Biological Actions of Artemisinin: Insights from Medicinal Chemistry Studies
Abstract
:1. Introduction
2. The History of Artemisinin
3. Physical and Chemical Properties of Artemisinin
4. Artemisinins and Iron
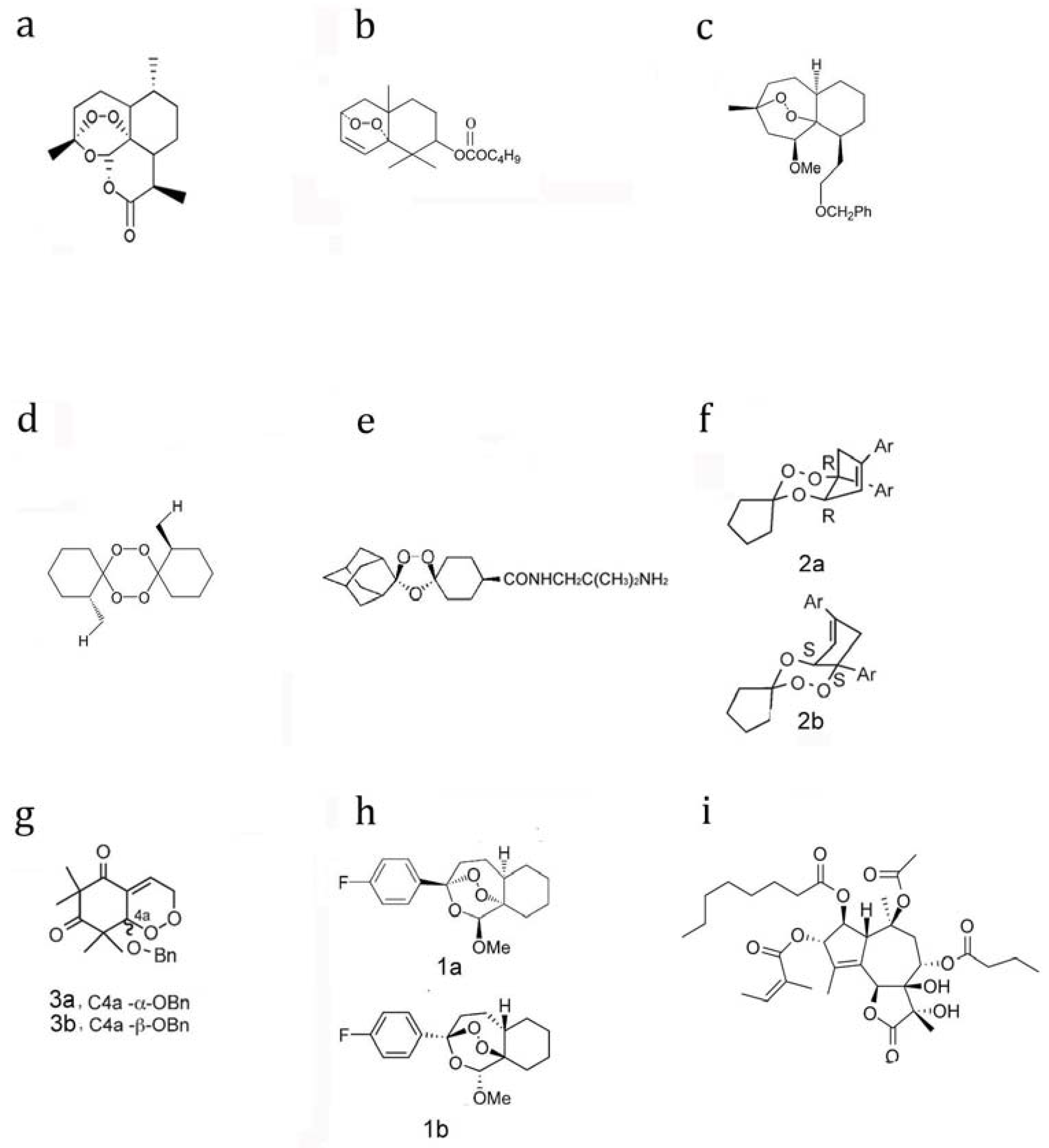
5. The Structure-Efficacy Relationship of Artemisinins and Other Endoperoxidic Antimalarials
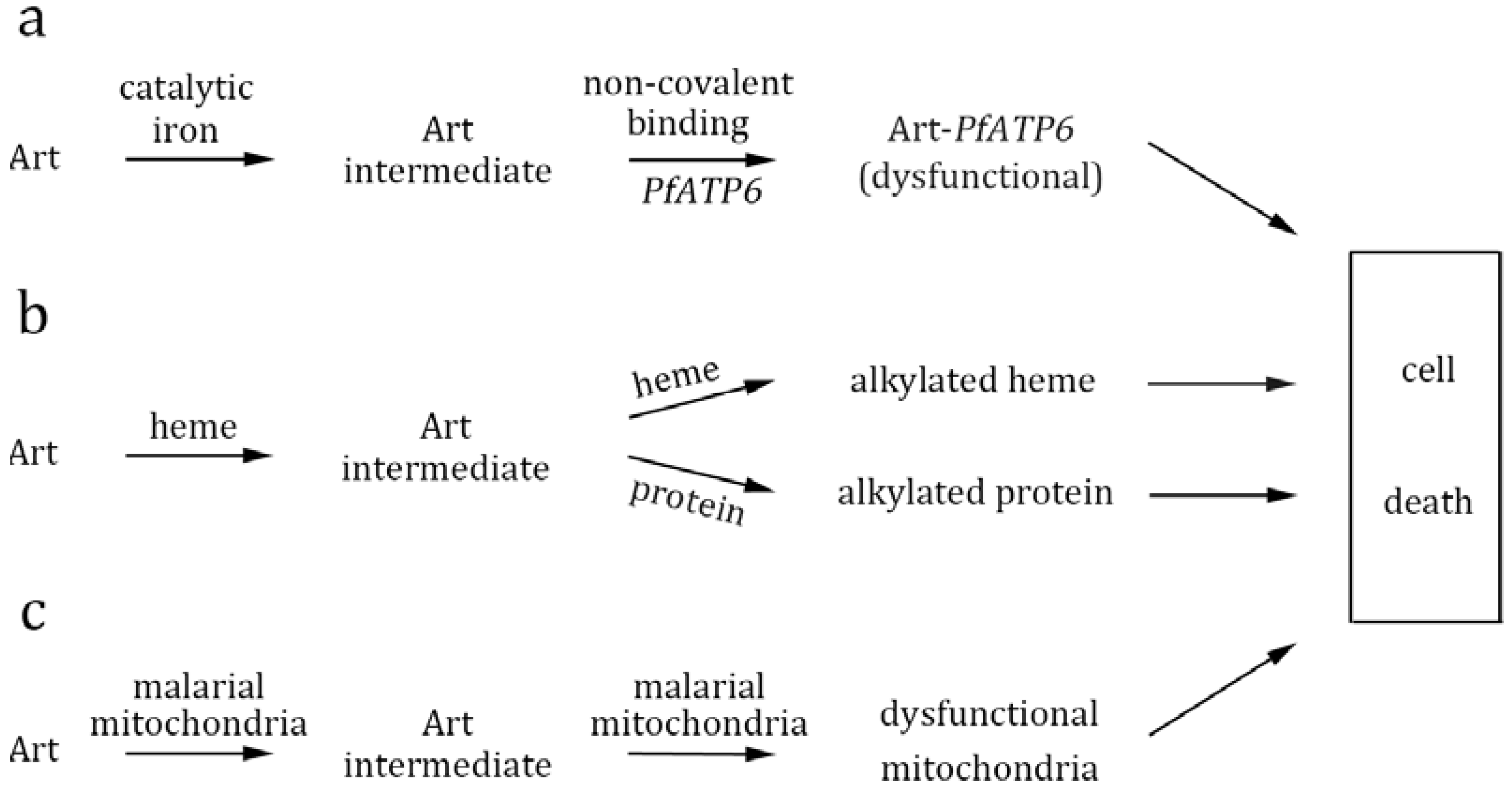
6. Mechanistic Insights from Chemistry Studies and Proposed Biological Models
6.1. PfATP6 as the target
6.2. Heme as the activator and target
6.3. Mitochondria as the target
7. Mutations Affecting Artemisinin Resistance
8. Conclusions
Acknowledgements
References
- Enserink, M. Epidemiology: Lower malaria numbers reflect better estimates and a glimmer of hope. Science 2008, 321, 1620. [Google Scholar] [CrossRef] [PubMed]
- ter Kuile, F.; White, N.J.; Holloway, P.; Pasvol, G.; Krishna, S. Plasmodium falciparum: In vitro studies of the pharmacodynamic properties of drugs used for the treatment of severe malaria. Exp. Parasitol. 1993, 76, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Sen, R.; Bandyopadhyay, S.; Dutta, A.; Mandal, G.; Ganguly, S.; Saha, P.; Chatterjee, M. Artemisinin triggers induction of cell-cycle arrest and apoptosis in Leishmania donovani promastigotes. J. Med. Microbiol. 2007, 56, 1213–1218. [Google Scholar] [CrossRef] [PubMed]
- Utzinger, J.; N’Goran, E.K.; N’Dri, A.; Lengeler, C.; Xiao, S.; Tanner, M. Oral artemether for prevention of Schistosoma mansoni infection: Randomised controlled trial. Lancet 2000, 355, 1320–1325. [Google Scholar] [CrossRef]
- Utzinger, J.; Xiao, S.H.; Tanner, M.; Keiser, J. Artemisinins for schistosomiasis and beyond. Curr. Opin. Investig. Drugs 2007, 8, 105–116. [Google Scholar] [PubMed]
- Jones-Brando, L.; D’Angelo, J.; Posner, G.H.; Yolken, R. In vitro inhibition of Toxoplasma gondii by four new derivatives of artemisinin. Antimicrob. Agents Chemother. 2006, 50, 4206–4208. [Google Scholar] [CrossRef] [PubMed]
- Dunay, I.R.; Chan, W.C.; Haynes, R.K.; Sibley, L.D. Artemisone and artemiside control acute and reactivated toxoplasmosis in a murine model. Antimicrob. Agents Chemother. 2009, 53, 4450–4456. [Google Scholar] [CrossRef] [PubMed]
- Romero, M.R.; Serrano, M.A.; Vallejo, M.; Efferth, T.; Alvarez, M.; Marin, J.J. Antiviral effect of artemisinin from Artemisia annua against a model member of the Flaviviridae family, the bovine viral diarrhoea virus (BVDV). Planta Med. 2006, 72, 1169–1174. [Google Scholar] [CrossRef] [PubMed]
- Willoughby, J.A., Sr.; Sundar, S.N.; Cheung, M.; Tin, A.S.; Modiano, J.; Firestone, G.L. Artemisinin blocks prostate cancer growth and cell cycle progression by disrupting Sp1 interactions with the cyclin-dependent kinase-4 (CDK4) promoter and inhibiting CDK4 gene expression. J. Biol. Chem. 2009, 284, 2203–2213. [Google Scholar] [CrossRef] [PubMed]
- Li, P.C.; Lam, E.; Roos, W.P.; Zdzienicka, M.Z.; Kaina, B.; Efferth, T. Artesunate derived from traditional Chinese medicine induces DNA damage and repair. Cancer Res. 2008, 68, 4347–4351. [Google Scholar] [CrossRef] [PubMed]
- Svensson, U.S.; Ashton, M. Identification of the human cytochrome P450 enzymes involved in the in vitro metabolism of artemisinin. Br. J. Clin. Pharmacol. 1999, 48, 528–535. [Google Scholar] [CrossRef] [PubMed]
- The Qinghaosu Antimalarial Coordinating Research Group. Antimalaria studies on Qinghaosu (English). Chin. Med. J. 1979, 92, 811–816. [Google Scholar]
- Rosenthal, A.S.; Chen, X.; Liu, J.O.; West, D.C.; Hergenrother, P.J.; Shapiro, T.A.; Posner, G.H. Malaria-infected mice are cured by a single oral dose of new dimeric trioxane sulfones which are also selectively and powerfully cytotoxic to cancer cells. J. Med. Chem. 2009, 52, 1198–1203. [Google Scholar] [CrossRef] [PubMed]
