Abstract
Three phenylpropenoyl sucroses – sibiricose A5 (1), A6 (2) and 3′,6-disinapoyl sucrose (3) – were isolated from the 30% EtOH extract of Polygala tenuifolia, which displayed antidepressant-like action. HPLC analysis indicated that the three phenylpropenoyl sucroses could be absorbed into serum. From the serum pharmacochemistry point of view, these three phenylpropenoyl sucroses might prevent or relieve depression.
1. Introduction
Polygala tenuifolia (local name: Yuan zhi) is widely distributed in China, and its root is an important herb used in Traditional Chinese Medicine to treat depression. A number of investigations had been carried out to identify the compounds responsible of the biological activity of Yuan zhi [1,2,3,4]. Some phenylpropenoyl sucroses from Yuan zhi became hot research topics because of their bioactivity of preventing memory disorders [5,6,7,8]. The understanding and diagnosis of many diseases mainly relies on the metabolic profiling of biological fluids such as urine and plasma, as their compositions reflect the biochemical status of a living organism and imply information about the biological processes associated with pathological conditions. The application of high-performance liquid chromatography (HPLC) for untargeted plasma metabolic profiling with direct comparison with authentic samples in metabonomic research has attracted much attention in recent years [9,10]. In order to research the untargeted plasma metabolite profiling of phenylpropenoyl sucroses, we examined the metabolites in rat serum using HPLC by direct comparison with authentic samples after the rats are administered orally with Yuan zhi aqueous extracts. As a result, sibiricose A5 (1), A6 (2) and 3′,6-disinapoyl sucrose (3) were absorbed into serum, as we report here for the first time.
2. Results and Discussion
2.1. Identification of Compounds 1 to 3
The 30% EtOH extract of Yuan zhi exhibited more notable activity in the antidepressive assay than the 50%, 70% and 90% EtOH extracts [11]. The proportion of phenylpropenoyl sucroses in the 30% EtOH fraction of Yuan zhi is 44.88%, which is calculated by their peak area ratio in HPLC-DAD. Thus, we focus on the phenylpropenoyl sucroses-enriched fraction in this study.
Repeated column chromatography (CC) over Sephadex LH-20 and silica gel column led to the isolation of three phenylpropenoyl sucroses 1-3 (Figure 1) from this fraction. These compounds were determined to be sibiricose A5 (1) [12], A6 (2) [12] and 3′,6-disinapoyl sucrose (3) [13], respectively, by comparison of their spectroscopic data with reported literature values and authentic samples. The full NMR assignments of 1, 2 and 3 (Table 1) were achieved by the detailed 2D NMR analysis.
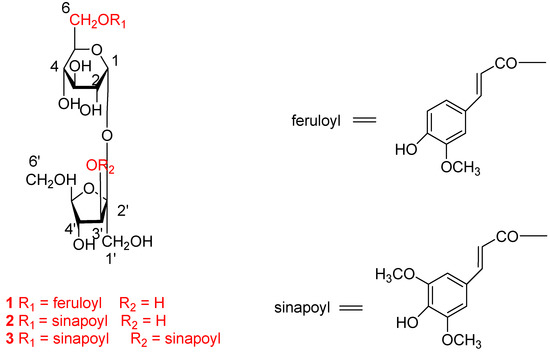
Figure 1.
Structures of compounds 1-3.
Reference samples of sibiricose A5 (1), A6 (2) and 3′,6-disinapoyl sucrose (3) were prepared by repeated CC on Sephadex LH-20, ODS-A, silica gel and RP-HPLC of the three crude samples.

Table 1.
1H-NMR (500 and 400 MHZ) and 13C-NMR (125 and 100 MHz) data of compounds 1-3 in CDCl3 (δ in ppm, J in Hz).
