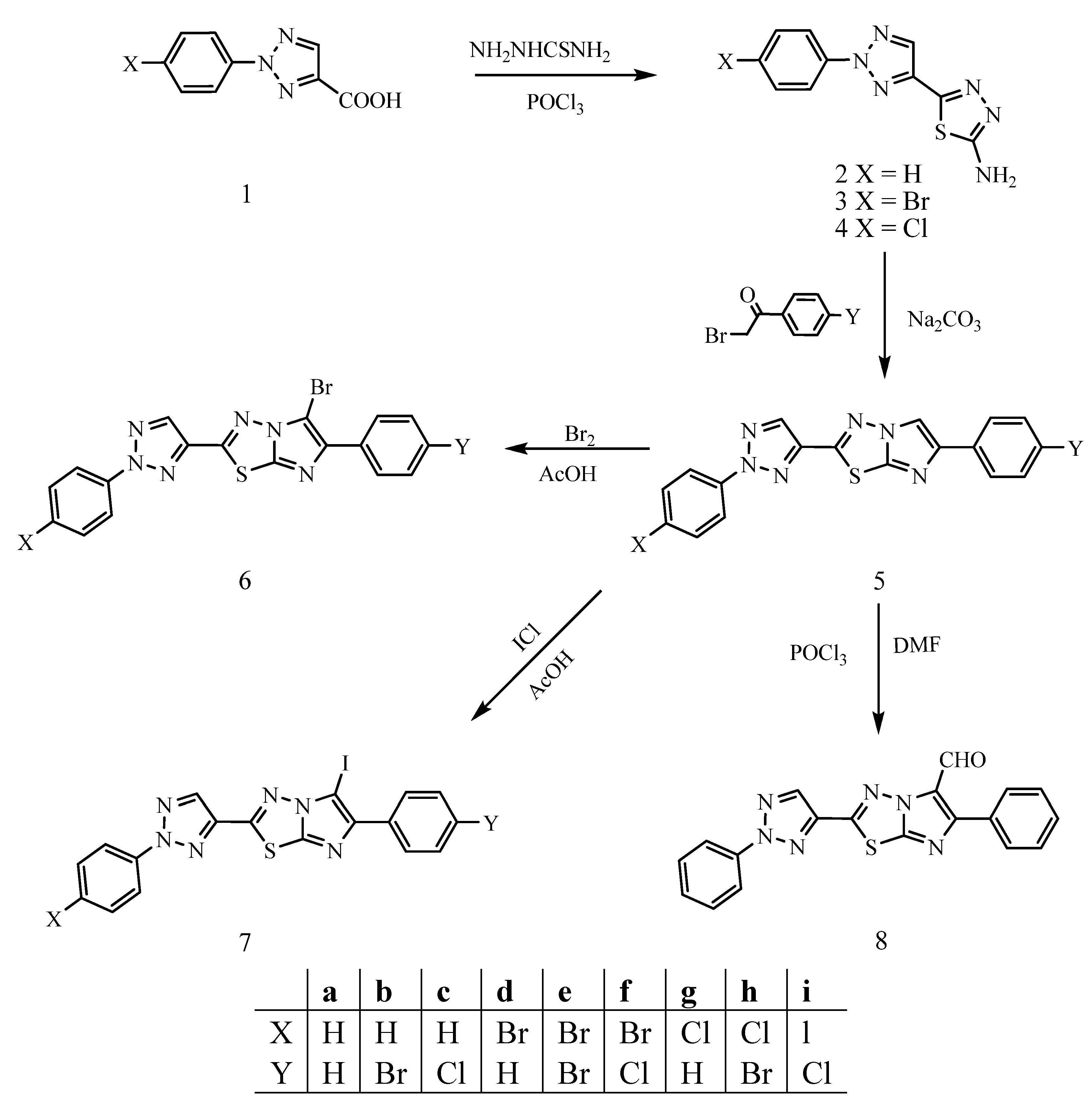
Synthesis and Antibacterial Activities of Novel Imidazo[2,1-b]-1,3,4-thiadiazoles
Abstract
:1. Introduction
2. Results and Discussion
| Cpd. No. | IR (cm−1) | 1H-NMR Chemical Shift (δ/ppm) * | ||||||
|---|---|---|---|---|---|---|---|---|
| C=N | C=N | C=N | C=C | C-S-C | ArH | Imidazole proton | Triazole proton | |
| (thiadiazole ring) | (imidazole ring) | (triazole ring) | (imidazole ring) | (thiadiazole ring) | (m) | (s, 1H) | (s, 1H) | |
| 5a | 1638 | 1594 | 1534 | 1490 | 690 | 7.33–8.14 (10H) | 8.07 | 8.33 |
| 5b | 1635 | 1595 | 1532 | 1490 | 669 | 7.61–8.84 (9H) | 7.50 | 8.46 |
| 5c | 1634 | 1594 | 1536 | 1490 | 669 | |||
| 5d | 1629 | 1527 | 1540 | 1844 | 690 | |||
| 5e | 1631 | 1555 | 1528 | 1488 | 669 | |||
| 5f | 1626 | 1600 | 1529 | 1488 | 669 | 7.42–8.11 (8H) | 8.06 | 8.50 |
| 5g | 1651 | 1601 | 1538 | 1486 | 690 | |||
| 5h | 1649 | 1565 | 1533 | 1487 | 671 | |||
| 5i | 1652 | 1599 | 1533 | 1485 | 670 | |||
| 6a | 1640 | 1597 | 1545 | 1489 | 662 | 7.25–8.14 (10H) | 8.41 | |
| 6b | 1641 | 1594 | 1522 | 1487 | 669 | 7.44–8.15 (9H) | 8.42 | |
| 6c | 1637 | 1595 | 1524 | 1488 | 669 | 7.42–8.14 (9H) | 8.41 | |
| 6d | 1632 | 1549 | 1526 | 1436 | 686 | 7.35–8.06 (9H) | 8.42 | |
| 6e | 1632 | 1552 | 1522 | 1486 | 669 | 7.57–8.03 (8H) | 8.42 | |
| 6f | 1628 | 1545 | 1521 | 1686 | 669 | 7.42–8.04 (8H) | 8.42 | |
| 6g | 1645 | 1550 | 1510 | 1488 | 686 | 7.36–8.10 (9H) | 8.42 | |
| 6h | 1642 | 1568 | 1519 | 1487 | 672 | 7.49–8.09 (8H) | 8.41 | |
| 6i | 1641 | 1585 | 1522 | 1488 | 672 | 7.42–8.09 (8H) | 8.41 | |
| 7a | 1634 | 1595 | 1532 | 1488 | 663 | 7.37–8.15 (10H) | 8.43 | |
| 7b | 1640 | 1594 | 1527 | 1489 | 668 | 7.42–8.14 (9H) | 8.41 | |
| 7c | 1636 | 1596 | 1530 | 1489 | 669 | 7.43–8.14 (9H) | 8.41 | |
| 7d | 1642 | 1547 | 1522 | 1486 | 685 | |||
| 7e | 1633 | 1565 | 1535 | 1485 | 671 | 7.58–8.03 (8H) | 8.41 | |
| 7f | 1633 | 1565 | 1521 | 1487 | 671 | 7.43–8.03 (8H) | 8.42 | |
| 7g | 1639 | 1545 | 1509 | 1488 | 685 | 7.39–8.11 (9H) | 8.42 | |
