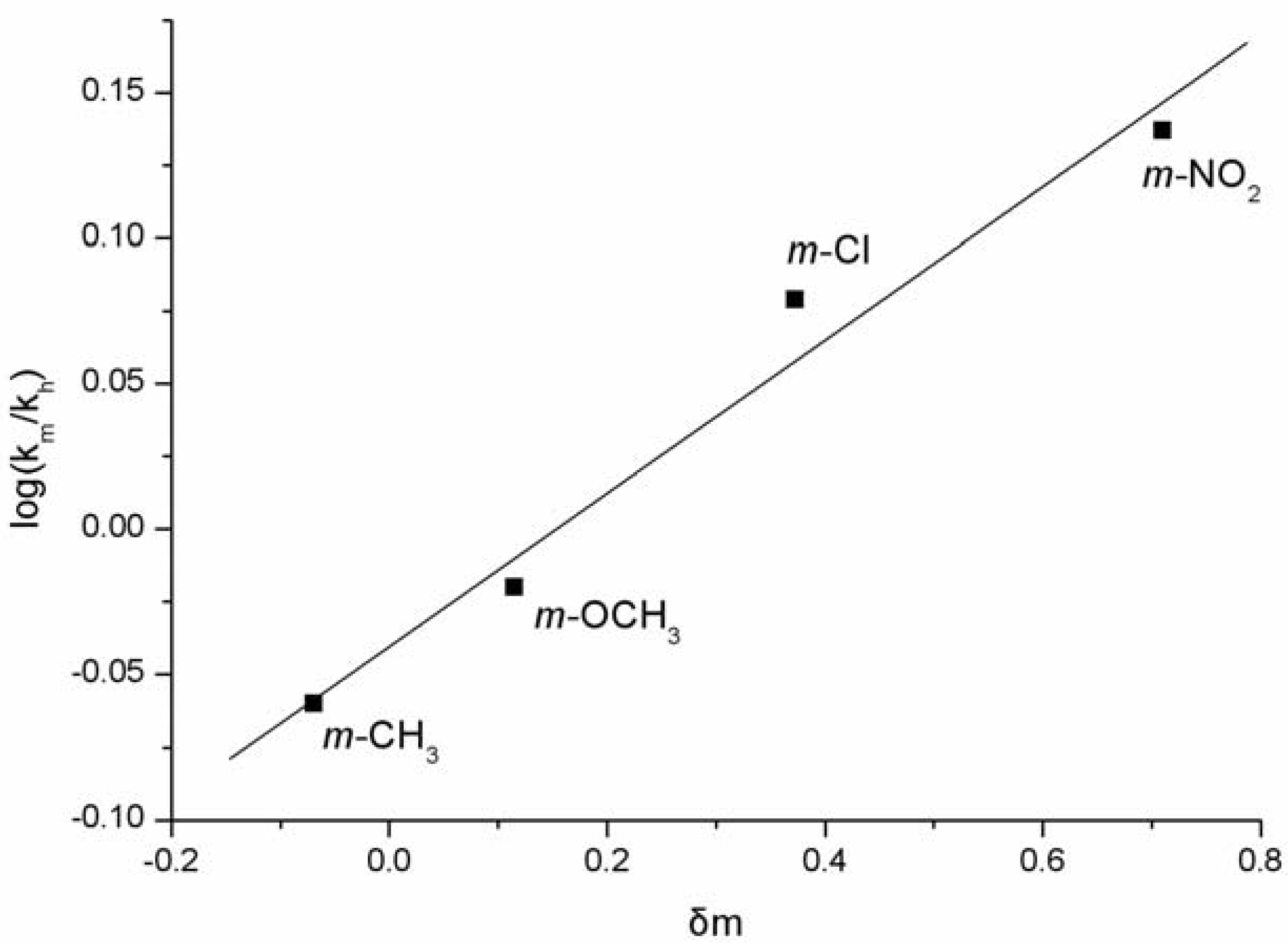
3.2. General Procedure for the Preparation of Bis(indolyl)Methanes 2-4
To a solution of aryl benzaldehyde (0.5 mmol) and RuCl3·3H2O (0.05 mmol) in benzene (1 mL) was added indole (1.0 mmol) under air atmosphere and the mixture was stirred at room temperature (monitored by TLC). Then, the reaction mixture was concentrated under reduced pressure. The residue was purified by flash chromatography on silica gel (eluent:EtOAc/PE = 1:4) to yield the corresponding product.
3,3’-Bis-indolyl phenylmethane (
2a). Pink solid, mp: 126–127 °C (EtOAc/PE = 1:4) (lit [
29], 125–127 °C). IR (KBr) ν
max: 3417, 3065, 1513, 1454 cm
−1.
1H-NMR (CDCl
3):
δ 7.82 (s, 2H, N–H), 7.43 (d,
J = 7.9 Hz, 2H, Ar–H), 7.40–7.36 (m, 3H, Ar–H), 7.35–7.29 (m, 3H, Ar–H), 7.28–7.24 (m, 1H, Ar–H), 7.21 (dt,
J = 0.8, 7.9 Hz, 2H, Ar–H), 7.05 (dt,
J = 0.8, 7.9 Hz, 2H, Ar–H), 6.61 (d,
J = 1.5 Hz, 2H, Ar–H), 5.92 (s, 1H).
