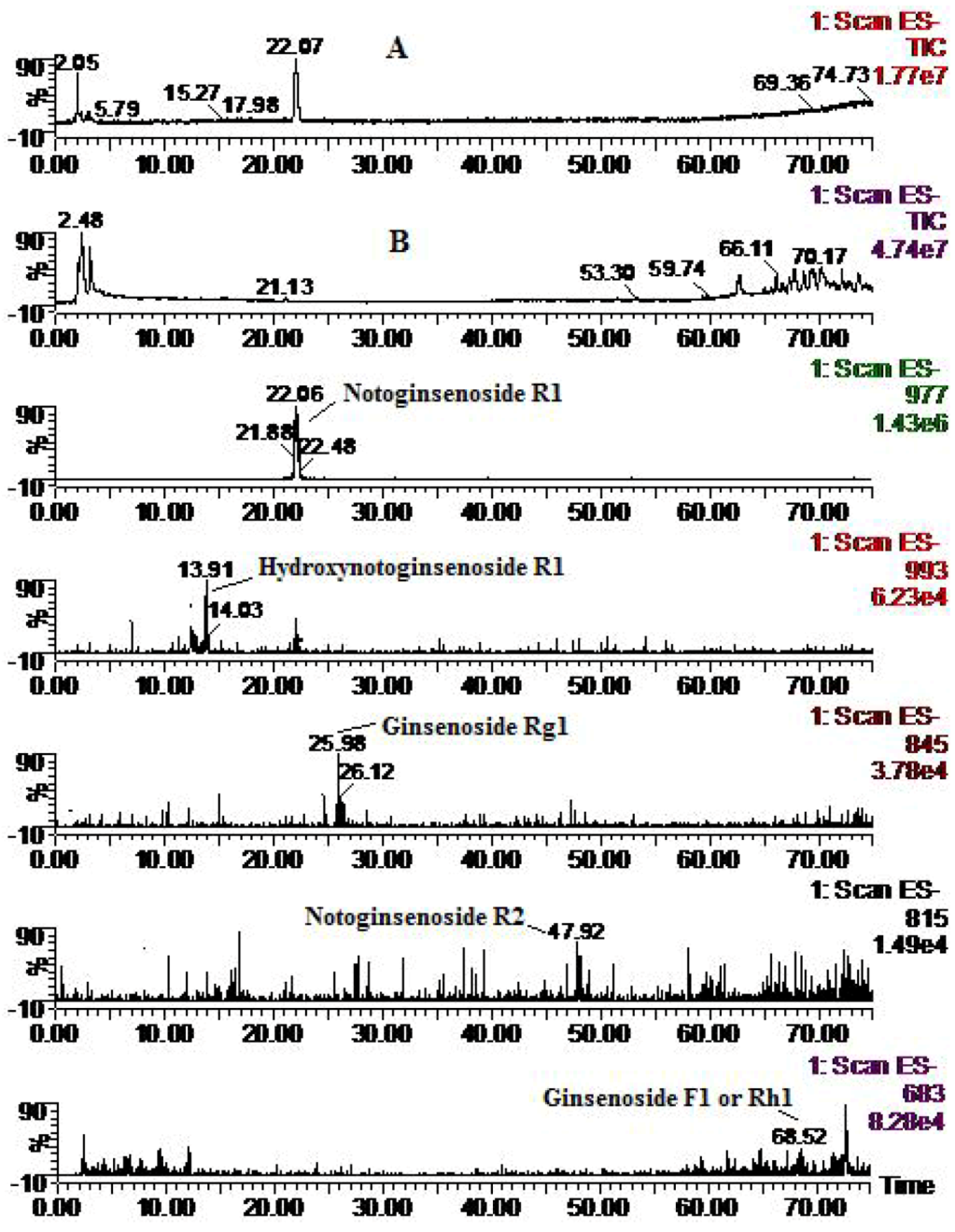
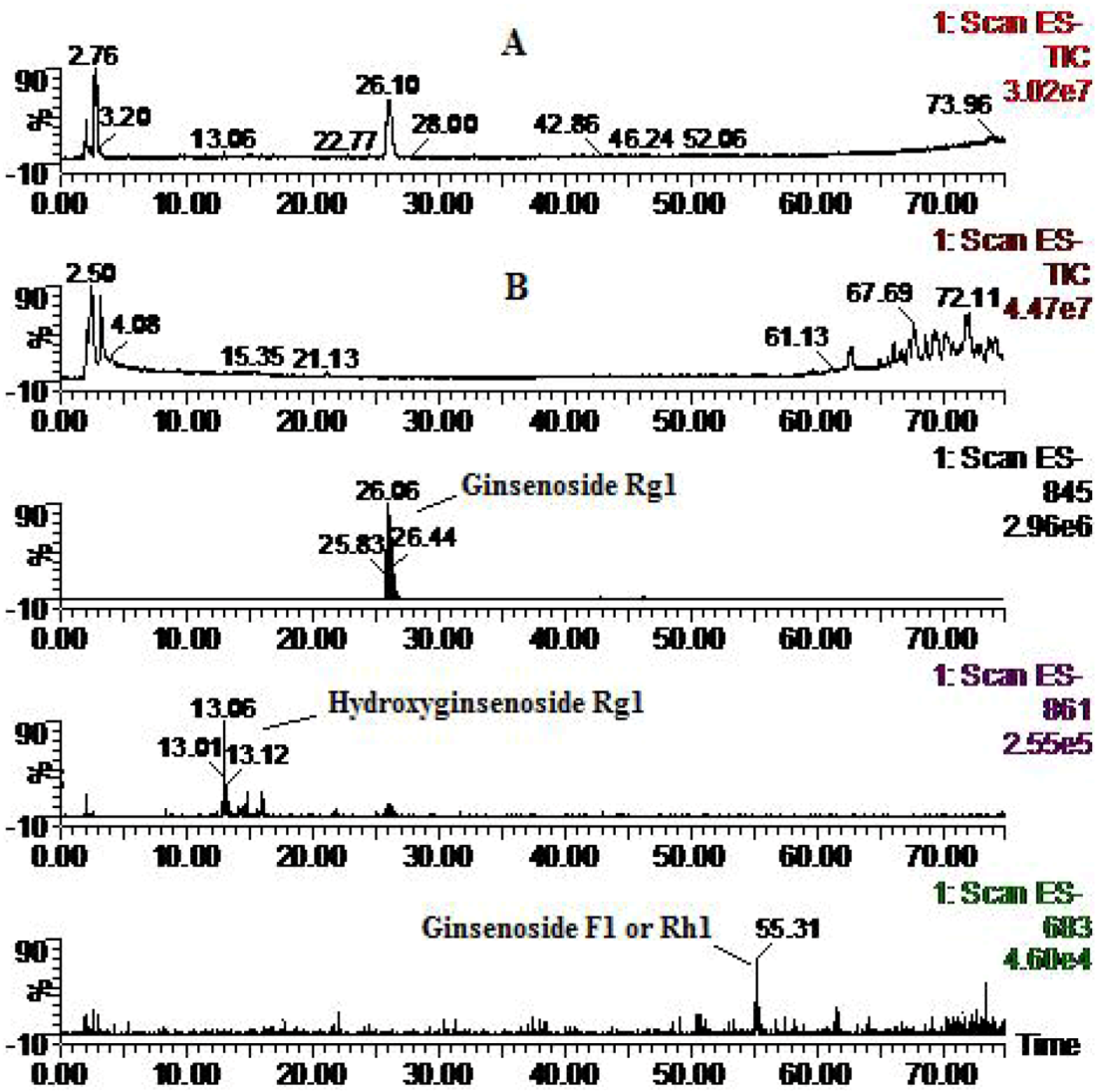
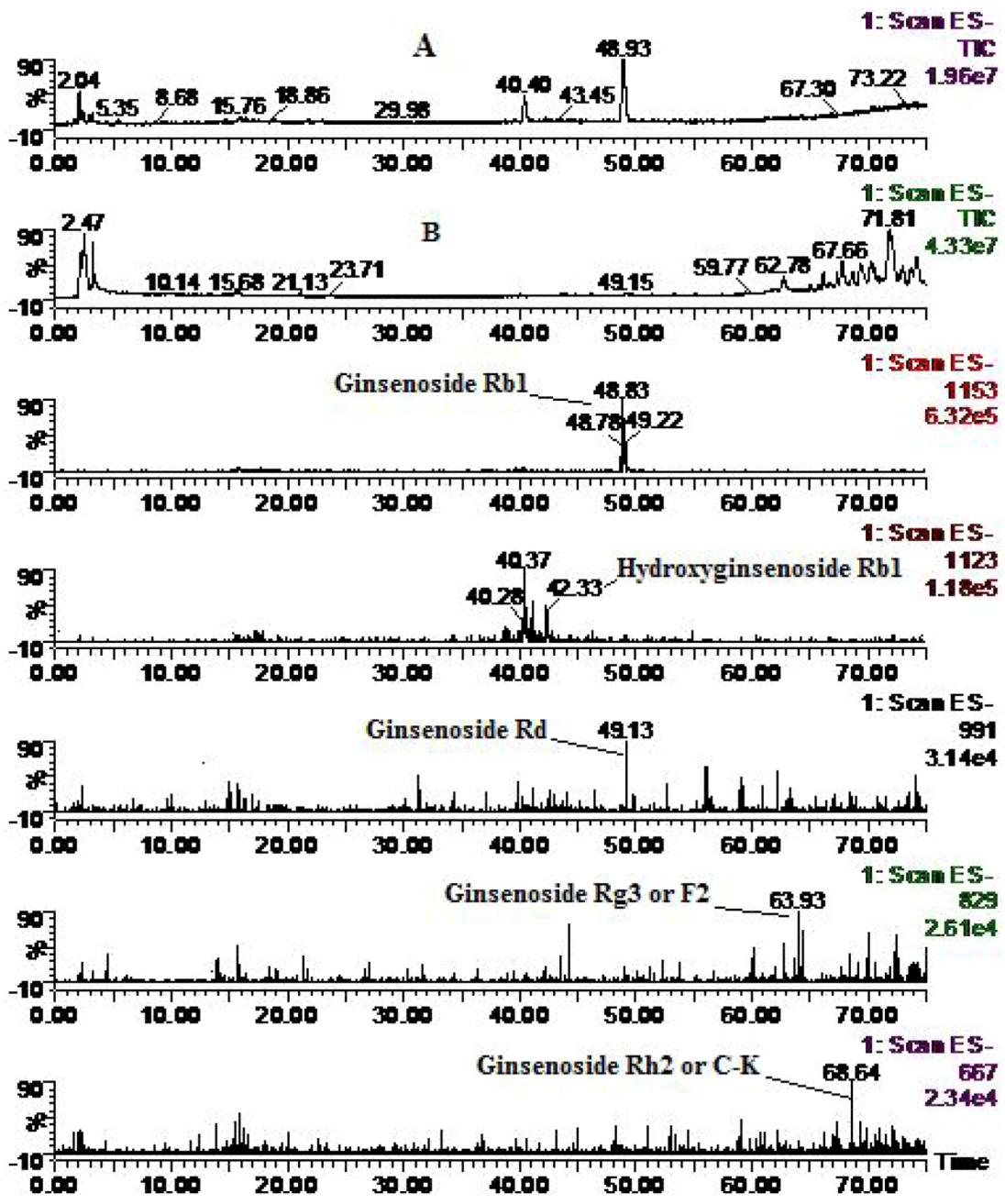
| Compounds | Retention time (min) | quasi-molecular ions peak | MW | Metabolite presumed | Zebrafish | Mammalian metabolism (references) | ||
| [M−H]− | [M+HCOO]− | solution | body | |||||
| R1 | 21.91 | 931.84 | 977.73 | 932.8 | Notoginsenoside R1 | + | + | [14,15,16] |
| 13.91 | 947.71 | 993.46 | 948.7 | Hydroxynotoginsenoside R1 | + | [14] | ||
| 25.98 | 799.81 | 845.83 | 800.8 | Ginsenoside Rg1 | + | [14, 16] | ||
| 48.07 | 769.24 | 815.54 | 770.2 | Notoginsenoside R2 | + | [15] | ||
| 68.52 | 637.21 | 683.1 | 638.2 | Ginsenoside F1 or Rh1 | + | [14,15,16] | ||
| Rg1 | 26.1 | 799.81 | 845.83 | 800.8 | Ginsenoside Rg1 | + | + | [15,16,17,18,19] |
| 13.06 | 861.77 | 816.8 | Hydroxyginsenoside Rg1 | + | [29] | |||
| 55.31 | 637.69 | 683.23 | 638.7 | Ginsenoside F1 or Rh1 | + | [17,18,19,20,21] | ||
| Rb1 | 48.93 | 1107.78 | 1153.46 | 1108.9 | Ginsenoside Rb1 | + | + | [25, 28] |
| 42.16 | 1123.03 | 1169.47 | 1124 | Hydroxyginsenoside Rb1 | + | [28] | ||
| 49.13 | 944.86 | 991.26 | 946 | Ginsenoside Rd | + | + | [25, 28] | |
| 63.98 | 783.87 | 829.98 | 784.8 | Ginsenoside Rg3 or F2 | + | [25, 28] | ||
