Revealing the Power of the Natural Red Pigment Lycopene
Abstract
:1. Introduction
2. Lycopene in Food and Its Properties
| Molecular formula | C40H56 |
| Molecular weight | 536.85 Da |
| Melting point | 172–175 ºC |
| Crystal form | Long red needles separate from a mixture of carbon disulfide and ethanol |
| Powder form | Dark reddish-brown |
| Solubility | Soluble in chloroform, hexane, benzene, carbon disulfide, acetone, petroleum ether and oil; |
| Insoluble in water, ethanol and methanol | |
| Stability | Sensitive to light, oxygen, high temperature, acids, catalyst and metal ions |
3. Lycopene Absorption, Transportation and Distribution in Human
3.1. Absorption
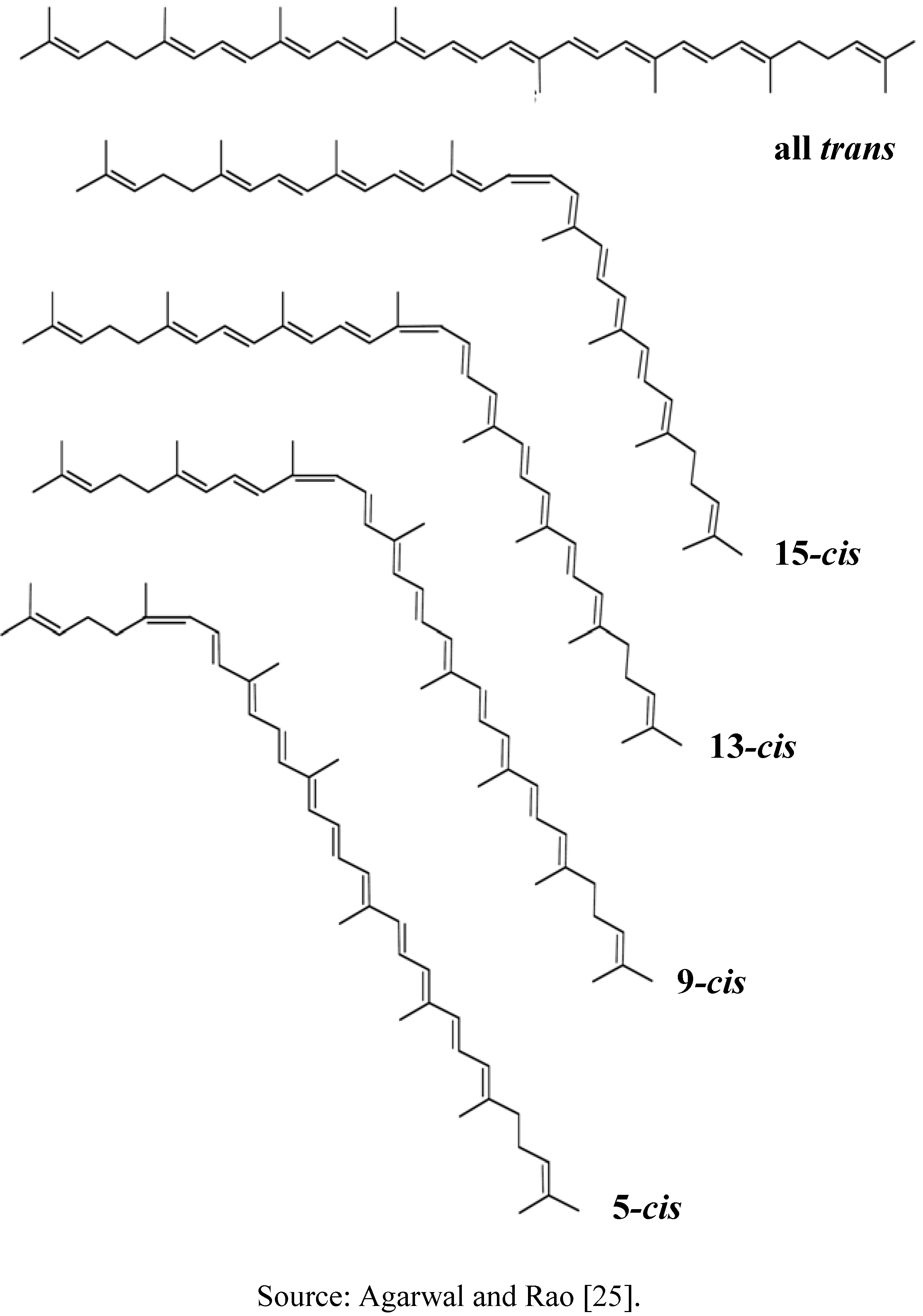
3.2. Transportation
| References | Country | Plasma lycopene levels (µmol/L) | |
|---|---|---|---|
| Male | Female | ||
| [49] | UK | - | 0.32 ± 0.12 |
| [50] | USA | 0.82 ± 0.38 | 0.76 ± 0.32 |
| [51] | France | 0.66 (0.18-1.47) | 0.66 (0.31-2.06) |
| Republic of Ireland | 0.73 (0.09-2.12) | 0.57 (0.09-0.65) | |
| The Netherland | 0.54 (0.08-1.72) | 0.53 (0.04-1.98) | |
| Spain | 0.53 (0.21-1.16) | 0.51 (0.07-1.72) | |
| Ireland | 0.30 ± 0.13 | 0.25 ± 0.11 | |
| [52] | Italy (Varese/Turin) | 1.03 ± 0.43 | 0.90 ± 0.37 |
| Italy (Florence) | 1.01 ± 0.37 | 0.90 ± 0.36 | |
| Italy (Ragusa/Naples) | 1.29 ± 0.46 | 1.32 ± 0.46 | |
| Greece (Athens) | 0.90 ± 0.38 | 0.87 ± 0.47 | |
| Spain (Granada) | 0.69 ± 0.40 | 0.69 ± 0.33 | |
| Spain (Murcia) | 0.66 ± 0.30 | 0.74 ± 0.35 | |
| Northern Spain | 0.53 ± 0.31 | 0.43 ± 0.29 | |
| UK (vegetarians) | 0.98 ± 0.45 | 0.89 ± 0.44 | |
| UK (Cambridge) | 0.72 ± 0.30 | 0.77 ± 0.38 | |
| Germany (Potsdam) | 0.60 ± 0.30 | 0.69 ± 0.33 | |
| Germany (Heidelberg) | 0.62 ± 0.31 | 0.54 ± 0.25 | |
| The Netherlands | 0.54 ± 0.33 | 0.47 ± 0.26 | |
| Denmark | 0.58 ± 0.34 | 0.53 ± 0.29 | |
| Sweden (Malmö) | 0.46 ± 0.24 | 0.52 ± 0.27 | |
| Sweden (Umeå) | 0.56 ±0.37 | 0.44 ± 0.25 | |
| [53] | Japan | 0.11 (0.04-0.33) | 0.20 (0.08-0.52) |
| [54] | Thailand | 0.46 ± 0.33 | 0.74 ± 0.38 |
3.3. Distribution
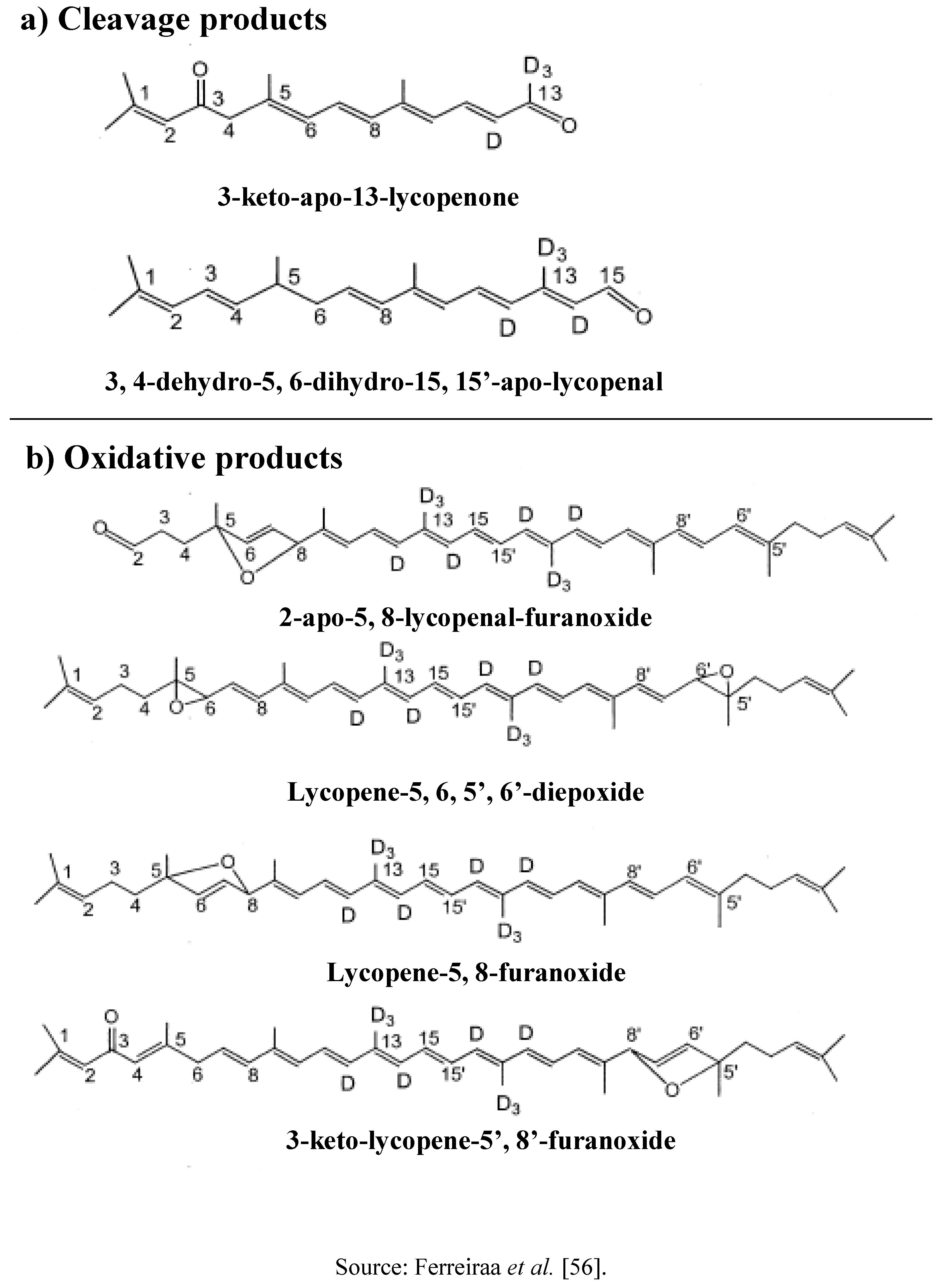
4. Lycopene as Antioxidant and Its Mechanism of Function

 ROO-lycopene•
ROO-lycopene•  ROO-lycopene-OO•
ROO-lycopene-OO• ROO-lycopene-OOH + R•
ROO-lycopene-OOH + R• ROO•
ROO• inactive products
inactive products NO2- + Lycopene+•
NO2- + Lycopene+• [CCl3O2- Lycopene]•
[CCl3O2- Lycopene]•  CCl3O2- + Lycopene+•
CCl3O2- + Lycopene+•  Lycopene -• + O2
Lycopene -• + O2  Lycopene• + ROOH
Lycopene• + ROOH5. Interaction of Lycopene with Other Antioxidants
 TOH + Lycopene+˙
TOH + Lycopene+˙ Lycopene + ASCH˙ + H+
Lycopene + ASCH˙ + H+ Lycopene + ASCH˙- + H+
Lycopene + ASCH˙- + H+ Carotenoid + Lycopene+˙
Carotenoid + Lycopene+˙ α-TOH + Lycopene+˙
α-TOH + Lycopene+˙ α-TO˙ + Lycopene
α-TO˙ + Lycopene α-TO˙ + Lycopene
α-TO˙ + Lycopene δ-TOH + Lycopene+˙
δ-TOH + Lycopene+˙ α-TOH- + Lycopene+˙
α-TOH- + Lycopene+˙ Lycopene + Asc-˙+ H+
Lycopene + Asc-˙+ H+6. Preventive Effect of Lycopene toward Diseases
6.1. Oxidative stress
6.3. Cancers
6.4. Diabetes
6.5. Other health benefits and safety aspect
| Lycopene doses | Method | Impairment | Improvement | Lit. cited |
|---|---|---|---|---|