13C-NMR (CDCl
3):
δ 144.0, 136.7, 128.8, 128.3, 127.1, 126.2, 123.7, 121.9, 120.0, 119.7, 119.2, 111.1, 40.2. MS (EI, 70 eV):
m/z = 322 (M
+, 20), 245 (75), 206 (100), 77 (10).
3,3’-Bis-indolyl-(3-methylphenyl)methane (2b). Pink solid, mp: 98–99 °C (EtOAc/PE = 1:4). IR (KBr) νmax: 3406, 3049, 1610, 1458, 1419, 745 cm−1. 1H-NMR (CDCl3): δ 7.88 (s, 2H, N–H), 7.42 (d, J = 7.9 Hz, 2H, Ar–H), 7.36 (d, J = 8.2 Hz, 2H, Ar–H), 7.20–7.14 (m, 5H, Ar–H), 7.05–6.99 (m, 3H, Ar–H), 6.66 (d, J = 1.6 Hz, 2H, Ar–H), 5.85 (s, 1H), 2.30 (s, 3H, CH3). 13C-NMR (CDCl3): δ 143.9, 137.6, 136.7, 129.5, 128.1, 127.1, 126.9, 125.8, 123.6, 121.9, 120.0, 119.8, 119.2, 111.0, 40.1, 21.5. MS (EI, 70 eV): m/z = 336 (M+, 30), 245 (100), 221 (30). Anal. calcd. for C24H20N2: C, 85.68; H, 5.99; N, 8.33. Found C, 85.30; H, 5.87; N, 8.05.
3,3’-Bis-indolyl-(4-methylphenyl)methane (
2c). Pink solid, mp: 93–95 °C (EtOAc/PE = 1:4) (lit [
30], 94–96 °C). IR (KBr) ν
max: 3410, 3046, 1457, 743 cm
−1.
1H-NMR (CDCl
3):
δ 7.89 (s, 2H, N–H), 7.42 (d,
J = 7.9 Hz, 2H, Ar–H), 7.36 (d,
J = 8.2 Hz, 2H, Ar–H), 7.25 (d,
J = 8.0 Hz, 2H, Ar–H), 7.18 (dt,
J = 1.0, 8.2 Hz, 2H, Ar–H), 7.10 (d,
J = 7.9 Hz, 2H, Ar–H), 7.02 (dt,
J = 1.0, 8.0 Hz, 2H, Ar–H), 6.66 (dd,
J = 2.2, 0.7 Hz, 2H, Ar-H), 5.87 (s, 1H), 2.34 (s, 3H, CH
3).
13C-NMR (CDCl
3):
δ 141.0, 136.7, 135.5, 128.9, 128.6, 127.1, 123.5, 121.9, 120.0, 119.9, 119.2, 111.0, 39.8, 21.1. MS (EI, 70 eV):
m/z = 336 (M
+, 35), 245 (100), 220 (35), 116 (10).
3,3’-Bis-indolyl-(3-methoxyphenyl)methane (2d). Pink solid, mp: 188.5–189.5 °C (EtOAc/PE = 1:4). IR (KBr) νmax: 3410, 3046, 2923, 1487, 1441, 1263, 1152, 1049, 745 cm−1. 1H-NMR (CDCl3): δ 7.91 (s, 2H, N–H), 7.42 (d, J = 7.8 Hz, 2H, Ar–H), 7.36 (d, J = 8.2 Hz, 2H, Ar–H), 7.22 (d, J = 7.8 Hz, 1H, Ar–H), 7.18 (dt, J = 0.9, 8.2 Hz, 2H, Ar–H), 7.02 (dt, J = 0.9, 7.8 Hz, 2H, Ar–H), 6.97 (d, J = 7.7 Hz, 1H, Ar–H), 6.93 (t, J = 2.2 Hz, 1H, Ar–H), 6.77 (dq, J = 0.6, 7.7 Hz, 1H, Ar–H), 6.68 (dd, J = 2.2, 0.6 Hz, 2H, Ar–H), 5.87 (s, 1H), 2.34 (s, 3H, CH3), 3.75 (s, 3H, OCH3). 13C-NMR (CDCl3): δ 159.6, 145.7, 136.7, 129.1, 127.1, 123.6, 121.9, 121.3, 119.9, 119.6, 119.2, 114.7, 111.3, 111.0, 55.1, 40.2. MS (EI, 70 eV): m/z = 352 (M+, 35), 337 (35), 321 (8), 245 (100), 130 (40). Anal. calcd for C24H20N2O: C, 81.79; H, 5.72; N, 7.95. Found C, 81.52; H, 5.33; N, 7.66.