| 7h | 1640 | 1595 | 1555 | 1487 | 669 | 7.49–8.09 (8H) | 8.41 | |
| 7i | 1639 | 1598 | 1565 | 1486 | 670 | 7.43–8.10 (8H) | 8.41 | |
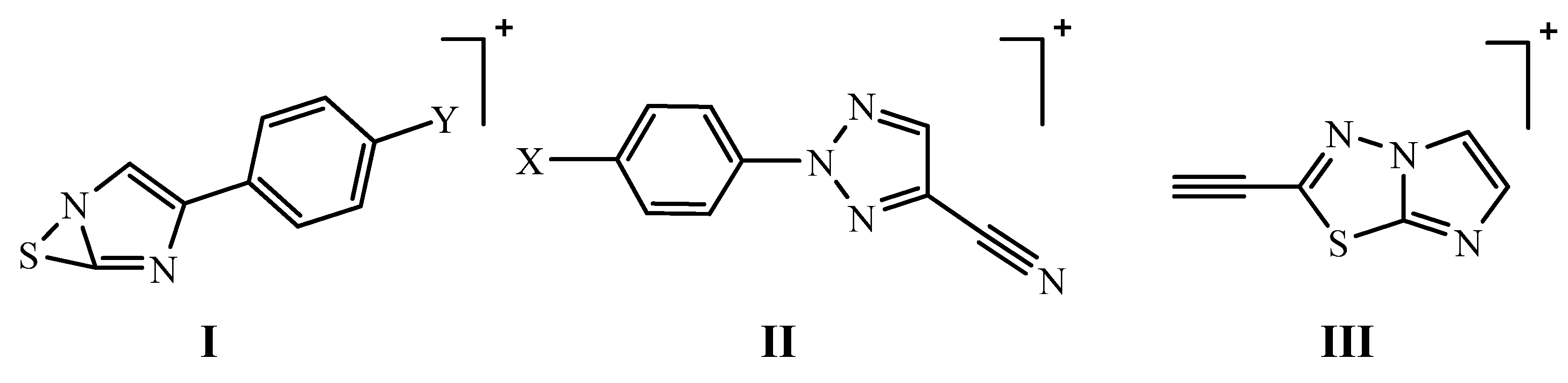
| Cpd. No. | M | I | II | III |
|---|---|---|---|---|
| 5a | 33 | 2 | 11 | 8 |
| 5b | 100,78 | 10 | 70 | 5 |
| 5g | 21 | 5 | 36 | 4 |
| 5i | 29 | 24 | 40 | 4 |
| 6a | 69 | 100 | 34 | 18 |
| 6d | 7 | 57 | 100 | 12 |
| 6f | 8 | 40 | 8 | 7 |
| 6g | 22 | 100 | 29 | 19 |
| 6i | 5 | 35 | 34 | 1 |
| 7a | 100 | 73 | 22 | 18 |
| 7e | 39 | 16,15 | 44,46 | 2 |
| 7g | 46 | 100 | 18 | 57 |
| 7i | 14 | 36 | 18 | 1 |
Biological screening: antimicrobial activity tests
| Test microorganisms | S | C | Ps | E |
|---|---|---|---|---|
| Test compounds | Average inhibition zone diameter in mms | |||
| Imepenam | 30 | - | 30 | 26 |
| Ampicillin | 30 | - | - | - |
| Clotrimazole | - | 40 | - | - |
| 2 | - | 18 | 22 | 17 |
| 3 | - | 18 | 22 | 16 |
| 4 | - | 18 | 22 | 16 |
| 5a | - | 18 | 22 | 17 |
| 5b | - | 18 | 22 | 18 |
| 5c | - | 18 | 22 | 16 |
| 5d | - | 18 | 22 | 20 |
| 5e | - | 18 | 22 | 16 |
| 5f | - | 18 | 22 | 18 |
| 5g | - | 18 | 22 | 16 |
| 5h | - | 19 | 22 | 18 |
| 5i | - | 18 | 23 | 16 |
| 6a | - | 19 | 22 | 17 |
| 6b | - | 18 | 23 | 16 |
| 6c | - | 18 | 22 | 17 |
| 6d | - | 18 | 23 | 17 |
| 6e | - | 18 | 22 | 16 |
| 6f | - | 18 | 22 | 17 |
| 6g | - | 18 | 22 | 17 |
| 6h | - | 18 | 22 | 20 |
| 6i | - | 18 | 22 | 16 |
| 7a | - | 18 | 22 | 19 |
| 7b | - | 18 | 26 | 19 |
| 7c | - | 18 | 22 | 16 |
| 7d | - | 18 | 22 | 16 |
| 7e | - | 19 | 22 | 17 |
| 7f | - | 19 | 22 | 17 |
| 7g | - | 19 | 22 | 16 |
| 7h | - | 18 | 22 | 17 |
| 7i | - | 18 | 22 | 16 |
| 8 | - | 18 | 22 | 19 |
| DMF | - | 18 | 22 | 16 |
3. Experimental
3.1. General
3.2.1. 2-Amino-5-(2-aryl-2H-1,2,3-triazol-4-yl)-1,3,4-thiadiazoles (2-4)
3.2.2. 6-Aryl-2-(2-aryl-2H-1,2,3-triazol-4-yl)imidazo[2,1-b]-1,3,4-thiadiazoles 5a-i
3.2.3. 6-Aryl-2-(2-aryl-2H-1,2,3-triazol-4-yl)-5-haloimidazo[2,1-b]-1,3,4-thiadiazoles 6a-i and 7a-i