- Woodard, L.E.; Chang, W.; Chen, X.; Liu, J.O.; Shapiro, T.A.; Posner, G.H. Malaria-infected mice live until at least day 30 after a new monomeric trioxane combined with mefloquine are administered together in a single low oral dose. J. Med. Chem. 2009, 52, 7458–7462. [Google Scholar] [CrossRef] [PubMed]
- The Research Group of Artemisinin Structure. A new sesquiterpene lactone-artemisinin. Chin. Sci. Bull. 1977, 22, 142. [Google Scholar]
- Xu, X.X.; Zhu, J.; Huang, D.; Zhou, W.S. Studies on structure and syntheses of arteannuin and related compound X. The stereocontrolled syntheses of arteannuin and deoxyarteannuin from arteannuic acid. Acta Chim. Sinica 1983, 41, 574–576. [Google Scholar]
- Schmid, G.; Hofheinz, W. Total synthesis of qinghaosu. J. Am. Chem. Soc. 1983, 105, 624–625. [Google Scholar] [CrossRef]
- Tsuruta, H.; Paddon, C.J.; Eng, D.; Lenihan, J.R.; Horning, T.; Anthony, L.C.; Regentin, R.; Keasling, J.D.; Renninger, N.S.; Newman, J.D. High-level production of amorpha-4,11-diene, a precursor of the antimalarial agent artemisinin, in Escherichia coli. PLoS One 2009, 4, e4489. [Google Scholar] [CrossRef] [PubMed]
- Arsenault, P.R.; Wobbe, K.K.; Weathers, P.J. Recent advances in artemisinin production through heterologous expression. Curr. Med. Chem. 2008, 15, 2886–2896. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.A.; Ji, R.Y. A quantitative structure-activity study on artemisinine analogues. Acta Pharmacol. Sin. 1982, 3, 55–60. [Google Scholar]
- Meshnick, S.R.; Taylor, T.E.; Kamchonwongpaisan, S. Artemisinin and the antimalarial endoperoxides: From herbal remedy to targeted chemotherapy. Microbiol. Rev. 1996, 60, 301–315. [Google Scholar] [PubMed]
- Lin, A.J.; Klayman, D.L. Thermal rearrangement and decomposition products of artemisinin (qinghaosu). J. Org. Chem. 1985, 50, 4504–4508. [Google Scholar] [CrossRef]
- Liu, J.M.; Ni, M.Y.; Fan, J.F.; Tu, Y.Y. Structure and reaction of arteannuin. Acta Chimica Sinica 1979, 37, 129–140. [Google Scholar]
- Zhou, W.S.; Wen, Y.C. Studies on structure and synthesis of arteannuin and its related compounds.VI the structures of arteannuin degradation products. Acta Chim. Sin. 1984, 42, 455–459. [Google Scholar]
- Liu, J.M.; Ni, M.Y.; Fan, J.F.; Tu, Y.Y. Structure and reaction of arteannuin. Acta Chim. Sin. 1979, 37, 129–141. [Google Scholar]
- Li, Y.; Yu, P.L.; Chen, Y.X. Studies on analoges of qinghaosu-some acidic degradations of qinghaosu (English). Kexue Tongbao 1986, 31, 1038–1040. [Google Scholar]
- Levander, O.A.; Ager, A.L., Jr.; Morris, V.C.; May, R.G. Qinghaosu, dietary vitamin E, selenium, and cod-liver oil: Effect on the susceptibility of mice to the malarial parasite Plasmodium yoelii. Am. J. Clin. Nutr. 1989, 50, 346–352. [Google Scholar] [CrossRef] [PubMed]
- Meshnick, S.R.; Tsang, T.W.; Lin, F.B.; Pan, H.Z.; Chang, C.N.; Kuypers, F.; Chiu, D.; Lubin, B. Activated oxygen mediates the antimalarial activity of qinghaosu. Prog. Clin. Biol. Res. 1989, 313, 95–104. [Google Scholar] [PubMed]
- Slater, A.F. Malaria pigment. Exp. Parasitol. 1992, 74, 362–365. [Google Scholar] [CrossRef]
- Meshnick, S.R.; Thomas, A.; Ranz, A.; Xu, C.M.; Pan, H.Z. Artemisinin (qinghaosu): The role of intracellular hemin in its mechanism of antimalarial action. Mol. Biochem. Parasitol. 1991, 49, 181–189. [Google Scholar] [CrossRef]
- Zhang, F.; Gosser, D.K., Jr.; Meshnick, S.R. Hemin-catalyzed decomposition of artemisinin (qinghaosu). Biochem. Pharmacol. 1992, 43, 1805–1809. [Google Scholar] [CrossRef]
