Inhibitors of Microglial Neurotoxicity: Focus on Natural Products
Abstract
:1. Introduction
2. Natural Products and Their Active Constituents as Inhibitors of Microglia-Mediated Neurotoxicity
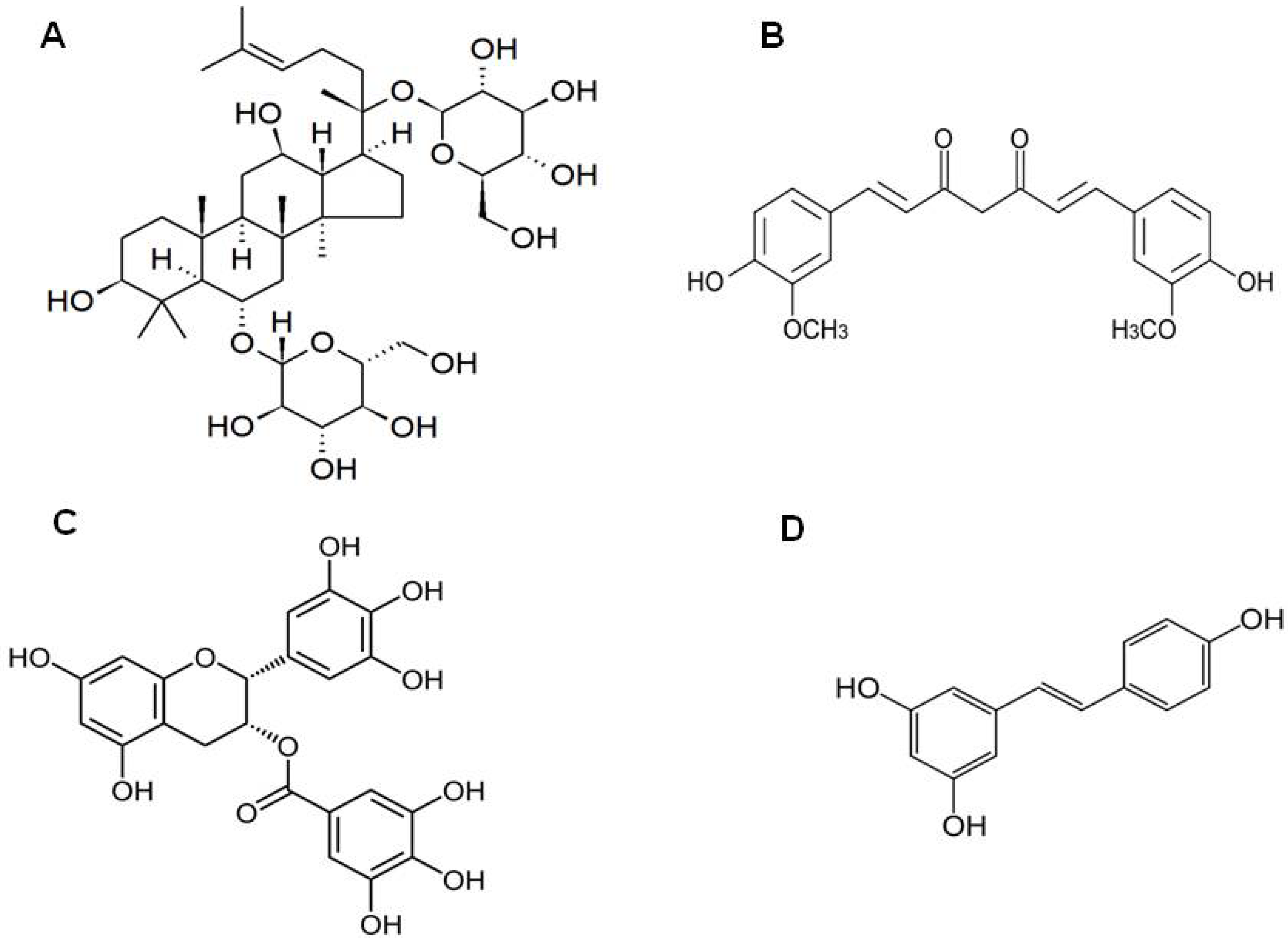
3. Ginsenosides from Panax ginseng
4. Curcumin from Curcuma longa
5. Epigallocatechin-3-Gallate (EGCG) from Camellia sinensis
6. Resveratrol
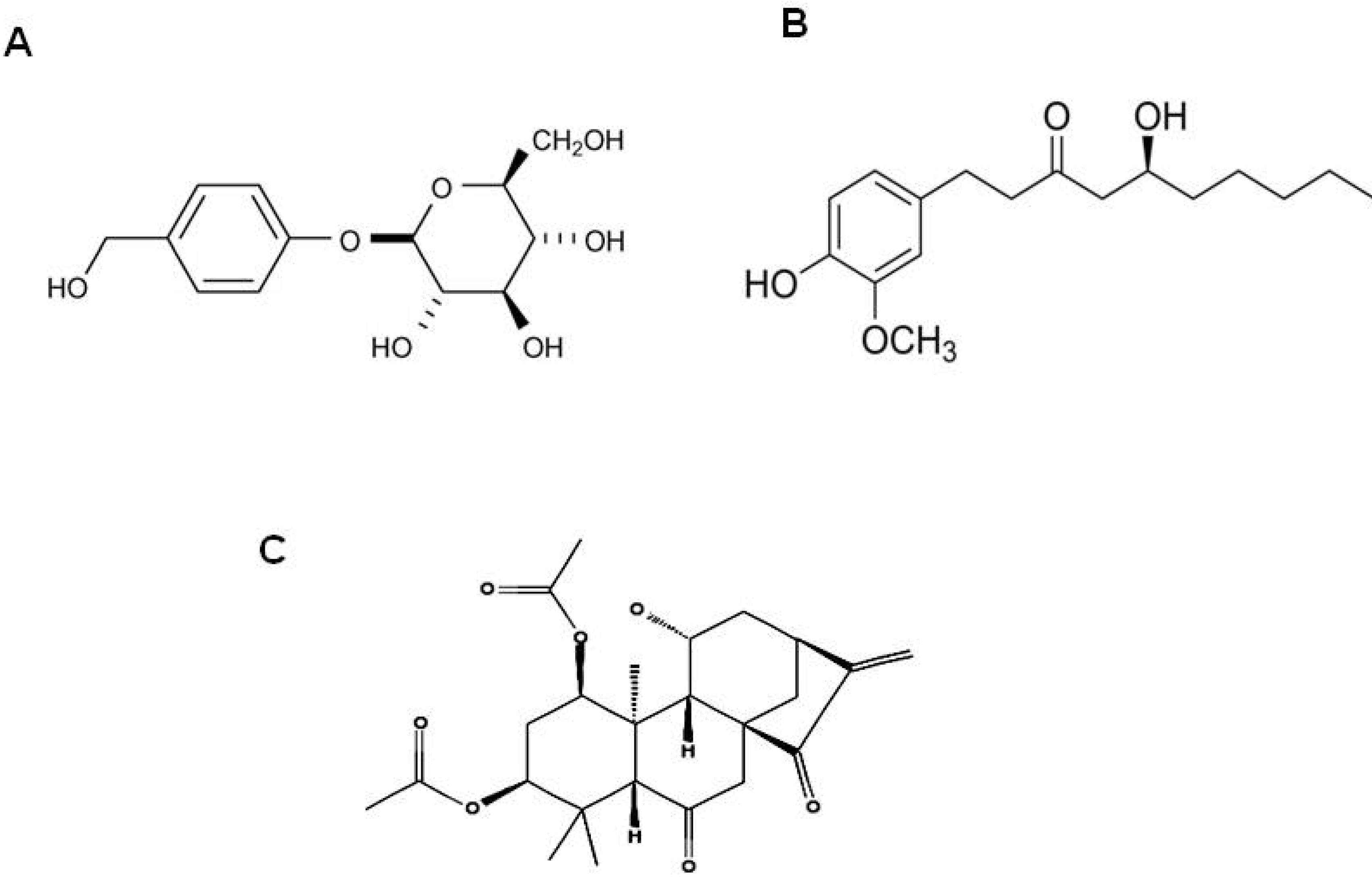
7. Gastrodin from Gastrodia Elata
8. Gingerol from Zingiber officinale
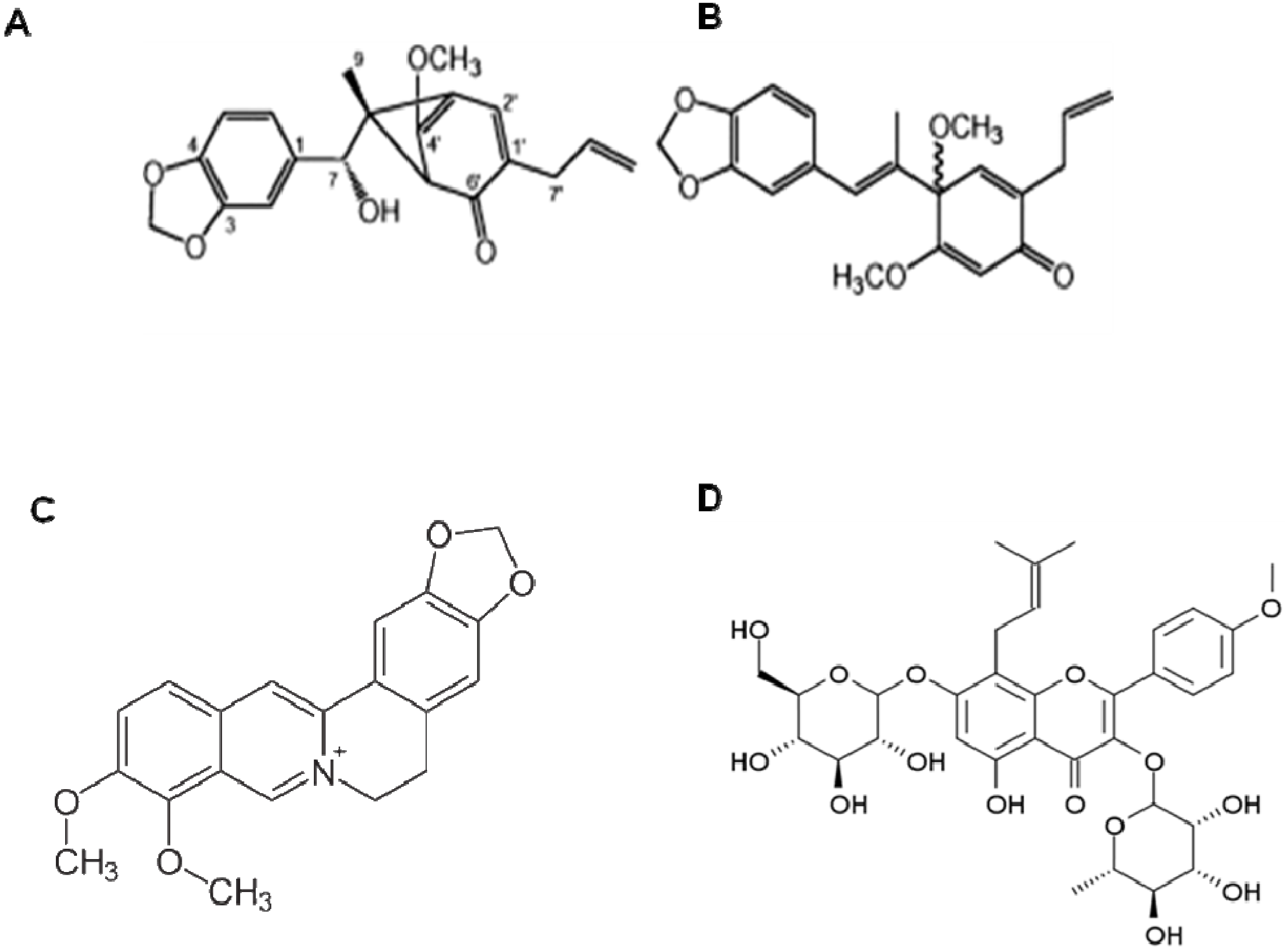
9. Obovatol from Magnolia Obovata
10. Other Natural Products and Components
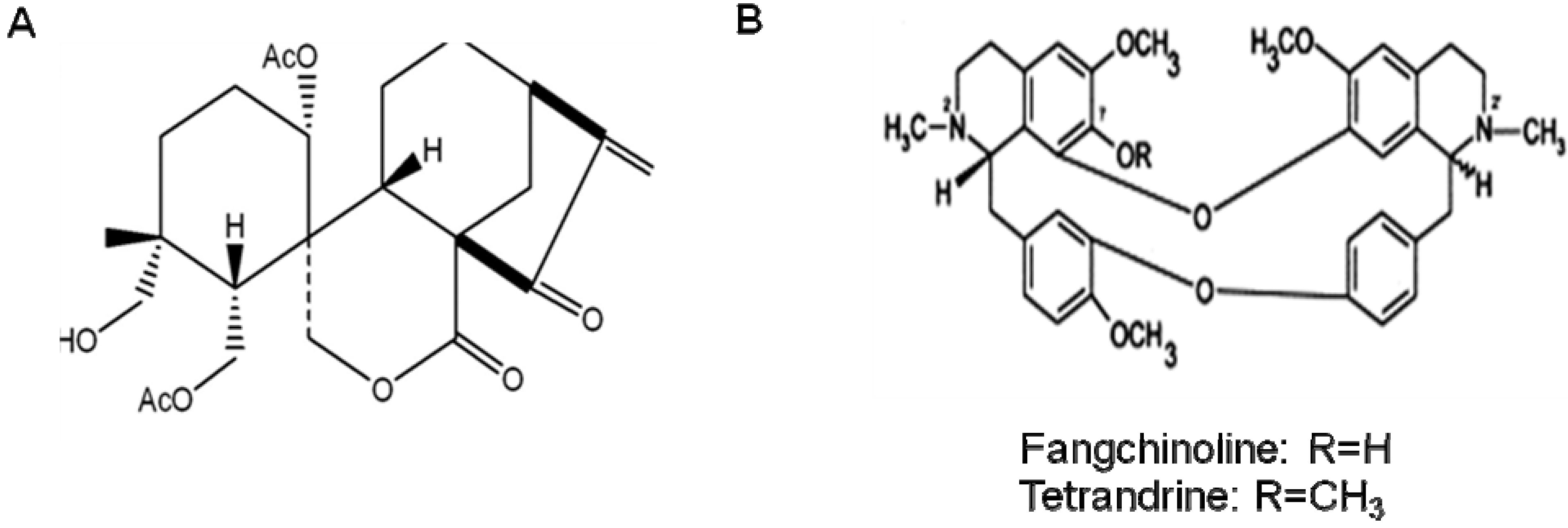
11. Conclusions
Acknowledgements
References
- Boje, K.M.; Arora, P.K. Microglial-produced nitric oxide and reactive nitrogen oxides mediate neuronal cell death. Brain Res. 1992, 587, 250–256. [Google Scholar] [CrossRef]
- Chao, C.C.; Hu, S.; Ehrlich, L.; Peterson, P.K. Interleukin-1 and tumor necrosis factor-alpha synergistically mediate neurotoxicity: involvement of nitric oxide and of N-methyl-D-aspartate receptors. Brain Behav. Immun. 1995, 9, 355–365. [Google Scholar] [CrossRef]
- Chao, C.C.; Hu, S.; Molitor, T.W.; Shaskan, E.G.; Peterson, P.K. Activated microglia mediate neuronal cell injury via a nitric oxide mechanism. J. Immunol. 1992, 149, 2736–2741. [Google Scholar]
- Jeohn, G.H.; Kong, L.Y.; Wilson, B.; Hudson, P.; Hong, J.S. Synergistic neurotoxic effects of combined treatments with cytokines in murine primary mixed neuron/glia cultures. J. Neuroimmunol. 1998, 85, 1–10. [Google Scholar] [CrossRef]
- McGuire, S.O.; Ling, Z.D.; Lipton, J.W.; Sortwell, C.E.; Collier, T.J.; Carvey, P.M. Tumor necrosis factor alpha is toxic to embryonic mesencephalic dopamine neurons. Exp. Neurol. 2001, 169, 219–230. [Google Scholar] [CrossRef]
- Xie, Z.; Wei, M.; Morgan, T.E.; Fabrizio, P.; Han, D.; Finch, C.E.; Longo, V.D. Peroxynitrite mediates neurotoxicity of amyloid beta-peptide1-42- and lipopolysaccharide-activated microglia. J. Neurosci. 2002, 22, 3484–3492. [Google Scholar]
- Glass, C.K.; Saijo, K.; Winner, B.; Marchetto, M.C.; Gage, F.H. Mechanisms underlying inflammation in neurodegeneration. Cell 2010, 140, 918–934. [Google Scholar] [CrossRef]
- Ransohoff, R.M.; Perry, V.H. Microglial physiology: unique stimuli, specialized responses. Annu. Rev. Immunol. 2009, 27, 119–145. [Google Scholar] [CrossRef]
- Cagnin, A.; Myers, R.; Gunn, R.N.; Lawrence, A.D.; Stevens, T.; Kreutzberg, G.W.; Jones, T.; Banati, R.B. In vivo visualization of activated glia by [11C] (R)-PK11195-PET following herpes encephalitis reveals projected neuronal damage beyond the primary focal lesion. Brain 2001, 124, 2014–2027. [Google Scholar] [CrossRef]
- Chen, M.K.; Baidoo, K.; Verina, T.; Guilarte, T.R. Peripheral benzodiazepine receptor imaging in CNS demyelination: functional implications of anatomical and cellular localization. Brain 2004, 127, 1379–1392. [Google Scholar] [CrossRef]
- Turner, M.R.; Cagnin, A.; Turkheimer, F.E.; Miller, C.C.; Shaw, C.E.; Brooks, D.J.; Leigh, P.N.; Banati, R.B. Evidence of widespread cerebral microglial activation in amyotrophic lateral sclerosis: an [11C](R)-PK11195 positron emission tomography study. Neurobiol. Dis. 2004, 15, 601–609. [Google Scholar] [CrossRef]
- Chen, L.W.; Wang, Y.Q.; Wei, L.C.; Shi, M.; Chan, Y.S. Chinese herbs and herbal extracts for neuroprotection of dopaminergic neurons and potential therapeutic treatment of Parkinson's disease. CNS Neurol. Disord. Drug Targets 2007, 6, 273–281. [Google Scholar] [CrossRef]
- Ho, Y.S.; Yu, M.S.; Lai, C.S.; So, K.F.; Yuen, W.H.; Chang, R.C. Characterizing the neuroprotective effects of alkaline extract of Lycium barbarum on beta-amyloid peptide neurotoxicity. Brain Res. 2007, 1158, 123–134. [Google Scholar] [CrossRef]
- Lee, H.; Kim, Y.O.; Kim, H.; Kim, S.Y.; Noh, H.S.; Kang, S.S.; Cho, G.J.; Choi, W.S.; Suk, K. Flavonoid wogonin from medicinal herb is neuroprotective by inhibiting inflammatory activation of microglia. FASEB J. 2003, 17, 1943–1944. [Google Scholar]
- Li, F.Q.; Wang, T.; Pei, Z.; Liu, B.; Hong, J.S. Inhibition of microglial activation by the herbal flavonoid baicalein attenuates inflammation-mediated degeneration of dopaminergic neurons. J. Neural Transm. 2005, 112, 331–347. [Google Scholar] [CrossRef]
- Shimazawa, M.; Chikamatsu, S.; Morimoto, N.; Mishima, S.; Nagai, H.; Hara, H. Neuroprotection by Brazilian Green Propolis against In vitro and In vivo Ischemic Neuronal Damage. Evid. Based Complement. Alternat. Med. 2005, 2, 201–207. [Google Scholar] [CrossRef]
