Comparative Anti-Infectious Bronchitis Virus (IBV) Activity of (-)-Pinene: Effect on Nucleocapsid (N) Protein
Abstract
1. Introduction
2. Results and Discussion
2.1. Effects of (-)-pinenes against IBV by MTT assay
| Compound | CC50a (mM) | TD0b (mM) | IBV (Gray strain) | |
|---|---|---|---|---|
| IC50c (mM) | SId | |||
| (-)-α-Pinene | >10.0 | 7.88 ± 0.06 | 0.98 ± 0.25 | >10.20 |
| (-)-β-Pinene | >10.0 | 6.09 ± 0.31 | 1.32 ± 0.11 | >7.58 |
| Ribavirin | >1.0 | 0.78 ± 0.15 | 0.118 ± 0.02 | >8.47 |
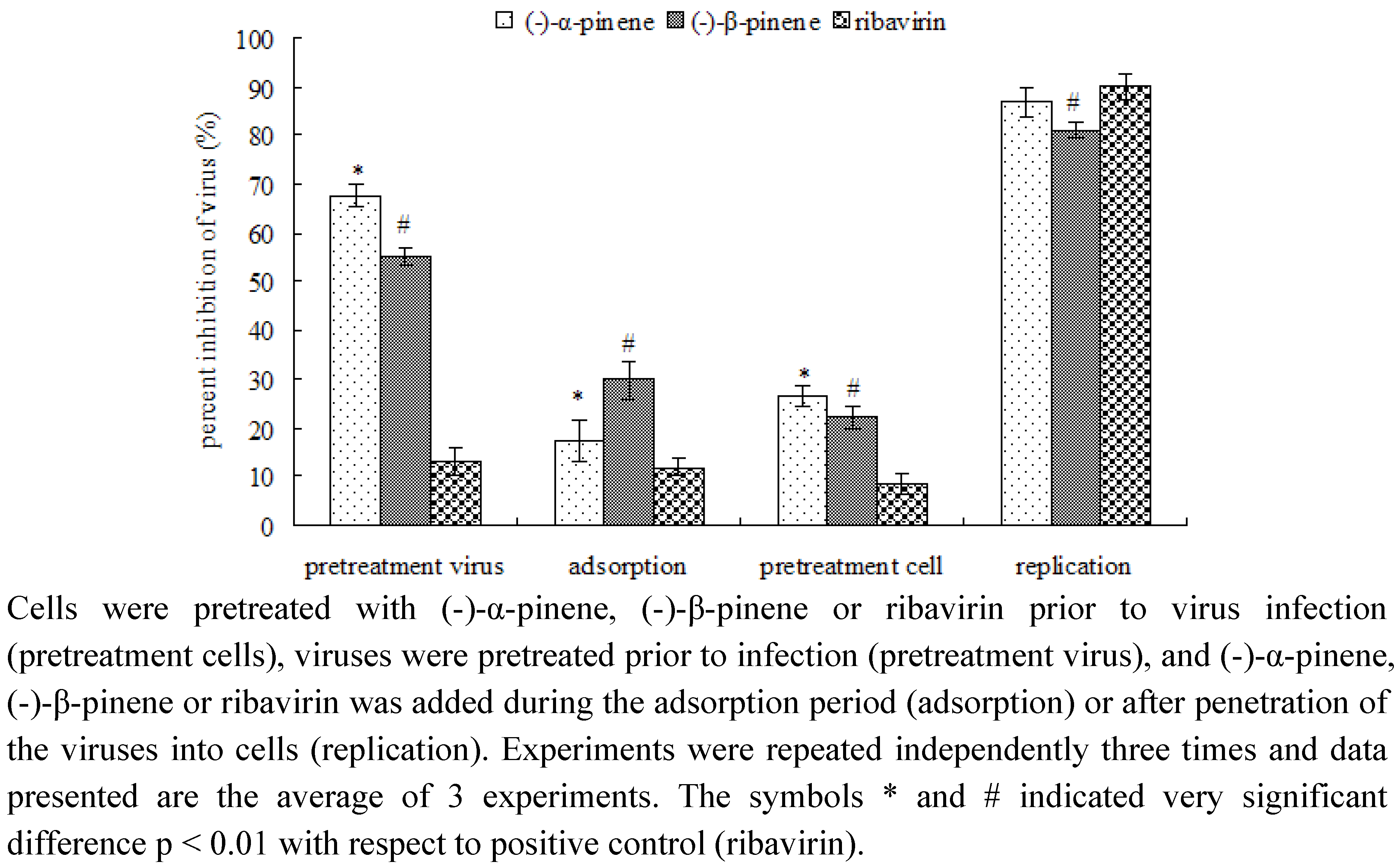
2.2. Mode of anti-IBV activity by MTT assay
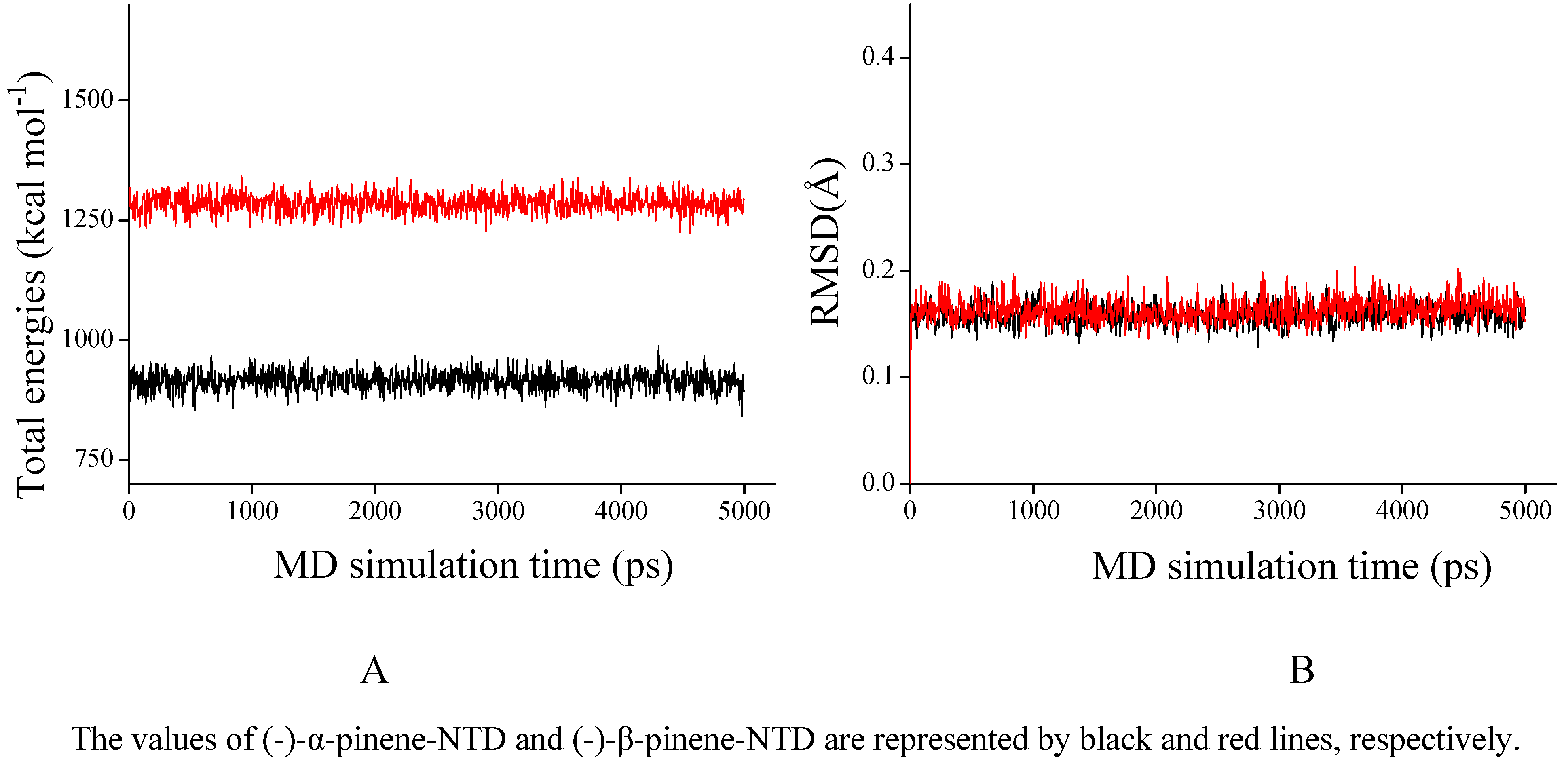
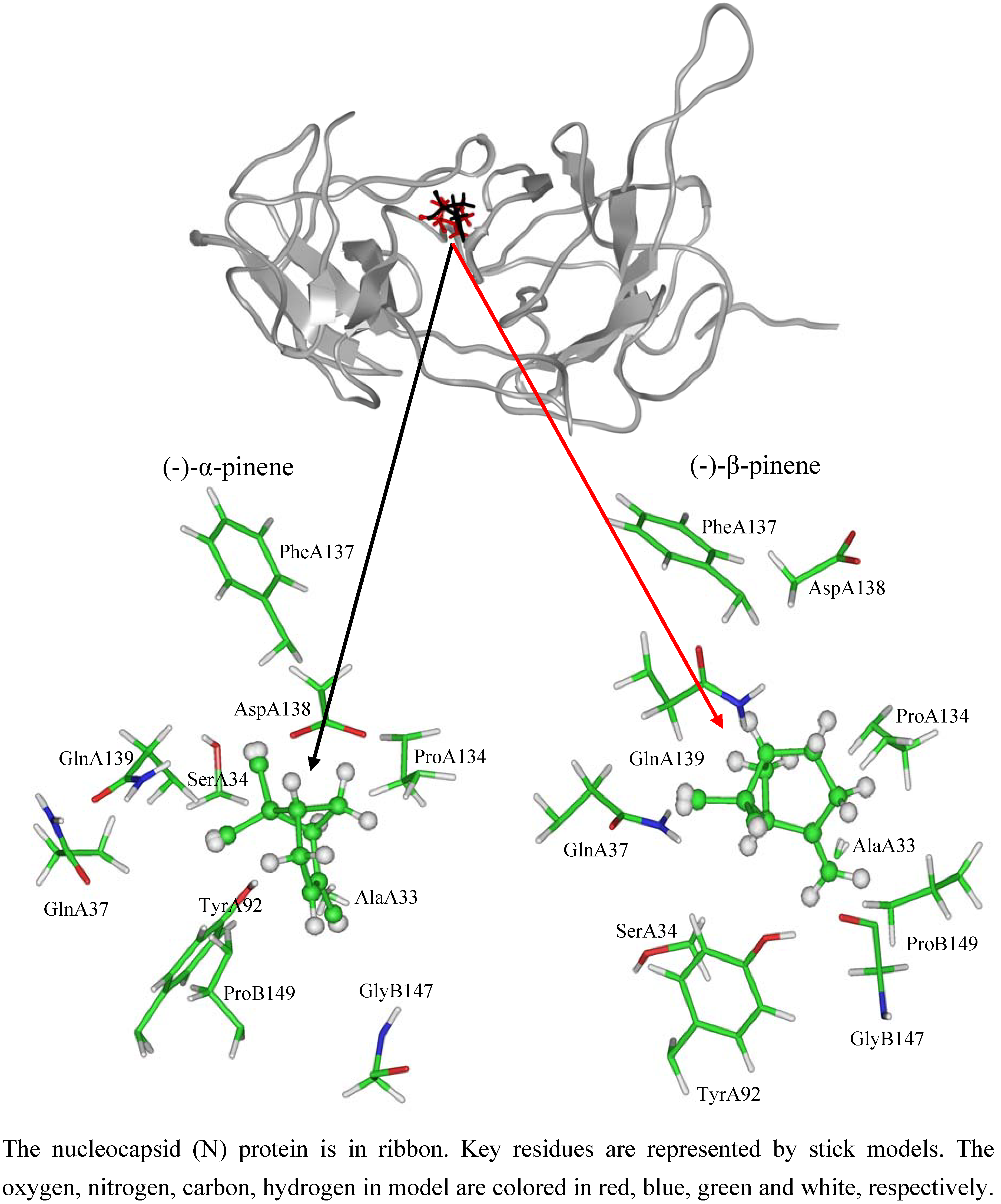
2.3. NTD inhibitory activities of (-)-pinenes
| (-)-α-pinene | (-)-β-pinene | |||||
|---|---|---|---|---|---|---|
| Residue | EvdW | Eele | Esum | EvdW | Eele | Esum |
| AlaA33 | — | — | — | −1.81 | −0.36 | −2.17 |
| SerA34 | −1.61 | −0.18 | −1.79 | −3.03 | −0.10 | −3.13 |
| PheA36 | — | — | — | −1.07 | −0.50 | −1.57 |
| GlnA37 | −1.57 | 0.07 | −1.50 | −2.22 | −0.11 | −2.33 |
| TyrA92 | −3.86 | −2.46 | −6.32 | −3.45 | −0.14 | −3.59 |
| ProA134 | −2.53 | −0.12 | −2.65 | −2.96 | −0.42 | −3.38 |
| PheA137 | −2.66 | 0.08 | −2.58 | −2.00 | −0.23 | −2.23 |
| AspA138 | −1.43 | −0.94 | −2.37 | −2.35 | −0.31 | −2.66 |
| GlnA139 | −2.21 | −0.43 | −2.64 | −2.42 | −0.23 | −2.65 |
| TyrA140 | −1.66 | 0.08 | −1.58 | — | — | — |
| AspB146 | −1.00 | −0.89 | −1.89 | — | — | — |
| GlyB147 | −0.77 | −0.23 | −1.00 | −2.47 | 0.01 | −2.46 |
| GlyB148 | — | — | — | −1.58 | 0.55 | −1.03 |
