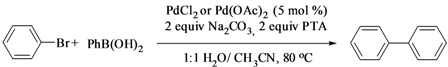The Suzuki Reaction in Aqueous Media Promoted by P, N Ligands
Abstract
:1. Introduction
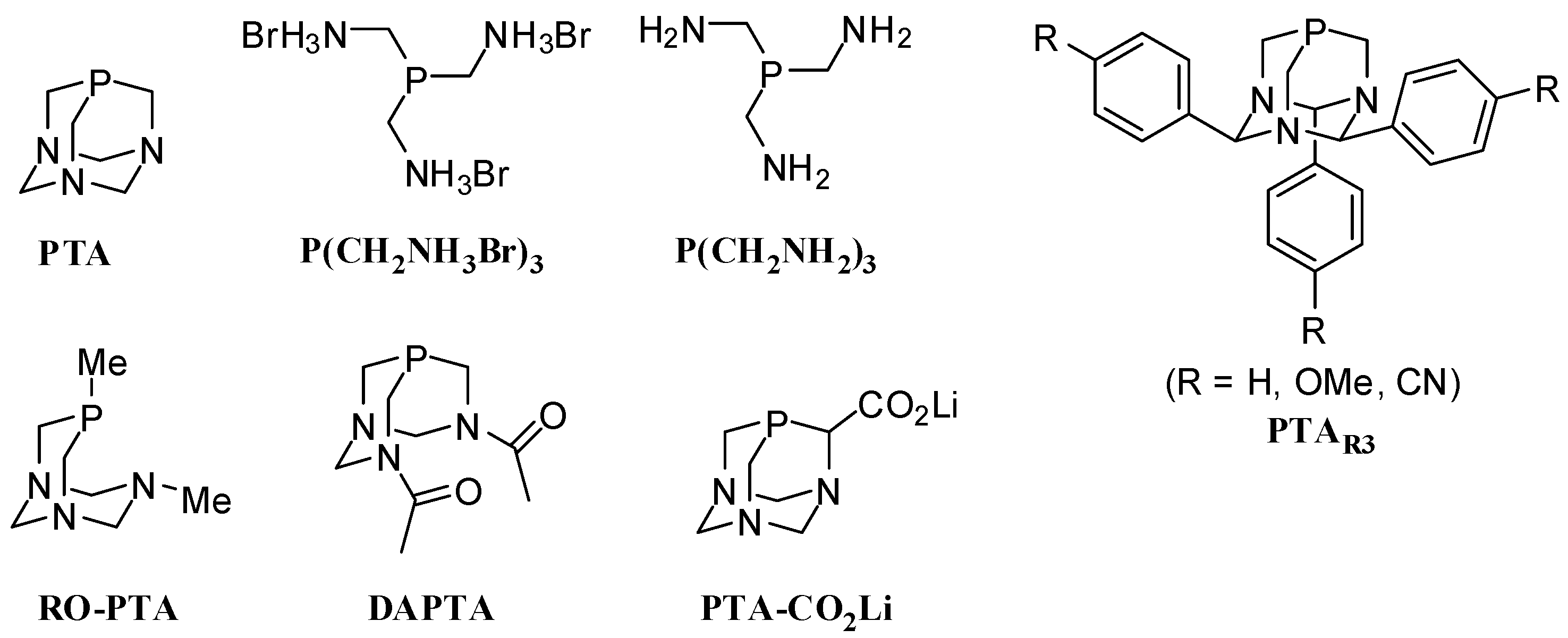
2. Results and Discussion
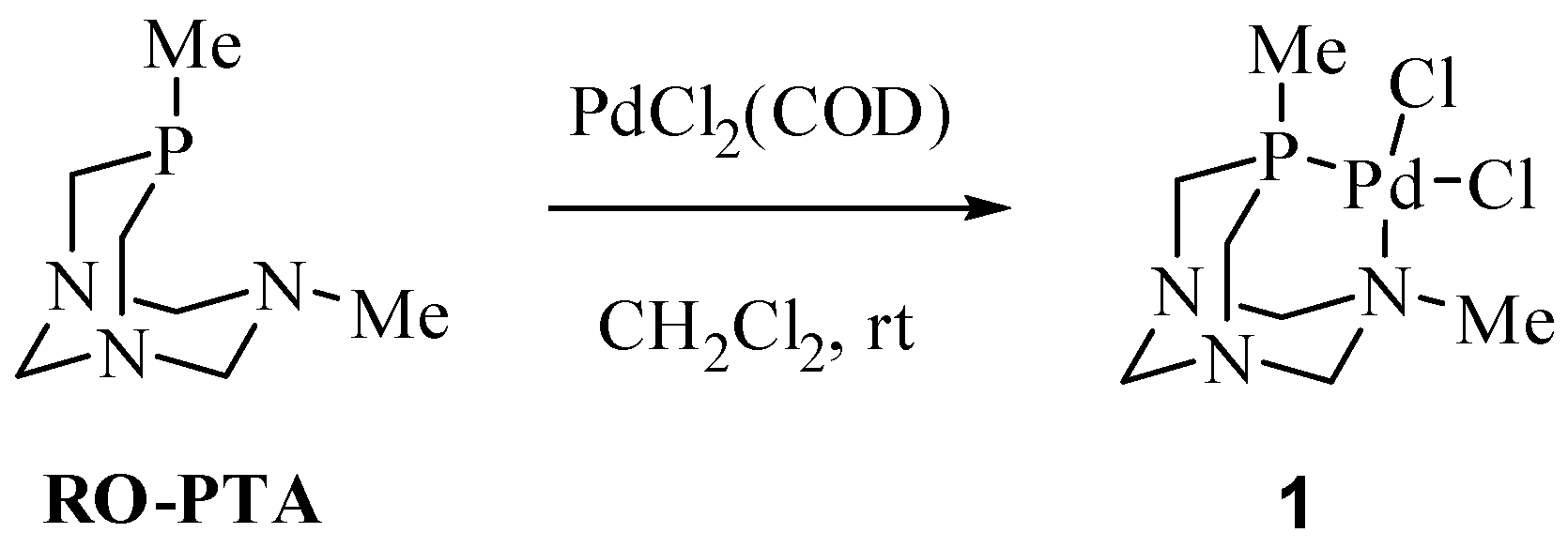
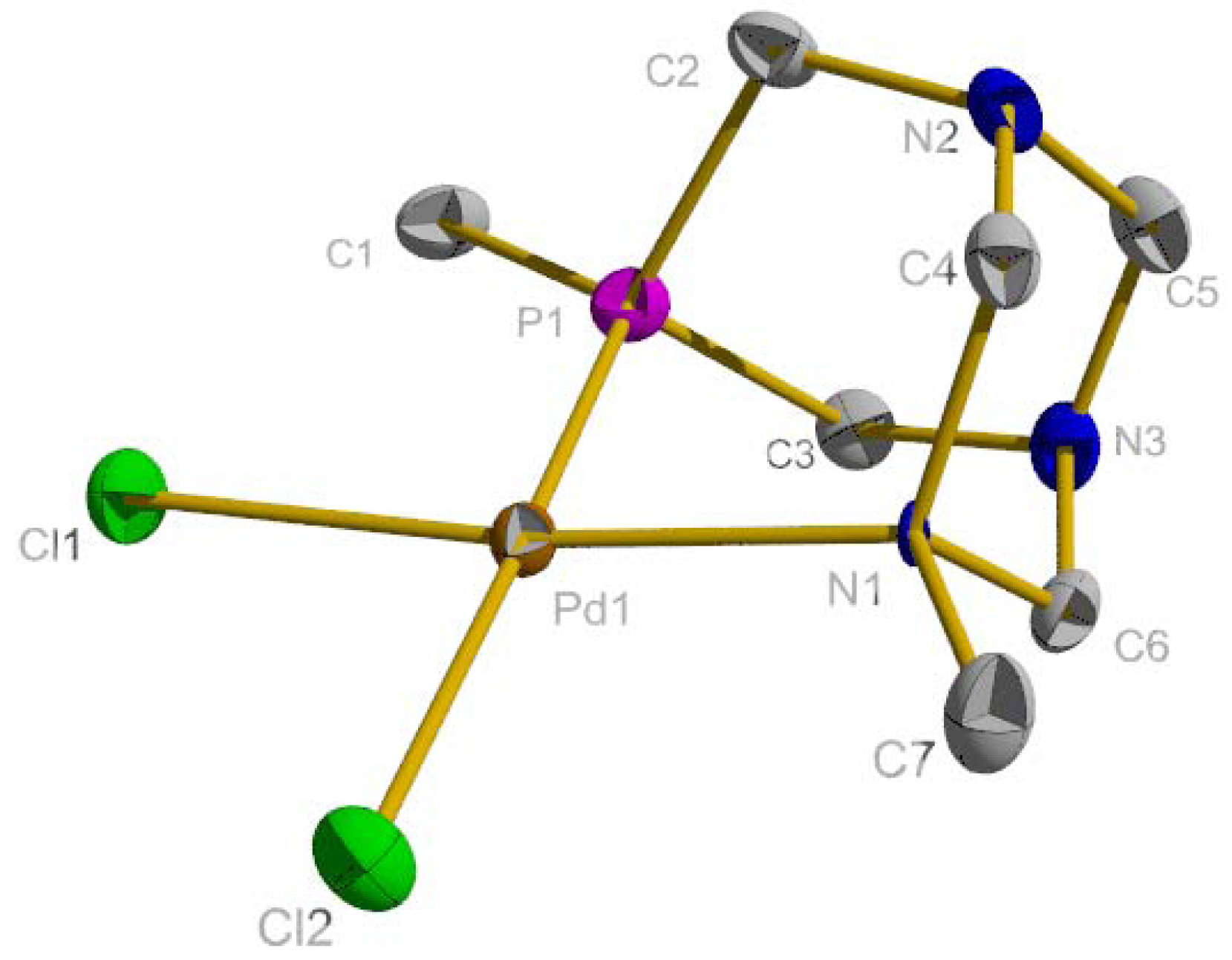
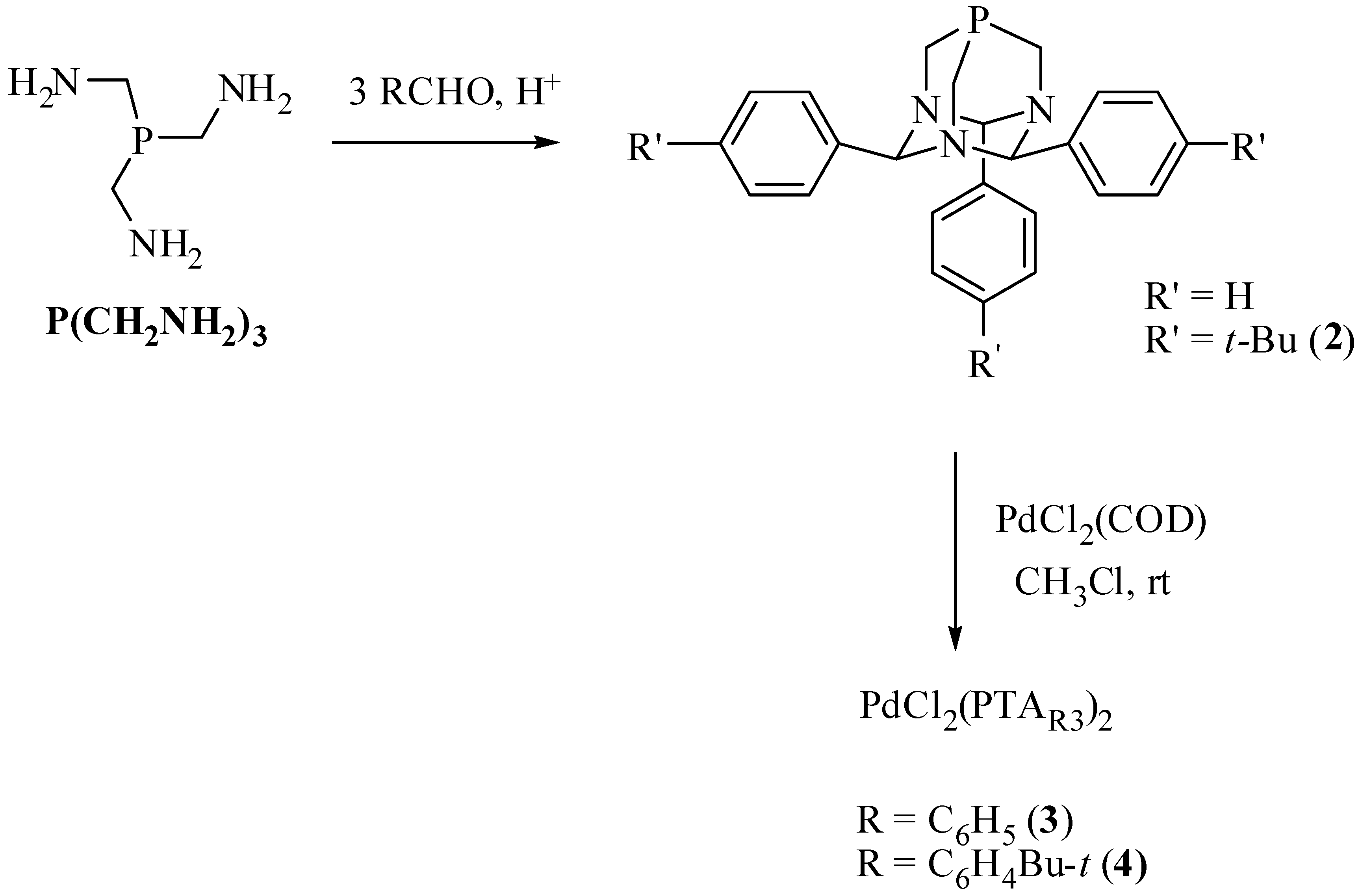
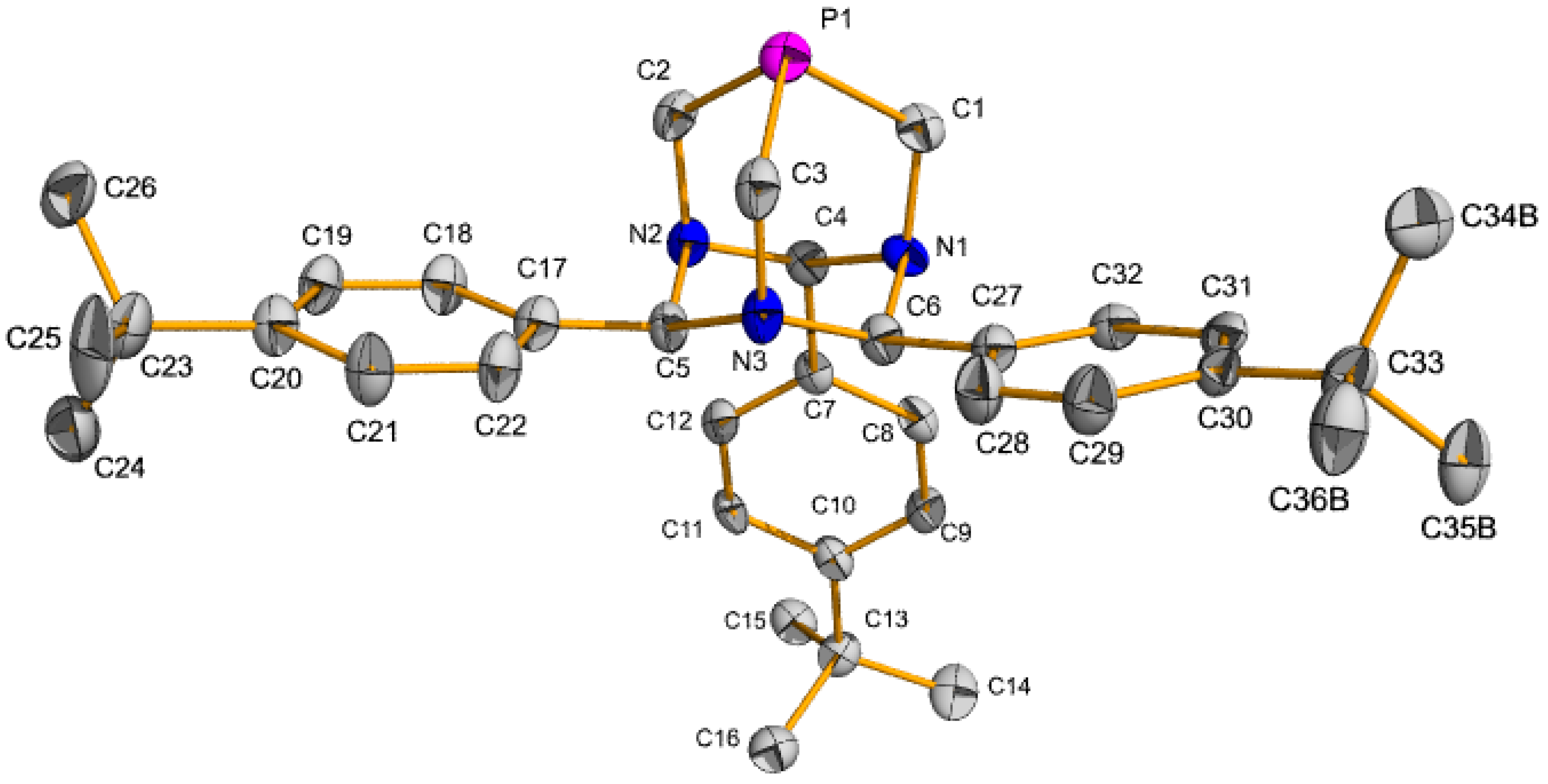
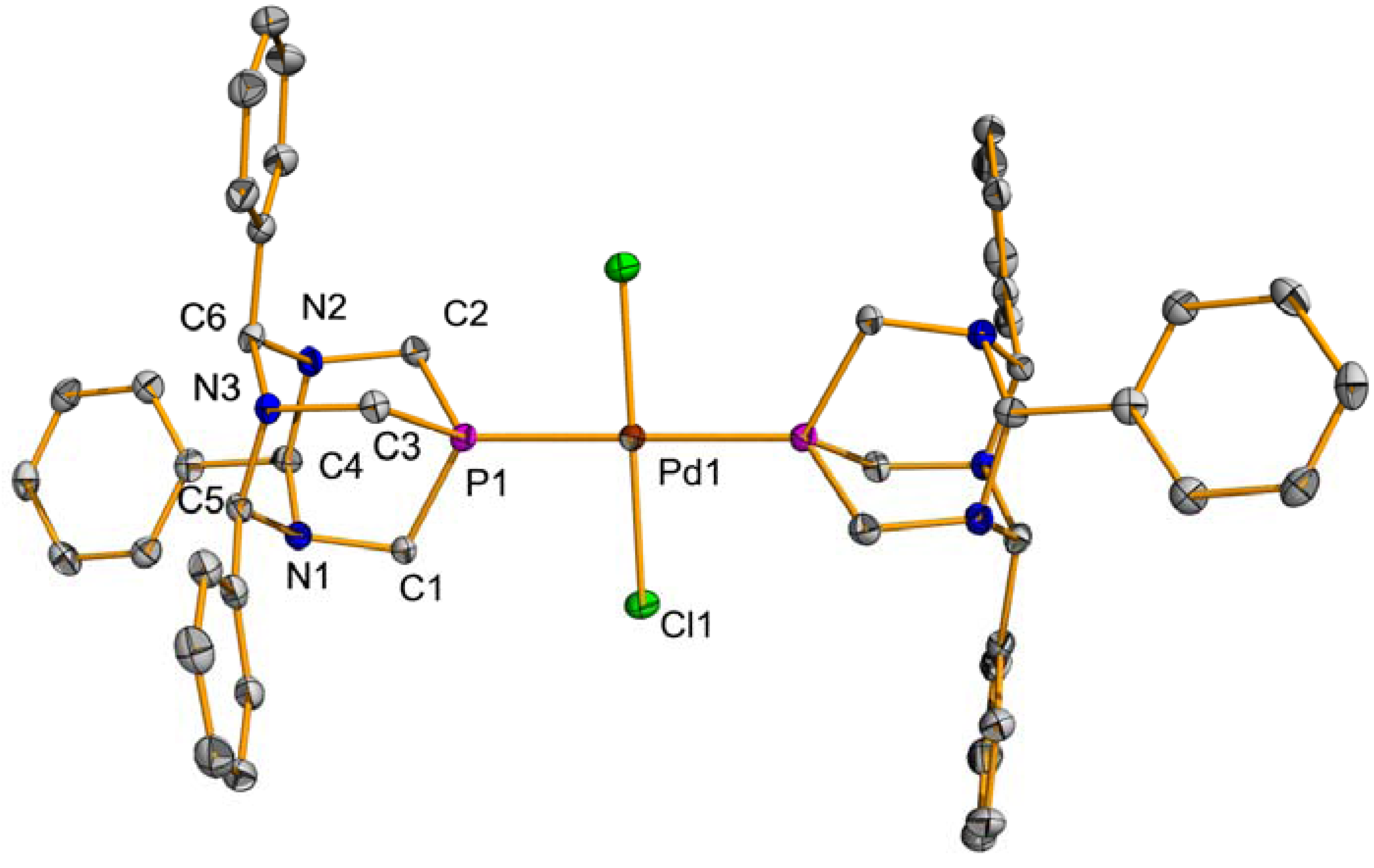
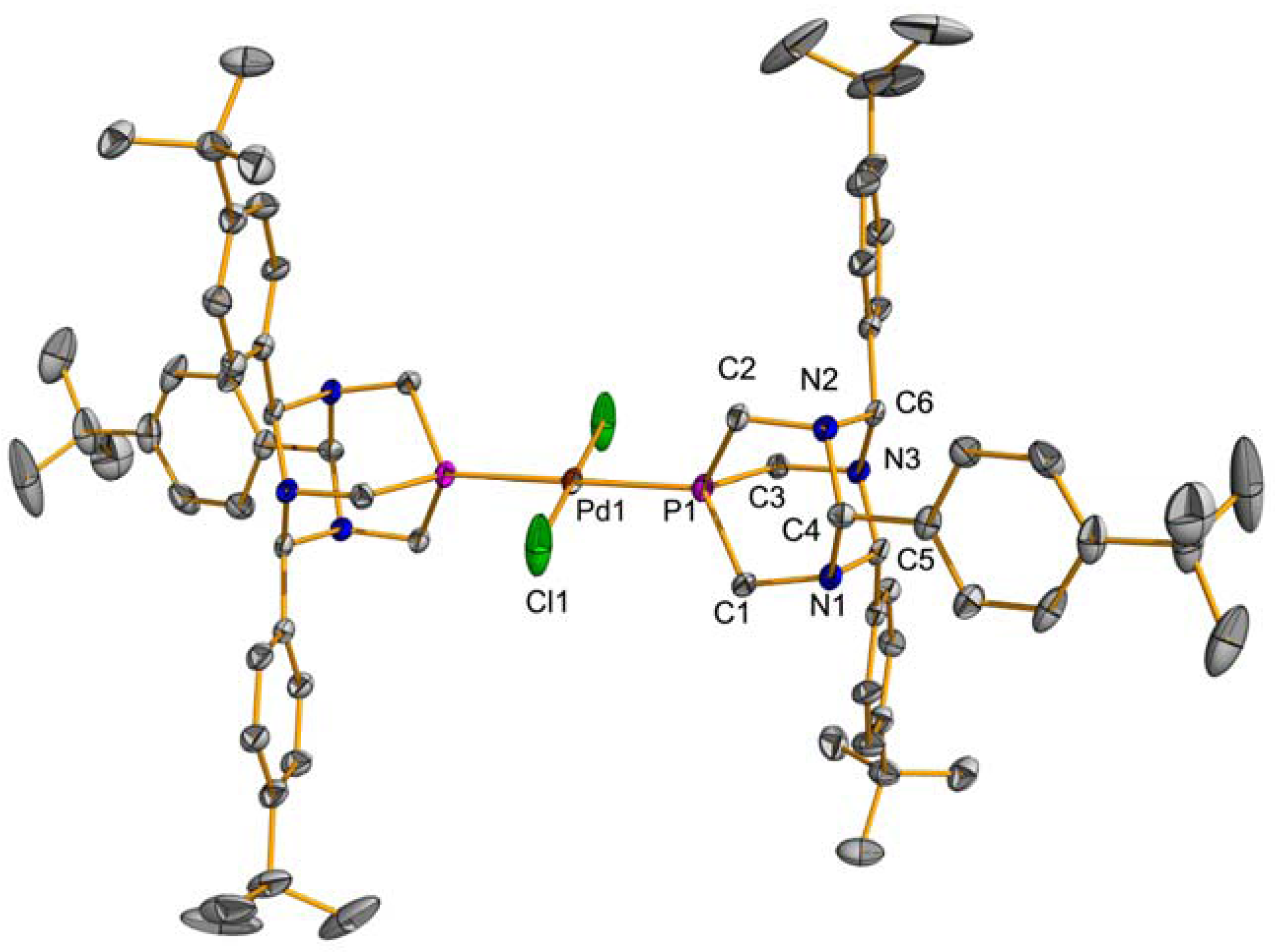
2.1. Suzuki-Miyaura Coupling
| Entry | Catalyst | Time (h) | % Yield a |
|---|---|---|---|
| 1 | Pd(OAc)2PTA2 | 24 | 36.3 |
| 2 | Pd(OAc)2PTA2 | 48 | 63.3 |
| 3 | Pd(OAc)2PTA2 | 72 | 66 |
| 4 b | Pd(OAc)2PTA2 | 24 | 3.0 |
| 5 b | Pd(OAc)2PTA2 | 48 | 4.9 |
| 6 | PdCl2PTA2 | 48 | 51.5 |
| 7 b | PdCl2PTA2 | 48 | 5.0 |
| Entry | Precatalyst | Ligand | Pd:L | Yield (%) a |
|---|---|---|---|---|
| 1 | Pd(OAc)2 | DAPTA | 1:2 | 40 |
| 2 | Pd(OAc)2 | DAPTA | 1:3 | 42 |
| 3 | Pd(OAc)2 | PTAPh3 | 1:2 | 65 |
| 4 | Pd(OAc)2 | 2 | 1:2 | 56 |
| 5 | 3 | - | 1:2 | 58 |
| 6 | 4 | - | 1:2 | 60 |
| 7 | Pd(OAc)2 | PTA-CO2Li | 1:1 | 60 |
| 8 | Pd(OAc)2 | PTA-CO2Li | 1:2 | 76 |
| 9 | Pd(OAc)2 | PTA-CO2Li | 1:3 | 75 |
| 10 | Pd(OAc)2 | P(CH2NH3Br)3 | 1:1 | 74 |
| 11 | Pd(OAc)2 | P(CH2NH3Br)3 | 1:2 | 77 |
| 12 | Pd(OAc)2 | P(CH2NH3Br)3 | 1:3 | 78 |
| 13 | Pd(OAc)2 | P(CH2NH2)3 | 1:1 | 77 |
| 14 | Pd(OAc)2 | P(CH2NH2)3 | 1:2 | 80 |
| 15 | Pd(OAc)2 | RO-PTA | 1:2 | 86 |
| 16 | Pd(OAc)2 | RO-PTA | 1:3 | 82 |
| 17 b | Pd(OAc)2 | RO-PTA | 1:2 | 52 |
| 18 | PdCl2 | RO-PTA | 1:2 | 89 |
| 19 | PdCl2 | RO-PTA | 1:3 | 85 |
| 20 | 1 | - | 1:1 | 91 |
| 21 | Pd(OAc)2 | - | - | 88 |
| 22 | PdCl2 | - | - | 80 |
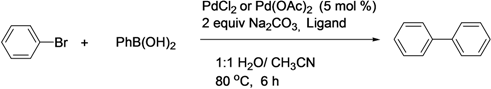
| Entry b | Bromide | Precatalyst | Yield (%) a |
|---|---|---|---|
| 1 |  | Pd(OAc)2 | 84 |
| 2 | 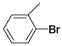 | Pd(OAc)2 | 65 |
| 3 |  | Pd(OAc)2 | <5 |
| 4 | 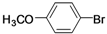 | Pd(OAc)2 | 50 |
| 5 | 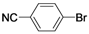 | Pd(OAc)2 | 90 |
| 6 | 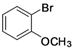 | Pd(OAc)2 | 55 |
| 7 | 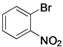 | Pd(OAc)2 | 77 |
3. Experimental
3.1. General
3.2. General Procedure for the Suzuki Coupling Reaction Aryl Halides and Arylboronic Acids
3.3. Mercury Experiment
3.4. X-ray Crystallography
| 1 | 2 | 3 | 4 | |
|---|---|---|---|---|
| Empirical Formula | C7H16Cl2N3PPd | C36H48N3P | C52H58Cl2N6OP2Pd | C74H98Cl8N6P2Pd |
