3. Experimental
3.1. General
Melting points (ºC) were measured in open glass capillaries using a Branstead 9001 Electrothermal melting point apparatus and are uncorrected. NMR spectra were obtained on a Bruker AC 500 Ultra Shield NMR spectrometer (Fällanden, Switzerland) operating at 500.13 MHz for 1H and 125.76 MHz for 13C; the chemical shifts are expressed in δ (ppm) downfield from tetramethylsilane (TMS) as internal standard; coupling constants (J) are expressed in Hz. Electrospray ionization mass spectra (ESI-MS) were recorded on a Waters QuatroMicro triple quadrupole tandem mass spectrometer at 4.0 and 3.5 kV for positive and negative ions, respectively. Elemental analyses (C, H, N, S) were in full agreement with the proposed structures within ±0.4% of the theoretical values. Monitoring the reactions and checking the purity of the final products were carried out by thin layer chromatography (TLC) using silica gel precoated aluminum sheets (60 F254, Merck) and visualization with ultraviolet light (UV) at 365 and 254 nm. The bacterial strains and Candida albicans fungus were obtained from the Institute of Fermentation of Osaka (IFO), Osaka, Japan. The reference drugs ampicillin trihydrate (CAS 7177-48-2), clotrimazole (CAS 23593-75-1) and indomethacin (CAS 53-86-1) were obtained from Sigma-Aldrich Chemie GmbH, Taufkirchen, Germany. The Sprauge-Dawley rats were purchased from local animal house (Abu-Rawash, Giza, Egypt). The animal experiments for the determination of the anti-inflammatory activity were carried out in agreement with the pertinent legal and ethical standards of the international guidelines.
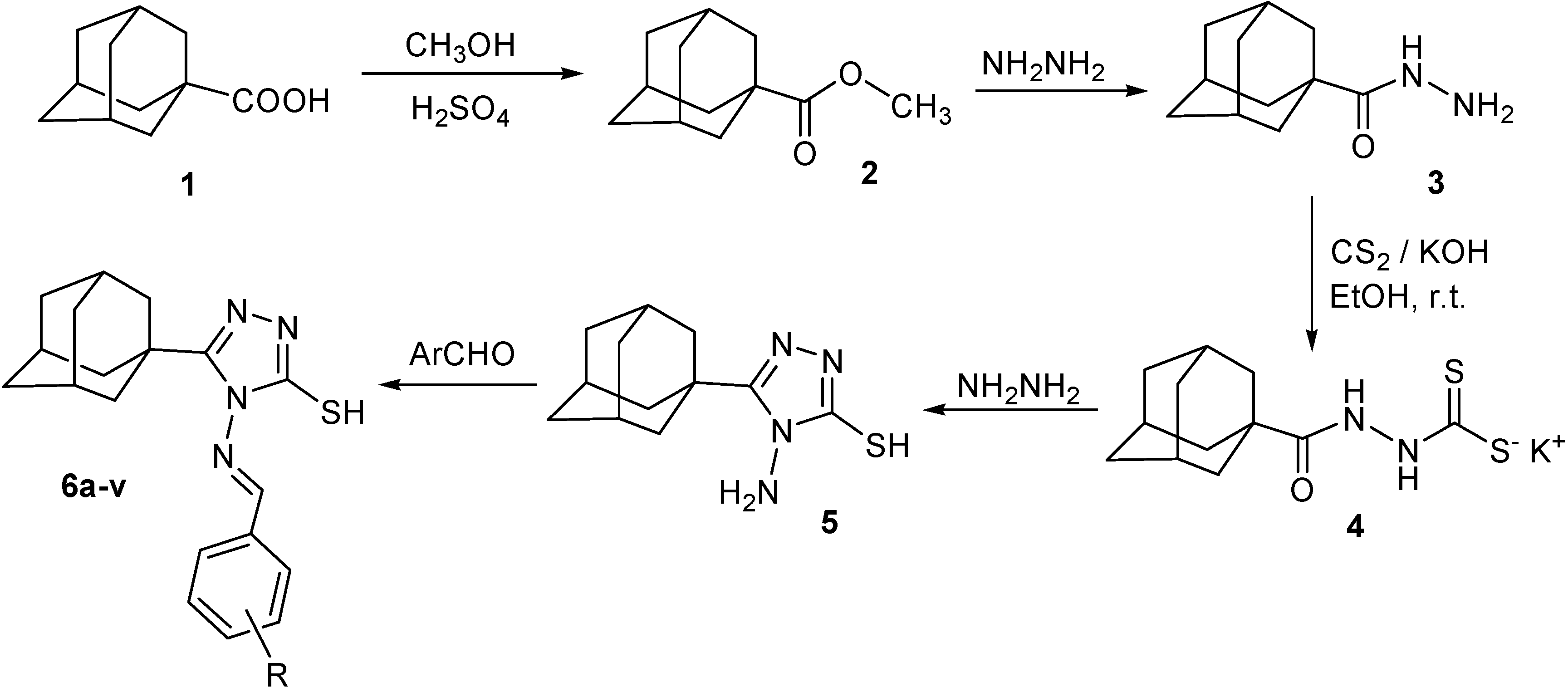
3.2. 5-(1-Adamantyl)-4-arylideneamino-3-mercapto-1,2,4-triazoles 6a–v
Method A: A mixture of the appropriate aromatic aldehyde (2.0 mmol) and 5-(1-adamantyl)-4-amino-3-mercapto-1,2,4-triazole (
5, 0.5 g, 2.0 mmol) in absolute ethanol (10 mL) was heated under reflux for 5 h. On cooling, the separated solid was filtered, washed with cold ethanol (5 mL), dried and crystallized (
Table 1).
Method B(compounds
6l,
6m,
6r,
6s,
6u and
6v): A mixture of the appropriate aromatic aldehyde (2.0 mmol) and 5-(1-adamantyl)-4-amino-3-mercapto-1,2,4-triazole (
5, 0.5 g, 2 mmol) in acetic acid (8 mL) was heated under reflux for 4 h. On cooling, the separated solid was filtered, washed with cold ethanol (5 mL), dried and crystallized (
Table 1).
5-(1-Adamantyl)-4-benzylideneamino-3-mercapto-1,2,4-triazole 6a: 1H-NMR (DMSO-d6): δ 1.69–1.73 (m, 6H, adamantane-H), 2.02 (s, 3H, adamantane-H), 2.07 (s, 6H, adamantane-H), 7.58–7.65 (m, 3H, Ar-H), 7.92 (d, 2H, Ar-H, J = 7.0 Hz), 9.71 (s, 1H, CH=N), 13.82 (s, 1H, SH). 13C-NMR: 27.72, 35.28, 36.47, 38.61 (adamantane-C), 128.94, 129.78, 132.67, 133.17 (Ar-C), 156.04, 162.50 (triazole C-5 & CH=N), 165.02 (triazole C-3). MS, m/z (Rel. Int.): 338 (M+, 51), 261 (12), 236 (26), 235 (100), 221 (14), 202 (17), 135 (15), 104 (12), 90 (11), 77 (6).
5-(1-Adamantyl)-4-(2-fluorobenzylideneamino)-3-mercapto-1,2,4-triazole 6b: 1H-NMR (DMSO-d6): δ 1.71 (s, 6H, adamantane-H), 2.02 (s, 3H, adamantane-H), 2.08 (s, 6H, adamantane-H), 7.40–7.45 (m, 2H, Ar-H), 7.67–7.71 (m, 1H, Ar-H), 8.04 (t, 1H, Ar-H, J = 7.0 Hz), 10.18 (s, 1H, CH=N), 13.85 (s, 1H, SH). 13C-NMR: 27.77, 35.36, 36.49, 38.37 (adamantane-C), 117.0, 120.45, 125.92, 127.86, 135.25, 156.88 (Ar-C), 156.20, 162.44 (triazole C-5 & CH=N), 163.18 (triazole C-3). MS, m/z (Rel. Int.): 356 (M+, 44), 339 (25), 297 (17), 266(24), 260 (20), 235 (100), 234 (60), 220 (31), 135 (25), 122 (26), 108 (29), 96 (10).
5-(1-Adamantyl)-4-(4-fluorobenzylideneamino)-3-mercapto-1,2,4-triazole 6c: 1H-NMR (DMSO-d6): δ 1.71 (s, 6H, adamantane-H), 2.01 (s, 3H, adamantane-H), 2.07 (s, 6H, adamantane-H), 7.34 (d, 2H, Ar-H, J = 6.5 Hz), 7.94 (d, 2H, Ar-H, J = 6.5 Hz), 9.71 (s, 1H, CH=N), 13.81 (s, 1H, SH). 13C-NMR: 27.73, 35.28, 36.47, 38.63 (adamantane-C), 116.40, 129.28, 131.09, 163.95 (Ar-C), 156.02, 162.54 (triazole C-5 & CH=N), 165.37 (triazole C-3). MS, m/z (Rel. Int.): 356 (M+, 17), 235 (100), 234 (58), 220 (14), 135 (34), 122 (22), 121 (62), 107 (18), 95 (44).
5-(1-Adamantyl)-4-(2-chlorobenzylideneamino)-3-mercapto-1,2,4-triazole 6d: 1H-NMR (DMSO-d6): δ 1.73 (s, 6H, adamantane-H), 2.03 (s, 3H, adamantane-H), 2.09 (s, 6H, adamantane-H), 7.58–7.70 (m, 3H, Ar-H), 8.15 (d, 1H, Ar-H, J = 7.5 Hz), 10.48 (s, 1H, CH=N), 13.89 (s, 1H, SH). 13C-NMR: 27.76, 35.41, 36.51, 38.70 (adamantane-C), 127.77, 128.64, 130.40, 130.96, 134.46, 135.69 (Ar-C), 156.27, 158.73 (triazole C-5 & CH=N), 162.44 (triazole C-3). MS, m/z (Rel. Int.): 374 (M+ +2, 25), 372 (M+, 100), 337 (34), 261 (25), 249 (26), 235 (58), 234 (48), 221 (20), 140 (32), 135 (21), 125 (26), 110 (12).
