2.2. Reducing Power
The reducing power assay determines the electron-donating ability of antioxidants using the potassium ferricyanide reduction method. The presence of reducers in the test solution leads to the reduction of the Fe
3+/ferricyanide complex to the ferrous form. The reducing capacity of a compound may serve as a significant indicator of its potential antioxidant activity. The reducing powers of three extracts from
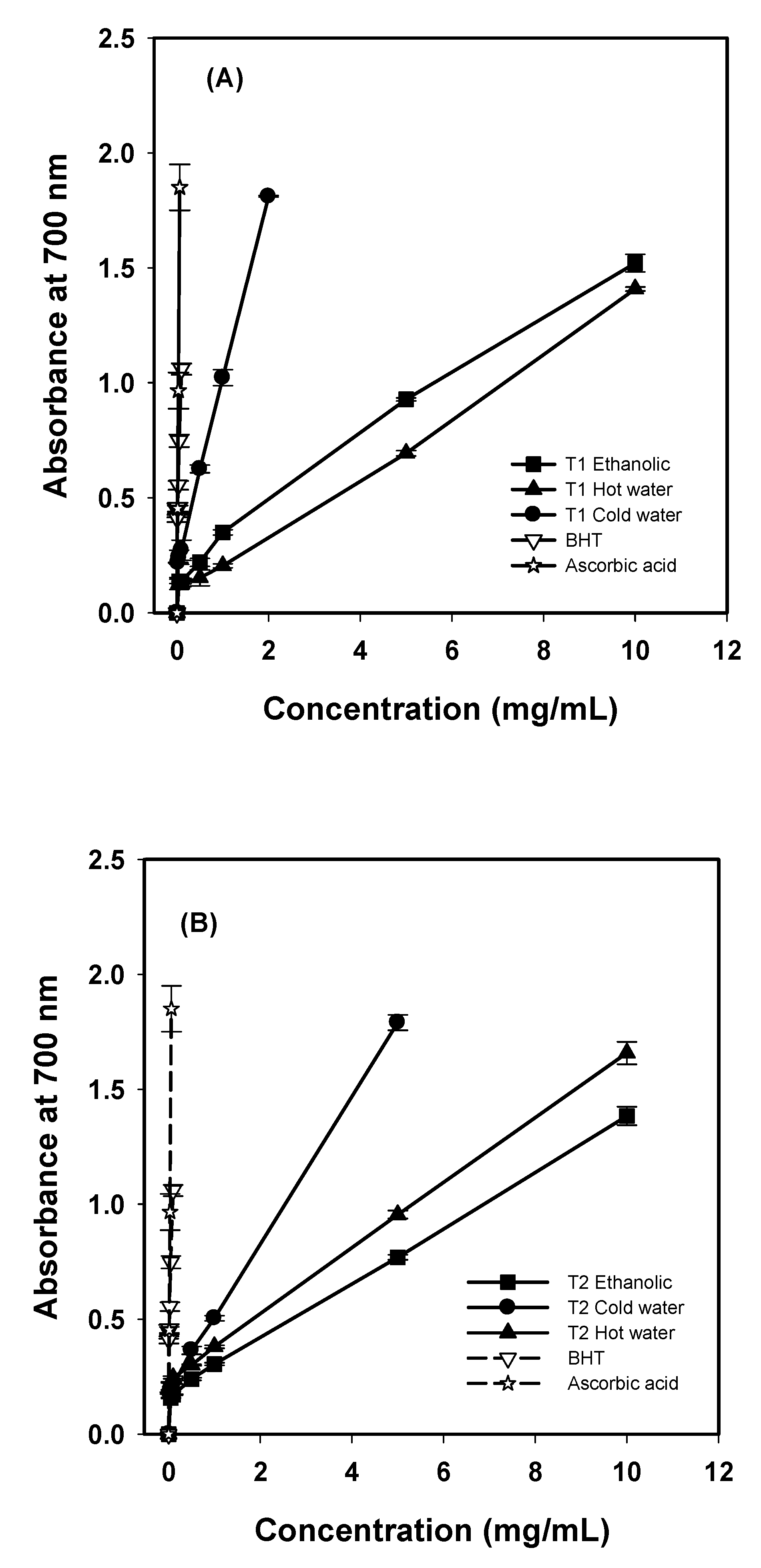
G. frondosa T1 and T2 increased rapidly with the increase in concentration, as shown in
Figure 1.