- Meshnick, S.R.; Yang, Y.Z.; Lima, V.; Kuypers, F.; Kamchonwongpaisan, S.; Yuthavong, Y. Iron-dependent free radical generation from the antimalarial agent artemisinin (qinghaosu). Antimicrob. Agents Chemother. 1993, 37, 1108–1114. [Google Scholar] [CrossRef] [PubMed]
- Posner, G.H.; Oh, C.H. A Regiospecifically O-18 Labeled 1,2,4-trioxane, a simple chemical-model system to probe the mechanism(s) for the antimalarial activity of artemisinin Qinghaosu). J. Am. Chem. Soc. 1992, 114, 8328–8329. [Google Scholar] [CrossRef]
- O’Neill, P.M.; Posner, G.H. A medicinal chemistry perspective on artemisinin and related endoperoxides. J. Med. Chem. 2004, 47, 2945–2964. [Google Scholar] [CrossRef] [PubMed]
- Golenser, J.; Domb, A.; Leshem, B.; Kremsner, P.; Luty, A. Iron chelators as drugs against malaria pose a potential risk. Redox Rep. 2003, 8, 268–271. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.L.; Yang, Y.Z.; Meshnick, S.R. The interaction of artemisinin with malarial hemozoin. Mol. Biochem. Parasitol. 1994, 63, 121–128. [Google Scholar] [CrossRef]
- Cheng, F.; Shen, J.; Luo, X.; Zhu, W.; Gu, J.; Ji, R.; Jiang, H.; Chen, K. Molecular docking and 3-D-QSAR studies on the possible antimalarial mechanism of artemisinin analogues. Bioorg. Med. Chem. 2002, 10, 2883–2891. [Google Scholar] [CrossRef]
- Posner, G.H.; Wang, D.; Cumming, J.N.; Oh, C.H.; French, A.N.; Bodley, A.L.; Shapiro, T.A. Further evidence supporting the importance of and the restrictions on a carbon-centered radical for high antimalarial activity of 1,2,4-trioxanes like artemisinin. J. Med. Chem. 1995, 38, 2273–2275. [Google Scholar] [CrossRef] [PubMed]
- Kamchonwongpaisan, S.; Meshnick, S.R. The mode of action of the antimalarial artemisinin and its derivatives. Gen. Pharmacol. 1996, 27, 587–592. [Google Scholar] [CrossRef]
- Sibmooh, N.; Udomsangpetch, R.; Kujoa, A.; Chantharaksri, U.; Mankhetkorn, S. Redox reaction of artemisinin with ferrous and ferric ions in aqueous buffer. Chem. Pharm. Bull. (Tokyo) 2001, 49, 1541–1546. [Google Scholar] [CrossRef] [PubMed]
- Eckstein-Ludwig, U.; Webb, R.J.; Van Goethem, I.D.; East, J.M.; Lee, A.G.; Kimura, M.; O’Neill, P.M.; Bray, P.G.; Ward, S.A.; Krishna, S. Artemisinins target the SERCA of Plasmodium falciparum. Nature 2003, 424, 957–961. [Google Scholar] [CrossRef] [PubMed]
- Stocks, P.A.; Bray, P.G.; Barton, V.E.; Al-Helal, M.; Jones, M.; Araujo, N.C.; Gibbons, P.; Ward, S.A.; Hughes, R.H.; Biagini, G.A.; Davies, J.; Amewu, R.; Mercer, A.E.; Ellis, G.; O’Neill, P.M. Evidence for a common non-heme chelatableiron-dependent activation mechanism for semisynthetic and synthetic endoperoxide antimalarial drugs. Angew. Chem. Int. Ed. Engl. 2007, 46, 6278–6283. [Google Scholar] [CrossRef] [PubMed]
- Haynes, R.K.; Ho, W.Y.; Chan, H.W.; Fugmann, B.; Stetter, J.; Croft, S.L.; Vivas, L.; Peters, W.; Robinson, B.L. Highly antimalaria-active artemisinin derivatives: Biological activity does not correlate with chemical reactivity. Angew. Chem. Int. Ed. Engl. 2004, 43, 1381–1385. [Google Scholar] [CrossRef] [PubMed]
- Morel, I.; Cillard, J.; Lescoat, G.; Sergent, O.; Pasdeloup, N.; Ocaktan, A.Z.; Abdallah, M.A.; Brissot, P.; Cillard, P. Antioxidant and free radical scavenging activities of the iron chelators pyoverdin and hydroxypyrid-4-ones in iron-loaded hepatocyte cultures: Comparison of their mechanism of protection with that of desferrioxamine. Free Radic Biol. Med. 1992, 13, 499–508. [Google Scholar] [CrossRef]
- Barbouti, A.; Doulias, P.T.; Zhu, B.Z.; Frei, B.; Galaris, D. Intracellular iron, but not copper, plays a critical role in hydrogen peroxide-induced DNA damage. Free Radic Biol. Med. 2001, 31, 490–498. [Google Scholar] [CrossRef]