| Fw | 350.50 | 553.74 | 1022.28 | 1523.52 |
| T(K) | 100(2) | 100(2) | 100(2) | 100(2) |
| λ(Ǻ) | 0.71073 | 0.71073 | 0.71073 | 0.71073 |
| cryst syst | Monoclinic | Triclinic | Triclinic | Triclinic |
| space group | P21/c | P-1 | P-1 | P-1 |
| a(Ǻ) | 8.60830(10 | 8.2876(8) | 11.10810(10) | 12.0121(3) |
| b(Ǻ) | 11.5593(2) | 11.8367(12) | 14.6003(2) | 12.7019(3) |
| c(Ǻ) | 12.0933(2) | 17.4678(19) | 16.9813(2) | 13.9564(3) |
| α(deg) | 90 | 97.424(7) | 113.3050(10) | 116.1830(10) |
| β(deg) | 107.1980(10) | 100.306(7) | 102.5090(10) | 94.1170(10) |
| γ(deg) | 90 | 104.442(8) | 100.2660(10) | 99.6960(10) |
| V(Ǻ3) | 1149.55(3) | 1605.2(3) | 2359.92(5) | 1857.75(8) |
| Z | 4 | 2 | 2 | 1 |
| Dcalc (Mg/m3) | 2.025 | 1.146 | 1.439 | 1.362 |
| abs coeff (mm−1) | 2.184 | 0.114 | 0.620 | 0.626 |
| cryst size (mm3) | 0.07 × 0.06 × 0.03 | 0.56 × 0.56 × 0.04 | 0.12 × 0.08 × 0.07 | 0.31 × 0.09 × 0.05 |
| θ data collect (deg) | 2.48 to 26.37 | 1.81 to 22.50 | 1.38 to 27.43 | 1.65 to 27.55 |
| Index ranges | −10 ≤ h ≤ 9 | −8 ≤ h ≤ 8 | −14 ≤ h ≤ 14 | −15 ≤ h ≤ 15 |
| −14 ≤ k ≤13 | −12 ≤ k ≤ 12 | −18 ≤ k ≤ 18 | −16 ≤ k ≤ 16 | |
| −14 ≤ l ≤ 15 | −18 ≤ l ≤ 18 | −21 ≤ l ≤ 21 | −18 ≤ l ≤ 18 | |
| reflns collected | 21262 | 10984 | 59870 | 30379 |
| indep reflns | 2351 Rint = 0.0452 | 4121 Rint = 0.0822 | 10747 Rint = 0.0622 | 8530 Rint = 0.0674 |
| abs correction | SADABS | SADABS | SADABS | SADABS |
| data/restraints/param | 2351/0/127 | 4121/0/418 | 10747/0/582 | 8530/0/421 |
| GOF F2 | 1.088 | 0.930 | 1.033 | 1.051 |
| final R indices | R1 = 0.0343 | R1 = 0.0643, | R1 = 0.0424 | R1 = 0.0810 |
| [ I □ 2σ(I)] | wR2 = 0.0761 | wR2 = 0.1190 | wR2 = 0.0898 | wR2 = 0.2168 |
| R indicies | R1 = 0.0462 | R1 = 0.1289 | R1 = 0.0755 | R1 = 0.1200 |
| (all data) | wR2 = 0.0791 | wR2 = 0.1363 | wR2 = 0.1046 | wR2 = 0.2426 |
| CCDC no. | 827747 | 827746 | 827745 | 827744 |
4. Conclusions
Acknowledgments
References and Notes
- Jana, R.; Pathak, T.P.; Sigman, M.S. Advances in transition metal (Pd, Ni, Fe)-catalyzed cross-coupling reactions using alkyl-organometallics as reaction partners. Chem. Rev. 2011, 111, 1417–1492. [Google Scholar]
- Liu, S.; Xiao, J. Toward green catalytic synthesis-Transition metal-catalyzed reactions in non-conventional media. J. Mol. Catal. A: Chem. 2007, 270, 1–43. [Google Scholar] [CrossRef]
- Beletskaya, I.P.; Cheprakov, A.V. The Heck reaction as a sharpening stone of palladium catalysis. Chem. Rev. 2000, 100, 3009–3066. [Google Scholar]