- Suk, K.; Lee, H.; Kang, S.S.; Cho, G.J.; Choi, W.S. Flavonoid baicalein attenuates activation-induced cell death of brain microglia. J. Pharmacol. Exp. Ther. 2003, 305, 638–645. [Google Scholar] [CrossRef]
- Corbit, R.M.; Ferreira, J.F.; Ebbs, S.D.; Murphy, L.L. Simplified extraction of ginsenosides from American ginseng (Panax quinquefolius L.) for high-performance liquid chromatography-ultraviolet analysis. J. Agric. Food Chem. 2005, 53, 9867–9873. [Google Scholar] [CrossRef]
- Kitts, D.D.; Wijewickreme, A.N.; Hu, C. Antioxidant properties of a North American ginseng extract. Mol. Cell. Biochem. 2000, 203, 1–10. [Google Scholar] [CrossRef]
- Yun, T.K. Panax ginseng--a non-organ-specific cancer preventive? Lancet Oncol. 2001, 2, 49–55. [Google Scholar] [CrossRef]
- Park, S.; Yeo, M.; Jin, J.H.; Lee, K.M.; Jung, J.Y.; Choue, R.; Cho, S.W.; Hahm, K.B. Rescue of Helicobacter pylori-induced cytotoxicity by red ginseng. Dig. Dis. Sci. 2005, 50, 1218–1227. [Google Scholar] [CrossRef]
- Volate, S.R.; Davenport, D.M.; Muga, S.J.; Wargovich, M.J. Modulation of aberrant crypt foci and apoptosis by dietary herbal supplements (quercetin, curcumin, silymarin, ginseng and rutin). Carcinogenesis 2005, 26, 1450–1456. [Google Scholar] [CrossRef]
- Kang, K.A.; Kim, Y.W.; Kim, S.U.; Chae, S.; Koh, Y.S.; Kim, H.S.; Choo, M.K.; Kim, D.H.; Hyun, J.W. G1 phase arrest of the cell cycle by a ginseng metabolite, compound K, in U937 human monocytic leukamia cells. Arch. Pharm. Res. 2005, 28, 685–690. [Google Scholar] [CrossRef]
- Kakizoe, T. Asian studies of cancer chemoprevention: latest clinical results. Eur. J. Cancer 2000, 36, 1303–1309. [Google Scholar] [CrossRef]
- Shin, H.R.; Kim, J.Y.; Yun, T.K.; Morgan, G.; Vainio, H. The cancer-preventive potential of Panax ginseng: a review of human and experimental evidence. Cancer Causes Control 2000, 11, 565–576. [Google Scholar] [CrossRef]
- Ichikawa, T.; Li, J.; Nagarkatti, P.; Nagarkatti, M.; Hofseth, L.J.; Windust, A.; Cui, T. American ginseng preferentially suppresses STAT/iNOS signaling in activated macrophages. J. Ethnopharmacol. 2009, 125, 145–150. [Google Scholar] [CrossRef]
- Rhule, A.; Navarro, S.; Smith, J.R.; Shepherd, D.M. Panax notoginseng attenuates LPS-induced pro-inflammatory mediators in RAW264.7 cells. J. Ethnopharmacol. 2006, 106, 121–128. [Google Scholar] [CrossRef]
- Park, J.S.; Park, E.M.; Kim, D.H.; Jung, K.; Jung, J.S.; Lee, E.J.; Hyun, J.W.; Kang, J.L.; Kim, H.S. Anti-inflammatory mechanism of ginseng saponins in activated microglia. J. Neuroimmunol. 2009, 209, 40–49. [Google Scholar] [CrossRef]
- Keum, Y.S.; Han, S.S.; Chun, K.S.; Park, K.K.; Park, J.H.; Lee, S.K.; Surh, Y.J. Inhibitory effects of the ginsenoside Rg3 on phorbol ester-induced cyclooxygenase-2 expression, NF-kappaB activation and tumor promotion. Mutat. Res. 2003, 523-524, 75–85. [Google Scholar] [CrossRef]
- Ahn, J.Y.; Choi, I.S.; Shim, J.Y.; Yun, E.K.; Yun, Y.S.; Jeong, G.; Song, J.Y. The immunomodulator ginsan induces resistance to experimental sepsis by inhibiting Toll-like receptor-mediated inflammatory signals. Eur. J. Immunol. 2006, 36, 37–45. [Google Scholar] [CrossRef]
- Lin, W.M.; Zhang, Y.M.; Moldzio, R.; Rausch, W.D. Ginsenoside Rd attenuates neuroinflammation of dopaminergic cells in culture. J. Neural Transm. Suppl. 2007, 105–112. [Google Scholar]
- Zhang, B.; Hata, R.; Zhu, P.; Sato, K.; Wen, T.C.; Yang, L.; Fujita, H.; Mitsuda, N.; Tanaka, J.; Samukawa, K.; Maeda, N.; Sakanaka, M. Prevention of ischemic neuronal death by intravenous infusion of a ginseng saponin, ginsenoside Rb(1), that upregulates Bcl-x(L) expression. J. Cereb. Blood Flow Metab. 2006, 26, 708–721. [Google Scholar] [CrossRef]
- Pendurthi, U.R.; Williams, J.T.; Rao, L.V. Inhibition of tissue factor gene activation in cultured endothelial cells by curcumin. Suppression of activation of transcription factors Egr-1, AP-1, and NF-kappa B. Arterioscler. Thromb. Vasc. Biol. 1997, 17, 3406–3413. [Google Scholar] [CrossRef]
- Weber, W.M.; Hunsaker, L.A.; Gonzales, A.M.; Heynekamp, J.J.; Orlando, R.A.; Deck, L.M.; Vander Jagt, D.L. TPA-induced up-regulation of activator protein-1 can be inhibited or enhanced by analogs of the natural product curcumin. Biochem. Pharmacol. 2006, 72, 928–940. [Google Scholar] [CrossRef]
- Hanai, H.; Iida, T.; Takeuchi, K.; Watanabe, F.; Maruyama, Y.; Andoh, A.; Tsujikawa, T.; Fujiyama, Y.; Mitsuyama, K.; Sata, M.; Yamada, M.; Iwaoka, Y.; Kanke, K.; Hiraishi, H.; Hirayama, K.; Arai, H.; Yoshii, S.; Uchijima, M.; Nagata, T.; Koide, Y. Curcumin maintenance therapy for ulcerative colitis: randomized, multicenter, double-blind, placebo-controlled trial. Clin. Gastroenterol. Hepatol. 2006, 4, 1502–1506. [Google Scholar] [CrossRef]