5-(1-Adamantyl)-4-(4-chlorobenzylideneamino)-3-mercapto-1,2,4-triazole 6e: 1H-NMR (DMSO-d6): δ 1.70 (s, 6H, adamantane-H), 2.01 (s, 3H, adamantane-H), 2.06 (s, 6H, adamantane-H), 7.66 (d, 2H, Ar-H, J = 6.5 Hz), 7.93 (d, 2H, Ar-H, J = 6.5 Hz), 9.76 (s, 1H, CH=N), 13.81 (s, 1H, SH). 13C-NMR: 27.73, 35.29, 36.46, 38.63 (adamantane-C), 129.98, 130.56, 131.56, 137.89 (Ar-C), 156.04, 162.53 (triazole C-5 & CH=N), 163.66 (triazole C-3). MS, m/z (Rel. Int.): 374 (M+ +2, 10), 373 (M+ +1, 40), 372 (M+, 23), 371 (100).
5-(1-Adamantyl)-4-(2-bromobenzylideneamino)-3-mercapto-1,2,4-triazole 6f: 1H-NMR (DMSO-d6): δ 1.71 (s, 6H, adamantane-H), 2.01 (s, 3H, adamantane-H), 2.09 (s, 6H, adamantane-H), 7.99 (d, 2H, Ar-H, J = 8.5), 7.86 (d, 2H, Ar-H, J = 8.5 Hz), 9.75 (s, 1H, CH=N), 13.82 (s, 1H, SH). 13C-NMR: 27.80, 35.30, 36.53, 38.64 (adamantane-C), 126.89, 130.71, 131.89, 132.93 (Ar-C), 156.04, 162.51 (triazole C-5 & CH=N), 162.73 (triazole C-3). MS, m/z (Rel. Int.): 418 (M+ +2, 20), 417 (M+ +1, 100), 416 (M+, 20), 415 (100).
5-(1-Adamantyl)-4-(2-hydroxybenzylideneamino)-3-mercapto-1,2,4-triazole 6g: 1H-NMR (DMSO-d6): δ 1.69–1.73 (m, 6H, adamantane-H), 2.01 (s, 3H, adamantane-H), 2.07 (s, 6H, adamantane-H), 6.97–7.02 (m, 2H, Ar-H), 7.42–7.46 (m, 1H, Ar-H), 7.90–7.92 (m, 1H, Ar-H), 9.91 (s, 1H, CH=N), 10.55 (s, 1H, OH), 13.46 (s, 1H, SH). 13C-NMR: 27.74, 35.29, 36.47, 38.61 (adamantane-C), 117.26, 118.95, 120.26, 127.24, 134.72, 158.97 (Ar-C), 156.01, 161.63 (triazole C-5 & CH=N), 162.49 (triazole C-3). MS, m/z (Rel. Int.): 355 (M+ +1, 7), 354 (M+, 28), 353 (100).
5-(1-Adamantyl)-4-(4-hydroxybenzylideneamino)-3-mercapto-1,2,4-triazole 6h: 1H-NMR (DMSO-d6): δ 1.67–1.71 (m, 6H, adamantane-H), 2.01 (s, 3H, adamantane-H), 2.06 (s, 6H, adamantane-H), 6.94 (d, 2H, Ar-H, J = 8.5 Hz), 7.76 (d, 2H, Ar-H, J = 8.5 Hz), 9.36 (s, 1H, CH=N), 10.38 (s, 1H, OH), 13.70 (s, 1H, SH). 13C-NMR: 27.73, 35.21, 36.48, 38.57 (adamantane-C), 116.65, 123.51, 131.18, 162.51 (Ar-C), 155.91, 162.32 (triazole C-5 & CH=N), 165.79 (triazole C-3). MS, m/z (Rel. Int.): 355 (M+ +1, 6), 354 (M+, 26), 353 (100).
5-(1-Adamantyl)-4-(4-methylbenzylideneamino)-3-mercapto-1,2,4-triazole 6i: 1H-NMR (DMSO-d6): δ 1.69–1.73 (m, 6H, adamantane-H), 2.01 (s, 3H, adamantane-H), 2.06 (s, 6H, adamantane-H), 2.40 (s, 3H, CH3), 7.39 (d, 2H, Ar-H, J = 7.5 Hz), 7.81 (d, 2H, Ar-H, J = 7.5 Hz), 9.61 (s, 1H, CH=N), 13.77 (s, 1H, SH). 13C-NMR: 21.72 (CH3), 27.73, 35.26, 36.47, 38.60 (adamantane-C), 128.96, 129.99, 130.37, 143.57 (Ar-C), 156.0, 162.51 (triazole C-5 & CH=N), 165.19 (triazole C-3). MS, m/z (Rel. Int.): 351 (M+ -1, 14), 234 (100), 233 (32).
5-(1-Adamantyl)-4-(2-methoxybenzylideneamino)-3-mercapto-1,2,4-triazole 6j: 1H-NMR (DMSO-d6): δ 1.67–1.74 (m, 6H, adamantane-H), 2.02 (s, 3H, adamantane-H), 2.07 (s, 6H, adamantane-H), 3.89 (s, 3H, OCH3), 7.13–7.23 (m, 2H, Ar-H), 7.60–7.63 (m, 1H, Ar-H), 8.01 (d, 1H, Ar-H, J = 8.0 Hz), 10.02 (s, 1H, CH=N), 13.76 (s, 1H, SH). 13C-NMR: 27.74, 35.30, 36.49, 38.60 (adamantane-C), 56.54 (OCH3), 112.99, 120.74, 121.59, 126.69, 134.97, 159.84 (Ar-C), 156.13 (triazole C-5), 159.81 (CH=N), 162.39 (triazole C-3). MS, m/z (Rel. Int.): 369 (M+ +1, 7), 368 (M+, 23), 367 (100).
5-(1-Adamantyl)-4-(4-methoxybenzylideneamino)-3-mercapto-1,2,4-triazole 6k: 1H-NMR (DMSO-d6): δ 1.67–1.73 (m, 6H, adamantane-H), 2.01 (s, 3H, adamantane-H), 2.06 (s, 6H, adamantane-H), 3.86 (s, 3H, OCH3), 7.13 (d, 2H, Ar-H, J = 8.5 Hz), 7.87 (d, 2H, Ar-H, J = 8.5 Hz), 9.50 (s, 1H, CH=N), 13.79 (s, 1H, SH). 13C-NMR: 27.73, 35.24, 36.48, 38.59 (adamantane-C), 56.04 (OCH3), 115.32, 125.11, 130.91, 163.43 (Ar-C), 155.95, 162.50 (triazole C-5 & CH=N), 165.12 (triazole C-3). MS, m/z (Rel. Int.): 369 (M+ +1, 29), 368 (M+, 76), 353 (33), 259 (33), 249 (37), 236 (51), 235 (49), 234 (29), 135 (54), 133 (100), 108 (112).
5-(1-Adamantyl)-4-(2-nitrobenzylideneamino)-3-mercapto-1,2,4-triazole 6l: 1H-NMR (DMSO-d6): δ 1.72 (s, 6H, adamantane-H), 2.01 (s, 3H, adamantane-H), 2.07 (s, 6H, adamantane-H), 7.85 (t, 1H, Ar-H, J = 7.5 Hz), 7.94 (t, 1H, Ar-H, J = 7.5 Hz), 8.17 (d, 2H, Ar-H, J = 8.0 Hz), 10.43 (s, 1H, CH=N), 13.90 (s, 1H, SH). 13C NMR: 27.76, 35.39, 36.42, 38.63 (adamantane-C), 125.40, 126.87, 129.43, 133.54, 134.53, 149.29 (Ar-C), 156.26, 159.20 (triazole C-5 & CH=N), 162.65 (triazole C-3). MS, m/z (Rel. Int.): 384 (M+ +1, 7), 383 (M+, 24), 382 (100).
5-(1-Adamantyl)-4-(4-nitrobenzylideneamino)-3-mercapto-1,2,4-triazole 6m: 1H-NMR (DMSO-d6): δ 1.71 (s, 6H, adamantane-H), 2.01 (s, 3H, adamantane-H), 2.06 (s, 6H, adamantane-H), 7.89 (d, 2H, Ar-H, J = 8.4 Hz), 8.32(d, 2H, Ar-H, J = 8.4 Hz), 10.21 (s, 1H, CH=N), 13.88 (s, 1H, SH). 13C NMR: 27.73, 35.21, 36.40, 38.61 (adamantane-C), 124.12, 128.83, 136.55, 150.22 (Ar-C), 155.66, 161.26 (triazole C-5 & CH=N), 162.85 (triazole C-3).
5-(1-Adamantyl)-4-(4-dimethylaminobenzylideneamino)-3-mercapto-1,2,4-triazole 6n: 1H-NMR (DMSO-d6): δ 1.66–1.71 (m, 6H, adamantane-H), 2.01 (s, 3H, adamantane-H), 2.06 (s, 6H, adamantane-H), 3.04 (s, 6H, CH3), 6.83 (d, 2H, Ar-H, J = 8.5 Hz), 7.71 (d, 2H, Ar-H, J = 9.0 Hz), 9.21 (s, 1H, CH=N), 13.74 (s, 1H, SH). 13C NMR: 27.74, 35.18, 36.50, 38.55 (adamantane-C), 39.89 (CH3), 112.20, 119.31, 130.66, 153.72 (Ar-C), 155.88, 162.51 (triazole C-5 & CH=N), 165.99 (triazole C-3). MS, m/z (Rel. Int.): 382 (M+ +1, 8), 381 (M+, 36), 379 (100).