| Cpd. No. | Mp (°C) | Yield (%) | Formula | Calcd. % | Found % | ||||
|---|---|---|---|---|---|---|---|---|---|
| C | H | N | C | H | N | ||||
| 5a | 225-226 | 60 | C18H12N6S | 62.77 | 3.51 | 24.40 | 62.50 | 3.42 | 24.43 |
| 5b | 253-254 | 62 | C18H11BrN6S | 51.07 | 2.62 | 19.85 | 51.00 | 2.32 | 19.92 |
| 5c | 257-258 | 61 | C18H11ClN6S | 57.07 | 2.93 | 22.18 | 57.11 | 2.86 | 22.20 |
| 5d | 285-286 | 61 | C18H11BrN6S | 51.07 | 2.62 | 19.85 | 51.15 | 2.40 | 20.06 |
| 5e | 281-282 | 60 | C18H10 Br2N6S | 43.05 | 2.01 | 16.73 | 43.07 | 2.00 | 16.81 |
| 5f | 276-277 | 66 | C18H10BrClN6S | 47.23 | 2.20 | 18.36 | 47.31 | 2.12 | 18.02 |
| 5g | 279-280 | 58 | C18H11ClN6S | 57.07 | 2.93 | 22.18 | 57.10 | 2.92 | 22.21 |
| 5h | 267-268 | 55 | C18H10BrClN6S | 47.23 | 2.20 | 18.36 | 47.19 | 2.26 | 18.41 |
| 5i | 271-272 | 58 | C18H10 Cl2N6S | 52.31 | 2.44 | 20.33 | 52.40 | 2.40 | 20.21 |
| 6a | 221-222 | 80 | C18H11BrN6S | 51.07 | 2.62 | 19.85 | 51.12 | 2.42 | 20.03 |
| 6b | 261-262 | 80 | C18H10 Br2N6S | 43.05 | 2.01 | 16.73 | 43.12 | 2.00 | 16.92 |
| 6c | 255-256 | 80 | C18H10BrClN6S | 47.23 | 2.20 | 18.36 | 47.21 | 2.15 | 18.38 |
| 6d | 265-266 | 82 | C18H10 Br2N6S | 43.05 | 2.01 | 16.73 | 43.05 | 2.01 | 16.73 |
| 6e | 246-247 | 76 | C18H9 Br3N6S | 37.21 | 1.56 | 14.46 | 37.22 | 1.81 | 14.51 |
| 6f | 248-249 | 80 | C18H9Br2ClN6S | 40.29 | 1.69 | 15.66 | 40.01 | 1.81 | 15.62 |
| 6g | 257-258 | 80 | C18H10BrClN6S | 47.23 | 2.20 | 18.36 | 47.12 | 2.25 | 18.39 |
| 6h | 259-260 | 80 | C18H9Br2ClN6S | 0.29 | 1.69 | 15.66 | 40.42 | 1.72 | 15.52 |
| 6i | 237-238 | 80 | C18H9 BrCl2N6S | 43.93 | 1.84 | 17.08 | 43.90 | 1.87 | 16.90 |
| 7a | 270-271 | 85 | C18H11IN6S | 45.97 | 2.36 | 17.87 | 45.82 | 2.39 | 17.85 |
| 7b | 251-252 | 83 | C18H10BrIN6S | 39.37 | 1.84 | 15.30 | 39.40 | 1.90 | 15.21 |
| 7c | 251-252 | 82 | C18H10ClIN6S | 42.83 | 2.00 | 16.65 | 42.90 | 2.03 | 16.82 |
| 7d | 273-274 | 85 | C18H10BrIN6S | 39.37 | 1.84 | 15.30 | 39.21 | 1.61 | 15.42 |
| 7e | 251-252 | 80 | C18H9Br2IN6S | 34.42 | 1.44 | 13.38 | 34.50 | 1.49 | 13.42 |