Figure 1.
Reducing power of various extracts from G. frondosa T1 (A), and G. frondosa T2 (B). Each value is expressed as mean ± S.D. (n = 3).
Figure 1.
Reducing power of various extracts from G. frondosa T1 (A), and G. frondosa T2 (B). Each value is expressed as mean ± S.D. (n = 3).
The reducing powers of cold-water extracts from both
G. frondosa T1 and T2 were excellent. At 1 mg/mL, the reducing powers of cold-water extracts T1 and T2 were 1.02 and 0.50, respectively, higher than that of ethanolic (0.35 and 0.30 for
G. frondosa T1 and T2, respectively) and hot-water extracts (0.20 and 0.38 for
G. frondosa T1 and T2, respectively). The reducing powers of
G. frondosa T1 and T2 were 1.81 at 2 mg/mL and 1.75 at 5 mg/mL, respectively, and were higher than that of
H. marmoreus fruit bodies, which is reported to be 0.99 at 5 mg/mL [
11]. However, the reducing power of ascorbic acid was 0.97 at 0.03 mg/mL and that of BHT was 1.06 at 0.1 mg/mL.
The reducing power of hot-water extracts from
G. frondosa T1 and
G. frondosa T2 was 0.69 and 0.95 at 5 mg/mL, respectively. Mau
et al. [
12] have reported that hot-water extracts of mature and baby Ling chihs revealed the reducing powers of 1.08 and 1.04 at 5 mg/mL, respectively. In addition, the hot-water extract of
Agrocybe cylindracea strain B exhibited a reducing power of 0.87 at 5 mg/mL [
13].
G. frondosa T1 and
G. frondosa T2 hot-water extracts seem to be lower in reducing power, as shown in
Figure 1, when compared to those reported by Mau
et al. [
14]. Furthermore,
G. frondosa T2 hot-water extract was higher than the same extract from
Agrocybe cylindracea strain B in reducing power.
As to ethanolic extracts, the reducing powers of
G. frondosa T1 and T2 were 0.93 and 0.77 at 5 mg/mL, respectively. Tsai
et al. [
14] reported that the reducing powers of ethanolic extracts were 0.37, 0.25 and 0.61 at 5 mg/mL for
A. blazei,
A. cylindracea and
B. edulis, respectively. Apparently, the reducing powers of ethanolic extracts from
G. frondosa T1 and T2 were more effective than those previously reported.