5-(1-Adamantyl)-4-(2,6-difluorobenzylideneamino)-3-mercapto-1,2,4-triazole 6o: 1H-NMR (DMSO-d6): δ 1.71 (s, 6H, adamantane-H), 2.01 (s, 3H, adamantane-H), 2.09 (s, 6H, adamantane-H), 7.30–7.34 (m, 2H, Ar-H), 7.67-7.72 (m, 1H, Ar-H), 10.37 (s, 1H, CH=N), 13.87 (s, 1H, SH). 13C NMR: 27.30, 34.89, 35.87, 37.67 (adamantane-C), 109.74, 112.67, 134.86, 160.06 (Ar-C), 155.87, 160.06 (triazole C-5 & CH=N), 162.11 (triazole C-3). MS, m/z (Rel. Int.): 375 (M+ +1, 10), 374 (M+, 33), 343 (14), 236 (37), 235 (100), 234 (43), 220 (17), 139 (40), 135 (18), 126 (18), 113 (13).
5-(1-Adamantyl)-4-(2-chloro-6-fluorobenzylideneamino)-3-mercapto-1,2,4-triazole 6p: 1H-NMR (DMSO-d6): δ 1.71 (s, 6H, adamantane-H), 2.01 (s, 3H, adamantane-H), 2.09 (s, 6H, adamantane-H), 7.44–7.48 (m, 1H, Ar-H), 7.52 (d, 1H, Ar-H, J = 8.0 Hz), 7.63–7.67 (m, 1H, Ar-H), 10.54 (s, 1H, CH=N), 13.89 (s, 1H, SH). 13C NMR: 27.79, 35.41, 36.36, 38.23 (adamantane-C), 116.52, 119.14, 127.17, 134.57, 135.99, 162.59 (Ar-C), 156.41, 160.51 (triazole C-5 & CH=N), 162.21 (triazole C-3). MS, m/z (Rel. Int.): 392 (M+ +2, 8), 390 (M+, 20), 371 (12), 355 (22), 336 (9), 236 (56), 235 (100), 234 (18), 220 (26), 156 (33), 135 (14), 129 (7), 127 (22).
5-(1-Adamantyl)-4-(2,6-dichlorobenzylideneamino)-3-mercapto-1,2,4-triazole 6q: 1H-NMR (DMSO-d6): δ 1.68 (s, 6H, adamantane-H), 1.99 (s, 3H, adamantane-H), 2.09 (s, 6H, adamantane-H), 7.53–7.68 (m, 3H, Ar-H), 10.63 (s, 1H, CH=N), 13.93 (s, 1H, SH). 13C NMR: 27.77, 35.48, 36.38, 38.38 (adamantane-C), 128.33, 130.42, 133.60, 135.32 (Ar-C), 156.43, 158.38 (triazole C-5 & CH=N), 162.46 (triazole C-3). MS, m/z (Rel. Int.): 410 (M+ +4, 4), 409 (M+ +3, 16), 408 (M+ +2, 18), 407 (M+ +1, 74), 406 (M+, 22), 405 (100).
5-(1-Adamantyl)-4-(2,4-dichlorobenzylideneamino)-3-mercapto-1,2,4-triazole 6r: 1H-NMR (DMSO-d6): δ 1.72 (s, 6H, adamantane-H), 2.03 (s, 3H, adamantane-H), 2.08 (s, 6H, adamantane-H), 7.67–7.69 (m, 1H, Ar-H), 7.84 (s, 1H, Ar-H), 8.12 (d, 1H, Ar-H, J = 8.0 Hz), 10.50 (s, 1H, CH=N), 13.89 (s, 1H, SH). 13C NMR: 27.76, 35.41, 36.49, 38.71 (adamantane-C), 128.91, 129.12, 129.49, 130.48, 136.44, 138.29 (Ar-C), 156.26, 157.41 (triazole C-5 & CH=N), 162.44 (triazole C-3). MS, m/z (Rel. Int.): 410 (M+ +4, 3), 409 (M+ +3, 15), 408 (M+ +2, 17), 407 (M+ +1, 69), 406 (M+, 22), 405 (100).
5-(1-Adamantyl)-4-(3,4-dichlorobenzylideneamino)-3-mercapto-1,2,4-triazole 6s: 1H-NMR (DMSO-d6): δ 1.70–1.72 (m, 6H, adamantane-H), 2.02 (s, 3H, adamantane-H), 2.06 (s, 6H, adamantane-H), 7.85–7.93 (m, 2H, Ar-H), 8.13 (s, 1H, Ar-H), 9.88 (s, 1H, CH=N), 13.86 (s, 1H, SH). 13C NMR: 27.74, 35.33, 36.47, 38.67 (adamantane-C), 128.34, 130.61, 132.19, 132.74, 133.40, 135.64 (Ar-C), 156.09, 161.65 (triazole C-5 & CH=N), 162.56 (triazole C-3). MS, m/z (Rel. Int.): 410 (M+ +4, 3), 409 (M+ +3, 15), 408 (M+ +2, 17), 407 (M+ +1, 73), 406 (M+, 23), 405 (100).
5-(1-Adamantyl)-4-(3,4-dimethoxybenzylideneamino)-3-mercapto-1,2,4-triazole 6t: 1H-NMR (DMSO-d6): δ 1.67–1.74 (m, 6H, adamantane-H), 2.02 (s, 3H, adamantane-H), 2.08 (s, 6H, adamantane-H), 3.84 (s, 3H, OCH3), 3.86 (s, 3H, OCH3), 7.15 (d, 1H, Ar-H, J = 8.5 Hz), 7.47 (d, 1H, Ar-H, J = 8.5 Hz), 7.50 (s, 1H, Ar-H), 9.56 (s, 1H, CH=N), 13.74 (s, 1H, SH). 13C NMR: 27.76, 35.28, 36.52, 38.64 (adamantane-C), 56.0 (OCH3), 56.28 (OCH3), 110.09, 112.34, 124.20, 125.25, 149.74, 153.35 (Ar-C), 156.01, 162.46 (triazole C-5 & CH=N), 164.54 (triazole C-3). MS, m/z (Rel. Int.): 399 (M+ +1, 7), 398 (M+, 24), 397 (100).
5-(1-Adamantyl)-4-(2,4-dinitrobenzylideneamino)-3-mercapto-1,2,4-triazole 6u: 1H-NMR (DMSO-d6): δ 1.72 (s, 6H, adamantane-H), 2.02 (s, 3H, adamantane-H), 2.08 (s, 6H, adamantane-H), 8.41 (d, 1H, Ar-H, J = 8.5 Hz), 8.65 (d, 1H, Ar-H, J = 8.5 Hz), 9.03 (s, 1H, Ar-H), 11.51 (s, 1H, CH=N), 13.46 (s, 1H, SH). 13C NMR: 27.80, 34.80, 36.52, 38.36 (adamantane-C), 120.79, 128.01, 129.52, 131.72, 135.05, 147.74 (Ar-C), 157.09, 157.86 (triazole C-5 & CH=N), 167.73 (triazole C-3). MS, m/z (Rel. Int.): 451 (M+ +Na, 4), 450 (11), 451 (48), 436 (100), 428 (M+, 3), 427 (10).
5-(1-Adamantyl)-4-(4,5-dimethoxy-2-nitrobenzylideneamino)-3-mercapto-1,2,4-triazole 6v: 1H-NMR (DMSO-d6): δ 1.70–1.74 (m, 6H, adamantane-H), 2.02 (s, 3H, adamantane-H), 2.10 (s, 6H, adamantane-H), 3.96 (s, 3H, OCH3), 3.97 (s, 3H, OCH3), 7.64 (s, 1H, Ar-H), 7.75 (s, 1H, Ar-H), 10.56 (s, 1H, CH=N), 13.86 (s, 1H, SH). 13C NMR: 27.77, 35.44, 36.49, 38.76 (adamantane-C), 56.67 (OCH3), 57.05 (OCH3), 108.73, 109.16, 120.95, 143.12, 151.89, 153.10 (Ar-C), 156.28, 157.92 (triazole C-5 & CH=N), 162.56 (triazole C-3). MS, m/z (Rel. Int.): 444 (M+ +1, 7), 443 (M+, 24), 442 (100).