- Alonso, F.; Beletskaya, I.P.; Yus, M. Non-conventional methodologies for transition-metal catalysed carbon-carbon coupling: a critical review. Part 2: The Suzuki reaction. Tetrahedron 2008, 64, 3047–3101. [Google Scholar]
- De Souza, M.V.N. Stille reaction: An important tool in the synthesis of complex natural products. Curr. Org. Syn. 2006, 3, 313–326. [Google Scholar] [CrossRef]
- Espinet, P.; Echavarren, A.M. The mechanisms of the Stille reaction. Angew. Chem. Int. Ed. Engl. 2004, 43, 4704–4734. [Google Scholar]
- Doucet, H.; Hierso, J.-C. Palladium-based catalytic systems for the synthesis of conjugated enynes by Sonogashira reactions and related alkynylations. Angew. Chem. Int. Ed. Engl. 2007, 46, 834–871. [Google Scholar] [CrossRef]
- Chinchilla, R.; Najera, C. The Sonogashira reaction: A booming methodology in synthetic organic chemistry. Chem. Rev. 2007, 107, 874–922. [Google Scholar]
- Schlummer, B.; Scholz, U. Palladium-catalyzed C–N and C–O coupling—A practical guide from an industrial vantage point. Adv. Synth. Catal. 2004, 346, 1599–1626. [Google Scholar]
- Hartwig, J.F. Carbon-heteroatom bond-forming reductive eliminations of amines, ethers, and sulfides. Acc. Chem. Res. 1998, 31, 852–860. [Google Scholar] [CrossRef]
- Casalnuovo, A.L.; Calabrese, J.C. Palladium-catalyzed alkylations in aqueous media. J. Am. Chem. Soc. 1990, 112, 4324–4330. [Google Scholar] [CrossRef]
- Shaughnessy, K.H. Beyond TPPTS: New approaches to the development of efficient palladium-catalyzed aqueous-phase cross-coupling reactions. Eur. J. Org. Chem. 2006, 1827–1835. [Google Scholar] [CrossRef]
- Zhou, J.; Li, X.; Sun, H. An efficient and recyclable water-soluble cyclopalladated complex for aqueous Suzuki reactions under aerial conditions. J. Organomet. Chem. 2010, 695, 297–303. [Google Scholar]
- Liu, C.; Ni, Q.; Hu, P.; Qiu, J. Oxygen-promoted PdCl2-catalyzed ligand-free Suzuki reaction in aqueous media. Org. Biomol. Chem. 2011, 9, 1054–1060. [Google Scholar]
- Wang, J.; Song, G.; Peng, Y. Reusable Pd nanoparticles immobilized on functional ionic liquid co-polymerized with styrene for Suzuki reactions in water-ethanol solution. Tetrahedron Lett. 2011, 52, 1477–1480. [Google Scholar] [CrossRef]