The reducing capacity may be a significant index of antioxidant activity. Various mechanisms related to antioxidant activities were suggested, including chain initiation, binding of transition metal ion catalysts, decomposition of peroxides, prevention of continued hydrogen abstraction, reductive capacity, and radical scavenging [
15].
2.3. Scavenging Ability on DPPH Radicals
Stable DPPH radicals are widely used to evaluate the antioxidant activities of proton-donating substances according to hydrogen donating ability. DPPH radicals accept electrons or hydrogen radicals to form stable diamagnetic molecules. The antioxidant activity of substances can be expressed as the reduction capability of DPPH radical at 517 nm [
16]. This chromogen radical compound can directly react with antioxidants and the procedure is simple, rapid and sensitive. Hence, the model of scavenging the stable DPPH radical is a widely used method to evaluate the free radical scavenging ability in variety of samples. Generally, methanolic and/or ethanolic extracts were more effective in scavenging abilities than hot-water extract for most mushroom species reported previously [
17]. Similar observations were found in this study. The ethanolic extracts from
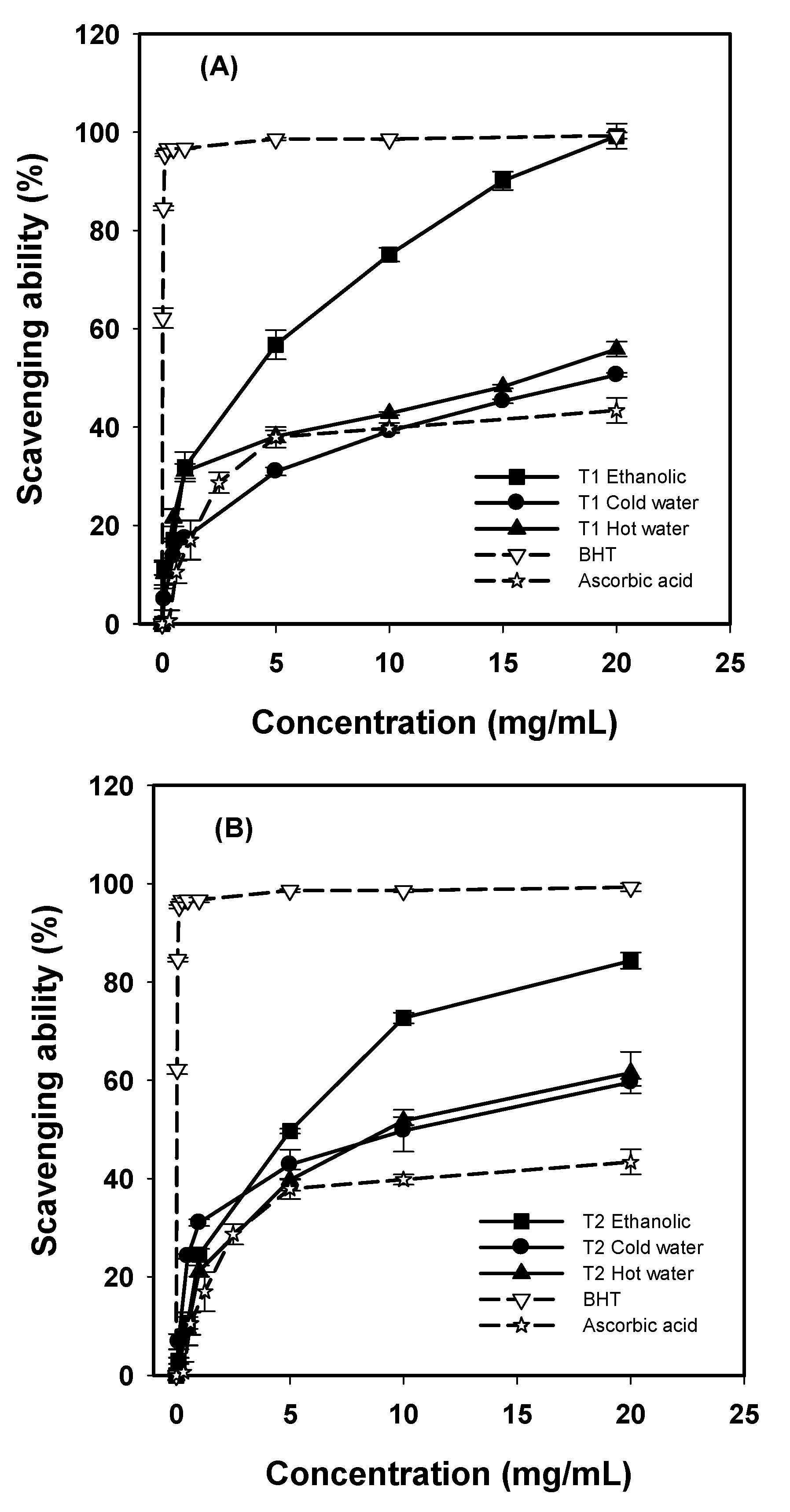
G. frondosa T1 and T2 showed higher scavenging ability on DPPH radicals than water extracts (
Figure 2). The ethanolic extracts of
G. frondosa T1 and T2 scavenged DPPH radicals by 99.19% and 84.36% at 20 mg/mL, respectively. For cold-water extracts, the scavenging abilities of
G. frondosa T1 and T2 were 50.62% and 59.58% respectively, at 20 mg/mL. However, for the hot-water extracts, the scavenging abilities of
G. frondosa T1 and T2 was 55.87% and 61.56% respectively, at 20 mg/mL. The BHT had considerably high scavenging ability of 95.34% at 0.125 mg/mL. In addition, ascorbic acid revealed a lower scavenging ability of 43.39% at 20 mg/mL.
Figure 2.
Scavenging ability of various extracts from G. frondosa T1 (A), and G. frondosa T2 (B) on DPPH radicals. Each value is expressed as mean ± S.D. (n = 3).
Figure 2.
Scavenging ability of various extracts from G. frondosa T1 (A), and G. frondosa T2 (B) on DPPH radicals. Each value is expressed as mean ± S.D. (n = 3).