| 7f | 243-244 | 85 | C18H9BrClIN6S | 37.04 | 1.55 | 14.40 | 37.24 | 1.52 | 14.43 |
| 7g | 285-286 | 82 | C18H10ClIN6S | 42.83 | 2.00 | 16.65 | 42.72 | 1.95 | 16.62 |
| 7h | 255-256 | 84 | C18H9BrClIN6S | 37.04 | 1.55 | 14.40 | 36.92 | 1.61 | 14.41 |
| 7i | 253-254 | 80 | C18H9Cl2IN6S | 40.10 | 1.68 | 15.59 | 40.20 | 1.72 | 15.51 |
3.2.4. 5-Formyl-6-phenyl-2-(2-phenyl-2H-1,2,3-triazol-4-yl)imidazo[2,1-b]-1,3,4-thiadiazole (8)
3.3. Antibacterial Activity Tests
4. Conclusions
References
- Gujrel, M.S.; Patnaik, P.M.; Kaul, R.; Parikh, H.K.; Conradt, C.; Tamhankar, C.P.; Daftary, G.V. Efficacy of hydrolytic enzymes in preventing radiation therapy-induced side effects in patients with head and neck cancers. Cancer Chemother. Pharmacol. 2001, 47, S23–S28. [Google Scholar] [CrossRef]
- Stockler, M.R.; Wilcken, N.J.C.; Coates, A. Chemotherapy for Advanced Breast Cancer – How Long Should it Continue? Brest Cancer Res. Treat. 2003, 81, 49–52. [Google Scholar] [CrossRef]
- Senff-Ribeiro, A.; Echevarria, A.; Silva, E.F.; Veiga, S.S.; Oliveria, M.B. Antimelanoma activity of 1,3,4-thiadiazolium mesoionics: a structure-activity relationship study. Anticancer Drugs 2004, 15, 269–275. [Google Scholar] [CrossRef]
- Rzeski, W.; Matysiak, J.; Kandefer-Szerszen, M. Anticancer, neuroprotective and computational studies of 2-amino-1,3,4-thiadiazole based compound. Bioorg. Med. Chem. 2007, 15, 3201–3207. [Google Scholar] [CrossRef]
- Khalil, M.A.; El-Sayed, O.A.; El-Shamy, H.A. Synthesis and Antimicrobial Evaluation of Novel Oxa(thia)diazolylquinolines and Oxa(thia)diazepino[7,6-b]quinolines. Arch. Pharm. (Weinheim) 1993, 326, 489–492. [Google Scholar] [CrossRef]
- Lamani, R.S.; Shatty, N.S.; Kamble, R.R.; Khazi, I.A.M. Synthesis and antimicrobial studies of novel methylene bridged benzisoxazolylimidazo[2,1-b][1,3,4]thiadiazole derivatives. Eur. J. Med. Chem. 2009, 44, 2828–2833. [Google Scholar]