- Mejías, N.; Pleixats, R.; Shafir, A.; Medio-Simón, M.; Asensio, G. Water-soluble palladium nanoparticles: Click synthesis and applications as a recyclable catalyst in Suzuki cross-couplings in aqueous media. Eur. J. Org. Chem. 2010, 5090–5099. [Google Scholar]
- Monopoli, A.; Nacci, A.; Calò, V.; Ciminale, F.; Cotugno, P.; Mangone, A.; Giannossa, L.C.; Azzone, P.; Cioffi, N. Palladium/zirconium oxide nanocomposite as a highly recyclable catalyst for C–C coupling reactions in water. Molecules 2010, 15, 4511–4525. [Google Scholar] [CrossRef]
- Shaughnessy, K.H. Hydrophilic ligands and their application in aqueous-phase metal-catalyzed reactions. Chem. Rev. 2009, 109, 643–710. [Google Scholar]
- Casalnuovo, A.L.; Calabrese, J.C. Palladium-catalyzed alkylations in aqueous media. J. Am. Chem. Soc. 1990, 112, 4324–4330. [Google Scholar] [CrossRef]
- Genet, J.P.; Blart, E.; Savignac, M. Palladium-catalyzed cross-coupling reactions in a homogeneous aqueous medium. Synlett 1992, 715–717. [Google Scholar]
- Ueda, M.; Nishimura, M.; Miyaura, N. A palladium-catalyzed biaryl coupling of arylboronic acids in aqueous media using a gluconamide-substituted triphenylphosphine (GLCAphos) ligand. Synlett 2000, 6, 856–858. [Google Scholar]
- Moore, L.R.; Shaughnessy, K.H. Efficient aqueous-phase Heck and Suzuki couplings of aryl bromides using tri(4,6-dimethyl-3-sulfonatophenyl)phosphine trisodium salt (TXPTS). Org. Lett. 2004, 2, 225–228. [Google Scholar]
- Moore, L.R.; Western, E.C.; Craciun, R.; Spruell, J.M.; Dixon, D.A.; O’Halloran, K.P.; Shaughnessy, K.H. Sterically demanding, sulfonated, triarylphosphines: Application to palladium-catalyzed cross-coupling, steric and electronic properties, and coordination chemistry. Organometallics 2008, 27, 576–593. [Google Scholar]
- Wallow, T.I.; Novak, B.M. Highly efficient and accelerated Suzuki aryl couplings mediated by phosphine-free palladium sources. J. Org. Chem. 1994, 59, 5034–5037. [Google Scholar] [CrossRef]
- Wu, W.-Y.; Chen, S.-N.; Tsai, F.-Y. Recyclable and highly active cationic 2,2’-bipyridyl palladium(II) catalyst for Suzuki cross-coupling reaction in water. Tetrahedron Lett. 2006, 47, 9267–9270. [Google Scholar]
- Pawar, S.S.; Uppalla, L.S.; Shingare, M.S.; Thore, S.N. Sodium 2-(2-pyridin-3-ylethylamino)ethyl sulfonate: an efficient ligand and base for palladium-catalyzed Suzuki reaction in aqueous media. Tetrahedron Lett. 2008, 49, 5858–5862. [Google Scholar] [CrossRef]