At 20 mg/mL, the scavenging abilities of hot-water extracts from mature and baby Ling chih, mycelia on DPPH radicals were 73.8%, 80.1%, and 91.2%, respectively [
12]. Hot-water extracts of
A. blazei,
A. cylindracea and
B. edulis revealed scavenging abilities of 51.6%, 39.2% and 53.7%, respectively, at 20 mg/mL [
14]. The scavenging ability of hot-water extracts from
G. frondosa T1 and T2 were higher than that from
A. blazei,
A. cylindracea and
B. edulis, but were less effective than that from mature and baby Ling chih, mycelia.
Tsai
et al. [
14] reported that the scavenging abilities of three ethanolic extracts were 97.4%, 95.8% and 94.3%, respectively, at 20 mg/mL for
A. blazei,
A. cylindracea and
B. edulis. The scavenging ability of ethanolic extract from
G. frondosa T1 in this study was comparable to that previously reported.
High scavenging ability in ethanolic extract may attribute to high level of antioxidant components in the extract, which react rapidly with DPPH radicals and reduce most DPPH radical molecules. The results from this study indicated that ethanolic extracts were free radical scavengers and behaved possibly as primary antioxidants. Various extracts of G. frondosa T1 and T2 may react with free radicals, the major initiators of the autoxidation chain of fat, and accordingly terminate the chain reactions.
2.4. Chelating Abilities on Ferrous Ions
The chelating effects of various extracts on Fe
2+ were determined by the formation of ferrozine-Fe
2+ complexes. Chelating agents are able to capture ferrous ion before ferrozine, thus hindering the formation of ferrozine-Fe
2+. Spectrophotometic examination of ferrozine-Fe
2+ absorbance can accordingly be used to calculate ferrous ion chelating activity. The metal chelating capacity is important since it reduces the concentration of transition metals that may act as catalysts to generate the first few radicals to initiate the radical-mediated oxidative chain reactions in biological and/or food systems. Ion chelating agents also may inhibit the Fenton reaction and hydroperoxide decomposition [
18].
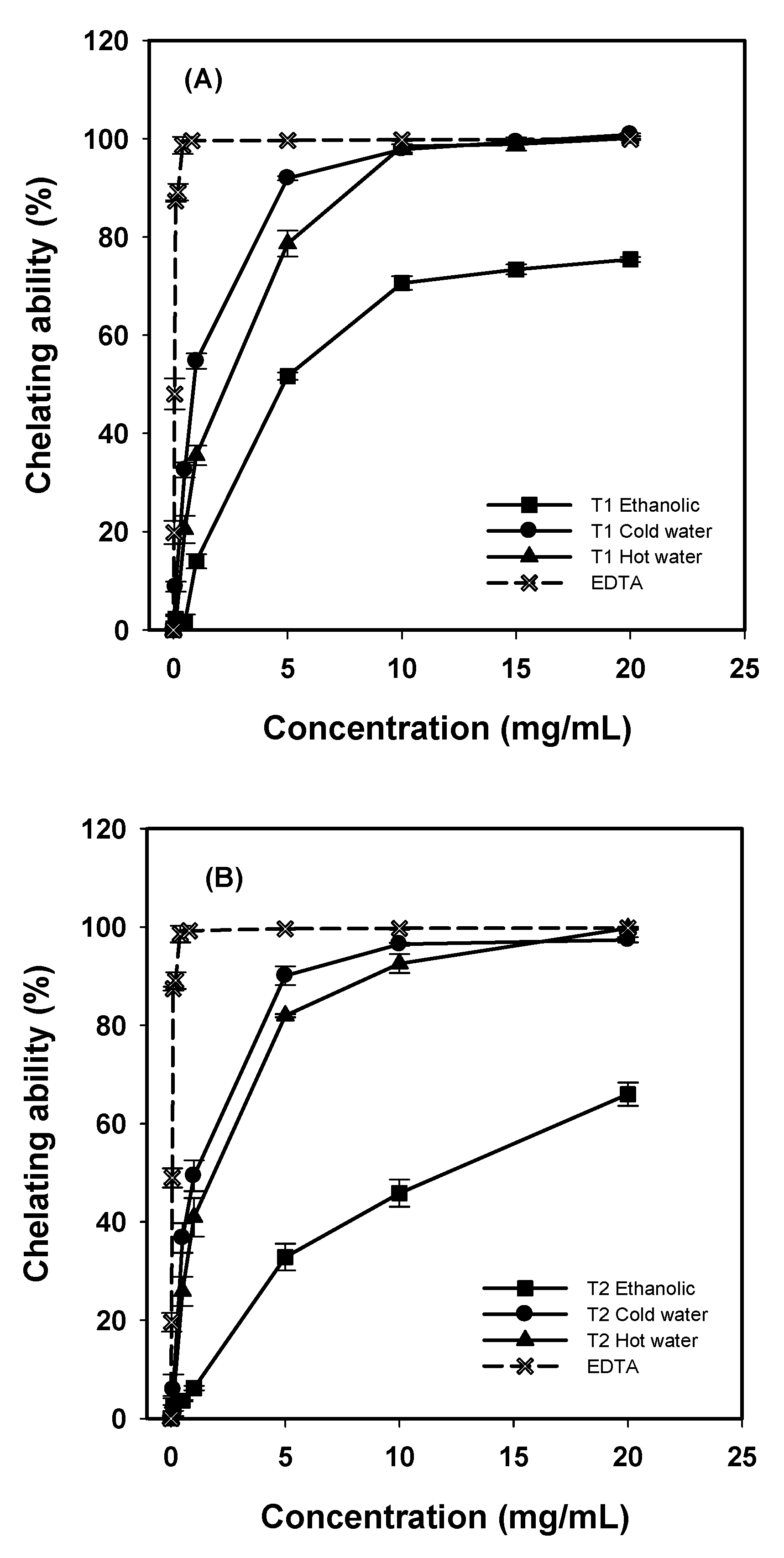
Chelating abilities of various extracts increased as concentration rose (
Figure 3). The chelating abilities of water extracts from