3,3’-Bis-indolyl-(3-chlorophenyl)methane (2e). Pink solid, mp: 64–68 °C (EtOAc/PE = 1:4). IR (KBr) νmax: 3412, 3046, 1458, 1418, 1094, 744 cm−1. 1H-NMR (CDCl3): δ 7.88 (s, 2H, N–H), 7.42–7.34 (m, 5H, Ar–H), 7.26–7.17 (m, 5H, Ar–H), 7.04 (dt, J = 0.8, 7.8 Hz, 2H, Ar–H), 6.62 (s, 2H, Ar–H), 5.86 (s, 1H). 13C-NMR (CDCl3): δ 146.2, 136.7, 134.0, 129.5, 128.8, 126.9, 126.8, 126.4, 123.6, 122.1, 119.7, 119.4, 119.0, 111.1, 40.0. MS (EI, 70 eV): m/z = 283 (25), 281 (100), 245 (80). Anal. calcd. for C23H17N2Cl: C, 77.41; H, 4.80; N, 7.85. Found C, 77.51; H, 4.67; N, 7.48.
3,3’-Bis-indolyl-(2-bromophenyl)methane (2f). Pink solid, mp: 89–91 °C (EtOAc/PE = 1:4). IR (KBr) νmax: 3411, 3043, 1443, 1022, 744 cm−1. 1H-NMR (CDCl3): δ 7.90 (s, 2H, N–H), 7.64 (d, J = 7.9 Hz, 1H, Ar–H), 7.42 (d, J = 7.9 Hz, 2H, Ar–H), 7.37 (d, J = 8.2 Hz, 2H, Ar–H), 7.25–7.14 (m, 4H, Ar–H), 7.10 (dt, J = 1.9, 7.9 Hz, 1H, Ar–H), 7.04 (dt, J = 0.9, 8.0 Hz, 2H, Ar–H), 6.62 (dd, J = 2.3, 0.9 Hz, 2H, Ar–H), 6.33 (s, 1H). 13C-NMR (CDCl3): δ 143.0, 136.7, 132.9, 130.5, 127.8, 127.3, 127.0, 124.8, 123.8, 122.0, 119.9, 119.3, 118.5, 111.1, 39.6. MS (EI, 70 eV): m/z = 351 (100), 349 (100), 245 (80). Anal. calcd. for C23H17N2Br: C, 68.84; H, 4.27; N, 6.98. Found C, 68.64; H, 4.05; N, 6.81.
3,3’-Bis-indolyl-(3-nitrophenyl)methane (
2g). Pink solid, mp: 262–264 °C (EtOAc/PE = 1:4) (lit [
29], 265–266 °C). IR (KBr) ν
max: 3410, 3053, 2924, 1524, 1455, 1346, 1092, 741 cm
−1.
1H-NMR (CDCl
3):
δ 8.23 (t,
J = 2.0 Hz, 1H, Ar–H), 8.10 (dq,
J = 1.0, 8.2 Hz, 1H, Ar–H), 8.01 (s, 2H, N–H), 7.71 (d,
J = 7.9 Hz, 1H, Ar–H), 7.46 (t,
J = 7.9 Hz, 1H, Ar–H), 7.39 (d,
J = 8.2 Hz, 2H, Ar–H), 7.37 (d,
J = 7.9 Hz, 2H, Ar–H), 7.22 (dt,
J = 0.9, 7.2 Hz, 2H, Ar–H), 7.04 (dt,
J = 0.9, 7.2 Hz, 2H, Ar–H), 6.68 (dd,
J = 2.0, 0.9 Hz, 2H, Ar–H), 6.01 (s, 1H).
13C-NMR (CDCl
3):
δ 148.5, 146.4, 136.7, 134.9, 129.2, 126.6, 123.7, 123.6, 122.3, 121.5, 119.6, 119.5, 118.3, 111.3, 40.0. MS (EI, 70 eV):
m/z = 367 (100), 321 (10), 245 (85), 122 (20). Anal calcd. for C
23H
17N
3O
2: C, 75.19; H, 4.66; N, 11.44. Found C, 75.07; H, 4.36; N, 11.14.
3,3’-Bis-(N-methylindolyl)phenylmethane (3a). Pink solid, mp: 185–187 °C (EtOAc/PE = 1:4). IR (KBr) νmax: 3046, 2930, 1607, 1474, 1329, 1125, 743 cm−1. 1H-NMR (CDCl3): δ 7.43 (d, J = 7.9 Hz, 2H, Ar–H), 7.39 (d, J = 8.6 Hz, 2H, Ar–H), 7.35–7.29 (m, 4H, Ar–H), 7.26-7.21 (m, 3H, Ar–H), 7.03 (dt, J = 0.8, 7.9 Hz, 2H, Ar–H), 6.57 (s, 2H, Ar–H), 5.93 (s, 1H), 3.71 (s, 6H, 2 × CH3). 13C-NMR (CDCl3): δ 144.5, 137.4, 128.7, 128.3, 128.2, 127.5, 126.0, 121.4, 120.1, 118.7, 118.3, 109.1, 40.1, 32.7. MS (EI, 70 eV): m/z = 350 (M+, 100), 273 (85), 220 (25), 130 (15). Anal. calcd. for C25H22N2: C, 85.68; H, 6.33; N, 7.99. Found C, 85.90; H, 6.58; N, 7.64.