- Huang, R.; Shaughnessy, K.H. Water-soluble palladacycles as precursors to highly recyclable catalysts for the Suzuki coupling of aryl bromides in aqueous solvents. Organometallics 2006, 25, 4105–4112. [Google Scholar] [CrossRef]
- Frost, B.J.; Mebi, C.A. Aqueous organometallic chemistry: Synthesis, structure, and reactivity of the water-soluble metal hydride CpRu(PTA)2H. Organometallics 2004, 23, 5317–5323. [Google Scholar] [CrossRef]
- Wong, G.W.; Harkreader, J.L.; Mebi, C.A.; Frost, B.J. Synthesis and coordination chemistry of a novel bidentate phosphine: 6-(Diphenylphosphino)-1,3,5-triaza-7-phophaadamantane (PTA-PPh2). Inorg. Chem. 2006, 45, 6748–6755. [Google Scholar]
- Huang, R.; Frost, B.J. Development of a series of P(CH2NCHR)3 and trisubstituted 1,3,5-triaza-7-phosphaadamantane ligands. Inorg. Chem. 2007, 46, 10962–10964. [Google Scholar] [CrossRef]
- Mebi, C.A.; Frost, B.J. Isomerization of trans-[Ru(PTA)4Cl2] to cis-[Ru(PTA)4Cl2] in water and organic solvent: Revisiting the chemistry of [Ru(PTA)4Cl2]. Inorg. Chem. 2007, 46, 7115–7120. [Google Scholar]
- Frost, B.J.; Harkreader, J.L.; Bautista, C.M. Synthesis and solid state structure of Co(II) complexes of O=PTA. Inorg. Chem. Comm. 2008, 11, 580–583. [Google Scholar] [CrossRef]
- Wong, G.W.; Lee, W.-C.; Frost, B.J. Insertion of CO2, ketones, and aldehydes into the C-Li bond of 1,3,5-triaza-7-phosphaadamantan-6-yllithium. Inorg. Chem. 2008, 47, 612–620. [Google Scholar]
- Frost, B.J.; Lee, W.-C.; Pal, K.; Kim, T.H.; VanDerveer, D.; Rabinovich, D. Synthesis, structure, and coordination chemistry of O=PTA and S=PTA with group 12 metals (PTA = 1,3,5-triaza-7-phosphaadamantane). Polyhedron 2010, 29, 2373–2380. [Google Scholar]
- Assmann, B.; Angermaier, K.; Paul, M.; Riede, J.; Schmidbaur, H. Synthesis of 7-alkyl/aryl-1,3,5-triaza-7-phosphoniaadamantane cations and their reductive cleavage to novel N-methyl-P-alkyl/aryl[3.3.1]bicyclononane ligands. Chem. Ber. 1995, 128, 891–900. [Google Scholar] [CrossRef]
- Siele, V.I. Some reactions of 1,3,5-Triaza-7-phosphaadamantane and its 7-oxide. J. Heterocycl. Chem. 1977, 14, 337–339. [Google Scholar] [CrossRef]
- Darensbourg, D.J.; Ortiz, C.G.; Kamplain, J.W. A new water-soluble phosphine derived from 1,3,5-triaza-7-phospaadamantane (PTA), 3,7-diacetyl-1,3,7-triaza-5-phosphabicyclo[3.3.1]nonane. Structural, bonding, and solubility properties. Organometallics 2004, 23, 1747–1754. [Google Scholar]