- Haynes, R.K.; Pai, H.H.O.; Voerste, A. Ring opening of artemisinin (qinghaosu) and dihydroartemisinin and interception of the open hydroperoxideswith formation of N-oxides-A chemicalmodel for antimalarialmode of action. Tetrahedron Lett. 1999, 40, 4715–4718. [Google Scholar] [CrossRef]
- Haynes, R.K.; Vonwiller, S.C. The behaviour of qinghaosu (artemisinin) in the presence of non-heme Iron(II) and (III). Tetrahedron Lett. 1996, 37, 257–260. [Google Scholar] [CrossRef]
- Haynes, R.K.; Chan, W.C.; Lung, C.-M.; Uhlemann, A.-C.; Eckstein, U.; Taramelli, D.; Parapini, S.; Monti, D.; Krishna, S. The Fe2-mediated decomposition, PfATP6 binding, and antimalarial activities of artemisone and other artemisinins: The unlikelihood of C-entered radicals as bioactive intermediates. ChemMedChem 2007, 2, 1480–1497. [Google Scholar] [PubMed]
- Posner, G.H.; Parker, M.H.; Northrop, J.; Elias, J.S.; Ploypradith, P.; Xie, S.; Shapiro, T.A. Orally active, hydrolytically stable, semisynthetic, antimalarial trioxanes in the artemisinin family. J. Med. Chem. 1999, 42, 300–304. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, P.M.; Searle, N.L.; Kan, K.W.; Storr, R.C.; Maggs, J.L.; Ward, S.A.; Raynes, K.; Park, B.K. Novel, potent, semisynthetic antimalarial carba analogues of the first-generation 1,2,4-trioxane artemether. J. Med. Chem. 1999, 42, 5487–5493. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Weiss, E.; Kyle, D.E.; Ziffer, H. Acid catalyzed Michael additions to artemisitene. Bioorg. Med. Chem. Lett. 2000, 10, 1601–1603. [Google Scholar] [CrossRef]
- Jung, M.; Lee, K.; Jung, H. First synthesis of (1)-deoxoartemisitene and its novel C-11 derivatives. Tetrahedron Lett. 2001, 42, 3997–4000. [Google Scholar] [CrossRef]
- Jefford, C.W.; McGoran, E.C.; Boukouvalas, J.; Richardson, G.; Robinson, B.L.; Peters, W. Synthesis of new 1,2,4-trioxanes and their antimalarial activity. Helv. Chim. Acta 1988, 1805–1812. [Google Scholar] [CrossRef]
- Jefford, C.W.; Velarde, J.A.; Bernardinelli, G.; Bray, D.H.; Warhurst, D.C.; Milhous, W.K. Synthesis, structure, and antimalarial activity of tricyclic 1,2,4-trioxanes related to artemisinin. Helv. Chim. Acta 1993, 76, 2775–2788. [Google Scholar] [CrossRef]
- Kepler, J.A.; Philip, A.; Lee, Y.W.; Musallam, H.A.; Carroll, F.I. Endoperoxides as potential antimalarial agents. J. Med. Chem. 1987, 30, 1505–1509. [Google Scholar] [CrossRef] [PubMed]
- Borstnik, K.; Paik, I.H.; Shapiro, T.A.; Posner, G.H. Antimalarial chemotherapeutic peroxides: Artemisinin, yingzhaosu A and related compounds. Int. J. Parasitol. 2002, 32, 1661–1667. [Google Scholar] [CrossRef]
- Vennerstrom, J.L.; Fu, H.N.; Ellis, W.Y.; Ager, A.L., Jr.; Wood, J.K.; Andersen, S.L.; Gerena, L.; Milhous, W.K. Dispiro-1,2,4,5-tetraoxanes: A new class of antimalarial peroxides. J. Med. Chem. 1992, 35, 3023–3027. [Google Scholar] [CrossRef] [PubMed]
- Peters, W.; Robinson, B.L.; Rossier, J.C.; Jefford, C.W. The chemotherapy of rodent malaria. XLVIII. The activities of some synthetic 1,2,4-trioxanes against chloroquine-sensitive and chloroquine-resistant parasites. Part 1: Studies leading to the development of novel cis-fused cyclopenteno derivatives. Ann. Trop.Med. Parasitol. 1993, 87, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Peters, W.; Robinson, B.L.; Rossier, J.C.; Misra, D.; Jefford, C.W.; Rossiter, J.C. The chemotherapy of rodent malaria. XLIX. The activities of some synthetic 1,2,4-trioxanes against chloroquine-sensitive and chloroquine-resistant parasites. Part 2: Structure-activity studies on cis-fused cyclopenteno-1,2,4-trioxanes (fenozans) against drug-sensitive and drug-resistant lines of Plasmodium berghei and P. yoelii ssp. NS in vivo. Ann. Trop. Med. Parasitol. 1993, 87, 9–16. [Google Scholar] [PubMed]