| ProB149 | −2.30 | −0.02 | −2.32 | −0.63 | −0.09 | −0.72 |
| TrpB155 | −1.85 | −0.08 | −1.93 | −1.57 | −0.04 | −1.61 |
3. Experimental
3.1. Materials
3.2. Cell cultures
3.3. Viruses
3.4. Cytotoxicity assay
3.5. Anti-IBV activity
3.6. Mode of anti-IBV activity
3.7. Flexible docking and MD simulations
3.8. Statistical analysis
4. Conclusions
Acknowledgements
References and Notes
- Lai, M.M.; Cavanagh, D. The molecular biology of coronaviruses. Adv. Virus. Res. 1997, 48, 1–100. [Google Scholar] [CrossRef]
- Reynolds, J.E.F. Martindale the Extra Pharmacopoeia; Royal Pharmaceutical Society of Great Britain: London, UK, 1996; Volume 31, p. 342. [Google Scholar]
- Pei, J.; Briles, W.E.; Collisson, E.W. Memory T cells protect chicks from acute infectious bronchitis virus infection. Virology 2003, 306, 376–384. [Google Scholar] [CrossRef]
- Mondal, S.P.; Cardona, C.J. Comparison of four regions in the replicase gene of heterologous infectious bronchitis virus strains. Virology 2004, 324, 238–248. [Google Scholar] [CrossRef] [Green Version]
- Bosch, B.J.; van der Zee, R.; de Haan, C.A.; Rottier, P.J. The coronavirus spike protein is a class I virus fusion protein: structural and functional characterization of the fusion core complex. J. Virol. 2003, 77, 8801–8811. [Google Scholar] [CrossRef]
- de Haan, C.A.; Kuo, L.; Masters, P.S.; Vennema, H.; Rottier, P.J. Coronavirus particle assembly: Primary structure requirements of the membrane protein. J. Virol. 1998, 72, 6838–6850. [Google Scholar]