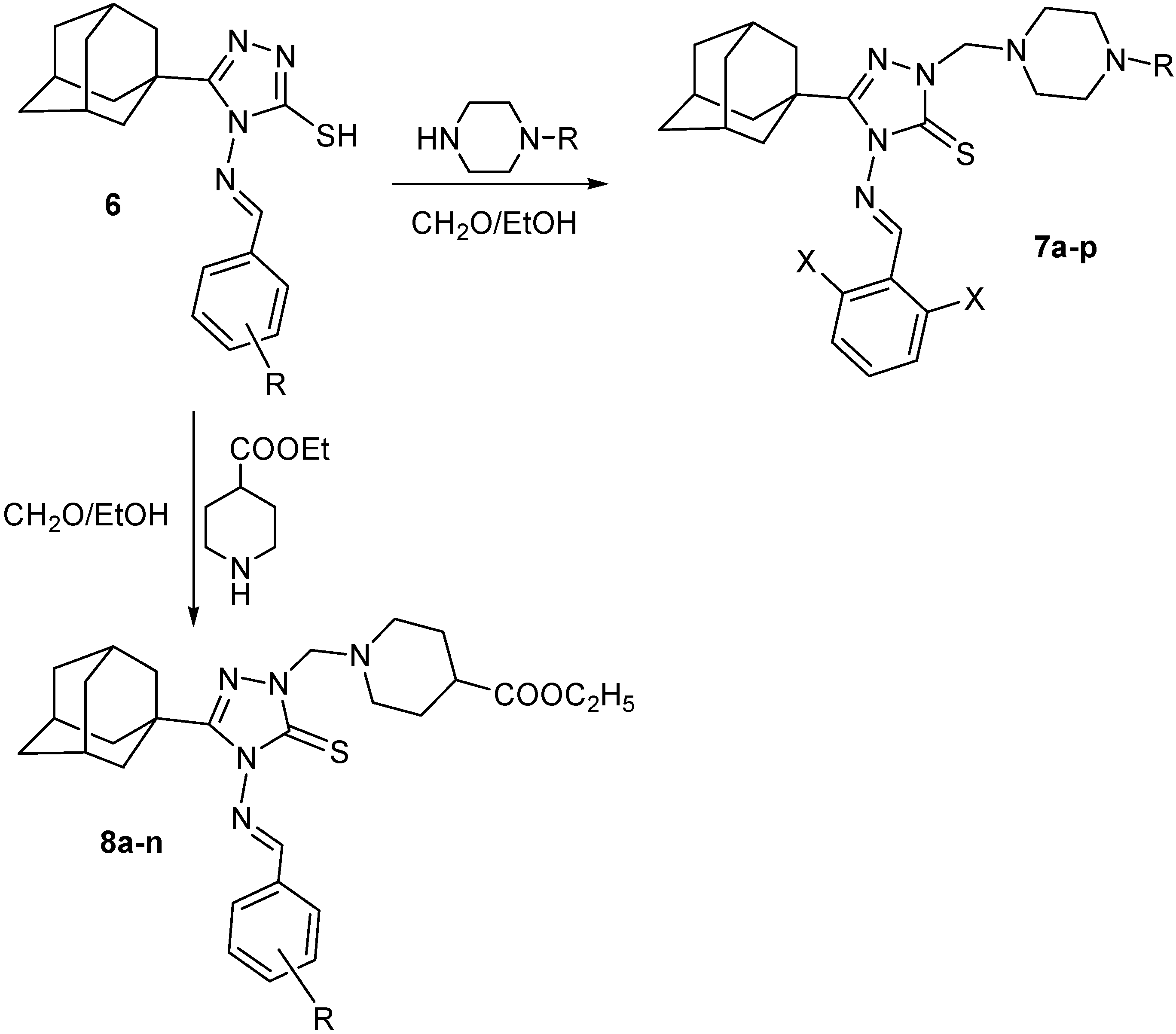
5-(1-Adamantyl)-4-arylideneamino-2-(4-substituted-1-piperazinylmethyl)-1,2,4-triazoline-3-thiones 7a–p
A mixture of the 5-(1-adamantyl)-4-(2,6-dihalobenzylideneamino)-3-mercapto-1,2,4-triazoles
6o or
6q (1.0 mmol), the appropriate
N-substituted piperazine (1.0 mmol) and 37% formaldehyde solution (1 mL), in ethanol (8 mL), was heated under reflux for 15 min when a clear solution was obtained. Stirring was continued for 12 h at room temperature and the mixture was allowed to stand overnight. Cold water (5 mL) was added and the reaction mixture was stirred for 20 min. The precipitated crude products were filtered, washed with water, dried, and crystallized (
Table 2).
5-(1-Adamantyl)-4-(2,6-difluorobenzylideneamino)-2-(4-methyl-1-piperazinylmethyl)-1,2,4-triazoline-3-thione 7a: 1H-NMR (CDCl3): 1.78 (s, 6H, adamantane-H), 2.08 (s, 3H, adamantane-H), 2.16 (s, 6H, adamantane-H), 2.30 (s, 3H, CH3), 2.47 (s, 4H, piperazine-H), 2.92 (s, 4H, piperazine-H), 5.18 (s, 2H, CH2), 7.02–7.07 (m, 2H, Ar-H), 7.46–7.48 (m. 1H, Ar-H), 10.64 (s, 1H, CH=N). 13C NMR: 27.95, 35.51, 36.45, 38.32 (adamantane-C), 46.05 (CH3), 55.05, 58.36 (piperazine-C), 68.68 (CH2), 110.87, 112.16, 133.21, 152.24 (Ar-C), 155.52, 160.99 (triazole C-5 & CH=N), 163.05 (C=S). MS, m/z (Rel. Int.): 488 (M+ +2, 30), 487 (M+ +1, 100).
5-(1-Adamantyl)-4-(2,6-difluorobenzylideneamino)-2-(4-ethyl-1-piperazinylmethyl)-1,2,4-triazoline-3-thione 7b: 1H-NMR (CDCl3): δ 1.07 (t, 3H, CH3, J = 7.0 Hz), 1.78 (s, 6H, adamantane-H), 2.08 (s, 3H, adamantane-H), 2.16 (s, 6H, adamantane-H), 2.40 (q, 2H, CH2CH3, J = 7.0 Hz), 2.48–2.49 (m, 4H, piperazine-H), 2.92 (s, 4H, piperazine-H), 5.18 (s, 2H, CH2), 7.02 (t, 2H, Ar-H, J = 8.5 Hz), 7.44–7.48 (m. 1H, Ar-H), 10.62 (s, 1H, CH=N). 13C NMR: 11.91 (CH3), 28.0, 35.50, 36.46, 38.33 (adamantane-C), 52.32 (CH2CH3), 50.42, 52.80 (piperazine-C), 68.79 (CH2), 110.89, 112.16, 133.20, 152.22 (Ar-C), 155.45, 161.0 (triazole C-5 & CH=N), 163.14 (C=S). MS, m/z (Rel. Int.): 502 (M+ +2, 32), 501 (M+ +1, 100).
5-(1-Adamantyl)-4-(2,6-difluorobenzylideneamino)-2-(4-ethoxycarbonyl-1-piperazinylmethyl)-1,2,4-triazoline-3-thione 7c: 1H-NMR (CDCl3): δ 1.24 (t, 3H, CH3, J = 7.0 Hz), 1.79 (s, 6H, adamantane-H), 2.09 (s, 3H, adamantane-H), 2.17 (s, 6H, adamantane-H), 2.81 (s, 4H, piperazine-H), 3.50 (s, 4H, piperazine-H), 4.10 (q, 2H, CH2CH3, J = 7.0 Hz), 5.16 (s, 2H, CH2), 7.02 (t, 2H, Ar-H, J = 7.0 Hz), 7.46–7.50 (m. 1H, Ar-H), 10.64 (s, 1H, CH=N). 13C NMR: 14.62 (CH3), 27.98, 35.55, 36.43, 38.35 (adamantane-C), 50.37, 52.39 (piperazine-C), 61.32 (CH2CH3), 68.97 (CH2), 110.81, 112.22, 133.29, 152.23 (Ar-C), 155.46, 155.70, 161.04 (C=O, triazole C-5 & CH=N), 163.20 (C=S). MS, m/z (Rel. Int.): 546 (M+ +2, 6), 445 (M+ +1, 20).
5-(1-Adamantyl)-4-(2,6-difluorobenzylideneamino)-2-(4-phenyl-1-piperazinylmethyl)-1,2,4-triazoline-3-thione 7d: 1H-NMR (CDCl3): δ 1.80 (s, 6H, adamantane-H), 2.10 (s, 3H, adamantane-H), 2.19 (s, 6H, adamantane-H), 3.04 (s, 4H, piperazine-H), 3.23 (s, 4H, piperazine-H), 5.24 (s, 2H, CH2), 6.89 (t, 1H, Ar-H, J = 7.0 Hz), 6.94 (d, 2H, Ar-H, J = 8.0 Hz), 7.03 (t. 2H, Ar-H, J = 8.5 Hz), 7.26–7.28 (m, 2H, Ar-H), 7.47–7.50 (m. 1H, Ar-H), 10.67 (s, 1H, CH=N). 13C NMR: 28.0, 35.56, 36.46, 38.37 (adamantane-C), 49.41, 50.55 (piperazine-C), 68.82 (CH2), 110.76, 112.19, 116.31, 119.88, 129.10, 133.27, 151.38, 152.24 (Ar-C), 155.64, 161.06 (triazole C-5 & CH=N), 163.22 (C=S). MS, m/z (Rel. Int.): 550 (M+ +2, 36), 549 (M+ +1, 100).
5-(1-Adamantyl)-4-(2,6-difluorobenzylideneamino)-2-[4-(4-fluorophenyl)-1-piperazinylmethyl]-1,2,4-triazoline-3-thione 7e: 1H-NMR (CDCl3): δ 1.80 (s, 6H, adamantane-H), 2.10 (s, 3H, adamantane-H), 2.19 (s, 6H, adamantane-H), 3.04 (s, 4H, piperazine-H), 3.14 (s, 4H, piperazine-H), 5.23 (s, 2H, CH2), 6.89–6.99 (m, 4H, Ar-H), 7.03 (t. 2H, Ar-H, J = 8.5 Hz), 7.48–7.49 (m, 1H, Ar-H), 10.66 (s, 1H, CH=N). 13C NMR: 28.0, 35.57, 36.46, 38.38 (adamantane-C), 50.40, 50.54 (piperazine-C), 68.78 (CH2), 110.85, 112.39, 115.41, 118.10, 133.28, 148.04, 152.24, 158.24 (Ar-C), 155.65 (CH=N), 161.06 (triazole C-5), 163.23 (C=S). MS, m/z (Rel. Int.): 568 (M+ +2, 33), 567 (M+ +1, 100).
5-(1-Adamantyl)-4-(2,6-difluorobenzylideneamino)-2-[4-(3-trifluoromethylphenyl)-1-piperazinylmethyl]-1,2,4-triazoline-3-thione 7f: 1H-NMR (CDCl3): δ 1.80 (s, 6H, adamantane-H), 2.10 (s, 3H, adamantane-H), 2.19 (s, 6H, adamantane-H), 3.03-3.04 (m, 4H, piperazine-H), 3.21-3.27 (m, 4H, piperazine-H), 5.24 (s, 2H, CH2), 7.04–7.13 (m, 5H, Ar-H), 7.34 (t. 1H, Ar-H, J = 8.0 Hz), 7.46–7.50 (m, 1H, Ar-H), 10.67 (s, 1H, CH=N). 13C NMR: 27.99, 35.57, 36.44, 38.38 (adamantane-C), 48.87, 50.36 (piperazine-C), 68.73 (CH2), 118.86 (CF3), 110.83, 112.36, 115.09, 123.22, 125.39, 129.53, 131.58, 133.31, 151.44, 152.27 (Ar-C), 155.71, 161.01 (triazole C-5 & CH=N), 163.25 (C=S). MS, m/z (Rel. Int.): 618 (M+ +2, 35), 617 (M+ +1, 100).