- Gadad, A.K.; Palkar, M.B.; Anand, K.; Noolvi, M.N.; Boreddy, T.S.; Wagwade, J. Synthesis and biological evaluation of 2-trifluromethyl/sulfonamido-5,6-diaryl substituted imidazo[2,1-b]-1,3,4-thiadiazoles: A novel class of cyclooxygenase-2-inhibitors. Bioorg. Med. Chem. 2008, 16, 276–283. [Google Scholar] [CrossRef]
- Joshi, K.C.; Pathak, V.N.; Panwar, M.P. Organic pesticides, Part XVIII: Synthesis and fungicidal activity of fluorine containing 2,5-disubstituted imidazo [2,1-b]-1,3,4-thiadiazole hydrobromides. J. Indian Chem. Soc. 1979, 56, 716–717. [Google Scholar]
- Jadhav, V.B.; Kulkarni, M.V.; Rasal, V.P.; Biradar, S.S.; Vinay, M.D. Synthesis and anti-inflammatory evaluation of methylene bridged benzofuranylimidazo[2,1-b][1,3,4]thiadiazoles. Eur. J. Med. Chem. 2008, 43, 1721–1729. [Google Scholar] [CrossRef]
- Kolavi, G.; Hegde, V.; Khazi, I.A. Intramolecularamidation: Synthesis of novel imidazo[2,1-b][1,3,4]thiadiazole and imidazo[2,1-b][1,3]thiazole fused diazepinones. Tetrahedron Lett. 2006, 47, 2811–2814. [Google Scholar] [CrossRef]
- Terzioglu, N.; Gürosy, A. Synthesis and anticancer evaluation of some new hydrazone derivatives of 2,6-dimethylimidazo[2,1-b][1,3,4]thiadiazole-5-carbohydrazide. Eur. J. Med. Chem. 2003, 38, 781–786. [Google Scholar] [CrossRef]
- Schenetti, M.L.; Taddei, F.; Greci, L.; Marchetti, L.; Milani, G.; Milani, G.; Andreetti, G.D.; Bocelli, G.; Sgarabotto, P. Secondary deuterium kinetic isotope effects in the solvolysis of 4-t-butylcyclohexyl and bicyclo[3.2.l]octan-3-y toluene-P-sulphonates. New results using 50% aqueous ethanol, acetic acid, and 97% aqueous hexafluoropropan-2-ol. J. Chem. Soc. Perkin Trans. II 1980, 2, 421–426. [Google Scholar]
- El-Shafei, A.K.; Sultan, A.; Vernin, G. A Convenient Synthesis of Polyfused Heterocyclic Systems from Heterocyclic Amines and 2,3-Dichloronaphthquinone Using Phase Transfer Catalysis. Heterocycles 1982, 19, 333–338. [Google Scholar] [CrossRef]