3,3’-Bis-(N-methylindolyl)-(3-methylphenyl)methane (3b). Pink waxy solid. IR (KBr) νmax: 3049, 2932, 1475, 1123, 737 cm−1. 1H-NMR (CDCl3): δ 7.42 (d, J = 7.9 Hz, 2H, Ar–H), 7.31 (d, J = 8.2 Hz, 2H, Ar–H), 7.25–7.14 (m, 5H, Ar–H), 7.07–6.99 (m, 3H, Ar–H), 6.56 (s, 2H, Ar–H), 5.87 (s, 1H), 3.70 (s, 6H, 2 × CH3), 2.32 (s, 3H, CH3). 13C-NMR (CDCl3): δ 144.4, 137.6, 137.4, 129.4, 128.2, 128.0, 127.5, 126.8, 125.7, 121.4, 120.1, 118.6, 118.4, 109.0, 40.0, 32.7, 21.6. MS (EI, 70 eV): m/z = 364 (M+, 95), 349 (85), 273 (100), 257 (25), 130 (20). Anal. calcd. for C26H24N2: C, 85.68; H, 6.64; N, 7.69. Found C, 85.30; H, 6.60; N, 7.36.
3,3’-Bis-(N-methylindolyl)-(4-methylphenyl)methane (3c). Pink solid, mp: 146–148 °C (EtOAc/PE = 1:4). IR (KBr) νmax: 3050, 2928, 1470, 1125, 745 cm−1. 1H-NMR (CDCl3): δ 7.40 (d, J = 7.9 Hz, 2H, Ar–H), 7.30 (d, J = 8.2 Hz, 2H, Ar–H), 7.26–7.17 (m, 4H, Ar–H), 7.09 (d, J = 7.9 Hz, 2H, Ar–H), 7.00 (dt, J = 1.0, 7.9 Hz, 2H, Ar–H), 6.54 (s, 2H, Ar–H), 5.85 (s, 1H), 3.69 (s, 6H, 2 × CH3), 2.32 (s, 3H, CH3). 13C-NMR (CDCl3): δ 141.4, 137.4, 135.4, 128.9, 128.5, 128.2, 127.5, 121.4, 120.1, 118.6, 118.5, 109.0, 39.6, 32.6, 21.1. MS (EI, 70 eV): m/z = 364 (M+, 15), 273 (100), 257 (60), 130 (60). Anal. calcd. for C26H24N2: C, 85.68; H, 6.64; N, 7.69. Found C, 85.44; H, 6.96; N, 7.32.
3,3’-Bis-(N-methylindolyl)-(3-methoxyphenyl)methane (3d). Pink solid, mp: 149–151 °C (EtOAc/PE = 1:4). IR (KBr) νmax: 3054, 2930, 1478, 1256, 1135, 740 cm−1. 1H-NMR (CDCl3): δ 7.42 (d, J = 7.9 Hz, 2H, Ar–H), 7.30 (d, J = 8.2 Hz, 2H, Ar–H), 7.22 (t, J = 7.9 Hz, 3H, Ar–H), 7.02 (dt, J = 0.9, 7.9 Hz, 2H, Ar–H), 6.97 (d, J = 7.9 Hz, 1H, Ar–H), 6.94 (t, J = 2.0 Hz, 1H, Ar–H), 6.78 (dd, J = 8.2, 2.0 Hz, 1H, Ar–H), 6.57 (s, 2H, Ar–H), 5.87 (s, 1H), 3.76 (s, 3H, OCH3), 3.70 (s, 6H, 2 × CH3). 13C-NMR (CDCl3): δ 159.6, 146.2, 137.4, 129.1, 128.2, 127.5, 121.4, 121.3, 120.0, 118.6, 118.1, 114.8, 111.1, 109.0, 55.1, 40.1, 32.7. MS (EI, 70 eV): m/z = 380 (M+, 65), 365 (85), 349 (30), 273 (100), 130 (25). Anal. calcd. for C26H24N2O: C, 82.07; H, 6.36; N, 7.36. Found C, 81.72; H, 5.96; N, 6.99.
3,3’-Bis-(N-methylindolyl)-(3-chlorophenyl)methane (3e). Pink solid, mp: 195–197 °C (EtOAc/PE = 1:4). IR (KBr) νmax: 3051, 2930, 1458, 1420, 1094, 743 cm−1. 1H-NMR (CDCl3): δ 7.45 (t, J = 7.9 Hz, 2H, Ar–H), 7.42 (s, 1H), 7.37 (d, J = 8.2 Hz, 2H, Ar–H), 7.36-7.25 (m, 5H, Ar–H), 7.10 (dt, J = 0.7, 7.9 Hz, 2H, Ar–H), 6.61 (s, 2H, Ar–H), 5.94 (s, 1H), 3.73 (s, 6H, 2 × CH3). 13C-NMR (CDCl3): δ 146.8, 137.5, 134.1, 129.5, 128.8, 128.3, 127.3, 127.0, 126.4, 121.6, 119.9, 118.9, 117.5, 109.2, 39.9, 32.7. MS (EI, 70 eV): m/z = 386 (M+, 20), 384 (M+, 60), 371 (5), 369 (15), 273 (100). Anal. calcd. for C25H21N2Cl: C, 78.01; H, 5.50; N, 7.28. Found C, 77.80; H, 5.50; N, 7.16.