5-(1-Adamantyl)-4-(2,6-difluorobenzylideneamino)-2-[4-(2-methoxylphenyl)-1-piperazinylmethyl]-1,2,4-triazoline-3-thione 7g: 1H-NMR (CDCl3): δ 1.80 (s, 6H, adamantane-H), 2.10 (s, 3H, adamantane-H), 2.20 (s, 6H, adamantane-H), 3.10 (s, 8H, piperazine-H), 3.87 (s, 3H, OCH3), 5.25 (s, 2H, CH2), 6.86 (d, 1H, Ar-H, J = 7.5 Hz), 6.92–6.98 (m, 2H, Ar-H), 7.0–7.07 (m, 3H, Ar-H), 7.47–7.49 (m, 1H, Ar-H), 10.63 (s, 1H, CH=N). 13C NMR: 28.02, 35.55, 36.48, 38.34 (adamantane-C), 50.72, 50.83 (piperazine-C), 55.27 (OCH3), 69.04 (CH2), 110.03, 112.18, 112.38, 118.28, 120.94, 123.02, 133.23, 141.34, 152.26, 152.33 (Ar-C), 155.55, 161.02 (triazole C-5 & CH=N), 163.21 (C=S). MS, m/z (Rel. Int.): 580 (M+ +2, 33), 579 (M+ +1, 92).
5-(1-Adamantyl)-4-(2,6-difluorobenzylideneamino)-2-(4-benzyl-1-piperazinylmethyl)-1,2,4-triazoline-3-thione 7h: 1H-NMR (CDCl3): δ 1.80 (s, 6H, adamantane-H), 2.09 (s, 3H, adamantane-H), 2.18 (s, 6H, adamantane-H), 2.41–2.52 (m, 4H, piperazine-H), 2.95 (s, 4H, piperazine-H), 3.52 (s, 2H, PhCH2), 5.17 (s, 2H, CH2), 7.03 (t, 2H, Ar-H, J = 8.5 Hz), 7.25–7.32 (m, 5H, Ar-H), 7.45–7.49 (m, 1H, Ar-H), 10.65 (s, 1H, CH=N). 13C NMR: 28.0, 35.52, 36.47, 38.36 (adamantane-C), 50.51, 53.12 (piperazine-C), 63.18 (PhCH2), 68.96 (CH2), 110.89, 112.17, 127.04, 128.18, 129.29, 133.11, 137.96, 152.11 (Ar-C), 155.47, 161.05 (triazole C-5 & CH=N), 163.19 (C=S). MS, m/z (Rel. Int.): 564 (M+ +2, 34), 563 (M+ +1, 100).
5-(1-Adamantyl)-4-(2,6-dichlorobenzylideneamino)-2-(4-methyl-1-piperazinylmethyl)-1,2,4-triazoline-3-thione 7i: 1H-NMR (CDCl3): δ 1.76 (s, 6H, adamantane-H), 2.07 (s, 3H, adamantane-H), 2.16 (s, 6H, adamantane-H), 2.33 (s, 3H, CH3), 2.50–2.54 (m, 4H, piperazine-H), 2.92–2.96 (m, 4H, piperazine-H), 5.17 (s, 2H, CH2), 7.22-7.48 (m, 3H, Ar-H), 10.80 (s, 1H, CH=N). 13C-NMR: 27.91, 35.54, 36.43, 38.44 (adamantane-C), 43.85 (CH3), 54.36, 56.42 (piperazine-C), 68.71 (CH2), 128.62, 129.43, 131.75, 136.18 (Ar-C), 155.42, 158.18 (triazole C-5 & CH=N), 163.44 (C=S). MS, m/z (Rel. Int.): 522 (M+ +4, 6), 520 (M+ +2, 55), 518 (M+, 100).
5-(1-Adamantyl)-4-(2,6-dichlorobenzylideneamino)-2-(4-ethyl-1-piperazinylmethyl)-1,2,4-triazoline-3-thione 7j: 1H-NMR (CDCl3): δ 1.08 (t, 3H, CH3, J = 7.0 Hz), 1.75 (s, 6H, adamantane-H), 2.06 (s, 3H, adamantane-H), 2.16–2.17 (m, 6H, adamantane-H), 2.42 (q, 2H, CH2CH3, J = 7.0 Hz), 2.52–2.53 (m, 4H, piperazine-H), 2.95 (s, 4H, piperazine-H), 5.19 (s, 2H, CH2), 7.32-7.37 (m, 1H, Ar-H), 7.42–7.47 (m. 2H, Ar-H), 10.74 (s, 1H, CH=N). 13C-NMR: 11.87 (CH3), 27.96, 35.55, 36.47, 38.51 (adamantane-C), 50.41 (CH2CH3), 52.31, 52.76 (piperazine-C), 68.93 (CH2), 129.32, 129.40, 131.53, 136.10 (Ar-C), 155.41, 157.82 (triazole C-5 & CH=N), 163.42 (C=S). MS, m/z (Rel. Int.): 536 (M+ +4, 5), 534 (M+ +2, 43), 532 (M+, 100).
5-(1-Adamantyl)-4-(2,6-dichlorobenzylideneamino)-2-(4-ethoxycarbonyl-1-piperazinylmethyl)-1,2,4-triazoline-3-thione 7k: 1H-NMR (CDCl3): δ 1.25 (t, 3H, CH3, J = 7.0 Hz), 1.76 (s, 6H, adamantane-H), 2.07 (s, 3H, adamantane-H), 2.16 (s, 6H, adamantane-H), 2.83 (s, 4H, piperazine-H), 3.51 (s, 4H, piperazine-H), 4.11 (q, 2H, CH2CH3, J = 7.0 Hz), 5.17 (s, 2H, CH2), 7.30–7.36 (m, 1H, Ar-H), 7.41–7.46 (m. 2H, Ar-H), 10.79 (s, 1H, CH=N). 13C-NMR: 14.64 (CH3), 27.94, 35.61, 36.44, 38.52 (adamantane-C), 50.30, 52.50 (piperazine-C), 61.33 (CH2CH3), 69.15 (CH2), 128.94, 129.37, 131.61, 136.09 (Ar-C), 155.30, 155.46, 157.76 (C=O, triazole C-5 & CH=N), 163.46 (C=S). MS, m/z (Rel. Int.): 599 (M+ +Na, 100), 577 (M+ +1, 7).
5-(1-Adamantyl)-4-(2,6-dichlorobenzylideneamino)-2-(4-phenyl-1-piperazinylmethyl)-1,2,4-triazoline-3-thione 7l: 1H-NMR (CDCl3): δ 1.77 (s, 6H, adamantane-H), 2.08 (s, 3H, adamantane-H), 2.18 (s, 6H, adamantane-H), 3.06 (s, 4H, piperazine-H), 3.23 (s, 4H, piperazine-H), 5.25 (s, 2H, CH2), 6.87 (t, 1H, Ar-H, J = 7.5 Hz), 6.95 (d, 2H, Ar-H, J = 8.0 Hz), 7.27–7.37 (m, 4H, Ar-H), 7.46–7.47 (m, 1H, Ar-H), 10.80 (s, 1H, CH=N). 13C-NMR: 27.96, 35.61, 36.46, 38.54 (adamantane-C), 49.45, 50.60 (piperazine-C), 69.0 (CH2), 116.35, 119.92, 129.06, 129.11, 129.37, 131.60, 136.12, 151.39 (Ar-C), 155.61, 156.09, 157.83 (triazole C-5 & CH=N), 163.48 (C=S). MS, m/z (Rel. Int.): 585 (M+ +4, 16), 583 (M+ +2, 77), 581 (M+, 100).
5-(1-Adamantyl)-4-(2,6-dichlorobenzylideneamino)-2-[4-(4-fluorophenyl)-1-piperazinylmethyl]-1,2,4-triazoline-3-thione 7m: 1H-NMR (CDCl3): δ 1.76 (s, 6H, adamantane-H), 2.08 (s, 3H, adamantane-H), 2.18 (s, 6H, adamantane-H), 3.05–3.06 (m, 4H, piperazine-H), 3.15 (s, 4H, piperazine-H), 5.24 (s, 2H, CH2), 6.88–6.91 (m, 2H, Ar-H), 6.96–6.99 (m, 2H, Ar-H), 7.33–7.36 (m, 1H, Ar-H), 7.45 (d, 2H, Ar-H, J = 7.0 Hz), 10.79 (s, 1H, CH=N). 13C-NMR: 27.96, 35.61, 36.46, 38.55 (adamantane-C), 50.44, 50.59 (piperazine-C), 68.95 (CH2), 115.43, 115.60, 118.08, 118.14, 129.37, 131.60, 136.11, 148.06 (Ar-C), 155.62, 157.83 (triazole C-5 & CH=N), 163.49 (C=S). MS, m/z (Rel. Int.): 602 (M+ +4, 23), 601 (M+ +3, 78), 600 (M+ +2, 32), 599 (M+ +1, 100).
5-(1-Adamantyl)-4-(2,6-dichlorobenzylideneamino)-2-[4-(3-trifluoromethylphenyl)-1-piperazinylmethyl]-1,2,4-triazoline-3-thione 7n: 1H-NMR (CDCl3): δ 1.77 (s, 6H, adamantane-H), 2.08 (s, 3H, adamantane-H), 2.18 (s, 6H, adamantane-H), 3.05–3.06 (m, 4H, piperazine-H), 3.27–3.28 (m, 4H, piperazine-H), 5.24 (s, 2H, CH2), 7.07 (t, 2H, Ar-H, J = 8.5 Hz), 7.14 (s, 1H, Ar-H), 7.34–7.37 (m, 2H, Ar-H), 7.46 (d, 2H, Ar-H, J = 8.0 Hz), 10.80 (s, 1H, CH=N). 13C-NMR: 27.94, 35.62, 36.44, 38.55 (adamantane-C), 48.89, 50.40 (piperazine-C), 68.91 (CH2), 118.89 (CF3), 112.43, 115.98, 123.22, 125.39, 129.02, 129.39, 129.55, 131.78, 136.12, 151.44 (Ar-C), 155.68, 157.86 (triazole C-5 & CH=N), 163.50 (C=S). MS, m/z (Rel. Int.): 652 (M+ +4, 16), 651 (M+ +3, 76), 650 (M+ +2, 32), 649 (M+ +1, 100).