| 0.2 mg/kg b.w. daily | In vivo–rats | Cataract | Significant delayed in the onset and progression of galactose cataract and reduced the incidence of selenite cataract. | 145 |
| 2.5, 5 and 10 mg/kg b.w. daily | In vivo–rats | Cognitive function | Significant improved in memory. | 143 |
| 60 mg/kg b.w. daily | In vivo–hyperlipemia rabbits | Lipid peroxidation injury | Significant reduced in the levels of serum TG and MDA, increase serum SOD activity, increase serum NO. | 146 |
| 0.1, 0.5, 1, 2 g/kg b.w. daily | In vivo– mouse ear oedema model | Swelling | Decreased swelling of the croton oil-induced ear. | 147 |
| 0, 5 and 10 μg/mLcarried by liposomes | In vitro–Calu-3 cells | Inflammation of cells infected by rhinovirus or exposed to lipopolysaccharide | Reduced the release of interleukin-6 and interferon-gamma induced protein-10. | 148 |
| 8 or 16 mg/kg/day by i.p. injection | In vivo–murine model of asthma | Ovalbumin-induced inflammation | Significant inhibition of the infiltration of inflammatory immunocytes into the bronchoalveolar lavage. | 149 |
| 2 mgtwice daily | In vivo–primigravida women | Pre-eclampsia and intrauterinegrowth retardation | Significant reduced in pre-eclamsia incidence and intrauterine growth retardation in the lycopene group compare to placebo group. | 150 |
| 9 mg/kg b.w. twice a day for 2 weeks | In vivo–rats | Chronic bacterial prostatitis | Significant decreased in bacterial growth and improvement of prostatic inflammation. | 151 |
| 0.025–2 mg per 20 mg b.w. | In vivo–white heterozygote mouse | X-ray radiation lesions | Moderate curative effect on the radiation lesions and increased survival rate | 152 |
7. Lycopene-Rich By-Products from Food Processing
| Country | By-products | References |
|---|---|---|
| Algeria | Tomato skin | 160 |
| Argentina | Tomato skin | 161 |
| Canada | Tomato skin | 162 |
| China, Canada | Tomato paste waste | 163 |
| China | Tomato paste waste | 164 |
| Tomato paste waste | 165 | |
| India | Mace (Myristica fragrans) | 166 |
| Tomato peels and seeds, tomato industrial waste | 167 | |
| Tomato skin | 20 | |
| Iraq | Tomato skin | 158 |
| Italy | Tomato peels and seeds | 168 |
| Tomato peels | 169 | |
| Hungary | Tomato pomace | 19 |
| Japan | Tomato skin | 18 |
| Portugal, Brazil | Tomato skin and seeds | 170 |
| Spain | Tomato peels | 171 |
| Taiwan | Tomato pulp waste | 172 |
| Turkey, Netherland | Tomato paste waste | 173 |
| USA | Tomato pomace | 174 |
8. Thermal Process on Lycopene Content
9. Conclusions
References and Notes
- Mortensen, A. Carotenoids and other pigments as natural colorants. Pure Appl. Chem. 2006, 78, 1477–1491. [Google Scholar] [CrossRef]
- Vogele, A.C. Effect of environmental factors upon the color of the tomato and the watermelon. Plant Physiol. 1937, 12, 929–955. [Google Scholar] [CrossRef]
- Rodriguez-Amaya, D.B.; Kimura, M. Carotenoids in foods. In Harvestplus Handbook for Carotenoid Analysis; IFPRI and CIAT: Washington, DC, USA, 2004; pp. 2–7. [Google Scholar]
- Rodriguez-Amaya, D.B. A Guide to Carotenoid Analysis in Foods; ILSI Press: Washington, D.C., USA, 2001; pp. 1–45. [Google Scholar]
- Focus on Pigments. World spends more than $50 M on lycopene red. Focus Pigm. 2007, 4, 3–4.