- He, L.F.; Chen, H.J.; Qian, L.H.; Chen, G.Y.; Buzby, J.S. Curcumin protects pre-oligodendrocytes from activated microglia in vitro and in vivo. Brain Res. 2010, 1339, 60–69. [Google Scholar] [CrossRef]
- Lim, G.P.; Chu, T.; Yang, F.; Beech, W.; Frautschy, S.A.; Cole, G.M. The curry spice curcumin reduces oxidative damage and amyloid pathology in an Alzheimer transgenic mouse. J. Neurosci. 2001, 21, 8370–8377. [Google Scholar]
- Kim, H.J.; Moon, K.D.; Lee, D.S.; Lee, S.H. Ethyl ether fraction of Gastrodia elata Blume protects amyloid beta peptide-induced cell death. J. Ethnopharmacol. 2003, 84, 95–98. [Google Scholar] [CrossRef]
- Wang, H.M.; Zhao, Y.X.; Zhang, S.; Liu, G.D.; Kang, W.Y.; Tang, H.D.; Ding, J.Q.; Chen, S.D. PPARgamma agonist curcumin reduces the amyloid-beta-stimulated inflammatory responses in primary astrocytes. J. Alzheimers Dis. 2010, 20, 1189–1199. [Google Scholar]
- Cole, G.M.; Teter, B.; Frautschy, S.A. Neuroprotective effects of curcumin. Adv. Exp. Med. Biol. 2007, 595, 197–212. [Google Scholar] [CrossRef]
- Frautschy, S.A.; Hu, W.; Kim, P.; Miller, S.A.; Chu, T.; Harris-White, M.E.; Cole, G.M. Phenolic anti-inflammatory antioxidant reversal of Abeta-induced cognitive deficits and neuropathology. Neurobiol. Aging 2001, 22, 993–1005. [Google Scholar] [CrossRef]
- Garcia-Alloz, M.; Dodwell, L.; Borelli, A.; Raju, S.; Backskai, B. In vivo reduction of plaque size in APPswe/PS1D9 mice treated with curcumin. Alzheimer's Dementia 2006, 2, S617. [Google Scholar]
- Yang, F.; Lim, G.P.; Begum, A.N.; Ubeda, O.J.; Simmons, M.R.; Ambegaokar, S.S.; Chen, P.P.; Kayed, R.; Glabe, C.G.; Frautschy, S.A.; Cole, G.M. Curcumin inhibits formation of amyloid beta oligomers and fibrils, binds plaques, and reduces amyloid in vivo. J. Biol. Chem. 2005, 280, 5892–5901. [Google Scholar]
- Neyestani, T.R.; Gharavi, A.; Kalayi, A. Selective effects of tea extract and its phenolic compounds on human peripheral blood mononuclear cell cytokine secretions. Int. J. Food Sci. Nutr. 2009, 60 (Suppl. 1), 79–88. [Google Scholar]
- Mandel, S.A.; Avramovich-Tirosh, Y.; Reznichenko, L.; Zheng, H.; Weinreb, O.; Amit, T.; Youdim, M.B. Multifunctional activities of green tea catechins in neuroprotection. Modulation of cell survival genes, iron-dependent oxidative stress and PKC signaling pathway. Neurosignals 2005, 14, 46–60. [Google Scholar] [CrossRef]
- Bazinet, L.; Labb, D.; Tremblay, A. Production of green tea EGC- and EGCG-enriched fractions by a two-step extraction procedure. Separation Purification Technol. 2007, 56, 53–56. [Google Scholar] [CrossRef]
- Hu, B.; Wang, L.; Zhou, B.; Zhang, X.; Sun, Y.; Ye, H.; Zhao, L.; Hu, Q.; Wang, G.; Zeng, X. Efficient procedure for isolating methylated catechins from green tea and effective simultaneous analysis of ten catechins, three purine alkaloids, and gallic acid in tea by high-performance liquid chromatography with diode array detection. J. Chromatogr. A 2009, 1216, 3223–3231. [Google Scholar] [CrossRef]
- Perva-Uzunalića, A.; Škergeta, M.; Kneza, Ž.; Weinreichb, B.; Ottob, F.; Grünerb, S. Extraction of active ingredients from green tea (Camellia sinensis): Extraction efficiency of major catechins and caffeine. Food Chem. 2006, 96, 597–605. [Google Scholar] [CrossRef]
- Chen, C.; Yu, R.; Owuor, E.D.; Kong, A.N. Activation of antioxidant-response element (ARE), mitogen-activated protein kinases (MAPKs) and caspases by major green tea polyphenol components during cell survival and death. Arch. Pharm. Res. 2000, 23, 605–612. [Google Scholar] [CrossRef]
- Hou, R.R.; Chen, J.Z.; Chen, H.; Kang, X.G.; Li, M.G.; Wang, B.R. Neuroprotective effects of (-)-epigallocatechin-3-gallate (EGCG) on paraquat-induced apoptosis in PC12 cells. Cell Biol. Int. 2008, 32, 22–30. [Google Scholar] [CrossRef]
- Rezai-Zadeh, K.; Shytle, D.; Sun, N.; Mori, T.; Hou, H.; Jeanniton, D.; Ehrhart, J.; Townsend, K.; Zeng, J.; Morgan, D.; Hardy, J.; Town, T.; Tan, J. Green tea epigallocatechin-3-gallate (EGCG) modulates amyloid precursor protein cleavage and reduces cerebral amyloidosis in Alzheimer transgenic mice. J. Neurosci. 2005, 25, 8807–8814. [Google Scholar]
- Okello, E.J.; Savelev, S.U.; Perry, E.K. In vitro anti-beta-secretase and dual anti-cholinesterase activities of Camellia sinensis L. (tea) relevant to treatment of dementia. Phytother. Res. 2004, 18, 624–627. [Google Scholar] [CrossRef]