G. frondosa T1 and T2 on ferrous ions were higher than that of ethanolic extracts. At 5–20 mg/mL, chelating abilities were 92.0–100%, 78.7–100%, and 51.7–75.4% for cold-water, hot-water and ethanolic extracts from
G. frondosa T1, respectively, and 90.1–97.4%, 81.9–99.7%, and 32.8–66.0% for that from
G. frondosa T2, respectively. Ethylenediaminetetraacetic acid (EDTA) exhibited a high chelating ability of 99.4% at 0.4 mg/mL.
Figure 3.
Chelating ability of various extracts from G. frondosa T1 (A), and G. frondosa T2 (B) on ferrous ions. Each value is expressed as mean ± S.D. (n = 3).
Figure 3.
Chelating ability of various extracts from G. frondosa T1 (A), and G. frondosa T2 (B) on ferrous ions. Each value is expressed as mean ± S.D. (n = 3).
At 5 mg/mL, chelating abilities were 66.6% and 94.1%, respectively, for cold-water extracts from
P. citrinopileatus and
H. marmoreus fruit bodies [
11]. Apparently, the chelating ability of
G. frondosa T1 cold-water extract was comparable to that of
H. marmoreus, and higher than that of
P. citrinopileatus. Tsai
et al. [
14] reported that chelating abilities of ethanolic extracts were 36.2–58.8%, 24.6–59.8%, and 37.4–61.8% for
A. blazei,
A. cylindracea and
B. edulis at 5–20 mg/mL, respectively. The chelating ability of
G. frondosa T1 ethanolic extract was obviously more effective than those reported previously. Apparently, the chelating ability of hot-water extracts of
G. frondosa T1 and T2 were higher than that reported previously. The results in this study suggest that extracts from
G. frondosa T1 and T2 show the noticeable chelating iron capacity and may act as chelating agents to prevent and/or reduce lipid peroxidation.
2.5. Scavenging Effect on Superoxide Anion
Superoxide anion, which is formed in almost all aerobic cells, is a major agent in the mechanism of oxygen toxicity [
19]. Oxygen anion (O
2•−) is also a highly reactive chemical species and can be generated under regular physiological conditions. In addition to direct attack of important biological molecules, O
2•− may also be involved in the formation of singlet oxygen and hydroxyl radicals, which may increase local oxidative stress and initiate cellular damage and pathological incidents such as arthritis and Alzheimer’s disease. Therefore, to study the scavenging of a superoxide anion is of great importance to human health.