- Rao, A.V.; Argawal, S. Role of lycopene as antioxidant carotenoid in the prevention of chronic diseases: A review. Nutr. Res. 1999, 19, 305–323. [Google Scholar] [CrossRef]
- Scordino, M.; Di Mauro, A.; Passerini, A.; Maccarone, E. Selective recovery of anthocyanins and hydroxycinnamates from a byproduct of citrus processing. J. Agric. Food Chem. 2005, 53, 651–658. [Google Scholar] [CrossRef]
- Amin, I.; Mukhrizah, O. Antioxidant capacity of methanolic and water extracts prepared from food-processing by-products. J. Sci. Food Agric. 2006, 86, 778–784. [Google Scholar] [CrossRef]
- Ajila, C.M.; Naidu, K.A.; Bhat, S.G.; Prasada Rao, U.J.S. Bioactive compounds and antioxidant potential of mango peel extract. Food Chem. 2007, 105, 982–988. [Google Scholar] [CrossRef]
- Abdalla, A.E.M.; Darwish, S.M.; Ayad, E.H.E.; El-Hamahmy, R.M. Egyptian mango by-product 2: Antioxidant and antimicrobial activities of extract and oil from mango seed kernel. Food Chem. 2007, 103, 1141–1152. [Google Scholar] [CrossRef]
- Correia, R.T.P.; Mccue, P.; Magalhães, M.M.A.; Macêdo, G.R.; Shetty, K. Phenolic antioxidant enrichment of soy flour supplemented guava waste by rizhopus oligosporus-mediated solid-state bioprocessing. J. Food Biochem. 2004, 28, 404–418. [Google Scholar] [CrossRef]
- Bernardino-Nicanor, A.; Anón, M.C.; Scilingo, A.A.; Dávila-Ortíz, G. Functional Properties of Guava Seed Glutelins. Journal of Agricultural and Food Chem. 2005, 53, 3613–3617. [Google Scholar] [CrossRef]
- Thongsombat, W.; Sirichote, A.; Chanthachum, S. The production of guava juice fortified with dietary fiber. Songklanakarin J. Sci. Technol. 2007, 29, 187–196. [Google Scholar]
- Kong, K.W.; Ismail, A.; Tan, C.P.; Rajab, N.F. Optimization of oven drying conditions for lycopene content and lipophilic antioxidant capacity in a by-product of the pink guava puree industry using response surface methodology. LWT-Food Sci. Technol. 2009. [Google Scholar] [CrossRef]
- Li, Y.; Guo, C.; Yang, J.; Wei, J.; Xu, J.; Cheng, S. Evaluation of antioxidant properties of pomegranate peel extract in comparison with pomegranate pulp extract. Food Chem. 2006, 96, 254–260. [Google Scholar] [CrossRef]
- Hajimahmoodi, M.; Oveisi, M.R.; Sadeghi, N.; Jannat, B.; Hadjibabaie, M.; Farahani, E.; Akrami, M.R.; Namdar, R. Antioxidant properties of peel and pulp hydro extract in ten Persian pomegranate cultivars. Pak. J. Biol. Sci. 2008, 11, 1600–1604. [Google Scholar] [CrossRef]
- Rozzi, N.L.; Singh, R.K.; Vierling, R.A.; Watkins, B.A. Supercritical fluid extraction of lycopene from tomato processing byproducts. J. Agric.Food Chem. 2002, 50, 2638–2643. [Google Scholar] [CrossRef]
- Topal, U.; Sasaki, M.; Goto, M.; Hayakawa, K. Extraction of lycopene from tomato skin with supercritical carbon dioxide: effect of operating conditions and solubility analysis. J. Agric.Food Chem. 2006, 54, 5604–5610. [Google Scholar] [CrossRef]
- Vagi, E.; Simandi, B.; Vasarhelyine, K.P.; Daood, H.; Kery, A.; Doleschall, F.; Nagy, B. Supercritical carbon dioxide extraction of carotenoids, tocopherols and sitosterols from industrial tomato by-products. J. Supercrit. Fluids 2007, 40, 218–226. [Google Scholar] [CrossRef]
- Kaur, D.; Wani, A.A.; Oberoi, D.P.S.; Sogi, D.S. Effect of extraction conditions on lycopene extractions from tomato processing waste skin using response surface methodology. Food Chem. 2008, 108, 771–718. [Google Scholar]
- Chantaro, P.; Devahastin, S.; Chiewchan, N. Production of antioxidant high dietary fiber powder from carrot peels. LWT-Food Sci. Technol. 2008, 41, 1987–1994. [Google Scholar] [CrossRef]
- Stah, W.; Sies, H. Lycopene: A biologically important carotenoid for humans? Arch. Biochem. Biophys. 1996, 336, 1–9. [Google Scholar] [CrossRef]
- Shi, J.; Le Maguer, M.; Bryan, M. Lycopene from tomatoes. In Functional Foods-Biochemical and Processing Aspects; Shi, J., Mazza, G., Le Maguer, M., Eds.; CRC Press: USA, 2002; pp. 135–168. [Google Scholar]
- Roldán-Gutiérrez, J.M.; Dolores Luque de Castro, M. Lycopene: The need for better methods for characterization and determination. Trends Anal. Chem. 2007, 26, 163–170. [Google Scholar] [CrossRef]
- Agarwal, S.; Rao, A.V. Tomato Lycopene and Low Density Lipoprotein Oxidation: A Human Dietary Intervention Study. Lipids 1998, 33, 981–984. [Google Scholar] [CrossRef]
- Krinsky, N.I.; Johnson, E.J. Carotenoid actions and their relation to health and disease. Mol. Aspects Med. 2005, 26, 459–516. [Google Scholar] [CrossRef]
- Clinton, S.K. Lycopene: chemistry, biology, and implications for human health and disease. Nutr. Rev. 1998, 56, 35–51. [Google Scholar] [CrossRef]
- During, A.; Harrison, E.H. Intestinal absorption and metabolism of carotenoids: Insights from cell culture. Arch. Biochem. Biophys. 2004, 430, 77–88. [Google Scholar] [CrossRef]
- During, A.; Harrison, E.H. An in vitro model to study the intestinal absorption of carotenoids. Food Res. Int. 2005, 38, 1001–1008. [Google Scholar] [CrossRef]
- Goñi, I.; Serrano, J.; Saura-Calixto, F. Bioaccessibility of β-carotene, lutein, and lycopene from fruits and vegetables. J. Agric. Food Chem. 2006, 54, 5382–5387. [Google Scholar] [CrossRef]
- Riedl, J.; Linseisen, J.; Hoffmann, J.; Wolfram, G. Some dietary fibers reduce the absorption of carotenoids in women. J. Nutr. 1999, 129, 2170–2176. [Google Scholar]
- Failla, M.L.; Chitchumroonchokchai, C.; Ishida, B.K. In vitro micellarization and intestinal cell uptake of cis isomers of lycopene exceed those of all-trans lycopene. J. Nutr. 2008, 138, 482–486. [Google Scholar]
- Richelle, M.; Bortlik, K.; Liardet, S.; Hager, C.; Lambelet, P.; Baur, M.; Applegate, L.A.; Offord, E.A. A food-based formulation provides lycopene with the same bioavailability to humans as that from tomato paste. J. Nutr. 2002, 132, 404–408. [Google Scholar]
- Re, R.; Fraser, P.D.; Long, M.; Bramley, P.M.; Rice-Evans, C. Isomerization of lycopene in the gastric milieu. Biochem. Biophys. Res. Commun. 2001, 281, 576–581. [Google Scholar] [CrossRef]
- Unlu, N.Z.; Bohn, T.; Francis, D.M.; Nagaraja, H.N.; Clinton, S.K.; Schwartz, S.J. Lycopene from heat-induced cis-isomer-rich tomato sauce is more bioavailable than from all-trans-rich tomato sauce in human subjects. Br. J. Nutr. 2007, 98, 140–146. [Google Scholar] [CrossRef]
- Karakaya, S.; Yilmaz, N. Lycopene content and antioxidant activity of fresh and processed tomatoes and in vitro bioavailability of lycopene. J. Sci. Food Agric. 2007, 87, 2342–2347. [Google Scholar] [CrossRef]
- Ahuja, K.D.K.; Pittaway, J.K.; Ball, M.J. Effects of olive oil and tomato lycopene combination on serum lycopene, lipid profile, and lipid oxidation. Nutrition 2006, 22, 259–265. [Google Scholar] [CrossRef]
- Brown, M.J.; Ferruzzi, M.G.; Nguyen, M.L.; Cooper, D.A.; Eldridge, A.L.; Schwartz, S.J.; White, W.S. Carotenoid bioavailability is higher from salads ingested with full-fat than with fat-reduced salad dressings as measured with electrochemical detection. Am. J. Clin. Nutr. 2004, 80, 396–403. [Google Scholar]
- Fielding, J.M.; Rowley, K.G.; Cooper, P.; O'Dea, K. Increases in plasma lycopene concentration after consumption of tomatoes cooked with olive oil. Asia-Pac J. Clin. Nutr. 2005, 14, 131–136. [Google Scholar]
- Cardinault, N.; Tyssandier, V.; Grolier, P.; Winklhofer-Roob, B.M.; Ribalta, J.; Bouteloup-Demange, C.; Rock, E.; Borel, P. Comparison of the postprandial chylomicron carotenoid responses in young and older subjects. Eur. J. Nutr. 2003, 42, 315–323. [Google Scholar] [CrossRef]