- Li, R.; Huang, Y.G.; Fang, D.; Le, W.D. (-)-Epigallocatechin gallate inhibits lipopolysaccharide-induced microglial activation and protects against inflammation-mediated dopaminergic neuronal injury. J. Neurosci. Res. 2004, 78, 723–731. [Google Scholar] [CrossRef]
- Salah, N.; Miller, N.J.; Paganga, G.; Tijburg, L.; Bolwell, G.P.; Rice-Evans, C. Polyphenolic flavanols as scavengers of aqueous phase radicals and as chain-breaking antioxidants. Arch. Biochem. Biophys. 1995, 322, 339–346. [Google Scholar] [CrossRef]
- LI, Y.; Ouyang, C.; Shu, S. Protective Effect of EGCG on Focal Cerebral Ischemia-Reperfusion Injury in Rats. Journal of Xianning College(Medical Sciences) 2006, 20, 373–376. [Google Scholar]
- Yang, S.; Zhu, G.; Xie, D. Epigallocatechin Gallate Ameliorates Accumulated β-amyloid Protein Caused by Oxidative Free Radicals in Cortex Neuron in vitro. J. Clinical Res. 2007, 24, 1065–1067. [Google Scholar]
- Mandel, S.A.; Amit, T.; Kalfon, L.; Reznichenko, L.; Weinreb, O.; Youdim, M.B. Cell signaling pathways and iron chelation in the neurorestorative activity of green tea polyphenols: special reference to epigallocatechin gallate (EGCG). J. Alzheimers Dis. 2008, 15, 211–222. [Google Scholar]
- Aktas, O.; Prozorovski, T.; Smorodchenko, A.; Savaskan, N.E.; Lauster, R.; Kloetzel, P.M.; Infante-Duarte, C.; Brocke, S.; Zipp, F. Green tea epigallocatechin-3-gallate mediates T cellular NF-kappa B inhibition and exerts neuroprotection in autoimmune encephalomyelitis. J. Immunol. 2004, 173, 5794–5800. [Google Scholar]
- Rietveld, A.; Wiseman, S. Antioxidant effects of tea: evidence from human clinical trials. J. Nutr. 2003, 133, 3285S–3292S. [Google Scholar]
- Baur, J.A.; Sinclair, D.A. Therapeutic potential of resveratrol: the in vivo evidence. Nat. Rev. Drug Discov. 2006, 5, 493–506. [Google Scholar]
- Bradamante, S.; Barenghi, L.; Villa, A. Cardiovascular protective effects of resveratrol. Cardiovasc. Drug Rev. 2004, 22, 169–188. [Google Scholar]
- Jang, M.; Cai, L.; Udeani, G.O.; Slowing, K.V.; Thomas, C.F.; Beecher, C.W.; Fong, H.H.; Farnsworth, N.R.; Kinghorn, A.D.; Mehta, R.G.; Moon, R.C.; Pezzuto, J.M. Cancer chemopreventive activity of resveratrol, a natural product derived from grapes. Science 1997, 275, 218–220. [Google Scholar]
- Das, S.; Das, D.K. Anti-inflammatory responses of resveratrol. Inflamm. Allergy Drug Targets 2007, 6, 168–173. [Google Scholar] [CrossRef]
- Bi, X.L.; Yang, J.Y.; Dong, Y.X.; Wang, J.M.; Cui, Y.H.; Ikeshima, T.; Zhao, Y.Q.; Wu, C.F. Resveratrol inhibits nitric oxide and TNF-alpha production by lipopolysaccharide-activated microglia. Int. Immunopharmacol. 2005, 5, 185–193. [Google Scholar] [CrossRef]
- Meng, X.L.; Yang, J.Y.; Chen, G.L.; Wang, L.H.; Zhang, L.J.; Wang, S.; Li, J.; Wu, C.F. Effects of resveratrol and its derivatives on lipopolysaccharide-induced microglial activation and their structure-activity relationships. Chem. Biol. Interact. 2008, 174, 51–59. [Google Scholar] [CrossRef]
- Candelario-Jalil, E.; de Oliveira, A.C.; Graf, S.; Bhatia, H.S.; Hull, M.; Munoz, E.; Fiebich, B.L. Resveratrol potently reduces prostaglandin E2 production and free radical formation in lipopolysaccharide-activated primary rat microglia. J. Neuroinflammation 2007, 4, 25. [Google Scholar] [CrossRef]
- Lorenz, P.; Roychowdhury, S.; Engelmann, M.; Wolf, G.; Horn, T.F. Oxyresveratrol and resveratrol are potent antioxidants and free radical scavengers: effect on nitrosative and oxidative stress derived from microglial cells. Nitric Oxide 2003, 9, 64–76. [Google Scholar] [CrossRef]
- Wang, Q.; Xu, J.; Rottinghaus, G.E.; Simonyi, A.; Lubahn, D.; Sun, G.Y.; Sun, A.Y. Resveratrol protects against global cerebral ischemic injury in gerbils. Brain Res. 2002, 958, 439–447. [Google Scholar] [CrossRef]
- Okawara, M.; Katsuki, H.; Kurimoto, E.; Shibata, H.; Kume, T.; Akaike, A. Resveratrol protects dopaminergic neurons in midbrain slice culture from multiple insults. Biochem. Pharmacol. 2007, 73, 550–560. [Google Scholar]
- Anekonda, T.S. Resveratrol--a boon for treating Alzheimer's disease? Brain Res. Rev. 2006, 52, 316–326. [Google Scholar] [CrossRef]
- Chen, J.; Zhou, Y.; Mueller-Steiner, S.; Chen, L.F.; Kwon, H.; Yi, S.; Mucke, L.; Gan, L. SIRT1 protects against microglia-dependent amyloid-beta toxicity through inhibiting NF-kappaB signaling. J. Biol. Chem. 2005, 280, 40364–40374. [Google Scholar]
- Parker, J.A.; Arango, M.; Abderrahmane, S.; Lambert, E.; Tourette, C.; Catoire, H.; Neri, C. Resveratrol rescues mutant polyglutamine cytotoxicity in nematode and mammalian neurons. Nat. Genet. 2005, 37, 349–350. [Google Scholar]