5-(1-Adamantyl)-4-(2,6-dichlorobenzylideneamino)-2-[4-(2-methoxyphenyl)-1-piperazinylmethyl]-1,2,4-triazoline-3-thione 7o: 1H-NMR (CDCl3): δ 1.77 (s, 6H, adamantane-H), 2.08 (s, 3H, adamantane-H), 2.19 (s, 6H, adamantane-H), 3.11 (s, 8H, piperazine-H), 3.87 (s, 3H, OCH3), 5.26 (s, 2H, CH2), 6.86 (d, 1H, Ar-H, J = 7.5 Hz), 6.93–7.04 (m, 3H, Ar-H), 7.33–7.36 (m, 1H, Ar-H) 7.45 (d, 2H, Ar-H, J = 8.0 Hz), 10.76 (s, 1H, CH=N). 13C-NMR: 27.98, 35.59, 36.49, 38.51 (adamantane-C), 50.76, 50.84 (piperazine-C), 55.28 (OCH3), 69.22 (CH2), 110.98, 118.29, 120.93, 123.04, 129.12, 129.34, 131.55, 136.12, 141.32, 152.27 (Ar-C), 155.50 (triazole C-5), 157.89 (CH=N), 163.49 (C=S). MS, m/z (Rel. Int.): 614 (M+ +4, 9), 613 (M+ +3, 35), 612 (M+ +2, 14), 611 (M+ +1, 46).
5-(1-Adamantyl)-4-(2,6-dichlorobenzylideneamino)-2-(4-benzyl-1-piperazinylmethyl)-1,2,4-triazoline-3-thione 7p: 1H-NMR (CDCl3): δ 1.76 (s, 6H, adamantane-H), 2.07 (s, 3H, adamantane-H), 2.17 (s, 6H, adamantane-H), 2.51 (s, 4H, piperazine-H), 2.92 (s, 4H, piperazine-H), 3.52 (s, 2H, PhCH2), 5.17 (s, 2H, CH2), 7.26–7.36 (m, 6H, Ar-H), 7.45 (d, 2H, Ar-H , J = 8.0 Hz), 10.78 (s, 1H, CH=N). 13C-NMR: 27.96, 35.56, 36.47, 38.53 (adamantane-C), 50.53, 53.14 (piperazine-C), 63.24 (PhCH2), 69.11 (CH2), 127.07, 128.20, 129.12, 129.33, 129.42, 131.54, 136.10, 137.90 (Ar-C), 155.45, 157.68 (triazole C-5 & CH=N), 163.45 (C=S). MS, m/z (Rel. Int.): 598 (M+ +4, 13), 597 (M+ +3, 76), 596 (M+ +2, 35), 595 (M+ +1, 100).
5-(1-Adamantyl)-4-arylideneamino-2-(4-ethoxycarbonyl-1-piperidylmethyl)-1,2,4-triazoline-3-thiones 8a–n
A mixture of the 5-(1-adamantyl)-4-arylideneamino-3-mercapto-1,2,4-triazole
6 (1.0 mmol), ethyl 4-piperidinecarboxylate (0.16 g, 1.0 mmol) and 37% formaldehyde solution (1 mL), in ethanol (8 mL), was heated under reflux for 20 min when a clear solution was obtained. Stirring was continued for 12 h at room temperature and the mixture was allowed to stand overnight. Cold water (5 mL) was added and the reaction mixture was stirred for 20 min. The precipitated crude products were filtered, washed with water, dried, and crystallized (
Table 3).
5-(1-Adamantyl)-4-benzylideneamino-2-(4-ethoxycarbonyl-1-piperidylmethyl)-1,2,4-triazoline-3-thione 8a: 1H-NMR (CDCl3): δ 1.24 (t, 3H, CH3, J = 7.0 Hz), 1.72–1.83 (m, 8H, 6 adamantane-H & 2 piperidine-H), 1.92–1.94 (m, 2H, piperidine-H), 2.10 (s, 3H, adamantane-H), 2.17 (s, 6H, adamantane-H), 2.20–2.25 (m, 1H, piperidine-4 H), 2.46–2.50 (m, 2H, piperidine-H), 3.20–3.22 (m, 2H, piperidine-H), 4.12 (q, 2H, CH2CH3, J = 7.0 Hz), 5.15 (s, 2H, CH2), 7.51–7.58 (m, 3H, Ar-H), 7.91 (d, 2H, Ar-H, J = 7.0 Hz), 10.08 (s, 1H, CH=N). 13C-NMR: 14.20 (CH3), 27.93, 35.44, 36.58, 38.77 (adamantane-C), 28.35 (piperidine C-3), 40.42 (piperidine C-4), 50.42 (piperidine C-2), 60.25 (CH2CH3), 69.63 (CH2), 128.72, 129.02, 132.28, 132.84 (Ar-C), 155.28, 162.26 (triazole C-5 & CH=N), 163.21 (C=S), 174.99 (C=O). MS, m/z (Rel. Int.): 509 (M+ +2, 21), 508 (M+ +1, 72).
5-(1-Adamantyl)-4-(2-fluorobenzylideneamino)-2-(4-ethoxycarbonyl-1-piperidylmethyl)-1,2,4-triazoline-3-thione 8b: 1H-NMR (CDCl3): δ 1.24 (t, 3H, CH3CH2, J = 7.0 Hz), 1.72–1.82 (m, 8H, 6 adamantane-H & 2 piperidine-H), 1.92–1.94 (m, 2H, piperidine-H), 2.10 (s, 3H, adamantane-H), 2.16 (s, 6H, adamantane-H), 2.20–2.25 (m, 1H, piperidine-4 H), 2.50–2.54 (m, 2H, piperidine-H), 3.19–3.22 (m, 2H, piperidine-H), 4.12 (q, 2H, CH2CH3, J = 7.0 Hz), 5.15 (s, 2H, CH2), 7.18 (t, 1H, Ar-H, J = 8.0 Hz), 7.30 (d, 1H, Ar-H, J = 7.5 Hz), 7.53–7.56 (m, 1H, Ar-H), 8.08-8.11 (m, 1H, Ar-H), 10.42 (s, 1H, CH=N). 13C-NMR: 14.20 (CH3), 27.93, 35.46, 36.56, 38.76 (adamantane-C), 28.35 (piperidine C-3), 40.77 (piperidine C-4), 50.42 (piperidine C-2), 60.25 (CH2CH3), 69.64 (CH2), 116.31, 120.89, 124.66, 127.57, 133.90, 155.82 (Ar-C), 155.28, 161.54 (triazole C-5 & CH=N), 163.31 (C=S), 174.99 (C=O). MS, m/z (Rel. Int.): (M+ +2, 33), 526 (M+ +1, 100).
5-(1-Adamantyl)-4-(2-chlorobenzylideneamino)-2-(4-ethoxycarbonyl-1-piperidylmethyl)-1,2,4-triazoline-3-thione 8c: 1H-NMR (CDCl3): δ 1.24 (t, 3H, CH3CH2, J = 7.0 Hz), 1.72–1.79 (m, 8H, 6 adamantane-H & 2 piperidine-H), 1.92–1.94 (m, 2H, piperidine-H), 2.11 (s, 3H, adamantane-H), 2.17 (s, 6H, adamantane-H), 2.21–2.25 (m, 1H, piperidine-4 H), 2.51-2.55 (m, 2H, piperidine-H), 3.20–3.22 (m, 2H, piperidine-H), 4.12 (q, 2H, CH2CH3, J = 7.0 Hz), 5.15 (s, 2H, CH2), 7.40 (t, 1H, Ar-H, J = 7.5 Hz), 7.46–7.51 (m, 2H, Ar-H), 8.19 (d, 1H, Ar-H, J = 7.5 Hz), 10.69 (s, 1H, CH=N). 13C-NMR: 14.20 (CH3), 27.93, 35.49, 36.58, 38.83 (adamantane-C), 28.35 (piperidine C-3), 40.78 (piperidine C-4), 50.43 (piperidine C-2), 60.25 (CH2CH3), 69.65 (CH2), 127.20, 127.60, 130.34, 130.85, 132.97, 136.68 (Ar-C), 155.28, 158.27 (triazole C-5 & CH=N), 163.37 (C=S), 174.99 (C=O). MS, m/z (Rel. Int.): 564 (M+ +Na, 100), 544 (23), 542 (54).
5-(1-Adamantyl)-4-(4-methylbenzylideneamino)-2-(4-ethoxycarbonyl-1-piperidylmethyl)-1,2,4-triazoline-3-thione 8d: 1H-NMR (CDCl3): δ 1.24 (t, 3H, CH3CH2, J = 7.0 Hz), 1.72–1.78 (m, 8H, 6 adamantane-H & 2 piperidine-H), 1.91–1.94 (m, 2H, piperidine-H), 2.09 (s, 3H, adamantane-H), 2.16 (s, 6H, adamantane-H), 2.20–2.25 (m, 1H, piperidine-4 H), 2.45 (s, 3H, PhCH3), 2.50–2.54 (m, 2H, piperidine-H), 3.19–3.22 (m, 2H, piperidine-H), 4.12 (q, 2H, CH2CH3, J = 7.0 Hz), 5.15 (s, 2H, CH2), 7.31 (d, 2H, Ar-H, J = 8.0 Hz), 7.80 (d, 2H, Ar-H, J = 8.0 Hz), 9.94 (s, 1H, CH=N). 13C-NMR: 14.20 (CH3), 21.71 (PhCH3), 27.93, 35.40, 36.58, 38.75 (adamantane-C), 28.35 (piperidine C-3), 40.78 (piperidine C-4), 50.41 (piperidine C-2), 60.24 (CH2CH3), 69.62 (CH2), 128.75, 129.76, 130.10, 143.05 (Ar-C), 155.24, 162.69 (triazole C-5 & CH=N), 163.21 (C=S), 174.99 (C=O). MS, m/z (Rel. Int.): 544 (M+ +Na, 100), 523 (M+ +2, 43), 522 (M+ +1, 76).