| 68.66 | 620.79 | 666.75 | 622 | Ginsenoside Rh2 or C-K | + | + | [25, 28] | |
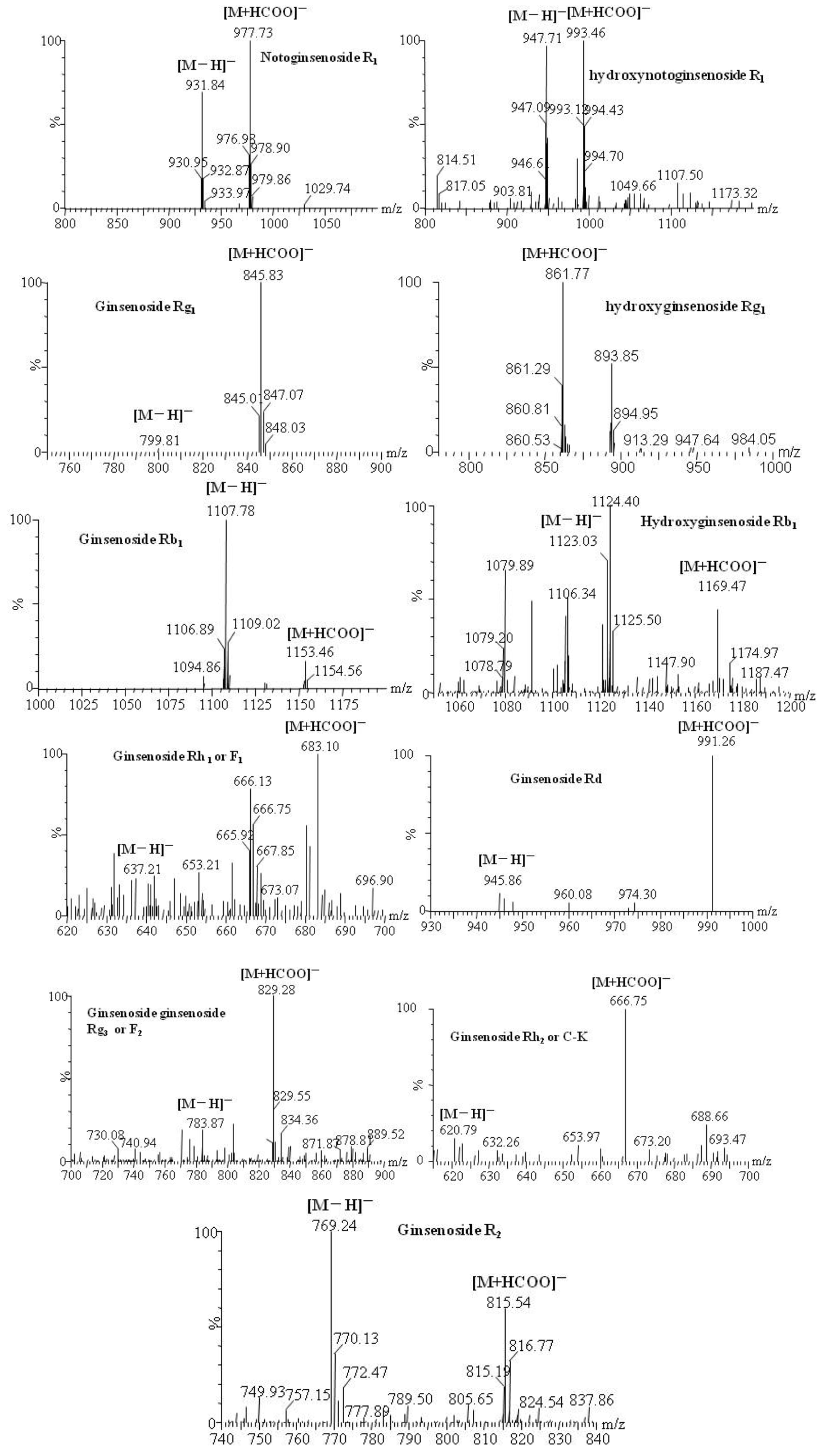
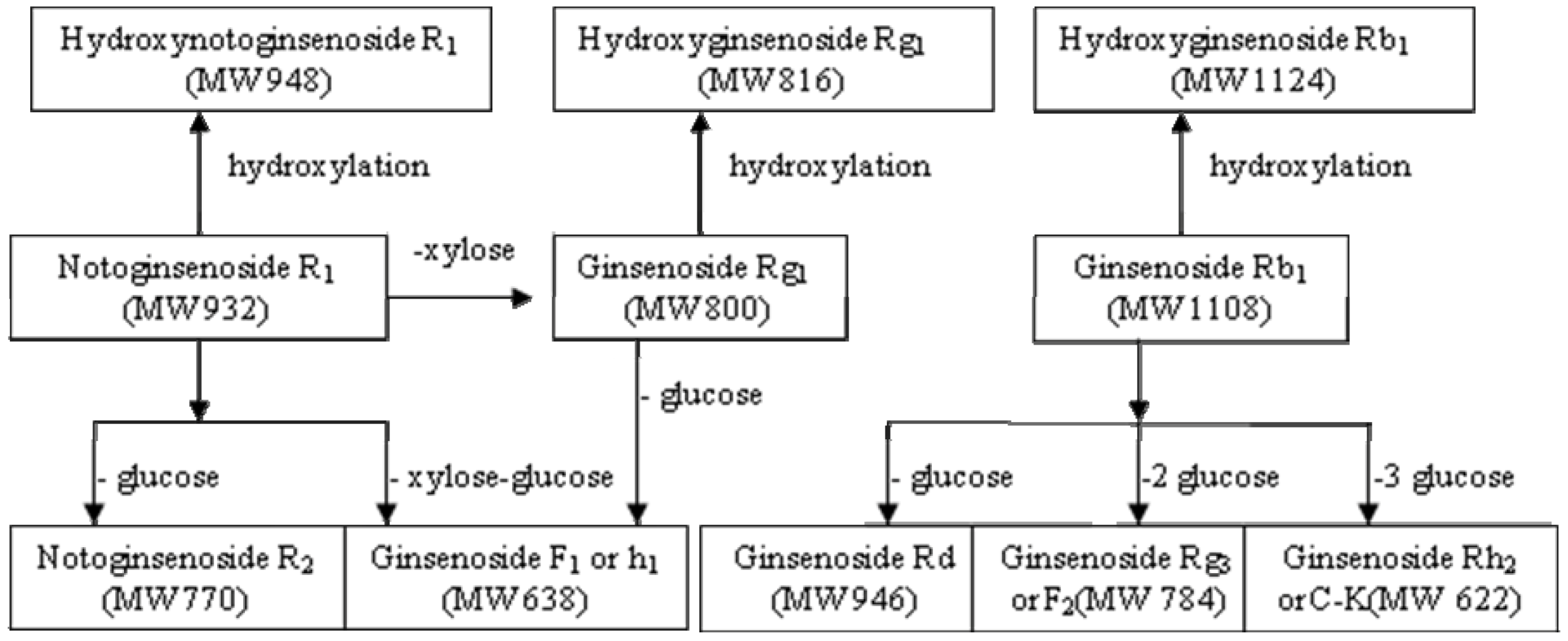
2.3. Rationality and Advantages of Metabolic Study with Zebrafish Compared to Existing Model
3. Experimental
3.1. Chemicals and Reagents
3.2. Animals
3.3. Instruments
3.4. Biological Sample Collection
3.5. Sample Preparation
3.6. Analysis Condition
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Goldsmith, P. Zebrafish as a pharmacological tool: The how, why and when. Curr. Opin. Pharmacol. 2004, 4, 504–512. [Google Scholar] [CrossRef]
- Kari, G.; Rodeck, U.; Dicker, A.P. Zebrafish: An emerging model system for human disease and drug discovery. Clin. Pharmacol. Ther. 2007, 82, 70–80. [Google Scholar] [CrossRef]
- Den Hertog, J. Chemical genetics: Drug screens in zebrafish. Biosci. Rep. 2005, 25, 289–297. [Google Scholar] [CrossRef]
- Zon, L.I.; Peterson, R.T. In vivo drug discovery in the zebrafish. Nat. Rev. Drug Discov. 2005, 4, 35–44. [Google Scholar] [CrossRef]
- Rubinstein, A.L. Zebrafish assays for drug toxicity screening. Expert Opin. Drug Metab. Toxicol. 2006, 2, 231–240. [Google Scholar] [CrossRef]