- Bournival, J.; Quessy, P.; Martinoli, M.G. Protective effects of resveratrol and quercetin against MPP+ -induced oxidative stress act by modulating markers of apoptotic death in dopaminergic neurons. Cell. Mol. Neurobiol. 2009, 29, 1169–1180. [Google Scholar] [CrossRef]
- Chen, C.Y.; Jang, J.H.; Li, M.H.; Surh, Y.J. Resveratrol upregulates heme oxygenase-1 expression via activation of NF-E2-related factor 2 in PC12 cells. Biochem. Biophys. Res. Commun. 2005, 331, 993–1000. [Google Scholar] [CrossRef]
- Lu, X.; Ji, C.; Xu, H.; Li, X.; Ding, H.; Ye, M.; Zhu, Z.; Ding, D.; Jiang, X.; Ding, X.; Guo, X. Resveratrol-loaded polymeric micelles protect cells from Abeta-induced oxidative stress. Int. J. Pharm. 2009, 375, 89–96. [Google Scholar] [CrossRef]
- Lee, M.K.; Kang, S.J.; Poncz, M.; Song, K.J.; Park, K.S. Resveratrol protects SH-SY5Y neuroblastoma cells from apoptosis induced by dopamine. Exp. Mol. Med. 2007, 39, 376–384. [Google Scholar]
- Tang, W. Gastrodia elata Bl. In Chinese Drugs of Plant Origin: Chemistry, Pharmacology, and Use in Traditional and Modern Medicine; Springer-Verlag: Berlin, Heidelberg, 1992; pp. 545–548. [Google Scholar]
- Zeng, X.; Zhang, S.; Zhang, L.; Zhang, K.; Zheng, X. A study of the neuroprotective effect of the phenolic glucoside gastrodin during cerebral ischemia in vivo and in vitro. Planta Med. 2006, 72, 1359–1365. [Google Scholar] [CrossRef]
- An, H.; Kim, I.S.; Koppula, S.; Kim, B.W.; Park, P.J.; Lim, B.O.; Choi, W.S.; Lee, K.H.; Choi, D.K. Protective effects of Gastrodia elata Blume on MPP+-induced cytotoxicity in human dopaminergic SH-SY5Y cells. J. Ethnopharmacol. 2010, 130, 290–298. [Google Scholar] [CrossRef]
- Huang, N.K.; Chern, Y.; Fang, J.M.; Lin, C.I.; Chen, W.P.; Lin, Y.L. Neuroprotective principles from Gastrodia elata. J. Nat. Prod. 2007, 70, 571–574. [Google Scholar] [CrossRef]
- Kim, S.T.; Kim, J.D.; Lyu, Y.S.; Lee, M.Y.; Kang, H.W. Neuroprotective effect of some plant extracts in cultured CT105-induced PC12 cells. Biol. Pharm. Bull. 2006, 29, 2021–2024. [Google Scholar] [CrossRef]
- Jung, J.W.; Yoon, B.H.; Oh, H.R.; Ahn, J.H.; Kim, S.Y.; Park, S.Y.; Ryu, J.H. Anxiolytic-like effects of Gastrodia elata and its phenolic constituents in mice. Biol. Pharm. Bull. 2006, 29, 261–265. [Google Scholar] [CrossRef]
- Ahn, E.K.; Jeon, H.J.; Lim, E.J.; Jung, H.J.; Park, E.H. Anti-inflammatory and anti-angiogenic activities of Gastrodia elata Blume. J. Ethnopharmacol. 2007, 110, 476–482. [Google Scholar] [CrossRef]
- Hsieh, C.L.; Chen, C.L.; Tang, N.Y.; Chuang, C.M.; Hsieh, C.T.; Chiang, S.Y.; Lin, J.G.; Hsu, S.F. Gastrodia elata BL mediates the suppression of nNOS and microglia activation to protect against neuronal damage in kainic acid-treated rats. Am. J. Chin. Med. 2005, 33, 599–611. [Google Scholar] [CrossRef]
- Huang, N.K.; Lin, Y.L.; Cheng, J.J.; Lai, W.L. Gastrodia elata prevents rat pheochromocytoma cells from serum-deprived apoptosis: the role of the MAPK family. Life Sci. 2004, 75, 1649–1657. [Google Scholar] [CrossRef]
- Baek, N.I.; Choi, S.Y.; Park, J.K.; Cho, S.W.; Ahn, E.M.; Jeon, S.G.; Lee, B.R.; Bahn, J.H.; Kim, Y.K.; Shon, I.H. Isolation and identification of succinic semialdehyde dehydrogenase inhibitory compound from the rhizome of Gastrodia elata Blume. Arch. Pharm. Res. 1999, 22, 219–224. [Google Scholar] [CrossRef]
- Wu, C.R.; Hsieh, M.T.; Huang, S.C.; Peng, W.H.; Chang, Y.S.; Chen, C.F. Effects of Gastrodia elata and its active constituents on scopolamine-induced amnesia in rats. Planta Med. 1996, 62, 317–321. [Google Scholar] [CrossRef]
- Wu, H.Q.; Xie, L.; Jin, X.N.; Ge, Q.; Jin, H.; Liu, G.Q. The effect of vanillin on the fully amygdala-kindled seizures in the rat. Yao Xue Xue Bao 1989, 24, 482–486. [Google Scholar]
- Hwang, S.M.; Lee, Y.J.; Kang, D.G.; Lee, H.S. Anti-inflammatory effect of Gastrodia elata rhizome in human umbilical vein endothelial cells. Am. J. Chin. Med. 2009, 37, 395–406. [Google Scholar] [CrossRef]
- Lee, Y.J.; Hwang, S.M.; Kang, D.G.; Kim, J.S.; Lee, H.S. Effect of Gastrodia elata on tumor necrosis factor-alpha-induced matrix metalloproteinase activity in endothelial cells. J. Nat. Med. 2009, 63, 463–467. [Google Scholar] [CrossRef]
- Heo, J.C.; Woo, S.U.; Son, M.; Park, J.Y.; Choi, W.S.; Chang, K.T.; Kim, S.U.; Yoon, E.K.; Kim, Y.H.; Shin, H.M.; Lee, S.H. Anti-tumor activity of Gastrodia elata Blume is closely associated with a GTP-Ras-dependent pathway. Oncol. Rep. 2007, 18, 849–853. [Google Scholar]
- Hsieh, M.T.; Wu, C.R.; Chen, C.F. Gastrodin and p-hydroxybenzyl alcohol facilitate memory consolidation and retrieval, but not acquisition, on the passive avoidance task in rats. J. Ethnopharmacol. 1997, 56, 45–54. [Google Scholar] [CrossRef]