3,3’-Bis-(N-methylindolyl)-(2-bromophenyl)methane (3f). Pink solid, mp: 247–249 °C (EtOAc/PE = 1:4). IR (KBr) νmax: 3046, 2926, 1457, 1227, 1023, 792 cm−1. 1H-NMR (CDCl3): δ 7.63 (dd, J = 7.9, 1.2 Hz, 1H, Ar–H), 7.42 (d, J = 7.9 Hz, 2H, Ar–H), 7.31 (d, J = 8.2 Hz, 2H, Ar–H), 7.28–7.20 (m, 3H, Ar–H), 7.17 (dt, J = 1.2, 7.6 Hz, 1H, Ar–H), 7.09 (dt, J = 1.8, 7.6 Hz, 1H, Ar–H), 7.03 (dt, J = 0.9, 7.9 Hz, 2H, Ar–H), 6.51 (s, 2H, Ar–H), 6.33 (s, 1H), 3.70 (s, 6H, 2 × CH3). 13C-NMR (CDCl3): δ 143.4, 137.5, 132.8, 130.5, 128.5, 127.7, 127.4, 127.2, 124.8, 121.5, 120.0, 118.7, 117.0, 109.1, 39.4, 32.7. MS (EI, 70 eV): m/z = 430 (M+, 20), 428 (M+, 20), 350 (55), 273 (100), 130 (30). Anal. calcd. for C25H21N2Br: C, 69.94; H, 4.93; N, 6.52. Found C, 69.82; H, 4.55; N, 6.54.
3,3’-Bis-(N-methylindolyl)-(3-nitrophenyl)methane (3g). Yellow solid, mp: 157–159 °C (EtOAc/PE = 1:4). IR (KBr) νmax: 3063, 2926, 1525, 1474, 1349, 743 cm−1. 1H-NMR (CDCl3): δ 8.27 (t, J = 1.9 Hz, 1H, Ar–H), 8.12 (dd, J = 8.2, 1.9 Hz, 1H, Ar–H), 7.74 (d, J = 7.9 Hz, 1H, Ar–H), 7.47 (t, J = 7.9 Hz, 1H, Ar–H), 7.41 (d, J = 7.9 Hz, 2H, Ar–H), 7.38 (d, J = 8.2 Hz, 2H, Ar–H), 7.28 (dt, J = 0.9, 7.9 Hz, 2H, Ar–H), 7.18 (dt, J = 0.9, 7.9 Hz, 2H, Ar–H), 6.61 (s, 2H, Ar–H), 6.05 (s, 1H), 3.75 (s, 6H, 2 × CH3). 13C-NMR (CDCl3): δ 148.5, 146.9, 137.5, 134.9, 129.1, 128.4, 127.1, 123.6, 121.9, 121.4, 119.7, 119.0, 116.8, 109.4, 40.0, 32.8. MS (EI, 70 eV): m/z = 395 (M+, 80), 380 (5), 349 (5), 273 (100), 122 (5). Anal. calcd. for C25H21N3O2: C, 75.93; H, 5.35; N, 10.63. Found C, 75.76; H, 4.98; N, 10.54.
3,3’-Bis-(2-methylindolyl)-(3-methylphenyl)methane (4a). Pink solid, mp: 181–184 °C (EtOAc/PE = 1:4). IR (KBr) νmax: 3383, 2915, 1677, 1607, 1459, 740 cm−1. 1H-NMR (DMSO-d6): δ 10.71 (s, 2H, N–H), 7.19 (d, J = 8.0 Hz, 2H, Ar–H), 7.11 (t, J = 8.0 Hz, 1H, Ar–H), 7.03–6.96 (m, 3H, Ar–H), 6.87 (dt, J = 0.9, 8.0 Hz, 2H, Ar–H), 6.81 (d, J = 8.0 Hz, 2H, Ar–H), 6.66 (dt, J = 0.9, 8.0 Hz, 2H, Ar–H), 5.87 (s, 1H), 2.19 (s, 3H, CH3), 2.04 (s, 6H, 2 × CH3). 13C-NMR (DMSO-d6): δ 144.65, 137.20, 135.48, 132.44, 129.81, 128.76, 128.20, 126.88, 126.24, 119.93, 118.92, 118.34, 112.69, 110.73, 39.00, 21.64, 12.38. MS (EI, 70 eV): m/z = 364 (M+, 15), 349 (100), 234 (40), 130 (70). Anal. calcd. for C26H24N2: C, 85.68; H, 6.64; N, 7.69. Found C, 85.50; H, 6.91; N, 7.45.