- Vennerstrom, J.L.; Dong, Y.; Andersen, S.L.; Ager, A.L., Jr.; Fu, H.; Miller, R.E.; Wesche, D.L.; Kyle, D.E.; Gerena, L.; Walters, S.M.; Wood, J.K.; Edwards, G.; Holme, A.D.; McLean, W.G.; Milhous, W.K. Synthesis and antimalarial activity of sixteen dispiro-1,2,4, 5-tetraoxanes: Alkyl-substituted 7,8,15,16-tetraoxadispiro[5.2.5.2]hexadecanes. J. Med. Chem. 2000, 43, 2753–2758. [Google Scholar] [CrossRef] [PubMed]
- Posner, G.H.; Jeon, H.B.; Parker, M.H.; Krasavin, M.; Paik, I.H.; Shapiro, T.A. Antimalarial simplified 3-aryltrioxanes: Synthesis and preclinical efficacy/toxicity testing in rodents. J. Med. Chem. 2001, 44, 3054–3058. [Google Scholar] [CrossRef] [PubMed]
- Posner, G.H.; Cumming, J.N.; Woo, S.H.; Ploypradith, P.; Xie, S.; Shapiro, T.A. Orally active antimalarial 3-substituted trioxanes: New synthetic methodology and biological evaluation. J. Med. Chem. 1998, 41, 940–951. [Google Scholar] [CrossRef] [PubMed]
- Vennerstrom, J.L.; Arbe-Barnes, S.; Brun, R.; Charman, S.A.; Chiu, F.C.; Chollet, J.; Dong, Y.; Dorn, A.; Hunziker, D.; Matile, H.; McIntosh, K.; Padmanilayam, M.; Santo Tomas, J.; Scheurer, C.; Scorneaux, B.; Tang, Y.; Urwyler, H.; Wittlin, S.; Charman, W.N. Identification of an antimalarial synthetic trioxolane drug development candidate. Nature 2004, 430, 900–904. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, P.M.; Rawe, S.L.; Borstnik, K.; Miller, A.; Ward, S.A.; Bray, P.G.; Davies, J.; Oh, C.H.; Posner, G.H. Enantiomeric 1,2,4-trioxanes display equivalent in vitro antimalarial activity versus Plasmodium falciparum malaria parasites: Implications for the molecular mechanism of action of the artemisinins. ChemBioChem 2005, 6, 2048–2054. [Google Scholar] [CrossRef] [PubMed]
- Jefford, C.W. Synthesis, structure, and antimalarial activity of some enantiomerically pure, cis-fused cyclopenteno-1,2,4-trioxanes. Helv. Chim. Acta 1995, 78, 647–662. [Google Scholar] [CrossRef]
- Najjar, F.; Gorrichon, L.; Baltas, M.; Andre-Barres, C.; Vial, H. Alkylation of natural endoperoxide G3-factor. Synthesis and antimalarial activity studies. Org. Biomol. Chem. 2005, 3, 1612–1614. [Google Scholar] [CrossRef] [PubMed]
- Joris, J.; Costello, A.; Dubner, R.; Hargreaves, K.M. Opiates suppress carrageenan-induced edema and hyperthermia at doses that inhibit hyperalgesia. Pain 1990, 43, 95–103. [Google Scholar] [CrossRef]
- Olliaro, P.L.; Haynes, R.K.; Meunier, B.; Yuthavong, Y. Possible modes of action of the artemisinin-type compounds. Trends Parasitol. 2001, 17, 122–126. [Google Scholar] [CrossRef]
- del Pilar Crespo, M.; Avery, T.D.; Hanssen, E.; Fox, E.; Robinson, T.V.; Valente, P.; Taylor, D.K.; Tilley, L. Artemisinin and a series of novel endoperoxide antimalarials exert early effects on digestive vacuole morphology. Antimicrob. Agents Chemother. 2008, 52, 98–109. [Google Scholar] [CrossRef] [PubMed]
- Jung, M.; Kim, H.; Nam, K.Y.; No, K.T. Three-dimensional structure of Plasmodium falciparum Ca2+ -ATPase(PfATP6) and docking of artemisinin derivatives to PfATP6. Bioorg. Med. Chem. Lett. 2005, 15, 2994–2997. [Google Scholar] [CrossRef] [PubMed]
- Garah, F.B.; Stigliani, J.L.; Cosledan, F.; Meunier, B.; Robert, A. Docking studies of structurally diverse antimalarial drugs targeting PfATP6: No correlation between in silico binding affinity and in vitro antimalarial activity. ChemMedChem 2009, 4, 1469–1479. [Google Scholar] [CrossRef] [PubMed]