- Jung, T.Y.; Suh, S.I.; Lee, H.; Kim, I.S.; Kim, H.J.; Yoo, H.S.; Lee, S.R. Protective effects of several components of Gastrodia elata on lipid peroxidation in gerbil brain homogenates. Phytother. Res. 2007, 21, 960–964. [Google Scholar] [CrossRef]
- Kim, H.J.; Lee, S.R.; Moon, K.D. Ether fraction of methanol extracts of Gastrodia elata, medicinal herb protects against neuronal cell damage after transient global ischemia in gerbils. Phytother. Res. 2003, 17, 909–912. [Google Scholar] [CrossRef]
- Liu, J.; Mori, A. Antioxidant and free radical scavenging activities of Gastrodia elata Bl. and Uncaria rhynchophylla (Miq.) Jacks. Neuropharmacology 1992, 31, 1287–1298. [Google Scholar] [CrossRef]
- Sun, X.F.; Wang, W.; Wang, D.Q.; Du, G.Y. Research progress of neuroprotective mechanisms of Gastrodia elata and its preparation. Zhongguo Zhong Yao Za Zhi 2004, 29, 292–295. [Google Scholar]
- Ojemann, L.M.; Nelson, W.L.; Shin, D.S.; Rowe, A.O.; Buchanan, R.A. Tian ma, an ancient Chinese herb, offers new options for the treatment of epilepsy and other conditions. Epilepsy Behav. 2006, 8, 376–383. [Google Scholar] [CrossRef]
- Afzal, M.; Al-Hadidi, D.; Menon, M.; Pesek, J.; Dhami, M.S. Ginger: an ethnomedical, chemical and pharmacological review. Drug Metabol. Drug Interact. 2001, 18, 159–190. [Google Scholar]
- Awang, D. Ginger. Can. Pharmaceut. J. 1992, 125, 309–311. [Google Scholar]
- Leung, A. Encyclopedia of Common Natural Ingredients Used in Food, Drugs and Cosmetics. In Encyclopedia of Common Natural Ingredients Used in Food, Drugs and Cosmetics; John Wiley and Sons: New York, 1980. [Google Scholar]
- Tripathi, S.; Bruch, D.; Kittur, D.S. Ginger extract inhibits LPS induced macrophage activation and function. BMC Complement. Altern. Med. 2008, 8, 1. [Google Scholar] [CrossRef]
- Grzanna, R.; Phan, P.; Polotsky, A.; Lindmark, L.; Frondoza, C.G. Ginger extract inhibits beta-amyloid peptide-induced cytokine and chemokine expression in cultured THP-1 monocytes. J. Altern. Complement. Med. 2004, 10, 1009–1013. [Google Scholar] [CrossRef]
- Jung, H.W.; Yoon, C.H.; Park, K.M.; Han, H.S.; Park, Y.K. Hexane fraction of Zingiberis Rhizoma Crudus extract inhibits the production of nitric oxide and proinflammatory cytokines in LPS-stimulated BV2 microglial cells via the NF-kappaB pathway. Food Chem. Toxicol. 2009, 47, 1190–1197. [Google Scholar] [CrossRef]
- Kim, S.O.; Kundu, J.K.; Shin, Y.K.; Park, J.H.; Cho, M.H.; Kim, T.Y.; Surh, Y.J. [6]-Gingerol inhibits COX-2 expression by blocking the activation of p38 MAP kinase and NF-kappaB in phorbol ester-stimulated mouse skin. Oncogene 2005, 24, 2558–2567. [Google Scholar] [CrossRef]
- Tripathi, S.; Maier, K.G.; Bruch, D.; Kittur, D.S. Effect of 6-gingerol on pro-inflammatory cytokine production and costimulatory molecule expression in murine peritoneal macrophages. J. Surg. Res. 2007, 138, 209–213. [Google Scholar] [CrossRef]
- Lee, S.K.; Kim, H.N.; Kang, Y.R.; Lee, C.W.; Kim, H.M.; Han, D.C.; Shin, J.; Bae, K.; Kwon, B.M. Obovatol inhibits colorectal cancer growth by inhibiting tumor cell proliferation and inducing apoptosis. Bioorg. Med. Chem. 2008, 16, 8397–8402. [Google Scholar]
- Pyo, M.K.; Lee, Y.; Yun-Choi, H.S. Anti-platelet effect of the constituents isolated from the barks and fruits of Magnolia obovata. Arch. Pharm. Res. 2002, 25, 325–328. [Google Scholar] [CrossRef]
- Ito, K.; Iida, T.; Ichino, K.; Tsunezuka, M.; Hattori, M.; Namba, T. Obovatol and obovatal, novel biphenyl ether lignans from the leaves of Magnolia obovata Thunb. Chem. Pharm. Bull. (Tokyo) 1982, 30, 3347–3353. [Google Scholar] [CrossRef]
- Choi, M.S.; Lee, S.H.; Cho, H.S.; Kim, Y.; Yun, Y.P.; Jung, H.Y.; Jung, J.K.; Lee, B.C.; Pyo, H.B.; Hong, J.T. Inhibitory effect of obovatol on nitric oxide production and activation of NF-kappaB/MAP kinases in lipopolysaccharide-treated RAW 264.7cells. Eur. J. Pharmacol. 2007, 556, 181–189. [Google Scholar] [CrossRef]
- Seo, J.J.; Lee, S.H.; Lee, Y.S.; Kwon, B.M.; Ma, Y.; Hwang, B.Y.; Hong, J.T.; Oh, K.W. Anxiolytic-like effects of obovatol isolated from Magnolia obovata: involvement of GABA/benzodiazepine receptors complex. Prog. Neuropsychopharmacol. Biol. Psychiatry 2007, 31, 1363–1369. [Google Scholar] [CrossRef]
- Ock, J.; Han, H.S.; Hong, S.H.; Lee, S.Y.; Han, Y.M.; Kwon, B.M.; Suk, K. Obovatol attenuates microglia-mediated neuroinflammation by modulating redox regulation. Br. J. Pharmacol. 2010, 159, 1646–1662. [Google Scholar] [CrossRef]
- Jung, B.; Shin, M. Encyclopedia of illustrated Korea Natural Drugs. In Encyclopedia of illustrated Korea Natural Drugs; Yong Lim Sa: Seoul, 1990; pp. 845–846. [Google Scholar]