3,3’-Bis-(2-methylindolyl)-(3-methoxylphenyl)methane (4b). Pink solid, mp: 147–150 °C (EtOAc/PE = 1:4). IR (KBr) νmax: 3385, 1594, 1459, 1147, 744 cm−1. 1H-NMR (DMSO-d6): δ 10.73 (s, 2H, N–H), 7.21 (d, J = 8.0 Hz, 2H, Ar–H), 7.16 (t, J = 8.0 Hz, 1H, Ar–H), 6.88 (dt, J = 0.9, 8.0 Hz, 2H, Ar–H), 6.83 (d, J = 8.0 Hz, 2H, Ar–H), 6.79–6.72 (m, 3H, Ar–H), 6.67 (dt, J = 0.9, 8.0 Hz, 2H, Ar–H), 5.88 (s, 1H), 3.62 (s, 3H, OCH3), 2.07 (s, 6H, 2 × CH3). 13C-NMR (DMSO-d6): δ 159.55, 146.39, 135.49, 132.47, 129.31, 128.71, 121.71, 119.97, 118.93, 118.36, 115.48, 112.57, 110.98, 110.75, 55.27, 39.05, 12.36. MS (EI, 70 eV): m/z = 380 (M+, 95), 365 (35), 349 (45), 273 (100), 130 (35). Anal. calcd. for C26H24N2O: C, 82.07; H, 6.36; N, 7.36. Found C, 81.85; H, 6.02; N, 7.17.
3.3. General Procedure for the Preparation of Bis(thienyl)methanes 5a-5g
To a solution of aryl benzaldehyde (0.5 mmol) and RuCl3·3H2O (0.05 mmol ) in ethylene glycol dimethyl ether (1 mL) was added 2-methylthiophene (1.0 mmol) under air atmosphere and the mixture was stirred at 80 °C (monitored by TLC). Then, the reaction mitxure was concentrated under reduced pressure. The residue was purified by flash chromatography on silica gel (eluent: EtOAc/PE = 1:8) to yield the corresponding product.
5,5’-Bis-(2-methylthienyl)phenylmethane (5a). Yellow waxy solid. IR (KBr) νmax: 3059, 2919, 1525, 1448, 1225, 794 cm−1. 1H-NMR (MHz, CDCl3): δ 7.33–7.28 (m, 4H), 7.27–7.21 (m, 1H), 6.61–6.55 (m, 4H), 5.67 (s, 1H), 2.41 (s, 6H, 2 × CH3). 13C-NMR (CDCl3): δ 145.3, 143.8, 139.1, 128.4, 128.3, 127.0, 125.7, 124.5, 47.8, 15.4. MS (EI, 70 eV): m/z = 284 (M+, 100), 269 (95), 207 (50), 187 (20), 97 (5), 77 (5). HRESIMS calcd. for [C17H16S2 + H]+: 285.4469; found: 285.4466.
5,5’-Bis-(2-methylthienyl)-(3-methylphenyl)methane (5b). Yellow waxy solid. IR (KBr) νmax: 3058, 2919, 2859, 1446, 800, 755 cm−1. 1H-NMR (CDCl3): δ 7.19 (t, J = 7.5 Hz, 1H), 7.13–7.03 (m, 3H), 6.58 (dd, J = 0.5, 3.5 Hz, 2H), 6.54 (dd, J = 1.0, 3.5 Hz, 2H), 5.66 (s, 1H), 2.44 (s, 6H, 2 × CH3), 2.35 (s, 3H, CH3). 13C-NMR (CDCl3): δ 145.4, 143.7, 139.0, 138.0, 129.0, 128.3, 127.8, 125.6, 125.3, 124.5, 47.8, 21.5, 15.4. MS (EI, 70 eV): m/z = 298 (M+, 98), 283 (100), 201 (15), 91 (5), 77 (5). HRESIMS calcd. for [C18H18S2 + H]+: 299.4735; found: 299.4733.
5,5’-Bis-(2-methylthienyl)-(4-methylphenyl)methane (5c). Yellow waxy solid. IR (KBr) νmax: 3062, 2920, 1533, 1448, 745 cm−1. 1H-NMR (CDCl3): δ 7.24 (d, J = 7.9 Hz, 2H), 7.16 (d, J = 7.9 Hz, 2H), 6.63 (d, J = 3.4 Hz, 2H), 6.61 (d, J = 3.4 Hz, 2H), 5.68 (s, 1H), 2.45 (s, 6H, 2 × CH3), 2.37 (s, 3H, CH3). 13C-NMR (CDCl3): δ 145.5, 140.9, 138.9, 136.5, 129.1, 128.1, 125.5, 124.4, 47.4, 21.0, 15.3. MS (EI, 70 eV): m/z = 298 (M+, 90), 283 (100), 201 (20), 91 (5), 77 (5). Anal. calcd. for C18H18S2: C, 72.43; H, 6.08. Found C, 72.80; H, 6.43.
5,5’-Bis-(2-methylthienyl)-(3-methoxyphenyl)methane (5d). Yellow waxy solid. IR (KBr) νmax: 2922, 1599, 1487, 1448, 1265, 1156, 1046 cm−1. 1H-NMR (CDCl3): δ 7.26 (t, J = 7.9 Hz, 1H), 6.94 (d, J = 8.1 Hz, 1H), 6.89 (t, J = 2.0 Hz, 1H), 6.82 (dd, J = 0.6, 8.1 Hz, 1H), 6.63 (dd, J = 0.5, 3.5 Hz, 2H), 6.58 (dd, J = 1.0, 3.5 Hz, 2H), 5.67 (s, 1H), 3.80 (s, 3H, OCH3), 2.44 (s, 6H, 2 × CH3). 13C-NMR (CDCl3): δ 159.6, 145.4, 145.0, 139.1, 129.4, 125.7, 124.5, 120.8, 114.3, 112.1, 55.2, 47.8, 15.4. MS (EI, 70 eV): m/z = 314 (M+, 100), 299 (15), 283 (10), 207 (90), 122 (15). Anal. calcd. for C18H18S2O: C, 68.75; H, 5.77. Found C, 68.91; H, 5.84.