- Sukardi, H.; Chng, H.T.; Chan, E.C.Y.; Gong, Z.Y.; Lam, S.H. Zebrafish for drug toxicity screening: Bridging the in vitro cell-based models and in vivo mammalian models. Expert Opin. Drug Metab. Toxicol. 2011, 7, 579–589. [Google Scholar] [CrossRef]
- Liang, A. Zebrafish—Useful model for pharmacodynamics and toxicity screening of traditional Chinese medicine. Chin. J. Chin. Mater. Med. 2009, 34, 2839–2842. [Google Scholar]
- Lv, A.J.; Yang, Z.X.; Liu, H.; Hu, X.C.; Zhang, Y.H.; Cheng, C. Isolation of intestinal bacteria from zebrafish (Daniorerio) and their physiological and biochemical characteristics. Chin. Agric. Sci. Bull. 2010, 26, 412–415. [Google Scholar]
- Collodi, P.; Miranda, C.; Zhao, X.; Buhler, D.; Barnes, D. Induction of zebrafish (Brachydanio rerio) P450 in vivo and in cell culture. Xenobiotica 1994, 24, 487–493. [Google Scholar] [CrossRef]
- Wang, L.; Yao, J.H.; Xue, J.L. Advances in the vertebrate P450s superfamily in transgenic animals. Int. J. Genet. 2006, 29, 424–428. [Google Scholar]
- Wei, Y.J.; Ning, Q.; Jia, X.B.; Gong, Z.A. Thoughts and methods for metabolic study of Chinese materia medica based on zebraf ish model. Chin. Trad. Herb. Drugs 2009, 40, 1009–1011. [Google Scholar]