5-(1-Adamantyl)-4-(2-hydroxybenzylideneamino)-2-(4-ethoxycarbonyl-1-piperidylmethyl)-1,2,4-triazoline-3-thione 8e: 1H-NMR (CDCl3): δ 1.25 (t, 3H, CH3CH2, J = 7.0 Hz), 1.73–1.79 (m, 8H, 6 adamantane-H & 2 piperidine-H), 1.92–1.95 (m, 2H, piperidine-H), 2.10 (s, 9H, adamantane-H), 2.22–2.24 (m, 1H, piperidine-4 H), 2.51–2.55 (m, 2H, piperidine-H), 3.19–3.21 (m, 2H, piperidine-H), 4.12 (q, 2H, CH2CH3, J = 7.0 Hz), 5.15 (s, 2H, CH2), 7.03–7.06 (m, 1H, Ar-H), 7.08 (d, 1H, Ar-H, J = 8.5 Hz), 7.44–7.51 (m, 2H, Ar-H), 9.69 (s, 1H, CH=N), 10.45 (s, 1H, OH). 13C-NMR: 14.20 (CH3), 27.80, 35.35, 36.29, 38.97 (adamantane-C), 28.32 (piperidine C-3), 40.73 (piperidine C-4), 50.42 (piperidine C-2), 60.28 (CH2CH3), 70.22 (CH2), 116.09, 117.56, 120.09, 133.60, 134.69, 169.08 (Ar-C), 154.28, 160.08 (triazole C-5 & CH=N), 164.01 (C=S), 174.94 (C=O). MS, m/z (Rel. Int.): 546 (M+ +Na, 100), 525 (M+ +2, 26), 524 (M+ +1, 84).
5-(1-Adamantyl)-4-(4-hydroxybenzylideneamino)-2-(4-ethoxycarbonyl-1-piperidylmethyl)-1,2,4-triazoline-3-thione 8f: 1H-NMR (CDCl3): δ 1.25 (t, 3H, CH3CH2, J = 7.0 Hz), 1.75–1.78 (m, 8H, 6 adamantane-H & 2 piperidine-H), 1.94–1.96 (m, 2H, piperidine-H), 2.07 (s, 3H, adamantane-H), 2.12 (s, 6H, adamantane-H), 2.24–2.28 (m, 1H, piperidine-4 H), 2.52–2.56 (m, 2H, piperidine-H), 3.21–3.23 (m, 2H, piperidine-H), 4.13 (q, 2H, CH2CH3, J = 7.0 Hz), 5.14 (s, 2H, CH2), 6.92 (d, 2H, Ar-H, J = 8.5 Hz), 7.72 (d, 2H, Ar-H, J = 8.5 Hz), 9.55 (s, 1H, CH=N). 13C-NMR: 14.17 (CH3), 27.88, 35.37, 36.52, 38.66 (adamantane-C), 28.15 (piperidine C-3), 40.72 (piperidine C-4), 50.43 (piperidine C-2), 58.53 (CH2CH3), 69.64 (CH2), 116.24, 124.64, 130.91, 163.01 (Ar-C), 155.38, 160.42 (triazole C-5 & CH=N), 164.17 (C=S), 175.32 (C=O). MS, m/z (Rel. Int.): 525 (M+ +2, 33), 524 (M+ +1, 100).
5-(1-Adamantyl)-4-(4-methoxybenzylideneamino)-2-(4-ethoxycarbonyl-1-piperidylmethyl)-1,2,4-triazoline-3-thione 8g: 1H-NMR (CDCl3): δ 1.24 (t, 3H, CH3CH2, J = 7.0 Hz), 1.72–1.79 (m, 8H, 6 adamantane-H & 2 piperidine-H), 1.91–1.93 (m, 2H, piperidine-H), 2.09 (s, 3H, adamantane-H), 2.16 (s, 6H, adamantane-H), 2.20–2.24 (m, 1H, piperidine-4 H), 2.50-2.53 (m, 2H, piperidine-H), 3.19–3.21 (m, 2H, piperidine-H), 3.90 (s, 3H, OCH3), 4.12 (q, 2H, CH2CH3, J = 7.0 Hz), 5.15 (s, 2H, CH2), 7.02 (d, 2H, Ar-H, J = 8.5 Hz), 7.86 (d, 2H, Ar-H , J = 8.5 Hz), 9.81 (s, 1H, CH=N). 13C-NMR: 14.20 (CH3), 27.93, 35.37, 36.58, 38.74 (adamantane-C), 28.35 (piperidine C-3), 40.79 (piperidine C-4), 55.48 (piperidine C-2), 60.23 (CH2CH3), 69.64 (CH2), 114.54, 125.35, 130.59, 162.76 (Ar-C), 155.18, 162.69 (triazole C-5 & CH=N), 163.21 (C=S), 174.99 (C=O). MS, m/z (Rel. Int.): 560 (M+ +Na, 100), 539 (M+ +2, 38), 538 (M+ +1, 86).
5-(1-Adamantyl)-4-(2,6-difluorobenzylideneamino)-2-(4-ethoxycarbonyl-1-piperidylmethyl)-1,2,4-triazoline-3-thione 8h: 1H-NMR (CDCl3): δ 1.24 (t, 3H, CH3CH2, J = 7.0 Hz), 1.71–1.80 (m, 8H, 6 adamantane-H & 2 piperidine-H), 1.92–1.94 (m, 2H, piperidine-H), 2.09 (s, 3H, adamantane-H), 2.18 (s, 6H, adamantane-H), 2.21–2.25 (m, 1H, piperidine-4 H), 2.50–2.54 (m, 2H, piperidine-H), 3.19–3.21 (m, 2H, piperidine-H), 4.12 (q, 2H, CH2CH3, J = 7.0 Hz), 5.14 (s, 2H, CH2), 7.03 (t, 2H, Ar-H, J = 8.5 Hz), 7.45–7.49 (m, 1H, Ar-H), 10.70 (s, 1H, CH=N). 13C-NMR: 14.19 (CH3), 28.0, 35.53, 36.46, 38.41 (adamantane-C), 28.35 (piperidine C-3), 40.77 (piperidine C-4), 50.42 (piperidine C-2), 60.25 (CH2CH3), 69.43 (CH2), 110.89, 112.37, 133.21, 161.04 (Ar-C), 155.53, 160.99 (triazole C-5 & CH=N), 163.21 (C=S), 174.98 (C=O). MS, m/z (Rel. Int.): 566 (M+ +Na, 100), 545 (M+ +2, 18), 544 (M+ +1, 61).
5-(1-Adamantyl)-4-(2-chloro-6-fluorobenzylideneamino)-2-(4-ethoxycarbonyl-1-piperidylmethyl)-1,2,4-triazoline-3-thione 8i: 1H-NMR (CDCl3): δ 1.24 (t, 3H, CH3CH2, J = 7.0 Hz), 1.71–1.79 (m, 8H, 6 adamantane-H & 2 piperidine-H), 1.92–1.94 (m, 2H, piperidine-H), 2.09 (s, 3H, adamantane-H), 2.18 (s, 6H, adamantane-H), 2.21–2.25 (m, 1H, piperidine-4 H), 2.51–2.55 (m, 2H, piperidine-H), 3.20–3.22 (m, 2H, piperidine-H), 4.12 (q, 2H, CH2CH3, J = 7.0 Hz), 5.15 (s, 2H, CH2), 7.14–7.17 (m, 2H, Ar-H), 7.33 (d, 1H, Ar-H, J = 7.5 Hz), 10.87 (s, 1H, CH=N). 13C-NMR: 14.19 (CH3), 28.0, 35.55, 36.46, 38.40 (adamantane-C), 28.36 (piperidine C-3), 40.77 (piperidine C-4), 50.43 (piperidine C-2), 60.24 (CH2CH3), 69.46 (CH2), 115.35, 119.81, 126.25, 132.52, 137.10, 162.85 (Ar-C), 155.11 (CH=N), 160.76 (triazole C-5), 163.05 (C=S), 174.99 (C=O). MS, m/z (Rel. Int.): 582 (M+ +Na, 100), 562 (29), 561 (M+ +2, 21), 560 (M+ +1, 63).
5-(1-Adamantyl)-4-(2,6-dichlorobenzylideneamino)-2-(4-ethoxycarbonyl-1-piperidylmethyl)-1,2,4-triazoline-3-thione 8j: 1H-NMR (CDCl3): δ 1.24 (t, 3H, CH3CH2, J = 7.0 Hz), 1.72–1.81 (m, 8H, 6 adamantane-H & 2 piperidine-H), 1.92–1.95 (m, 2H, piperidine-H), 2.07 (s, 3H, adamantane-H), 2.17 (s, 6H, adamantane-H), 2.21–2.26 (m, 1H, piperidine-4 H), 2.53–2.57 (m, 2H, piperidine-H), 3.20–3.22 (m, 2H, piperidine-H), 4.12 (q, 2H, CH2CH3, J = 7.0 Hz), 5.15 (s, 2H, CH2), 7.33–7.36 (m, 1H, Ar-H), 7.45 (d, 2H, Ar-H, J = 8.0 Hz), 10.84 (s, 1H, CH=N). 13C-NMR: 14.20 (CH3), 27.96, 35.59, 36.46, 38.58 (adamantane-C), 28.36 (piperidine C-3), 40.77 (piperidine C-4), 50.44 (piperidine C-2), 60.24 (CH2CH3), 69.62 (CH2), 128.77, 129.36, 131.54, 136.08 (Ar-C), 155.52, 157.56 (triazole C-5 & CH=N), 163.22 (C=S), 174.99 (C=O). MS, m/z (Rel. Int.): 598 (M+ +Na, 100), 578 (52), 577 (M+ +2, 23), 576 (M+ +1, 73).