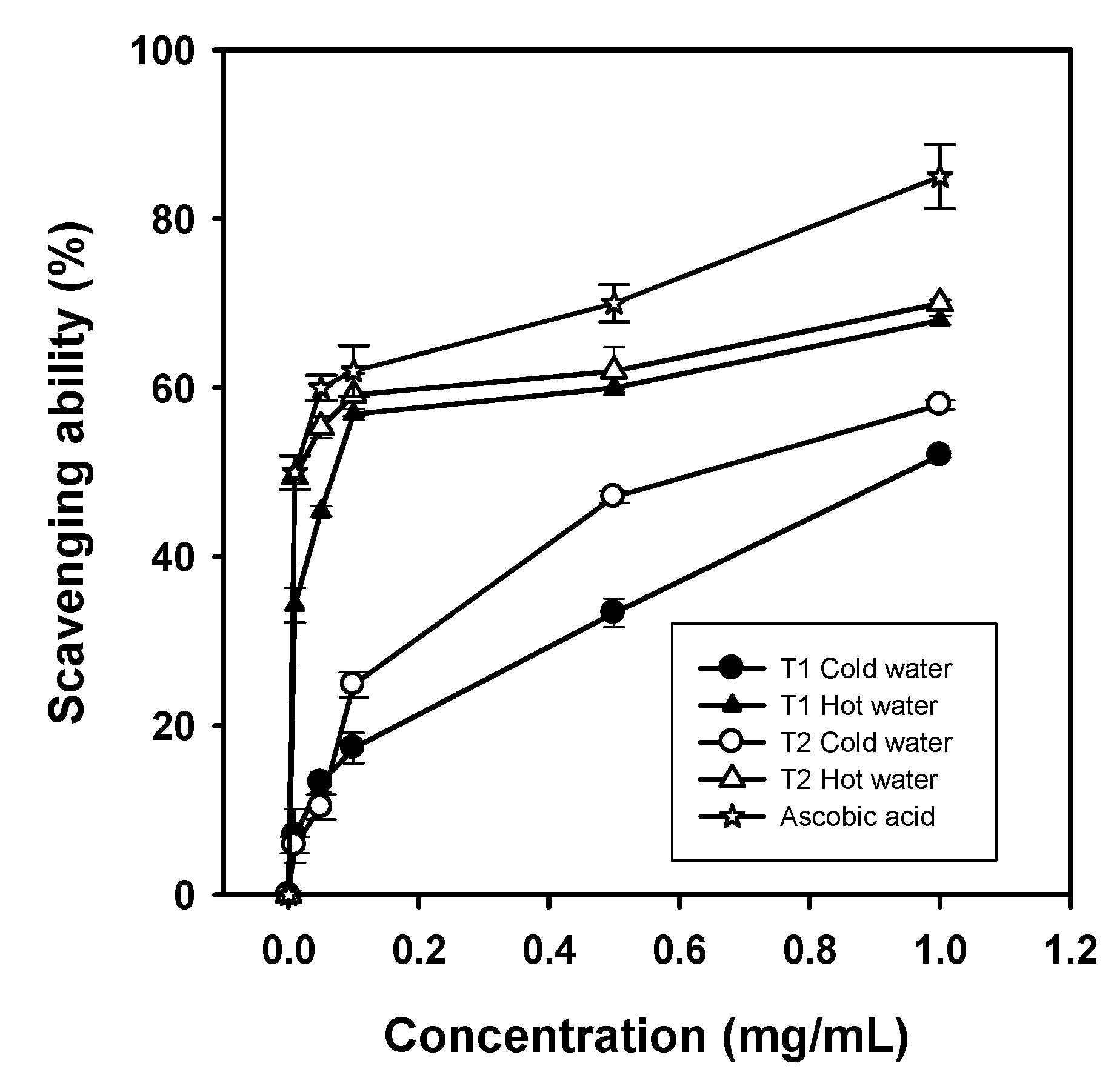
Superoxide radicals are generated in phenazine methosulfate-nicotinamide adenine dinucleotide (PMS/NADH) system by the reduction of nitroblue tetrazolium (NBT). For cold-water extracts,
G. frondosa T1 and T2 showed scavenging abilities of 33.4% and 47.1% at 0.5 mg/mL, respectively. Scavenging abilities of
G. frondosa T1 and T2 hot-water extracts were 56.9% and 62 at 0.5 mg/mL, respectively. In general, the scavenging abilities of superoxide radicals for both extracts were in the descending order of
G. frondosa T2 hot-water >
G. frondosa T1 hot-water >
G. frondosa T2 cold-water >
G. frondosa T1 cold-water extracts. However, the scavenging ability of L-ascorbic acid was up to 85% at 1 mg/mL, whereas the scavenging abilities of superoxide radicals of ethanolic extracts from both
G. frondosa T1 and T2 were not detected (not shown in
Figure 4). Song and Yen [
19] demonstrated that the superoxide scavenging abilities were 40.2% and 30.0% for water and methanol extracts of mycelia from
Antrodia camphorata at 200 µg/mL, respectively. Superoxide anions indirectly initiate lipid oxidation as superoxides and hydrogen peroxides serve as the precursors of singlet oxygen and hydroxyl radicals. As the result, both