- Stahl, W.; Sies, H. Antioxidant activity of carotenoids. Mol. Aspects Med. 2003, 24, 345–351. [Google Scholar] [CrossRef]
- Boileau, T.W.-M.; Boileau, A.C.; Erdman, J.W., Jr. Bioavailability of all-trans and cis-isomers of lycopene. Exp. Bio. Med. 2002, 227, 914–919. [Google Scholar]
- Erdman, J.W., Jr. How do nutritional and hormonal status modify the bioavailability, uptake, and distribution of different isomers of lycopene? J. Nutr. 2005, 135, 2046S–2047S. [Google Scholar]
- Goralczyk, R.; Siler, U. The Role of Lycopene in Human Health. In Phytochemicals in Health and Disease; Bao, Y., Fenwick, R., Eds.; Marcel Dekker Inc: New York, NY, USA, 2004; pp. 285–309. [Google Scholar]
- Zaripheh, S.; Boileau, T.W.-M.; Lila, M.A.; Erdman, J.W. Jr. [14C]-lycopene and [14C]-labeled polar products are differentially distributed in tissues of F344 rats prefed lycopene. J. Nutr. 2003, 133, 4189–4195. [Google Scholar]
- Su, Q.; Rowley, K.G.; Balazs, N.D.H. Carotenoids: Separation methods applicable to biological samples. J. Chromatogr. B 2002, 781, 393–418. [Google Scholar] [CrossRef]
- Porrini, M.; Riso, P.; Brusamolino, A.; Berti, C.; Guarnieri, S.; Visioli, F. Daily intake of a formulated tomato drink affects carotenoid plasma and lymphocyte concentrations and improves cellular antioxidant protection. Br. J. Nutr. 2005, 93, 93–99. [Google Scholar] [CrossRef]
- Stimpson, J.P.; Lackan, N.A. Serum carotenoid levels vary by marital status. J. Am. Dietetic Assoc. 2007, 107, 1581–1585. [Google Scholar] [CrossRef]
- Scott, K.J.; Thurnham, D.I.; Hart, D.J.; Bingham, S.A.; Day, K. The correlation between the intake of lutein, lycopene and beta-carotene from vegetables and fruits, and blood plasma concentrations in a group of women aged 50-65 years in the UK. Br. J. Nutr. 1996, 75, 409–418. [Google Scholar] [CrossRef]
- Michaud, D.S.; Giovannucci, E.L.; Ascherio, A.; Rimm, E.B.; Forman, M.R.; Sampson, L.; Willett, W.C. Associations of plasma carotenoid concentrations and dietary intake of specific carotenoids in samples of two prospective cohort studies using a new carotenoid database. Cancer Epidemiol. Biomarkers Prev. 1998, 7, 283–290. [Google Scholar]
- Olmedilla, B.; Granado, F.; Southon, S.; Wright, A.J.; Blanco, I.; Gil-Martinez, E.; Berg, H.; Corridan, B.; Roussel, A.M.; Chopra, M.; Thurnham, D.I. Serum concentrations of carotenoids and vitamins A, E, and C in control subjects from five European countries. Br. J. Nutr. 2001, 85, 227–238. [Google Scholar] [CrossRef]
- Al-Delaimyl, W.K.; Van Kappel, A.L.; Ferrari, P.; Slimani, N.; Steghens, J.-P.; Bingham, S.; Johansson, I.; Wallström, P.; Overvad, K.; Tjønneland, A.; Key, T.J.; Welch, A.A.; Bueno-de-Mesquita, H.B.; Peeters, P.H.M.; Boeing, H.; Linseisen, J.; Clavel-Chapelon, F.; Guibout, C.; Navarro, C.; Quirós, J.R.; Palli, D.; Celentano, E.; Trichopoulou, A.; Benetou, V.; Kaaks, R.; Riboli, E. Plasma levels of six carotenoids in nine European countries: Report from the European Prospective Investigation into Cancer and Nutrition (EPIC). Publ. Health Nutr. 2004, 7, 713–722. [Google Scholar]
- Ozasa, K.; Ito, Y.; Suzuki, K.; Watanabe, Y.; Wakai, K.; Tamakoshi, A. Association of serum carotenoid concentration and dietary habits among the JACC Study subjects. J. Epidemiol. 2005, 15, S220–S227. [Google Scholar] [CrossRef]
- Boonsiri, P.; Pooart, J.; Tangrassameeprasert, R.; Hongsprabhas, P. Serum β-carotene, lycopene and α-tocopherol levels of healthy people in northeast Thailand. Asia-Pac. J. Clin. Nutr. 2007, 16, 47–51. [Google Scholar]
- Lindshield, B.L.; Canene-Adams, K.; Erdman, J.W., Jr. Lycopenoids: Are lycopene metabolites bioactive? Arch. Biochem. Biophys. 2007, 458, 136–140. [Google Scholar] [CrossRef]
- Ferreira, A.L.D.A.; Yeum, K.-J.; Russell, R.M.; Krinsky, N.I.; Tang, G. Enzymatic and oxidative metabolites of lycopene. J. Nutr. Biochem. 2003, 14, 531–540. [Google Scholar] [CrossRef]
- Kim, S.-J.; Nara, E.; Kobayashi, H.; Terao, J.; Nagao, A. Formation of cleavage products by autoxidation of lycopene. Lipids 2001, 36, 191–199. [Google Scholar] [CrossRef]
- Hu, K.Q.; Liu, C.; Ernst, H.; Krinsky, N.I.; Russell, R.M.; Wang, X.D. The biochemical characterization of ferret carotene-9',10'-monooxygenase catalyzing cleavage of carotenoids in vitro and in vivo. J. Biol. Chem. 2006, 281, 19327–19338. [Google Scholar] [CrossRef]
- Britton, G. Structure and properties of carotenoids in relation to function. FASEB J. 1995, 9, 1551–1558. [Google Scholar]
- Young, A.J.; Lowe, G.M. Antioxidant and prooxidant properties of carotenoids. Arch. Biochem. Biophys. 2001, 385, 20–27. [Google Scholar] [CrossRef]
- Christensen, R.L. The Photochemistry of Carotenoids; Frank, H.A., Young, A.J., Britton, G., Cogdell, R.J., Eds.; Kluwer Academic: Dordrecht, The Netherlands, 1999; pp. 137–157. [Google Scholar]
- Di Mascio, P.; Kaiser, S.; Sies, H. Lycopene as the most efficient biological carotenoid singlet oxygen quencher. Arch. Biochem. Biophys. 1989, 274, 532–538. [Google Scholar] [CrossRef]
- Cantrell, A.; McGarvey, D.J.; Truscott, T.G.; Rancan, F.; Böhm, F. Singlet oxygen quenching by dietary carotenoids in a model membrane environment. Arch. Biochem. Biophys. 2003, 412, 47–54. [Google Scholar] [CrossRef]
- El-Agamey, A.; Lowe, G.M.; McGarvey, D.J.; Mortensen, A.; Phillip, D.M.; Truscott, T.G.; Young, A.J. Carotenoid radical chemistry and antioxidant or pro-oxidant properties. Arch. Biochem. Biophys. 2004, 430, 37–48. [Google Scholar] [CrossRef]
- Mortensen, A.; Skibsted, L.H.; Truscott, T.G. The interaction of dietary carotenoids with radical species. Arch. Biochem. Biophys. 2001, 385, 13–19. [Google Scholar] [CrossRef]
- Krinsky, N.I.; Yeum, K.J. Carotenoid-radical interactions. Biochem. Biophys. Res. Commun. 2003, 305, 754–760. [Google Scholar] [CrossRef]
- Burton, G.W.; Ingold, K.U. β-Carotene: An unusual type of lipid antioxidant. Science 1984, 224, 569–573. [Google Scholar]
- Kiokias, S.; Gordon, M.H. Antioxidant properties of carotenoids in vitro and in vivo. Food Rev. Int. 2004, 20, 99–121. [Google Scholar] [CrossRef]
- Mortensen, A.; Skibsted, L.H. Reactivity of β-carotene towards peroxyl radicals studied by laser flash and steady-state photolysis. FEBS Lett. 1998, 426, 392–396. [Google Scholar] [CrossRef]
- Conn, P.F.; Lambert, C.; Land, E.J.; Schalch, W.; Truscott, T.G. Carotene-oxygen radical interactions. Free Radical Res. Commun. 1992, 16, 401–408. [Google Scholar] [CrossRef]
- Johansson, L.B.; Lindblom, G.; Wieslander, Å.; Arvidson, G. Orientation of β-carotene and retinal in lipid bilayers. FEBS Lett. 1981, 128, 97–99. [Google Scholar] [CrossRef]
- Van de Ven, M.; Kattenberg, M.; van Ginkel, G.; Levine, Y.K. Study of the orientational ordering of carotenoids in lipid bilayers by resonance-Raman spectroscopy. Biophys. J. 1984, 45, 1203–1209. [Google Scholar] [CrossRef]
- Lowe, G.M.; Booth, L.A.; Young, A.J.; Bilton, R.F. Lycopene and β-carotene protect against oxidative damage in HT29 cells at low concentrations but rapidly lose this capacity at higher doses. Free Radical Res. 1999, 30, 141–151. [Google Scholar] [CrossRef]
- Liu, D.; Shi, J.; Colina Ibarra, A.; Kakuda, Y.; Jun Xue, S. The scavenging capacity and synergistic effects of lycopene, vitamin E, vitamin C, and β-carotene mixtures on the DPPH free radical. LWT-Food Sci. Technol. 2008, 41, 1344–1349. [Google Scholar] [CrossRef]