- Lee, C. Illustrated Flora of Korea. In Illustrated Flora of Korea; Hyang Moon Sa: Seoul, 1989; p. 661. [Google Scholar]
- Hong, S.S.; Lee, S.A.; Han, X.H.; Jin, H.Z.; Lee, J.H.; Lee, D.; Lee, J.J.; Hong, J.T.; Kim, Y.; Ro, J.S.; Hwang, B.Y. Kaurane diterpenoids from Isodon excisus inhibit LPS-induced NF-kappaB activation and NO production in macrophage RAW264.7 cells. J. Nat. Prod. 2007, 70, 632–636. [Google Scholar] [CrossRef]
- Ko, H.M.; Koppula, S.; Kim, B.W.; Kim, I.S.; Hwang, B.Y.; Suk, K.; Park, E.J.; Choi, D.K. Inflexin attenuates proinflammatory responses and nuclear factor-kappaB activation in LPS-treated microglia. Eur. J. Pharmacol. 2010, 633, 98–106. [Google Scholar] [CrossRef]
- Kim, H.S.; Lim, J.Y.; Sul, D.; Hwang, B.Y.; Won, T.J.; Hwang, K.W.; Park, S.Y. Neuroprotective effects of the new diterpene, CBNU06 against beta-amyloid-induced toxicity through the inhibition of NF-kappaB signaling pathway in PC12 cells. Eur. J. Pharmacol. 2009, 622, 25–31. [Google Scholar] [CrossRef]
- Zhang, R.; Xu, S.; Cai, Y.; Zhou, M.; Zuo, X.; Chan, P. Ganoderma lucidum Protects Dopaminergic Neuron Degeneration Through Inhibition of Microglial Activation. Evid. Based Complement. Alternat. Med. 2009.
- Neto, C.C. Cranberry and blueberry: evidence for protective effects against cancer and vascular diseases. Mol. Nutr. Food Res. 2007, 51, 652–664. [Google Scholar] [CrossRef]
- Seeram, N.P.; Adams, L.S.; Zhang, Y.; Lee, R.; Sand, D.; Scheuller, H.S.; Heber, D. Blackberry, black raspberry, blueberry, cranberry, red raspberry, and strawberry extracts inhibit growth and stimulate apoptosis of human cancer cells in vitro. J. Agric. Food Chem. 2006, 54, 9329–9339. [Google Scholar] [CrossRef]
- Zhu, Y.; Bickford, P.C.; Sanberg, P.; Giunta, B.; Tan, J. Blueberry opposes beta-amyloid peptide-induced microglial activation via inhibition of p44/42 mitogen-activation protein kinase. Rejuvenation Res. 2008, 11, 891–901. [Google Scholar] [CrossRef]
- Lu, D.Y.; Tang, C.H.; Chen, Y.H.; Wei, I.H. Berberine suppresses neuroinflammatory responses through AMP-activated protein kinase activation in BV-2 microglia. J. Cell. Biochem. 2010, 110, 697–705. [Google Scholar] [CrossRef]
- Wang, Y.K.; Huang, Z.Q. Protective effects of icariin on human umbilical vein endothelial cell injury induced by H2O2 in vitro. Pharmacol. Res. 2005, 52, 174–182. [Google Scholar] [CrossRef]
- Liang, H.R.; Vuorela, P.; Vuorela, H.; Hiltunen, R. Isolation and immunomodulatory effect of flavonol glycosides from Epimedium hunanense. Planta Med. 1997, 63, 316–319. [Google Scholar] [CrossRef]
- Chung, B.H.; Kim, J.D.; Kim, C.K.; Kim, J.H.; Won, M.H.; Lee, H.S.; Dong, M.S.; Ha, K.S.; Kwon, Y.G.; Kim, Y.M. Icariin stimulates angiogenesis by activating the MEK/ERK- and PI3K/Akt/eNOS-dependent signal pathways in human endothelial cells. Biochem. Biophys. Res. Commun. 2008, 376, 404–408. [Google Scholar] [CrossRef]
- Ding, L.; Liang, X.G.; Hu, Y.; Zhu, D.Y.; Lou, Y.J. Involvement of p38MAPK and reactive oxygen species in icariin-induced cardiomyocyte differentiation of murine embryonic stem cells in vitro. Stem Cells Dev. 2008, 17, 751–760. [Google Scholar] [CrossRef]
- Wu, J.F.; Dong, J.C.; Xu, C.Q. [Effects of icariin on inflammation model stimulated by lipopolysaccharide in vitro and in vivo]. Zhongguo Zhong Xi Yi Jie He Za Zhi 2009, 29, 330–334. [Google Scholar]
- Liu, M.H.; Sun, J.S.; Tsai, S.W.; Sheu, S.Y.; Chen, M.H. Icariin protects murine chondrocytes from lipopolysaccharide-induced inflammatory responses and extracellular matrix degradation. Nutr. Res. 2010, 30, 57–65. [Google Scholar] [CrossRef]
- Zeng, K.W.; Fu, H.; Liu, G.X.; Wang, X.M. Icariin attenuates lipopolysaccharide-induced microglial activation and resultant death of neurons by inhibiting TAK1/IKK/NF-kappaB and JNK/p38 MAPK pathways. Int. Immunopharmacol. 2010, 10, 668–678. [Google Scholar] [CrossRef]
- Li, B.; Tian, X. Enmein type diterpenoids from Isodon japonica. Phytochemistry 2001, 58, 543–546. [Google Scholar] [CrossRef]
- Hong, S.S.; Lee, S.A.; Han, X.H.; Hwang, J.S.; Lee, C.; Lee, D.; Hong, J.T.; Kim, Y.; Lee, H.; Hwang, B.Y. ent-Kaurane diterpenoids from Isodon japonicus. J. Nat. Prod. 2008, 71, 1055–1058. [Google Scholar] [CrossRef]
- Lim, J.Y.; Won, T.J.; Hwang, B.Y.; Kim, H.R.; Hwang, K.W.; Sul, D.; Park, S.Y. The new diterpene isodojaponin D inhibited LPS-induced microglial activation through NF-kappaB and MAPK signaling pathways. Eur. J. Pharmacol. 2010, 642, 10–18. [Google Scholar] [CrossRef]