G. frondosa T1 and T2 water extracts may be the potential scavengers of superoxide anions to prevent lipid oxidation.
2.6. EC50 Values in Antioxidant Properties
Antioxidant properties investigated in this study are expressed as EC
50 values for comparison (
Table 1). Higher EC
50 values indicate lower effectiveness in antioxidant properties. Effectiveness in reducing power was in a descending order of cold-water > ethanolic > hot-water extracts. As to the scavenging ability on DPPH radicals, various extracts were effective in the order of ethanolic > hot-water > cold-water extracts. The ethanolic extract with EC
50 of 5.51 mg/mL was more effective than that of hot-water extract (17.36 mg/mL) and cold-water extract (19.42 mg/mL) from
G. frondosa T1. Effectiveness in chelating ability on ferrous ions was in a descending order of cold-water > hot-water > ethanolic extracts. EC
50 values in chelating effect on ferrous ions were 0.88, 2.33, and 4.82 mg/mL for cold-water extract, hot-water extract, and ethanolic extract from
G. frondosa T1, respectively. The three extracts of
G. frondosa T1 showed dominant chelating effects on ferrous ions, and the similar trend was found in three extracts of
G. frondosa T2.
Figure 4.
Scavenging ability of cold-water and hot-water extracts from G. frondosa T1 and T2 on superoxide radicals. Each value is expressed as mean ± S.D. (n = 3).
Figure 4.
Scavenging ability of cold-water and hot-water extracts from G. frondosa T1 and T2 on superoxide radicals. Each value is expressed as mean ± S.D. (n = 3).
As to superoxide radical scavenging, the effectiveness was in a descending order of hot-water > cold-water extracts with G. frondosa T2 being more effective than T1. EC50 values in scavenging ability on superoxide radical were 0.95 and 0.62 mg/mL for cold-water extracts and 0.07 and 0.01 for hot-water extracts from G. frondosa T1 and T2, respectively. These results suggest that both extracts exhibited good scavenging superoxide radical activities as evidenced by their low EC50 values (<1 mg/mL).
In general, EC50 values of the three extracts of G. frondosa T1 and T2 were below 20 mg/mL for all antioxidant properties investigated in this study. Accordingly, the extracts of G. frondosa T1 and T2 exhibited effective antioxidant properties. These results imply that extracts of G. frondosa T1 and T2 may have potential benefit to protect human beings from oxidative damage.
2.7. Antioxidant Components
Several potential antioxidant components including total phenols, flavonoids, tocopherols, ascorbic acid and β-carotene were determined (
Table 2). Ascorbic acid was mainly found in water extracts from
G. frondosa T1 (2.90–5.45 mg/mL) and T2 (5.60–5.76 mg/mL). However, β-carotene was not detected in water extracts. The
α-tocopherol was mainly found in ethanolic extracts, and the content of tocopherols in ethanolic extracts from
G. frondosa T1 and T2, were 24.38 and 3.87 mg/g, respectively, which was considerably high in mushrooms reported previously.
Table 1.
EC50 values of various extracts from Grifola frondosa T1 (A) and T2 (B) for antioxidant properties.
Table 1.
EC50 values of various extracts from Grifola frondosa T1 (A) and T2 (B) for antioxidant properties.
| EC50 values (mg/mL) * |
|---|
| (A) | T1 | (B) | T2 |
|---|
| Ethanolic | Cold water | Hot water | Ethanolic | Cold water | Hot water |
|---|
| Reducing power ** | 2.43 ± 0.01a | 0.37 ± 0.04b | 3.15 ± 0.01c | 1.88 ± 0.00a | 0.99 ± 0.01b | 2.74 ± 0.01c |
| Scavenging effect on DPPH radicals ** | 3.51 ± 0.02a | 19.42 ± 0.00b | 17.36 ± 0.01c | 5.07 ± 0.02a | 10.25 ± 0.02b | 9.27 ± 0.00c |
| Chelating effect on ferrous ions ** | 4.82 ± 0.02a | 0.88 ± 0.08b | 2.33 ± 0.05c | 12.07 ± 0.09a | 1.06 ± 0.03b | 1.88 ± 0.01c |
| Scavenging ability on superoxide anion ** | ND *** | 0.95 ± 0.01a | 0.07 ± 0.00b | ND | 0.62 ± 0.02a | 0.01 ± 0.00b |
Table 2.