- Shi, J.; Qu, Q.; Kakuda, Y.; Xue, S.J.; Jiang, Y.; Koide, S.; Shim, Y.-Y. Investigation of the antioxidant and synergistic activity of lycopene and other natural antioxidants using LAME and AMVN model systems. J. Food Compos. Anal. 2007, 20, 603–608. [Google Scholar] [CrossRef]
- Truscott, T.G. β-carotene and disease: A suggested pro-oxidant and anti-oxidant mechanism and speculations concerning its role in cigarette smoking. J. Photochem. Photobiol. B: Biol. 1996, 35, 233–235. [Google Scholar] [CrossRef]
- Yeum, K.-J.; Russell, R.M.; Krinsky, N.I.; Aldini, G. Biomarkers of antioxidant capacity in the hydrophilic and lipophilic compartments of human plasma. Arch. Biochem. Biophys. 2004, 430, 97–103. [Google Scholar] [CrossRef]
- Stahl, W.; Junghans, A.; de Boer, B.; Driomina, E.S.; Briviba, K.; Sies, H. Carotenoid mixtures protect multilamellar liposomes against oxidative damage: synergistic effects of lycopene and lutein. FEBS Lett. 1998, 427, 305–308. [Google Scholar] [CrossRef]
- Edge, R.; Land, E.J.; McGarvey, D.; Mulroy, L.; Truscott, T.G. Relative one electron reduction potentials of carotenoid radical cations and the interactions of carotenoids with the vitamin E radical cation. J. Am. Chem. Soc. 1998, 120, 4087–4090. [Google Scholar]
- Edge, R.; Truscott, T.G. The Photochemistry of Carotenoids; Frank, H.A., Young, A.J., Britton, G., Cogdell, R.J., Eds.; Kluwer Academic: Dordrecht, The Netherlands, 1999; pp. 223–234. [Google Scholar]
- Böhm, F.; Edge, R.; Land, E.J.; McGarvey, D.J.; Truscott, T.G. Carotenoids enhance vitamin E antioxidant efficiency. J. Am. Chem. Soc. 1997, 119, 611–622. [Google Scholar]
- Mortensen, A.; Skibsted, L.H. Relative stability of carotenoid radical cations and homologue tocopheroxyl radicals: A real time kinetic study of antioxidant hierarchy. FEBS Lett. 1997, 417, 261–266. [Google Scholar] [CrossRef]
- Truscott, T.G. Synergistic effects of antioxidant vitamins. In Functions of Vitamins beyond Recommended Dietary Allowances; Walter, P., Hornig, D., Moser, U., Eds.; Karger AG: Basel, 2001; Volume 55, pp. 68–79. [Google Scholar]
- Shixian, Q.; Dai, Y.; Kakuda, Y.; Shi, J.; Mittal, G.; Yeung, D.; Jiang, Y. Synergistic anti-oxidative effects of lycopene with other bioactive compounds. Food Rev. Int. 2005, 21, 295–311. [Google Scholar] [CrossRef]
- Castro, I.A.; Moraes Barros, S.B.; Lanfer Marquez, U.M.; Motizuki, M.; Higashi Sawada, T.C. Optimization of the antioxidant capacity of a mixture of carotenoids and α-tocopherol in the development of a nutritional supplement. Food Res. Int. 2005, 38, 861–866. [Google Scholar] [CrossRef]
- Rao, A.V.; Rao, L.G. Carotenoids and human health. Pharmacol. Res. 2007, 55, 207–216. [Google Scholar] [CrossRef]
- Astley, S.B.; Hughes, D.A.; Wright, A.J.A.; Elliott, R.M.; Southon, S. DNA damage and susceptibility to oxidative damage in lymphocytes: effects of carotenoids in vitro and in vivo. Br. J. Nutr. 2004, 91, 53–61. [Google Scholar] [CrossRef]
- Liu, F.; Zhang, Z.-Z.; Wu, M.; Shu, Y. Study on the protective effect of tomato juice on DNA damage in cells. J. Sichuan Univ. (Medical Science Edition) 2007, 38, 18–21. [Google Scholar]
- Scolastici, C.; Alves de Lima, R.O.; Barbisan, L.F.; Ferreira, A.L.A.; Ribeiro, D.A.; Salvadori, D.M.F. Antigenotoxicity and antimutagenicity of lycopene in HepG2 cell line evaluated by the comet assay and micronucleus test. Toxicol. in Vitro 2008, 22, 510–514. [Google Scholar] [CrossRef]
- Matos, H.R.; Di Mascio, P.; Medeiros, M.H.G. Protective effect of lycopene on lipid peroxidation and oxidative DNA damage in cell culture. Arch. Biochem. Biophys. 2000, 383, 56–59. [Google Scholar] [CrossRef]
- Srinivasan, M.; Sudheer, A.R.; Pillai, K.R.; Kumar, P.R.; Sudhakaran, P.R.; Menon, V.P. Lycopene as a natural protector against γ-radiation induced DNA damage, lipid peroxidation and antioxidant status in primary culture of isolated rat hepatocytes in vitro. Biochim. Biophys. Acta-Gen. Sub. 2007, 1770, 659–665. [Google Scholar] [CrossRef]
- Matos, H.R.; Marques, S.A.; Gomes, O.F.; Silva, A.A.; Heimann, J.C.; Di Mascio, P.; Medeiros, M.H.G. Lycopene and β-carotene protect in vivo iron-induced oxidative stress damage in rat prostate. Braz. J. Med. Biol. Res. 2006, 39, 203–210. [Google Scholar]
- Watters, J.L.; Satia, J.A.; Kupper, L.L.; Swenberg, J.A.; Schroeder, J.C.; Switzer, B.R. Associations of antioxidant nutrients and oxidative DNA damage in healthy African-American and White adults. Cancer Epidemiol. Biomark. Prev. 2007, 16, 1428–1436. [Google Scholar] [CrossRef]
- Riso, P.; Pinder, A.; Santangelo, A.; Porrini, M. Does tomato consumption effectively increase the resistance of lymphocyte DNA to oxidative damage? Am. J. Clin. Nutr. 1999, 69, 712–718. [Google Scholar]
- Zhao, X.; Aldini, G.; Johnson, E.J.; Rasmussen, H.; Kraemer, K.; Woolf, H.; Musaeus, N.; Krinsky, N.I.; Russell, R.M.; Yeum, K.-J. Modification of lymphocyte DNA damage by carotenoid supplementation in postmenopausal women. Am. J. Clin. Nutr. 2006, 83, 163–169. [Google Scholar]
- Böhm, F.; Edge, R.; Burke, M.; Truscott, T.G. Dietary uptake of lycopene protects human cells from singlet oxygen and nitrogen dioxide: ROS components from cigarette smoke. J. Photochem. Photobiol. B: Biol. 2001, 64, 176–178. [Google Scholar] [CrossRef]
- Tinkler, J.H.; Bohm, F.; Schalch, W.; Truscott, T.G. Dietary carotenoids protect human cells from damage. J. Photochem. Photobiol. B: Biol. 1994, 26, 283–285. [Google Scholar] [CrossRef]
- Riso, P.; Visioli, F.; Erba, D.; Testolin, G.; Porrini, M. Lycopene and vitamin C concentrations increase in plasma and lymphocytes after tomato intake. Effects on cellular antioxidant protection. Eur. J. Clin. Nutr. 2004, 58, 1350–1358. [Google Scholar] [CrossRef]
- Riso, P.; Visioli, F.; Grande, S.; Guarnieri, S.; Gardana, C.; Simonetti, P.; Porrini, M. Effect of a tomato-based drink on markers of inflammation, immunomodulation, and oxidative stress. J. Agric. Food Chem. 2006, 54, 2563–2566. [Google Scholar] [CrossRef]
- Rao, A.V.; Shen, H. Effect of low dose lycopene intake on lycopene bioavailability and oxidative stress. Nutr. Res. 2002, 22, 1125–1131. [Google Scholar] [CrossRef]
- Rao, L.G.; Mackinnon, E.S.; Josse, R.G.; Murray, T.M.; Strauss, A.; Rao, A.V. Lycopene consumption decreases oxidative stress and bone resorption markers in postmenopausal women. Osteoporos. Int. 2007, 18, 109–115. [Google Scholar] [CrossRef]
- Visioli, F.; Riso, P.; Grande, S.; Galli, C.; Porrini, M. Protective activity of tomato products on in vivo markers of lipid oxidation. Eur. J. Nutr. 2003, 42, 201–206. [Google Scholar] [CrossRef]
- Misra, R.; Mangi, S.; Joshi, S.; Mittal, S.; Gupta, S.K.; Pandey, R.M. LycoRed as an alternative to hormone replacement therapy in lowering serum lipids and oxidative stress markers: A randomized controlled clinical trial. J. Obstet. Gynaecol. Res. 2006, 32, 299–304. [Google Scholar] [CrossRef]
- Jacob, K.; Periago, M.J.; Böhm, V.; Berruezo, G.R. Influence of lycopene and vitamin C from tomato juice on biomarkers of oxidative stress and inflammation. Br. J. Nutr. 2008, 99, 137–146. [Google Scholar]
- WHO. Cardiovascular Diseases: World Heart Day 2009. Available online: http://www.who.int/cardiovascular_diseases/en/, accessed on 23 September 2009.