- Uhlemann, A.C.; Cameron, A.; Eckstein-Ludwig, U.; Fischbarg, J.; Iserovich, P.; Zuniga, F.A.; East, M.; Lee, A.; Brady, L.; Haynes, R.K.; Krishna, S. A single amino acid residue can determine the sensitivity of SERCAs to artemisinins. Nat. Struct. Mol. Biol. 2005, 12, 628–629. [Google Scholar] [CrossRef] [PubMed]
- Jambou, R.; Legrand, E.; Niang, M.; Khim, N.; Lim, P.; Volney, B.; Ekala, M.T.; Bouchier, C.; Esterre, P.; Fandeur, T.; Mercereau-Puijalon, O. Resistance of Plasmodium falciparum field isolates to in-vitro artemether and point mutations of the SERCA-type PfATPase6. Lancet 2005, 366, 1960–1963. [Google Scholar] [CrossRef]
- Cojean, S.; Hubert, V.; Le Bras, J.; Durand, R. Resistance to dihydroartemisinin. Emerg. Infect. Dis. 2006, 12, 1798–1799. [Google Scholar] [CrossRef] [PubMed]
- Jefford, C.W. New developments in synthetic peroxidic drugs as artemisinin mimics. Drug Discov. Today 2007, 12, 487–495. [Google Scholar] [CrossRef] [PubMed]
- Robert, A.; Coppel, Y.; Meunier, B. Alkylation of heme by the antimalarial drug artemisinin. Chem. Comm. 2002, 414–415. [Google Scholar] [CrossRef]
- Pandey, A.V.; Tekwani, B.L.; Singh, R.L.; Chauhan, V.S. Artemisinin, an endoperoxide antimalarial, disrupts the hemoglobin catabolism and heme detoxification systems in malarial parasite. J. Biol. Chem. 1999, 274, 19383–19388. [Google Scholar] [CrossRef] [PubMed]
- Robert, A.; Benoit-Vical, C.; Claparols, C.; Meunier, B. The antimalarial drug artemisinin alkylates heme in infected mice. Proc. Natl. Acad. Sci. 2005, 102, 13676–13680. [Google Scholar] [CrossRef] [PubMed]
- Kannan, R.; Kumar, K.; Sahal, D.; Kukreti, S.; Chauhan, V.S. Reaction of artemisinin with haemoglobin: Implications for antimalarial activity. Biochem. J. 2005, 385, 409–418. [Google Scholar] [CrossRef] [PubMed]
- Kannan, R.; Sahal, D.; Chauhan, V.S. Heme-artemisinin adducts are crucial mediators of the ability of artemisinin to inhibit heme polymerization. Chem. Biol. 2002, 9, 321–332. [Google Scholar] [CrossRef]
- Loup, C.; Lelievre, J.; Benoit-Vical, F.; Meunier, B. Trioxaquines and heme-artemisinin adducts inhibit the in vitro formation of hemozoin better than chloroquine. Antimicrob. Agents Chemother. 2007, 51, 3768–3770. [Google Scholar] [CrossRef] [PubMed]
- Francis, S.E.; Sullivan, D.J.; Goldberg, D.E. Hemoglobin metabolism in the malarial parasite Plasmodium falciparum. Annu. Rev. Microbiol. 1997, 51, 97–123. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, M.; Wittlin, S.; Nehrbass-Stuedli, A.; Dong, Y.; Wang, X.; Hemphill, A.; Matile, H.; Brun, R.; Vennerstrom, J.L. Peroxide Bond-dependent antiplasmodial specificity of artemisinin and OZ277 (RBx11160). Antimicrob. Agents Chemother. 2007, 51, 2991–2993. [Google Scholar] [CrossRef] [PubMed]
- Elliott, D.A.; McIntosh, M.T.; Hosgood III, H.D.; Chen, S.; Zhang, G.; Baevova, P.; Joiner, K.A. Four distinct pathways of hemoglobin uptake in the malaria parasite Plasmodium falciparum. Proc. Natl. Acad. Sci. USA 2008, 105, 2463–2468. [Google Scholar] [CrossRef] [PubMed]
- Paitayatat, S.; Tarnchompoo, B.; Thebtaranonth, Y.; Yuthavong, Y. Correlation of antimalarial activity of artemisinin derivatives with binding affinity with ferroprotoporphyrin IX. J. Med. Chem. 1997, 40, 633–638. [Google Scholar] [CrossRef] [PubMed]
- Peters, W.; Li, Z.L.; Robinson, B.L.; Warhurst, D.C. The chemotherapy of rodent malaria, XL. The action of artemisinin and related sesquiterpenes. Ann. Trop. Med. Parasitol. 1986, 80, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Wittner, M.; Lederman, J.; Tanowitz, H.B.; Rosenbaum, G.S.; Weiss, L.M. Atovaquone in the treatment of Babesia microti infections in hamsters. Am. J. Trop. Med. Hyg. 1996, 55, 219–222. [Google Scholar] [CrossRef] [PubMed]
