Peppermint Oil Decreases the Production of Virulence-Associated Exoproteins by Staphylococcus aureus
Abstract
:1. Introduction
2. Results and Discusssion
2.1. Chemical composition of peppermint oil
2.2. Effects of peppermint oil on S. aureus growth
2.3. Peppermint oil decreases α-toxin, TSST-1, SEA and SEB levels in S. aureus culture supernatants
2.4. Peppermint oil inhibits hemolytic activities in S. aureus culture supernatants
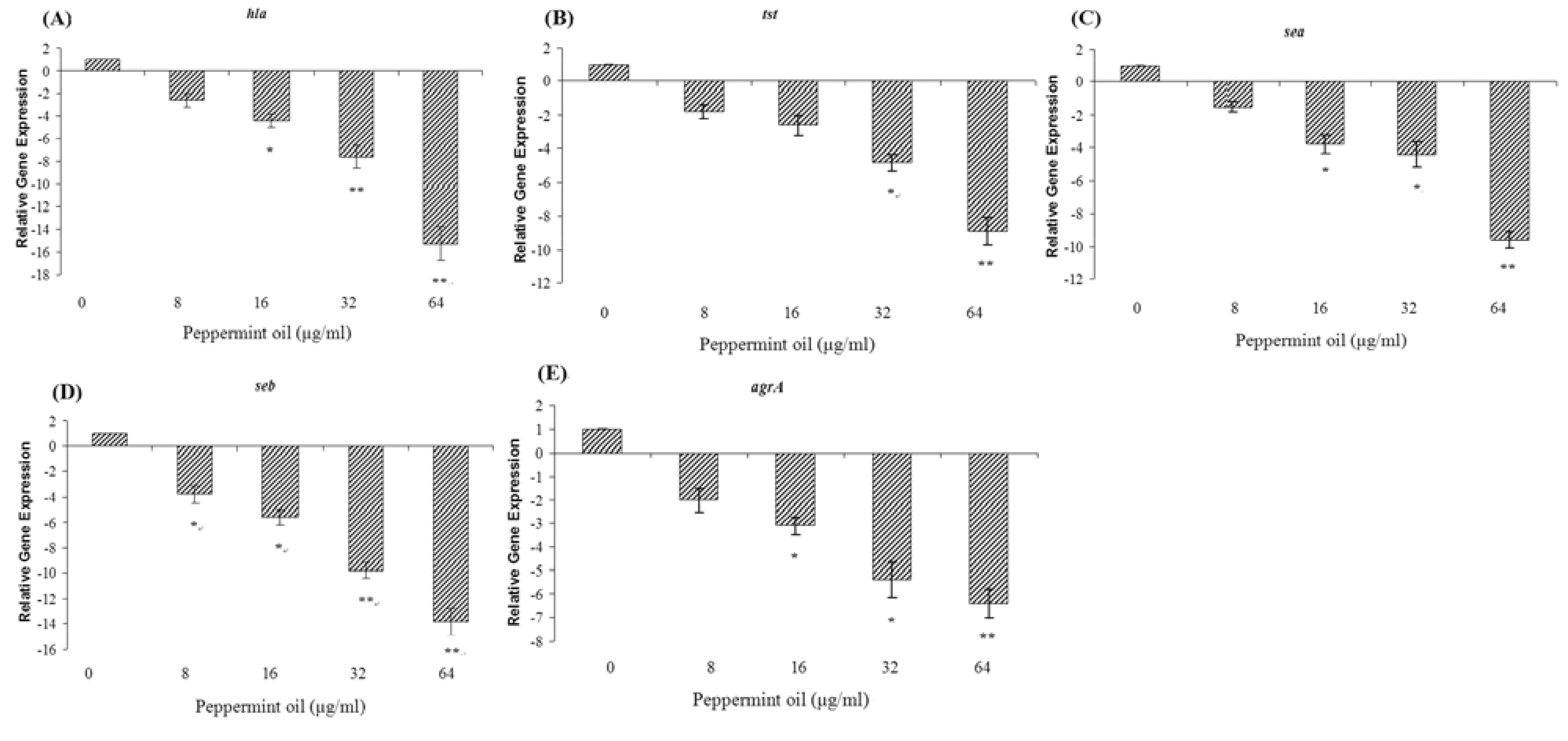
2.5. Peppermint oil inhibits the transcription of hla, tst, sea, seb, and agrA by S. aureus
2.6. Discussion
3. Experimental
3.1. Bacterial strains and reagents
3.2. Plant material and essential oil extraction
3.3. Gas chromatography/mass spectral analysis
3.4. MIC determination
3.5. Growth curves
3.6. Growth conditions
3.7. Western blot analysis
3.8. Hemolysin assay
3.9. Quantitative RT-PCR
3.10. Statistical analysis
4. Conclusions
Acknowledgements
References and Notes
- Boyle-Vavra, S.; Daum, R.S. Community-acquired methicillin-resistant Staphylococcus aureus: the role of Panton-Valentine leukocidin. Lab. Invest. 2007, 87, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S.D.; DeLeo, F.R. An update on community-associated MRSA virulence. Curr. Opin. Pharmacol. 2009, 9, 545–551. [Google Scholar] [CrossRef] [PubMed]
- Schlievert, P.M.; Strandberg, K.L.; Lin, Y.C.; Peterson, M.L.; Leung, D.Y. Secreted virulence factor comparison between methicillin-resistant and methicillin-sensitive Staphylococcus aureus, and its relevance to atopic dermatitis. J. Allergy Clin. Immunol. 2010, 125, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Itakura, J.; Watanabe, M.; Ohta, M.; Sato, Y.; Yamaya, Y. Inactivation of staphylococcal enterotoxin-A with an electrolyzed anodic solution. J. Agric. Food Chem. 2002, 50, 230–234. [Google Scholar] [CrossRef] [PubMed]
- Rasooly, R.; Do, P.M.; Friedman, M. Inhibition of biological activity of staphylococcal enterotoxin A (SEA) by apple juice and apple polyphenols. J. Agric. Food Chem. 2010, 58, 5421–5426. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.C.; Schlievert, P.M.; Anderson, M.J.; Fair, C.L.; Schaefers, M.M.; Muthyala, R.; Peterson, M.L. Glycerol monolaurate and dodecylglycerol effects on Staphylococcus aureus and toxic shock syndrome toxin-1 in vitro and in vivo. PLoS One 2009, 9, e7499. [Google Scholar] [CrossRef] [PubMed]
- Dinges, M.M.; Orwin, P.M.; Schlievert, P.M. Exotoxins of Staphylococcus aureus. Clin. Microbiol. Rev. 2000, 13, 16–34. [Google Scholar] [CrossRef] [PubMed]
- Grigoleit, H.G.; Grigoleit, P. Gastrointestinal clinical pharmacology of peppermint oil. Phytomedicine 2005, 12, 607–611. [Google Scholar] [CrossRef] [PubMed]
- Beesley, A.; Hardcastle, J.; Hardcastle, P.T.; Taylor, C.J. Influence of peppermint oil on absorptive and secretory processes in rat small intestine. Gut 1996, 39, 214–219. [Google Scholar] [CrossRef] [PubMed]