- Torgova, S.I.; Shtykov, N.M.; Abolin, A.G.; Karamysheva, L.A.; Ivashchenko, A.L.; Barnik, M.I. Liquid crystal imidazo[2,1-b]-1,3,4-thiadiazoles. I. Synthesis and mesomorphic and spectral characteristics of 2,6-disubstituted imidazo[2,1-b]-1,3,4-thiadiazoles. J. Org. Chem. USSR (Engl. Transl.) 1988, 24, 1177. [Google Scholar]
- Amery, W.K.; Hoerig, C.H.; Fenichel, R.I.; Chirigos, M.A. Immune Modulation Agents and their Mechanism; Mercel Dekker: New York, NY, USA, 1984; pp. 383–408. [Google Scholar]
- Torgova, S.I.; Abolin, A.G.; Roitman, K.B.; Karamysheva, L.A.; Ivashchenko, A.V. Liquid crystal imidazo[2,1-b]-1,3,4-thiadiazoles II. Synthesis and mesomorphic and spectral properties of liquid crystalline 5-substituted imidazo[2,1-b]-1,3,4-thiadiazoles. J. Org. Chem. USSR (Engl. Transl.) 1988, 24, 172. [Google Scholar]
- Terzioglu, N.; Gürsoy, A. Synthesis and anticancer evaluation of some new hydrazone derivatives of 2,6-dimethylimidazo[2,1-b][1,3,4]thiadiazole-5-carbohydrazide. Eur. J. Med. Chem. 2003, 38, 781–786. [Google Scholar] [CrossRef]
- Kolavi, G.; Hegde, V.; Khazi, I.A.; Gadad, P. Synthesis and evaluation of antitubercular activity of imidazo[2,1-b][1,3,4]thiadiazole derivatives. Bioorg. Med. Chem. 2006, 14, 3069–3080. [Google Scholar]
- Gadad, A.K.; Mahajanshetti, C.S.; Nimbalkar, S.; Raichurkar, A. Synthesis and antibacterial activity of some 5-guanylhydrazone/thiocyanato-6-arylimidazo[2,1-b]-1,3,4-thiadiazole-2-sulfonamide derivatives. Eur. J. Med. Chem. 2000, 35, 853–857. [Google Scholar] [CrossRef]
- Andotra, C.S.; Langer, T.C.; Kotha, A. Synthesis and Antifungal Activity of some Substituted 1,3,4-Thiadiazolo[3,2-a]-s-triazin-5-phenyl-7-thiones and Imidazo-[2,1-b]-1,3,4-thiadiazol-5-ones. J. Indian Chem. Soc. 1997, 74, 125–127. [Google Scholar]
- Khazi, I.A.M.; Mahajanshetti, C.S.; Gadad, A.K.; Tarnalli, A.D.; Sultanpur, C.M. Synthesis and anticonvulsant and analgesic activities of some 6-substituted imidazo[2,1-b]-1,3,4-thiadiazole-2-sulfonamides and their 5-bromo derivatives. Arzneium-Forsch. Drug Res. 1996, 46, 949–952. [Google Scholar]