- Frank, A.W.; Daigle, D.J. Triacidic salts of tris(aminomethly)phosphines and their oxides. Phosphorus Sulfur Relat. Elem. 1981, 10, 255–259. [Google Scholar]
- Darensbourg, D.J.; Stafford, N.W.; Joo, F.; Reibenspies, J.H. Water-soluble organometallic compounds. 5. The regio-selective catalytic hydrogenation of unsaturated aldehydes to saturated aldehydes in an aqueous two-phase solvent system using 1,3,5-triaza-7-phosphaadamantane complexes of rhodium. J. Organomet. Chem. 1995, 488, 99–108. [Google Scholar]
- Darensbourg, D.J.; Joo, F.; Katho, A.; Stafford, J.N.W.; Benyei, A.; Reibenspies, J.H. Nucleophilic addition of a water-soluble phosphine to aldehydes. Isolation of (1-hydroxyalkyl)phosphonium salts and the crystal structure of the (1-methoxy-1-benzyl)(m-sulfonatophenyl)diphenyl phosphonium salt. Inorg. Chem. 1994, 33, 175–177. [Google Scholar] [CrossRef]
- Darensbourg, D.J.; Joo, F.; Kannisto, M.; Katho, A.; Reibenspies, J.H.; Daigle, D.J. Water-soluble organometallic compunds. 4. Catalytic hydrogenation of aldehydes in an aqueous two-phase solvent system using a 1,3,5-triaza-7-phosphaadamantane complex of ruthenium. Inorg. Chem. 1994, 33, 200–208. [Google Scholar]
- Darensbourg, D.J.; Joo, F.; Kannisto, M.; Katho, A.; Reibenspies, J.H. Water-soluble organometallic compounds. 2. Catalytic hydrogenation of aldehydes and olefins by new water-soluble 1,3,5-triaza-7-phosphaadamantane complexes of ruthenium and rhodium. Organometallics 1992, 11, 1990–1993. [Google Scholar] [CrossRef]
- Kovacs, J.; Todd, T.D.; Reibenspies, J.H.; Joo, F.; Darensbourg, D.J. Water-soluble organometallic compounds. 9. Catalytic hydrogenation and selective isomerization of olefins by water-soluble analogues of Vaska's complex. Organometallics 2000, 19, 3963–3969. [Google Scholar] [CrossRef]
- Phillips, A.D.; Gonsalvi, L.; Romerosa, A.; Vizza, F.; Peruzzini, M. Coordination chemistry of 1,3,5-triaza-7-phophaadamantane (PTA): Transition metal complexes and related catalytic, medicinal, and photoluminescent applications. Coord. Chem. Rev. 2004, 248, 955–993. [Google Scholar]
- Bravo, J.; Bolano, S.; Gonsalvi, L.; Peruzzini, M. Coordination chemistry of 1,3,5-triaza-7-phosphaadamantane (PTA) and derivatives, Part II. The quest for tailoring ligands, complexes and related applications. Coord. Chem. Rev. 2010, 254, 555–607. [Google Scholar]