- Sesso, H.D.; Buring, J.E.; Norkus, E.P.; Gaziano, J.M. Plasma lycopene, other carotenoids, and retinol and the risk of cardiovascular disease in wome. Am. J. Clin. Nutr. 2004, 79, 47–53. [Google Scholar]
- Klipstein-Grobusch, K.; Launer, L.J.; Geleijnse, J.M.; Boeing, H.; Hofman, A.; Witteman, J.C.M. Serum carotenoids and atherosclerosis The Rotterdam Study. Atherosclerosis 2000, 148, 49–56. [Google Scholar] [CrossRef]
- Rissanen, T.H.; Voutilainen, S.; Nyyssönen, K.; Salonen, J.T. Lycopene, atherosclerosis, and coronary heart disease. Exp. Biol. Med. 2002, 227, 900–907. [Google Scholar]
- Hu, M.-Y.; Li, Y.-L.; Jiang, C.-H.; Liu, Z.-Q.; Qu, S.-L.; Huang, Y.-M. Comparison of lycopene and fluvastatin effects on atherosclerosis induced by a high-fat diet in rabbits. Nutrition 2008, 24, 1030–1038. [Google Scholar] [CrossRef]
- Engelhard, Y.N.; Gazer, B.; Paran, E.; Sheva, B. Natural antioxidants from tomato extract reduce blood pressure in patients with grade-1 hypertension: A double-blind, placebo-controlled pilot study. Am. Heart J. 2006, 151, 100–106. [Google Scholar]
- Rissanen, T.H.; Voutilainen, S.; Nyyssönen, K.; Salonen, R.; Kaplan, G.A.; Salonen, J.T. Serum lycopene concentrations and carotid atherosclerosis: the Kuopio Ischaemic Heart Disease Risk Factor Study. Am. J. Clin. Nutr. 2003, 77, 133–138. [Google Scholar]
- Ito, Y.; Kurata, M.; Suzuki, K.; Hamajima, N.; Hishida, H.; Aoki, K. Cardiovascular Disease Mortality and Serum Carotenoid Levels: A Japanese Population-based Follow-up Study. J. Epidemiol. 2006, 16, 154–160. [Google Scholar] [CrossRef]
- Osganian, S.K.; Stampfer, M.J.; Rimm, E.; Spiegelman, D.; Manson, J.E.; Willett, W.C. Dietary carotenoids and risk of coronary artery disease in women. Am. J. Clin. Nutr. 2003, 77, 1390–1399. [Google Scholar]
- Sesso, H.D.; Liu, S.; Gaziano, J.M.; Burin, J.E. Dietary Lycopene, Tomato-Based Food Products and Cardiovascular Disease in Women. J. Nutr. 2003, 133, 2336–2341. [Google Scholar]
- Salman, H.; Bergman, M.; Djaldetti, M.; Bessler, H. Lycopene affects proliferation and apoptosis of four malignant cell lines. Biomed. Pharmacother. 2007, 61, 366–369. [Google Scholar] [CrossRef]
- Scolastici, C.; Alves de Lima, R.O.; Barbisan, L.F.; Ferreira, A.L.A.; Ribeiro, D.A.; Salvadori, D.M.F. Lycopene activity against chemically induced DNA damage in Chinese hamster ovary cells. Toxicol. in Vitro 2007, 21, 840–845. [Google Scholar] [CrossRef]
- Zhang, J.; Dhakal, I.; Stone, A.; Ning, B.; Greene, G.; Lang, N.P.; Kadlubar, F.F. Plasma carotenoids and prostate cancer: A population-based case-control study in Arkansas. Nutr. Cancer 2007, 59, 46–53. [Google Scholar] [CrossRef]
- Mohanty, N.K.; Saxena, S.; Singh, U.P.; Goyal, N.K.; Arora, R.P. Lycopene as a chemopreventive agent in the treatment of high-grade prostate intraepithelial neoplasia. Urol. Oncol.-Semin. Ori. 2005, 23, 383–385. [Google Scholar] [CrossRef]
- Kanagaraj, P.; Vijayababu, M.R.; Ravisankar, B.; Anbalagan, J.; Aruldhas, M.M.; Arunakaran, J. Effect of lycopene on insulin-like growth factor-I, IGF binding protein-3 and IGF type-I receptor in prostate cancer cells. J. Cancer Res. Clin. Oncology 2007, 133, 351–359. [Google Scholar] [CrossRef]
- Gunasekera, R.S.; Sewgobind, K.; Desai, S.; Dunn, L.; Black, H.S.; McKeehan, W.L.; Patil, B. Lycopene and lutein inhibit proliferation in rat prostate carcinoma cells. Nutr. Cancer 2007, 58, 171–177. [Google Scholar] [CrossRef]
- Stacewicz-Sapuntzakis, M.; Bowen, P.E. Role of lycopene and tomato products in prostate health. Biochim. Biophys. Acta-Mol. Basis Dis. 2005, 1740, 202–205. [Google Scholar]
- Hadley, C.W.; Miller, E.C.; Schwartz, S.J.; Clinton, S.K. Tomatoes, lycopene, and prostate cancer: Progress and promise. Exp. Biol. Med. 2002, 227, 869–880. [Google Scholar]
- Giovannucci, E.; Rimm, E.B.; Liu, Y.; Stampfer, M.J.; Willett, W.C. A Prospective Study of Tomato Products, Lycopene, and Prostate Cancer Risk. J. Nat. Cancer Inst. 2002, 94, 391–398. [Google Scholar] [CrossRef]
- Kirsh, V.A.; Mayne, S.T.; Peters, U.; Chatterjee, N.; Leitzmann, M.F.; Dixon, L.B.; Urban, D.A.; Crawford, E.D.; Hayes, R.B. A prospective study of lycopene and tomato product intake and risk of prostate cancer. Cancer Epidemiol. Biomark. Prev. 2006, 15, 92–98. [Google Scholar] [CrossRef]
- Jatoi, A.; Burch, P.; Hillman, D.; Vanyo, J.M.; Dakhil, S.; Nikcevich, D.; Rowland, K.; Morton, R.; Flynn, P.J.; Young, C.; Tan, W. A tomato-based, lycopene-containing intervention for androgen-independent prostate cancer: Results of a phase II study from the North Central Cancer Treatment Group. Urology 2007, 69, 289–294. [Google Scholar]
- Key, T.J.; Appleby, P.N.; Allen, N.E.; Travis, R.C.; Roddam, A.W.; Jenab, M.; Egevad, L.; Tjonneland, A.; Johnsen, N.F.; Overvad, K.; Linseisen, J.; Rohrmann, S.; Boeing, H.; Pischon, T.; Psaltopoulou, T.; Trichopoulou, A.; Trichopoulos, D.; Palli, D.; Vineis, P.; Tumino, R.; Berrino, F.; Kiemeney, L.A.L.M.; Bueno-De-Mesquita, H.B.; Quiros, J.R.; Gonzalez, C.A.; Martinez, C.; Larranaga, N.; Chirlaque, M.D.; Ardanaz, E.; Stattin, P.; Hallmans, G.; Khaw, K.T.; Bingham, S.; Slimani, N.; Ferrari, P.; Rinaldi, S.; Riboli, E. Plasma carotenoids, retinol, and tocopherols and the risk of prostate cancer in the European Prospective Investigation into Cancer and Nutrition study. Am. J. Clin. Nutr. 2007, 86, 672–681. [Google Scholar]
- Peters, U.; Leitzmann, M.F.; Chatterjee, N.; Wang, Y.; Albanes, D.; Gelmann, E.P.; Friesen, M.D.; Riboli, E.; Hayes, R.B. Serum lycopene, other carotenoids, and prostate cancer risk: a nested case-control study in the prostate, lung, colorectal, and ovarian cancer screening trial. Cancer Epidemiol. Biomark. Prev. 2007, 16, 962–968. [Google Scholar] [CrossRef]
- Jian, L.; Lee, A.H.; Binns, C.W. Tea and lycopene protect against prostate cancer. Asia-Pac. J. Clin. Nutr. 2007, 16, 453–457. [Google Scholar]
- Hwang, E.-S.; Lee, H.J. Inhibitory effects of lycopene on the adhesion, invasion, and migration of SK-Hep1 human hepatoma cells. Exp. Biol. Med. 2006, 231, 322–327. [Google Scholar]
- Burgess, L.C.; Rice, E.; Fischer, T.; Seekins, J.R.; Burgess, T.P.; Sticka, S.J.; Klatt, K. Lycopene has limited effect on cell proliferation in only two of seven human cell lines (both cancerous and noncancerous) in an in vitro system with doses across the physiological range. Toxicol. in Vitro 2008, 22, 1297–1300. [Google Scholar] [CrossRef]