- 22. Andreani, A.; Leoni, A.; locatelli, A.; Morigi, R.; Rambaldi, M.; Simon, W.A.; Senn-Bilfinger, J. Synthesis and antisecretory activity of 6-substituted 5- cyanomethylimidazo[2,1-b]thiazoles and 2,6-dimethyl-5- hydroxymethylimidazo[2,1-b][1,3,4]thiadiazole. Arzneium-Forsch. Drug Res. 2000, 50, 550–553. [Google Scholar]
- Salehi, A.H.; Morris, S.J.; Ho, W.C.; Dickson, K.M.; Milutionovic, S.; Durkin, G.; Gillerd, J.W.; Barker, P.A. AEG3482 is an antiapoptotic compound that inhibits Jun kinase activity and cell death through induced expression of heat shock protein 70. Chem. Biol. 2006, 13, 213–223. [Google Scholar] [CrossRef]
- Andreani, A.; Bonazzi, D.; Rambaldi, M.; Fabbri, G.; Rainsford, K.D. 5,6-Disubstituted imidazo[2,1-b]thiazoles as potential anti-inflammatory agents II. Eur. J. Med. Chem. 1982, 17, 271–274. [Google Scholar]
- Andreani, A.; Rambaldi, M.; Mascellani, G.; Bossa, R.; Galatulas, I. Synthesis and cardiotonic activity of 5,6-disubstituted imidazo[2,1-b]thiazoles. Eur. J. Med. Chem. 1986, 21, 451–453. [Google Scholar]
- Andreani, A.; Rambaldi, M.; Mascellani, G.; Rugarli, P. Synthesis and diuretic activity of imidazo[2,1-b]thiazoleacetohydrazones. Eur. J. Med. Chem. 1987, 22, 19–22. [Google Scholar] [CrossRef]
- Andreani, A.; Rambaldi, M.; Locatelli, A.; Andreani, F. 5-Formylimidazo[2,1-b]thiazoles and derivatives with herbicidal activity. Collect. Czech. Chem. Commu. 1991, 56, 2436–2437. [Google Scholar] [CrossRef]
- Carpenter, J.W.; Mee, J.D.; Heseltine, D.W. Direct-positive silver halide immulsions. (Eastman Kodak Co.) Ger. Offen.1,804,465 1969. [Google Scholar]
- Carpenter, J.W.; Mee, J.D.; Heseltine, D.W. Polymethine dyes as electron acceptors and spectral sensitizers for direct-positive silver halide emulsions. U.S. Patent 3,615,639,26, 26 October 1971. [Google Scholar]
- Carpenter, J.W.; Mee, J.D.; Heseltine, D.W. Cyanine dyes with fused imidazolo nuclei. U.S. Patent 3,809,691, 7 May 1974. (Eastman Kodak Co.). [Google Scholar]