- Wei, Y.J. Analysis of chemical and metabolic components of Fufang Danshen prescription. Nanjing: China Pharm. Univ. 2007, 21, 797–809. [Google Scholar]
- National Commission of Chinese Pharmacopoeia. In Pharmacopoeia of the Peoples Republic of China; Chemical Industry Press: Beijing, China, 2010; pp. 11–12.
- Mao, X.J. Study on the fingerprints, assay, pharmacokinetics and metabolites of the Panax notoginsenosides. Nanjing: China Pharm. Univ. 2005. [Google Scholar]
- Chen, G.T.; Yang, M. Metabolites of notoginsenoside R1 in rats. Lishizhen Med. Mater. Med. Res. 2010, 21, 485–487. [Google Scholar]
- Ruan, J.Q.; Leong, W.I.; Yan, R.; Wang, Y.T. Characterization of metabolism and in vitro permeability study of notoginsenoside R1 from Radix Notoginseng. J. Agric. Food Chem. 2010, 58, 5770–5776. [Google Scholar] [CrossRef]
- Han, B.H.; Park, M.H.; Han, Y.N.; Woo, L.K.; Sankawa, U.; Yahara, S.; Tanaka, O. Degradation of ginseng saponins under mild acidic conditions. Planta Med. 1982, 44, 146–149. [Google Scholar] [CrossRef]
- Liu, Q.; Feng, Z.Y.; Zheng, L.J.; Zhang, X. HPLC detects degradation products of ginsenosides in the mild and acidic conditions. J. Dalian Med. Univ. 2000, 22, 55–56, 69. [Google Scholar]