- Asawamahasakda, W.; Ittarat, I.; Chang, C.C.; McElroy, P.; Meshnick, S.R. Effects of antimalarials and protease inhibitors on plasmodial hemozoin production. Mol. Biochem. Parasitol. 1994, 67, 183–191. [Google Scholar] [CrossRef]
- Haynes, R.K.; Monti, D.; Taramelli, D.; Basilico, N.; Parapini, S.; Olliaro, P. Artemisinin antimalarials do not inhibit hemozoin formation. Antimicrob. Agents Chemother. 2003, 47, 1175. [Google Scholar] [CrossRef] [PubMed]
- Meshnick, S.R. Artemisinin and heme. Antimicrob. Agents Chemother. 2003, 47, 2712; author reply 2712–2713. [Google Scholar] [CrossRef] [PubMed]
- Coghi, P.; Basilico, N.; Taramelli, D.; Chan, W.C.; Haynes, R.K.; Monti, D. Interaction of Artemisinins with Oxyhemoglobin Hb-FeII, Hb-FeII, CarboxyHb-FeII, Heme-FeII, and Carboxyheme FeII: Significance for Mode of action and implications for therapy of cerebral malaria. ChemMedChem 2009, 4, 2045–2053. [Google Scholar] [CrossRef] [PubMed]
- Asawamahasakda, W.; Ittarat, I.; Pu, Y.M.; Ziffer, H.; Meshnick, S.R. Reaction of antimalarial endoperoxides with specific parasite proteins. Antimicrob. Agents Chemother. 1994, 38, 1854–1858. [Google Scholar] [CrossRef] [PubMed]
- Bhisutthibhan, J.; Pan, X.Q.; Hossler, P.A.; Walker, D.J.; Yowell, C.A.; Carlton, J.; Dame, J.B.; Meshnick, S.R. The Plasmodium falciparum translationally controlled tumor protein homolog and its reaction with the antimalarial drug artemisinin. J. Biol. Chem. 1998, 273, 16192–16198. [Google Scholar] [CrossRef] [PubMed]
- Chae, J.; Choi, I.; Kim, C. Homology modeling and molecular docking study of translationally controlled tumor protein and artemisinin. Arch. Pharm. Res. 2006, 29, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Mo, W.; Shen, D.; Sun, L.; Wang, J.; Lu, S.; Gitschier, J.M.; Zhou, B. Yeast model uncovers dual roles of mitochondria in action of artemisinin. PLoS Genet. 2005, 1, e36. [Google Scholar] [CrossRef] [PubMed]
- Ginsburg, H.; Divo, A.A.; Geary, T.G.; Boland, M.T.; Jensen, J.B. Effects of mitochondrial inhibitors on intraerythrocytic Plasmodium falciparum in in vitro cultures. J. Protozool. 1986, 33, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, I.K.; Rottenberg, H.; Vaidya, A.B. Atovaquone, a broad spectrum antiparasitic drug, collapses mitochondrial membrane potential in a malarial parasite. J. Biol. Chem. 1997, 272, 3961–3966. [Google Scholar] [CrossRef] [PubMed]
- Hartwig, C.L.; Rosenthal, A.S.; D’ Angelo, J.; Griffin, C.E.; Posner, G.H.; Cooper, R.A. Accumulation of artemisinin trioxane derivatives within neutral lipids of Plasmodium falciparum malaria parasites is endoperoxide-dependent. Biochem. Pharmacol. 2009, 77, 322–336. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.L. Use of nuclear techniques in the study of the antimalarial action of Qinghaosu and dihydro-Qinghaosu. Nucl. Techniq. 1984, 2, 23–25. [Google Scholar]
- Maeno, Y.; Toyoshima, T.; Fujioka, H.; Ito, Y.; Meshnick, S.R.; Benakis, A.; Milhous, W.K.; Aikawa, M. Morphologic effects of artemisinin in Plasmodium falciparum. Am. J. Trop. Med. Hyg. 1993, 49, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Ellis, D.S.; Li, Z.L.; Gu, H.M.; Peters, W.; Robinson, B.L.; Tovey, G.; Warhurst, D.C. The chemotherapy of rodent malaria, XXXIX. Ultrastructural changes following treatment with artemisinine of Plasmodium berghei infection in mice, with observations of the localization of [3H]-dihydroartemisinine in P. falciparum in vitro. Ann. Trop. Med. Parasitol. 1985, 79, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.B.; Jacobs, G.; Liang, D.S.; Aikawa, M. Qinghaosu-induced changes in the morphology of Plasmodium inui. Am. J. Trop. Med. Hyg. 1985, 34, 424–428. [Google Scholar] [CrossRef] [PubMed]