- Müller, K.; Zucoloto, S.; Albuquerque Jr., R.F.; Vannucchi, H. Lack of inhibitory effect of lycopene on dysplastic lesions induced by 7,12-dimethyl-benz[a]anthracene in hamster buccal pouch. Nutr. Res. 2007, 27, 574–579. [Google Scholar] [CrossRef]
- Huang, J.P.; Zhang, M.; Holman, C.D.J.; Xie, X. Dietary carotenoids and risk of breast cancer in Chinese women. Asia-Pac. J. Clin. Nutr. 2007, 16, 437–442. [Google Scholar]
- Sesso, H.D.; Buring, J.E.; Zhang, S.M.; Norkus, E.P.; Gaziano, J.M. Dietary and plasma lycopene and the risk of breast cancer. Cancer Epidemiol. Biomark. Prev. 2005, 14, 1074–1081. [Google Scholar] [CrossRef]
- Larsson, S.C.; Bergkvist, L.; Näslund, I.; Rutegård, J.; Wolk, A. Vitamin A, retinol, and carotenoids and the risk of gastric cancer: a prospective cohort study. Am. J. Clin. Nutr. 2007, 85, 497–503. [Google Scholar]
- Lian, F.; Smith, D.E.; Ernst, H.; Russell, R.M.; Wang, X.-D. Apo-10′-lycopenoic acid inhibits lung cancer cell growth in vitro, and suppresses lung tumorigenesis in the A/J mouse model in vivo. Carcinogenesis 2007, 28, 1567–1574. [Google Scholar] [CrossRef]
- Guttenplan, J.B.; Chen, M.; Kosinska, W.; Thompson, S.; Zhao, Z.; Leonard, A. Cohen Effects of a lycopene-rich diet on spontaneous and benzo[a]pyrene-induced mutagenesis in prostate, colon and lungs of the lacZ mouse. Cancer Lett. 2001, 164, 1–6. [Google Scholar] [CrossRef]
- Coyne, T.; Ibiebele, T.I.; Baade, P.D.; Dobson, A.; McClintock, C.; Dunn, S.; Leonard, D.; Shaw, J. Diabetes mellitus and serum carotenoids: Findings of a population-based study in Queensland, Australia. Am. J. Clin. Nutr. 2005, 82, 685–693. [Google Scholar]
- Polidori, M.C.; Mecocci, P.; Stahl, W.; Parente, B.; Cecchetti, R.; Cherubini, A.; Cao, P.; Sies, H.; Senin, U. Plasma levels of lipophilic antioxidants in very old patients with Type 2 diabetes. Diabetes/Metabolism Res. Rev. 2000, 16, 15–19. [Google Scholar] [CrossRef]
- Ylönen, K.; Alfthan, G.; Groop, L.; Saloranta, C.; Aro, A.; Virtanen, S.M. Dietary intakes and plasma concentrations of carotenoids and tocopherols in relation to glucose metabolism in subjects at high risk of type 2 diabetes: The Botnia Dietary Study. Am. J. Clin. Nutr. 2003, 77, 1434–1441. [Google Scholar]
- Riso, P.; Brusamolino, A.; Martinetti, A.; Porrini, M. Effect of a tomato drink intervention on insulin-like growth factor (IGF)-1 serum levels in healthy subjects. Nutr.Cancer 2006, 55, 157–162. [Google Scholar] [CrossRef]
- Wang, L.; Liu, S.; Pradhan, A.D.; Manson, J.E.; Buring, J.E.; Gaziano, J.M.; Sesso, H.D. Plasma Lycopene, Other Carotenoids, and the Risk of Type 2 Diabetes in Wome. Am. J. Epidemiol. 2006, 164, 576–585. [Google Scholar] [CrossRef]
- Kuhad, A.; Sethi, R.; Chopra, K. Lycopene attenuates diabetes-associated cognitive decline in rats. Life Sci. 2008, 83, 128–134. [Google Scholar] [CrossRef]
- Kumar, P.; Kumar, A. Effect of lycopene and epigallocatechin-3-gallate against 3-nitropropionic acid induced cognitive dysfunction and glutathione depletion in rat: A novel nitric oxide mechanism. Food Chem. Toxicol. 2009, 47, 2522–2530. [Google Scholar] [CrossRef]
- Akbaraly, N.T.; Faure, H.; Gourlet, V.; Favier, A.; Berr, C. Plasma carotenoid levels and cognitive performance in an elderly population: Results of the EVA Study. J. Gerontol.-Ser. A 2007, 62, 308–316. [Google Scholar] [CrossRef]
- Gupta, S.K.; Trivedi, D.; Srivastava, S.; Joshi, S.; Halder, N.; Verma, S.D. Lycopene attenuates oxidative stress induced experimental cataract development: An in vitro and in vivo study. Nutrition 2003, 19, 794–799. [Google Scholar] [CrossRef]
- Tang, X.Y.; Yang, X.D.; Wang, L.; Sun, W.Q.; Gut, F.; He, H.; Ding, L.; Qu, S.L.; Xiao, C. To study the mechanisms and the effects of lycopene on lipid peroxidation injure in hyperlipemia rabbits. Atherosclerosis 2006, 7 (Suppl.), 521. [Google Scholar]
- Zhao, Y.; Yu, W.; Hu, W.; Yuan, Y. Anti-inflammatory and anticoagulant activities of lycopene in mice. Nutr. Res. 2003, 23, 1591–1595. [Google Scholar] [CrossRef]
- Saedisomeolia, A.; Wood, L.G.; Garg, M.L.; Gibson, P.G.; Wark, P.A.B. Lycopene enrichment of cultured airway epithelial cells decreases the inflammation induced by rhinovirus infection and lipopolysaccharide. J. Nutr. Biochem. 2009, 20, 577–585. [Google Scholar] [CrossRef]
- Lee, C.-M.; Chang, J.-H.; Moon, D.-O.; Choi, Y.H.; Choi, I.-W.; Park, Y.-M.; Kim, G.-Y. Lycopene suppresses ovalbumin-induced airway inflammation in a murine model of asthma. Biochem. Biophys. Res. Commun. 2008, 374, 248–252. [Google Scholar] [CrossRef]
- Sharma, J.B.; Kumar, A.; Kumar, A.; Malhotra, M.; Arora, R.; Prasad, S.; Batra, S. Effect of lycopene on pre-eclampsia and intra-uterine growth retardation in primigravidas. Int. J. Gynecol. Obstet. 2003, 81, 257–262. [Google Scholar] [CrossRef]
- Han, C.H.; Yang, C.H.; Sohn, D.W.; Kim, S.W.; Kang, S.H.; Cho, Y.-H. Synergistic effect between lycopene and ciprofloxacin on a chronic bacterial prostatitis rat model. Int. J. Antimicrob. Agents 2008, 31, 102–107. [Google Scholar] [CrossRef]
- Forssberg, A.; Lingen, C.; Ernster, L.; Linberg, O. Modification of the X-irradiation syndrome by lycopene. Exp. Cell Res. 1959, 16, 7–14. [Google Scholar] [CrossRef]
- Riccioni, G.; Bucciarelli, T.; Mancini, B.; Di Ilio, C.; Della Vecchia, R.; D'Orazio, N. Plasma lycopene and antioxidant vitamins in asthma: The PLAVA Study. J. Asthma 2007, 44, 429–432. [Google Scholar] [CrossRef]
- De Pablo, P.; Dietrich, T.; Karlson, E.W. Antioxidants and other novel cardiovascular risk factors in subjects with rheumatoid arthritis in a large population sample. Arthritis Care Res. 2007, 57, 953–962. [Google Scholar] [CrossRef]
- Trumbo, P.R. Are there adverse effects of lycopene exposure? J. Nutr. 2005, 135, 2060S–2061S. [Google Scholar]
- Christian, M.S.; Schulte, S.; Hellwig, J. Developmental (embryo-fetal toxicity/teratogenicity) toxicity studies of synthetic crystalline lycopene in rats and rabbits. Food Chem. Toxicol. 2003, 41, 773–783. [Google Scholar] [CrossRef]
- Shao, A.; Hathcock, J.N. Risk assessment for the carotenoids lutein and lycopene. Regul. Toxicol. Pharm. 2006, 45, 289–298. [Google Scholar] [CrossRef]