- Wang, Y.; Liu, T.H.; Wang, W.; Wang, B.X. Studies on the metabolism of ginsenoside Rg1 by intestinal bacteria. China J. Chin. Mater. Med. 2001, 26, 188–191. [Google Scholar]
- Wang, Y.; Liu, T.H.; Wang, W.; Wang, B.X. Studies on the metabolism of ginsenoside Rg1 by intestinal bacteria and its absorbed metabolites in rat and human sera. Acta Pharm. Sin. 2000, 35, 284–288. [Google Scholar]
- Chen, G.T.; Gao, H.Y.; Song, Y.; Yang, M.; Guo, D.A.; Wu, L.J. Metabolic study of ginsenoside Rg1 in rat. Mod. Chin. Med. 2008, 10, 37–40. [Google Scholar]
- Feng, L.; Hu, C.J.; Yu, L.Y. Pharmacokinetic study of ginsenoside Rg1 and its metabolite. Acta Pharm. Sin. 2010, 45, 636–640. [Google Scholar]
- Akao, T.; Kida, H.; Kanaoka, M.; Hattori, M.; Kobashi, K. Intestinal bacterial hydrolysis is required for the appearance of compound K in rat plasma after oral administration of ginsenoside Rb1 from Panax ginseng. J. Pharm. Pharmacol. 1998, 50, 1155. [Google Scholar]
- Odani, T.; Tanizawa, H.; Takino, Y. Studies on the absorption, distribution, excretion and metabolism of ginseng saponins. III. The absorption, distribution and excretion of ginsenoside Rb1 in the rat. Chem. Pharm. Bull. 1983, 31, 1059–1066. [Google Scholar] [CrossRef]