- Krogstad, D.A.; Cho, J.; DeBoer, A.J.; Klitzke, J.A.; Sanow, W.R.; Williams, H.A.; Halfen, J.A. Platinum (II) and Palladium(II) 1,3,5-triaza-7-phosphaadamantane (PTA) complexes as intramolecular hydroamination catalysts in aqueous and organic media. Inorg. Chim. Acta 2006, 359, 136–148. [Google Scholar] [CrossRef]
- Tang, X.; Zhang, B.; He, Z.; Gao, R.; He, Z. 1,3,5-Triaza-7-phosphadamantane (PTA): A practical and versatile nucleophilic phosphine organocatalyst. Adv. Synth. Catal. 2007, 349, 2007–2017. [Google Scholar] [CrossRef]
- He, Z.; Tang, X.; Chen, Y.; He, Z. The First air-stable and efficient nucleophilic trialkylphophine organocatalyst for the Baylis-Hillman reaction. Adv. Synth. Catal. 2006, 348, 413–417. [Google Scholar] [CrossRef]
- Ruiz, J.; Cutillas, N.; Lopez, F.; Lopez, G.; Bautista, D. A Copper and amine-free Sonogashira reaction of aryl halides catalyzed by 1,3,5-triaza-7-phosphaadamantane palladium systems. Organometallics 2006, 25, 5768–5773. [Google Scholar]
- García-Álvarez, J.; Díez, J.; Gimeno, J. A highly efficient copper(I) catalyst for the 1,3-dipolar cycloaddition of azides with terminal and 1-iodoalkynes in water: Regioselective synthesis of 1,4-disubstituted and 1,4,5-trisubstituted 1,2,3-triazoles. Green Chem. 2010, 12, 2127–2130. [Google Scholar] [CrossRef]
- Komine, N.; Ishiguro, K.; Kanai, S.; Hirano, M.; Komiya, S. Branch selective allylation of acetylacetone catalyzed by Water-soluble rhodium complex catalyst. J. Organomet. Chem. 2011, 696, 1927–1930. [Google Scholar] [CrossRef]
- Strotman, N.A.; Chobanian, H.R.; He, J.; Guo, Y.; Dormer, P.G.; Jones, C.M.; Steves, J.E. Catalyst-controlled regioselective Suzuki coupling at both postitions of dihaloimidazoles, dihalooxazoles, and dihalothiazoles. J. Org. Chem. 2010, 75, 1733–1739. [Google Scholar] [CrossRef]
- Caporali, M.; Bianchini, C.; Bolaño, S.; Bosquain, S.S.; Gonsalvi, L.; Oberhauser, W.; Rossin, A.; Peruzzini, M. Synthesis and characterization of κ1-P and κ1-P,N palladium (II) complexes of the open cage water soluble aminophosphine PTN. Inorg. Chim. Acta 2008, 361, 3017–3023. [Google Scholar]
- Weng, Z.; Teo, S.; Hor, T.S. A. metal unsaturated and ligand hemilability in Suzuki coupling. Acc. Chem. Res. 2007, 40, 676–684. [Google Scholar] [CrossRef]
- Liu, C.; Ni, Q.; Bao, F.; Qiu, J. A simple and efficient protocol for a palladium-catalyzed ligand-free Suzuki reaction at room temperature in aqueous DMF. Green Chem. 2011, 13, 1260–1266. [Google Scholar] [CrossRef]