- Kawai, S.; Kano, S.; Suzuki, M. Morphologic effects of artemether on Plasmodium falciparum in Aotus trivirgatus. Am. J. Trop. Med. Hyg. 1993, 49, 812–818. [Google Scholar] [CrossRef] [PubMed]
- Dondorp, A.M.; Nosten, F.; Yi, P.; Das, D.; Phyo, A.P.; Tarning, J.; Lwin, K.M.; Ariey, F.; Hanpithakpong, W.; Lee, S.J.; Ringwald, P.; Silamut, K.; Imwong, M.; Chotivanich, K.; Lim, P.; Herdman, J.; An, S.S.; Yeung, S.; Singhasivanon, P.; Day, N.P.; Lindegardh, N.; Socheat, D. Artemisinin resistance in Plasmodium falciparum malaria. N. Engl. J. Med. 2009, 361, 455–467. [Google Scholar] [CrossRef] [PubMed]
- Noedl, H.; Se, Y.; Schaecher, K.; Smith, B.L.; Socheat, D.; Fukuda, M.M. Evidence of artemisinin-resistant malaria in western Cambodia. N. Engl. J. Med. 2008, 359, 2619–2620. [Google Scholar] [CrossRef] [PubMed]
- Noedl, H.; Socheat, D.; Satimai, W. Artemisinin-resistant malaria in Asia. N. Engl. J. Med. 2009, 361, 540–541. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Zhou, B. Exploring artemisinins protein targets in yeast. J. Tsinghua Univ. (Sci. & Tech.) 2008, 43, 408–411. [Google Scholar]
- Price, R.N.; Uhlemann, A.C.; Brockman, A.; McGready, R.; Ashley, E.; Phaipun, L.; Patel, R.; Laing, K.; Looareesuwan, S.; White, N.J.; Nosten, F.; Krishna, S. Mefloquine resistance in Plasmodium falciparum and increased pfmdr1 gene copy number. Lancet 2004, 364, 438–447. [Google Scholar] [CrossRef]
- Sidhu, A.B.; Verdier-Pinard, D.; Fidock, D.A. Chloroquine resistance in Plasmodium falciparum malaria parasites conferred by pfcrt mutations. Science 2002, 298, 210–213. [Google Scholar] [CrossRef] [PubMed]
- Pickard, A.L.; Wongsrichanalai, C.; Purfield, A.; Kamwendo, D.; Emery, K.; Zalewski, C.; Kawamoto, F.; Miller, R.S.; Meshnick, S.R. Resistance to antimalarials in Southeast Asia and genetic polymorphisms in pfmdr1. Antimicrob. Agents Chemother. 2003, 47, 2418–2423. [Google Scholar] [CrossRef] [PubMed]
- Afonso, A.; Hunt, P.; Cheesman, S.; Alves, A.C.; Cunha, C.V.; do Rosario, V.; Cravo, P. Malaria parasites can develop stable resistance to artemisinin but lack mutations in candidate genes atp6 (encoding the sarcoplasmic and endoplasmic reticulum Ca2+ ATPase), tctp, mdr1, and cg10. Antimicrob. Agents Chemother. 2006, 50, 480–489. [Google Scholar] [CrossRef] [PubMed]
- Inselburg, J. Induction and isolation of artemisinine-resistant mutants of Plasmodium falciparum. Am. J. Trop. Med. Hyg. 1985, 34, 417–418. [Google Scholar] [CrossRef] [PubMed]
- Peters, W.; Robinson, B.L. The chemotherapy of rodent malaria. LVI. Studies on the development of resistance to natural and synthetic endoperoxides. Ann. Trop. Med. Parasitol. 1999, 93, 325–329. [Google Scholar] [CrossRef] [PubMed]
- Meshnick, S.R. Artemisinin: Mechanisms of action, resistance and toxicity. Int. J. Parasitol. 2002, 32, 1655–1660. [Google Scholar] [CrossRef]
Sample Availability: Not available. |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, J.; Zhou, B. Biological Actions of Artemisinin: Insights from Medicinal Chemistry Studies. Molecules 2010, 15, 1378-1397. https://doi.org/10.3390/molecules15031378
Li J, Zhou B. Biological Actions of Artemisinin: Insights from Medicinal Chemistry Studies. Molecules. 2010; 15(3):1378-1397. https://doi.org/10.3390/molecules15031378
Chicago/Turabian StyleLi, Jian, and Bing Zhou. 2010. "Biological Actions of Artemisinin: Insights from Medicinal Chemistry Studies" Molecules 15, no. 3: 1378-1397. https://doi.org/10.3390/molecules15031378
APA StyleLi, J., & Zhou, B. (2010). Biological Actions of Artemisinin: Insights from Medicinal Chemistry Studies. Molecules, 15(3), 1378-1397. https://doi.org/10.3390/molecules15031378