- El Khadem, H.; Elshfei, Z.M. The Scope and Mechanism of Carbohydrate Osotriazole Formation. Part II. The Action of Oxidising Agents on Osazones and Osotriazoles. J. Chem. Soc. 1958, 3117–3119. [Google Scholar] [CrossRef]
- Karabasanagouda, T.; Adhikari, A.V.; Shetty, N.S. Synthesis and antimicrobial activities of some novel 1,2,4-triazolo[3,4-b]-1,3,4-thiadiazoles and 1,2,4-triazolo[3,4-b]-1,3,4-thiadiazines carrying thioalkyl and sulphonyl phenoxy moieties. Eur. J. Med. Chem. 2007, 42, 521–529. [Google Scholar] [CrossRef]
- Karegoudar, P.; Prasad, D.J.; Ashok, M.; Mahalinga, M.; Poojary, B.; Holla, B.S. Synthesis, antimicrobial and anti-inflammatory activities of some 1,2,4-triazolo[3,4-b][1,3,4]thiadiazoles and 1,2,4-triazolo[3,4-b][1,3,4]thiadiazines bearing trichlorophenyl moiety. Eur. J. Med. Chem. 2008, 43, 808–815. [Google Scholar] [CrossRef]
- Finegold, S.M.; Martin, W.J. Bailey and Scott’s Diagnostic Microbiology, 6th ed; C.V. Mosby Co.: St. Louis, MO, USA, 1982. [Google Scholar]
- Sample Availability: Samples of the compounds (1-8) are available from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Atta, K.F.M.; Farahat, O.O.M.; Ahmed, A.Z.A.; Marei, M.G. Synthesis and Antibacterial Activities of Novel Imidazo[2,1-b]-1,3,4-thiadiazoles. Molecules 2011, 16, 5496-5506. https://doi.org/10.3390/molecules16075496
Atta KFM, Farahat OOM, Ahmed AZA, Marei MG. Synthesis and Antibacterial Activities of Novel Imidazo[2,1-b]-1,3,4-thiadiazoles. Molecules. 2011; 16(7):5496-5506. https://doi.org/10.3390/molecules16075496
Chicago/Turabian StyleAtta, Kamal F. M., Omaima O.M. Farahat, Alaa Z. A. Ahmed, and Mohamed G. Marei. 2011. "Synthesis and Antibacterial Activities of Novel Imidazo[2,1-b]-1,3,4-thiadiazoles" Molecules 16, no. 7: 5496-5506. https://doi.org/10.3390/molecules16075496
APA StyleAtta, K. F. M., Farahat, O. O. M., Ahmed, A. Z. A., & Marei, M. G. (2011). Synthesis and Antibacterial Activities of Novel Imidazo[2,1-b]-1,3,4-thiadiazoles. Molecules, 16(7), 5496-5506. https://doi.org/10.3390/molecules16075496