Antioxidant contents of extracts from Grifola frondosa T1 (A) and T2 (B).
Table 2.
Antioxidant contents of extracts from Grifola frondosa T1 (A) and T2 (B).
| Contents (mg/g) * |
|---|
| (A) | T1 | (B) | T2 |
|---|
| Ethanolic | Cold water | Hot water | Ethanolic | Cold water | Hot water |
|---|
| Total phenols ** | 19.61 ± 1.69a | 39.78 ± 1.86b | 30.78 ± 0.42c | 15.48 ± 1.78a | 38.96 ± 1.14b | 42.30 ± 1.64c |
| Flavonoid ** | 3.05 ± 0.10a | 1.09 ± 0.06b | 1.19 ± 0.05c | 0.11 ± 0.03a | 0.52 ± 0.04b | 0.76 ± 0.04c |
| Ascorbic acid ** | ND *** | 5.45 ± 0.40a | 2.90 ± 0.35b | ND | 5.76 ± 0.43a | 5.60 ± 0.25b |
| α-Tocopherol ** | 24.38 ± 1.98a | 2.82±0.38b | ND | 3.87 ± 0.21 | ND | ND |
| β-Carotene ** | ND | ND | ND | ND | ND | ND |
Flavonoids act as antioxidant agents by direct free radical scavenging, transition metal chelation and maintenance of endogenous antioxidants, such as glutathione and superoxide dismutase systems. Total flavonoid content was 1.09–3.05 mg/g and 0.11–0.76 mg/g for G. frondosa T1 and T2, respectively.
Phenols are known to be the effective antioxidants in plants due to their hydroxyl groups. Typical phenols with antioxidant activity have been characterized as phenolic acids and flavonoids. Phenolic acids are repeatedly implicated as natural antioxidants in fruits, vegetables and other plants [
16]. Total phenols were found in three extracts of
G. frondosa T1 (19.61–39.78 mg/g) or T2 (15.48–42.30 mg/g). Water extracts contained more total phenols than ethanolic extracts did. The results from this study were similar to the results reported by Lee
et al. [
11]. In addition, there was a strong correlation between the contents of total phenols and EC
50 values of chelating ferrous ions ability with the correlation coefficients (
R2) being 0.992 and 0.968 for
G. frondosa T1 and T2, respectively.
Polyphenols, such as BHT and gallate, are known to be the effective antioxidants. The high content of total polyphenolics in
G. frondosa T1 and T2 may be the key components accounting for the high reducing power, metal chelating activity and superoxide anion scavenging ability. Numerous studies have conclusively shown that consumption of foods high in phenolic content reduce the risk of heart disease [
16]. However, other specific active components that may also contribute in part to the antioxidant properties of these extracts from
G. frondosa T1 and T2, and will require further isolation and identification.
2.8. β-Glucan Content
According to the published literature, the polysaccharide purified from maitake mushroom extracts had very strong anti-tumor capabilities [
3]. Clinical studies had demonstrated that this glucan activates macrophages, subsequently increasing T-cell cascade to elevate the immune defense mechanism in the body [
3]. However, β-(1→3)-D-glucan with β-(1→6) branched chain had been proved to have the greatest anti-tumor effect [
3]. The β-glucan is used in enhancing defense mechanisms by activating macrophages, natural killer cells and T lymphocytes in immune system to promote the body resistance to the tumors. For
G. frondosa T1, the ethanolic extract β-glucan content, 13.25 ± 1.38%, was slightly higher than that of the hot-water extract (10.69 ± 0.82% glucan) and the cold-water extract (10.04 ± 1.39% glucan). The results of the
G. frondosa T2 extracts indicated that the ethanolic extract, containing 18.73 ± 1.68% β-glucan, was significantly higher (
P < 0.05) than that of the cold-water extract (9.26 ± 0.30% glucan) and hot-water extract (10.26 ± 0.06% glucan). Polysaccharides extracted from several mushrooms had reported to contain antioxidant properties by their free radical scavenging ability [
21].
G. frondosa T1 and T2 extracts contained polysaccharides that may contribute in part to the antioxidant properties.