- Chen, X.; Zhou, Q.L.; Wang, B.X. The Metabolism of ginsenoside Rb1 by intestinal bacteria. Acta Pharm. Sin. 1999, 34, 410–414. [Google Scholar]
- Chen, X.; Zhou, Q.L.; Wang, B.X. Studies on the metabolism of ginsenoside Rb1 by intestinal bacteria absorbed metabolites in rat and human sera. Acta Pharm. Sin. 1999, 34, 481–483. [Google Scholar]
- Ma, J.S.; Zhou, Q.Y.; Fei, X.F.; Sun, Y.; Wang, B.X. Metabolism of ginsenoside Rb1 and panaxadiolsaponins by fungi. Acta Pharm. Sin. 2001, 36, 603–605. [Google Scholar]
- Yang, L.; Xu, S.J.; Zeng, X.; Wu, Z.F.; Deng, Y.H.; Liu, Y.M.; Deng, S.G.; Ou, R.M. In vivo rat metabolism studies of ginsenoside Rb1. Chem. J. Chin. Univ. 2006, 27, 1042–1044. [Google Scholar]
- Wei, Y.J.; Li, P.; Shu, B.; Li, H.J.; Peng, Y.R.; Song, Y.; Chen, J.; Yi, L. Analysis of chemical and metabolic components in traditional Chinese medicinal combined prescription containing Radix Salvia miltiorrhiza and Radix Panaxnotoginseng by LC-ESI-MS methods. Biomed. Chromatogr. 2007, 21, 797–809. [Google Scholar] [CrossRef]
- Tawab, M.A.; Bahr, U.; Karas, M.; Wurglics, M.; Schubert-Zsilavecz, M. Degradation of ginsenosides in humans after oral administration. Drug Metab. Dispos. 2003, 31, 1065–1071. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds notoginsenoside R1, ginsenoside Rg1 and ginsenoside Rb1 are available from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wei, Y.; Li, P.; Fan, H.; Peng, Y.; Liu, W.; Wang, C.; Shu, L.; Jia, X. Metabolism Study of Notoginsenoside R1, Ginsenoside Rg1 and Ginsenoside Rb1 of Radix Panax Notoginseng in Zebrafish. Molecules 2011, 16, 6621-6633. https://doi.org/10.3390/molecules16086621
Wei Y, Li P, Fan H, Peng Y, Liu W, Wang C, Shu L, Jia X. Metabolism Study of Notoginsenoside R1, Ginsenoside Rg1 and Ginsenoside Rb1 of Radix Panax Notoginseng in Zebrafish. Molecules. 2011; 16(8):6621-6633. https://doi.org/10.3390/molecules16086621
Chicago/Turabian StyleWei, Yingjie, Ping Li, Hongwei Fan, Yunru Peng, Wei Liu, Changmei Wang, Luan Shu, and Xiaobin Jia. 2011. "Metabolism Study of Notoginsenoside R1, Ginsenoside Rg1 and Ginsenoside Rb1 of Radix Panax Notoginseng in Zebrafish" Molecules 16, no. 8: 6621-6633. https://doi.org/10.3390/molecules16086621
APA StyleWei, Y., Li, P., Fan, H., Peng, Y., Liu, W., Wang, C., Shu, L., & Jia, X. (2011). Metabolism Study of Notoginsenoside R1, Ginsenoside Rg1 and Ginsenoside Rb1 of Radix Panax Notoginseng in Zebrafish. Molecules, 16(8), 6621-6633. https://doi.org/10.3390/molecules16086621