- Fischer, F.; Stegen, C.F.; Masters, P.S.; Samsonoff, W.A. Analysis of constructed E gene mutants of mouse hepatitis virus confirms a pivotal role for E protein in coronavirus assembly. J. Virol. 1998, 72, 7885–7894. [Google Scholar]
- Robbins, S.G.; Frana, M.F.; McGowan, J.J.; Boyle, J.F.; Holmes, K.V. RNA-binding proteins of coronavirus MHV: Detection of monomeric and multimeric N protein with an RNA overlay-protein blot assay. Virology 1986, 150, 402–410. [Google Scholar] [CrossRef]
- Compton, S.R.; Rogers, D.B.; Holmes, K.V.; Fertsch, D.; Remenick, J.; McGowan, J.J. In vitro replication of mouse hepatitis virus strain A59. J. Virol. 1987, 61, 1814–1820. [Google Scholar]
- Baric, R.S.; Nelson, G.W.; Fleming, J.O.; Deans, R.J.; Keck, J.G.; Casteel, N.; Stohlman, S.A. Interactions between coronavirus nucleocapsid protein and viral RNAs: Implications for viral transcription. J. Virol. 1988, 62, 4280–4287. [Google Scholar]
- Jayaram, H.; Fan, H.; Bowman, B.R.; Ooi, A.; Jayaram, J.; Collisson, E.W.; Lescar, J.; Prasad, B.V. X-ray structures of the N- and C-terminal domains of a coronavirus nucleocapsid protein: Implications for nucleocapsid formation. J. Virol. 2006, 80, 6612–6620. [Google Scholar]
- Saif, L.J. Coronavirus immunogens. Vet. Microbiol. 1993, 37, 285–297. [Google Scholar] [CrossRef]
- Tan, Y.W.; Fang, S.; Fan, H.; Lescar, J.; Liu, D.X. Amino acid residues critical for RNA-binding in the N-terminal domain of the nucleocapsid protein are essential determinants for the infectivity of coronavirus in cultured cells. Nucleic. Acids. Res. 2006, 34, 4816–4825. [Google Scholar] [CrossRef]