- Charrois, T.L.; Hrudey, J.; Gardiner, P.; Vohra, S. Peppermint oil. Pediatr. Rev. 2006, 27, e49–e51. [Google Scholar] [CrossRef] [PubMed]
- Edris, A.E.; Farrag, E.S. Antifungal activity of peppermint and sweet basil essential oils and their major aroma constituents on some plant pathogenic fungi from the vapor phase. Nahrung 2003, 47, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Imai, H.; Osawa, K.; Yasuda, H.; Hamashima, H.; Arai, T.; Sasatsu, M. Inhibition by the essential oils of peppermint and spearmint of the growth of pathogenic bacteria. Microbios 2001, 1, 31–39. [Google Scholar]
- Novick, R.P. Autoinduction and signal transduction in the regulation of staphylococcal virulence. Mol. Microbiol. 2003, 48, 1429–1449. [Google Scholar] [CrossRef] [PubMed]
- Cornaglia, G.; Rossolini, G.M. Forthcoming therapeutic perspectives for infections due to multidrug-resistant Gram-positive pathogens. Clin. Microbiol. Infect. 2009, 15, 218–223. [Google Scholar] [CrossRef] [PubMed]
- Smith-Palmer, A.; Stewart, J.; Fyfe, L. Influence of subinhibitory concentrations of plant essential oils on the production of enterotoxins A and B and alpha-toxin by Staphylococcus aureus. J. Med. Microbiol. 2004, 53, 1023–1027. [Google Scholar] [CrossRef] [PubMed]
- Escaich, S. Antivirulence as a new antibacterial approach for chemotherapy. Curr. Opin. Chem. Biol. 2008, 12, 400–408. [Google Scholar] [CrossRef] [PubMed]
- Bernardo, K.; Pakulat, N.; Fleer, S.; Schnaith, A.; Utermöhlen, O.; Krut, O.; Müller, S.; Krönke, M. Subinhibitory concentrations of linezolid reduce Staphylococcus aureus virulence factor expression. Antimicrob. Agents Chemother. 2004, 48, 546–555. [Google Scholar] [CrossRef] [PubMed]
- Cegelski, L.; Marshall, G.R.; Eldridge, G.R.; Hultgren, S.J. The biology and future prospects of antivirulence therapies. Nat. Rev. Microbiol. 2008, 6, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Herbert, S.; Barry, P.; Novick, R.P. Subinhibitory clindamycin differentially inhibits transcription of exoprotein genes in Staphylococcus aureus. Infect. Immun. 2001, 69, 2996–3003. [Google Scholar] [CrossRef] [PubMed]
- Koszczol, C.; Bernardo, K.; Krönke, M.; Krut, O. Subinhibitory quinupristin/dalfopristin attenuates virulence of Staphylococcus aureus. J. Antimicrob. Chemother. 2006, 58, 564–574. [Google Scholar] [CrossRef] [PubMed]
- Kluytmans, J.A. Methicillin-resistant Staphylococcus aureus in food products: cause for concern or case for complacency? Clin. Microbiol. Infect. 2010, 16, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Kim, Y.S.; Shin, D.H. Antimicrobial synergistic effect of linolenic acid and monoglyceride against Bacillus cereus and Staphylococcus aureus. J. Agric. Food Chem. 2002, 50, 2193–2199. [Google Scholar] [CrossRef] [PubMed]