- Al-Wandawi, H.; Abdul-Rahman, M.; Al-Shaikhly, K. Tomato processing wastes as essential raw materials source. J. Agric. Food Chem. 1985, 33, 804–807. [Google Scholar] [CrossRef]
- Krishna, G. Carotene and tocopherol in agro-industrial by-products and wastes of the tropics. Agric. Wastes 1985, 12, 235–239. [Google Scholar] [CrossRef]
- Benakmoum, A.; Abbeddou, S.; Ammouche, A.; Kefalas, P.; Gerasopoulos, D. Valorisation of low quality edible oil with tomato peel waste. Food Chem. 2008, 110, 684–690. [Google Scholar] [CrossRef]
- Naviglio, D.; Caruso, T.; Iannece, P.; Aragòn, A.; Santini, A. Characterization of high purity lycopene from tomato wastes using a new pressurized extraction approach. J. Agric. Food Chem. 2008, 56, 6227–6231. [Google Scholar]
- Kassama, L.S.; Shi, J.; Mittal, G.S. Optimization of supercritical fluid extraction of lycopene from tomato skin with central composite rotatable design model. Sep. Purif. Technol. 2008, 60, 278–284. [Google Scholar] [CrossRef]
- Yang, S.X.; Shi, W.; Zeng, J. Modelling the supercritical fluid extraction of lycopene from tomato paste waste using neuro-fuzzy approaches. Lect. Notes Comput. Sci. 2004, 3174, 880–885. [Google Scholar] [CrossRef]
- Xi, J. Effect of high pressure processing on the extraction of lycopene in tomato paste waste. Chem. Eng. Technol. 2006, 29, 736–739. [Google Scholar] [CrossRef]
- Jun, X. Application of high hydrostatic pressure processing of food to extracting lycopene from tomato paste waste. High Pressure Res. 2006, 26, 33–41. [Google Scholar] [CrossRef]
- Dhas, P.H.A.; John Zachariah, T.; Rajesh, P.N.; Subramannian, S. Effect of blanching and drying on quality of mace (Myristica Fragrans). J. Food Sci. Technol. 2004, 41, 306–308. [Google Scholar]
- Choudhari, S.M.; Ananthanarayan, L. Enzyme aided extraction of lycopene from tomato tissues. Food Chem. 2007, 102, 77–81. [Google Scholar] [CrossRef]
- Sandei, L.; Leoni, C. Exploitation of by-products (solid wastes) from tomato processing to obtain high value antioxidants. Acta Horticult. 2006, 724, 249–257. [Google Scholar]
- Lavecchia, R.; Zuorro, A. Improved lycopene extraction from tomato peels using cell-wall degrading enzymes. Eur. Food Res. Technol. 2008, 228, 153–158. [Google Scholar] [CrossRef]
- Nobre, B.P.; Pessoa, F.L.P.; Palavra, A.F.; Mendes, R.L. Supercritical CO2 extraction of lycopene from tomato industrial waste. In CHISA 2006-17th International Congress of Chemical and Process Engineering, Prague, Czech Republic, 21-31 August 2006.
- Calvo, M.M.; García, M.L.; Selgas, M.D. Dry fermented sausages enriched with lycopene from tomato peel. Meat Sci. 2008, 80, 167–172. [Google Scholar] [CrossRef]
- Chiu, Y.T.; Chiu, C.P.; Chien, J.T.; Ho, G.H.; Yang, J.; Chen, B.H. Encapsulation of lycopene extract from tomato pulp waste with gelatin and poly(γ-glutamic acid) as carrier. J. Agric. Food Chem. 2007, 55, 5123–5130. [Google Scholar]
- Baysal, T.; Ersus, S.; Starmans, D.A.J. Supercritical CO2 extraction of β-carotene and lycopene from tomato paste waste. J. Agric. Food Chem. 2000, 48, 5507–5511. [Google Scholar] [CrossRef]
- Altan, A.; McCarthy, K.L.; Maskan, M. Evaluation of snack foods from barley-tomato pomace blends by extrusion processing. J. Food Eng. 2008, 84, 231–242. [Google Scholar] [CrossRef]
- Schieber, A.; Stintzing, F.C.; Carle, R. By-products of plant food processing as a source of functional compounds-recent developments. Trends Food Sci. Technol. 2001, 12, 401–413. [Google Scholar] [CrossRef]
- Naviglio, D.; Pizzolongo, F.; Ferrara, L.; Naviglio, B.; Aragòn, A.; Santini, A. Extraction of pure lycopene from industrial tomato waste in water using the extractor Naviglio. Afr. J. Food Sci. 2008, 2, 37–44. [Google Scholar]
- Knoblich, M.; Anderson, B.; Latshaw, D. Analyses of tomato peel and seed byproducts and their use as a source of carotenoids. J. Sci. Food Agric. 2005, 85, 1166–1170. [Google Scholar]
- Lee, M.T.; Chen, B.H. Stability of lycopene during heating and illumination in a model system. Food Chem. 2002, 78, 425–432. [Google Scholar] [CrossRef]
- Chang, C.H.; Lin, H.Y.; Chang, C.Y.; Liu, Y.C. Comparisons on the antioxidant properties of fresh, freeze-dried and hot-air-dried tomatoes. J. Food Eng. 2006, 77, 478–485. [Google Scholar] [CrossRef]
- Kerkhofs, N.S.; Lister, C.E.; Savage, G.P. Change in Colour and Antioxidant Content of Tomato Cultivars Following Forced-Air Drying. Plant Foods Human Nutr. 2005, 60, 117–121. [Google Scholar] [CrossRef]
- Toor, R.K.; Savage, G.P. Effect of semi-drying on the antioxidant components of tomatoes. Food Chem. 2006, 94, 90–97. [Google Scholar] [CrossRef]
- Shi, J.; Le Maguer, M.; Kakuda, Y.; Liptay, A.; Niekamp, F. Lycopene degradation and isomerization in tomato dehydration. Food Res. Int. 1999, 32, 15–21. [Google Scholar] [CrossRef]
- Giovanelli, G.; Zanoni, B.; Lavelli, V.; Nani, R. Water sorption, drying and antioxidant properties of dried tomato products. J. Food Eng. 2002, 52, 135–141. [Google Scholar] [CrossRef]
- Hsu, K.C. Evaluation of processing qualities of tomato juice induced by thermal and pressure processing. LWT-Food Sci. Technol. 2008, 41, 450–459. [Google Scholar] [CrossRef]
- Goula, A.M.; Adamopoulos, K.G.; Chatzitakis, P.C.; Nikas, V.A. Prediction of lycopene degradation during a drying process of tomato pulp. J. Food Eng. 2006, 74, 37–46. [Google Scholar] [CrossRef]
© 2010 by the authors;
Share and Cite
Kong, K.-W.; Khoo, H.-E.; Prasad, K.N.; Ismail, A.; Tan, C.-P.; Rajab, N.F. Revealing the Power of the Natural Red Pigment Lycopene. Molecules 2010, 15, 959-987. https://doi.org/10.3390/molecules15020959
Kong K-W, Khoo H-E, Prasad KN, Ismail A, Tan C-P, Rajab NF. Revealing the Power of the Natural Red Pigment Lycopene. Molecules. 2010; 15(2):959-987. https://doi.org/10.3390/molecules15020959
Chicago/Turabian StyleKong, Kin-Weng, Hock-Eng Khoo, K. Nagendra Prasad, Amin Ismail, Chin-Ping Tan, and Nor Fadilah Rajab. 2010. "Revealing the Power of the Natural Red Pigment Lycopene" Molecules 15, no. 2: 959-987. https://doi.org/10.3390/molecules15020959
APA StyleKong, K.-W., Khoo, H.-E., Prasad, K. N., Ismail, A., Tan, C.-P., & Rajab, N. F. (2010). Revealing the Power of the Natural Red Pigment Lycopene. Molecules, 15(2), 959-987. https://doi.org/10.3390/molecules15020959