- Yang, Z.W.; Wu, N.; Fu, F.J.; Yang, G.; Wang, W.; Zu, Y.G.; Efferth, T. Anti-infectious bronchitis virus (IBV) activity of 1,8-cineole: Effect on nucleocapsid (N) protein. J. Biomol. Struct. Dyn. 2010, 28, 323–330. [Google Scholar] [CrossRef]
- Nelson, G.W.; Stohlman, S.A.; Tahara, S.M. High affinity interaction between nucleocapsid protein and leader/intergenic sequence of mouse hepatitis virus RNA. J. Gen. Virol. 2000, 81, 181–188. [Google Scholar]
- Fan, H.; Ooi, A.; Tan, Y.W.; Wang, S.; Fang, S.; Liu, D.X.; Lescar, J. The nucleocapsid protein of coronavirus infectious bronchitis virus: crystal structure of its N-terminal domain and multimerization properties. Structure 2005, 13, 1859–1868. [Google Scholar] [CrossRef]
- Saller, R.; Reichling, J.; Hellenbrecht, D. Phytotherapiee Klinische, Pharmakologische und Pharmazeutische Grundlagen; Haug-Verlag: Heidelberg, Germany, 1995. [Google Scholar]
- Hammer, K.A.; Carson, C.F.; Riley, T.V. Antimicrobial activity of essential oils and other plant extracts. J. Appl. Microbiol. 1999, 86, 985–990. [Google Scholar] [CrossRef]
- Reichling, J.; Wildi, E.; Wink, M. Trends in Medicinal Plant Research; Romneya-Verlag: Dossenheim, Germany, 2001. [Google Scholar]
- Mourey, A.; Canillac, N. Anti-Listeria monocytogenes activity of essential oils components of conifers. Food Control 2002, 13, 289–292. [Google Scholar] [CrossRef]
- Hyatt, D.C.; Croteau, R. Mutational analysis of a monoterpene synthase reaction: Altered catalysis through directed mutagenesis of (-)-pinene synthase from Abies grandis. Arch. Biochem. Biophys. 2005, 439, 222–233. [Google Scholar] [CrossRef]