5,5’-Bis-(2-methylthienyl)-(3-chlorophenyl)methane (5e). Yellow waxy solid. IR (KBr) νmax: 3063, 2919, 1473, 1262, 1095, 1034, 802 cm−1. 1H-NMR (CDCl3): δ 7.33 (s, 1H), 7.28–7.21 (m, 3H), 6.64–6.59 (m, 4H), 5.68 (s, 1H), 2.46 (s, 6H, 2 × CH3). 13C-NMR (CDCl3): δ 145.8, 144.3, 139.4, 134.3, 129.7, 128.5, 127.2, 126.6, 125.9, 124.7, 47.4, 15.4. MS (EI, 70 eV): m/z = 320 (M+, 24), 318 (M+, 99), 305 (25), 303 (100), 283 (10), 223 (8), 221 (25), 207 (99), 113 (7), 111 (20). Anal. calcd. for C17H15S2Cl: C, 64.03; H, 4.74. Found C, 64.06; H, 4.92.
5,5’-Bis-(2-methylthienyl)-(2-bromophenyl)methane (5f). Yellow waxy solid. IR (KBr) νmax: 3063, 2923, 2856, 1442, 1229, 1028, 795 cm−1. 1H-NMR (CDCl3): δ 7.55 (dd, J = 7.9, 1.2 Hz, 1H), 7.32 (dd, J = 7.9, 1.8 Hz, 1H), 7.25 (dt, J = 1.2, 7.9 Hz, 1H), 7.10 (dt, J = 1.8, 7.9 Hz, 1H), 6.59–6.54 (m, 4H), 6.13 (s, 1H), 2.42 (s, 6H, 2 × CH3). 13C-NMR (CDCl3): δ 143.8, 143.0, 139.3, 132.9, 130.1, 128.6, 127.6, 126.2, 124.6, 124.4, 46.7, 15.4. . MS (EI, 70 eV): m/z = 364 (M+, 100), 362 (M+, 100), 349 (60), 347 (60), 283 (25), 207 (60), 97 (25). Anal. calcd. for C17H15S2Br: C, 56.20; H, 4.16. Found C, 56.53; H, 4.47.
5,5’-Bis-(2-methylthienyl)-(3-nitrophenyl)methane (5g). Yellow waxy solid. IR (KBr) νmax: 3061, 2918, 1529, 1350, 804 cm−1. 1H-NMR (CDCl3): δ 8.18 (t, J = 1.9 Hz, 1H), 8.13 (dq, J = 0.9, 8.2 Hz, 1H), 7.66 (d, J = 7.9 Hz, 1H), 7.50 (t, J = 7.9 Hz, 1H), 6.63-6.58 (m, 4H), 5.80 (s, 1H), 2.44 (s, 6H, 2 × CH3). 13C-NMR (CDCl3): δ 148.4, 145.9, 143.4, 139.9, 134.4, 129.4, 126.2, 124.8, 123.3, 122.1, 47.3, 15.4. MS (EI, 70 eV): m/z = 329 (M+, 100), 314 (96), 283 (5), 232 (5), 207 (70), 97 (5), 77 (5). Anal. calcd. for C17H15NO2S2: C, 61.98; H, 4.59; N, 4.25. Found C, 62.37; H, 4.63; N, 4.45.
3.4. General Procedure for the Preparation of Bis(fur-2-yl)methanes 6a-6f
To a cooled (0 °C) solution of aryl benzaldehyde (0.5 mmol) and RuCl3·3H2O (0.05 mmol) in ethylene glycol dimethyl ether (1 mL) was added 2-methylfuran (6.0 mmol) under air atmosphere and the mixture was placed into refrigerator to stay without stirring at 5 °C. The mixture was shaken for several seconds every day to ensure homodispersity (monitored by TLC). The reaction mitxure was then concentrated under reduced pressure. The residue was purified by flash chromatography on silica gel (eluent: EtOAc/PE = 1:8) to yield the corresponding product.
5,5’-Bis-(2-methylfuryl)-(3-methylphenyl)methane (6a). Waxy solid. IR (KBr) νmax: 2922, 1608, 1449, 1137, 1021, 779 cm−1. 1H-NMR (CDCl3): δ 7.24 (t, J = 7.5 Hz, 1H), 7.11 (s, 2H), 7.09 (s, 1H), 5.93 (d, J = 3.3 Hz, 2H), 5.91 (d, J = 3.3 Hz, 2H), 5.35 (s, 1H), 2.37 (s, 3H, CH3), 2.29 (s, 6H, 2 × CH3). 13C-NMR (CDCl3): δ 153.0, 151.4, 139.9, 138.0, 129.1, 128.3, 127.8, 125.5, 108.1, 106.1, 45.1, 21.5, 13.6. MS (EI, 70 eV): m/z = 266 (M+, 60), 251 (100), 175 (60). HRESIMS calcd. for [C18H18O2 + H]+: 267.3423; found: 267.3417.