- Grigoleit, H.G.; Grigoleit, P. Pharmacology and preclinical pharmacokinetics of peppermint oil. Phytomedicine 2005, 12, 612–616. [Google Scholar] [CrossRef] [PubMed]
- Goerke, C.; Fluckiger, U.; Steinhuber, A.; Zimmerli, W.; Wolz, C. Impact of the regulatory loci agr, sarA and sae of Staphylococcus aureus on the induction of alpha-toxin during device-related infection resolved by direct quantitative transcript analysis. Mol. Microbiol. 2001, 40, 1439–1447. [Google Scholar] [CrossRef] [PubMed]
- Chan, P.F.; Foster, S.J. Role of SarA in virulence determinant production and environmental signal transduction in Staphylococcus aureus. J. Bacteriol. 1998, 180, 6232–6241. [Google Scholar] [PubMed]
- Arvidson, S.; Tegmark, K. Regulation of virulence determinants in Staphylococcus aureus. Int. J. Food Microbiol. 2001, 291, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.Q.; Lei, J.C.; Yu, H.; Cai, X.; Zou, G.L. Chemical composition and antimicrobial activity of the essential oil of Scutellaria barbata. Phytochemistry 2004, 65, 881–884. [Google Scholar] [CrossRef] [PubMed]
- Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing; Fifteenth informational supplement, CLSI/NCCLS document M100-S15; CLSI: Wayne, PA, USA, 2005. [Google Scholar]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature (London) 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Qiu, J.; Wang, D.; Xiang, H.; Feng, H.; Jiang, Y.; Xia, L.; Dong, J.; Lu, J.; Yu, L.; Deng, X. Subinhibitory concentrations of thymol reduce enterotoxins A and B and alpha-hemolysin production in Staphylococcus aureus isolates. PLoS One 2010, 5, e9736. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.; Jiang, Y.; Xia, L.; Xiang, H.; Feng, H.; Pu, S.; Huang, N.; Yu, L.; Deng, X. Subinhibitory concentrations of licochalcone A decrease alpha-toxin production in both methicillin-sensitive and methicillin-resistant Staphylococcus aureus isolates. Lett. Appl. Microbiol. 2010, 50, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Xiang, H.; Qiu, J.Z.; Wang, D.C.; Jiang, Y.S.; Xia, L.J.; Deng, X.M. Influence of magnolol on the secretion of alpha-toxin by Staphylococcus aureus. Molecules 2010, 15, 1679–1689. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |


| Compound | Retention time (min) | Relative contents (%) |
|---|---|---|
| α-Pinene | 4.383 | 1.72 |
| β-Pinene | 5.017 | 1.17 |
| 3-Octanol | 5.150 | 1.5 |
| Limonene | 5.717 | 0.9 |
| Eucalyptol | 5.792 | 0.3 |
| Isopulegol | 7.575 | 1.5 |
| Menthone | 7.692 | 15.98 |
| Isomenthone | 7.842 | 7.89 |
| Neomenthol | 7.900 | 5.96 |
| Menthol | 8.017 | 48.44 |
| p-Menth-1-en-8-ol | 8.283 | 0.63 |
| n-Valeric acid cis-3-hexenyl ester | 8.767 | 0.3 |
| Pulegone | 8.958 | 1.29 |