- Tsuchiya, Y.; Shimizu, M.; Hiyama, Y.; Itoh, K.; Hashimoto, Y.; Nakayama, M.; Horie, T.; Morita, N. Antiviral activity of natural occurring flavonoids in vitro. Chem. Pharm. Bull (Tokyo) 1985, 33, 3881–3886. [Google Scholar] [CrossRef]
- Leyssen, P.; Balzarini, J.; De Clercq, E.; Neyts, J. The predominant mechanism by which ribavirin exerts its antiviral activity in vitro against flaviviruses and paramyxoviruses is mediated by inhibition of IMP dehydrogenase. J. Virol. 2005, 79, 1943–1947. [Google Scholar] [CrossRef]
- Schnitzler, P.; Schneider, S.; Stintzing, F.C.; Carle, R.; Reichling, J. Efficacy of an aqueous Pelargonium sidoides extract against herpesvirus. Phytomedicine 2008, 15, 1108–1116. [Google Scholar] [CrossRef]
- Okamoto, M.; Okamoto, T.; Baba, M. Inhibition of human immunodeficiency virus type 1 replication by combination of transcription inhibitor K-12 and other antiretroviral agents in acutely and chronically infected cells. Antimicrob. Agents Chemother. 1999, 43, 492–497. [Google Scholar]
- Head, J.D.; Zerner, M.C. A Broyden—Fletcher—Goldfarb—Shanno optimization procedure for molecular geometries. Chem. Phys. Lett. 1985, 122, 264–270. [Google Scholar] [CrossRef]
- Yang, Z.W.; Yang, G.; Zu, Y.G.; Fu, Y.J.; Zhou, L.J. The conformational analysis and proton transfer of the neuraminidase inhibitors: a theoretical study. Phys. Chem. Chem. Phys. 2009, 11, 10035–10041. [Google Scholar]
- Yang, Z.W.; Zu, Y.G.; Wu, X.M.; Liu, C.B.; Yang, G. A computational investigation on the interaction mechanisms of neuraminidases and 3-(3-pentyloxy)benzoic acid. Acta Chimica. Sinica. 2010, 14, 1370–1378. [Google Scholar]