| Position | 1 | 2 | 3 | |||
|---|---|---|---|---|---|---|
| δC | δH | δC | δH | δC | δH | |
| Glycone moiety | ||||||
| Glc-1 | 93.3 | 5.36 (d, 4) | 93.3 | 5.37 (d, 4) | 92.9 | 5.50 (d, 3.7) |
| 2 | 73.1 | 3.53 (d, 10.0, 4.0) | 73.1 | 3.51 (d, 10.0, 4.0) | 73.1 | 3.50 (m) |
| 3 | 74.9 | 3.69 (dd, 10.0, 9.0) | 75.0 | 3.75 (dd, 10.0, 9.5) | 75.0 | 3.69 (t, 9.3) |
| 4 | 71.2 | 3.38 (dd, 9.0, 9.0) | 71.2 | 3.34 (dd, 9.5, 9.0) | 71.9 | 3.38 (m) |
| 5 | 73.8 | 3.88 (m) | 74.5 | 3.89 (m) | 72.8 | 4.29 (m) |
| 6 | 62.3 | 3.77 (dd, 12.0, 4.5)3.81 (m) | 62.4 | 4.31 (dd, 12.0, 4.5)4.52 (m) | 65.6 | 4.48 (br d, 10.0)4.06 (m) |
| Fru-1 | 65.4 | 3.60 (d, 12.0) | 65.4 | 3.62 (d, 12.0) | 65.4 | 3.60 (d, 12.0) |
| 3.69 (d, 12.0) | 3.65 (d, 12.0) | 3.63 (d, 12.0) | ||||
| 2 | 104.4 | 104.8 | 104.8 | |||
| 3 | 79.7 | 5.40 (d, 8.0) | 79.6 | 5.46 (d, 8.0) | 79.6 | 5.58 (d, 7.5) |
| 4 | 74.8 | 4.33 (dd, 8.0, 7.5) | 74.5 | 4.07 (dd, 8.0, 7.5) | 74.4 | 4.51 (t, 8.0) |
| 5 | 84.1 | 3.88 (m) | 84.2 | 3.88 (m) | 84.2 | 4.10 (m) |
| 6 | 62.9 | 3.84 (m) | 62.9 | 3.89 (m) | 63.7 | 3.99 (m) |
| 3.84 (m) | 3.80 (m) | 4.20 (m) | ||||
| Aglycone moiety | ||||||
| R1-1 | 127.5 | 129.5 | 125.1 | |||
| 2 | 112.1 | 6.74 (d, 2.0) | 107.1 | 6.90 (s) | 106.8 | 7.12 (s) |
| 3 | 149.6 | 149.5 | 149.0 | |||
| 4 | 149.0 | 130.7 | 140.5 | |||
| 5 | 116.5 | 6.68 (d, 8.0) | 149.5 | 149.0 | ||
| 6 | 124.3 | 6.59 (dd, 8.0, 2.0) | 107.1 | 6.90 (s) | 106.8 | 7.12 (s) |
| 7 | 147.7 | 7.71 (d, 15.6) | 147.2 | 7.64 (d, 15.6) | 146.0 | 8.09 (d, 16.0) |
| 8 | 115.1 | 6.36 (d, 15.6) | 115.8 | 6.38 (d, 15.6) | 115.2 | 6.90 (d, 16.0) |
| 9 | 168.3 | 169.1 | 166.8 | |||
| -OCH3 | 56.6 | 3.86 (s) | 56.8 | 3.87 (s) | 56.4 | 3.80 (s) |
| R2-1 | 125.2 | |||||
| 2 | 106.9 | 7.12 (s) | ||||
| 3 | 149.2 | |||||
| 4 | 140.5 | |||||
| 5 | 149.2 | |||||
| 6 | 107.9 | 7.12 (s) | ||||
| 7 | 146.7 | 7.98 (d, 15.6) | ||||
| 8 | 115.6 | 6.67 (d, 15.6) | ||||
| 9 | 167.7 | |||||
| -OCH3 | 56.9 | 3.80 (s) | ||||
2.2. Untargeted Plasma Metabolite Profiling of Yuan zhi
The aqueous extract of P. tenuifolia (2.5 g crude drug/mL) was administered orally to male Sprague-Dawley (SD) rats at a dose of 1.56 g/kg. The orbital blood samples (3 mL) of the rats were collected at 60 min post-dose. The serum containing the extracts of P. tenuifolia and blank control serum were analyzed by HPLC at 330 nm after a series of preconditioning (Figure 2). As shown in the Figure, HPLC analysis indicated that three phenylpropenoyl sucroses sibiricose A5 (1), A6 (2) and 3′,6-disinapoyl sucrose (3) of Yuanzhi, can be absorbed into serum. Our feasibility study showed the proportions of sibiricose A5 (1), A6 (2) and 3′,6-disinapoyl sucrose (3) in the fraction were 11.06%, 8.89%, 24.29%, respectively, by calculating their peak area ratio in HPLC-DAD. It was also reported that the fraction containing these three oligosaccharide polyesters as main chemical constituents had obvious antidepressant activity [11].
In addition, it is reported that 3′,6-disinapoyl sucrose (3) can protect the SH-SY5Y human neuroblastoma cells from lesion, and sibiricose A5 (1) can protect the PC12 cells from the damage induced by glutamic acid [11,14]. Therefore, from the point of view of serum pharmacochemistry, our results indicate that the three phenylpropenoyl sucroses might prevent or relieve depression.