5-(1-Adamantyl)-4-(2,4-dichlorobenzylideneamino)-2-(4-ethoxycarbonyl-1-piperidylmethyl)-1,2,4-triazoline-3-thione 8k: 1H-NMR (CDCl3): δ 1.24 (t, 3H, CH3CH2, J = 7.0 Hz), 1.72–1.83 (m, 8H, 6 adamantane-H & 2 piperidine-H), 1.91–1.94 (m, 2H, piperidine-H), 2.11 (s, 3H, adamantane-H), 2.16 (s, 6H, adamantane-H), 2.20–2.25 (m, 1H, piperidine-4 H), 2.50–2.54 (m, 2H, piperidine-H), 3.19–3.21 (m, 2H, piperidine-H), 4.12 (q, 2H, CH2CH3, J = 7.0 Hz), 5.14 (s, 2H, CH2), 7.40 (d, 1H, Ar-H, J = 8.5 Hz), 7.53 (s, 1H, Ar-H), 8.11 (d, 1H, Ar-H, J = 8.5 Hz), 10.73 (s, 1H, CH=N). 13C-NMR: 14.20 (CH3), 27.91, 35.50, 36.57, 38.86 (adamantane-C), 28.34 (piperidine C-3), 40.76 (piperidine C-4), 50.42 (piperidine C-2), 60.26 (CH2CH3), 69.62 (CH2), 127.88, 128.28, 129.55, 130.16, 137.15, 138.58 (Ar-C), 155.23, 156.68 (triazole C-5 & CH=N), 163.33 (C=S), 174.96 (C=O). MS, m/z (Rel. Int.): 598 (M+ +Na, 100), 578 (56), 577 (M+ +2, 24), 576 (M+ +1, 78).
5-(1-Adamantyl)-4-(3,4-dichlorobenzylideneamino)-2-(4-ethoxycarbonyl-1-piperidylmethyl)-1,2,4-triazoline-3-thione 8l: 1H-NMR (CDCl3): δ 1.24 (t, 3H, CH3CH2, J = 7.0 Hz), 1.71–1.84 (m, 8H, 6 adamantane-H & 2 piperidine-H), 1.91–1.93 (m, 2H, piperidine-H), 2.12 (s, 3H, adamantane-H), 2.15 (s, 6H, adamantane-H), 2.20–2.24 (m, 1H, piperidine-4 H), 2.49–2.53 (m, 2H, piperidine-H), 3.18–3.20 (m, 2H, piperidine-H), 4.11 (q, 2H, CH2CH3, J = 7.0 Hz), 5.13 (s, 2H, CH2), 7.59 (d, 1H, Ar-H, J = 8.0 Hz), 7.72 (d, 1H, Ar-H, J = 8.0 Hz), 7.98 (s, 1H, Ar-H), 10.29 (s, 1H, CH=N). 13C-NMR: 14.20 (CH3), 27.90, 35.50, 36.57, 38.86 (adamantane-C), 28.33 (piperidine C-3), 40.74 (piperidine C-4), 50.41 (piperidine C-2), 60.27 (CH2CH3), 69.63 (CH2), 127.44, 130.03, 131.16, 133.01, 133.67, 136.40 (Ar-C), 155.25, 158.17 (triazole C-5 & CH=N), 163.20 (C=S), 174.94 (C=O). MS, m/z (Rel. Int.): 598 (M+ +Na, 100), 577 (M+ +2, 20), 576 (M+ +1, 67).
5-(1-Adamantyl)-4-(3,4-dimethoxybenzylideneamino)-2-(4-ethoxycarbonyl-1-piperidylmethyl)-1,2,4-triazoline-3-thione 8m: 1H-NMR (CDCl3): δ 1.24 (t, 3H, CH3CH2, J = 7.0 Hz), 1.76-1.82 (m, 8H, 6 adamantane-H & 2 piperidine-H), 1.91-1.93 (m, 2H, piperidine-H), 2.09 (s, 3H, adamantane-H), 2.17 (s, 6H, adamantane-H), 2.20-2.24 (m, 1H, piperidine-4 H), 2.49-2.54 (m, 2H, piperidine-H), 3.19-3.21 (m, 2H, piperidine-H), 3.96 (s, 3H, OCH3), 3.98 (s, 3H, OCH3), 4.11 (q, 2H, CH2CH3, J = 7.0 Hz), 5.15 (s, 2H, CH2), 6.79 (d, 1H, Ar-H, J = 8.0 Hz), 7.40 (d, 1H, Ar-H, J = 8.0 Hz), 7.54 (s, 1H, Ar-H), 9.85 (s, 1H, CH=N). 13C-NMR: 14.19 (CH3), 27.94, 35.39, 36.61, 38.80 (adamantane-C), 28.35 (piperidine C-3), 40.78 (piperidine C-4), 50.41 (piperidine C-2), 55.92 (OCH3), 56.08 (OCH3), 60.24 (CH2CH3), 69.63 (CH2), 109.19, 110.96, 124.42, 125.61, 149.60, 152.98 (Ar-C), 155.13, 162.48 (triazole C-5 & CH=N), 163.18 (C=S), 174.98 (C=O). MS, m/z (Rel. Int.): 590 (M+ +Na, 100), 569 (M+ +2, 26), 568 (M+ +1, 84).
5-(1-Adamantyl)-4-(4,5-dimethoxy-2-nitrobenzylideneamino)-2-(4-ethoxycarbonyl-1-piperidylmethyl)-1,2,4-triazoline-3-thione 8n: 1H-NMR (CDCl3): δ 1.24 (t, 3H, CH3CH2, J = 7.0 Hz), 1.73–1.80 (m, 8H, 6 adamantane-H & 2 piperidine-H), 1.92–1.94 (m, 2H, piperidine-H), 2.08 (s, 3H, adamantane-H), 2.15 (s, 6H, adamantane-H), 2.21–2.23 (m, 1H, piperidine-4 H), 2.51–2.55 (m, 2H, piperidine-H), 3.18–3.21 (m, 2H, piperidine-H), 4.04 (s, 3H, OCH3), 4.05 (s, 3H, OCH3), 4.11 (q, 2H, CH2CH3, J = 7.0 Hz), 5.14 (s, 2H, CH2), 7.65 (s, 1H, Ar-H), 7.63 (s, 1H, Ar-H), 10.54 (s, 1H, CH=N). 13C-NMR: 14.20 (CH3), 27.89, 35.42, 36.51, 38.81 (adamantane-C), 28.34 (piperidine C-3), 40.75 (piperidine C-4), 50.41 (piperidine C-2), 56.60 (OCH3), 56.68 (OCH3), 60.25 (CH2CH3), 69.85 (CH2), 107.82, 110.0, 122.50, 142.33, 151.45, 153.30 (Ar-C), 155.03, 160.57 (triazole C-5 & CH=N), 163.57 (C=S), 174.99 (C=O). MS, m/z (Rel. Int.): 635 (M+ +Na, 100), 614 (M+ +2, 23), 613 (M+ +1, 68).
Determination of the antimicrobial activity by the agar disc-diffusion method. Sterile filter paper discs (8 mm diameter) were moistened with the compound solution in dimethylsulphoxide of specific concentration (200 μg/disc), the antibacterial antibiotics Gentamicin and Ampicillin trihydrate (100 μg/disc) and the antifungal drug Clotrimazole (100 μg/disc) were carefully placed on the agar culture plates that had been previously inoculated separately with the microorganisms. The plates were incubated at 37 ºC, and the diameter of the growth inhibition zones were measured after 24 h in case of bacteria and 48 h in case of Candida albicans.
Determination of minimal inhibitory concentration (MIC). Compounds 6h, 6o, 7n, 8a, 8e, 8f and 8m, Gentamicin, Ampicillin trihydrate and Clotrimazole were dissolved in dimethylsulphoxide at concentration of 128 μg/mL. The twofold dilutions of the solution were prepared (128, 64, 32, …, 0.5 μg/mL). The microorganism suspensions at 106 CFU/mL (colony forming unit/ml) concentrations were inoculated to the corresponding wells. The plates were incubated at 36 ºC for 24 and 48 h for the bacteria and Candida albicans, respectively. The MIC values were determined as the lowest concentration that completely inhibited visible growth of the microorganism as detected by unaided eye.
Determination of the anti-inflammatory activity. Male Sprague-Dawley rats weighing 140–190 g were maintained at room temperature (20–23 ºC). The animals were randomly divided into 42 groups each of 5 animals. The animals were housed with food and water
ad libitum and allowed to be accustomed to their environment for two days before testing. Each group was injected with the specific dose of the test compound (20 and 40 mg/kg), or Indomethacin (5 mg/kg) intraperitoneally as a uniform suspension in 1 ml of 0.5% (w/v) aqueous carboxymethyl cellulose solution, one hour before injection of 0.1 mL of carrageenan (1% solution in normal saline) into the plantar tissue of the right hind paw. The left hind paw was injected with 0.1 mL of normal saline solution. Four hours after carrageenan injection, the volume of paw oedema (mL) was determined using water plethysmometer. The percentage protection against inflammation was calculated as follows:
Where Vc is the mean percentage increase in paw volume in the absence of the test compound (control) and Vd is the mean percentage increase in paw volume after injection of the test compound. The values are expressed as the mean percentage reduction ± S.E.M. Statistical significance between the control and treated groups was performed using the Student “t” test.