- Daigle, D.J.; Pepperman, A.B., Jr.; Vail, S.L. Synthesis of a monphosphorus analog of hexamethylenetetramine. J. Heterocycl. Chem. 1974, 11, 407–408. [Google Scholar]
- Drew, D.; Doyle, J.R. Cyclic diolefin complexes of platinum and palladium. Inorg. Synth. 1990, 28, 346–349. [Google Scholar] [CrossRef]
- Arvela, R.K.; Leadbeater, N.E. Suzuki coupling of aryl chlorides with phenylboronic acid in water, using microwave heating with simultaneous cooling. Org. Lett. 2005, 7, 2101–2104. [Google Scholar] [CrossRef]
- Huang, R.; Shaughnessy, K.H. Water-soluble palladacycles as precursors to highly recyclable catalysts for the Suzuki coupling of aryl bromides in aqueous solvents. Organometallics 2006, 25, 4105–4112. [Google Scholar] [CrossRef]
- Wolfe, J.P.; Singer, R.A.; Yang, B.H.; Buchwald, S.L. Highly active palladium catalysts for suzuki coupling reactions. J. Am. Chem. Soc. 1999, 121, 9550–9561. [Google Scholar] [CrossRef]
- Hiyama, J.-H.; Li, C.-C.; Deng, C.-L.; Liu, W.-J.; Xie, Y.-X. Pd(OAc)2/DABCO as an inexpensive and efficient catalytic system for Hiyama cross-coupling reactions of aryl halides with aryltrimethoxysilanes. Synthesis 2005, 18, 3039–3044. [Google Scholar]
- Sheldrick, G.M. SHELXTL: Structure Determination Software Suite, version 6.10; Bruker AXS: Madison, WI, USA, 2001. [Google Scholar]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. Sect. A 2008, 64, 112–122. [Google Scholar] [CrossRef]
- Sample Availability: Samples of most compounds are available from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Weeden, J.A.; Huang, R.; Galloway, K.D.; Gingrich, P.W.; Frost, B.J. The Suzuki Reaction in Aqueous Media Promoted by P, N Ligands. Molecules 2011, 16, 6215-6231. https://doi.org/10.3390/molecules16086215
Weeden JA, Huang R, Galloway KD, Gingrich PW, Frost BJ. The Suzuki Reaction in Aqueous Media Promoted by P, N Ligands. Molecules. 2011; 16(8):6215-6231. https://doi.org/10.3390/molecules16086215
Chicago/Turabian StyleWeeden, Jason A., Rongcai Huang, Kathryn D. Galloway, Phillip W. Gingrich, and Brian J. Frost. 2011. "The Suzuki Reaction in Aqueous Media Promoted by P, N Ligands" Molecules 16, no. 8: 6215-6231. https://doi.org/10.3390/molecules16086215
APA StyleWeeden, J. A., Huang, R., Galloway, K. D., Gingrich, P. W., & Frost, B. J. (2011). The Suzuki Reaction in Aqueous Media Promoted by P, N Ligands. Molecules, 16(8), 6215-6231. https://doi.org/10.3390/molecules16086215