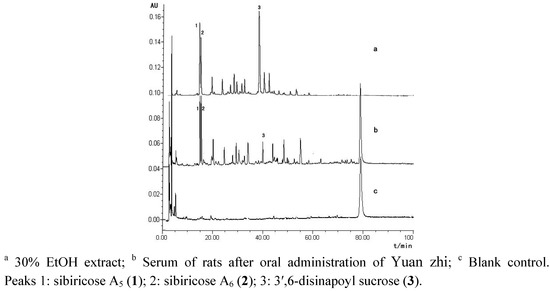
Figure 2.
Plasma metabolite HPLC profiles of Yuan zhi extract.
3. Experimental
3.1. General
Column chromatography (CC) was performed on Sephadex LH-20 (Pharmacia Fine Chemical Co., Ltd.), SP825 (Mitsuishi Chemical Co.), Chromatorex ODS (Fuji Silysia Chemical Co., Ltd.) and silica gel (Qingdao Haiyang Chemical Co.). TLC was carried on silica gel G precoated plates (Qingdao Haiyang Chemical Co.) with CHCl3-MeOH-H2O (9:1:0.1 or 7:3:0.5). The spots were detected by spraying with 10% H2SO4 ethanol solution followed by heating. 1H- and 13C-NMR, HSQC and HMBC spectra were recorded in DMSO-d6 with Bruker AM-400 and DRX-500 spectrometers. The HPLC was performed with a Waters 2695 HPLC system on a Agela Venusil MP-C18 column (5 μm, 250 × 4.6 mm, Agela, China) and with column temperature set at 30 °C, using a diode-array detection (Shimadzu SPD-M10A).
3.2. Plant Material
The roots of P. tenuifolia were collected from Shanxi province, China, and identified by Yuting Chen (Beijing University of Chinese Medicine).
3.3. Extraction and Isolation
Dried plant material (300 g) of P. tenuifolia was refluxed with ethanol for 3 h (4 times). After removal of the organic solvent under reduced pressure, the aqueous fraction was concentrated to a small volume (100 mL) and subjected to a SP825 macroporous adsorptive resin column eluting with H2O and 30% EtOH to afford two fractions. The concentrated 30% EtOH was applied to CC on silica gel (CHCl3-MeOH-H2O, 9:1:0.1-7:3:0.5), Sephadex LH-20 and ODS-A, eluting with H2O-MeOH (1:0-0:1) to afford compounds 1 (14 mg), 2 (6 mg) and 3 (15 mg), respectively.
3.4. HPLC Analysis
The plasma sample (0.5 mL) was placed in a 5 mL polypropylene tube, and monopotassium phosphate (0.2 mL) and acetonitrile (1.5 mL) were added to the tube. The tube was vortexed-mixed for 90 s. The precipitated protein was removed by centrifugation at 4,000 rpm for 30 min. The organic layer was transferred to another tube, and evaporated to dryness by N2 at 50 °C. The residue was added 0.2 mL distilled water to dissolve, and then filtered through a 0.45 μm Millipore filter film. Aliquots of 20 μL were injected into the HPLC system. The mobile phase consisted of A, CH3CN; B, H2O (0.1% phosphoric acid); gradient: 11-16% A linear in 10 min; 16-22% A linear in 10 min; 22-23% A linear in 5 min; 23-28% A linear in 9 min; 28% A linear in 11 min; 28-33% A linear in 10 min; 33-39% A linear in 15 min; 39-42% A linear in 15 min; 42-55% A linear in 10 min; 55-70% A linear in 5 min; isocratic on 70-11% A for 10 min. The mobile phase was degassed automatically using the electronic degasser system. The flow rate was 1.0 mL/min. The optimum wavelength was set at 330 nm [15].
3.5. Animal Experiments
Male SD rats (250 ± 20 g body weight) were provided by Vital River Experimental Animal Co., Ltd. (license number: SCXK2006-0009). Rats were kept in an environmentally controlled breeding room for three days before starting the experiment and were fed with standard laboratory food and water. SD rats had free access to water but no food for 12 h before the experiments. The extract of Yuan zhi was administered orally to rats at a dose of 1.56 g/kg, and the blood samples (3 mL) were collected at 60 min post-dose. The plasma samples were immediately separated by centrifugation at 4,000 rpm for 10 min.
Acknowledgements
This work was supported by Important National Science & Technology Specific Projects during the 11th Five-Year Plan Period (NO. 2009ZX09502-006).