5,5’-Bis-(2-methylfuryl)-(4-methylphenyl)methane (6b). Waxy solid. IR (KBr) νmax: 2922, 1607, 1510, 1448, 1130, 1014, 775 cm−1. 1H-NMR (CDCl3): δ 7.20 (d, J = 8.2 Hz, 2H), 7.16 (d, J = 8.2 Hz, 2H), 5.92 (d, J = 3.5 Hz, 2H), 5.90 (d, J = 3.5 Hz, 2H), 5.35 (s, 1H), 2.37 (s, 3H, CH3), 2.28 (s, 6H, 2 × CH3). 13C-NMR (CDCl3): δ 153.1, 151.4, 137.1, 136.5, 129.2, 128.3, 108.1, 106.1, 44.8, 21.1, 13.6. MS (EI, 70 eV): m/z = 266 (M+, 100), 251 (20), 185 (15), 175 (55). HRESIMS calcd. for [C18H18O2 + H]+: 267.3423; found: 267.3422.
5,5’-Bis-(2-methylfuryl)-(3-methoxyphenyl)methane (6c). Waxy solid. IR (KBr) νmax: 2922, 1600, 1262, 1151, 771 cm−1. 1H-NMR (CDCl3): δ 7.26 (t, J = 7.8 Hz, 1H), 6.87 (d, J = 7.8 Hz, 1H), 6.84–6.80 (m, 2H), 5.91 (d, J = 3.8 Hz, 2H), 5.89 (d, J = 3.8 Hz, 2H), 5.33 (s, 1H), 3.79 (s, 3H, OCH3), 2.27 (s, 6H, 2 × CH3). 13C-NMR (CDCl3): δ 159.7, 152.7, 151.4, 141.6, 129.3, 120.8, 114.3, 112.2, 108.2, 106.1, 55.1, 45.1, 13.6. MS (EI, 70 eV): m/z = 282 (M+, 100), 251 (80), 175 (60). HRESIMS calcd. for [C18H19O3 + H]+: 283.3417; found: 283.3411.
5,5’-Bis-(2-methylfuryl)-(3-chlorophenyl)methane (6d). Waxy solid. IR (KBr) νmax: 2923, 1624, 1437, 1131 cm−1. 1H-NMR (CDCl3): δ 7.26–7.23 (m, 3H), 7.16–7.14 (m, 1H), 5.91 (d, J = 3.2 Hz, 2H), 5.89 (d, J = 3.2 Hz, 2H), 5.32 (s, 1H), 2.26 (s, 6H, 2 × CH3). 13C-NMR (CDCl3): δ 152.0, 151.7, 142.0, 134.2, 129.7, 128.5, 127.2, 126.6, 108.5, 106.2, 44.7, 13.6. MS (EI, 70 eV): m/z = 288 (M+, 20), 286 (M+, 60), 273 (M+, 5), 271 (M+, 15), 175 (100). HRESIMS calcd. for [C17H16O2Cl + H]+: 287.7607; found: 287.7601.
5,5’-Bis-(2-methylfuryl)-(2-bromophenyl)methane (6e). Waxy solid. IR (KBr) νmax: 2926, 1462, 1131, 1021, 747 cm−1. 1H-NMR (CDCl3): δ 7.58 (dd, J = 7.9, 1.0 Hz, 1H), 7.27 (dt, J = 1.0, 7.9 Hz, 1H), 7.21 (dd, J = 7.9, 1.8 Hz, 1H), 7.14 (dt, J = 1.8, 7.9 Hz, 1H), 5.91 (d, J = 3.0 Hz, 2H), 5.86 (d, J = 3.0 Hz, 2H), 5.83 (s, 1H), 2.27 (s, 6H, 2 × CH3). 13C-NMR (CDCl3): δ 151.7, 151.6, 139.1, 132.9, 130.1, 128.5, 127.5, 124.5, 108.8, 106.1, 44.4, 13.6. MS (EI, 70 eV): m/z = 332 (M+, 100), 330 (M+, 100), 317 (20), 315 (20), 175 (65). HRESIMS calcd. for [C17H16O2Br + H]+: 332.2117; found: 332.2109.
5,5’-Bis-(2-methylfuryl)-(3-nitrophenyl)methane (6f). Waxy solid. IR (KBr) νmax: 2922, 1528, 1348, 1132, 781 cm−1. 1H-NMR (CDCl3): δ 8.13–8.11 (m, 2H), 7.59 (d, J = 7.9 Hz, 1H), 7.48 (dt, J = 2.3, 7.9 Hz, 1H), 5.94 (d, J = 3.0 Hz, 2H), 5.92 (d, J = 3.0 Hz, 2H), 5.44 (s, 1H), 2.25 (s, 6H, 2 × CH3). 13C-NMR (CDCl3): δ 152.1, 151.1, 148.4, 142.2, 134.6, 129.3, 123.4, 122.2, 108.8, 106.3, 44.7, 13.6. MS (EI, 70 eV): m/z = 297 (M+, 90), 282 (15), 175 (100). HRESIMS calcd. for [C17H16NO4 + H]+: 298.3132; found: 298.3129.