| 2-Cyclohexen-1-one | 9.033 | 1.55 |
| 1-Decanol | 9.200 | 0.49 |
| Menthyl acetate | 9.642 | 3.17 |
| γ-Elemene | 11.150 | 0.41 |
| Caryophyllene | 11.683 | 0.44 |
| Caryophyllene oxide | 13.900 | 0.54 |
| Organism | Antimicrobial agent | MIC (μg/mL)a | ||
|---|---|---|---|---|
| Range | 50% | 90% | ||
| MSSA (8) | Peppermint oil | 64-128 | 64 | 128 |
| Oxacillin | 0.5-4 | 2 | 2 | |
| Gentamycin | 0.06-32 | 2 | 4 | |
| MRSA (20) | Peppermint oil | 64-256 | 128 | 128 |
| Oxacillin | 16->256 | 128 | 256 | |
| Gentamycin | 0.125-128 | 8 | 32 | |
| S. aureus strain | Hemolysis (%) of rabbit erythrocytes by culture supernatanta | ||||
|---|---|---|---|---|---|
| 0 | 8 µg/mL | 16 µg/mL | 32 µg/mL | 64 µg/mL | |
| ATCC 29213 | 100% | 76.4% ± 5.2 | 37.3% ± 7.2* | 24.2% ± 6.5** | noneb |
| MRSA 2985 | 100% | 81.5% ± 7.2 | 34.6% ± 5.3* | 18.6% ± 4.4** | none |
| MRSA 3701 | 100% | 70.4% ± 8.3 | 25.3% ± 5.8** | 10.8% ± 3.2** | none |
| Primer | Sequence | Genomic Location |
|---|---|---|
| 16S rRNA-fw | 5'-GCTGCCCTTTGTATTGTC-3' | 287-305 |
| 16S rRNA-rv | 5'-AGATGTTGGGTTAAGTCCC-3' | 446-465 |
| hla –fw | 5'-TTGGTGCAAATGTTTC-3' | 485-501 |
| hla-rv | 5'-TCACTTTCCAGCCTACT-3' | 569-586 |
| sea-fw | 5'-ATGGTGCTTATTATGGTTATC-3' | 335-356 |
| sea-rv | 5'-CGTTTCCAAAGGTACTGTATT-3' | 477-498 |
| seb-fw | 5'-TGTTCGGGTATTTGAAGATGG-3' | 480-501 |
| seb-rv | 5'-CGTTTCATAAGGCGAGTTGTT-3' | 612-633 |
| tst-fw | 5'-ACCCCTGTTCCCTTATCATC-3' | 73-93 |
| tst-rv | 5'-AAAAGCGTCAGACCCACTAC-3' | 159-180 |
| agrA- fw | 5'-TGATAATCCTTATGAGGTGCTT-3' | 111-133 |
| agrA- rv | 5'-CACTGTGACTCGTAACGAAAA-3' | 253-274 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, J.; Dong, J.; Qiu, J.-Z.; Wang, J.-F.; Luo, M.-J.; Li, H.-E.; Leng, B.-F.; Ren, W.-Z.; Deng, X.-M. Peppermint Oil Decreases the Production of Virulence-Associated Exoproteins by Staphylococcus aureus. Molecules 2011, 16, 1642-1654. https://doi.org/10.3390/molecules16021642
Li J, Dong J, Qiu J-Z, Wang J-F, Luo M-J, Li H-E, Leng B-F, Ren W-Z, Deng X-M. Peppermint Oil Decreases the Production of Virulence-Associated Exoproteins by Staphylococcus aureus. Molecules. 2011; 16(2):1642-1654. https://doi.org/10.3390/molecules16021642
Chicago/Turabian StyleLi, Jing, Jing Dong, Jia-Zhang Qiu, Jian-Feng Wang, Ming-Jing Luo, Hong-En Li, Bing-Feng Leng, Wen-Zhi Ren, and Xu-Ming Deng. 2011. "Peppermint Oil Decreases the Production of Virulence-Associated Exoproteins by Staphylococcus aureus" Molecules 16, no. 2: 1642-1654. https://doi.org/10.3390/molecules16021642
APA StyleLi, J., Dong, J., Qiu, J.-Z., Wang, J.-F., Luo, M.-J., Li, H.-E., Leng, B.-F., Ren, W.-Z., & Deng, X.-M. (2011). Peppermint Oil Decreases the Production of Virulence-Associated Exoproteins by Staphylococcus aureus. Molecules, 16(2), 1642-1654. https://doi.org/10.3390/molecules16021642