- Yang, Z.; Nie, Y.; Yang, G.; Zu, Y.; Fu, Y.; Zhou, L. Synergistic effects in the designs of neuraminidase ligands: Analysis from docking and molecular dynamics studies. J. Theor. Biol. 2010, 267, 363–374. [Google Scholar] [CrossRef]
- Accelrys Inc. InsightII, Version 2005, Accelrys Inc.: San Diego, CA, USA.
- Accelrys Inc. Affinity User Guide, Accelrys Inc.: San Diego, CA, USA, 2005.
- Sample Availability: Samples of (-)-α-pinene and (-)-β-pinene are available on request from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yang, Z.; Wu, N.; Zu, Y.; Fu, Y. Comparative Anti-Infectious Bronchitis Virus (IBV) Activity of (-)-Pinene: Effect on Nucleocapsid (N) Protein. Molecules 2011, 16, 1044-1054. https://doi.org/10.3390/molecules16021044
Yang Z, Wu N, Zu Y, Fu Y. Comparative Anti-Infectious Bronchitis Virus (IBV) Activity of (-)-Pinene: Effect on Nucleocapsid (N) Protein. Molecules. 2011; 16(2):1044-1054. https://doi.org/10.3390/molecules16021044
Chicago/Turabian StyleYang, Zhiwei, Nan Wu, Yuangang Zu, and Yujie Fu. 2011. "Comparative Anti-Infectious Bronchitis Virus (IBV) Activity of (-)-Pinene: Effect on Nucleocapsid (N) Protein" Molecules 16, no. 2: 1044-1054. https://doi.org/10.3390/molecules16021044
APA StyleYang, Z., Wu, N., Zu, Y., & Fu, Y. (2011). Comparative Anti-Infectious Bronchitis Virus (IBV) Activity of (-)-Pinene: Effect on Nucleocapsid (N) Protein. Molecules, 16(2), 1044-1054. https://doi.org/10.3390/molecules16021044