References
- Kako, M.; Miura, T.; Nishiyama, Y.; Ichimaru, M.; Moriyasu, M.; Atsushi, K. Hypoglycemic activity of some triterpenoid glycosides. J. Nat. Prod. 1997, 60, 604–605. [Google Scholar] [CrossRef]
- Nagai, T.; Suzuki, Y.; Kiyohara, H.; Susa, E.; Kato, T.; Nagamine, T.; Hagiwara, Y.; Tamura, S.; Yabe, T.; Aizawa, C.; et al. Onjisaponins, from the root of Polygala tenuifolia Willdenow, as effective adjuvants for nasal influenza and diphtheria-pertussis-tetanus vaccines. Vaccine 2001, 19, 4824–4834. [Google Scholar] [CrossRef]
- Yoshikawa, M.; Murakami, T.; Ueno, T.; Kadoya, M.; Matsuda, H.; Yamahara, J.; Murakami, N. Bioactive saponins and glycosides. 1. Senegae radix. (1): E-senegasaponins a and b and Z-senegasaponins a and b, their inhibitory effect on alcohol absorption and hypoglycemic activity. Chem. Pharm. Bull. 1995, 43, 350–352. [Google Scholar] [CrossRef]
- Campos, R.O.P.; Santos, A.R.S.; Vaz, Z.R.; Pinheiro, T.R.; Pizzolatti, M.G.; Filho, V.C.; Monache, F.D.; Yunes, R.A.; Calixto, J.B. Antinociceptive properties of the hydroalcoholic extract and prelininary study of a xanthone isolated from Polygala cyparissias (Polygalaceae). Life Sci. 1997, 61, 1619–1630. [Google Scholar] [CrossRef]
- Yukinobu, I.; Shigefumi, T.; Mitsuo, T.; Humito, K.; Kouin, T.; Takuji, Y.; Masaki, A. Cognitive improving and cerebral protective effects of acylated oligosaccharides in Polygala tenuifolia. Biol. Pharm. Bull. 2004, 27, 1081–1085. [Google Scholar] [CrossRef]
- Miyase, T.; Iwata, Y.; Ueno, A. Tenuifolioses A-F, oligosaccharide multi-esters from the roots of Polygala Tenuifolia Willd. Chem. Pharm. Bull. 1991, 39, 3082–3084. [Google Scholar] [CrossRef]
- Kobayashi, S.; Miyase, T.; Noguchi, H. Polyphenolia glycosides and oligosaccharide multiesters from the roots of Polygala dalmaisiana. J. Nat. Prod. 2002, 65, 319–328. [Google Scholar] [CrossRef]
- Chang, H.T.; Tu, P.F. New oligosaccharide ester and xanthone C-glucosides from Polygala Telephioides. Helv. Chim. Acta 2007, 90, 944–950. [Google Scholar] [CrossRef]
- Zuo, F.; Zhou, Z.M.; Yan, M.Z.; Liu, M.L.; Xiong, Y.L.; Zhang, Q.; Song, H.Y.; Ye, W.H. Metabolism of constituents in Huangqin-Tang, a prescription in traditional Chinese medicine, by human intestinal flora. Biol. Pharm. Bull. 2002, 25, 558–563. [Google Scholar] [CrossRef]
- Song, R.; Xu, L.; Xu, F.G.; Li, Z.; Dong, H.J.; Tian, Y.; Zhang, Z.J. In vivo metabolism study of rhubarb decoction in rat using high-performance liquid chromatography with UV photodiode-array and mass-spectrometric detection: A strategy for systematic analysis of metabolites from traditional Chinese medicines in biological samples. J. Chromatogr. A 2010, 1217, 7144–7152. [Google Scholar]
- Tu, H.H.; Liu, P.; Mu, L.; Liao, H.B.; Xie, T.T.; Ma, L.H.; Liu, Y.M. Study on antidepressant components of sucrose ester from Polygala tenuifolia. Zhongguo Zhong Yao Za Zhi 2008, 33, 1278–1280. [Google Scholar]
- Miyase, T.; Ioguchi, H.; Chen, X.M. Sucrose ester and xanth one C-glycosides from the roots of Polygala sibirica. J. Nat. Prod. 1999, 62, 993–996. [Google Scholar] [CrossRef]
- Miyase, T.; Ueno, A. Sucrose derivatives from the roots of Polygala tenuifolia. Nat. Med. (Tokyo) 1993, 47, 267–278. [Google Scholar]
- Liu, P.; Hu, Y.; Guo, D.H.; Wang, D.X.; Tu, H.H.; Ma, L.; Xie, T.T.; Kong, L.Y. Potential antidepressant properties of radix polygalae (Yuanzhi). Phytomedicine 2010, 17, 794–799. [Google Scholar] [CrossRef]
- Yong, J.; Na, Z.; Zheng, C.; Peng, F.T. Fingerprint of polygala tenuifolia by high performance liquid chromatography. Yao Xue Xue Bao 2006, 41, 179–183. [Google Scholar]
- Sample Availability: Samples of the compounds are available from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).