- Tak, P.P.; Firestein, G.S. NF-kappaB: a key role in inflammatory diseases. J. Clin. Invest. 2001, 107, 7–11. [Google Scholar] [CrossRef]
- Teh, B.S.; Seow, W.K.; Li, S.Y.; Thong, Y.H. Inhibition of prostaglandin and leukotriene generation by the plant alkaloids tetrandrine and berbamine. Int. J. Immunopharmacol. 1990, 12, 321–326. [Google Scholar] [CrossRef]
- Xue, Y.; Wang, Y.; Feng, D.C.; Xiao, B.G.; Xu, L.Y. Tetrandrine suppresses lipopolysaccharide-induced microglial activation by inhibiting NF-kappaB pathway. Acta Pharmacol. Sin. 2008, 29, 245–251. [Google Scholar] [CrossRef]
- Klingelhoefer, S.; Obertreis, B.; Quast, S.; Behnke, B. Antirheumatic effect of IDS 23, a stinging nettle leaf extract, on in vitro expression of T helper cytokines. J. Rheumatol. 1999, 26, 2517–2522. [Google Scholar]
- Konrad, A.; Mahler, M.; Arni, S.; Flogerzi, B.; Klingelhofer, S.; Seibold, F. Ameliorative effect of IDS 30, a stinging nettle leaf extract, on chronic colitis. Int. J. Colorectal Dis. 2005, 20, 9–17. [Google Scholar] [CrossRef]
- Teucher, T.; Obertreis, B.; Ruttkowski, T.; Schmitz, H. Cytokine secretion in whole blood of healthy subjects following oral administration of Urtica dioica L. plant extract. Arzneimittelforschung 1996, 46, 906–910. [Google Scholar]
- Cheon, B.S.; Kim, Y.H.; Son, K.S.; Chang, H.W.; Kang, S.S.; Kim, H.P. Effects of prenylated flavonoids and biflavonoids on lipopolysaccharide-induced nitric oxide production from the mouse macrophage cell line RAW 264.7. Planta Med. 2000, 66, 596–600. [Google Scholar] [CrossRef]
- Chi, Y.S.; Jong, H.G.; Son, K.H.; Chang, H.W.; Kang, S.S.; Kim, H.P. Effects of naturally occurring prenylated flavonoids on enzymes metabolizing arachidonic acid: cyclooxygenases and lipoxygenases. Biochem. Pharmacol. 2001, 62, 1185–1191. [Google Scholar] [CrossRef]
- Kim, H.K.; Cheon, B.S.; Kim, Y.H.; Kim, S.Y.; Kim, H.P. Effects of naturally occurring flavonoids on nitric oxide production in the macrophage cell line RAW 264.7 and their structure-activity relationships. Biochem. Pharmacol. 1999, 58, 759–765. [Google Scholar]
- Krol, W.; Czuba, Z.P.; Threadgill, M.D.; Cunningham, B.D.; Pietsz, G. Inhibition of nitric oxide (NO.) production in murine macrophages by flavones. Biochem. Pharmacol. 1995, 50, 1031–1035. [Google Scholar] [CrossRef]
- Rezai-Zadeh, K.; Ehrhart, J.; Bai, Y.; Sanberg, P.R.; Bickford, P.; Tan, J.; Shytle, R.D. Apigenin and luteolin modulate microglial activation via inhibition of STAT1-induced CD40 expression. J. Neuroinflammation 2008, 5, 41. [Google Scholar] [CrossRef]
- Sung, B.; Pandey, M.K.; Aggarwal, B.B. Fisetin, an inhibitor of cyclin-dependent kinase 6, down-regulates nuclear factor-kappaB-regulated cell proliferation, antiapoptotic and metastatic gene products through the suppression of TAK-1 and receptor-interacting protein-regulated IkappaBalpha kinase activation. Mol. Pharmacol. 2007, 71, 1703–1714. [Google Scholar] [CrossRef]
- Maher, P.; Akaishi, T.; Abe, K. Flavonoid fisetin promotes ERK-dependent long-term potentiation and enhances memory. Proc. Natl. Acad. Sci. U S A 2006, 103, 16568–16573. [Google Scholar]
- Hanneken, A.; Lin, F.F.; Johnson, J.; Maher, P. Flavonoids protect human retinal pigment epithelial cells from oxidative-stress-induced death. Invest. Ophthalmol. Vis. Sci. 2006, 47, 3164–3177. [Google Scholar] [CrossRef]
- Park, H.H.; Lee, S.; Oh, J.M.; Lee, M.S.; Yoon, K.H.; Park, B.H.; Kim, J.W.; Song, H.; Kim, S.H. Anti-inflammatory activity of fisetin in human mast cells (HMC-1). Pharmacol. Res. 2007, 55, 31–37. [Google Scholar]
- Zheng, L.T.; Ock, J.; Kwon, B.M.; Suk, K. Suppressive effects of flavonoid fisetin on lipopolysaccharide-induced microglial activation and neurotoxicity. Int. Immunopharmacol. 2008, 8, 484–494. [Google Scholar] [CrossRef]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Choi, D.K.; Koppula, S.; Suk, K. Inhibitors of Microglial Neurotoxicity: Focus on Natural Products. Molecules 2011, 16, 1021-1043. https://doi.org/10.3390/molecules16021021
Choi DK, Koppula S, Suk K. Inhibitors of Microglial Neurotoxicity: Focus on Natural Products. Molecules. 2011; 16(2):1021-1043. https://doi.org/10.3390/molecules16021021
Chicago/Turabian StyleChoi, Dong Kug, Sushruta Koppula, and Kyoungho Suk. 2011. "Inhibitors of Microglial Neurotoxicity: Focus on Natural Products" Molecules 16, no. 2: 1021-1043. https://doi.org/10.3390/molecules16021021
APA StyleChoi, D. K., Koppula, S., & Suk, K. (2011). Inhibitors of Microglial Neurotoxicity: Focus on Natural Products. Molecules, 16(2), 1021-1043. https://doi.org/10.3390/molecules16021021






